1. Introduction
Across the American continent, arid zones exist in many countries, such as Canada, the United States of America, Mexico, Bolivia, Venezuela, Brazil, Argentina, Chile, and Peru [
1]. In these countries, livestock are an important asset and an integral part of agriculture and farming systems [
2]. Many pastoralist communities comprising small ruminants have been established in these arid regions and depend on local ranges for their livelihood [
3]. However, current climatic volatility involves global warming, resulting in prolonged periods of dry weather and poor soil nutrient quality, affecting grassland plant species and threatening rangeland livestock production in arid zones [
1,
3]. Nevertheless, even though livestock production faces considerable challenges from climate change, more significant global threats to the future of humanity, such as food security, require the use of the entire range potential in arid zones [
4].
Thus, in the last decade, research has endeavored to identify plant species in grasslands adapted to alkaline soils, highly porous soils, and drier conditions with comparatively higher leaf drought tolerance [
5]. However, most plant species with these characteristics are identified as weedy or invasive; hence, grassland specialists recommend their elimination or removal [
6]. Interestingly, one of these plant genera is Salsola, which comprises halophyte plants and annual semi-dwarf to dwarf shrubs, including woody trees. Murshid et al. [
6] described Salsola as being rich in many classes of phytoconstituents, including flavonoids, phenolics, saponins, and volatile constituents. Nevertheless, the phytochemical composition and biological consequences of this genus have received little attention; only a few species from the genus Salsola have been examined for chemical and biological applications [
6,
7]. The most commonly reported species are
Salsola vermiculata,
Salsola cyclophylla, and
Salsola komarovii [
8]. Nevertheless, these species do not exist on the American continent, the most abundant being
Salsola tragus [
6]. However, government agencies describe little botanical information about this species [
9,
10]. Hence, the chemical characterization and biological applications of
Salsola tragus on the American continent are unknown.
Little information on the genus Salsola has been linked to herbal medicine applications. El-Bassossy et al. [
11] reported a pharmacological study that described the chemical constituents of the aerial parts of
Salsola kali. The authors concluded that
Salsola kali extract exhibited promise as a therapeutic source due to its increased antioxidant, anti-inflammatory, and cytotoxic activities. In addition, Arrekhi et al. [
12] and Mohammed et al. [
13] proposed that the metabolites and nutrients potentially convert the Salsola genus into an optimistic target for herbivore feed sources in arid zones. However, little information on animal performance in diets has been published using family species from the genus Salsola. Arrekhi et al. [
12] evaluated the forage potential of Salsola turcomanica and found that the plant’s growth and developmental stage determines the total digestive nutrients, fiber content, and crude protein. They concluded that an acceptable forage quality occurs during the young plant phase when
Salsola turcomanica has chemical values comparable with traditional forages such as
Medicago sativa. Based on these descriptions, the genus Salsola has excellent potential for use in ruminant feed. Nevertheless, the chemical composition of each species in the genus is accompanied by phytoconstituents that might modify the rumen microbiota [
14]. Therefore, the quality of meat or milk will depend on the chemical compounds and metabolites donated to the ruminant system by the particular species [
15].
Dietary flavonoids and phenolic compounds have been reported to increase serum growth hormone levels [
16], improve the immune system and antioxidant enzymes activity in the blood serum [
13], and promote muscle tissue synthesis in ruminants [
8,
11]. Therefore, different studies have proposed that photogenic compounds or phytoconstituents have a wide range of applications to provide insights into the biochemical balance of muscle tissue [
17]. This is because of the cascade of chemical pathways that involve proteins, carbohydrates, lipids, and minerals that impact meat color, tenderness, and flavor [
13]. In recent years, muscle physiology and meat science research has increasingly incorporated metabolomic analysis of the longissimus muscle metabolome, establishing connections to the chemical pathways within muscle tissue [
18]. Different scientific reports have characterized the effects of diet on the fattening phase and meat tastiness [
19,
20]. Nevertheless, few studies have employed metabolomics approaches to evaluate new sources of forage, such as arid plants that grow in saline soils, and their impact on meat production in small ruminants [
15]. Thus, we hypothesized that the herbal compounds of
Salsola tragus have specific metabolites with properties that can modify the metabolome during muscle growth. Thus, the objectives of the current study were (1) to characterize the natural compounds of
Salsola tragus via gas chromatography coupled with mass spectrometry (GC-MS), (2) evaluate the effect of
Salsola tragus as a source of forage on lamb growth and cellular damage from oxidative stress during the fattening phase, and (3) determine the impact of
Salsola tragus on the metabolomic meat interaction in lambs.
4. Discussion
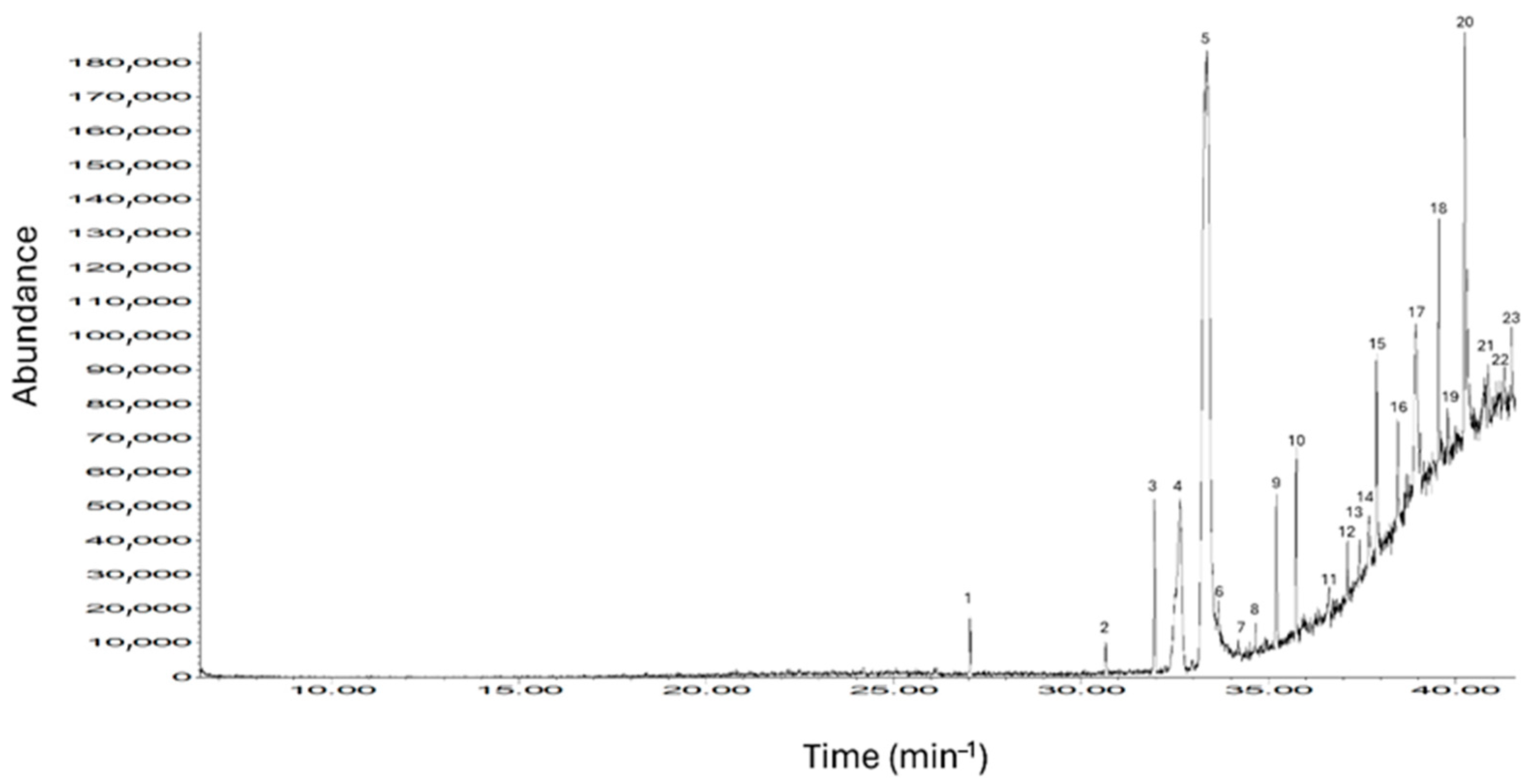
Twenty-three bioactive compounds were detected in
Salsola tragus, some of which have potential implications for ruminant metabolism and muscle formation [
6]. Previous studies [
6,
11,
37] provide solid evidence that the identified metabolites, such as tricosane, heneicosane, and octacosane, have a potential role in modulating lipid metabolism due to their hydrophobic nature and low reactivity [
38]. Additionally, sterol-based compounds such as cholest-5-en-3-ol (cholesterol derivative) might contribute to the modulation of membrane integrity and sterol biosynthesis pathways, which are critical for cellular function in muscle tissue [
39]. Additionally, several phenolic derivatives, including N-[2′-(3″,4″-dihydroxyphenyl)-2′-hydroxyethyl]-3-(4‴-methoxyphenyl)prop-2-enamide and isorhamnetin-(3β)-d-diglucuronate dimethyl ester, were also detected. These compounds are known for their antioxidant properties in desert plants [
40] and might support oxidative stress regulation, thereby influencing the redox status in muscle tissues [
6,
37]. Notably, several unique compounds, including 5H-isoindolo [1,2-b][3] benzazepin-5-one,7,8,13,13a-tetrahydro-10-hydroxy-3,4,12-trimethoxy-, and N,N-diisopropyl-4,6-bis-(methoxycarbonyl)-4,6-dimethylcyclohept-1-enecarboxamide, were identified for the first time in a forage context. These findings suggest a potential for novel metabolic pathways or bioactivities that warrant further exploration.
The presence of ferulic acid and its derivatives, such as S-(−)-trans-N-feruloyloctopamine and N-trans-feruloyltyramine, further highlights the functional benefits of
Salsola tragus as a forage source. Ferulic acid derivatives have been associated with anti-inflammatory effects and improved metabolic health in ruminants, as documented in studies exploring other forage plants like alfalfa and ryegrass [
41,
42,
43]. Specifically, N-trans-feruloyltyramine, an inhibitor of cyclooxygenases 1 and 2, mitigates inflammation, while its antioxidant and radical-scavenging properties contribute to cellular homeostasis [
6,
37]. Nevertheless, Soberón et al. [
44] described how ferulic acid and its derivatives modulate the microbiota in the rumen through a complex formed with arabinoxylans and lignin in forage cell walls in ester and ether covalent forms. According to this, Wang et al. [
42] reported that after ruminants ingest forages with ferulic acid conjugates, these cross-linkages form physical and chemical barriers to protect cell wall carbohydrates from microbial attack and enzymatic hydrolysis, increasing the capacity to break down the ester linkages within forage cell walls by secreting feruloyl and p-coumaroyl esterase, resulting in the release of free ferulic acid and an improvement in cell wall digestibility.
Concerning the productive performance of the lambs, no effect was observed on the ADG kg/d
−1 and DMI kg/d
−1 in lambs fed
Salsola tragus as the source of forage compared to the lambs fed sorghum stover in their diet. This consistency suggests that, at the tested inclusion levels,
Salsola tragus provided comparable or digestible nutrients relative to sorghum stover. This implies that the lambs were able to maintain an adequate dry matter intake. The unique nutritional profile of
Salsola tragus was not negatively impaired and potentially even supported the metabolic processes crucial for muscle growth. Different studies have proposed that plants of the genus Salsola increase ruminant productivity based on their nutrient values. Nevertheless, there is a lack of data on the use of plants of the genus Salsola during the fattening phase in ruminants [
6,
15]. The few reported studies have been linked to extensive systems in rangelands. Osman et al. [
45] evaluated the use of
Salsola vermiculata as a source of forage in Awassi ewes, in which grazed rangelands cover different types of forage. The authors concluded that rangelands in the presence of
Salsola vermiculata increased productive performance and economic benefits because
Salsola vermiculata maintains an amount of nutrients comparable to traditional forages during the year, allowing ewes to intake quality forage. Hanif et al. [
37] and ElNaggar et al. [
8] also reported that in the case of
Salsola vermiculata, the plant is highly palatable in the early stage of its growth for goats and sheep. These descriptions might be comparable to those of the current study because the DMI was maintained for the experimental period. In addition, the form of the
Salsola tragus particles used in the current study was observed by Sokolowska-Krzaczek et al. [
46], and Ali et al. [
47], where the authors emphasized that the plant has to be dried in the early stage as hay bait straw and later ground without spines or hardening. Based on these recommendations, Asaadi et al. [
48] concluded that in ruminants,
Salsola arbusculiformis has relatively high digestibility and metabolic energy during vegetative growth. As per Murshid et al. [
6], the benefits of animals using the genus Salsola are related to their secondary metabolites and phytochemical composition at different stages of growth.
In line with previous studies, the in vitro and estimated kinetics parameter model for lambs fed
Salsola tragus as a source of forage showed a quadratic effect for the reduction in the volume of gas produced when lambs received the two treatments that included
Salsola tragus. According to various studies, plants that contain ferulic acids might reduce the volume of gases emitted to the atmosphere to modulate microorganisms in the rumen, promoting sustainability and potential mitigation through adjusted inclusion rates of
Salsola tragus [
42,
49]. Wang et al. [
42] presented an extensive review of the potential relationship between esters or ethers linked to ferulic acid and fiber digestion in ruminants. The authors pointed out that, in conjunction with specific bacteria that naturally produce ferulic acid, gas production can be decreased by changes in the digestibility of the fiber, leading to a delay in the fermentation process in the rumen. These observations might be related to the increase in lag time in the 30-Salsola treatment. Wang et al. [
42] and Rosa-Prates et al. [
50] reported that different plant extracts prolong the lag time in the rumen by substantially depressing the activity of cellulolytic ruminal bacteria, such as
Selenomonas ruminantium, which have been reported to be capable of metabolizing ferulic acid. Nevertheless, the findings in the current study are restricted and only permit theorizing whether the secondary metabolites of
Salsola tragus have the capacity to increase lag time and prolong the period of microbial activity, ultimately producing greater methane emissions over a longer time frame. Finally, the quadratic effects observed in the estimated ME and mmol SCFA analysis might be explained by the greater concentration of methane at 48 h of gas accumulation and the lag time in the two treatments that included
Salsola tragus as a source of forage compared to the treatment with sorghum stover. Beauchemin et al. [
51] described that methane emissions and energy generated in the rumen by fermentation are commonly higher, partly because of greater fiber content, slow retention lag time, and in specific cases, the secondary metabolites in the source of forage. To our knowledge, from all the volatile components and chemical composition of
Salsola tragus, only ferulic acid and its secondary metabolites have been reported as modifiers of fiber digestibility in the rumen, while also improving the energy rate to potentiate the development of muscle. Lynch et al. [
52] concluded that ferulic acid could increase forage digestibility and the availability of cell wall carbohydrates to cellulolytic bacteria, potentially improving the substrate but increasing hydrogen production [
53]. Despite these promising results, more species of the genus Salsola should be investigated to characterize their fermentation and gas production effects on different ruminants when the genus is utilized as a unique source of forage in arid regions.
Despite the relatively minor effects of treatments on productive performance, such as DMI and ADG, the 30-Salsola treatment promotes muscle tissue formation to increase loin eye depth and tends to grow wider loins. In addition, the 30-Salsola treatment augmented the percentage of protein in LT muscle tissue compared to the other treatments. Even though the potential of the genus Salsola to be used as a source of forage has been described [
6,
11,
36], there is a lack of studies that have verified its effect on meat production. However, this response can be attributed to the increased CP in the diets where
Salsola tragus was included and the fact that lambs fed with the same treatments maintained their DMI during the current study, generating, as we described previously, different fermentation parameters in the rumen. In addition, the greater amount of CP in both diets with
Salsola tragus might increase the net energy to produce muscle growth by improving carbohydrate digestion and using secondary metabolites. Other halophyte plant species, such as
Salsola tragus, have been identified as forage sources with positive effects similar to traditional forages on muscle development during different ruminant productive periods [
54,
55]. Ahmed et al. [
56] concluded that
Atriplex nummularia and
Acacia saligna positively impact carcass weight and the CP content of the meat from Barki lambs when they were fed with a mix of both plants. The authors also concluded that changes in the CP of meat could be linked to the highest level of CP present in both plants compared to traditional forage.
In contrast to the increase in muscle development and CP content, fat thickness and crude fat content did not differ between treatments. These results support the diverse studies that have postulated that halophyte plants decrease carcass fat content and increase lean meat quantity compared to ruminants that are finished with a high-concentrate diet [
55,
56,
57]. This indicates that meat from sheep fed
Salsola tragus is nutritionally desirable, as this source of forage linearly reduces the amount of intramuscular fat and increases CP when it is included in the diet. These results could be further studied and used as commercial tools in arid systems of sheep meat production using
Salsola tragus as forage. Some initiatives have already been implemented in some regions of the American continent, aiming at these objectives, which will be invaluable in fully ascertaining the financial benefits of incorporating
Salsola tragus into livestock diets [
54,
58,
59].
The single-cell gel electrophoresis or comet assay has become a valuable tool in livestock production for assessing DNA damage and genotoxicity [
60]. In our study, this technique allowed us to evaluate the integrity of genetic material in individual cells, providing insights into the potential impacts of
Salsola tragus as a source of forage for lambs during the fattening phase. In livestock, Pu et al. [
61] proposed that the comet assay should be applied to farm animal models to investigate the effects of factors such as heat stress, mycotoxin exposure, and oxidative stress on DNA stability with respect to the type of ingredients in diets. Applying this analysis to lambs elucidates that
Salsola tragus is effective in promoting animal health and productivity compared to sources of traditional forage. Nevertheless, further research is necessary to evaluate the inclusion of
Salsola tragus as a forage source at different moments of livestock production.
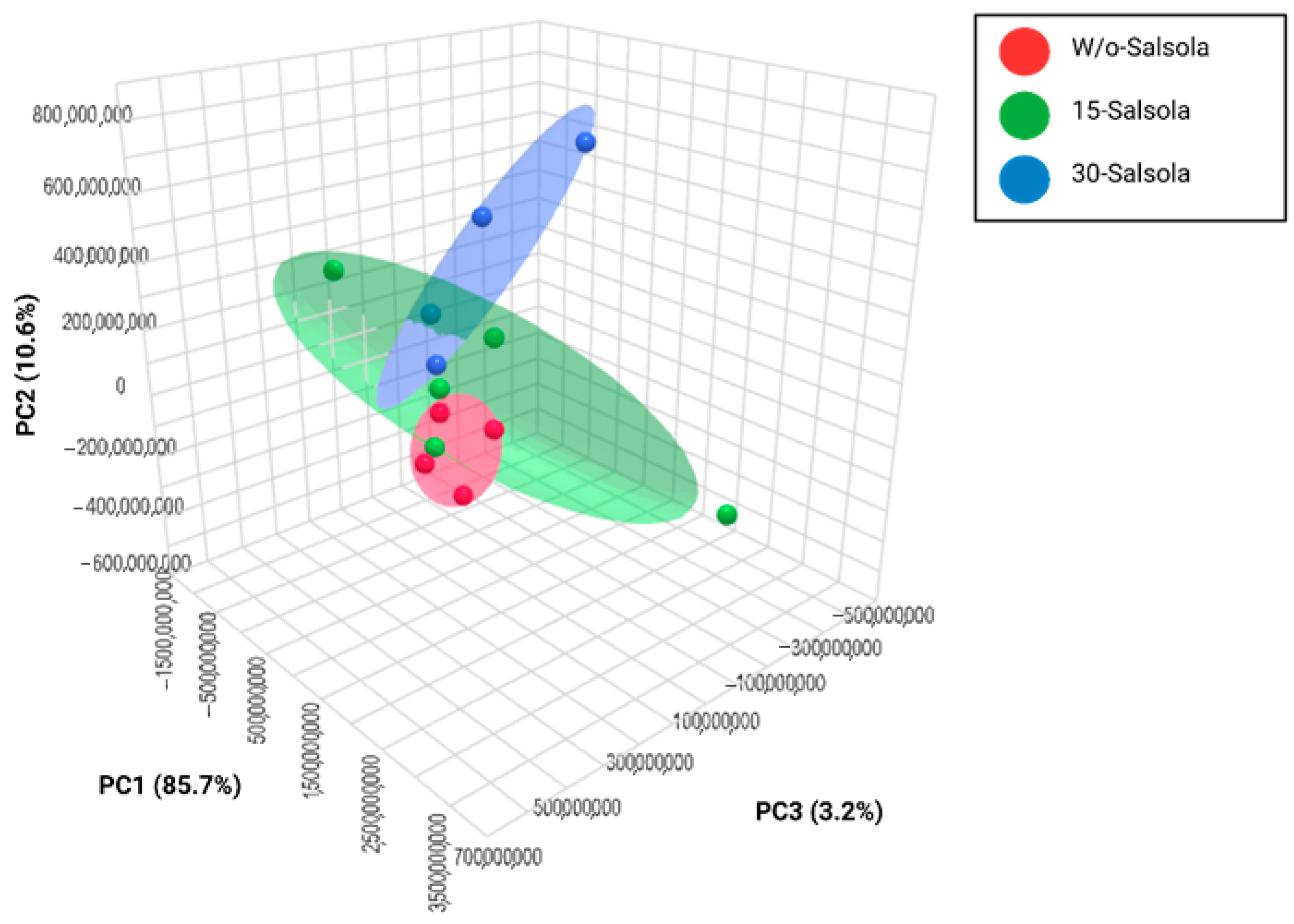
The sources of nutrients in feed systems are pivotal in determining the quality of raw meat, particularly in relation to the composition of fatty acids in meat. As noted by Wang et al. [
62], lipids and metabolites are key components that influence the taste, flavor, tenderness, and juiciness of cooked meat [
63]. The volatile compounds present in raw meat contribute to both the odor of raw lamb meat and the flavor of cooked meat. Metabolomic analysis is employed to rapidly screen small molecular metabolites under specific conditions, such as in muscle tissue, muscular cells, and fat adipocytes, and has been utilized to assess muscle mass growth and meat quality [
61]. Based on the GC-MS analysis, we found several important metabolites that showed not only a potential as biomarkers for discriminating the specific source of forage but also as indicators of meat quality, as illustrated in the PCA. The results obtained from the PCA also showed distinct metabolite profiles in the LT muscle of lambs fed the 15-Salsola treatment. This implies that the metabolomic analysis was able to authenticate all three sources of forage. In recent research, sheep meat from grazing or feedlots has been tested to understand the role of VOCs in determining flavor, tenderness, and shelf life [
64,
65]. The identified metabolite markers provide valuable information for future targeted studies. Subsequent research should aim to validate these VOCs in the metabolic pathways in meat modified by
Salsola tragus in meat, using different ruminant animal models.
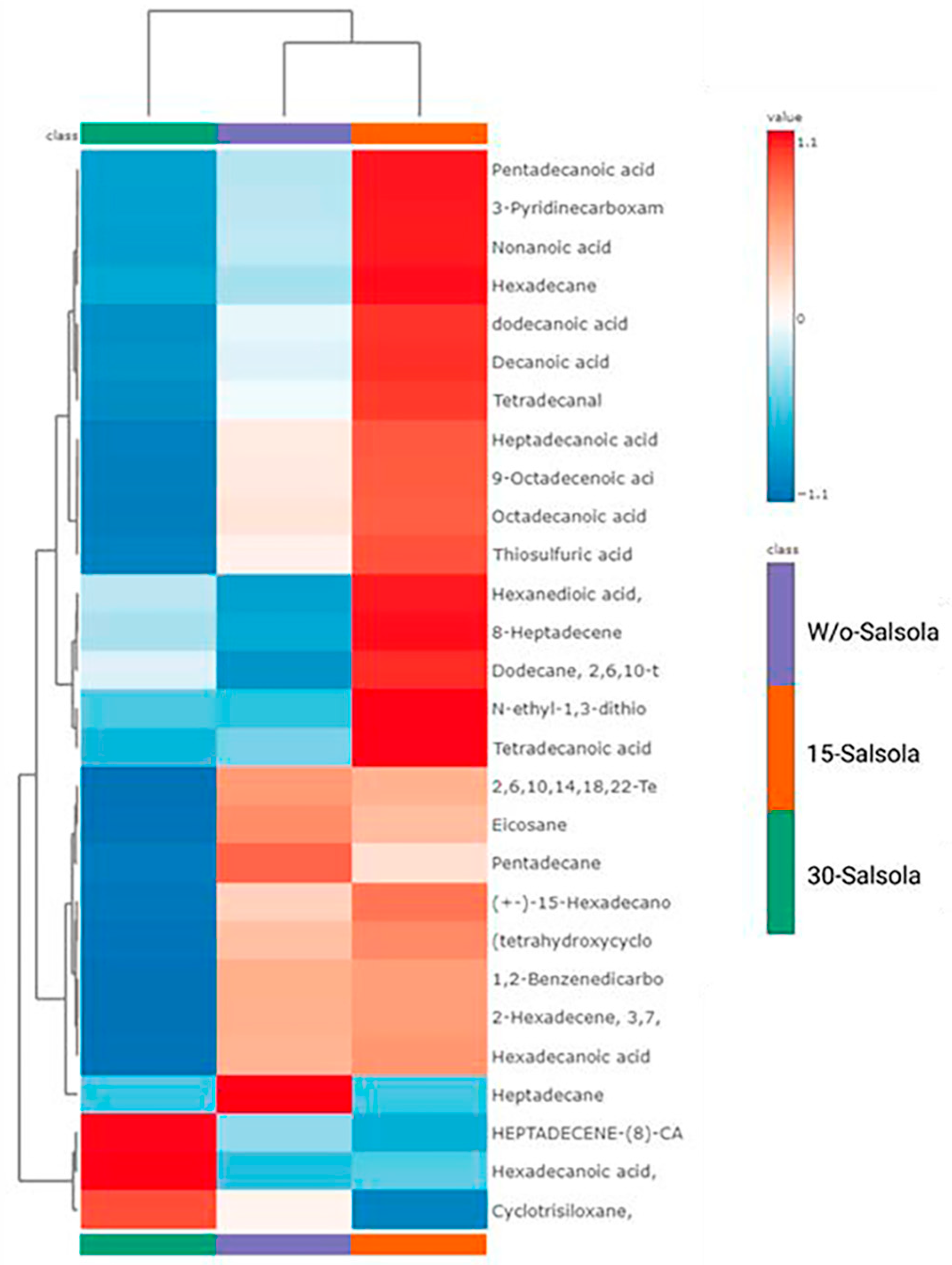
In our study, the 15-Salsola treatment showed the highest correlations between the 15 metabolites with the greatest importance index in LT tissue of lambs, mainly contributing to the discrimination between treatments. According to Strandvik [
66], the detection of volatile compounds in lamb meat and their relationship with better flavor has been linked for the greater part to the source of forage, independent of the amount of concentrate fed. This is because forages reduce certain types of unpleasant odors in meat via the types of volatile compounds that translocate in the form of lipids in the muscle [
62,
65]. Despite the fact that few studies have focused on volatile compounds in sheep meat and their relationship with different sources of forage, different authors have postulated that different silage-, alfalfa-, or hay-feeding regimes could modify decanoic acid content in meat, which is a medium-chain fatty acid associated with fatty acid metabolic modulations [
64,
65]. The increased abundance of decanoic acid determined using GC–MS-based metabolomics might confirm an altered fatty acid metabolism based on other fatty acids with a higher correlation to the 15-Salsola treatment. Fatty acids (FAs), particularly octadecanoic acid, 9-octadecanoic acid, and hexadecanoic acid, play a significant role in meat composition and quality. Curiously, the 15-Salsola treatment was positively correlated with previous FAs. Tánori-Lozano et al. [
49] found a source of ferulic acid might modify the fatty acid profile of the meat of lambs, such as octadecanoic acid and hexadecanoic acid, which are the primary saturated fatty acids found in meat. In addition, the 15-Salsola treatment increased its correlation with hexadecanoic acid, which is particularly prevalent and one of the most abundant fatty acids in various meat sources [
62,
65]. Finally, the utilization of the 30-Salsola or W/o-Salsola treatments as a forage source did not demonstrate a positive correlation with 9-Octadecanoic acid, a monounsaturated fatty acid that is also prevalent in meat. This compound is one of the primary substances released during cooking and contributes significantly to the distinctive aroma and flavor of cooked meat [
65,
67,
68]. Various forage sources have the potential to influence the fatty acid composition and volatile profiles of lamb meat, as well as certain biochemical properties of muscle, including protein content, texture, and shear force [
66,
67,
68]. The present study demonstrates that
Salsola tragus can beneficially alter volatile compounds in meat, suggesting that its inclusion in diets is viable in arid regions where traditional forage production is challenging. Furthermore, the data indicate that lambs fed fresh
Salsola tragus are capable of accumulating a greater quantity and diversity of plant-derived fatty acids or metabolites, such as ferulic acid, which may positively affect lipid metabolism. However, additional research is necessary to substantiate the essential roles and functions of these altered components in meat chemistry.