From Screening to Laboratory Scale-Up: Bioremediation Potential of Mushroom Strains Grown on Olive Mill Wastewater
Abstract
1. Introduction
2. Materials and Methods
2.1. Fungal Strains
2.2. Measurement of Mycelial Linear Growth Rate
2.3. Liquid Cultures Preparation and Mycelial Analyses
2.4. Data Analysis
3. Results
3.1. Preliminary Experiment on Kinetics Growth in Petri Dishes with Different OMW Concentrations
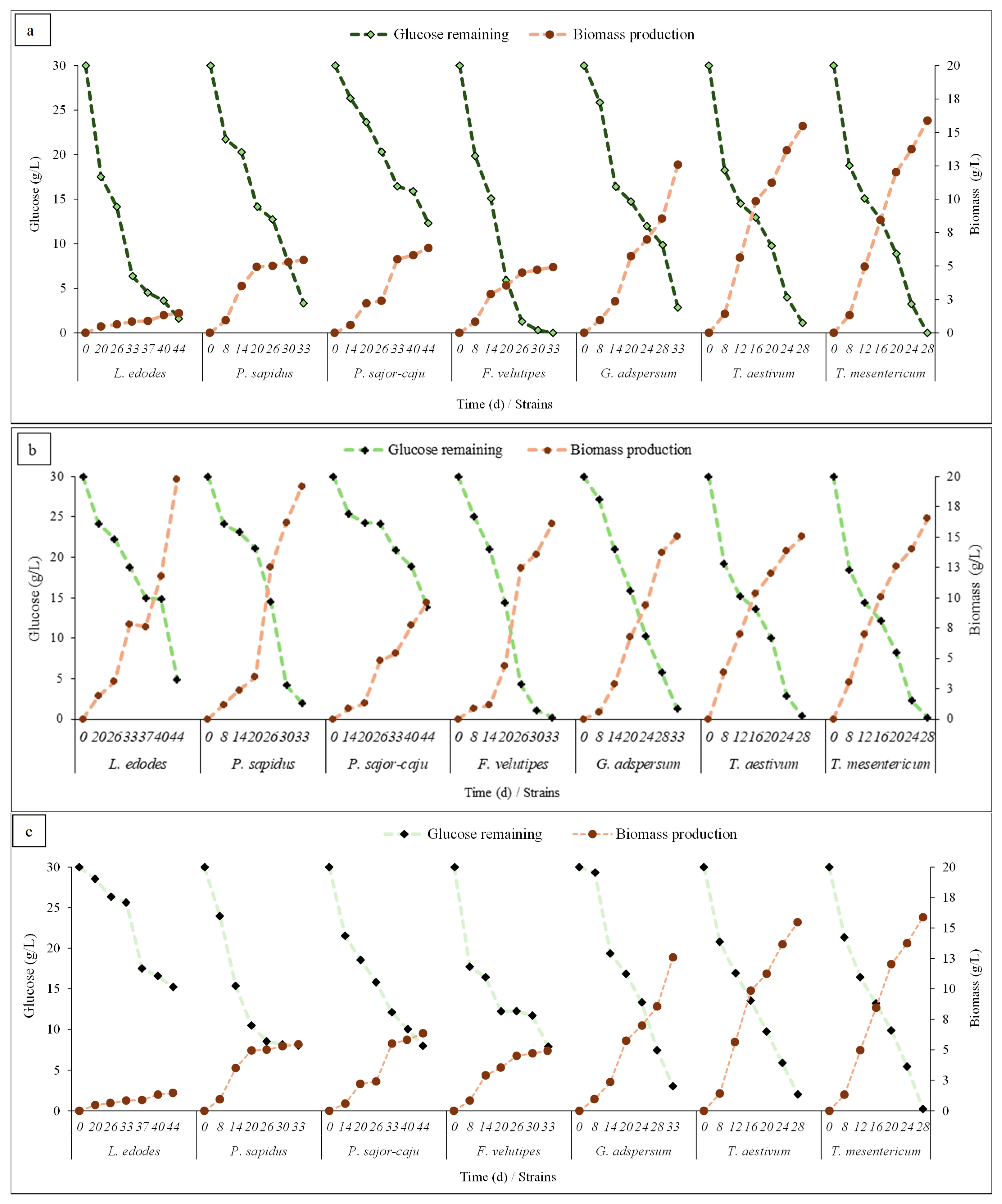
3.2. Growth Kinetics of Fungal Strains in Different OMW Concentrations
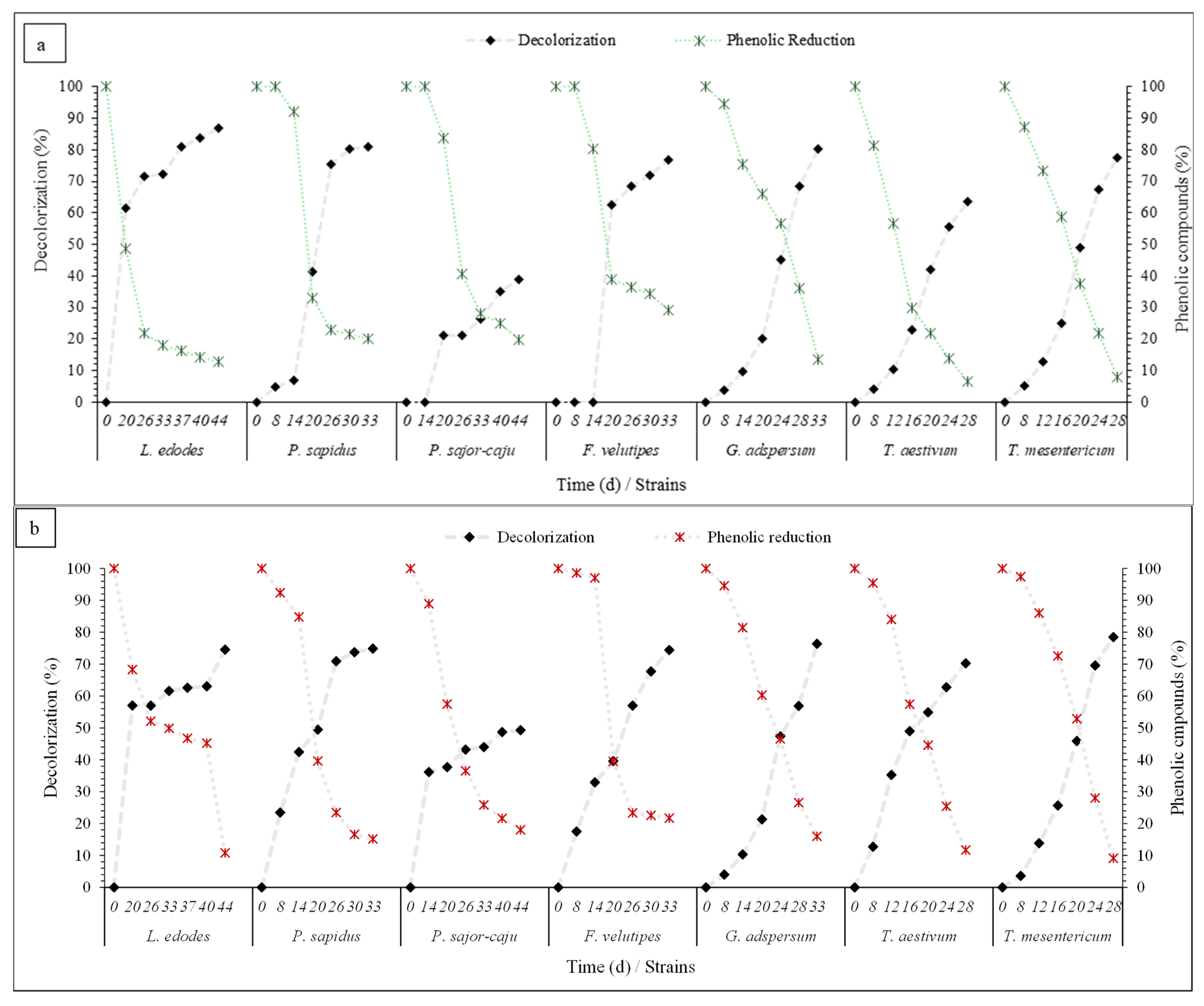
3.3. Fungal Efficiency in Phenolics Compounds Removal and Decolorization
3.4. Effect of OMW Concentration on Polysaccharide Production and Lipid Content of Fungi
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- FAOSTAT—Food and Agriculture Organization of the United Nations. Crops and Livestock Products: Olive Oil Production, 2022. Available online: https://www.fao.org/faostat/en/#data/QCL (accessed on 6 July 2025).
- Enaime, G.; Dababat, S.; Wichern, M.; Lübken, M. Olive mill wastes: From wastes to resources. Environ. Sci. Pollut. Res. 2024, 31, 20853–20880. [Google Scholar] [CrossRef] [PubMed]
- Mantzavinos, D.; Kalogerakis, N. Treatment of Olive Mill Effluents. Environ. Int. 2005, 31, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Rozzi, A.; Malpei, F. Treatment and Disposal of Olive Mill Effluents. Int. Biodeterior. Biodegrad. 1996, 38, 135–144. [Google Scholar] [CrossRef]
- Kapellakis, I.E.; Tsagarakis, K.P.; Avramaki, C.; Angelakis, A.N. Olive Mill Wastewater Management in River Basins: A Case Study in Greece. Agric. Water Manag. 2006, 82, 354–370. [Google Scholar] [CrossRef]
- Diamantis, I.; Melanouri, E.-M.; Dedousi, M.; Panagopoulou, I.; Papanikolaou, S.; Stoforos, N.G.; Diamantopoulou, P. Sustainable and Eco-Friendly Conversions of Olive Mill Wastewater-Based Media by Pleurotus pulmonarius Cultures. Fermentation 2022, 8, 129. [Google Scholar] [CrossRef]
- Diamantopoulou, P.; Gardeli, C.; Papanikolaou, S. Impact of Olive Mill Wastewaters on the Physiological Behavior of a Wild-Type New Ganoderma resinaceum Isolate. Environ. Sci. Pollut. Res. 2021, 28, 20570–20585. [Google Scholar] [CrossRef]
- El-Abbassi, A.; Saadaoui, N.; Kiai, H.; Raiti, J.; Hafidi, A. Potential Applications of Olive Mill Wastewater as Biopesticide for Crops Protection. Sci. Total Environ. 2017, 576, 10–21. [Google Scholar] [CrossRef]
- Dedousi, M.; Fourtaka, K.; Melanouri, E.-M.; Argyropoulos, D.; Psallida, C.; Diamantis, I.; Papanikolaou, S.; Diamantopoulou, P. Detoxification of Molasses and Production of Mycelial Mass and Valuable Metabolites by Morchella Species. Appl. Sci. 2021, 11, 9481. [Google Scholar] [CrossRef]
- Papanikolaou, S.; Rontou, M.; Belka, A.; Athenaki, M.; Gardeli, C.; Mallouchos, A.; Kalantzi, O.; Koutinas, A.A.; Kookos, I.K.; Zeng, A.-P.; et al. Conversion of Biodiesel-Derived Glycerol into Biotechnological Products of Industrial Significance by Yeast and Fungal Strains. Eng. Life Sci. 2017, 17, 262–281. [Google Scholar] [CrossRef]
- Sarris, D.; Philippoussis, A.; Mallouchos, A.; Diamantopoulou, P. Valorization of Low-Cost, Carbon-Rich Substrates by Edible Ascomycetes and Basidiomycetes Grown on Liquid Cultures. FEMS Microbiol. Lett. 2020, 367, fnaa168. [Google Scholar] [CrossRef]
- Morillo, J.A.; Antizar-Ladislao, B.; Monteoliva-Sánchez, M.; Ramos-Cormenzana, A.; Russell, N.J. Bioremediation and Biovalorisation of Olive-Mill Wastes. Appl. Microbiol. Biotechnol. 2009, 82, 25–39. [Google Scholar] [CrossRef]
- Sampedro, I.; Marinari, S.; D’Annibale, A.; Grego, S.; Ocampo, J.A.; García-Romera, I. Organic Matter Evolution and Partial Detoxification in Two-Phase Olive Mill Waste Colonized by White-Rot Fungi. Int. Biodeterior. Biodegrad. 2007, 60, 116–125. [Google Scholar] [CrossRef]
- Thomas, L.; Mago, P. Unearthing the Therapeutic Benefits of Culinary-medicinal Mushrooms for Humans: Emerging Sustainable Bioresources of 21st Century. J. Basic Microbiol. 2024, 64, e2400127. [Google Scholar] [CrossRef]
- Zhao, J.; Hu, Y.; Qian, C.; Hussain, M.; Liu, S.; Zhang, A.; He, R.; Sun, P. The Interaction between Mushroom Polysaccharides and Gut Microbiota and Their Effect on Human Health: A Review. Biology 2023, 12, 122. [Google Scholar] [CrossRef]
- Jesenak, M.; Majtan, J.; Rennerova, Z.; Kyselovic, J.; Banovcin, P.; Hrubisko, M. Immunomodulatory Effect of Pleuran (β-Glucan from Pleurotus ostreatus) in Children with Recurrent Respiratory Tract Infections. Int. Immunopharmacol. 2013, 15, 395–399. [Google Scholar] [CrossRef]
- Jesenak, M.; Hrubisko, M.; Chudek, J.; Bystron, J.; Rennerova, Z.; Diamant, Z.; Majtan, J.; Emeryk, A. Beneficial Effects of Pleuran on Asthma Control and Respiratory Tract-Infection Frequency in Children with Perennial Asthma. Sci. Rep. 2025, 15, 7146. [Google Scholar] [CrossRef]
- Hamuro, J.; Chihara, G. Immunopotentiation by the Antitumor Polysaccharide Lentinan. In Pharmacology; Hadden, J.W., Szentivanyi, A., Eds.; Springer: Boston, MA, USA, 1985; pp. 285–307. [Google Scholar] [CrossRef]
- Wasser, S.P.; Weis, A.L. Therapeutic Effects of Substances Occurring in Higher Basidiomycetes Mushrooms: A Modern Perspective. Crit. Rev. Immunol. 1999, 19, 65–96. [Google Scholar] [CrossRef]
- Deveci, E.; Çayan, F.; Tel-Çayan, G.; Duru, M.E. Structural Characterization and Determination of Biological Activities for Different Polysaccharides Extracted from Tree Mushroom Species. J. Food Biochem. 2019, 43, 12965. [Google Scholar] [CrossRef]
- Mfopa, A.; Mediesse, F.K.; Mvongo, C.; Nkoubatchoundjwen, S.; Lum, A.A.; Sobngwi, E.; Kamgang, R.; Boudjeko, T. Antidyslipidemic Potential of Water-Soluble Polysaccharides of Ganoderma applanatum in MACAPOS-2-Induced Obese Rats. Evid. Based Complement. Alternat. Med. 2021, 2021, 2452057. [Google Scholar] [CrossRef]
- Du, B.; Peng, F.; Xie, Y.; Wang, H.; Wu, J.; Liu, C.; Yang, Y. Optimization Extraction and Antioxidant Activity of Crude Polysaccharide from Chestnut Mushroom (Agrocybe aegerita) by Accelerated Solvent Extraction Combined with Response Surface Methodology (ASE-RSM). Molecules 2022, 27, 2380. [Google Scholar] [CrossRef] [PubMed]
- Jing, H.; Zhang, Q.; Liu, M.; Zhang, J.; Zhang, C.; Li, S.; Ren, Z.; Gao, Z.; Liu, X.; Jia, L. Polysaccharides with Antioxidative and Antiaging Activities from Enzymatic-extractable Mycelium by Agrocybe aegerita (Brig.) Sing. Evid. Based Complement. Alternat. Med. 2018, 2018, 1584647. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Nam, K.; Zahra, Z.; Farooqi, M.Q.U. Potentials of Truffles in Nutritional and Medicinal Applications: A Review. Fungal Biol. Biotechnol. 2020, 7, 9. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Wang, X.-H.; Li, H.-M.; Wang, S.-H.; Chen, T.; Yuan, Z.-P.; Tang, Y.-J. Isolation and Characterization of Polysaccharides with the Antitumor Activity from Tuber Fruiting Bodies and Fermentation System. Appl. Microbiol. Biotechnol. 2014, 98, 1991–2002. [Google Scholar] [CrossRef]
- Sande, D.; de Oliveira, G.P.; Moura, M.A.F.E.; de Martins, B.A.; Lima, M.T.N.S.; Takahashi, J.A. Edible Mushrooms as a Ubiquitous Source of Essential Fatty Acids. Food Res. Int. 2019, 125, 108524. [Google Scholar] [CrossRef]
- Kalač, P. Chemical composition and nutritional value of European species of wild growing mushrooms: A review. Food Chem. 2009, 113, 9–16. [Google Scholar] [CrossRef]
- Diamantopoulou, P.; Fourtaka, K.; Melanouri, E.M.; Dedousi, M.; Diamantis, I.; Gardeli, C.; Papanikolaou, S. Examining the Impact of Substrate Composition on the Biochemical Properties and Antioxidant Activity of Pleurotus and Agaricus Mushrooms. Fermentation 2023, 9, 689. [Google Scholar] [CrossRef]
- Günç Ergönül, P.; Akata, I.; Kalyoncu, F.; Ergönül, B. Fatty Acid Compositions of Six Wild Edible Mushroom Species. Sci. World J. 2013, 2013, 163964. [Google Scholar] [CrossRef]
- Holighaus, G.; Rohlfs, M. Volatile and Non-Volatile Fungal Oxylipins in Fungus-Invertebrate Interactions. Fungal Ecol. 2019, 38, 28–36. [Google Scholar] [CrossRef]
- Kües, U.; Khonsuntia, W.; Subba, S.; Dörnte, B. Volatiles in Communication of Agaricomycetes. In Physiology and Genetics; Anke, T., Schüffler, A., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 149–212. [Google Scholar] [CrossRef]
- Von Arx, M.; Schmidt-Büsser, D.; Guerin, P.M. Plant Volatiles Enhance Behavioral Responses of Grapevine Moth Males, Lobesia Botrana to Sex Pheromone. J. Chem. Ecol. 2012, 38, 222–225. [Google Scholar] [CrossRef] [PubMed]
- Miller, G.L. Use of Dinitrosalicylic Acid Reagent for Determination of Reducing Sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Slinkard, K.; Singleton, V.L. Total Phenol Analysis: Automation and Comparison with Manual Methods. Am. J. Enol. Vitic. 1977, 28, 49–55. [Google Scholar] [CrossRef]
- Tsioulpas, A. Phenolic Removal in Olive Oil Mill Wastewater by Strains of Pleurotus spp. in Respect to Their Phenol Oxidase (Laccase) Activity. Bioresour. Technol. 2002, 84, 251–257. [Google Scholar] [CrossRef]
- Diamantopoulou, P.; Papanikolaou, S.; Katsarou, E.; Komaitis, M.; Aggelis, G.; Philippoussis, A. Mushroom Polysaccharides and Lipids Synthesized in Liquid Agitated and Static Cultures. Part II: Study of Volvariella volvacea. Appl. Biochem. Biotechnol. 2012, 167, 1890–1906. [Google Scholar] [CrossRef]
- Folch, J.; Lees, M.; Stanley, S.G.H. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [CrossRef] [PubMed]
- Zervakis, G.I.; Koutrotsios, G.; Katsaris, P. Composted versus Raw Olive Mill Waste as Substrates for the Production of Medicinal Mushrooms: An Assessment of Selected Cultivation and Quality Parameters. BioMed Res. Int. 2013, 2013, 546830. [Google Scholar] [CrossRef] [PubMed]
- Lakhtar, H.; Ismaili-Alaoui, M.; Philippoussis, A.; Perraud-Gaime, I.; Roussos, S. Screening of Strains of Lentinula edodes Grown on Model Olive Mill Wastewater in Solid and Liquid State Culture for Polyphenol Biodegradation. Int. Biodeterior. Biodegrad. 2010, 64, 167–172. [Google Scholar] [CrossRef]
- Bevilacqua, A.; Cibelli, F.; Raimondo, M.L.; Carlucci, A.; Lops, F.; Sinigaglia, M.; Corbo, M.R. Fungal Bioremediation of Olive Mill Wastewater: Using a Multi-Step Approach to Model Inhibition or Stimulation. J. Sci. Food Agric. 2017, 97, 461–468. [Google Scholar] [CrossRef]
- Cibelli, F.; Bevilacqua, A.; Raimondo, M.L.; Campaniello, D.; Carlucci, A.; Ciccarone, C.; Sinigaglia, M.; Corbo, M.R. Evaluation of Fungal Growth on Olive-Mill Wastewaters Treated at High Temperature and by High-Pressure Homogenization. Front. Microbiol. 2017, 8, 2515. [Google Scholar] [CrossRef]
- Mulinacci, N.; Romani, A.; Galardi, C.; Pinelli, P.; Giaccherini, C.; Vincieri, F.F. Polyphenolic Content in Olive Oil Waste Waters and Related Olive Samples. J. Agric. Food Chem. 2001, 49, 3509–3514. [Google Scholar] [CrossRef]
- Dinakarkumar, Y.; Ramakrishnan, G.; Gujjula, K.R.; Vasu, V.; Balamurugan, P.; Murali, G. Fungal Bioremediation: An Overview of the Mechanisms, Applications and Future Perspectives. Environ. Chem. Ecotoxicol. 2024, 6, 293–302. [Google Scholar] [CrossRef]
- Matos, A.J.F.S.; Bezerra, R.M.F.; Dias, A.A. Screening of Fungal Isolates and Properties of Ganoderma applanatum Intended for Olive Mill Wastewater Decolourization and Dephenolization. Lett. Appl. Microbiol. 2007, 45, 270–275. [Google Scholar] [CrossRef]
- Koutrotsios, G.; Zervakis, G.I. Comparative Examination of the Olive Mill Wastewater Biodegradation Process by Various Wood-Rot Macrofungi. BioMed Res. Int. 2014, 2014, 482937. [Google Scholar] [CrossRef] [PubMed]
- Diamantopoulou, P.; Papanikolaou, S.; Kapoti, M.; Komaitis, M.; Aggelis, G.; Philippoussis, A. Mushroom Polysaccharides and Lipids Synthesized in Liquid Agitated and Static Cultures. Part I: Screening Various Mushroom Species. Appl. Biochem. Biotechnol. 2012, 167, 536–551. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.Y.; Park, Y.; Ahn, J.K.; Ka, K.H.; Park, S.Y. Factors Influencing the Production of Endopolysaccharide and Exopolysaccharide from Ganoderma applanatum. Enzyme Microb. Technol. 2007, 40, 249–254. [Google Scholar] [CrossRef]
- Maziero, R.; Cavazzoni, V.; Bononi, V.L.R. Screening of Basidiomycetes for the Production of Exopolysaccharide and Biomass in Submerged Culture. Rev. Microbiol. 1999, 30, 77–84. [Google Scholar] [CrossRef]
- Kim, H.O.; Lim, J.M.; Joo, J.H.; Kim, S.W.; Hwang, H.J.; Choi, J.W.; Yun, J.W. Optimization of Submerged Culture Condition for the Production of Mycelial Biomass and Exopolysaccharides by Agrocybe cylindracea. Bioresour. Technol. 2005, 96, 1175–1182. [Google Scholar] [CrossRef]
- Wasser, S. Medicinal Mushrooms as a Source of Antitumor and Immunomodulating Polysaccharides. Appl. Microbiol. Biotechnol. 2002, 60, 258–274. [Google Scholar] [CrossRef]
- Zerva, A.; Zervakis, G.I.; Christakopoulos, P.; Topakas, E. Degradation of Olive Mill Wastewater by the Induced Extracellular Ligninolytic Enzymes of Two Wood-Rot Fungi. J. Environ. Manag. 2017, 203, 791–798. [Google Scholar] [CrossRef]
- Ntougias, S.; Baldrian, P.; Ehaliotis, C.; Nerud, F.; Antoniou, T.; Merhautová, V.; Zervakis, G.I. Biodegradation and Detoxification of Olive Mill Wastewater by Selected Strains of the Mushroom Genera Ganoderma and Pleurotus. Chemosphere 2012, 88, 620–626. [Google Scholar] [CrossRef]
- D’Annibale, A.; Casa, R.; Pieruccetti, F.; Ricci, M.; Marabottini, R. Lentinula edodes Removes Phenols from Olive-Mill Wastewater: Impact on Durum Wheat (Triticum durum Desf.) Germinability. Chemosphere 2004, 54, 887–894. [Google Scholar] [CrossRef]
- Fraij, A.; Massadeh, M. Use of Pleurotus Sajor-caju for the Biotreatment of Olive Mill Wastewater. Rom. Biotechnol. Lett. 2015, 20, 10611–10617. [Google Scholar]
- Diamantopoulou, P.; Papanikolaou, S.; Komaitis, M.; Aggelis, G.; Philippoussis, A. Patterns of Major Metabolites Biosynthesis by Different Mushroom Fungi Grown on Glucose-Based Submerged Cultures. Bioprocess Biosyst. Eng. 2014, 37, 1385–1400. [Google Scholar] [CrossRef]
- Miles, P.G.; Chang, S.-T. Mushrooms, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2004. [Google Scholar] [CrossRef]
- Tang, Y.-J.; Zhu, L.-L.; Liu, R.-S.; Li, H.-M.; Li, D.-S.; Mi, Z.-Y. Quantitative Response of Cell Growth and Tuber Polysaccharides Biosynthesis by Medicinal Mushroom Chinese Truffle Tuber sinense to Metal Ion in Culture Medium. Bioresour. Technol. 2008, 99, 7606–7615. [Google Scholar] [CrossRef]
- Beopoulos, A.; Mrozova, Z.; Thevenieau, F.; Le Dall, M.-T.; Hapala, I.; Papanikolaou, S.; Chardot, T.; Nicaud, J.-M. Control of Lipid Accumulation in the Yeast Yarrowia lipolytica. Appl. Environ. Microbiol. 2008, 74, 7779–7789. [Google Scholar] [CrossRef] [PubMed]
- Ratledge, C. Fatty Acid Biosynthesis in Microorganisms Being Used for Single Cell Oil Production. Biochimie 2004, 86, 807–815. [Google Scholar] [CrossRef]
- Papanikolaou, S.; Aggelis, G. Lipids of Oleaginous Yeasts. Part I: Biochemistry of Single Cell Oil Production. Eur. J. Lipid Sci. Technol. 2011, 113, 1031–1051. [Google Scholar] [CrossRef]
- Sancholle, M.; Lösel, D.M.; Laruelle, E. Lipids in Fungal Biotechnology. In Genetics and Biotechnology; Springer: New York, NY, USA, 2004; pp. 391–423. [Google Scholar]
- Miric, O.; Lalic, V.Z.; Miletic, D.I. The Composition of Some Lipid Fractions (Phospholipids, Triglycerides, Free Fatty Acids, Sterols) of Wild Growing Edible Mushrooms. Hrana Ishr. Yugosl. 1985, 26, 123–128. [Google Scholar]
- Bellou, S.; Triantaphyllidou, I.-E.; Aggeli, D.; Elazzazy, A.M.; Baeshen, M.N.; Aggelis, G. Microbial Oils as Food Additives: Recent Approaches for Improving Microbial Oil Production and Its Polyunsaturated Fatty Acid Content. Curr. Opin. Biotechnol. 2016, 37, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Dzurendova, S.; Zimmermann, B.; Tafintseva, V.; Kohler, A.; Ekeberg, D.; Shapaval, V. The Influence of Phosphorus Source and the Nature of Nitrogen Substrate on the Biomass Production and Lipid Accumulation in Oleaginous Mucoromycota Fungi. Appl. Microbiol. Biotechnol. 2020, 104, 8065–8076. [Google Scholar] [CrossRef]
- Kavishree, S.; Hemavathy, J.; Lokesh, B.R.; Shashirekha, M.N.; Rajarathnam, S. Fat and Fatty Acids of Indian Edible Mushrooms. Food Chem. 2008, 106, 597–602. [Google Scholar] [CrossRef]
- Confortin, F.G.; Marchetto, R.; Bettin, F.; Camassola, M.; Salvador, M.; Dillon, A.J.P. Production of Pleurotus sajor-caju Strain PS-2001 Biomass in Submerged Culture. J. Ind. Microbiol. Biotechnol. 2008, 35, 1149–1155. [Google Scholar] [CrossRef] [PubMed]
- Dimou, D.M.; Georgala, A.; Komaitis, M.; Aggelis, G. Mycelial Fatty Acid Composition of Pleurotus spp. and Its Application in the Intrageneric Differentiation. Mycol. Res. 2002, 106, 925–929. [Google Scholar] [CrossRef]
- Smiderle, F.R.; Ruthes, A.C.; Van Arkel, J.; Chanput, W.; Iacomini, M.; Wichers, H.J.; Van Griensven, L.J. Polysaccharides from Agaricus bisporus and Agaricus brasiliensis Show Similarities in Their Structures and Their Immunomodulatory Effects on Human Monocytic THP-1 Cells. BMC Complement. Altern. Med. 2011, 11, 58. [Google Scholar] [CrossRef] [PubMed]
| Strain | AMRL |
|---|---|
| Agrocybe cylindracea | 101 |
| A. cylindracea | 104 |
| Lentinula edodes | 125 |
| L. edodes | 126 |
| Pleurotus sapidus | 156-1 |
| P. sapidus | 156-2 |
| Pleurotus sajor-caju | 197 |
| Flammulina velutipes | 271 |
| Ganoderma adspersum | 315 |
| Tuber aestivum | 364 |
| Tuber mesentericum | 365 |
| OMW (% v/v)/ Species (Strains) | 0 | 5 | 10 | 15 | 20 | 25 | 30 | 35 | 40 | 45 | 50 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| A. cylindracea AMRL 101 | 3.20 ± 0.13 | 3.32 ± 0.05 | 3.11 ± 0.18 | 2.87 ± 0.10 | 2.54 ± 0.31 | 2.69 ± 0.05 | 2.86 ± 0.15 | 3.12 ± 0.04 | 2.63 ± 0.15 | 2.33 ± 0.16 | 2.05 ± 0.11 |
| A. cylindracea AMRL 104 | 3.11 ± 0.36 | 3.40 ± 0.03 | 3.57 ± 0.25 | 3.48 ± 0.11 | 3.18 ± 0.07 | 2.81 ± 0.09 | 2.98 ± 0.03 | 3.00 ± 0.04 | 2.67 ± 0.06 | 2.30 ± 0.03 | 2.27 ± 0.05 |
| L. edodes AMRL 125 | 3.63 ± 0.13 | 4.76 ± 0.11 | 3.83 ± 0.22 | 4.04 ± 0.68 | 4.59 ± 0.32 | 2.02 ± 0.14 | 3.33 ± 0.29 | 3.59 ± 0.09 | 2.80 ± 0.51 | 1.80 ± 0.03 | 1.55 ± 0.04 |
| L. edodes AMRL 126 | 3.84 ± 0.21 | 4.26 ± 0.27 | 5.22 ± 0.27 | 4.79 ± 0.05 | 4.59 ± 0.30 | 2.90 ± 0.32 | 3.52 ± 0.33 | 3.68 ± 0.04 | 3.17 ± 0.37 | 2.21 ± 0.10 | 2.31 ± 0.33 |
| P. sapidus AMRL 156-1 | 2.86 ± 0.20 | 4.96 ± 0.02 | 4.89 ± 0.14 | 4.90 ± 0.06 | 4.40 ± 0.55 | 3.83 ± 0.04 | 3.87 ± 0.03 | 3.89 ± 0.03 | 3.75 ± 0.06 | 3.76 ± 0.21 | 3.79 ± 0.13 |
| P. sapidus AMRL 156-2 | 4.58 ± 0.25 | 4.49 ± 0.03 | 4.42 ± 0.06 | 4.47 ± 0.05 | 4.72 ± 0.01 | 3.83 ± 0.07 | 3.84 ± 0.12 | 3.90 ± 0.07 | 3.88 ± 0.03 | 4.88 ± 0.24 | 5.12 ± 0.23 |
| P. sajor-caju AMRL 197 | 4.05 ± 1.22 | 6.45 ± 0.30 | 6.36 ± 0.15 | 6.10 ± 0.03 | 6.28 ± 0.07 | 5.06 ± 0.12 | 6.31 ± 0.93 | 5.45 ± 0.08 | 5.08 ± 0.08 | 4.26 ± 0.08 | 4.25 ± 0.10 |
| F. velutipes AMRL 271 | 6.60 ± 0.07 | 6.28 ± 0.46 | 6.44 ± 0.03 | 6.18 ± 0.48 | 5.86 ± 0.48 | 3.52 ± 0.10 | 4.73 ± 0.04 | 4.72 ± 0.07 | 4.87 ± 0.52 | 4.07 ± 0.06 | 4.02 ± 0.06 |
| G. adspersum AMRL 315 | 12.05 ± 0.07 | 12.51 ± 0.07 | 14.57 ± 0.46 | 12.22 ± 0.03 | 12.15 ± 0.48 | 11.94 ± 0.48 | 10.61 ± 0.10 | 12.51 ± 0.04 | 13.25 ± 0.07 | 10.13 ± 0.52 | 11.79 ± 0.06 |
| T. aestivum AMRL 364 | 7.39 ± 0.07 | 6.21 ± 0.07 | 6.24 ± 0.46 | 6.38 ± 0.03 | 5.97 ± 0.48 | 7.00 ± 0.48 | 6.98 ± 0.10 | 6.66 ± 0.04 | 7.24 ± 0.07 | 8.39 ± 0.52 | 8.95 ± 0.06 |
| T. mesentericum AMRL 365 | 10.31 ± 0.07 | 9.10 ± 0.07 | 8.16 ± 0.46 | 9.07 ± 0.03 | 10.25± 0.48 | 9.41 ± 0.48 | 9.21 ± 0.10 | 9.30± 0.04 | 8.68 ± 0.07 | 8.88 ± 0.52 | 8.32 ± 0.06 |
| Stains | Days | 0% v/v OMW | 15% v/v OMW | 35% v/v OMW | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| L | IPS | L | IPS | L | IPS | |||||||||
| (% w/w) | g/L | (% w/w) | g/L | (% w/w) | g/L | (% w/w) | g/L | (% w/w) | g/L | (% w/w) | g/L | |||
| L. edodes | 33 | c | 13.35 ± 0.22 | 1.18 ± 0.04 | 27.58 ± 0.27 | 2.44 ± 0.03 | 6.74 ± 0.27 | 1.00 ± 0.17 | 36.69 ± 0.50 | 5.46 ± 0.07 | 5.26 ± 0.24 | 0.41 ± 0.04 | 32.49 ± 0.08 | 2.49 ± 0.08 |
| AMRL 126 | 44 | a, b | 7.11 ± 0.34 | 0.74 ± 0.06 | 39.45 ± 0.16 | 4.13 ± 0.02 | 6.79 ± 0.31 | 1.02 ± 0.08 | 22.99 ± 0.52 | 3.46 ± 0.08 | 4.23 ± 0.26 | 0.71 ± 0.05 | 34.52 ± 0.03 | 4.52 ± 0.03 |
| P. sapidus | 20 | c | 9.72 ± 0.16 | 0.48 ± 0.02 | 37.82 ± 0.19 | 1.87 ± 0.01 | 8.19 ± 0.08 | 0.57 ± 0.12 | 29.90 ± 0.48 | 2.09 ± 0.03 | 7.92 ± 0.08 | 0.19 ± 0.01 | 31.06 ± 0.01 | 1.06 ± 0.01 |
| AMRL 156-2 | 33 | a, b | 7.68 ± 0.09 | 0.40 ± 0.03 | 38.10 ± 0.48 | 2.02 ± 0.03 | 3.12 ± 0.03 | 0.35 ± 0.06 | 36.08 ± 0.10 | 4.00 ± 0.01 | 3.60 ± 0.02 | 0.48 ± 0.05 | 33.92 ± 0.10 | 3.92 ± 0.10 |
| P. sajor-caju | 26 | c | 13.61 ± 0.34 | 0.33 ± 0.05 | 25.48 ± 0.01 | 0.61 ± 0.00 | 8.83 ± 0.07 | 0.51 ± 0.14 | 36.05 ± 2.66 | 2.08 ± 0.15 | 5.60 ± 0.19 | 0.27 ± 0.04 | 31.26 ± 0.01 | 1.26 ± 0.01 |
| AMRL 197 | 44 | a, b | 7.16 ± 0.29 | 0.45 ± 0.01 | 39.36 ± 0.32 | 2.50 ± 0.02 | 7.13 ± 0.06 | 0.45 ± 0.12 | 42.21 ± 0.45 | 2.66 ± 0.03 | 6.73 ± 0.12 | 0.65 ± 0.08 | 33.42 ± 0.04 | 3.42 ± 0.04 |
| F. velutipes | 20 | c | 16.68 ± 0.26 | 0.59 ± 0.04 | 33.64 ± 0.75 | 1.19 ± 0.03 | 10.49 ± 0.31 | 1.23 ± 0.07 | 27.43 ± 0.35 | 3.22 ± 0.04 | 14.87 ± 0.36 | 0.66 ± 0.03 | 31.18 ± 0.06 | 1.18 ± 0.06 |
| AMRL 271 | 33 | a, b | 7.65 ± 0.13 | 0.31 ± 0.02 | 44.13 ± 0.16 | 1.81 ± 0.10 | 6.90 ± 0.19 | 0.38 ± 0.02 | 32.51 ± 1.09 | 1.78 ± 0.06 | 3.16 ± 0.11 | 0.39 ± 0.01 | 33.92 ± 0.00 | 3.92 ± 0.00 |
| G. adspersum | 20 | c | 10.68 ± 0.23 | 0.61 ± 0.05 | 34.74 ± 0.68 | 2.00 ± 0.03 | 11.69 ± 0.28 | 0.92 ± 0.01 | 37.13 ± 0.83 | 2.91 ± 0.09 | 12.70 ± 0.16 | 0.86 ± 0.02 | 32.58 ± 0.93 | 2.21 ± 0.09 |
| AMRL 315 | 33 | a, b | 8.52 ± 0.20 | 1.07 ± 0.06 | 46.23 ± 0.15 | 5.81 ± 0.10 | 9.90 ± 0.15 | 1.41 ± 0.04 | 42.81 ± 0.85 | 6.09 ± 0.20 | 11.62 ± 0.10 | 1.75 ± 0.03 | 39.89 ± 0.91 | 6.00 ± 0.11 |
| T. aestivum | 20 | c | 14.28 ± 0.16 | 1.60 ± 0.08 | 32.88 ± 0.65 | 3.69 ± 0.09 | 12.42 ± 0.33 | 1.47 ± 0.03 | 31.42 ± 0.87 | 3.72 ± 0.07 | 14.22 ± 0.33 | 1.71 ± 0.07 | 31.83 ± 0.88 | 3.82 ± 0.21 |
| AMRL 364 | 28 | a, b | 9.85 ± 0.31 | 1.52 ± 0.06 | 43.85 ± 0.20 | 6.78 ± 0.30 | 10.93 ± 0.18 | 1.62 ± 0.07 | 43.51 ± 1.20 | 6.46 ± 0.11 | 13.26 ± 0.22 | 1.99 ± 0.04 | 39.87 ± 0.92 | 5.98 ± 0.31 |
| T. mesentericum | 20 | c | 15.88 ± 0.30 | 1.90 ± 0.07 | 34.42 ± 0.58 | 4.14 ± 0.14 | 13.99 ± 0.29 | 1.68 ± 0.05 | 32.52 ± 0.83 | 3.90 ± 0.09 | 15.63 ± 0.28 | 1.96 ± 0.03 | 41.82 ± 0.89 | 5.26 ± 0.28 |
| AMRL 365 | 28 | a, b | 12.65 ± 0.12 | 2.01 ± 0.07 | 45.53 ± 0.17 | 7.23 ± 0.30 | 11.83 ± 0.16 | 1.94 ± 0.08 | 42.55 ± 1.03 | 6.98 ± 0.29 | 13.60 ± 0.21 | 2.25 ± 0.05 | 43.92 ± 0.83 | 7.27 ± 0.22 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Diamantis, I.; Stamatiadis, S.; Melanouri, E.-M.; Papanikolaou, S.; Diamantopoulou, P. From Screening to Laboratory Scale-Up: Bioremediation Potential of Mushroom Strains Grown on Olive Mill Wastewater. Biomass 2025, 5, 50. https://doi.org/10.3390/biomass5030050
Diamantis I, Stamatiadis S, Melanouri E-M, Papanikolaou S, Diamantopoulou P. From Screening to Laboratory Scale-Up: Bioremediation Potential of Mushroom Strains Grown on Olive Mill Wastewater. Biomass. 2025; 5(3):50. https://doi.org/10.3390/biomass5030050
Chicago/Turabian StyleDiamantis, Ilias, Spyridon Stamatiadis, Eirini-Maria Melanouri, Seraphim Papanikolaou, and Panagiota Diamantopoulou. 2025. "From Screening to Laboratory Scale-Up: Bioremediation Potential of Mushroom Strains Grown on Olive Mill Wastewater" Biomass 5, no. 3: 50. https://doi.org/10.3390/biomass5030050
APA StyleDiamantis, I., Stamatiadis, S., Melanouri, E.-M., Papanikolaou, S., & Diamantopoulou, P. (2025). From Screening to Laboratory Scale-Up: Bioremediation Potential of Mushroom Strains Grown on Olive Mill Wastewater. Biomass, 5(3), 50. https://doi.org/10.3390/biomass5030050









