Algorithmic Reconstruction of Multimodal Copper Wire Explosion Products from Extinction Spectra
Abstract
1. Introduction
2. Materials and Methods
2.1. Wire Explosion Setup and Optical Characterization
2.2. Input Datasets for the Spectral Decomposition
2.3. Computational Methods
- The scaling factors , , and for copper, copper(I) oxide, and copper(II) oxide, respectively;
- The mean and standard deviation of the copper size distribution;
- The logarithmic means and , and the logarithmic standard deviations and , of the oxide distributions;
- The weight of the baseline.
3. Results
3.1. Evaluation Uncertainty of Extinction Spectrum Deconvolution
3.2. Voltage-Dependent Trends in the WE Products
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CuNP | Copper Nanoparticle |
| EA | Evolutionary Algorithm |
| EF | Exploded Fraction |
| GS | Grid Search |
| IR | Infrared |
| NP | Nanoparticle |
| SM | Semimanual (fitting) |
| UV | Ultraviolet |
| VIS | Visible |
| WE | Wire Explosion |
References
- Khodashenas, B.; Ghorbani, H.R. Synthesis of Copper Nanoparticles: An Overview of the Various Methods. Korean J. Chem. Eng. 2014, 31, 1105–1109. [Google Scholar] [CrossRef]
- Purohit, P.; Samadi, A.; Bendix, P.M.; Laserna, J.J.; Oddershede, L.B. Optical Trapping Reveals Differences in Dielectric and Optical Properties of Copper Nanoparticles Compared to Their Oxides and Ferrites. Sci. Rep. 2020, 10, 1198. [Google Scholar] [CrossRef]
- Zamir Anvari, J.; Karimzadeh, R.; Mansour, N. Thermo-Optic Properties and Nonlinear Responses of Copper Nanoparticles in Polysiloxane oil. J. Opt. 2010, 12, 035212. [Google Scholar] [CrossRef]
- Crisan, M.C.; Teodora, M.; Lucian, M. Copper Nanoparticles: Synthesis and Characterization, Physiology, Toxicity and Antimicrobial Applications. Appl. Sci. 2021, 12, 141. [Google Scholar] [CrossRef]
- Zhang, P.; Sun, Q.; Fang, S.; Guo, H.; Liu, K.; Zhang, L.; Zhu, Q.; Wang, M. Fabrication of Nano Copper Highly Conductive and Flexible Printed Electronics by Direct Ink Writing. ACS Appl. Mater. Interfaces 2025, 17, 1847–1860. [Google Scholar] [CrossRef]
- Pathak, R.; Punetha, V.D.; Bhatt, S.; Punetha, M. A Review on Copper-Based Nanoparticles as a Catalyst: Synthesis and Applications in Coupling Reactions. J. Mater. Sci. 2024, 59, 6169–6205. [Google Scholar] [CrossRef]
- Devaraji, M.; Thanikachalam, P.V.; Elumalai, K. The Potential of Copper Oxide Nanoparticles in Nanomedicine: A Comprehensive Review. Biotechnol. Notes 2024, 5, 80–99. [Google Scholar] [CrossRef]
- Shankar, S.; Rhim, J.-W. Effect of Copper Salts and Reducing Agents on Characteristics and Antimicrobial Activity of Copper Nanoparticles. Mater. Lett. 2014, 132, 307–311. [Google Scholar] [CrossRef]
- Cioffi, N.; Ditaranto, N.; Torsi, L.; Sabbatini, L. Approaches to Synthesis and Characterization of Spherical and Anisotropic Copper Nanomaterials. In Nanotechnologies for the Life Sciences; Kumar, C.S.S.R., Ed.; Wiley: Hoboken, NJ, USA, 2008. [Google Scholar]
- Amendola, V.; Meneghetti, M. Laser Ablation Synthesis in Solution and Size Manipulation of Noble Metal Nanoparticles. Phys. Chem. Chem. Phys. 2009, 11, 3805. [Google Scholar] [CrossRef]
- Goncharova, D.A.; Kharlamova, T.S.; Lapin, I.N.; Svetlichnyi, V.A. Chemical and Morphological Evolution of Copper Nanoparticles Obtained by Pulsed Laser Ablation in Liquid. J. Phys. Chem. C 2019, 123, 21731–21742. [Google Scholar] [CrossRef]
- Begildayeva, T.; Lee, S.J.; Yu, Y.; Park, J.; Kim, T.H.; Theerthagiri, J.; Ahn, A.; Jung, H.J.; Choi, M.Y. Production of Copper Nanoparticles Exhibiting Various Morphologies via Pulsed Laser Ablation in Different Solvents and Their Catalytic Activity for Reduction of Toxic Nitroaromatic Compounds. J. Hazard. Mater. 2021, 409, 124412. [Google Scholar] [CrossRef]
- Barai, K.; Tiwary, C.S.; Chattopadhyay, P.P.; Chattopadhyay, K. Synthesis of Free Standing Nanocrystalline Cu by Ball Milling at Cryogenic Temperature. Mater. Sci. Eng. A 2012, 558, 52–58. [Google Scholar] [CrossRef]
- Saxena, R.; Kotnala, S.; Bhatt, S.C.; Uniyal, M.; Rawat, B.S.; Negi, P.; Riyal, M.K. A Review on Green Synthesis of Nanoparticles toward Sustainable Environment. Sustain. Chem. Clim. Action 2025, 6, 100071. [Google Scholar] [CrossRef]
- Abuzeid, H.M.; Julien, C.M.; Zhu, L.; Hashem, A.M. Green Synthesis of Nanoparticles and Their Energy Storage, Environmental, and Biomedical Applications. Crystals 2023, 13, 1576. [Google Scholar] [CrossRef]
- Thrush, B.A. The Exploding Wire Phenomenon. Nature 1960, 187, 726–727. [Google Scholar] [CrossRef]
- Lerner, M.I.; Shamanskii, V.V. Synthesis of Nanoparticles by High-Power Current Pulses. J. Struct. Chem. 2004, 45, S111–S114. [Google Scholar] [CrossRef]
- Kim, W.; Park, J.; Suh, C.; Lee, J.; Kim, J.; Oh, Y.-J. A New Method for the Production of Alloy Nanoparticles by Electrical Wire Explosion. Mater. Trans. 2007, 48, 1973–1974. [Google Scholar] [CrossRef]
- Sen, P.; Ghosh, J.; Abdullah, A.; Kumar, P. Vandana Preparation of Cu, Ag, Fe and Al Nanoparticles by the Exploding Wire Technique. J. Chem. Sci. 2003, 115, 499–508. [Google Scholar] [CrossRef]
- Pervikov, A.; Toropkov, N.; Kazantsev, S.; Bakina, O.V.; Glazkova, E.; Lerner, M. Preparation of Nano/Micro Bimodal Aluminum Powder by Electrical Explosion of Wires. Materials 2021, 14, 6602. [Google Scholar] [CrossRef]
- Lee, Y.S.; Tay, W.H.; Yap, S.L.; Wong, C.S.; Ahmad, Z.; Chia, S.-P.; Ratnavelu, K.; Muhamad, M.R. Copper Nanoparticle Synthesis by the Wire Explosion Technique. AIP Conf. Proc. 2009, 1150, 452–455. [Google Scholar]
- Oreshkin, V.I.; Baksht, R.B. Wire Explosion in Vacuum. IEEE Trans. Plasma Sci. 2020, 48, 1214–1248. [Google Scholar] [CrossRef]
- Park, S.; Lee, H.; Chung, K.-J. Liquid–Vapor Phase Transition Process during Underwater Electrical Wire Explosion. AIP Adv. 2023, 13, 065002. [Google Scholar] [CrossRef]
- Aravinth, S.; Sankar, B.; Chakravarthi, S.R.; Sarathi, R. Generation and Characterization of Nano Tungsten Oxide Particles by Wire Explosion Process. Mater. Charact. 2011, 62, 248–255. [Google Scholar] [CrossRef]
- Maqsood, K.; Ullah, S.; Rehman, A.U.; Bashir, H.; Shah, S.S.A.; Nazir, M.A. Recent Progress of Metallic Nanomaterials in Energy, Biomedical and Environment Sector. Next Res. 2025, 2, 100887. [Google Scholar] [CrossRef]
- Pervikov, A.; Lerner, M.; Krukovskii, K. Structural Characteristics of Copper Nanoparticles Produced by the Electric Explosion of Wires with Different Structures of Metal Grains. Curr. Appl. Phys. 2017, 17, 201–206. [Google Scholar] [CrossRef]
- Tkachenko, S.I.; Vorob’ev, V.S.; Malyshenko, S.P. The Nucleation Mechanism of Wire Explosion. J. Phys. Appl. Phys. 2004, 37, 495–500. [Google Scholar] [CrossRef]
- Lee, Y.S.; Bora, B.; Yap, S.L.; Wong, C.S. Effect of Ambient Air Pressure on Synthesis of Copper and Copper Oxide Nanoparticles by Wire Explosion Process. Curr. Appl. Phys. 2012, 12, 199–203. [Google Scholar] [CrossRef]
- Kwon, Y.-S.; Jung, Y.-H.; Yavorovsky, N.A.; Illyn, A.P.; Kim, J.-S. Ultra-Fine Powder by Wire Explosion Method. Scr. Mater. 2001, 44, 2247–2251. [Google Scholar] [CrossRef]
- Tokoi, Y.; Cho, H.-B.; Suzuki, T.; Nakayama, T.; Suematsu, H.; Niihara, K. Particle Size Determining Equation in Metallic Nanopowder Preparation by Pulsed Wire Discharge. Jpn. J. Appl. Phys. 2013, 52, 055001. [Google Scholar] [CrossRef]
- Bora, B.; Wong, C.S.; Bhuyan, H.; Lee, Y.S.; Yap, S.L.; Favre, M. Understanding the Mechanism of Nanoparticle Formation in Wire Explosion Process. J. Quant. Spectrosc. Radiat. Transf. 2013, 117, 1–6. [Google Scholar] [CrossRef]
- Kwon, Y.S.; An, V.V.; Ilyin, A.P.; Tikhonov, D.V. Properties of Powders Produced by Electrical Explosions of Copper–Nickel Alloy Wires. Mater. Lett. 2007, 61, 3247–3250. [Google Scholar] [CrossRef]
- Ju Park, E.; Won Lee, S.; Bang, I.C.; Park, H.W. Optimal Synthesis and Characterization of Ag Nanofluids by Electrical Explosion of Wires in Liquids. Nanoscale Res. Lett. 2011, 6, 223. [Google Scholar] [CrossRef]
- Kinemuchi, Y.; Sangurai, C.; Suzuki, T.; Suematsu, H.; Jiang, W.; Yatsui, K. Synthesis of Nanosize Powders of Aluminum Nitride by Pulsed Wire Discharge. In Proceedings of the PPPS-2001 Pulsed Power Plasma Science 2001. 28th IEEE International Conference on Plasma Science and 13th IEEE International Pulsed Power Conference. Digest of Papers (Cat. No.01CH37251), Las Vegas, NV, USA, 17–22 June 2001; IEEE: New York, NY, USA, 2001; Volume 2, pp. 1830–1833. [Google Scholar]
- Peng, C.; Wang, J.; Zhou, N.; Sun, G. Fabrication of Nanopowders by Electrical Explosion of a Copper Wire in Water. Curr. Appl. Phys. 2016, 16, 284–287. [Google Scholar] [CrossRef]
- Tkachenko, S.I.; Kuskova, N.I. Dynamics of Phase Transitions at Electrical Explosion of Wire. J. Phys. Condens. Matter 1999, 11, 2223–2232. [Google Scholar] [CrossRef]
- Antony, J.K.; Vasa, N.J.; Chakravarthy, S.R.; Sarathi, R. Understanding the Mechanism of Nano-Aluminum Particle Formation by Wire Explosion Process Using Optical Emission Technique. J. Quant. Spectrosc. Radiat. Transf. 2010, 111, 2509–2516. [Google Scholar] [CrossRef]
- Minin, I.V.; Uchaikin, S.; Rogachev, A.; Starý, O. (Eds.) Progress in Material Science and Engineering; Studies in Systems, Decision and Control; Springer International Publishing: Cham, Switzerland, 2021; Volume 351. [Google Scholar]
- Liu, H.; Zhao, J.; Zhang, R.; Guo, C.; Zhang, Q. Dimensional Effects of Electrically Exploding Aluminum Wires in Argon Gas: Experimental Investigation. J. Appl. Phys. 2020, 128, 073301. [Google Scholar] [CrossRef]
- Park, E.; Park, H.W.; Lee, J. Synthesis of Hierarchical Copper Oxide Composites Prepared via Electrical Explosion of the Wire in Liquids Method. Colloids Surf. A Physicochem. Eng. Asp. 2015, 482, 710–717. [Google Scholar] [CrossRef]
- Égerházi, L.; Kovács, B.; Szörényi, T. Spectroscopic Quantification of the Nanoparticle Production Efficiency of Copper Wire Explosion. J. Appl. Phys. 2021, 129, 195902. [Google Scholar] [CrossRef]
- Égerházi, L.; Szörényi, T. The Potential of Wire Explosion in Nanoparticle Production in Terms of Reproducibility. Materials 2024, 17, 3450. [Google Scholar] [CrossRef]
- Bäck, T.; Schwefel, H.-P. An Overview of Evolutionary Algorithms for Parameter Optimization. Evol. Comput. 1993, 1, 1–23. [Google Scholar] [CrossRef]
- Slowik, A.; Kwasnicka, H. Evolutionary Algorithms and Their Applications to Engineering Problems. Neural Comput. Appl. 2020, 32, 12363–12379. [Google Scholar] [CrossRef]
- Belete, D.M.; Huchaiah, M.D. Grid Search in Hyperparameter Optimization of Machine Learning Models for Prediction of HIV/AIDS Test Results. Int. J. Comput. Appl. 2022, 44, 875–886. [Google Scholar] [CrossRef]
- Stuke, A.; Rinke, P.; Todorović, M. Efficient Hyperparameter Tuning for Kernel Ridge Regression with Bayesian Optimization. Mach. Learn. Sci. Technol. 2021, 2, 035022. [Google Scholar] [CrossRef]
- Laven, P. MiePlot Software Download Site. Available online: http://www.philiplaven.com/mieplot.htm (accessed on 11 July 2024).
- Bohren, C.F.; Huffman, D.R. Absorption and Scattering of Light by Small Particles, 1st ed.; Wiley: Hoboken, NJ, USA, 1998. [Google Scholar]
- Johnson, P.B.; Christy, R.W. Optical Constants of the Noble Metals. Phys. Rev. B 1972, 6, 4370–4379. [Google Scholar] [CrossRef]
- Segelstein, D.J. The Complex Refractive Index of Water. M.Sc. Thesis, University of Missiouri-Kansas City, Kansas City, MO, USA, 1981. [Google Scholar]
- Tahir, D.; Tougaard, S. Electronic and Optical Properties of Cu, CuO and Cu2O Studied by Electron Spectroscopy. J. Phys. Condens. Matter 2012, 24, 175002. [Google Scholar] [CrossRef]
- Palik, E.D.; Palik, E.D. Handbook of Optical Constants of Solids, Author and Subject Indices for Volumes I, II, and III, 1st ed.; Elsevier: Amsterdam, The Netherlands, 1997. [Google Scholar]
- Drobny, V.F.; Pulfrey, L. Properties of Reactively-Sputtered Copper Oxide Thin Films. Thin Solid Films 1979, 61, 89–98. [Google Scholar] [CrossRef]
- Meyer, B.K.; Polity, A.; Reppin, D.; Becker, M.; Hering, P.; Klar, P.J.; Sander, T.; Reindl, C.; Benz, J.; Eickhoff, M.; et al. Binary Copper Oxide Semiconductors: From Materials towards Devices. Phys. Status Solidi B 2012, 249, 1487–1509. [Google Scholar] [CrossRef]
- Creighton, J.A.; Eadon, D.G. Ultraviolet–Visible Absorption Spectra of the Colloidal Metallic Elements. J. Chem. Soc. Faraday Trans. 1991, 87, 3881–3891. [Google Scholar] [CrossRef]
- Desarkar, H.S.; Kumbhakar, P.; Mitra, A.K. Effect of Ablation Time and Laser Fluence on the Optical Properties of Copper Nano Colloids Prepared by Laser Ablation Technique. Appl. Nanosci. 2012, 2, 285–291. [Google Scholar] [CrossRef]
- Kluczyk, K.; Jacak, W.A. Size Effect in Plasmon Resonance of Metallic Nanoparticles: RPA versus COMSOL. Acta Phys. Pol. A 2016, 129, A-83–A-86. [Google Scholar] [CrossRef]
- Scaffardi, L.B.; Schinca, D.C.; Lester, M.; Videla, F.A.; Santillán, J.M.J.; Ekeroth, R.M.A. Size-Dependent Optical Properties of Metallic Nanostructures. In UV-VIS and Photoluminescence Spectroscopy for Nanomaterials Characterization; Kumar, C., Ed.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 179–229. [Google Scholar]
- Melikyan, A.; Minassian, H. On Surface Plasmon Damping in Metallic Nanoparticles. Appl. Phys. B 2004, 78, 453–455. [Google Scholar] [CrossRef][Green Version]
- Xu, Y.; Deng, L.; Kuang, D.; Xie, H.; Shan, D.; Wang, X.; Huang, S.; Luo, H. Cu@C Core-Shell Nanoparticles with Efficient Optical Absorption: DDA-Based Simulation and Experimental Validation. Results Phys. 2020, 16, 102885. [Google Scholar] [CrossRef]
- Pò, C.L.; Iacono, V.; Boscarino, S.; Grimaldi, M.G.; Ruffino, F. Monte Carlo Approach to the Evaluation of Nanoparticles Size Distribution from the Analysis of UV-Vis-NIR Spectra. Micromachines 2023, 14, 2208. [Google Scholar] [CrossRef]
- Quintero, M.; Manrique-Moreno, M.; Riascos, H.; Torres-Palma, R.A.; Castro-Narvaez, S.; Ávila-Torres, Y.P. Laser Ablation for the Synthesis of Cu/Cu2O/CuO and Its Development as Photocatalytic Material for Escherichia coli Detoxification. Int. J. Mol. Sci. 2024, 25, 6817. [Google Scholar] [CrossRef]
- Mohammadparast, F.; Ramakrishnan, S.B.; Khatri, N.; Tirumala, R.T.A.; Tan, S.; Kalkan, A.K.; Andiappan, M. Cuprous Oxide Cubic Particles with Strong and Tunable Mie Resonances for Use as Nanoantennas. ACS Appl. Nano Mater. 2020, 3, 6806–6815. [Google Scholar] [CrossRef]
- Tyurnina, A.E.; Shur, V.Y.; Kozin, R.V.; Kuznetsov, D.K.; Pryakhina, V.I.; Burban, G.V. Synthesis and Investigation of Stable Copper Nanoparticle Colloids. Phys. Solid State 2014, 56, 1431–1437. [Google Scholar] [CrossRef]
- Ramakrishnan, S.B.; Khatri, N.; Addanki Tirumala, R.T.; Mohammadparast, F.; Karuppasamy, K.; Kalkan, A.K.; Andiappan, M. Cupric Oxide Mie Resonators. J. Phys. Chem. C 2022, 126, 16272–16279. [Google Scholar] [CrossRef]
- Ghodselahi, T.; Vesaghi, M.A.; Shafiekhani, A. Study of Surface Plasmon Resonance of Cu@Cu2O Core–Shell Nanoparticles by Mie Theory. J. Phys. D Appl. Phys. 2009, 42, 015308. [Google Scholar] [CrossRef]
- Liu, Z.; Xu, B.; Cheng, Y.; Si, M.; Chu, X.; Sun, M.; Fang, Y. Spectral Analysis of Oxidation on Localized Surface Plasmon Resonance of Copper Nanoparticles Thin Film. Spectrochim. Acta. A. Mol. Biomol. Spectrosc. 2023, 303, 123202. [Google Scholar] [CrossRef] [PubMed]
- Maiwald, T.; Timmer, J. Dynamical Modeling and Multi-Experiment Fitting with PottersWheel. Bioinformatics 2008, 24, 2037–2043. [Google Scholar] [CrossRef]
- Oliveto, P.S.; Witt, C. On the Runtime Analysis of the Simple Genetic Algorithm. Theor. Comput. Sci. 2014, 545, 2–19. [Google Scholar] [CrossRef]
- Bergstra, J.; Bengio, Y. Random Search for Hyper-Parameter Optimization. J. Mach. Learn. Res. 2012, 13, 281–305. [Google Scholar]
- Talam, S.; Karumuri, S.R.; Gunnam, N. Synthesis, Characterization, and Spectroscopic Properties of ZnO Nanoparticles. ISRN Nanotechnol. 2012, 2012, 372505. [Google Scholar] [CrossRef]
- Hong, S.-J.; Mun, H.-J.; Kim, B.-J.; Kim, Y.-S. Characterization of Nickel Oxide Nanoparticles Synthesized under Low Temperature. Micromachines 2021, 12, 1168. [Google Scholar] [CrossRef]
- Kayed, K. The Optical Properties of Individual Silver Nanoparticles in Ag/Ag2O Composites Synthesized by Oxygen Plasma Treatment of Silver Thin Films. Plasmonics 2020, 15, 1439–1449. [Google Scholar] [CrossRef]
- Sun, W.; Wei, W.; Liu, Q.; Yan, T.; Wang, Q.; Lin, H.; Tao, C.; Zhang, D.; Hong, R. Ag–Ag2O Composite Structure with Tunable Localized Surface Plasmon Resonance as Ultrastable, Sensitive and Cost-Effective SERS Substrate. J. Alloys Compd. 2020, 839, 155729. [Google Scholar] [CrossRef]
- Amendola, V.; Meneghetti, M. Size Evaluation of Gold Nanoparticles by UV−vis Spectroscopy. J. Phys. Chem. C 2009, 113, 4277–4285. [Google Scholar] [CrossRef]
- Gharibshahi, E.; Saion, E.; Ashraf, A.; Gharibshahi, L. Size-Controlled and Optical Properties of Platinum Nanoparticles by Gamma Radiolytic Synthesis. Appl. Radiat. Isot. 2017, 130, 211–217. [Google Scholar] [CrossRef]
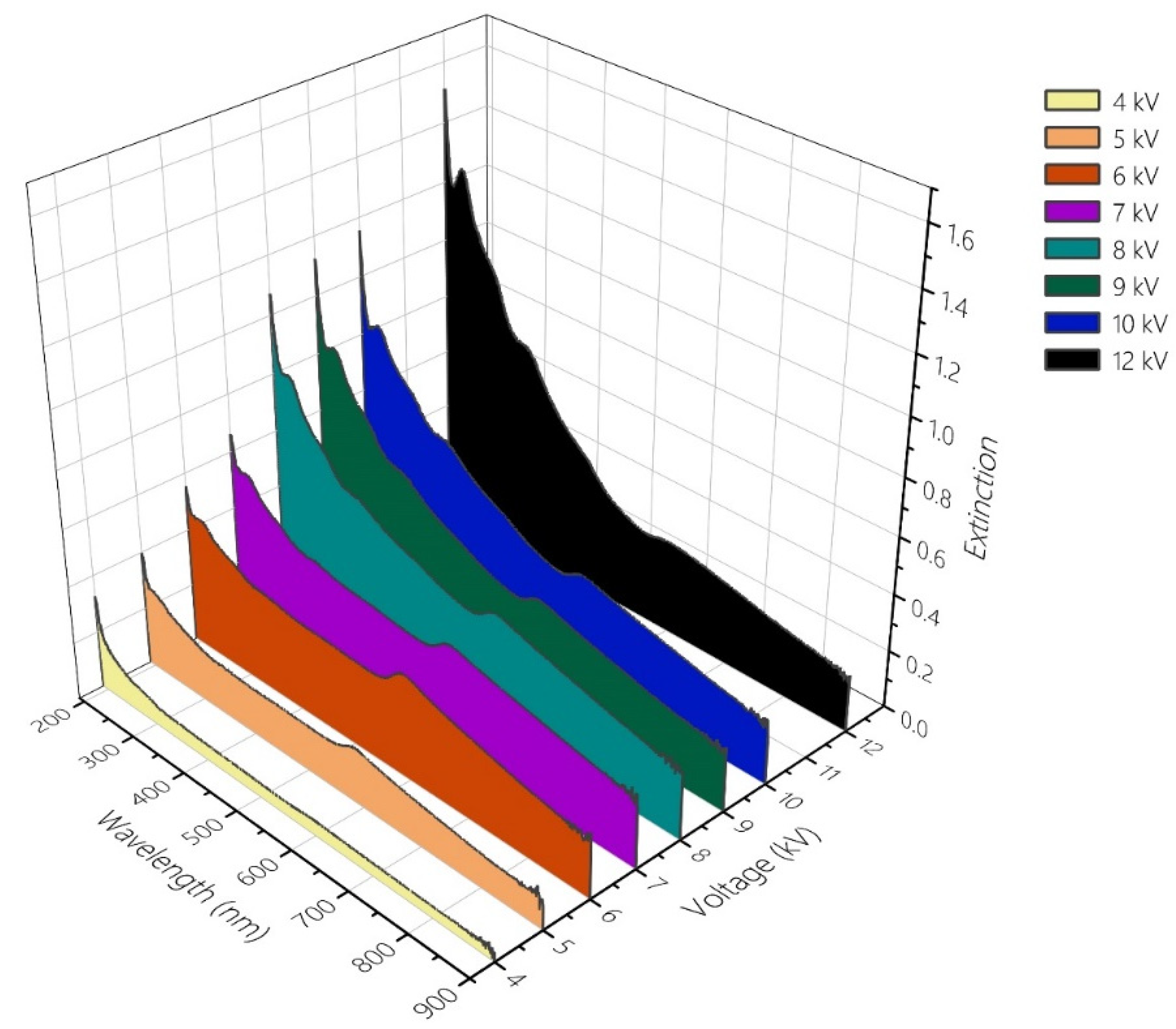




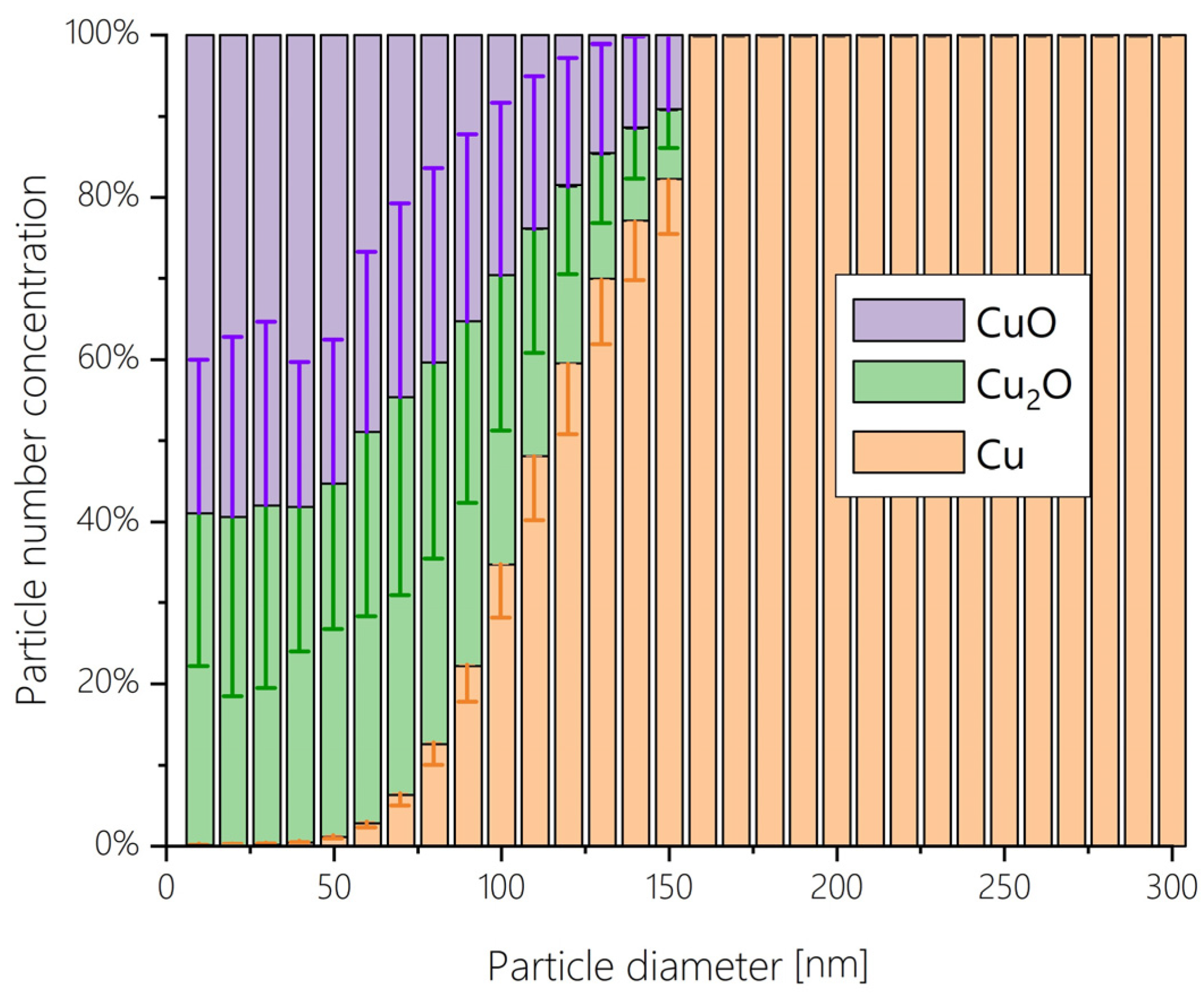
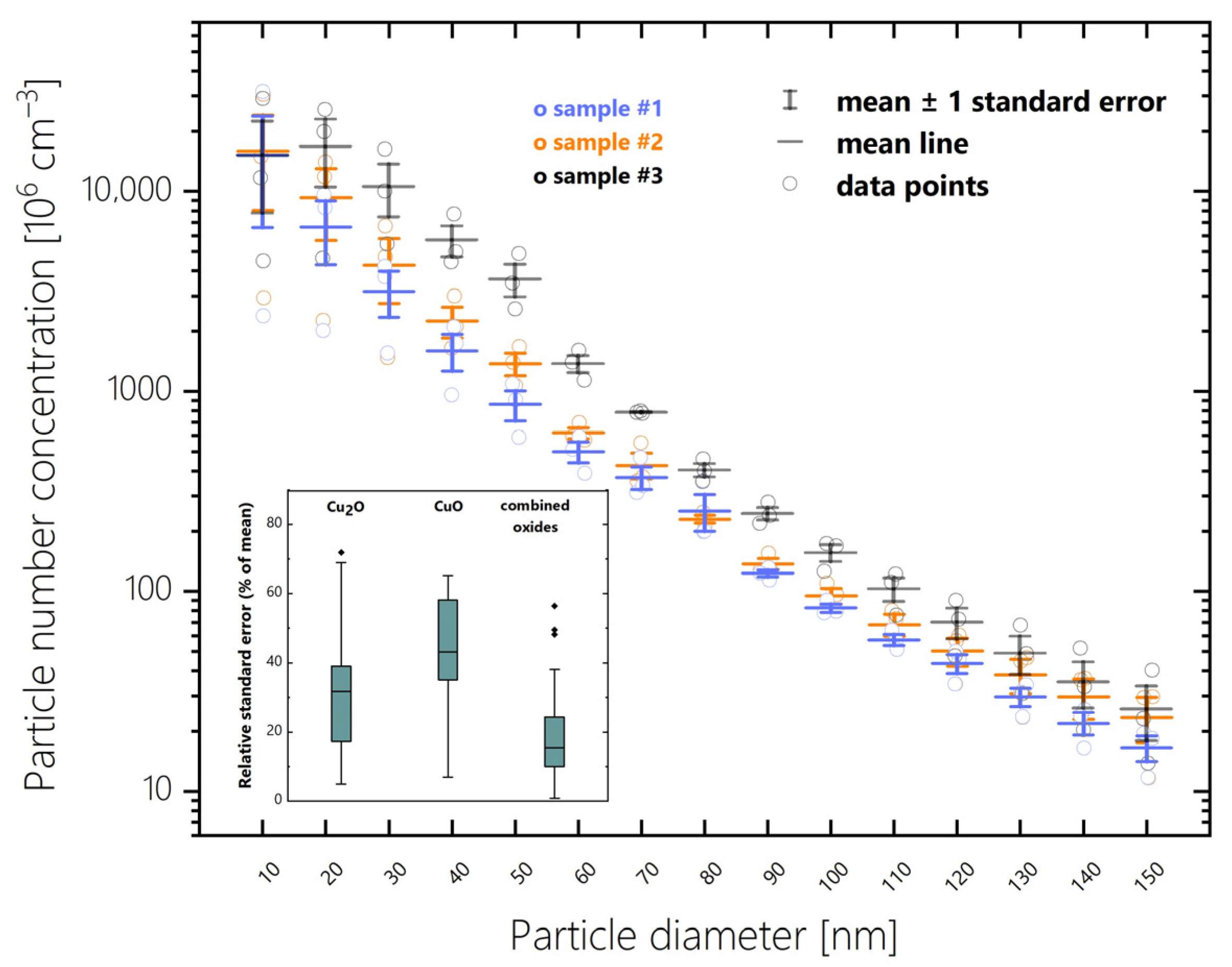

| Manual Fitting [42] | Semimanual Fitting | Evolutionary Algorithm | Grid Search | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| NRMSD | NRMSD | Cu Mean | Cu STD | NRMSD | Cu Mean | Cu STD | NRMSD | Cu Mean | Cu STD | |
| sample #1 | 1.81% | 1.90% | 145.4 nm | 146.3 nm | 2.03% | 143.8 nm | 149.6 nm | 1.95% | 145.9 nm | 140.5 nm |
| sample #2 | 2.27% | 2.42% | 160.1 nm | 118.5 nm | 2.09% | 169.6 nm | 122.1 nm | 1.98% | 161.5 nm | 142.7 nm |
| sample #3 | 1.73% | 1.71% | 152.2 nm | 137 nm | 2.42% | 151.3 nm | 148.3 nm | 1.89% | 150.4 nm | 147.2 nm |
| Discharge Voltage | NRMSD | Cu Mean | Cu STD |
|---|---|---|---|
| 4 kV | 6.62 ± 1.47% | 164 ± 33 nm | 116 ± 92 nm |
| 5 kV | 3.41 ± 0.77% | 123 ± 41 nm | 203 ± 146 nm |
| 6 kV | 2.00 ± 0.06% | 156 ± 5 nm | 139 ± 5 nm |
| 7 kV | 1.85 ± 0.19% | 177 ± 13 nm | 111 ± 28 nm |
| 8 kV | 1.81 ± 0.11% | 171 ± 1 nm | 151 ± 69 nm |
| 9 kV | 2.40 ± 0.86% | 178 ± 15 nm | 108 ± 8 nm |
| 10 kV | 2.26 ± 0.38% | 181 ± 15 nm | 112 ± 4 nm |
| 12 kV | 4.31 ± 0.55% | 172 ± 64 nm | 95 ± 61 nm |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Égerházi, L.; Griechisch, E.; Szörényi, T. Algorithmic Reconstruction of Multimodal Copper Wire Explosion Products from Extinction Spectra. Micro 2026, 6, 14. https://doi.org/10.3390/micro6010014
Égerházi L, Griechisch E, Szörényi T. Algorithmic Reconstruction of Multimodal Copper Wire Explosion Products from Extinction Spectra. Micro. 2026; 6(1):14. https://doi.org/10.3390/micro6010014
Chicago/Turabian StyleÉgerházi, László, Erika Griechisch, and Tamás Szörényi. 2026. "Algorithmic Reconstruction of Multimodal Copper Wire Explosion Products from Extinction Spectra" Micro 6, no. 1: 14. https://doi.org/10.3390/micro6010014
APA StyleÉgerházi, L., Griechisch, E., & Szörényi, T. (2026). Algorithmic Reconstruction of Multimodal Copper Wire Explosion Products from Extinction Spectra. Micro, 6(1), 14. https://doi.org/10.3390/micro6010014







