Experimental Characterization of Water Condensation Processes on Self-Assembled Monolayers Using a Quartz Crystal Microbalance with Energy Dissipation Monitoring
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Self-Assembled Monolayers (SAMs) on QCM-D Sensors and Water Contact Angle Measurements
2.2. QCM-D Measurement
2.3. QCM-D Data Analysis
2.3.1. Determination of the Mass and Thickness of Adsorbed Water on SAMs
2.3.2. Analysis of Viscoelasticity of Adsorbed Water
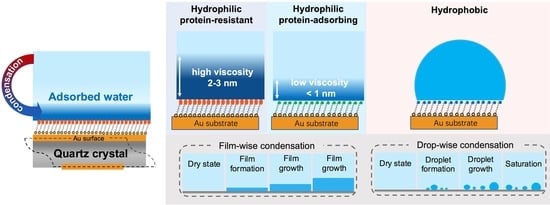
3. Results and Discussion
3.1. Kinetics of the Water Condensation onto SAMs
3.2. Interactions between Adsorbed Water and SAMs
3.3. The Viscoelasticity of Adsorbed Water on SAMs
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- James, M.; Ciampi, S.; Darwish, T.A.; Hanley, T.L.; Sylvester, S.O.; Gooding, J.J. Nanoscale Water Condensation on Click-Functionalized Self-Assembled Monolayers. Langmuir 2011, 27, 10753–10762. [Google Scholar] [CrossRef] [PubMed]
- James, M.; Darwish, T.A.; Ciampi, S.; Sylvester, S.O.; Zhang, Z.; Ng, A.; Justin Gooding, J.; Hanley, T.L. Nanoscale Condensation of Water on Self-Assembled Monolayers. Soft Matter 2011, 7, 5309–5318. [Google Scholar] [CrossRef]
- Bhardwaj, R.; Agrawal, A. How Coronavirus Survives for Days on Surfaces. Phys. Fluids 2020, 32, 111706. [Google Scholar] [CrossRef] [PubMed]
- Fedel, M.; Olivier, M.; Poelman, M.; Deflorian, F.; Rossi, S.; Druart, M.-E. Corrosion Protection Properties of Silane Pre-Treated Powder Coated Galvanized Steel. Prog. Org. Coat. 2009, 66, 118–128. [Google Scholar] [CrossRef]
- Barkhudarov, P.M.; Shah, P.B.; Watkins, E.B.; Doshi, D.A.; Brinker, C.J.; Majewski, J. Corrosion Inhibition Using Superhydrophobic Films. Corros. Sci. 2008, 50, 897–902. [Google Scholar] [CrossRef]
- Chen, L.; Qian, L. Role of Interfacial Water in Adhesion, Friction, and Wear—A Critical Review. Friction 2021, 9, 1–28. [Google Scholar] [CrossRef]
- Khawaji, A.D.; Kutubkhanah, I.K.; Wie, J.-M. Advances in Seawater Desalination Technologies. Desalination 2008, 221, 47–69. [Google Scholar] [CrossRef]
- Gomez-Garcia, F.; Gauthier, D.; Flamant, G. Design and Performance of a Multistage Fluidised Bed Heat Exchanger for Particle-Receiver Solar Power Plants with Storage. Appl. Energy 2017, 190, 510–523. [Google Scholar] [CrossRef]
- Muselli, M.; Beysens, D.; Milimouk, I. A Comparative Study of Two Large Radiative Dew Water Condensers. J. Arid Environ. 2006, 64, 54–76. [Google Scholar] [CrossRef]
- Phan, A.; Ho, T.A.; Cole, D.R.; Striolo, A. Molecular Structure and Dynamics in Thin Water Films at Metal Oxide Surfaces: Magnesium, Aluminum, and Silicon Oxide Surfaces. J. Phys. Chem. C 2012, 116, 15962–15973. [Google Scholar] [CrossRef]
- Wang, C.; Lu, H.; Wang, Z.; Xiu, P.; Zhou, B.; Zuo, G.; Wan, R.; Hu, J.; Fang, H. Stable Liquid Water Droplet on a Water Monolayer Formed at Room Temperature on Ionic Model Substrates. Phys. Rev. Lett. 2009, 103, 137801. [Google Scholar] [CrossRef] [PubMed]
- Santos, S.; Verdaguer, A.; Souier, T.; Thomson, N.H.; Chiesa, M. Measuring the True Height of Water Films on Surfaces. Nanotechnology 2011, 22, 465705. [Google Scholar] [CrossRef] [PubMed]
- Rykaczewski, K.; Scott, J.H.J.; Rajauria, S.; Chinn, J.; Chinn, A.M.; Jones, W. Three Dimensional Aspects of Droplet Coalescence during Dropwise Condensation on Superhydrophobic Surfaces. Soft Matter 2011, 7, 8749–8752. [Google Scholar] [CrossRef]
- Rykaczewski, K.; Scott, J.H.J. Methodology for Imaging Nano-to-Microscale Water Condensation Dynamics on Complex Nanostructures. ACS Nano 2011, 5, 5962–5968. [Google Scholar] [CrossRef]
- Rykaczewski, K. Microdroplet Growth Mechanism during Water Condensation on Superhydrophobic Surfaces. Langmuir 2012, 28, 7720–7729. [Google Scholar] [CrossRef]
- Rykaczewski, K.; Osborn, W.A.; Chinn, J.; Walker, M.L.; Scott, J.H.J.; Jones, W.; Hao, C.; Yao, S.; Wang, Z. How Nanorough Is Rough Enough to Make a Surface Superhydrophobic during Water Condensation? Soft Matter 2012, 8, 8786–8794. [Google Scholar] [CrossRef]
- Rykaczewski, K.; Paxson, A.T.; Anand, S.; Chen, X.; Wang, Z.; Varanasi, K.K. Multimode Multidrop Serial Coalescence Effects during Condensation on Hierarchical Superhydrophobic Surfaces. Langmuir 2013, 29, 881–891. [Google Scholar] [CrossRef]
- Bhattacharya, D.; Bosman, M.; Mokkapati, V.R.S.S.; Leong, F.Y.; Mirsaidov, U. Nucleation Dynamics of Water Nanodroplets. Microsc. Microanal. 2014, 20, 407–415. [Google Scholar] [CrossRef]
- Niu, D.; Tang, G.H. The Effect of Surface Wettability on Water Vapor Condensation in Nanoscale. Sci. Rep. 2016, 6, 19192. [Google Scholar] [CrossRef]
- Yang, S.; Zhang, Y.; Chen, L. Molecular Dynamics Study on the Effect of Surface Wettability on the Performance of Water Vapor Condensation. AIP Adv. 2019, 9, 025031. [Google Scholar] [CrossRef]
- Ranathunga, D.T.S.; Shamir, A.; Dai, X.; Nielsen, S.O. Molecular Dynamics Simulations of Water Condensation on Surfaces with Tunable Wettability. Langmuir 2020, 36, 7383–7391. [Google Scholar] [CrossRef] [PubMed]
- Benes, E. Improved Quartz Crystal Microbalance Technique. J. Appl. Phys. 1984, 56, 608–626. [Google Scholar] [CrossRef]
- Su, J.; Charmchi, M.; Sun, H. A Study of Drop-Microstructured Surface Interactions during Dropwise Condensation with Quartz Crystal Microbalance. Sci. Rep. 2016, 6, 35132. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, T.; Tanaka, Y.; Koide, Y.; Tanaka, M.; Hara, M. Mechanism Underlying Bioinertness of Self-Assembled Monolayers of Oligo(Ethyleneglycol)-Terminated Alkanethiols on Gold: Protein Adsorption, Platelet Adhesion, and Surface Forces. Phys. Chem. Chem. Phys. 2012, 14, 10196–10206. [Google Scholar] [CrossRef]
- Hayashi, T.; Hara, M. Nonfouling Self-Assembled Monolayers: Mechanisms Underlying Protein and Cell Resistance. Curr. Phys. Chem. 2011, 1, 90–98. [Google Scholar] [CrossRef]
- Björklund, S.; Kocherbitov, V. Humidity Scanning Quartz Crystal Microbalance with Dissipation Monitoring Setup for Determination of Sorption-Desorption Isotherms and Rheological Changes. Rev. Sci. Instrum. 2015, 86, 055105. [Google Scholar] [CrossRef]
- Sheng, Q.; Sun, J.; Wang, Q.; Wang, W.; Wang, H.S. On the Onset of Surface Condensation: Formation and Transition Mechanisms of Condensation Mode. Sci. Rep. 2016, 6, 30764. [Google Scholar] [CrossRef]
- Maruyama, S.; Kimura, T. A Study on Thermal Resistance over a Solid-Liquid Interface by the Molecular Dynamics Method. Available online: https://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.37.2275&rep=rep1&type=pdf (accessed on 18 February 2022).
- He, Y.; Chang, Y.; Hower, J.C.; Zheng, J.; Chen, S.; Jiang, S. Origin of Repulsive Force and Structure/Dynamics of Interfacial Water in OEG-Protein Interactions: A Molecular Simulation Study. Phys. Chem. Chem. Phys. 2008, 10, 5539–5544. [Google Scholar] [CrossRef]
- Mondarte, E.A.Q.; Zamarripa, E.M.M.; Chang, R.; Wang, F.; Song, S.; Tahara, H.; Hayashi, T. Interphase Protein Layers Formed on Self-Assembled Monolayers in Crowded Biological Environments: Analysis by Surface Force and Quartz Crystal Microbalance Measurements. Langmuir 2022, 38, 1324–1333. [Google Scholar] [CrossRef]
- Schwendel, D.; Hayashi, T.; Dahint, R.; Pertsin, A.; Grunze, M.; Steitz, R.; Schreiber, F. Interaction of Water with Self-Assembled Monolayers: Neutron Reflectivity Measurements of the Water Density in the Interface Region. Langmuir 2003, 19, 2284–2293. [Google Scholar] [CrossRef]
- Sekine, T.; Asatyas, S.; Sato, C.; Morita, S.; Tanaka, M.; Hayashi, T. Surface Force and Vibrational Spectroscopic Analyses of Interfacial Water Molecules in the Vicinity of Methoxy-Tri(Ethylene Glycol)-Terminated Monolayers: Mechanisms Underlying the Effect of Lateral Packing Density on Bioinertness. J. Biomater. Sci. Polym. Ed. 2017, 28, 1231–1243. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Hayashi, T.; Morita, S. The Roles of Water Molecules at the Biointerface of Medical Polymers. Polym. J. 2013, 45, 701–710. [Google Scholar] [CrossRef]
- Chang, R.; Asatyas, S.; Lkhamsuren, G.; Hirohara, M.; Mondarte, E.A.Q.; Suthiwanich, K.; Sekine, T.; Hayashi, T. Water near Bioinert Self-Assembled Monolayers. Polym. J. 2018, 50, 563–571. [Google Scholar] [CrossRef]
- Hayashi, T. Water at Interfaces: Its Behavior and Roles in Interfacial Phenomena. Chem. Lett. 2021, 50, 1173–1180. [Google Scholar] [CrossRef]
- Tanaka, M.; Morita, S.; Hayashi, T. Role of Interfacial Water in Determining the Interactions of Proteins and Cells with Hydrated Materials. Colloids Surf. B Biointerfaces 2021, 198, 111449. [Google Scholar] [CrossRef]
- Shao, Q.; He, Y.; White, A.D.; Jiang, S. Difference in Hydration between Carboxybetaine and Sulfobetaine. J. Phys. Chem. B 2010, 114, 16625–16631. [Google Scholar] [CrossRef]
- Pertsin, A.J.; Hayashi, T.; Grunze, M. Grand Canonical Monte Carlo Simulations of the Hydration Interaction between Oligo(Ethylene Glycol)-Terminated Alkanethiol Self-Assembled Monolayers. J. Phys. Chem. B 2002, 106, 12274–12281. [Google Scholar] [CrossRef]
- Dalvi, V.H.; Rossky, P.J. Molecular Origins of Fluorocarbon Hydrophobicity. Proc. Natl. Acad. Sci. USA 2010, 107, 13603–13607. [Google Scholar] [CrossRef]
- Mezger, M.; Sedlmeier, F.; Horinek, D.; Reichert, H.; Pontoni, D.; Dosch, H. On the Origin of the Hydrophobic Water Gap: An X-Ray Reflectivity and MD Simulation Study. J. Am. Chem. Soc. 2010, 132, 6735–6741. [Google Scholar] [CrossRef]
- Stein, M.J.; Weidner, T.; McCrea, K.; Castner, D.G.; Ratner, B.D. Hydration of Sulphobetaine and Tetra(Ethylene Glycol)-Terminated Self-Assembled Monolayers Studied by Sum Frequency Generation Vibrational Spectroscopy. J. Phys. Chem. B 2009, 113, 11550–11556. [Google Scholar] [CrossRef][Green Version]
- Araki, Y.; Sekine, T.; Chang, R.; Hayashi, T.; Onishi, H. Molecular-Scale Structures of the Surface and Hydration Shell of Bioinert Mixed-Charged Self-Assembled Monolayers Investigated by Frequency Modulation Atomic Force Microscopy. RSC Adv. 2018, 8, 24660–24664. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, T.; Tanaka, Y.; Usukura, H.; Hara, M. Behavior of Hydroxide Ions in Vicinity of Self-Assembled Monolayers of Alkanethiols on Metals. E-J. Surf. Sci. Nanotechnol. 2009, 7, 601–605. [Google Scholar] [CrossRef][Green Version]
- Zheng, J.; Li, L.; Tsao, H.-K.; Sheng, Y.-J.; Chen, S.; Jiang, S. Strong Repulsive Forces between Protein and Oligo (Ethylene Glycol) Self-Assembled Monolayers: A Molecular Simulation Study. Biophys. J. 2005, 89, 158–166. [Google Scholar] [CrossRef] [PubMed]






| Abbreviation | Chemical Structure | Static Water Contact Angle (°) |
|---|---|---|
| C8 | HS-(CH2)7-CH3 | 109 (2.3) 1 |
| CF3 | HS-(CH2)2-(CF2)7CF3 | 114 (3.5) |
| OH | HS–(CH2)11–OH | 17 (0.9) |
| EG3-OH | HS–(CH2)11–(O–CH2–CH2)3–OH | 37 (1.7) |
| SB | HS-(CH2)11-N+(CH3)2-(CH2)3-SO3− | 19 (1.4) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, S.; Latag, G.V.; Mondarte, E.A.Q.; Chang, R.; Hayashi, T. Experimental Characterization of Water Condensation Processes on Self-Assembled Monolayers Using a Quartz Crystal Microbalance with Energy Dissipation Monitoring. Micro 2022, 2, 513-523. https://doi.org/10.3390/micro2030033
Song S, Latag GV, Mondarte EAQ, Chang R, Hayashi T. Experimental Characterization of Water Condensation Processes on Self-Assembled Monolayers Using a Quartz Crystal Microbalance with Energy Dissipation Monitoring. Micro. 2022; 2(3):513-523. https://doi.org/10.3390/micro2030033
Chicago/Turabian StyleSong, Subin, Glenn Villena Latag, Evan Angelo Quimada Mondarte, Ryongsok Chang, and Tomohiro Hayashi. 2022. "Experimental Characterization of Water Condensation Processes on Self-Assembled Monolayers Using a Quartz Crystal Microbalance with Energy Dissipation Monitoring" Micro 2, no. 3: 513-523. https://doi.org/10.3390/micro2030033
APA StyleSong, S., Latag, G. V., Mondarte, E. A. Q., Chang, R., & Hayashi, T. (2022). Experimental Characterization of Water Condensation Processes on Self-Assembled Monolayers Using a Quartz Crystal Microbalance with Energy Dissipation Monitoring. Micro, 2(3), 513-523. https://doi.org/10.3390/micro2030033