Abstract
Ethylene, a gaseous phytohormone, is emerging as a central player in the intricate web of plant developmental processes from germination to senescence under optimal and stressed conditions. The presence of ethylene has been noted in different plant parts, including the stems, leaves, flowers, roots, seeds, and fruits. This review aims to provide a comprehensive overview of the regulatory impact of ethylene on pivotal plant developmental processes, such as cell division and elongation, senescence, abscission, fruit and flower development, root hair formation, chloroplast maturation, and photosynthesis. The review also encompasses ethylene biosynthesis and signaling: a snapshot of the regulatory mechanisms governing ethylene production. Understanding of the impact of ethylene’s regulatory functions on plant developmental processes has significant implications for agriculture, biotechnology, and our fundamental comprehension of plant biology. This review underscores the potential of ethylene to revolutionize plant development and crop management.
1. Introduction
Plant development is influenced by several intrinsic and extrinsic factors that coordinate to regulate all processes in the life cycles of plants. Plant growth is one carefully regulated process that occurs throughout the vegetative phase. Plant growth regulators act as regulators of plant developmental processes and are crucial for the resilience of responses under stress. Ethylene (ET) is a versatile hormone involved in development, metabolism, and stress responses in plants. Researchers have shown manifold roles of this hormone in the development of plants, as a signaling agent, in leaf development, in senescence, in fruit ripening, in the promotion of germination, etc. ET is the second most fundamental unsaturated hydrocarbon. It exhibits peculiar dose-dependent actions under ideal and external disruptions for all its suppression or promotion responses like germination, ripening, growth, and senescence. For instance, a low concentration of ET facilitates the activation of defense signaling in plants; its high concentration seems to inhibit development in Triticum aestivum and Cucumis sativus [1,2,3]. Plants growing in compacted soil experience excessive accumulation and concentration of ET in root tissues, resulting in root growth inhibition [4]. Increasing evidence confirms that ET is crucial in plant architecture. In Zea mays, ACS7 (1-aminocyclopropane-1-carboxylic acid synthases 7) mutation brought about a dwarf phenotype with a larger leaf angle on Sdw3 (semi-dwarf3) due to increased ET synthesis [5].
ET plays a central role in the growth and development process, starting from seed germination to senescence. However, complex crosstalk between phytohormones like abscisic acid (ABA), gibberellin (GA), cytokinin, and auxin regulates the responses sometimes antagonistically or synergistically [6,7]. ET biosynthesis or signaling mutant showed differential responses to hormonal sensitivity and plant developmental processes. For example, ethylene overproduction 3 (eto3) and constitutive triple response 1 (ctr1) mutants correspond to ABA insensitivity, whereas ethylene receptor 1 (etr1), ethylene-insensitive 2 (ein2), and ethylene-insensitive 6 (ein6) show enhancement of ABA sensitivity [8]. The faulty hook formation in ET-insensitive mutant ein2 signifies the prominence of ET-mediated auxin biosynthesis for hook development and regulation [9]. Moreover, EIN3 synchronizes the expression of chlorophyll biosynthesis genes PORA/B (PROTOCHLOROPHYLLIDE OXIDOREDUCTASE A/B) [10]: the pigment-binding proteins LHC which are essential for photosynthesis initiation. EIN3/EIL1 chiefly regulates ethylene signaling responses, thereby tuning the range of transcriptional regulation depending on spatiotemporal and environmental conditions [11]. Exogenous application of ET has been found to increase the number of lateral roots, root fresh weight, and mineral content in the root and shoot system, along with the upregulation of auxin biosynthesis and transportation genes [12]. In addition, ET has a significant effect on flower development and sex differentiation. ACS gene family has been widely studied and characterized for its potential function in sex determination [13] Fruit ripening is another ET-dependent process associated with multiple biological events like respiration, pigment accumulation, ET production, change in texture, and overall building up of fruit quality traits. ET activity participates in gene expression responsible for the aforementioned changes during ripening [14].
Importantly, ET is produced in response to various biotic and abiotic environmental stimuli, indicating that it connects environmental change and developmental adaptation [15,16,17]. Thus, under both normal and adverse conditions, ET confers plant growth and development. Moreover, ET’s role in postharvest fruit and vegetable management, impacting quality and shelf life, has been extensively explored through biochemical, technological, molecular, and metabolomic investigations. Recent reviews reveal its diverse and crucial functions, tailored to specific fruits and vegetables, making it essential to understand and control ET effects for different postharvest products [18,19,20]. Considering ET’s importance, we have tried to assemble the literature on recent advances and molecular insight into ET-mediated responses in regulating different phases of the plant life cycle. The research on ET’s role in plant processes has increased in recent years and reviews have also been published [6,21,22,23,24]. The previously published reviews predominantly focused on individual developmental processes regulated by ET like root hair development, senescence, germination, abscission, and fruit ripening. However, this review presents a holistic and all-encompassing view of the diverse ET-regulated processes within a single comprehensive framework. This approach provides a unified perspective, consolidating insights from various specialized reviews, which can help researchers to gain a more thorough understanding of ET’s effect on plant growth and development. With the overview of ET synthesis, signaling, and regulation of its biosynthesis, the details on ET-regulated processes are explained in the following areas: cell division and elongation, leaf and flower development, root hair development, fruit ripening, chloroplast development, photosynthesis, senescence, and abscission.
2. Ethylene Biosynthesis and Signaling
Ethylene biosynthesis involves a two-step enzymatic pathway starting with methionine, which is converted into S-adenosyl-L-methionine (SAM or S-AdoMet) by S-AdoMet synthetase. This transformation utilizes one ATP molecule. Subsequently, S-AdoMet is enzymatically converted into 1-aminocyclopropane-1-carboxylic acid (ACC) and 5′-methylthioadenosine (MTA) by ACC synthase (ACS) [25]. Lastly, ET is generated through the oxidation of ACC, facilitated by ACC oxidase (ACO). In parallel, the Yang cycle operates to convert MTA, a byproduct of the second stage, back into methionine, ensuring an optimal methionine pool [26]. Under normal basal ET production levels, ACS is believed to control the rate-limiting step in biosynthesis [25]. Nevertheless, specific circumstances can lead to ACO becoming the limiting factor [27]. Figure 1a illustrates the biosynthesis of ET.
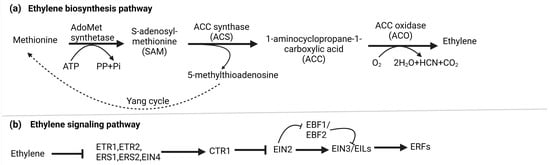
Figure 1.
(a) Biosynthesis of ethylene: In higher plants, ethylene is produced through the Methionine/Yang cycle. This involves converting methionine into 1-aminocyclopropane-1-carboxylic acid (ACC) using ACC synthase (ACS), and then ACC is further converted into ethylene by ACC oxidase (ACO). The Yang cycle recycles 5-methylthioadenosine to regenerate methionine, which is crucial for this process. (b) Signaling pathway of ethylene: Ethylene receptors (ETR1, ERS1, EIN4, ETR2, and ERS2) on the endoplasmic reticulum (ER) activate CTR1 kinase, which phosphorylates and degrades EIN2s C-terminal part, preventing its nucleus entry. Simultaneously, EBF1/2 transcripts produce F-box proteins that target EIN3/EIL transcription factors for degradation, blocking ethylene response. Ethylene binding deactivates CTR1, leading to EIN2 dephosphorylation. A protease cleaves EIN2s C-terminal part (EIN2-CEND) in the nucleus and cytoplasm. EIN2-CEND then enters the nucleus and binds to EIN3/EIL transcription factors, initiating gene expression for ethylene response.
The canonical ethylene signaling pathway begins when ethylene binds to ER-localized receptor proteins, such as ETHYLENE RESPONSE 1 (ETR1), ETHYLENE RESPONSE 2 (ETR2), ETHYLENE-INSENSITIVE 4 (EIN4), ETHYLENE RESPONSE SENSOR 1 (ERS1), and ERS2 in Arabidopsis, organized into two subfamilies defined by their ET binding and histidine kinase domains. Without ET, these receptors activate CONSTITUTIVE TRIPLE RESPONSE (CTR1), a Ser-Thr protein kinase on the ER membrane, which phosphorylates ETHYLENE-INSENSITIVE 2 (EIN2) C-terminal end (EIN2-CEND). F-box proteins ETP1 and ETP2 target phosphorylated EIN2 for degradation by the 26 S proteasome, preventing its nuclear translocation [28]. Similarly, ETHYLENE-INSENSITIVE 3 (EIN3) is subject to ubiquitin-mediated proteasomal degradation, guided by F-box proteins EBF1 and EBF2, thereby inhibiting ET responses. ET binds to its receptor when it is present, facilitated by copper ions from RESPONSIVE-TO-ANTAGONIST (RAN1). This binding deactivates CTR1, leading to the dephosphorylation and cleavage of EIN2-CEND [29]. EIN2-CEND subsequently obstructs the translation of EBF1 and EBF2 mRNA by transporting them to cytoplasmic P-bodies responsible for mRNA degradation. Furthermore, EIN2-CEND moves into the nucleus, activating EIN3 or EIL (EIN3-LIKE) transcription factors, culminating in the transcription of ET-responsive genes [30]. Furthermore, an alternative non-canonical signaling pathway has been suggested, which encompasses the regulatory proteins AHP (histidine-containing phosphotransfer proteins) and ARR (response regulator proteins) [31]. Figure 1b represents the summary of the process.
3. Snapshot of Regulation of Ethylene Biosynthesis
Since ACS and ACO are the exclusive enzymes involved in ET biosynthesis, most regulation concerning overall ET production revolves around modulating these pivotal enzymes’ transcription, translation, and protein stability. However, these enzymes are also affected by phytohormones and other stimuli like light and stress. In the early stages of research on the transcriptional regulation of ET biosynthesis, scientists uncovered the presence of a multigene family known as ACS genes, which exhibited distinct expression patterns in plants. Notably, four specific ACS genes in S. lycopersicum were pivotal in orchestrating the shift from autoinhibitory to autocatalytic ET production during fruit ripening [32]. Leading the charge as the initial regulator of ACS expression was the MADS-box transcription factor SlRIN, which directly bolstered the expression of selected S. lycopersicum ACS genes [33]. Over the years, numerous other transcription factors have emerged as key players, promoters, and inhibitors of ACS gene expression, influencing diverse growth processes. However, there is also supporting information suggesting that ET itself can directly influence ACS transcription. For instance, the ET response factor SlERF2/TERF2 of S. lycopersicum interacts with the promoter of NtACS3, thereby stimulating its expression [34]. The regulation of ET synthesis is further complicated by its interaction with light, phytohormones, and various biotic or abiotic stresses. The impact of light on ACS gene expression varies, contingent on developmental stages and light conditions. Notably, different ACS isozymes seem to have specific functions, with ACS5, 6, 8, and 9 primarily influencing ET production in dark-grown seedlings, while ACS2 and ACS4 assume control over ET production in well-lit conditions [35]. Mutations in the phytochrome genes PHYA and PHYB have been found to affect ET biosynthesis, with phyA mutants displaying a more pronounced increase in ET production. Furthermore, transgenic lines engineered to overexpress Arabidopsis PHYTOCHROME-INTERACTING FACTOR5 (PIF5) have exhibited enhanced ET production, underscoring the role of PIFs in the regulation of ET biosynthesis, which appears to be stage-dependent [35].
Much like their ACS counterparts, ACO genes are subject to transcriptional regulation. Notably, the S. lycopersicum HD-ZIP transcription factor SlHB-1 and the ripening regulator RIN directly stimulate ACO gene expression. Across various species, different classes of transcription factors have also been shown to regulate ACO. Interestingly, ET can directly control ACO expression through intricate feedback mechanisms mediated by ERF (ethylene response factor) proteins [36]. In S. lycopersicum, ACO1 experiences upregulation in response to white light pulses. This phenomenon propels ET production and serves as a reporting mechanism for ET responses, thus establishing a positive feedback loop. Additionally, ACO transcript levels surge after ACC treatment of light-grown seedlings. During phases of ripening or stress, when ACS activity is maximum, ACO activity becomes rate-limiting, prompting an upswing in ACO expression. This orchestrated feed-forward mechanism is pivotal in eliminating excess ACC when ACO activity imposes restrictions on ET production [35].
Transcriptional regulation of ET biosynthesis is complemented by posttranslational modifications of ACS proteins. ACS proteins share a conserved N terminus and catalytic core, there’s notable variability in the C terminus among different ACS isoforms. This variability leads to the classification of ACS proteins into three major groups in Arabidopsis: type 1 (ACS1, 2 and 6), type 2 (ACS4, 5, 8, 9, and 11), and type 3 (ACS7). Type 1 has a phosphorylation site for mitogen-activated protein kinases (MAPKs) and calcium-dependent protein kinases (CDPKs). Type 2 has a site for CDPKs and ETO1 (Ethylene Overproducer1)/ETO1-LIKE1 (EOL), an E3 ligase leading to degradation. Type 3 has no target site. Phosphorylation stabilizes type 1 ACS, promoting ET production. Conversely, dephosphorylation can lead to ACS degradation, although this effect depends on the type of ACS [36]. Moreover, phosphorylation can destabilize type 2 ACS, as exemplified by the phosphorylation of ACS5 (type 2) by Casein Kinase 1.8 (CK1.8), resulting in E3 ubiquitin ligases mediated degradation [37]. Light-triggered posttranslational regulation of type-2 ACSs, especially ACS5, regulating hypocotyl elongation during the dark-to-light shift [29]. PIF3 plays a key role, with ET stabilizing PIF3 in light. This light-stimulated stabilization of ACS enzymes increases ET production, potentially contributing to PIF3 stability and the subsequent promotion of ET-driven hypocotyl elongation under light exposure [38]. PP2A, a regulatory component, plays a role in posttranslational ACS stability regulation. PP2A-mediated dephosphorylation negatively affects ACS6 protein stability in the dark [39]. Posttranslational regulation of ACS also involves ubiquitination, with type 3 ACS7 being targeted for degradation through ubiquitination mediated by XBAT32 (XB3 orthologue 2 in Arabidopsis (Figure 2a). Protein Phosphatase 2C family members (PP2C’s) also play a role in stabilizing ACS7 [36].
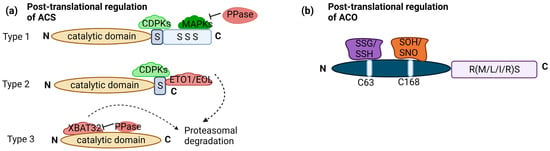
Figure 2.
Posttranslational regulation of ACS and ACO: (a) ACS enzymes can be categorized into three groups based on their phosphorylation sites: type 1 with both CDPK (calcium-dependent protein kinase) and MAPK (Mitogen-activated protein kinase) sites, type 2 with only CDPK sites, and type 3 with no C-terminal regulatory sites. Type 1 ACS is positively regulated by MAPKs and CDPKs, significantly boosting its activity and ethylene production. Phosphatases (PPase) are involved in dephosphorylation, negatively impacting protein stability. Type 2 ACSs are regulated by the ETO1-containing E3 ligase, recognizing the TOE (Target of ETO1) domain in their C-termini. CDPK phosphorylation likely plays a role in regulating this ubiquitination process and, thus, the stability of Type 2 ACS protein. XBAT32 directly binds to type 3 ACS, leading to protein degradation, while PPase plays a role in stabilizing. (b) ACO enzymes fall into three related groups based on amino acids in the RXS motif: type 1 with RMS intermediate residue, type 2 with R-L/I-S intermediate residue, and type 3 with RRS intermediate residue. Type 1 ACOs can undergo modifications at C63, including S-glutathionylation (SSG) and S-sulfhydration (SSH), as well as at C168, including S-sulfenylation (SOH) and S-nitrosylation (SNO). No such modifications have been observed in type 2 and type 3 ACOs. The figure is based on and modified from [36]. Dotted arrow—multistep pathway; solid arrow—promotion; flat-head arrow—inhibition.
While much is known about the posttranslational regulation of ACS, research on the posttranslational regulation of ACO has been comparatively limited. It is worth mentioning that the three categories of ACO can be distinguished based on the specific intermediate amino acid present within the conserved RXS motif. Type 1 ACOs contain an RMS intermediate residue, type 2 ACOs possess an R-L/I-S intermediate residue, and type 3 ACOs feature an RRS intermediate residue. ACO proteins undergo redox-specific posttranslational modifications. These modifications involve cysteine residues and include S-glutathionylation (SSG), S-sulfhydration (SSH) at cysteine (C63), S-sulfenylation (SOH), and S-nitrosylation (SNO) at C168. These modifications are recorded only in type 1 ACO (Figure 2b). Recent studies have highlighted the significance of these modifications in controlling ACO activity and structural stability [36].
4. Involvement of Ethylene in Plant Developmental Processes
Ethylene plays various roles in regulating cellular, molecular, and whole-plant metabolism. It influences the performance of plants under optimal and stressful environments by interacting with other signaling molecules. The impact of ET is contingent on its concentration within the cell and the plant’s sensitivity to this hormone. Ethylene has garnered attention due to its dual influence on controlling plant processes at both physiological and molecular levels [40,41].
4.1. Cell Division and Elongation
The role of ET in cell division is complex. Ethylene affects cell division depending on the specific tissue type and the internal and external signals at play. In certain situations, ET acts as a stimulator of cell division. For example, during the development of the apical hook and during the early stages of apical hook development, ethylene appears to play a synergistic role with auxins in stimulating cell division within the subepidermal layers. This collaborative effect is crucial for the bending of the apical hook. Specifically, ET is believed to facilitate the cell’s expansion in the hooks convex side contributing to its curvature [42]. Additionally, ET has been found to control cell division rate in vascular tissue, promoting vascular cell differentiation. Mutants like eto1 and eto2 (ethylene overproducer) enhance vascular cell division. Emphasizing the necessity of ET signaling transcription factors, ERF018 and ERF109 further boost cell division during vasculature development in Arabidopsis stem [43]. In this specific context, ET has a positive impact on cell division.
However, the role of ET in cell division is not uniform across all plant tissues. Ethylene’s effects on cell division in the root system are somewhat contradictory. Studies have shown that ET does not significantly alter the expression pattern of CYCLIN-DEPENDENT PROTEIN KINASE B1;1 (CYCB1;1), pointing out that it does not directly affect mitotic activity in the root. However, it is important to note that ET modulates cell division within the quiescent center, developing additional columella layers in the root cap [44,45]. Moreover, ET control over the root apical meristem (RAM) size and cell number depends on CULLIN3-type E3 ligases, demonstrating an ET-dependent regulatory mechanism [46]. In addition, loss of function mutant ctr1-2 results in a reduced meristematic zone. Conversely, ethylene-insensitive mutant etr1-1 exhibits an extended meristematic zone relative to wild-type plants [47]. Ethylene regulates RAM size through the canonical pathway involving CTR1, EIN2, and EIN3/EIL, and an alternative pathway with ARR1. This highlights ET’s complex role in controlling root apical meristem size and proliferation [48].
The impact of ET on leaf cell division is context-dependent. Under environmental stress, particularly when plants experience less than 10 h of osmotic stress, ET mediates transient and reversible cell cycle cessation. This effect is thought to involve the phosphorylation-mediated inactivation of CYCLIN-DEPENDENT KINASE A (CDKA), possibly via the MPK3/6 pathway. Notably, this cell cycle arrest operates independently of the EIN3 transcriptional control [49]. Cell cycle inhibition by ET in leaves is multifaceted; ET accumulation activates BOLITA, an ERF, activating type II TCP (TEOSINTE BRANCHED 1/CYCLOIDEA/PCF) genes [50]. These TCP proteins then bind to the promoter of RBR1 (RETINOBLASTOMA RELATED 1), phosphorylating E2Fa and repressing E2F target gene transcription, thereby impeding progression into the S-phase and cell division [51]. Furthermore, ET induces the expression of ERF5 and ERF6 in stressed leaves. ERF6, in turn, prompts the expression of GA2-OX6 (GA2-OXIDASE6), reducing active GA levels and accumulating DELLA proteins, which repress DEL1 and UVI4 (regulates the transition from the mitotic cell cycle to endoreduplication) gene expression, resulting in an early exit from the cell cycle [51,52]. Furthermore, ET restricts the mitotic cell cycle within the leaf petiole’s abaxial cells. This inhibition is achieved by suppressing the expression of CYCLIN2A;1, thereby partially playing a role in the hyponastic phenomenon [53]. These findings provide comprehensive insights into ET’s intricate control of cell division.
Cellular growth relies on key processes like the rearrangement of the cytoskeleton, the modification of the cell wall facilitated by cell-wall-remodeling enzymes, and water uptake via aquaporins. Cell elongation involves rearranging cortical microtubules (CMTs) perpendicular to the growth axis [54]. Ethylene rapidly alters CMT orientation in Arabidopsis roots and hypocotyls, inhibiting cell elongation and promoting radial swelling within 10 min [54]. Conversely, ET induces petiole elongation in well-lit conditions by reorienting CMT from longitudinal to transverse in abaxial cells [55], excluding adaxial cells. In ET-regulated cellular elongation, expansins (EXP) and xyloglucan endotransglycolases/hydrolases (XTH) play pivotal roles. ET induces EXP expression in Arabidopsis, influencing root hair formation and submergence–escape elongation [54]. However, in Arabidopsis leaf with elevated EIN2 expression, EXP3, and EXP5 expression was reduced, while in ein2 knockout plants, these genes were upregulated. Additionally, the expression of EXP1 and EXP5 was significantly suppressed in dwarfed BOLITA (ERF) gain-of-function plants [50,56]. These findings demonstrated a tissue-specific linkage between ET and expansins in the regulation of cell elongation. Ethylene regulates XTH expression during root hair initiation and submergence-induced hyponastic responses [54]. A report showed that ET serves as a negative regulator of cell elongation in the primary root of Arabidopsis by impeding the uptake, accumulation, and distribution of gibberellic acid in the endodermis of the elongation zone [57]. Additionally, cytokinin influences cell elongation, partly through an ET-dependent pathway, as evidenced by the reduced cell elongation response to cytokinins in the ethylene-insensitive ein2 mutant compared to the wild type [47].
4.2. Leaf Growth and Flower Development
Ethylene overproduction in Arabidopsis resulted in dwarfed plants with reduced growth [58,59]. Accordingly, when positive regulators of the ET signaling pathway in plants were mutated, these plants exhibited rosette leaves compared to control plants. Mutation of the endoplasmic reticulum (ER)-anchored protein EIN2, for instance, has been linked to increased growth [56]. On the contrary, mutations in components that inhibited ET signaling, like the receptors ETR1 and ERS1, resulted in stunted growth. Furthermore, when the negative regulators ARGOS or ARGOS-LIKE (ARL) were excessively expressed, it promoted leaf growth in Arabidopsis [60,61]. Another relevant study states that certain rhizosphere bacteria enhance plant growth by expressing ACC-DEAMINASE, reducing ACC levels in stressed plants [62]. However, lower concentrations of ET have been reported to promote leaf growth. For instance, Poa alpina and Poa compressa showed increased leaf elongation rates [63], and the primary leaves of Helianthus annuus were enlarged following ET exposure [64]. However, the effect was altered by increasing ET concentration. Furthermore, studies also showed that the application of ethephon increased the leaf area [65,66]. Certain reports also observed no change in the leaf area of ET-insensitive Arabidopsis, Nicotiana, and Petunia relative to ET-sensitive plants [67]. Thus, it can be inferred that the effect of ET on leaf growth and development was influenced by the concentration of ET used and the specific plant species under investigation [7,41].
The regulatory mechanism of sex determination in plants represents a model experiment system in the case of unisexual plants [68]. Considerable focus has been directed towards identifying genes responsible for regulating the development of male and female flowers. Although various plant hormones were investigated for their impact on the proportion of unisexual flowers, ET emerged as a prominent regulator of unisexual flower development [69]. The primary genes determining flower sex type encode crucial enzymes engaged in ethylene production, with Wiskott–Aldrich syndrome protein-interacting protein (WIP1), which encodes a C2H2 zinc finger transcription factor of the WIP family. The M (Monoecious) gene encodes ACS2, expressed in the carpel region of the female flower; its inactivation (m) leads to the development of a bisexual flower [70]. ACO2 has also been demonstrated to impact the formation of unisexual flowers by collaborating with ACS11 to promote the selective production of ET in the floral region; dysfunctional ACO2 results in the absence of female flowers [71]. Trebitsh et al. [72] discovered that the F (Female) locus contains an additional copy of ACS1, suggesting an initial connection between ET biosynthesis and female flower development. In Cucumis sativus, plants carrying the F locus duplication, ACS1G was expressed early in floral bud development, triggering ET burst when functioning with ACS2. Following the initiation of gynoecy, ET engages in the repression of WIP1 and activation of ACS2. This leads to the early expression of ACS1G, bypassing the requirement for ACS11 in ET production and establishing a dominant pathway for female floral development [70]. A study observed that in Cucumis melo, the WIP1, employed a corepressor TOPLESS to inhibit CRC (CRABS CLAW, carpel identity gene) expression through histone deacetylation, leading to male flower development. Disruption in the interaction between TOPLESS and WIP1 resulted in the expression of CRC, leading expression of the stamina inhibitor CmACS7, ultimately promoting female flower development [73]. Genome-wide analysis of the ET-responsive ACS gene family in Cucumis melo and C. lanatus revealed that ACS1s and ACS6s are mainly responsible for sex determination. Most of the genes are responsive to exogenous ethephon; however, ACS1, 9 and 10 show the opposite expression pattern [13]. Gene editing of ACO2 in Zea mays leads to decreased ET production during ear development. This alteration facilitates the enhancement of meristem and flower development, resulting in a significant boost of approximately 13.4% in grain yield per ear in hybrid lines [74]. Recent reports proposed that an integrated genetic network of both ET biosynthesis and receptor genes regulates sex determination. In C. pepo, the extent of ethylene insensitivity is influenced by both the potency and quantity of mutant alleles in at least three collaborating ETHYLENE RESPONSE (ETR) genes, and this level of ET insensitivity was a key factor in determining the ultimate sexual phenotype of the plant [75]. CpACO1A, CpACS11A, CpACO2B, and CpACS271A were found to be associated with female flowering transition and development as well as stamen arrest [76]. To understand how unisexual flowers develop in Cucumis melo, a mechanism was reported where ET originating from the carpel was sensed by ET receptors with distinct spatial expression in the stamen primordia. This leads to the activation of the CmEIN3/CmEIL1 ethylene signaling module within the stamen primordia. Consequently, this module triggers the expression of CmHB40, a transcription factor that downregulates genes essential for stamen development while upregulating genes associated with organ senescence [77]. Ethylene enhances petal maturation and flower opening through jasmonic acid biosynthesis but hinders ovary growth by reducing auxin production [76]. A recent study using the jasmonate-deficient mutant lox3a 9 (LIPOXYGENASE, gene associated with jasmonate synthesis) confirmed the interplay between jasmonate and ET in regulating the opening of male and female flowers during their development [78]. A study in Cucumis melo identified a novel gene, CmCPR5, within locus b, regulating bisexual flower development. CmCPR5 interacts with ET receptor CmETR1, impacting ET signaling. Malfunction of CmCPR5 prevents stamen inhibition, leading to bisexual flower formation [79]. The description is illustrated in Figure 3.
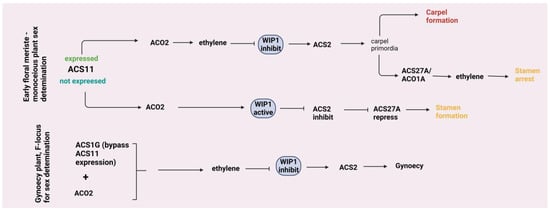
Figure 3.
Flower development: In monoecious Cucumis sativus (cucumber) plants, the presence or absence of ACS11 plays a crucial role in determining the sex of the flowers. When ACS11 is expressed in the floral meristem, it produces ethylene, leading to the development of female flowers by suppressing WIP1 (Wiskott–Aldrich syndrome protein-interacting protein, encodes a C2H2 zinc finger transcription factor) and activating ACS2. Later, ACO1A and ACS27A produce ethylene, causing stamen arrest. In buds without ACS11 expression, the male development program takes over. However, the presence of the F-gene (ACS1G) ensures the production of female flowers, conferring gynoecy, regardless of ACS11, by directly upregulating ACS2 through its interaction with ACO2 and promoting carpel development. Solid arrow—promotion; flat-head arrow—inhibition.
4.3. Root Hair Development
Root hairs represent an extensive network of epidermal cells within the root system, playing a vital role in nutrient uptake, anchoring the plant in the soil, and facilitating interactions with the environment in stationary plants. The plant hormone ET not only fosters the growth of these root hairs but also acts as a mediator for various signals that trigger the development of hair cells [21]. Ethylene’s role in Arabidopsis root hair formation is elucidated through ET biosynthesis mutants. Root hairs of eto1, eto2, and eto4 mutants develop longer hairs than wild-type ones on trichoblasts cells. Interestingly, the eto3 mutant stands out as it produces higher levels of ET than other eto alleles. Remarkably, it also triggers the development of hairs on atrichoblasts, which are typically hairless cells [80]. Mutations in genes involved in ET signaling confirm ET necessity for root hair elongation; for example, etr1 and ein2 receptor mutations yield shorter root hairs (50–70% of wild type), while ctr1 loss-of-function mutations produce longer root hairs. This underscores ET’s crucial role in controlling root hair length [80]. In Arabidopsis, root epidermal cell fate depends on their position relative to cortical cells. Hair (H) cells form over the junction of two cortical cells, while nonhair (N) cells develop over a single cortical cell. A transcriptional cascade drives the core regulatory network. The genes involved in root hair cell fate determination in root epidermal cells of Arabidopsis include TRANSPARENT TESTA GLABRA (TTG), GLABRA3 (GL3), ENHANCER OF GLABRA3 (EGL3), WEREWOLF (WER), GLABRA2 (GL2) and CAPRICE (CPC), and its homologs TRIPTYCHON (TRY) and ENHANCER OF TRY AND CPCs (ETC) [81]. The WER-GL3/EGL3-TTG complex induces GL2 and CPC expression in N-type cells, inhibiting root hair growth. CPC moves to adjacent H-type cells, competing with WER, inhibiting GL2 expression, and promoting H-type cell differentiation [81]. GLABRA 2 (GL2), a crucial transcription factor, maintains N cell fate by repressing a group of basic helix-loop-helix (bHLH) factors that promote root hair development in H cells. ROOT HAIR DEFECTIVE 6 (RHD6) and its homolog RHD6-LIKE 1 (RSL1) are key players in this process, positively regulating other bHLH factors (RSL2–5) that enhance root hair growth, with RSL4 being directly regulated by RHD6 [82]. The ET-activated transcription factors EIN3/EIL1 interact with ROOT HAIR DEFECTIVE 6 (RHD6)/RHD6-LIKE 1, leading to the direct coactivation of the RSL4 (ROOT HAIR DEFECTIVE 6-LIKE 4) gene, thereby enhancing the growth of root hairs [82]. A recent study showed that EIN3 influences the WER-GL3-TTG1 complex formation by competing with GL3 for TTG1 binding, consequently reducing GL2 transcription via diminished WER-GL3-TTG1 complex formation and inducing the development of root hairs [83]. Furthermore, MYB30, belonging to the MYB protein family, functions as a plant transcription factor with a negative role in root hair elongation. MYB30 directly interacts with the RSL4 promoter region, suppressing its transcription and inhibiting root hair elongation. Ethylene promotes the formation of a complex between EIN3 and MYB30 by decreasing the interaction between MYB30 and the RSL4 promoter. The activation of RSL4 transcription ultimately stimulates the elongation of root hair [84]. A study centered on C2H2 family proteins specifically highlighted the role of ZINC FINGER PROTEIN 5/6 (ZFP5/6) within this family. They confirmed that ZFP5/6 serves a dual function, acting as a downstream component of gibberellins, ethylene, and cytokinins while also operating as an upstream regulator of the CAPRICE (CPC)/GLABRA3/ENHANCER OF GLABRA3 (GL3/EGL3)/TRANSPARENT TESTA GLABRA1 (TTG1) complex in the control of root hair development [85]. The study aimed to understand ET’s role in root hair initiation when the normal differentiation pattern of root epidermal cells is disrupted and showed that CPC and TRY are crucial for root hair formation. Interestingly, exogenous ET triggered root hair formation in the cpc mutant at certain positions but did not affect the cpc try mutant. This suggests that ET-induced root hair initiation depends on the functionality of the CPC-TRY complex, even if it is partially functional [86]. Applying phytohormones like auxin and cytokinin induces root hair growth in a dependent manner. Ethylene’s influence on root hair development could be, at least partially, linked to the auxin pathway. RSL4 was pivotal in bridging the ET and auxin pathways during root hair development. Multiple AUXIN SIGNALING F-BOX genes induced by auxin directly bind to the promoter of RSL4, activating its expression and facilitating root hair elongation [87]. Additionally, the initiation of root hairs induced by low pH (acidification), which involves cortical microtubule randomization, was associated with elevated auxin synthesis and its subsequent effects [88]. Analysis of the transcriptome showed that both auxin and ET could increase the expression of 90% of the 208 genes associated with root hair growth and development [89]. In comparison to the wild type, the auxin-insensitive mutant axr1 had shorter root hairs, which could be restored with exogenous ACC. The ET-insensitive mutant ein2-1 exhibited inhibited root hair growth, which was ameliorated by applying exogenous naphthalene acetic acid. The ET overproducing mutant eto1 displayed longer root hairs, while the loss of AUX1 in the eto1 showed reduced root hair length [90,91,92]. An auxin efflux carrier PIN2 (PIN-FORMED2) plays a crucial role in creating the right auxin gradient in root tips, which is essential for root hair growth. A recent study introduces SAV4 (SHADE AVOIDANCE 4), a novel regulator that influences PIN2 abundance membrane clustering, and stability through direct interactions and regulates polar auxin transport. Studies showed that ET treatment increases the SAV4 protein level, which leads to a decrease in the breakdown of PIN2. The higher levels of PIN2 on the cell membrane of the epidermis enhance the transport of auxin towards the base of the root, consequently stimulating root hair development [93]. Often, ET and auxin appear to be closely interconnected, resulting in cooperative and complementary impacts on root hair development. Ethylene-induced auxin activity plays a substantial role in promoting root hair initiation. In contrast, the elongation of root hairs necessitates a synergistic interaction between auxin and ethylene, utilizing their canonical signaling pathways [92]. Figure 4 illustrates ethylene-regulated root hair development.
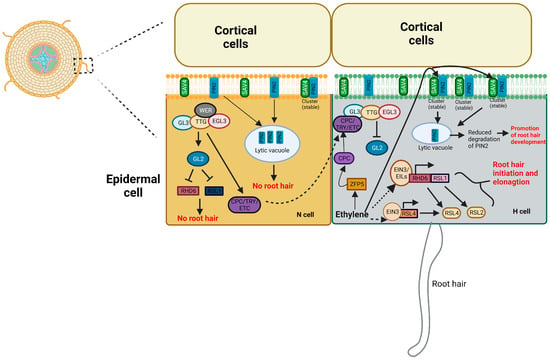
Figure 4.
Role of ethylene in root hair development: In N-type cells (nonhair) of the epidermis of the root, a transcription complex consisting of WEREWOLF (WER), GLABRA3 (GL3), ENHANCER OF GLABRA3 (EGL3), and TRANSPARENT TESTA GLABRA (TTG) known as WER-GL3/EGL3-TTG induces the expression of GLABRA2 (GL2) and CAPRICE (CPC). GL2 is responsible for determining the differentiation of N-type cells, and the hormone ethylene inhibits the transcription of GL2 in these cells. GL2, in turn, suppresses the expression of ROOT HAIR DEFECTIVE 6 (RHD6) and RHD6 LIKE1 (RSL1), which are positive regulators of root hair initiation and elongation. RHD6 and RSL1 promote the expression of RSL2 and RSL4, which further stimulate root hair elongation by upregulating genes responsible for elongation. CPC can move laterally from N-type cells to neighboring H-type (hair cell) cells of the root’s epidermis. CPC competes with WER for binding to the GL3/EGL3-TTG complex in H-type cells. Additionally, CPC, along with functionally redundant homologs TRIPTYCHON (TRY) and ENHANCER OF TRY AND CPCs (ETC1), inhibits the expression of GL2, which regulates the differentiation of H-type cells. ETH promotes the transcription of CPC by involving the transcription factor ZINC FINGER PROTEIN 5 (ZFP5). CPC, TRY, ETC1, TTG, GL3, and EGL3 collectively work to suppress the expression of GL2 in H-type cells. Furthermore, EIN3/EIL1 can form a complex with RHD6/RSL1, promoting root hair initiation. EIN3 also binds to the promoter region of RSL4, enhancing its transcription and promoting both root hair initiation and elongation. In addition, ethylene has a regulatory role in root hair development by affecting SAV4 (SHADE AVOIDANCE 4, regulator of auxin transport). SAV4 is responsible for managing the PIN2 (PIN-FORMED2) protein levels on the cell membrane, ensuring their stability and preventing degradation in the vacuole. Ethylene treatment boosts SAV4 levels, resulting in an increase in PIN2 proteins on the cell membrane. SAV4 interacts with PIN2 through PIN2HL (PIN2 hydrophilic region), potentially promoting PIN2 protein clustering and stability on the membrane. This process encourages the downward movement of auxin, ultimately leading to the growth of root hairs. Figure is an updated illustration from [81]. Dotted arrow—multistep pathway; solid arrow—promotion; flat-head arrow—inhibition.
4.4. Fruit Ripening
Ethylene plays a crucial role in the regulation of fruit ripening by orchestrating the expression of genes involved in various biological processes. These processes encompass pigment accumulation, respiration, ET production, texture change, and the overall enhancement of fruit quality traits [14]. Excess ET production is chiefly associated with transcriptional upregulation of ET biosynthesis genes (ACS and ACO). Until now, 14 ACS genes have been identified in the S. lycopersicum, among these, ACS2 and ACS4 showed important fruit-ripening functions [94]. Through genome-wide identification, six ACO genes have been identified in S. lycopersicum. Interestingly, during the pre-ripening stages, the expression levels of all ACO genes remain undetectable. However, ACO1 and ACO2 demonstrate high expression levels during the ripening process, while ACO4 exhibits a gradual and slight increase in expression. Upon ripening, the undetectable transcript level of ACO3, ACO5, and ACO6 suggests the least role in climacteric ET production [14]. During the climacteric stage, there is a well-known transition from system 1 to system 2, accompanied by a remarkable upregulation of ACS2, ACS4, ACO1, and ACO4. This upregulation leads to positive feedback regulation. In S. lycopersicum, the expressions of ACS1A, ACS3, ACS6, ACO1, and ACO4 are associated with system 1 of ethylene production, while the expressions of ACS2, ACS4, ACO1, and ACO4 are associated with system 2 [32,95].
Unlike climacteric fruits such as Solanum lycopersicum, non-climacteric fruits, such as Fragaria x ananassa (strawberry) do not rely on ET for the initiation and maintenance of the ripening process [96]. However, the measurement of ACC revealed that it is present in large quantities as strawberries progress towards the stages of fruit ripening. ACC accumulation in strawberry directly indicates higher expression of ACS genes. According to the expression analysis, the genes FaACS1 and FaACS26 exhibited ripening-specific expression in the receptacle tissues of strawberries. On the other hand, the genes FaACS17, FaACS21, FaACS19, and FaACS23 were considered to be achene-specific ripening-induced genes. Notably, certain ACS genes, including FaACS27 and FaACS29, displayed strong expression during the developmental stages of achenes, ranging from the green achene to the white achene stage. These findings suggest a strong association between ET and the onset and progression of fruit ripening [97].
Shan et al. [98] discovered the mechanism for maintaining a balance of MaNAC2, MaACS1, and MaACO1 levels in Musa acuminata. MaNAC1 and MaNAC2 suppress MaERF expression, which is recognized for its inhibitory effect on the genes involved in ET biosynthesis, namely MaACS1, and MaACO1. A RING E3 ligase MaXB3 interacts with MaNAC2, facilitating its ubiquitination-mediated degradation. This process inhibits the transcriptional repression mediated by MaNAC2. In addition, MaXB3 also targets proteasome degradation. Collectively, these findings unveil a regulatory cascade involving MaXB3, MaNACs, MaERF11, and MaACS1/MaACO1 that controls ET biosynthesis during ripening in Musa acuminata. MaNAC1 and MaNAC2 repress MaXB3, creating a feedback mechanism in the cascade. The AP2/ERF superfamily, a substantial class of transcription factors, plays a pivotal role in regulating fruit ripening. It targets genes associated with various processes, such as softening (POLYGALACTURONASE, PECTATE LYASE, EXPANSIN, XYLOGLUCAN ENDOTRANSGLUCOSYLASE/HYDROLASE), chlorophyll degradation (CHLOROPHYLL-B-REDUCTASE, NON-YELLOW COLORING, PHEOPHORBIDEα OXYGENASE, RED CHLOROPHYLL CATABOLITE REDUCTASE RCCR), anthocyanin accumulation (CHALCONE SYNTHASE, DIHYDROFLAVONOL-4-REDUCTASE), carotenoid accumulation (LYCOPENEβ-CYCLASE, PHYTOENE DESATURASE, β-CAROTENE HYDROXYLASE, ZEAXANTHIN EPOXIDASE, 9-CIS-EPOXYCAROTENOID DIOXYGENASE, CAROTENOID CLEAVAGE DIOXYGENASE), and flavor development (ALCOHOL DEHYDROGENASE, TERPENE SYNTHASE, ALCOHOL ACYLTRANSFERASE) [99]. A finding demonstrated that ET response factor ERF D7 triggers the activation of SlARF2A/B (auxin response factor) controlling fruit ripening in S. lycopersicum [100]. Positive feedback regulation is observed in PpIAA1 and PpERF4 in promoting fruit ripening in Prunus by enhancing the expression of ripening regulator genes. PpERF4 activates transcription of PpIAA1 and PpACO1 and PpIAA1 enhances transcription of PpACS1 [101]. A recent study using genetic analysis on S. lycopersicum highlighted that ET promoted auxin production in seeds for cell division and fruit growth. Conversely, during fruit ripening, system-2 ET regulates the process through a complex network, including SlEIN2, SlEILs, and crucial developmental transcription factors NOR (NON-RIPENING), RIN (RIPENING INHIBITOR), and FUL1 (FRUITFULL1). This underscores the significance of ET and its regulatory network in shaping S. lycopersicum fruit development and ripening [24]. A recent study recorded that the trihelix transcription factor SlGT31 enhances the transcription of ACS4 and ACO1 and acts as a positive modulator of fruit ripening [102]. Studies have shown that posttranslational modifications also play a role in fruit ripening. For instance, a recent report observed SPINDLY (O-glycosylation enzymes) collaborated with EIN2 to enhance the process of ET signaling, which in turn promoted the ripening of fruits in S. lycopersicum plants [103]. Histone modifiers, like histone deacetylases, are enlisted by AP2/ERFs to regulate fruit ripening. In the case of Malus, MdHDA19 was brought into the MdERF4–MdTPL4 complex and hindered fruit ripening by decreasing MdACSa expression [104]. In S. lycopersicum, the protein complex SlERF.F12–TPL2–HDA1/HDA3 suppressed the expression of ripening-related genes, including ACS2, ACS4 [105]. Figure 5 shows the summary of the fruit ripening process.
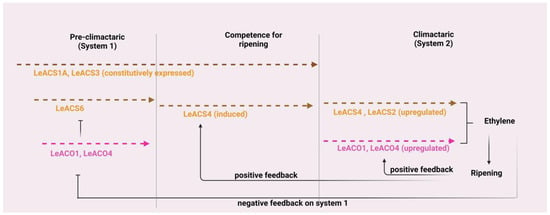
Figure 5.
Fruit ripening: In Solanum persicum (tomato) fruit, the pre-climacteric phase involves system 1 ethylene response, which is likely controlled by continuously active genes, LeACS1A and LeACS3, along with negatively regulated LeACS6 genes, which interact with existing Le ACO1 and LeACO4 mRNAs. As the fruit reaches competence, the expression of LeACS1A and LeACS6 is delayed, while LeACS4 expression is induced. Upon entering the climacteric stage, there is a transition to system 2, characterized by a significant increase in LeACS2, LeACS4, LeACO1, and LeACO4 mRNA levels due to positive feedback regulation. This surge in ethylene production leads to negative feedback on system 1, resulting in balanced ethylene production during the climacteric stage. The illustration is based on and modified from [32]. Dotted arrow—induction of specific gene and length of arrow defines the limit up to which gene expression occurs; solid arrow—promotion; flat-head arrow—inhibition.
4.5. Chloroplast Development
When seeds start to sprout in conditions lacking light, they activate a specific developmental program called skotomorphogenesis. In the case of plants like Arabidopsis, which undergoes hypogeal germination (germination below the soil surface), an enhancement in the length of the hypocotyl (the embryonic stem) occurs due to cell elongation. This elongation helps push the cotyledons against the mechanical pressure of the soil, allowing them to reach the light source. In the course of the process, proplastids, the precursors to all plastids, undergo both proliferation and differentiation. This transformation leads them to become etioplasts within the cotyledon cells, thus preparing plants for photosynthetic apparatus during the dark-to-light transition [106]. Etioplasts have a semi-crystalline membrane cluster called the prolamellar body (PLB). This structure contains essential components for photosynthesis, including prothylakoid membranes, protochlorophyllide, and protochlorophyllide oxidoreductase, a light-dependent enzyme that transforms protochlorophyllide into chlorophyllide [106]. In Arabidopsis, phytochromes (phyA and phyB) influence seedling photomorphogenesis. Etiolated seedlings produce phytochromes in their inactive Pr form in the cytoplasm. When exposed to light, Pr converts to the active Pfr form and moves to the nucleus [107]. This light-induced transformation triggers the photoactivated phytochromes’ degradation of certain bHLH transcription factors, known as PHYTOCHROME INTERACTING FACTORS (PIFs). PIF3 and its homologs PIF1, PIF4, and PIF5 play a crucial role in maintaining skotomorphogenesis and inhibiting light-induced chloroplast development [108].
Ethylene’s regulatory functions hinge on two transcription factors, EIN3 and EIL1. During seedling emergence, the photomorphogenic regulator CONSTITUTIVE PHOTOMORPHOGENIC1 (COP1) senses light fluctuations, while ET processes mechanical stress cues, collaboratively influencing EIN3 protein levels. In the nucleus transcription of PhANGs (PHOTOSYNTHESIS-ASSOCIATED NUCLEAR GENE) is suppressed by PIFs in coordination with other protein EIN3. COP1, an E3 ligase, targets EBF1/2 for degradation, stabilizes PIF/EIN3, and degrades photomorphogenesis stimulating transcription factors like HY5 (ELONGATED HYPOCOTYL5) [109,110,111]. The perception of light by photoreceptors initiates chloroplast biogenesis and the shift toward photoautotrophic growth. Light causes a transformation in the structure of phytochrome (phy), converting it from an inactive state to an active nuclear form. This active form then initiates the degradation of the PIFs (PHYTOCHROME INTERACTING FACTORS) [111]. Crucially, the photoreceptor phyB has directly influenced EIN3 in a light-dependent manner, promoting EIN3 degradation through enhanced interactions with EBF1/2 upon exposure to light [109]. When exposed to light, COP1 is prevented from entering the nucleus, and transcription factors like HY5 become more stable. Consequently, this leads to an upsurge in the expression of PhANGs [111]. EIN3/EILs inhibit protochlorophyllide (Pchlide) biosynthesis through PIF3 and EIN3/EILs directly trigger the expression of PROTOCHLOROPHYLLIDE OXIDOREDUCTASE A (PORA) and PORB in the cotyledon safeguarding against photooxidative damage during de-etiolation [10,112]. The EIN3-PIF3 module significantly suppresses LIGHT-HARVESTING COMPLEX (LHC) gene expression in darkness, preventing premature thylakoid membrane development. Overexpressing LHC genes in dark conditions leads to partially formed thylakoid membranes. This strict repression maintains the prolamellar body and safeguards against photooxidation upon exposure to light [113]. Thus, EIN3-PIF3 interdependence ensures proper etioplast-to-chloroplast development, enabling seedlings to adapt to changing environmental conditions during emergence. Thus, ethylene signaling oversees chloroplast development by preserving the correct levels of protochlorophyllide and PORA/PORB enzymes, thus preventing harmful photooxidative damage during de-etiolation. Figure 6 depicts the role of ethylene in chloroplast development.
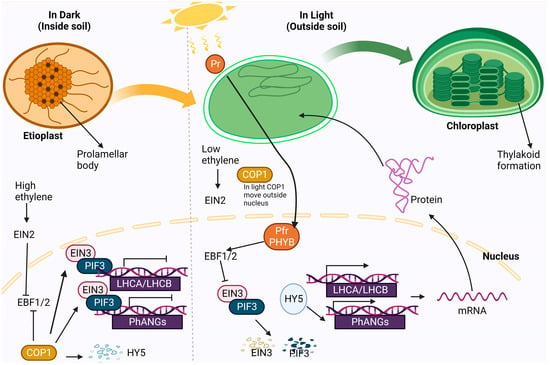
Figure 6.
Role of ethylene in chloroplast development: During the early growth of seedlings in the dark underground, mechanical pressure triggers the production of a plant hormone called ethylene. This leads to an increase in the concentration of a protein called EIN3 in the nucleus of plant cells. EIN3 (ETHYLENE-INSENSITIVE3) and another protein called PIF3 (PHYTOCHROME INTERACTING FACTORS), which is stabilized in the dark, directly interact and bind to the promoters of PhANGs (PHOTOSYNTHESIS-ASSOCIATED NUCLEAR GENE) and LHC (LIGHT-HARVESTING COMPLEX) genes. This interaction strongly suppresses the expression of these genes and helps maintain prolamellar bodies in etioplasts. In the absence of light, a key photoreceptor called PHYB Pr remains inactive, and a regulatory protein called COP1 (CONSTITUTIVE PHOTOMORPHOGENIC1) becomes active. COP1 controls the levels of various proteins involved in plant development, including EBF1/2 (F-box protein), and it stabilizes PIFs and EIN3 while degrading transcription factors that promote photomorphogenesis, such as HY5 (ELONGATED HYPOCOTYL5).
When the etiolated seedlings emerge into the light, removing mechanical pressure reduces ethylene production and EIN3 levels. At the same time, light activates PHYB Pfr, which prevents COP1 from entering the nucleus. This, in turn, leads to the rapid degradation of PIFs and EIN3, promoting the transition of prolamellar bodies to thylakoid membranes and stabilizing transcription factors like HY5. Consequently, there is an increase in the expression of LHC and PhANGs genes involved in chloroplast maturation, facilitating the development of chloroplasts for photosynthesis. The illustration is based on [111,113]. Solid arrow—promotion; flat-head arrow—inhibition
4.6. Photosynthesis
Photosynthesis is the foundational production mechanism for life to thrive on our planet. This mechanism not only boosts carbon acquisition but also leads to the enhancement of both crop yield and overall quality. The intricate regulation of this process involves a diverse range of signals, from environmental stressors to the influential role played by phytohormones. These phytohormones are pivotal in governing photosynthesis across biochemical and physiological contexts, operating through varied molecular mechanisms. As a result, photosynthesis remains a highly dynamic and finely tuned process, shaped by many factors. Many factors affect photosynthesis, like stomatal movement.
4.6.1. Stomatal Regulation
Stomata play a pivotal role in managing the exchange of gases between the leaf’s interior and the atmosphere by regulating turgor pressure in guard cells. These dynamic stomatal adjustments facilitate the precise assimilation of CO2 during photosynthesis, all while mitigating water loss through transpiration. ET’s regulation of stomatal responses is characterized by its tendency to manifest dual and sometimes conflicting roles [62]. Ethylene induces stomatal closure in wild Arabidopsis by triggering H2O2 synthesis in guard cells, this process relies on NADPH oxidase AtrbohF. ETR1 mediates ethylene and H2O2 signaling in guard cells via EIN2 and ARR2-dependent pathway(s), identifying AtrbohF as a key mediator of stomatal responses to ET [114]. Ethylene-insensitive mutants (etr1-1) of Arabidopsis showed a smaller number of stomata and reduced stomatal conductance indicating ET influences the stomatal development process and negatively affects stomatal conductance [67,115]. Also, another study showed that the application of exogenous ET enhances stomatal conductance, photosynthesis, and growth in Brassica juncea plants under optimal and deficient nitrogen fertilization [116]. A recent study showed the effect of ET on stomatal movement. Elevated CO2 levels were observed to induce higher ET production in Arabidopsis rosettes. ACC-synthase octuple mutant, resulting in reduced ethylene biosynthesis, experiences disrupted stomatal responses triggered by elevated CO2. Ethylene-insensitive gain-of-function receptor mutants (etr1-1 and etr2-1) and signaling mutants (ein2-5 and ein2-1) retain unaltered stomatal responses to CO2 shifts. Conversely, ET receptor loss-of-function mutants (etr2-3; ein4-4; ers2-3, etr1-6; etr2-3; etr1-6) demonstrate notably accelerated stomatal reactions to CO2 changes. Further examination shows compromised stomatal closure in the ACC-synthase octuple mutant and hastened stomatal responses in etr1-6, etr2-3, and etr1-6 mutants, but not in etr2-3, ein4-4, or ers2-3 mutants. These findings imply the crucial roles of ET production and signaling elements in fine tuning and expediting stomatal conductance responses to CO2 and ABA [117]. A study explored the role of abscisic acid and ET in high relative air humidity triggers stomatal opening in S. lycopersicum leaves. Elevated relative humidity led to a slight decrease in ABA levels during darkness but significantly increased ET evolution. Ethephon enhanced conductance and stomatal aperture under moderate relative humidity, while amino-ethoxyvinylglycine (AVG) and ET receptor inhibition blocked stomatal opening during a shift to high relative humidity. Ethylene-insensitive mutant responses were reduced in high relative humidity conditions. Exogenous ABA spray countered conductance increase upon transferring to high relative humidity. These findings show that ET production and sensitivity play a role in high relative humidity-triggered stomatal opening in S. lycopersicum leaves [118]. Chen et al. [62] suggested that the decreased responsiveness of older leaves to ABA and soil drying in terms of stomatal closure is probably attributed to the modified sensitivity of stomata to ET, as opposed to ET production.
4.6.2. Chlorophyll Content
According to Ceusters and Van de Poel [119], the impact of ET on photosynthesis is contingent upon the age of the leaves. ET directly regulates the photosynthetic process in younger, non-senescing leaves, while in mature leaves, its influence is more indirect, primarily driving leaf senescence. Studies on ethylene-insensitive mutants of Arabidopsis (etr1-1) and Nicotiana showed a decline in the overall photosynthetic capacity of young non-senescing leaves of the plant due to a reduction in the expression of crucial photosynthesis-associated genes like CAB (CHLOROPHYLL A/B-BINDING PROTEIN) and the small subunit of Rubisco [120,121,122]. Grbic and Bleecker [122] found that, in etr1-1 mutants, there was a delay in the initiation of leaf senescence, which correlated with a postponement in the activation of SAGs (SENESCENCE-ASSOCIATED GENES). Furthermore, they observed elevated expression levels in genes associated with photosynthesis. When non-senescing leaves were treated with external ET, it reduced the expression of CAB and a subsequent decrease in chlorophyll content. These findings suggest that ET negatively regulates photosynthesis in non-senescing leaves, implying that a basal level of ET production and perception is necessary for normal photosynthetic function. In contrast, elevated levels of ET inhibit photosynthesis. In mature leaves prone to senescence, ethylene-insensitive mutants (etr1-1 and ein2-1) and S. lycopersicum typically display increased chlorophyll content [122,123]. This implies that ET contributes to establishing appropriate chlorophyll levels in non-senescing leaves while promoting chlorosis by inducing chlorophyll degradation in mature leaves. These observations strongly indicate ET participation in governing photosynthesis, specifically in leaves that have not yet begun the senescence process. Also, when combining data from various surveys conducted on different species, it becomes evident that a species-specific regulation of photosynthesis is influenced by ET [119].
4.6.3. Light Reaction
The process of photosynthesis transforms light energy into chemical energy. For this, absorption of light energy by PSII and PSI is essential to facilitate electron transport and reduction in CO2 in the chloroplast. But sometimes, excessive absorption can lead to photochemical damage due to excessive ROS generation. Plants employ protective mechanisms like non-photochemical quenching (NPQ) to prevent over-reduction in photosystems. Chen and Gallie [124] demonstrated that ET controls energy-dependent non-photochemical quenching in Arabidopsis by inhibiting the xanthophyll cycle. In the above study, Arabidopsis eto1-1 mutants (ethylene overproducing) exhibited reduced capacity to convert violaxanthin to zeaxanthin due to impaired violaxanthin de-epoxidase activity. This leads to elevated reactive oxygen species production and increased photosensitivity in response to high light in these plants. Analyzing the intricacies of chlorophyll fluorescence through pulse-amplitude modulation fluorimetry, Kim et al. [125] have shed light on the impact of ET signaling mutations on photosystem II (PSII) activity in Arabidopsis. Specifically, their study uncovers that ET-insensitive mutants (etr1-1) exhibit diminished PSII activity in comparison to their wild-type counterparts. Notably, etr1-1 mutant lines, which are often used for ET-related investigations, can carry a consequential secondary mutation in ACCUMULATION AND REPLICATION3 (a second mutation in etr1-1 mutant of Arabidopsis responsible for producing premature stop codon in ARC3), prompting the need for supplementary corroborative lines of evidence, particularly in photosynthesis research. Ethylene signaling mutants derived from the arc3 secondary mutation (etr1-1sg) also demonstrate reduced maximum quantum efficiency, prolonged chlorophyll fluorescence lifetime of PSII, and decreased quantum yield of PSII. According to Kim et al. [125], these findings underline the necessity of regular ET sensitivity for optimal photochemical efficiency of PSII, irrespective of any modifications in chloroplast structure resulting from the secondary arc3 mutation. A study by Wullschleger et al. [126] showed that ET exposure reduces electron transport capacity in Glycine max by over 30% within 4 h, leading to reduced CO2 assimilation. This effect is not tied to Rubisco activity decrease. Measurements also showed lowered efficiency of excitation energy capture in PSII after ethylene exposure. This impact was more substantial under higher light levels, indicating possible photoinhibition’s role in ethylene-induced CO2 assimilation reduction. In addition, another study showed that overexpression of CitERF13 in Nicotiana leaves leads to a substantial decline in Fv/Fm, effective quantum efficiency of both PSI and PSII, apparent quantum yield, and maximum rate of photosynthetic transport; however, the change in NPQ was less pronounced [127].
4.6.4. Dark Reaction
The available literature show that ET also controls the dark reaction of photosynthesis. Tholen et al. [120] demonstrated that as vegetative Nicotiana plants were cultivated under varying atmospheric CO2 concentrations, an inverse relationship was observed between glucose concentration within leaves and the expression of the Rubisco gene. This repression of gene expression was distinctly amplified by heightened glucose levels in plants insensitive to ethylene. The insensitivity to ET led to equivalent nitrogen allocations in light harvesting while experiencing diminished levels in electron transport and Rubisco. This, in turn, resulted in a noticeably diminished photosynthetic capacity in ethylene-insensitive transgenic Nicotiana plants compared to the wild type. These findings imply that the lack of ET perception enhanced the plants’ vulnerability to glucose, potentially due to escalated ABA concentrations. Ultimately, this increased susceptibility to endogenous glucose detrimentally affected Rubisco content and these plants’ carboxylation process and overall photosynthetic capacity. A similar decrease in the photosynthesis of Arabidopsis etr1 mutant was observed due to a decline in the content of Rubisco protein and expression level. Another study observed that overexpression of CitERF13 in tobacco leaves decreases the maximum rate of Rubisco carboxylase activity [127]. Nevertheless, ethephon treatment applied to ET-sensitive Brassica juncea plants increases Rubisco activity, stomatal conductance, and net photosynthesis compared to ET-insensitive plants. These results showed that ET sensitivity influences the photosynthesis of B. juncea plants [128]. Also, on evaluation of the influence of ethephon treatment on the photosynthesis of two B. juncea cultivars which have a contrast in their photosynthetic capacity, it became evident that alterations in net photosynthesis (Pn) in both cultivars resulted from effects on stomata and mesophyll. Ethephon-induced Pn was linked to ET emission. The high-capacity cultivar Varuna displayed a weaker ethephon response than the low-capacity RH30. RH30′s low Pn was attributed to minimal ET. Enhancing RH30′s ethylene levels could boost its lower photosynthetic capacity and increase Pn [129]. Ethylene affects Rubisco activases (RCAs), making carboxylation regulation more complex. RCAs are Rubisco chaperones. Recorded observations indicate that ethephon treatment increases carbonic anhydrase (CA) activity in B. juncea plants, helping maintain chloroplast pH and reducing photorespiration [66]. Nevertheless, reports observed that ET, at specific concentrations, increases photosynthesis and its related attributes, and beyond that concentration, it shows an inhibitory effect. Thus, it can be inferred that the effect of ET is concentration and sensitivity of species under study dependent [41,129]. In a study, Never-ripe (Nr) S. lycopersicum, which carries a loss-of-function mutation in the ET receptor SLETR3, exhibited a higher rate of carbon assimilation relative to their wild-type counterparts. Notably, there were no significant differences in stomatal conductance or chlorophyll parameters between Nr and wild-type plants. The heightened photosynthetic rates in Nr plants resulted in increased levels of glucose, fructose, idose, mannose, and myo-inositol at midday, while ribose levels decreased in Nr plants. These findings suggest that the enhanced carbon assimilation, elevated sugar levels, and overall growth improvement in Nr plants are most likely due to increased enzymatic activity in the Calvin cycle [130]. In summary, it can be deduced that maintaining basal levels of ET synthesis and signaling is essential for the normal regulation of Rubisco levels. However, elevated ethylene levels inhibit carbon fixation in most plant species.
4.7. Senescence
Leaf senescence is a highly programmed, regulated, and degenerative process. It is characterized by chlorophyll breakdown and degradation of macromolecules [131]. Studies reported increased ET production in senescent leaves with higher transcription rates of ET biosynthesis genes ACS and ACO [132]. On the other hand, the Octuple mutant of ACS genes showed a delayed senescence response [133]. Nevertheless, the recorded literature also reveal that its impact on growth and senescence processes varies depending on factors such as its concentration, timing of application, and the specific plant species involved [7,41].
A study revealed that EIN2 plays a role in regulating leaf senescence partially via microRNA164 (miR164) and ORESARA1 (ORE1, also named ANAC092 or NAC2) [134]. A recent study showed that EIN3 and ORE1 can directly regulate the expression of CHLOROPHYLL CATABOLIC GENES (CCGs), NONYELLOWING1 (NYE1, also known as STAY-GREEN1, SGR1), NONYELLOW COLORING1 (NYC1), and PHEOPHORBIDE A OXYGENASE (PAO), thereby initiating chlorophyll breakdown during leaf senescence [135]. An ETHYLENE-INSENSITIVE3-LIKE1 (EIL1) gene, GhLYI (LINT YIELD INCREASING), encodes a truncated protein that regulates senescence by directly targeting SENESCENCE-ASSOCIATED GENE 20 (SAG20) in Gossypium hirsutum [136]. Yu et al. [137] demonstrated that WRKY71 can directly enhance the expression of ET signaling pathway genes EIN2 and ORE1. Additionally, it promotes ET synthesis by directly stimulating ACS2, thereby accelerating leaf senescence in Arabidopsis. However, a recent study found that transcription factor SbWRKY50 in Sorghum bicolor hinders chlorophyll degradation by binding to chlorophyll catabolic gene promoters, notably repressing SbNYC1 (NON-YELLOW COLORING 1), thus suppressing leaf senescence [138]. Petal senescence is another phase in the flower developmental continuum accompanied by tissue differentiation, petal maturation, and finally, the growth and development of seeds coordinated by plant hormones [22]. Reports conducted so far indicate a close association of ET production with senescence in the Dianthus flower. In the Dianthus petal senescence induced by ET, the transcription factors DcHB30 and DcWRKY75 function antagonistically. They competitively modulate the expression of common target genes, specifically DcACS1, DcACO1, DcSAG12, and DcSAG29, resulting in a regulatory interplay that governs the process of petal senescence [139]. Another report observed that DcWRKY33 promotes petal senescence by activating genes involved in the biosynthesis of ET and ABA and the accumulation of ROS in Dianthus [140]. Ethylene-induced petal senescence in Dianthus is orchestrated by the transcription factor DcEIL3-1. Two genes, DcEBF1 and DcEBF2, regulate the process by influencing downstream target genes of DcEIL3-1. DcEBF1 and DcEBF2 also interact with DcEIL3-1 to promote its degradation through ubiquitination. This study uncovers the interplay between these factors, expanding our understanding of ET’s role in Dianthus petal aging [141]. RNA-sequencing analysis showed that the protein kinase RhCIPK6 played an important role in the petal senescence of the Rosa flower [142]. During natural senescence, the early expression of ACS and ACO in gynoecium than in petals indicates that ET is first produced in gynoecium followed by petals [143]. Moreover, removing gynoecium prevents increased production of petal ET and significantly prolongs the flower’s life [144,145]. Expression analyses of ACS and ACO in the floral organs of Dianthus caryophyllus, Petunia, Solanum lycopersicum, and Rosa hybrida have been performed [146,147,148,149]. Differential expression regulation has been observed for three cloned ACS genes, namely DcACS1, DcACS2, and DcACS3, in Dianthus flowers [150]. Okamoto et al. [151] revealed the differential expression of ACS and ACO genes in Delphinium grandiflorum. In this study, ET generation in the receptacle showed a significant increase during natural senescence, while the gynoecium exhibited only a slight enhancement in ET production. Ethylene application at low concentrations could trigger the expression of DcACS and DcACO in the short-lived varieties but not in the long-lived varieties of Dianthus flowers [152]. A report in Rosa hybrida claimed that RhACO1 was significantly correlated with vase life and ET sensitivity among 33 cultivars [153]. Although several transcription factors have been found to have favorable effects on ET biosynthesis, the regulation of ACS and ACO genes remains unclear. A study reported that upregulation of PLMYB308 (MYB transcription factor gene) is associated with ET application in herbaceous ponies. The gene silencing approach of PLMYB308 revealed delayed flower senescence and a dramatic increase in GA with reduced ET and ABA levels in petals [154]. Figure 7 summarizes the senescence process.
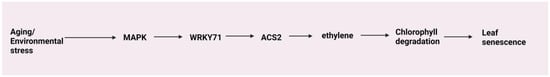
Figure 7.
Senescence process: WRKY71 is a senescence-associated gene induced by ethylene. WRKY71 can activate ethylene synthesis by directly upregulating ACS2; thus, WRKY71 and ACS2 form a positive feedback loop and promote senescence by activating downstream genes.
4.8. Abscission
The shedding of plant organs such as seedpods, leaves, floral organs, and fruits by detaching them at the abscission zone is called abscission [155]. Developmental and environmental changes can trigger abscission in plants and are mainly subjected to the crosstalk between two plant hormones, ET and auxin [156]. Ethylene regulates the gene expression pattern of enzymes involved in cell separation, like cellulases and pectinases [157]. Before abscission occurs, auxin is transported to the abscission zone to inhibit ET sensitivity in the cells. When abscission occurs, the organs undergoing abscission release ET, which is then followed by the detachment of leaves [23]. Ethylene present in the abscission zone initiates a signal transduction pathway that activates transcription factors and genes responsive to ET [158] which in turn, regulate abscission. In addition, the induction of ethylene production by methyl jasmonate was identified as the cause of fruit abscission in ‘Hamlin’ and ‘Valencia’ orange varieties [159]. For uniform ripening, external ET application promotes abscission in fruit crops. On the other hand, 2-aminoethoxyvinyl glycine (AVG), ET biosynthesis inhibitors are used to reduce abscission before harvest. Vesicle transport pathways genes in ethylene-induced AZ-C (calyx abscission zone) cells and adjacent FR (fruit rind) cells are responsible for citrus fruit abscission [160]. Hence, it appears that coordination between hydrolytic enzymes and ET production leads to plant organ abscission. The key ET biosynthetic enzymes ACS and ACO are found to be highly expressed during organ abscission, which facilitates ET production followed by activation of genes encoding cell-wall-remodeling enzymes [161,162]. Pollination upregulated the SlACS2 gene in S. lycopersicum [148]. In Petunia, pollination leads to a 20-fold increase in ET production autocatalytically from the stigmas contributing to wilting and eventually abscission [163]. Few studies have analyzed ACS and ACO expression in flowers displaying petal abscission. The expression of ACS and ACO genes in Pelargonium flowers has been partially assessed using RNA gel blot analysis. It has been observed that in ET-sensitive flowers, the application of exogenous ET and pollination can expedite sepal abscission in D. grandiflorum. cKNAT1, a KNOTTED-LIKE FROM ARABIDOPSIS THALIANA1 (KNAT1)-like protein from Litchi chinensis, controlling abscission by influencing production of ethylene. In vitro and in vivo assays showed that LcKNAT1 hinders the expression of LcACS/ACO genes by directly binding to their promoters and delaying abscission in Litchi chinensis [164]. The transcription factor LcERF10 acts as a positive regulator in Litchi fruitlet abscission [165]. Nonetheless, clarifying the interaction between ET and other hormones like auxin and gibberellins is essential for obtaining a better understanding of abscission.
5. Conclusions
In summary, ET is a remarkable and versatile plant hormone, wielding significant influence across a spectrum of plant developmental and physiological processes. It plays a pivotal role in a plant’s life cycle, including cell division, elongation, leaf growth, senescence, abscission, flower and fruit development, chloroplast maturation, and photosynthesis regulation. As our comprehension of ET involvement in plant development deepens, the potential for leveraging its properties to enhance crop performance and stress resilience becomes increasingly evident.
Ethylene’s capacity to finely regulate growth and development, modulate responses to environmental challenges, and control the maturation of fruits and flowers holds substantial promise for agriculture and food security, particularly in the face of a changing global climate. Nevertheless, it is worth noting that the specific mechanisms underlying ET regulation of photosynthesis, a fundamental process, remain largely unexplored. Investigating the downstream ET-responsive transcription factors governing photosynthesis-related gene expression is essential. Additionally, understanding the intricate interplay between ET and other plant hormones in orchestrating growth and development is crucial. Deciphering the cross-talk between ET and other phytohormones and their synergistic or antagonistic effects could offer valuable insights and allow precise manipulation of these hormone profiles through molecular techniques to elicit desired plant responses.
In the years to come, ongoing research into ET’s multifaceted role is likely to unveil novel avenues for steering plant growth and development, ultimately contributing to the advancement of sustainable agriculture and addressing the challenges posed by a rapidly changing climate. The captivating narrative of ethylene in the realm of plant biology still needs to be completed, offering a wealth of prospects for scientific discovery and practical application.
Author Contributions
Conceptualization, S.K. and N.A.K.; validation, N.A.K.; writing—original draft preparation, S.K. and A.F.A.; writing—review and editing, N.A.K.; visualization, S.K.; supervision, N.A.K. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Chang, C.; Wang, B.; Shi, L.; Li, Y.; Duo, L.; Zhang, W. Alleviation of salinity stress-induced inhibition of seed germination in cucumber (Cucumis sativus L.) by ethylene and glutamate. J. Plant Physiol. 2010, 167, 1152–1156. [Google Scholar] [CrossRef]
- Khan, M.I.R.; Iqbal, N.; Masood, A.; Per, T.S.; Khan, N.A. Salicylic acid alleviates adverse effects of heat stress on photosynthesis through changes in proline production and ethylene formation. Plant Signal. Behav. 2013, 8, e26374. [Google Scholar] [CrossRef]
- Riyazuddin, R.; Verma, R.; Singh, K.; Nisha, N.; Keisham, M.; Bhati, K.K.; Kim, S.T.; Gupta, R. Ethylene: A master regulator of salinity stress tolerance in plants. Biomolecules 2020, 10, 959. [Google Scholar] [CrossRef]
- Pandey, B.K.; Huang, G.; Bhosale, R.; Hartman, S.; Sturrock, C.J.; Jose, L.; Martin, O.C.; Karady, M.; Voesenek, L.A.C.J.; Ljung, K.; et al. Plant roots sense soil compaction through restricted ethylene diffusion. Science 2021, 371, 276–280. [Google Scholar] [CrossRef]
- Li, H.; Wang, L.; Liu, M.; Dong, Z.; Li, Q.; Fei, S.; Xiang, H.; Liu, B.; Jin, W. Maize plant architecture is regulated by the ethylene biosynthetic gene ZmACS7. Plant Physiol. 2020, 183, 1184–1199. [Google Scholar] [CrossRef] [PubMed]
- Ahammed, G.J.; Gantait, S.; Mitra, M.; Yang, Y.; Li, X. Role of ethylene crosstalk in seed germination and early seedling development: A review. Plant Physiol. Biochem. 2020, 151, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, N.; Khan, N.A.; Ferrante, A.; Trivellini, A.; Francini, A.; Khan, M.I.R. Ethylene role in plant growth, development and senescence. Interaction with other phytohormones. Front. Plant Sci. 2017, 8, 475. [Google Scholar] [CrossRef]
- Subbiah, V.; Reddy, K.J. Interactions between ethylene, abscisic acid and cytokinin during germination and seedling establishment in Arabidopsis. J. Biosci. 2010, 35, 451–458. [Google Scholar] [CrossRef]
- Li, H.; Johnson, P.; Stepanova, A.; Alonso, J.M.; Ecker, J.R. Convergence of signaling pathways in the control of differential cell growth in Arabidopsis. Dev. Cell 2004, 7, 193–204. [Google Scholar] [CrossRef]
- Zhong, S.; Zhao, M.; Shi, T.; Shi, H.; An, F.; Zhao, Q.; Guo, H. EIN3/EIL1 cooperate with PIF1 to prevent photo-oxidation and to promote greening of Arabidopsis seedlings. Proc. Natl. Acad. Sci. USA 2009, 106, 21431–21436. [Google Scholar] [CrossRef]
- Dolgikh, V.A.; Pukhovaya, E.M.; Zemlyanskaya, E.V. Shaping ethylene response: The role of EIN3/EIL1 transcription factors. Front. Plant Sci. 2019, 10, 1030. [Google Scholar] [CrossRef]
- Ceng-Hong, H.U.; Shi-Dong, Y.U.A.N.; Cui-Ling, T.O.N.G.; Zhang, D.J.; Huang, R.H. Ethylene modulates root growth and mineral nutrients levels in trifoliate orange through the auxin-signaling pathway. Not. Bot. Horti Agrobot. Cluj-Napoca 2023, 51, 13269. [Google Scholar]
- Wang, Z.; Yadav, V.; Yan, X.; Cheng, D.; Wei, C.; Zhang, X. Systematic genome-wide analysis of the ethylene-responsive ACS gene family: Contributions to sex form differentiation and development in melon and watermelon. Gene 2021, 805, 145910. [Google Scholar] [CrossRef]
- Liu, Y.; Tang, M.; Liu, M.; Su, D.; Chen, J.; Gao, Y.; Li, Z. The molecular regulation of ethylene in fruit ripening. Small Methods 2020, 4, 1900485. [Google Scholar] [CrossRef]
- Wang, J.; Huang, R. Modulation of ethylene and ascorbic acid on reactive oxygen species scavenging in plant salt response. Front. Plant Sci. 2019, 10, 319. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Bullock Jr, D.A.; Alonso, J.M.; Stepanova, A.N. To fight or to grow: The balancing role of ethylene in plant abiotic stress responses. Plants 2021, 11, 33. [Google Scholar] [CrossRef] [PubMed]
- Jasrotia, S.; Jastoria, R. Role of ethylene in combating biotic stress. In Ethylene in Plant Biology, 1st ed.; Singh, S., Husain, T., Singh, V.P., Tripathi, D.K., Prasad, S.M., Dubey, N.K., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2022; Volume 1, pp. 388–397. [Google Scholar]
- Cocetta, G.; Natalini, A. Ethylene: Management and breeding for postharvest quality in vegetable crops. A review. Front. Plant Sci. 2022, 13, 968315. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Seidi, F.; Zhang, T.; Jin, Y.; Xiao, H. Ethylene scavengers for the preservation of fruits and vegetables: A review. Food Chem. 2021, 337, 127750. [Google Scholar] [CrossRef]
- Hu, B.; Sun, D.W.; Pu, H.; Wei, Q. Recent advances in detecting and regulating ethylene concentrations for shelf-life extension and maturity control of fruit: A review. Trends Food Sci. Technol. 2019, 91, 66–82. [Google Scholar] [CrossRef]
- Yu, D.; Li, X.; Li, Y.; Ali, F.; Li, F.; Wang, Z. Dynamic roles and intricate mechanisms of ethylene in epidermal hair development in Arabidopsis and cotton. New Phytol. 2022, 234, 375–391. [Google Scholar] [CrossRef]
- Dar, R.A.; Nisar, S.; Tahir, I. Ethylene: A key player in ethylene sensitive flower senescence: A review. Sci. Hortic. 2021, 290, 110491. [Google Scholar] [CrossRef]
- Botton, A.; Ruperti, B. The yes and no of the ethylene involvement in abscission. Plants 2019, 8, 187. [Google Scholar] [CrossRef]
- Huang, W.; Hu, N.; Xiao, Z.; Qiu, Y.; Yang, Y.; Yang, J.; Guo, H. A molecular framework of ethylene-mediated fruit growth and ripening processes in tomato. Plant Cell 2022, 34, 3280–3300. [Google Scholar] [CrossRef]
- Adams, D.O.; Yang, S. Ethylene biosynthesis: Identification of 1-aminocyclopropane-1-carboxylic acid as an intermediate in the conversion of methionine to ethylene. Proc. Natl. Acad. Sci. USA 1979, 76, 170–174. [Google Scholar] [CrossRef] [PubMed]
- Kende, H. Ethylene biosynthesis. Annu. Rev. Plant Biol. 1993, 44, 283–307. [Google Scholar] [CrossRef]
- Houben, M.; Van de Poel, B. 1-Aminocyclopropane-1-carboxylic acid oxidase (ACO): The enzyme that makes the plant hormone ethylene. Front. Plant Sci. 2019, 10, 695. [Google Scholar] [CrossRef] [PubMed]
- Qiao, H.; Chang, K.N.; Yazaki, J.; Ecker, J.R. Interplay between ethylene, ETP1/ETP2 F-box proteins, and degradation of EIN2 triggers ethylene responses in Arabidopsis. Genes Dev. 2009, 23, 512–521. [Google Scholar] [CrossRef]
- Wen, X.; Zhang, C.; Ji, Y.; Zhao, Q.; He, W.; An, F.; Guo, H. Activation of ethylene signaling is mediated by nuclear translocation of the cleaved EIN2 carboxyl terminus. Cell Res. 2012, 22, 1613–1616. [Google Scholar] [CrossRef]
- Zhang, F.; Wang, L.; Qi, B.; Zhao, B.; Ko, E.E.; Riggan, N.D.; Qiao, H. EIN2 mediates direct regulation of histone acetylation in the ethylene response. Proc. Natl. Acad. Sci. USA 2017, 114, 10274–10279. [Google Scholar] [CrossRef]
- Binder, B.M. Ethylene signaling in plants. J. Biol. Chem. 2020, 295, 7710–7725. [Google Scholar] [CrossRef]
- Barry, C.S.; Llop-Tous, M.I.; Grierson, D. The regulation of 1-aminocyclopropane-1-carboxylic acid synthase gene expression during the transition from system-1 to system-2 ethylene synthesis in tomato. Plant Physiol. 2000, 123, 979–986. [Google Scholar] [CrossRef] [PubMed]
- Ito, Y.; Kitagawa, M.; Ihashi, N.; Yabe, K.; Kimbara, J.; Yasuda, J.; Ito, H.; Inakuma, T.; Hiroi, S.; Kasumi, T. DNA-binding specificity, transcriptional activation potential, and the rin mutation effect for the tomato fruit-ripening regulator RIN. Plant J. 2008, 55, 212–223. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhang, H.; Quan, R.; Wang, X.C.; Huang, R. Transcriptional regulation of the ethylene response factor LeERF2 in the expression of ethylene biosynthesis genes controls ethylene production in tomato and tobacco. Plant Physiol. 2009, 150, 365–377. [Google Scholar] [CrossRef] [PubMed]
- Harkey, A.F.; Yoon, G.M.; Seo, D.H.; DeLong, A.; Muday, G.K. Light modulates ethylene synthesis, signaling, and downstream transcriptional networks to control plant development. Front. Plant Sci. 2019, 10, 1094. [Google Scholar] [CrossRef] [PubMed]
- Pattyn, J.; Vaughan-Hirsch, J.; Van de Poel, B. The regulation of ethylene biosynthesis: A complex multilevel control circuitry. New Phytol. 2021, 229, 770–782. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.T.; Xue, H.W. Casein kinase 1 regulates ethylene synthesis by phosphorylating and promoting the turnover of ACS5. Cell Rep. 2014, 9, 1692–1702. [Google Scholar] [CrossRef] [PubMed]
- Seo, D.H.; Yoon, G.M. Light-induced stabilization of ACS contributes to hypocotyl elongation during the dark-to-light transition in Arabidopsis seedlings. Plant J. 2019, 98, 898–911. [Google Scholar] [CrossRef] [PubMed]
- Skottke, K.R.; Yoon, G.M.; Kieber, J.J.; DeLong, A. Protein phosphatase 2A controls ethylene biosynthesis by differentially regulating the turnover of ACC synthase isoforms. PLoS Genet. 2011, 7, e1001370. [Google Scholar] [CrossRef]
- Gautam, H.; Sehar, Z.; Khan, N.A. Ethylene: A gaseous signaling molecule with diverse roles. In The Plant Hormone Ethylene; Khan, N.A., Ferrante, A., Munné-Bosch, S., Eds.; Academic Press: Cambridge, MA, USA, 2023; pp. 1–13. [Google Scholar]
- Khan, N.A.; Khan, M.I.R.; Ferrante, A.; Poor, P. Ethylene: A key regulatory molecule in plants. Front. Plant Sci. 2017, 8, 1782. [Google Scholar] [CrossRef]
- Raz, V.; Koornneef, M. Cell division activity during apical hook development. Plant Physiol. 2001, 125, 219–226. [Google Scholar] [CrossRef]
- Etchells, J.P.; Provost, C.M.; Turner, S.R. Plant vascular cell division is maintained by an interaction between PXY and ethylene signalling. PLoS Genet. 2012, 8, e1002997. [Google Scholar] [CrossRef] [PubMed]
- Ruzicka, K.; Ljung, K.; Vanneste, S.; Podhorská, R.; Beeckman, T.; Friml, J.; Benková, E. Ethylene regulates root growth through effects on auxin biosynthesis and transport-dependent auxin distribution. Plant Cell 2007, 19, 2197–2212. [Google Scholar] [CrossRef] [PubMed]
- Ortega-Martínez, O.; Pernas, M.; Carol, R.J.; Dolan, L. Ethylene modulates stem cell division in the Arabidopsis thaliana root. Science 2007, 317, 507–510. [Google Scholar] [CrossRef] [PubMed]
- Thomann, A.; Lechner, E.; Hansen, M.; Dumbliauskas, E.; Parmentier, Y.; Kieber, J.; Scheres, B.; Genschik, P. Arabidopsis CULLIN3 genes regulate primary root growth and patterning by ethylene-dependent and -independent mechanisms. PLoS Genet. 2009, 5, e1000328. [Google Scholar] [CrossRef] [PubMed]
- Zluhan-Martínez, E.; López-Ruíz, B.A.; García-Gómez, M.L.; García-Ponce, B.; de la Paz Sánchez, M.; Álvarez-Buylla, E.R.; Garay-Arroyo, A. Integrative roles of phytohormones on cell proliferation, elongation and differentiation in the Arabidopsis thaliana primary root. Front. Plant Sci. 2021, 12, 659155. [Google Scholar] [CrossRef] [PubMed]
- Street, I.H.; Aman, S.; Zubo, Y.; Ramzan, A.; Wang, X.; Shakeel, S.N.; Kieber, J.J.; Schaller, G.E. Ethylene inhibits cell proliferation of the Arabidopsis root meristem. Plant Physiol. 2015, 169, 338–350. [Google Scholar] [CrossRef]
- Skirycz, A.; Claeys, H.; De Bodt, S.; Oikawa, A.; Shinoda, S.; Andriankaja, M.; Maleux, K.; Eloy, N.B.; Coppens, F.; Yoo, S.D.; et al. Pause-and-stop: The effects of osmotic stress on cell proliferation during early leaf development in Arabidopsis and a role for ethylene signaling in cell cycle arrest. Plant Cell 2011, 23, 1876–1888. [Google Scholar] [CrossRef]
- Marsch-Martinez, N.; Greco, R.; Becker, J.D.; Dixit, S.; Bergervoet, J.H.; Karaba, A.; de Folter, S.; Pereira, A. BOLITA, an Arabidopsis AP2/ERF-like transcription factor that affects cell expansion and proliferation/differentiation pathways. Plant Mol. Biol. 2006, 62, 825–843. [Google Scholar] [CrossRef]
- Dubois, M.; Van den Broeck, L.; Inzé, D. The pivotal role of ethylene in plant growth. Trends Plant Sci. 2018, 23, 311–323. [Google Scholar] [CrossRef]
- Claeys, H.; De Bodt, S.; Inzé, D. Gibberellins and DELLAs: Central nodes in growth regulatory networks. Trends Plant Sci. 2014, 19, 231–239. [Google Scholar] [CrossRef]
- Polko, J.K.; van Rooij, J.A.; Vanneste, S.; Pierik, R.; Ammerlaan, A.M.; Vergeer-van Eijk, M.H.; McLoughlin, F.; Gühl, K.; Van Isterdael, G.; Voesenek, L.A.; et al. Ethylene-mediated regulation of A2-type CYCLINs modulates hyponastic growth in Arabidopsis. Plant Physiol. 2015, 169, 194–208. [Google Scholar] [CrossRef] [PubMed]
- Van de Poel, B.; Smet, D.; Van Der Straeten, D. Ethylene and hormonal cross talk in vegetative growth and development. Plant Physiol. 2015, 169, 61–72. [Google Scholar] [CrossRef]
- Polko, J.K.; van Zanten, M.; van Rooij, J.A.; Marée, A.F.; Voesenek, L.A.; Peeters, A.J.; Pierik, R. Ethylene-induced differential petiole growth in Arabidopsis thaliana involves local microtubule reorientation and cell expansion. New Phytol. 2012, 193, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Feng, G.; Liu, G.; Xiao, J. The Arabidopsis EIN2 restricts organ growth by retarding cell expansion. Plant Signal. Behav. 2015, 10, e1017169. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Shani, E.; Weinstain, R.; Zhang, Y.; Castillejo, C.; Kaiserli, E.; Chory, J.; Estelle, M. Gibberellins accumulate in the elongating endodermal cells of Arabidopsis root. Proc. Natl. Acad. Sci. USA 2013, 110, 4834–4839. [Google Scholar] [CrossRef]
- Vogel, J.P.; Woeste, K.E.; Theologis, A.; Kieber, J.J. Recessive and dominant mutations in the ethylene biosynthetic gene ACS 5 of Arabidopsis confer cytokinin insensitivity and ethylene overproduction, respectively. Proc. Natl. Acad. Sci. USA 1998, 95, 4766–4771. [Google Scholar] [CrossRef] [PubMed]
- Qu, X.; Hall, B.P.; Gao, Z.; Schaller, G.E. A strong constitutive ethylene-response phenotype conferred on Arabidopsis plants containing null mutations in the ethylene receptors ETR1 and ERS1. BMC Plant Biol. 2007, 7, 3. [Google Scholar] [CrossRef]
- Rai, M.I.; Wang, X.; Thibault, D.M.; Kim, H.J.; Bombyk, M.M.; Binder, B.M.; Shakeel, S.N.; Schaller, G.E. The ARGOS gene family functions in a negative feedback loop to desensitize plants to ethylene. BMC Plant Biol. 2015, 15, 157. [Google Scholar] [CrossRef]
- Shi, J.; Habben, J.E.; Archibald, R.L.; Drummond, B.J.; Chamberlin, M.A.; Williams, R.W.; Lafitte, H.R.; Weers, B.P. Overexpression of ARGOS genes modifies plant sensitivity to ethylene, leading to improved drought tolerance in both Arabidopsis and maize. Plant Physiol. 2015, 169, 266–282. [Google Scholar] [CrossRef]
- Chen, L.; Dodd, I.C.; Davies, W.J.; Wilkinson, S. Ethylene limits abscisic acid- or soil drying-induced stomatal closure in aged wheat leaves. Plant Cell Environ. 2013, 36, 1850–1859. [Google Scholar] [CrossRef]
- Fiorani, F.; Bogemann, G.M.; Visser, E.J.; Lambers, H.; Voesenek, L.A. Ethylene emission and responsiveness to applied ethylene vary among Poa species that inherently differ in leaf elongation rates. Plant Physiol. 2002, 129, 1382–1390. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Reid, D.M. The role of endogenous ethylene in the expansion of Helianthus annuus leaves. Can. J. Bot. 1997, 75, 501–508. [Google Scholar] [CrossRef]
- Khan, N.A.; Mir, M.R.; Nazar, R.; Singh, S. The application of ethephon (an ethylene releaser) increases growth, photosynthesis and nitrogen accumulation in mustard (Brassica juncea L.) under high nitrogen levels. Plant Biol. 2008, 10, 534–538. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.A. The influence of exogenous ethylene on growth and photosynthesis of mustard (Brassica juncea) following defoliation. Sci. Hortic. 2005, 105, 499–505. [Google Scholar] [CrossRef]
- Tholen, D.; Voesenek, L.A.C.J.; Poorter, H. Ethylene insensitivity does not increase leaf area or relative growth rate in Arabidopsis, Nicotiana tabacum, and Petunia x hybrida. Plant Physiol. 2004, 134, 1803–1812. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.J.; Pannell, J.R. Sex determination: Separate sexes are a double turnoff in melons. Curr. Biol. 2016, 26, 171–174. [Google Scholar] [CrossRef] [PubMed]
- Yin, T.; Quinn, J.A. A Mechanistic Model of a Single Hormone Regulating Both Sexes in Flowering Plants. Bull. Torrey Bot. Club 1992, 119, 431–441. [Google Scholar] [CrossRef]
- Zhang, H.; Li, S.; Yang, L.; Cai, G.; Chen, H.; Gao, D.; Lin, T.; Cui, Q.; Wang, D.; Li, Z.; et al. Gain-of-function of the 1-aminocyclopropane-1-carboxylate synthase gene ACS1G induces female flower development in cucumber gynoecy. Plant Cell 2021, 33, 306–321. [Google Scholar] [CrossRef]
- Chen, H.; Sun, J.; Li, S.; Cui, Q.; Zhang, H.; Xin, F.; Wang, H.; Lin, T.; Gao, D.; Wang, S.; et al. An ACC oxidase gene essential for cucumber carpel development. Mol. Plant 2016, 9, 1315–1327. [Google Scholar] [CrossRef]
- Trebitsh, T.; Staub, J.E.; O’Neill, S.D. Identification of a 1-aminocyclopropane-1-carboxylic acid synthase gene linked to the female (F) locus that enhances female sex expression in cucumber. Plant Physiol. 1997, 113, 987–995. [Google Scholar] [CrossRef]
- Zhang, S.; Tan, F.Q.; Chung, C.H.; Slavkovic, F.; Devani, R.S.; Troadec, C.; Bendahmane, A. The control of carpel determinacy pathway leads to sex determination in cucurbits. Science 2022, 378, 543–549. [Google Scholar] [CrossRef] [PubMed]
- Ning, Q.; Jian, Y.; Du, Y.; Li, Y.; Shen, X.; Jia, H.; Zhang, Z. An ethylene biosynthesis enzyme controls quantitative variation in maize ear length and kernel yield. Nat. Commun. 2021, 12, 5832. [Google Scholar] [CrossRef] [PubMed]
- García, A.; Aguado, E.; Garrido, D.; Martínez, C.; Jamilena, M. Two androecious mutations reveal the crucial role of ethylene receptors in the initiation of female flower development in Cucurbita pepo. Plant J. 2020, 103, 1548–1560. [Google Scholar] [CrossRef]
- Cebrián, G.; Iglesias-Moya, J.; Romero, J.; Martínez, C.; Garrido, D.; Jamilena, M. The ethylene biosynthesis gene CpACO1A: A new player in the regulation of sex determination and female flower development in Cucurbita pepo. Front. Plant Sci. 2022, 12, 817922. [Google Scholar] [CrossRef] [PubMed]
- Rashid, D.; Devani, R.S.; Rodriguez-Granados, N.Y.; Abou-Choucha, F.; Troadec, C.; Morin, H.; Bendahmane, A. Ethylene produced in carpel primordia controls CmHB40 expression to inhibit stamen development. Nat. Plants 2023, 9, 1675–1687. [Google Scholar] [CrossRef] [PubMed]
- Cebrián, G.; Segura, M.; Martínez, J.; Iglesias-Moya, J.; Martínez, C.; Garrido, D.; Jamilena, M. Jasmonate-deficient mutant lox3a reveals crosstalk between jasmonate and ethylene in the differential regulation of male and female flower opening and early fruit development in Cucurbita pepo. J. Exp. Bot. 2023, 74, 1258–1274. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhang, S.; Yang, Y.; Li, Z.; Li, H.; Yu, R.; Wei, C. Novel bisexual flower control gene regulates sex differentiation in melon (Cucumis melo L.). J. Agric. Food Chem. 2022, 70, 15401–15414. [Google Scholar] [CrossRef] [PubMed]
- Dolan, L. The role of ethylene in root hair growth in Arabidopsis. J. Plant. Nutr. Soil Sci. 2001, 164, 141–145. [Google Scholar] [CrossRef]
- Li, M.; Zhu, Y.; Li, S.; Zhang, W.; Yin, C.; Lin, Y. Regulation of phytohormones on the growth and development of plant root hair. Front. Plant Sci. 2022, 13, 865302. [Google Scholar] [CrossRef]
- Feng, Y.; Xu, P.; Li, B.; Li, P.; Wen, X.; An, F.; Gong, Y.; Xin, Y.; Zhu, Z.; Wang, Y.; et al. Ethylene promotes root hair growth through coordinated EIN3/EIL1 and RHD6/RSL1 activity in Arabidopsis. Proc. Natl. Acad. Sci. USA 2017, 114, 13834–13839. [Google Scholar] [CrossRef]
- Qiu, Y.; Tao, R.; Feng, Y.; Xiao, Z.; Zhang, D.; Peng, Y.; Wen, X.; Wang, Y.; Guo, H. EIN3 and RSL4 interfere with an MYB–bHLH–WD40 complex to mediate ethylene-induced ectopic root hair formation in Arabidopsis. Proc. Natl. Acad. Sci. USA 2021, 118, e2110004118. [Google Scholar] [CrossRef]
- Xiao, F.; Gong, Q.; Zhao, S.; Lin, H.; Zhou, H. MYB30 and ETHYLENE INSENSITIVE3 antagonistically modulate root hair growth in Arabidopsis. Plant J. 2021, 106, 480–492. [Google Scholar] [CrossRef] [PubMed]
- Yan, A.; Wu, M.; Zhao, Y.; Zhang, A.; Liu, B.; Schiefelbein, J.; Gan, Y. Involvement of C2H2 zinc finger proteins in the regulation of epidermal cell fate determination in Arabidopsis. J. Integr. Plant Biol. 2014, 56, 1112–1117. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Huang, L.; Yan, A.; Liu, Y.; Liu, B.; Yu, C.; Zhang, A.; Schiefelbein, J.; Gan, Y. Multiple phytohormones promote root hair elongation by regulating a similar set of genes in the root epidermis in Arabidopsis. J. Exp. Bot. 2016, 67, 6363–6372. [Google Scholar] [CrossRef] [PubMed]
- Mangano, S.; Denita-Juarez, S.P.; Choi, H.S.; Marzol, E.; Hwang, Y.; Ranocha, P.; Velasquez, S.M.; Borassi, C.; Barberini, M.L.; Aptekmann, A.A.; et al. Molecular link between auxin and ROS-mediated polar growth. Proc. Natl. Acad. Sci. USA 2017, 114, 5289–5294. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, H.; Kawahara, A.; Inoue, Y. Ethylene promotes the induction by auxin of the cortical microtubule randomization required for low-pH-induced root hair initiation in lettuce (Lactuca sativa L.) seedlings. Plant Cell Physiol. 2003, 44, 932–940. [Google Scholar] [CrossRef][Green Version]
- Bruex, A.; Kainkaryam, R.M.; Wieckowski, Y.; Kang, Y.H.; Bernhardt, C.; Xia, Y.; Zheng, X.; Wang, J.Y.; Lee, M.M.; Benfey, P.; et al. A gene regulatory network for root epidermis cell differentiation in Arabidopsis. PLoS Genet. 2012, 8, 1002446. [Google Scholar] [CrossRef]
- Pitts, R.J.; Cernac, A.; Estelle, M. Auxin and ethylene promote root hair elongation in Arabidopsis. Plant J. 1998, 16, 553–560. [Google Scholar] [CrossRef]
- Strader, L.C.; Chen, G.L.; Bartel, B. Ethylene directs auxin to control root cell expansion. Plant J. 2010, 64, 874–884. [Google Scholar] [CrossRef]
- Muday, G.K.; Rahman, A.; Binder, B.M. Auxin and ethylene: Collaborators or competitors? Trends Plant Sci. 2012, 17, 181–195. [Google Scholar] [CrossRef]
- Chen, P.; Ge, Y.; Chen, L.; Yan, F.; Cai, L.; Zhao, H.; Lei, D.; Jiang, J.; Wang, M.; Tao, Y. SAV4 is required for ethylene-induced root hair growth through stabilizing PIN2 auxin transporter in Arabidopsis. New Phytol. 2022, 234, 1735–1752. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Pirrello, J.; Chervin, C.; Roustan, J.P.; Bouzayen, M. Ethylene control of fruit ripening: Revisiting the complex network of transcriptional regulation. Plant Physiol. 2015, 169, 2380–2390. [Google Scholar] [CrossRef] [PubMed]
- Nakatsuka, A.; Murachi, S.; Okunishi, H.; Shiomi, S.; Nakano, R.; Kubo, Y.; Inaba, A. Differential expression and internal feedback regulation of 1-aminocyclopropane-1-carboxylate synthase, 1-aminocyclopropane-1-carboxylate oxidase, and ethylene receptor genes in tomato fruit during development and ripening. Plant Physiol. 1998, 118, 1295–1305. [Google Scholar] [CrossRef] [PubMed]
- Symons, G.M.; Chua, Y.J.; Ross, J.J.; Quittenden, L.J.; Davies, N.W.; Reid, J.B. Hormonal changes during non-climacteric ripening in strawberry. J. Exp. Bot. 2012, 63, 4741–4750. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, R.K.; Motyka, V.; Pokorna, E.; Dobrev, P.I.; Lacek, J.; Shao, J.; Lewers, K.S.; Mattoo, A.K. Comprehensive profiling of endogenous phytohormones and expression analysis of 1-aminocyclopropane-1-carboxylic acid synthase gene family during fruit development and ripening in octoploid strawberry (Fragaria × ananassa). Plant Physiol. Biochem. 2003, 196, 186–196. [Google Scholar] [CrossRef] [PubMed]
- Shan, W.; Kuang, J.F.; Wei, W.; Fan, Z.Q.; Deng, W.; Li, Z.G.; Bouzayen, M.; Pirrello, J.; Lu, W.J.; Chen, J.Y. MaXB3 modulates MaNAC2, MaACS1, and MaACO1 stability to repress ethylene biosynthesis during banana fruit ripening. Plant Physiol. 2020, 184, 1153–1171. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Y.; Fan, Z.; Cui, Y.; Gu, X.; Chen, S.; Ma, H. APETALA2/ethylene responsive factor in fruit ripening: Roles, interactions and expression regulation. Front. Plant Sci. 2022, 13, 979348. [Google Scholar] [CrossRef]
- Gambhir, P.; Singh, V.; Parida, A.; Raghuvanshi, U.; Kumar, R.; Sharma, A.K. Ethylene response factor ERF. D7 activates auxin response factor 2 paralogs to regulate tomato fruit ripening. Plant Physiol. 2022, 190, 2775–2796. [Google Scholar] [CrossRef]
- Wang, X.; Pan, L.; Wang, Y.; Meng, J.; Deng, L.; Niu, L.; Zeng, W. PpIAA1 and PpERF4 form a positive feedback loop to regulate peach fruit ripening by integrating auxin and ethylene signals. Plant Sci. 2021, 313, 111084. [Google Scholar] [CrossRef]
- Fu, M.; Li, F.; Zhou, S.; Guo, P.; Chen, Y.; Xie, Q.; Hu, Z. Trihelix transcription factor SlGT31 regulates fruit ripening mediated by ethylene in tomato. J. Exp. Bot. 2023, 74, 5709–5721. [Google Scholar] [CrossRef]
- Xu, J.; Liu, S.; Cai, L.; Wang, L.; Dong, Y.; Qi, Z.; Zhou, Y. SPINDLY interacts with EIN2 to facilitate ethylene signalling-mediated fruit ripening in tomato. Plant Biotechnol. J. 2023, 21, 219–231. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Han, Z.; Wang, T.; Li, H.; Li, Q.; Wang, S.; Wu, T. Ethylene response factor MdERF4 and histone deacetylase MdHDA19 suppress apple fruit ripening through histone deacetylation of ripening-related genes. Plant Physiol. 2022, 188, 2166–2181. [Google Scholar] [CrossRef] [PubMed]
- Deng, H.; Chen, Y.; Liu, Z.; Liu, Z.; Shu, P.; Wang, R.; Liu, M. SlERF. F12 modulates the transition to ripening in tomato fruit by recruiting the co-repressor TOPLESS and histone deacetylases to repress key ripening genes. Plant Cell. 2022, 34, 1250–1272. [Google Scholar] [CrossRef] [PubMed]
- Cackett, L.; Luginbuehl, L.H.; Schreier, T.B.; Lopez-Juez, E.; Hibberd, J.M. Chloroplast development in green plant tissues: The interplay between light, hormone, and transcriptional regulation. New Phytol. 2022, 233, 2000–2016. [Google Scholar] [CrossRef]
- Rockwell, N.C.; Su, Y.S.; Lagarias, J.C. Phytochrome structure and signaling mechanisms. Annu. Rev. Plant Biol. 2006, 57, 837–858. [Google Scholar] [CrossRef]
- Leivar, P.; Monte, E. PIFs: Systems integrators in plant development. Plant Cell 2014, 26, 56–78. [Google Scholar] [CrossRef]
- Shi, H.; Liu, R.; Xue, C.; Shen, X.; Wei, N.; Deng, X.W.; Zhong, S. Seedlings transduce the depth and mechanical pressure of covering soil using COP1 and ethylene to regulate EBF1/EBF2 for soil emergence. Curr. Biol. 2016, 26, 139–149. [Google Scholar] [CrossRef]
- An, F.; Zhao, Q.; Ji, Y.; Li, W.; Jiang, Z.; Yu, X.; Zhang, C.; Han, Y.; He, W.; Liu, Y.; et al. Ethylene-induced stabilization of ETHYLENE INSENSITIVE3 and EIN3-LIKE1 is mediated by proteasomal degradation of EIN3 binding F-box 1 and 2 that requires EIN2 in Arabidopsis. Plant Cell 2010, 22, 2384–2401. [Google Scholar] [CrossRef]
- Hernández-Verdeja, T.; Vuorijoki, L.; Strand, A. Emerging from the darkness: Interplay between light and plastid signaling during chloroplast biogenesis. Physiol. Plant. 2020, 169, 397–406. [Google Scholar] [CrossRef]
- Zhong, S.; Shi, H.; Xue, C.; Wei, N.; Guo, H.; Deng, X.W. Ethylene-orchestrated circuitry coordinates a seedling’s response to soil cover and etiolated growth. Proc. Natl. Acad. Sci. USA 2014, 111, 3913–3920. [Google Scholar] [CrossRef]
- Liu, X.; Liu, R.; Li, Y.; Shen, X.; Zhong, S.; Shi, H. EIN3 and PIF3 form an interdependent module that represses chloroplast development in buried seedlings. Plant Cell 2017, 29, 3051–3067. [Google Scholar] [CrossRef]
- Desikan, R.; Last, K.; Harrett-Williams, R.; Tagliavia, C.; Harter, K.; Hooley, R.; Hancock, J.T.; Neill, S.J. Ethylene-induced stomatal closure in Arabidopsis occurs via AtrbohF-mediated hydrogen peroxide synthesis. Plant J. 2006, 47, 907–916. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, Y.; Sano, T.; Tamaoki, M.; Nakajima, N.; Kondo, N.; Hasezawa, S. Ethylene inhibits abscisic acid-induced stomatal closure in Arabidopsis. Plant Physiol. 2005, 138, 2337–2343. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, N.; Nazar, R.; Syeed, S.; Masood, A.; Khan, N.A. Exogenously-sourced ethylene increases stomatal conductance, photosynthesis, and growth under optimal and deficient nitrogen fertilization in mustard. J. Exp. Bot. 2011, 62, 4955–4963. [Google Scholar] [CrossRef]
- Azoulay-Shemer, T.; Schulze, S.; Nissan-Roda, D.; Bosmans, K.; Shapira, O.; Weckwerth, P.; Zamora, O.; Yarmolinsky, D.; Trainin, T.; Kollist, H.; et al. A role for ethylene signaling and biosynthesis in regulating and accelerating CO2-and abscisic acid-mediated stomatal movements in Arabidopsis. New Phytol. 2023, 238, 2460–2475. [Google Scholar] [CrossRef] [PubMed]
- Arve, L.E.; Torre, S. Ethylene is involved in high air humidity promoted stomatal opening of tomato (Lycopersicon esculentum) leaves. Funct. Plant Biol. 2015, 42, 376–386. [Google Scholar] [CrossRef] [PubMed]
- Ceusters, J.; Van de Poel, B. Ethylene exerts species-specific and age-dependent control of photosynthesis. Plant Physiol. 2018, 176, 2601–2612. [Google Scholar] [CrossRef]
- Tholen, D.; Pons, T.L.; Voesenek, L.A.C.J.; Poorter, H. Ethylene insensitivity results in down-regulation of Rubisco expression and photosynthetic capacity in tobacco. Plant Physiol. 2007, 144, 1305–1315. [Google Scholar] [CrossRef]
- Tholen, D.; Pons, T.L.; Voesenek, L.A.C.J.; Poorter, H. The role of ethylene perception in the control of photosynthesis. Plant Signal. Behav. 2008, 3, 108–109. [Google Scholar] [CrossRef]
- Grbic, V.; Bleecker, A.B. Ethylene regulates the timing of leaf senescence in Arabidopsis. Plant J. 1995, 8, 595–602. [Google Scholar] [CrossRef]
- Monteiro, C.C.; Carvalho, R.F.; Gratão, P.L.; Carvalho, G.; Tezotto, T.; Medici, L.O.; Peres, L.E.P.; Azevedo, R.A. Biochemical responses of the ethylene insensitive Never ripe tomato mutant subjected to cadmium and sodium stresses. Environ. Exp. Bot. 2011, 71, 306–320. [Google Scholar] [CrossRef]
- Chen, Z.; Gallie, D.R. Ethylene regulates energy-dependent non-photochemical quenching in Arabidopsis through repression of the xanthophyll cycle. PLoS ONE 2015, 10, e0144209. [Google Scholar] [CrossRef]
- Kim, G.D.; Cho, Y.H.; Yoo, S.D. Phytohormone ethylene-responsive Arabidopsis organ growth under light is in the fine regulation of Photosystem II deficiency-inducible AKIN10 expression. Sci. Rep. 2017, 7, 2767. [Google Scholar] [CrossRef] [PubMed]
- Wullschleger, S.D.; Hanson, P.J.; Gunderson, C.A. Assessing the influence of exogenous ethylene on electron transport and fluorescence quenching in leaves of Glycine max. Environ. Exp. Bot. 1992, 32, 449–455. [Google Scholar] [CrossRef]
- Xie, X.L.; Xia, X.J.; Kuang, S.; Zhang, X.L.; Yin, X.R.; Yu, J.Q.; Chen, K.S. A novel ethylene responsive factor CitERF13 plays a role in photosynthesis regulation. Plant Sci. 2017, 256, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, N.; Nazar, R.; Khan, M.I.R.; Khan, N.A. Variation in photosynthesis and growth of mustard cultivars: Role of ethylene sensitivity. Sci. Hortic. 2012, 135, 1–6. [Google Scholar] [CrossRef]
- Khan, N.A. An evaluation of the effects of exogenous ethephon, an ethylene releasing compound, on photosynthesis of mustard (Brassica juncea) cultivars that differ in photosynthetic capacity. BMC Plant Biol. 2004, 4, 21. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, V.L.; Pereira, A.M.; Pereira, A.S.; Silva, V.F.; Costa, L.C.; Bastos, C.E.; Ribeiro, D.M.; Caldana, C.; Sulpice, R.; Nunes-Nesi, A.; et al. Physiological and metabolic bases of increased growth in the tomato ethylene-insensitive mutant Never ripe: Extending ethylene signaling functions. Plant Cell Rep. 2021, 40, 1377–1393. [Google Scholar] [CrossRef]
- Woo, H.R.; Kim, H.J.; Lim, P.O.; Nam, H.G. Leaf senescence: Systems and dynamics aspects. Annu. Rev. Plant Biol. 2019, 70, 347–376. [Google Scholar] [CrossRef]
- van der Graaff, E.; Schwacke, R.; Schneider, A.; Desimone, M.; Flugge, U.I.; Kunze, R. Transcription analysis of Arabidopsis membrane transporters and hormone pathways during developmental and induced leaf senescence. Plant Physiol. 2006, 141, 776–792. [Google Scholar] [CrossRef]
- Tsuchisaka, A.; Yu, G.; Jin, H.; Alonso, J.M.; Ecker, J.R.; Zhang, X.; Gao, S.; Theologis, A. A combinatorial interplay among the 1-aminocyclopropane-1-carboxylate isoforms regulates ethylene biosynthesis in Arabidopsis thaliana. Genetics 2009, 183, 979–1003. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Woo, H.R.; Kim, J.; Lim, P.O.; Lee, I.C.; Choi, S.H.; Hwang, D.; Nam, H.G. Trifurcate feed-forward regulation of age-dependent cell death involving miR164 in Arabidopsis. Science 2009, 323, 1053–1057. [Google Scholar] [CrossRef] [PubMed]
- Qiu, K.; Li, Z.; Yang, Z.; Chen, J.; Wu, S.; Zhu, X.; Gao, S.; Gao, J.; Ren, G.; Kuai, B.; et al. EIN3 and ORE1 accelerate degreening during ethylene-mediated leaf senescence by directly activating chlorophyll catabolic genes in Arabidopsis. PLoS Genet. 2015, 11, e1005399. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zang, Y.; Chen, J.; Feng, S.; Zhang, Z.; Hu, Y.; Zhang, T. A truncated ETHYLENE INSENSITIVE3-like protein, GhLYI, regulates senescence in cotton. Plant Physiol. 2023, 193, 1177–1196. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Qi, Y.; Xu, J.; Dai, X.; Chen, J.; Dong, C.H.; Xiang, F. Arabidopsis WRKY71 regulates ethylene-mediated leaf senescence by directly activating EIN2, ORE1 and ACS2 genes. Plant J. 2021, 107, 1819–1836. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Zheng, Y.; Wang, J.; Wang, Z.; Yang, Z.; Chi, X.; Sun, B. Ethylene-responsive SbWRKY50 suppresses leaf senescence by inhibition of chlorophyll degradation in sorghum. New Phytol. 2023, 238, 1129–1145. [Google Scholar] [CrossRef]
- Xu, H.; Wang, S.; Larkin, R.M.; Zhang, F. The transcription factors DcHB30 and DcWRKY75 antagonistically regulate ethylene-induced petal senescence in carnation (Dianthus caryophyllus). J. Exp. Bot. 2022, 73, 7326–7343. [Google Scholar] [CrossRef]
- Wang, T.; Sun, Z.; Wang, S.; Feng, S.; Wang, R.; Zhu, C.; Zhang, F. DcWRKY33 promotes petal senescence in carnation (Dianthus caryophyllus L.) by activating genes involved in the biosynthesis of ethylene and abscisic acid and accumulation of reactive oxygen species. Plant J. 2023, 113, 698–715. [Google Scholar] [CrossRef]
- Zhu, C.; Huang, Z.; Sun, Z.; Feng, S.; Wang, S.; Wang, T.; Zhang, F. The mutual regulation between DcEBF1/2 and DcEIL3-1 is involved in ethylene induced petal senescence in carnation (Dianthus caryophyllus L.). Plant J. 2023, 114, 636–650. [Google Scholar] [CrossRef]
- Wu, Y.; Zuo, L.; Ma, Y.; Jiang, Y.; Gao, J.; Tao, J.; Chen, C. Protein Kinase RhCIPK6 Promotes Petal Senescence in Response to Ethylene in Rose (Rosa Hybrida). Genes 2022, 13, 1989. [Google Scholar] [CrossRef]
- Ten Have, A.; Woltering, E.J. Ethylene biosynthetic genes are differentially expressed during carnation (Dianthus caryophyllus L.) flower senescence. Plant Mol. Biol. 1997, 34, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Satoh, S. Ethylene Production and Petal Wilting during Senescence of Cut Carnation (Dianthus caryophyllus) Flowers and Prolonging Their Vase Life by Genetic Transformation. J. Jpn. Soc. Hortic. Sci. 2011, 80, 127–135. [Google Scholar] [CrossRef]
- Shibuya, K.; Yoshioka, T.; Hashiba, T.; Satoh, S. Role of the gynoecium in natural senescence of carnation (Dianthus caryophyllus L.) flowers. J. Exp. Bot. 2000, 51, 2067–2073. [Google Scholar] [CrossRef] [PubMed]
- Norikoshi, R.; Niki, T.; Ichimura, K. Differential regulation of two 1-aminocyclopropane-1-carboxylate oxidase (ACO) genes, including the additionally cloned DcACO2, during senescence in carnation flowers. Postharvest Biol. Technol. 2022, 183, 111752. [Google Scholar] [CrossRef]
- Tang, X.; Gomes, A.M.T.R.; Bhatia, A.; Woodson, W.R. Pistil specifc and ethylene-regulated expression of 1-aminocyclopropene-1-carboxylate oxidase genes on petunia flower. Plant Cell 1994, 6, 1227–1239. [Google Scholar] [CrossRef] [PubMed]
- Llop-Tous, I.; Barry, C.S.; Grierson, D. Regulation of ethylene biosynthesis in response to pollination in tomato flowers. Plant Physiol. 2000, 123, 971–978. [Google Scholar] [CrossRef] [PubMed]
- Xue, J.; Li, Y.; Tan, H.; Yang, F.; Ma, N.; Gao, J. Expression of ethylene biosynthetic and receptor genes in rose floral tissues during ethylene-enhanced flower opening. J. Exp. Bot. 2008, 59, 2161–2169. [Google Scholar] [CrossRef]
- Jones, M.L.; Woodson, W.R. Differential expression of three members of the 1-aminocyclopropane-1-carboxylate synthase gene family in carnation. Plant Physiol. 1999, 119, 755–764. [Google Scholar] [CrossRef]
- Okamoto, M.; Niki, T.; Azuma, M.; Shibuya, K.; Ichimura, K. Expression of ethylene biosynthesis genes in the gynoecium and receptacle associated with sepal abscission during senescence in Delphinium grandiflorum. Plant Growth Regul. 2022, 97, 593–609. [Google Scholar] [CrossRef]
- Tanase, K.; Onozaki, T. Regulation of ethylene-and senescence-related genes in pot carnation flowers during flower senescence. J. Hortic. 2016, 85, 254–263. [Google Scholar] [CrossRef]
- In, B.C.; Ha, S.T.; Lee, Y.S.; Lim, J.H. Relationships between the longevity, water relations, ethylene sensitivity, and gene expression of cut roses. Postharvest Biol. Technol. 2017, 131, 74–83. [Google Scholar] [CrossRef]
- Ji, X.; Wang, M.; Xu, Z.; Wang, K.; Sun, D.; Niu, L. PlMYB308 regulates flower senescence by modulating ethylene biosynthesis in Herbaceous Peony. Front. Plant Sci. 2022, 13, 872442. [Google Scholar] [CrossRef] [PubMed]
- Patharkar, O.R.; Walker, J.C. Advances in abscission signaling. J. Exp. Bot. 2018, 69, 733–740. [Google Scholar] [CrossRef] [PubMed]
- Meir, S.; Philosoph-Hadas, S.; Riov, J.; Tucker, M.L.; Patterson, S.E.; Roberts, J.A. Re-evaluation of the ethylene-dependent and-independent pathways in the regulation of floral and organ abscission. J. Exp. Bot. 2019, 70, 1461–1467. [Google Scholar] [CrossRef]
- Brown, K.M. Ethylene and abscission. Physiol. Plant. 1997, 100, 567–576. [Google Scholar] [CrossRef]
- Cheng, C.; Zhang, L.; Yang, X.; Zhong, G. Profiling gene expression in citrus fruit calyx abscission zone (AZ-C) treated with ethylene. Mol. Genet. Genom. 2015, 290, 1991–2006. [Google Scholar] [CrossRef]
- Hartmond, U.; Yuan, R.; Burns, J.K.; Grant, A.; Kender, W.J. Citrus fruit abscission induced by methyl-jasmonate. J. Am. Soc. Hortic. Sci. 2000, 125, 547–552. [Google Scholar] [CrossRef]
- Merelo, P.; Agustí, J.; Ventimilla, D.; Talón, M.; Tadeo, F.R. Vesicular trafficking in abscission zone cells during ethylene-promoted fruit abscission in citrus. Acta Hortic. 2019, 1230, 41–48. [Google Scholar] [CrossRef]
- Dal Cin, V.; Barbaro, E.; Danesin, M.; Murayama, H.; Velasco, R.; Ramina, A. Fruitlet abscission: A cDNA-AFLP approach to study genes differentially expressed during shedding of immature fruits reveals the involvement of a putative auxin hydrogen symporter in apple (Malus domestica L. Borkh). Gene 2009, 442, 26–36. [Google Scholar] [CrossRef]
- Li, J.; Zhu, H.; Yuan, R. Profiling the expression of genes related to ethylene biosynthesis, ethylene perception, and cell wall degradation during fruit abscission and fruit ripening in apple. J. Am. Soc. Hortic. Sci. 2010, 135, 391–401. [Google Scholar] [CrossRef]
- Woltering, E.J.; van Doorn, W.C. Role of ethylene in senescence of petals: Morphological and taxonomical relationships. J. Exp. Bot. 1998, 208, 1605–1616. [Google Scholar] [CrossRef]
- Zhao, M.; Li, C.; Ma, X.; Xia, R.; Chen, J.; Liu, X.; Ying, P.; Peng, M.; Wang, J.; Shi, C.L.; et al. KNOX protein KNAT1 regulates fruitlet abscission in litchi by repressing ethylene biosynthetic genes. J. Exp. Bot. 2020, 71, 4069–4082. [Google Scholar] [CrossRef]
- He, Z.; Ma, X.; Wang, F.; Li, J.; Zhao, M. LcERF10 functions as a positive regulator of litchi fruitlet abscission. Int. J. Biol. Macromol. 2023, 250, 126264. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).