Thermodynamic Properties as a Function of Temperature of AlMoNbV, NbTaTiV, NbTaTiZr, AlNbTaTiV, HfNbTaTiZr, and MoNbTaVW Refractory High-Entropy Alloys from First-Principles Calculations
Abstract
1. Introduction
Literature Review
2. Materials and Methods
2.1. Equation of State: Energy vs. Volume and Debye Model
2.2. Thermodynamic Properties
2.3. Computational Details
3. Results and Discussion
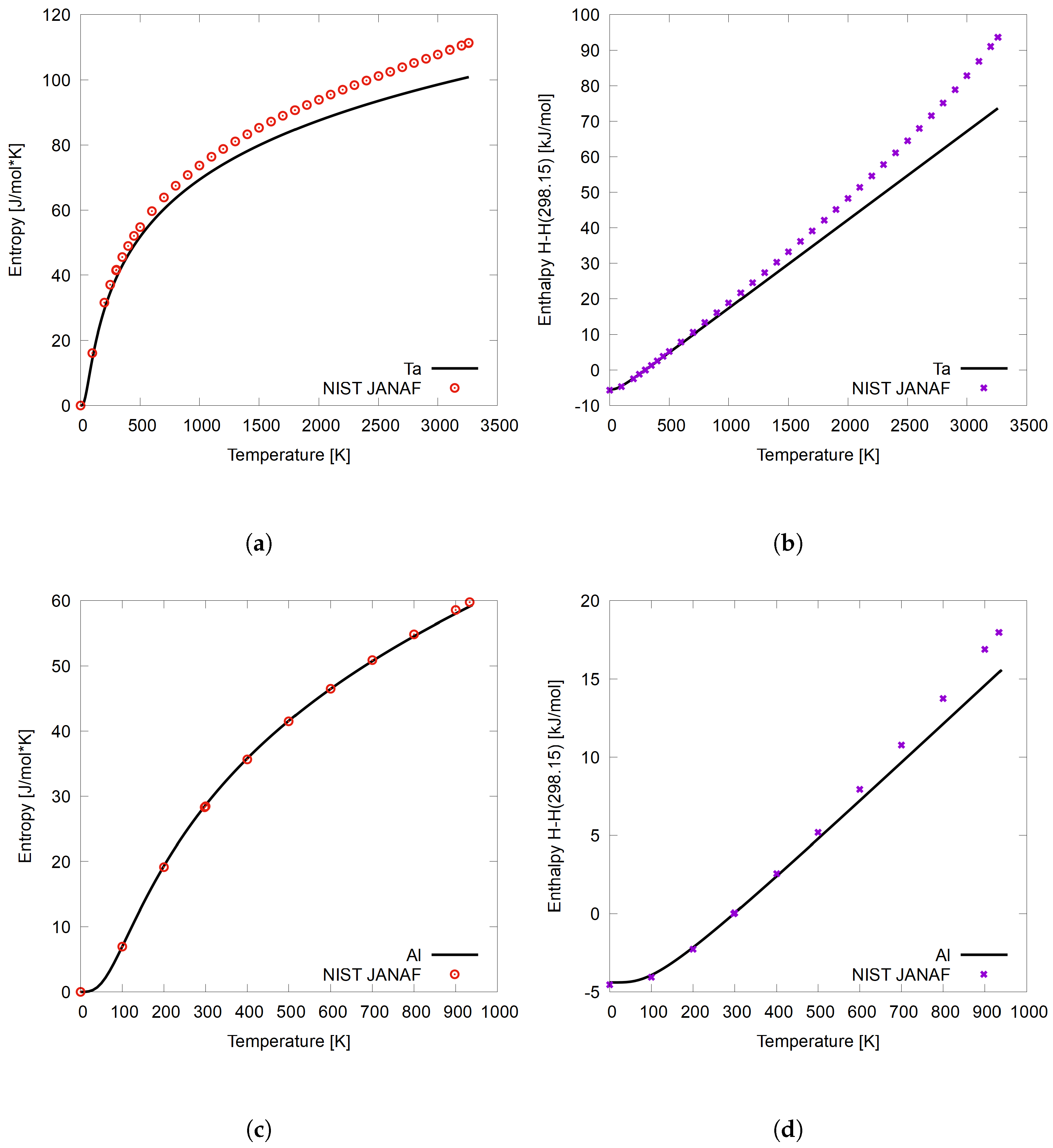
3.1. Methodology Validation on BCC Ta and FCC Al
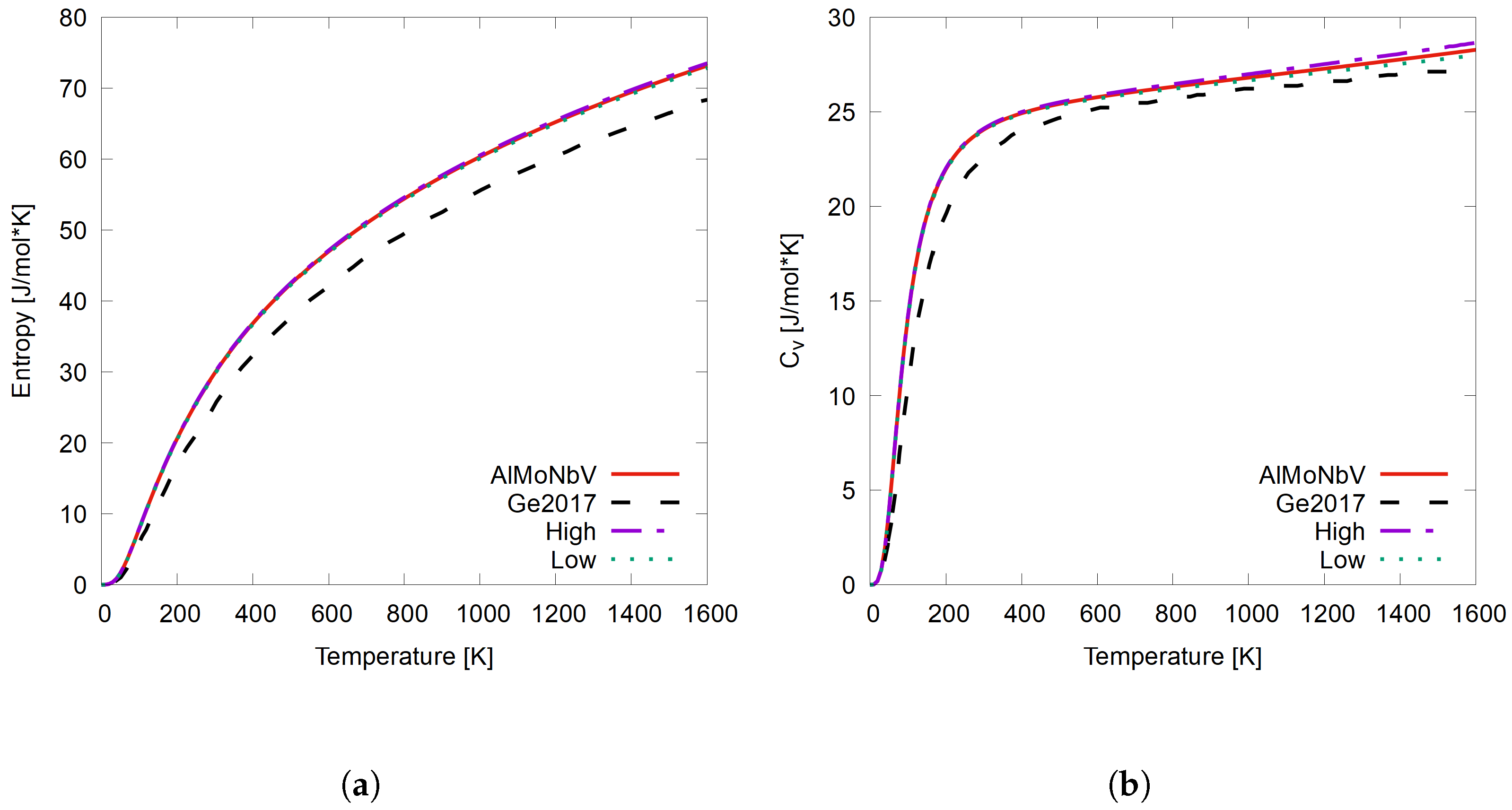
3.2. AlMoNbV—HEA Validation and Atomic Configuration Permutations
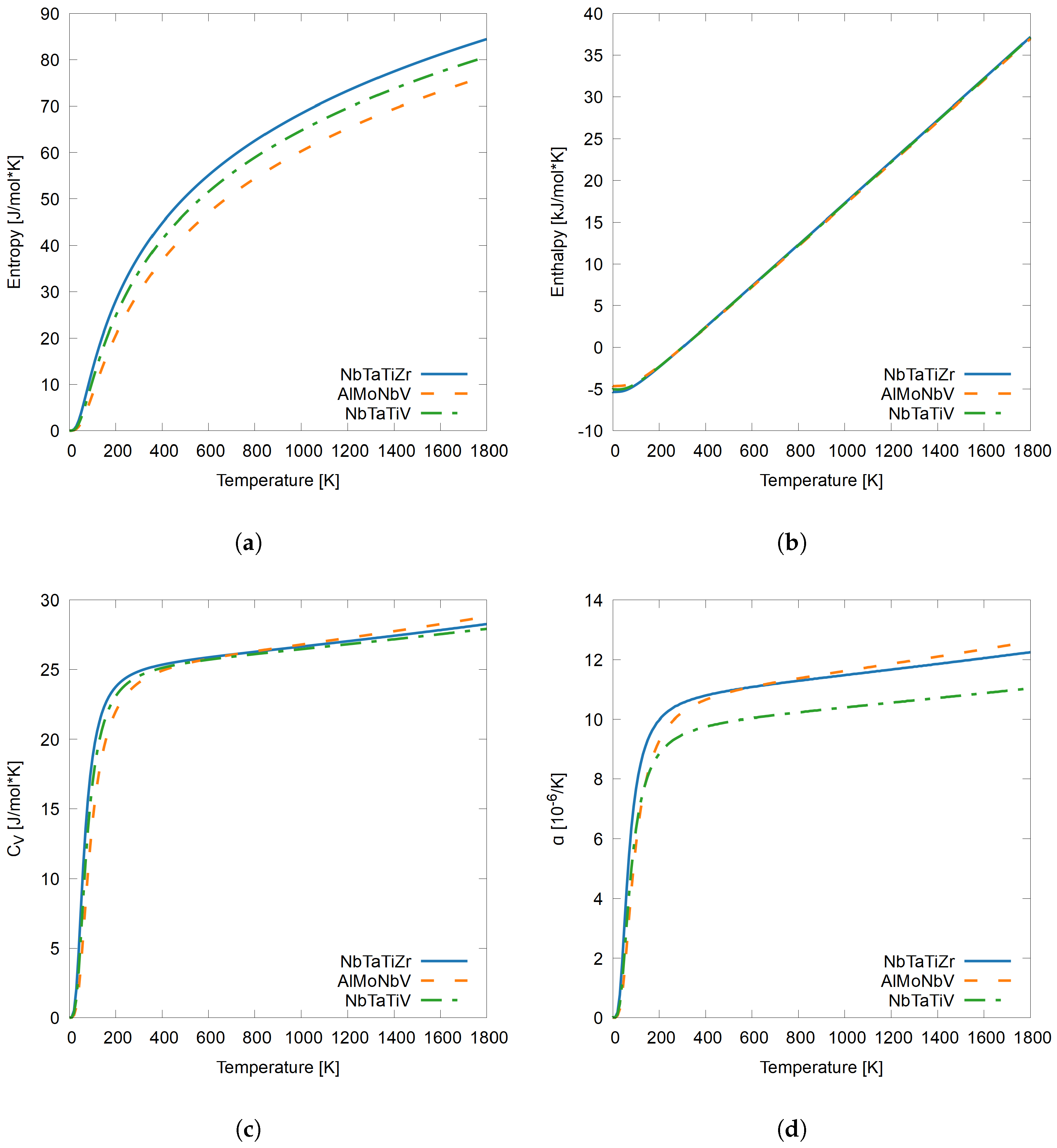
3.3. Thermodynamic Properties of RHEAs
3.3.1. Quaternary RHEAs
3.3.2. Quinary RHEAs
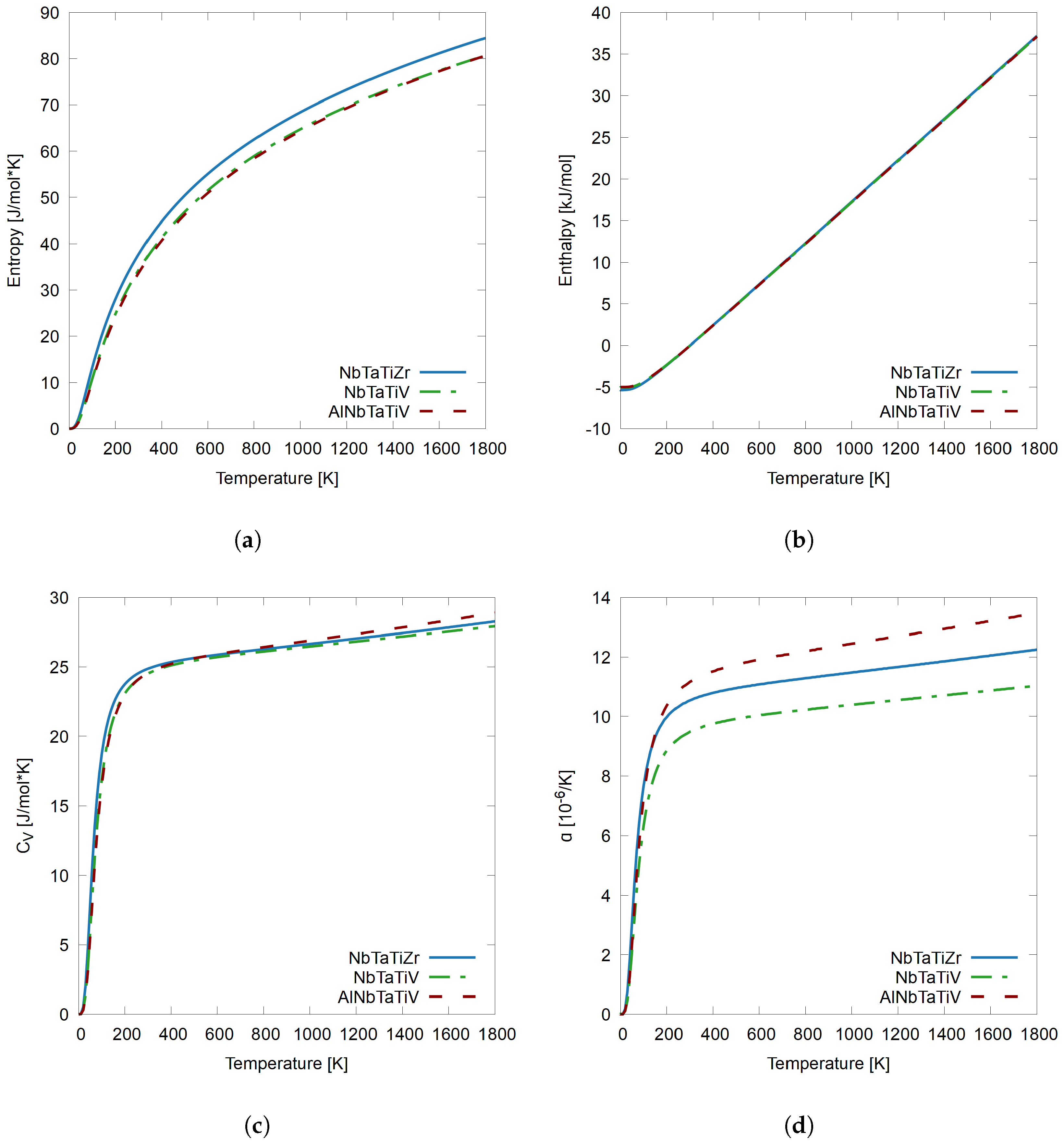
3.4. Comparing NbTaTiZr, NbTaTiV, and AlNbTaTiV
4. Conclusions
- Using DFT + SQS + Debye is a reliable method to use when calculating the finite temperature thermodynamic properties of an RHEA. This is because the difference between the highest and lowest calculated properties of entropy and heat capacity from all of the atomic configuration permutations of the quaternary system AlMoNbV vary by at most 1.7%. Only a few atomic permutation calculations are needed, depending on the number of chemical species in the alloy, which saves computational time and resources.
- The presence of Al and Zr elements with lower VEC in the RHEAs contributes to higher thermal expansion, while the presence of Mo, V, and W, elements with higher VEC contribute to lower thermal expansion, further validating the theory that lower VEC can lead to higher ductility in BCC refractory alloy design.
- In the RHEAs, the presence of Hf, an element with a VEC of four electrons, in the same way as other ductile metals such as Zr contributes to higher entropy and a higher compared to systems without Hf, especially at lower temperatures.
- At higher temperatures, Al contributes to the highest , which could be attributed to its capability for thermal expansion and low VEC of 3.
- V, Mo, and W elements with high VEC are the most structurally stable with the lowest thermal expansion, lowest , and entropy.
- The comparison of the compositionally similar systems as well as the comparison of quaternary and quinary systems offers valuable guidance for subsequent theoretical and experimental endeavors.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Correction Statement
Appendix A
| Type | GSE (eV/Atom) | (Å3/Atom) |
|---|---|---|
| AlMoNbV | −8.547 | 15.60 |
| AlMoVNb | −8.556 | 15.64 |
| AlNbMoV | −8.552 | 15.63 |
| AlNbVMo | −8.558 | 15.61 |
| AlVMoNb | −8.567 | 15.62 |
| AlVNbMo | −8.560 | 15.57 |
| MoAlNbV | −8.565 | 15.61 |
| MoAlVNb | −8.556 | 15.66 |
| MoNbAlV | −8.558 | 15.60 |
| MoNbVAl | −8.551 | 15.59 |
| MoVAlNb | −8.563 | 15.62 |
| MoVNbAl | −8.565 | 15.57 |
| NbAlMoV | −8.559 | 15.62 |
| NbAlVMo | −8.574 | 15.61 |
| NbMoAlV | −8.550 | 15.61 |
| NbMoVAl | −8.561 | 15.61 |
| NbVAlMo | −8.555 | 15.60 |
| NbVMoAl | −8.554 | 15.58 |
| VAlMoNb | −8.554 | 15.64 |
| VAlNbMo | −8.575 | 15.57 |
| VMoAlNb | −8.553 | 15.64 |
| VMoNbAl | −8.567 | 15.61 |
| VNbAlMo | −8.552 | 15.62 |
| VNbMoAl | −8.546 | 15.61 |
| AVG | −8.559 | 15.61 |
| STD | 0.007 | 0.023 |
| Max | −8.546 | 15.66 |
| Min | −8.575 | 15.57 |
References
- Miracle, D.B.; Senkov, O.N. A critical review of high entropy alloys and related concepts. Acta Mater. 2017, 122, 448–511. [Google Scholar] [CrossRef]
- Miracle, D.B.; Miller, J.D.; Senkov, O.N.; Woodward, C.; Uchic, M.D.; Tiley, J. Exploration and development of high entropy alloys for structural applications. Entropy 2014, 16, 494–525. [Google Scholar] [CrossRef]
- Senkov, O.N.; Wilks, G.; Scott, J.; Miracle, D.B. Mechanical properties of Nb25Mo25Ta25W25 and V20Nb20Mo20Ta20W20 refractory high entropy alloys. Intermetallics 2011, 19, 698–706. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, Y.; Qiao, Y.; Chen, G. Novel microstructure and properties of multicomponent CoCrCuFeNiTix alloys. Intermetallics 2007, 15, 357–362. [Google Scholar] [CrossRef]
- Senkov, O.N.; Miracle, D.B.; Chaput, K.J.; Couzinie, J.P. Development and exploration of refractory high entropy alloys—A review. J. Mater. Res. 2018, 33, 3092–3128. [Google Scholar] [CrossRef]
- Senkov, O.; Wilks, G.; Miracle, D.; Chuang, C.; Liaw, P. Refractory high-entropy alloys. Intermetallics 2010, 18, 1758–1765. [Google Scholar] [CrossRef]
- Philips, N.; Carl, M.; Cunningham, N. New opportunities in refractory alloys. Metall. Mater. Trans. A 2020, 51, 3299–3310. [Google Scholar] [CrossRef]
- Kwon, S.K.; Park, J.S.; Park, J.H. Influence of refractory-steel interfacial reaction on the formation behavior of inclusions in Ce-containing stainless steel. ISIJ Int. 2015, 55, 2589–2596. [Google Scholar] [CrossRef]
- Abyzov, V. Refractory cellular concrete based on phosphate binder from waste of production and recycling of aluminum. Procedia Eng. 2017, 206, 783–789. [Google Scholar] [CrossRef]
- Graddy, G.E., Jr.; Weirauch, D.A., Jr. Refractories Used for Aluminum Processing. In Alumina Chemicals: Science and Technology Handbook; Wiley: Hoboken, NJ, USA, 1990; pp. 489–493. [Google Scholar]
- Antusch, S.; Reiser, J.; Hoffmann, J.; Onea, A. Refractory Materials for Energy Applications. Energy Technol. 2017, 5, 1064–1070. [Google Scholar] [CrossRef]
- Pollock, T.M.; Tin, S. Nickel-based superalloys for advanced turbine engines: Chemistry, microstructure and properties. J. Propuls. Power 2006, 22, 361–374. [Google Scholar] [CrossRef]
- Wang, R.; Zhang, B.; Hu, D.; Jiang, K.; Liu, H.; Mao, J.; Jing, F.; Hao, X. Thermomechanical fatigue experiment and failure analysis on a nickel-based superalloy turbine blade. Eng. Fail. Anal. 2019, 102, 35–45. [Google Scholar] [CrossRef]
- Naser-Zoshki, H.; Kiani-Rashid, A.R.; Vahdati-Khaki, J. Design of a low density refractory high entropy alloy in non-equiatomic W–Mo–Cr–Ti–Al system. Vacuum 2020, 181, 109614. [Google Scholar] [CrossRef]
- Cutler, C.; Shaw, S. The interrelationship of γ’ size, grain size and mechanical properties in IN-939, a cast nickel-base superalloy. In Strength of Metals and Alloys; Elsevier: Amsterdam, The Netherlands, 1979; pp. 1357–1362. [Google Scholar]
- Yeh, J.W.; Lin, S.J. Breakthrough applications of high-entropy materials. J. Mater. Res. 2018, 33, 3129–3137. [Google Scholar] [CrossRef]
- Patel, K.; Sadeghilaridjani, M.; Pole, M.; Mukherjee, S. Hot corrosion behavior of refractory high entropy alloys in molten chloride salt for concentrating solar power systems. Sol. Energy Mater. Sol. Cells 2021, 230, 111222. [Google Scholar] [CrossRef]
- Hua, X.J.; Hu, P.; Xing, H.R.; Han, J.Y.; Ge, S.W.; Li, S.L.; He, C.J.; Wang, K.S.; Cui, C.J. Development and Property Tuning of Refractory High-Entropy Alloys: A Review. Acta Metall. Sin. English Lett. 2022, 2022, 1–35. [Google Scholar] [CrossRef]
- Zaddach, A.; Niu, C.; Koch, C.; Irving, D. Mechanical properties and stacking fault energies of NiFeCrCoMn high-entropy alloy. JOM 2013, 65, 1780–1789. [Google Scholar] [CrossRef]
- Zhang, Z.; Sheng, H.; Wang, Z.; Gludovatz, B.; Zhang, Z.; George, E.P.; Yu, Q.; Mao, S.X.; Ritchie, R.O. Dislocation mechanisms and 3D twin architectures generate exceptional strength-ductility-toughness combination in CrCoNi medium-entropy alloy. Nat. Commun. 2017, 8, 14390. [Google Scholar] [CrossRef]
- Huang, H.; Li, X.; Dong, Z.; Li, W.; Huang, S.; Meng, D.; Lai, X.; Liu, T.; Zhu, S.; Vitos, L. Critical stress for twinning nucleation in CrCoNi-based medium and high entropy alloys. Acta Mater. 2018, 149, 388–396. [Google Scholar] [CrossRef]
- Zhao, S.; Stocks, G.M.; Zhang, Y. Stacking fault energies of face-centered cubic concentrated solid solution alloys. Acta Mater. 2017, 134, 334–345. [Google Scholar] [CrossRef]
- Patriarca, L.; Ojha, A.; Sehitoglu, H.; Chumlyakov, Y. Slip nucleation in single crystal FeNiCoCrMn high entropy alloy. Scr. Mater. 2016, 112, 54–57. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhuang, Y.; Hu, A.; Kai, J.J.; Liu, C.T. The origin of negative stacking fault energies and nano-twin formation in face-centered cubic high entropy alloys. Scr. Mater. 2017, 130, 96–99. [Google Scholar] [CrossRef]
- Alkan, S.; Ojha, A.; Sehitoglu, H. Determination of latent hardening response for FeNiCoCrMn for twin-twin interactions. Acta Materialia 2018, 147, 149–164. [Google Scholar] [CrossRef]
- Niu, C.; LaRosa, C.R.; Miao, J.; Mills, M.J.; Ghazisaeidi, M. Magnetically-driven phase transformation strengthening in high entropy alloys. Nat. Commun. 2018, 9, 1363. [Google Scholar] [CrossRef] [PubMed]
- Ge, H.; Tian, F.; Wang, Y. Elastic and thermal properties of refractory high-entropy alloys from first-principles calculations. Comput. Mater. Sci. 2017, 128, 185–190. [Google Scholar] [CrossRef]
- Yang, M.; Shao, L.; Duan, J.M.; Chen, X.T.; Tang, B.Y. Temperature dependence of mechanical and thermodynamic properties of Ti (25+x) Zr25Nb25Ta (25-x)(x ≤ 20) refractory high entropy alloys: Influences of substitution of Ti for Ta. Phys. B Condens. Matter 2021, 606, 412851. [Google Scholar] [CrossRef]
- Song, H.; Tian, F.; Wang, D. Thermodynamic properties of refractory high entropy alloys. J. Alloy. Compd. 2016, 682, 773–777. [Google Scholar] [CrossRef]
- Yin, B.; Curtin, W.A. First-principles-based prediction of yield strength in the RhIrPdPtNiCu high-entropy alloy. NPJ Comput. Mater. 2019, 5, 14. [Google Scholar] [CrossRef]
- Ikeda, Y.; Grabowski, B.; Körmann, F. Ab initio phase stabilities and mechanical properties of multicomponent alloys: A comprehensive review for high entropy alloys and compositionally complex alloys. Mater. Charact. 2019, 147, 464–511. [Google Scholar] [CrossRef]
- Zunger, A.; Wei, S.H.; Ferreira, L.; Bernard, J.E. Special quasirandom structures. Phys. Rev. Lett. 1990, 65, 353. [Google Scholar] [CrossRef]
- Wei, S.H.; Ferreira, L.; Bernard, J.E.; Zunger, A. Electronic properties of random alloys: Special quasirandom structures. Phys. Rev. B 1990, 42, 9622. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, J.L.; Pomrehn, G.S.; Pribram-Jones, A.; Mahjoub, R.; Ferry, M.; Laws, K.J.; Bassman, L. Stacking fault energies of nondilute binary alloys using special quasirandom structures. Phys. Rev. B 2017, 95, 094112. [Google Scholar] [CrossRef]
- Zhao, S.; Weber, W.J.; Zhang, Y. Unique challenges for modeling defect dynamics in concentrated solid-solution alloys. JOM 2017, 69, 2084–2091. [Google Scholar] [CrossRef]
- De Jong, M.; Qi, L.; Olmsted, D.L.; van de Walle, A.; Asta, M. Calculations of planar defect energies in substitutional alloys using the special-quasirandom-structure approach. Phys. Rev. B 2016, 93, 094101. [Google Scholar] [CrossRef]
- Strother, J.D.; Hargather, C.Z. Statistical data set for first-principles calculations of stacking fault energies in an AlNbTaTiV high entropy alloy. Data Brief 2021, 34, 106670. [Google Scholar] [CrossRef]
- Kivy, M.B.; Zaeem, M.A. Generalized stacking fault energies, ductilities, and twinnabilities of CoCrFeNi-based face-centered cubic high entropy alloys. Scr. Mater. 2017, 139, 83–86. [Google Scholar] [CrossRef]
- Jiang, C.; Uberuaga, B.P. Efficient ab initio modeling of random multicomponent alloys. Phys. Rev. Lett. 2016, 116, 105501. [Google Scholar] [CrossRef] [PubMed]
- Senkov, O.; Scott, J.; Senkova, S.; Meisenkothen, F.; Miracle, D.; Woodward, C. Microstructure and elevated temperature properties of a refractory TaNbHfZrTi alloy. J. Mater. Sci. 2012, 47, 4062–4074. [Google Scholar] [CrossRef]
- Qi, L.; Chrzan, D.C. Tuning Ideal Tensile Strengths and Intrinsic Ductility of bcc Refractory Alloys. Phys. Rev. Lett. 2014, 112, 115503. [Google Scholar] [CrossRef]
- Wang, J.; Bai, S.; Tang, Y.; Li, S.; Liu, X.; Jia, J.; Ye, Y.; Zhu, L. Effect of the valence electron concentration on the yield strength of Ti–Zr–Nb–V high-entropy alloys. J. Alloy. Compd. 2021, 868, 159190. [Google Scholar] [CrossRef]
- Sheikh, S.; Shafeie, S.; Hu, Q.; Ahlström, J.; Persson, C.; Veselý, J.; Zýka, J.; Klement, U.; Guo, S. Alloy design for intrinsically ductile refractory high-entropy alloys. J. Appl. Phys. 2016, 120, 164902. [Google Scholar] [CrossRef]
- Yao, H.; Qiao, J.; Hawk, J.; Zhou, H.; Chen, M.; Gao, M. Mechanical properties of refractory high-entropy alloys: Experiments and modeling. J. Alloy. Compd. 2017, 696, 1139–1150. [Google Scholar] [CrossRef]
- Melnick, A.; Soolshenko, V. Thermodynamic design of high-entropy refractory alloys. J. Alloy. Compd. 2017, 694, 223–227. [Google Scholar] [CrossRef]
- Liao, M.; Liu, Y.; Min, L.; Lai, Z.; Han, T.; Yang, D.; Zhu, J. Alloying effect on phase stability, elastic and thermodynamic properties of Nb-Ti-V-Zr high entropy alloy. Intermetallics 2018, 101, 152–164. [Google Scholar] [CrossRef]
- Zheng, S.; Wang, S. First-Principles Design of Refractory High Entropy Alloy VMoNbTaW. Entropy 2018, 20, 965. [Google Scholar] [CrossRef]
- Dobbelstein, H.; Gurevich, E.L.; George, E.P.; Ostendorf, A.; Laplanche, G. Laser metal deposition of compositionally graded TiZrNbTa refractory high-entropy alloys using elemental powder blends. Addit. Manuf. 2019, 25, 252–262. [Google Scholar] [CrossRef]
- Bhandari, U.; Ghadimi, H.; Zhang, C.; Yang, S.; Guo, S. Predicting elastic constants of refractory complex concentrated alloys using machine learning approach. Materials 2022, 15, 4997. [Google Scholar] [CrossRef]
- Moruzzi, V.L.; Janak, J.F.; Schwarz, K. Calculated thermal properties of metals. Phys. Rev. B 1988, 37, 790–799. [Google Scholar] [CrossRef]
- Shang, S.L.; Wang, Y.; Kim, D.; Liu, Z.K. First-principles thermodynamics from phonon and Debye model: Application to Ni and Ni3Al. Comput. Mater. Sci. 2010, 47, 1040–1048. [Google Scholar] [CrossRef]
- Shang, S.; Böttger, A. A combined cluster variation method and ab initio approach to the γ-Fe [N]/γ’-Fe4N1- x phase equilibrium. Acta Mater. 2005, 53, 255–264. [Google Scholar] [CrossRef]
- Teter, D.; Gibbs, G.V.; Boisen, M.B., Jr.; Allan, D.; Teter, M. First-principles study of several hypothetical silica framework structures. Phys. Rev. B 1995, 52, 8064. [Google Scholar] [CrossRef]
- Debye, P. Zur Theorie der spezifischen Wärmen. Ann. Der Phys. 1912, 344, 789–839. [Google Scholar] [CrossRef]
- Abrahams, S.C.; Hsu, F.S.L. Debye temperatures and cohesive properties. J. Chem. Phys. 1975, 63, 1162–1165. [Google Scholar] [CrossRef]
- Degueldre, C.; Tissot, P.; Lartigue, H.; Pouchon, M. Specific heat capacity and Debye temperature of zirconia and its solid solution. Thermochim. Acta 2003, 403, 267–273. [Google Scholar] [CrossRef]
- Wang, X.; Bai, X.; Xiao, W.; Liu, Y.; Li, X.; Wang, J.; Peng, C.; Wang, L.; Wang, X. Calculation of Thermal Expansion Coefficient of Rare Earth Zirconate System at High Temperature by First Principles. Materials 2022, 15, 2264. [Google Scholar] [CrossRef] [PubMed]
- Kresse, G.; Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 1996, 6, 15–50. [Google Scholar] [CrossRef]
- Kresse, G.; Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 1996, 54, 11169–11186. [Google Scholar] [CrossRef]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized Gradient Approximation Made Simple. Phys. Rev. Lett. 1996, 77, 3865–3868. [Google Scholar] [CrossRef]
- Blöchl, P.E. Projector augmented-wave method. Phys. Rev. B 1994, 50, 17953. [Google Scholar] [CrossRef]
- Kresse, G.; Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 1999, 59, 1758–1775. [Google Scholar] [CrossRef]
- Kresse, G.; Marsman, M.; Furthmüller, J. VASP the Manual. Available online: https://www.vasp.at/wiki/index.php/The_VASP_Manual (accessed on 11 July 2023).
- Blochl, P.E.; Jepsen, O.; Andersen, O.K. Improved tetrahedron method for Brillouin-zone integrations. Phys. Rev. B 1994, 49, 16223–16233. [Google Scholar] [CrossRef]
- Van de Walle, A.; Tiwary, P.; De Jong, M.; Olmsted, D.; Asta, M.; Dick, A.; Shin, D.; Wang, Y.; Chen, L.Q.; Liu, Z.K. Efficient stochastic generation of special quasirandom structures. Calphad 2013, 42, 13–18. [Google Scholar] [CrossRef]
- Chase, M. NIST-JANAF Thermochemical Tables, 4th ed.; American Institute of Physics: College Park, MD, USA, 1998. [Google Scholar]
- He, X.; Bai, Y.; Zhu, C.; Sun, Y.; Li, M.; Barsoum, M. General trends in the structural, electronic and elastic properties of the M3AlC2 phases (M= transition metal): A first-principle study. Comput. Mater. Sci. 2010, 49, 691–698. [Google Scholar] [CrossRef]
- Khazaei, M.; Arai, M.; Sasaki, T.; Estili, M.; Sakka, Y. Trends in electronic structures and structural properties of MAX phases: A first-principles study on M2AlC (M= Sc, Ti, Cr, Zr, Nb, Mo, Hf, or Ta), M2AlN, and hypothetical M2AlB phases. J. Phys. Condens. Matter 2014, 26, 505503. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Ng, C.; Lu, J.; Liu, C. Effect of valence electron concentration on stability of fcc or bcc phase in high entropy alloys. J. Appl. Phys. 2011, 109, 103505. [Google Scholar] [CrossRef]
- Ikeda, Y.; Körmann, F.; Tanaka, I.; Neugebauer, J. Impact of chemical fluctuations on stacking fault energies of CrCoNi and CrMnFeCoNi high entropy alloys from first principles. Entropy 2018, 20, 655. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moreno, D.E.; Hargather, C.Z. Thermodynamic Properties as a Function of Temperature of AlMoNbV, NbTaTiV, NbTaTiZr, AlNbTaTiV, HfNbTaTiZr, and MoNbTaVW Refractory High-Entropy Alloys from First-Principles Calculations. Solids 2023, 4, 327-343. https://doi.org/10.3390/solids4040021
Moreno DE, Hargather CZ. Thermodynamic Properties as a Function of Temperature of AlMoNbV, NbTaTiV, NbTaTiZr, AlNbTaTiV, HfNbTaTiZr, and MoNbTaVW Refractory High-Entropy Alloys from First-Principles Calculations. Solids. 2023; 4(4):327-343. https://doi.org/10.3390/solids4040021
Chicago/Turabian StyleMoreno, Danielsen E., and Chelsey Z. Hargather. 2023. "Thermodynamic Properties as a Function of Temperature of AlMoNbV, NbTaTiV, NbTaTiZr, AlNbTaTiV, HfNbTaTiZr, and MoNbTaVW Refractory High-Entropy Alloys from First-Principles Calculations" Solids 4, no. 4: 327-343. https://doi.org/10.3390/solids4040021
APA StyleMoreno, D. E., & Hargather, C. Z. (2023). Thermodynamic Properties as a Function of Temperature of AlMoNbV, NbTaTiV, NbTaTiZr, AlNbTaTiV, HfNbTaTiZr, and MoNbTaVW Refractory High-Entropy Alloys from First-Principles Calculations. Solids, 4(4), 327-343. https://doi.org/10.3390/solids4040021






