Management of Type 2 and Post-Transplant Diabetes in Kidney Transplant Recipients: A Single-Center Clinical Experience with GLP-1 Receptor Agonists and SGLT-2 Inhibitors
Abstract
1. Introduction
2. Objectives
3. Material and Methods
3.1. Ethical Considerations
3.2. Study Design
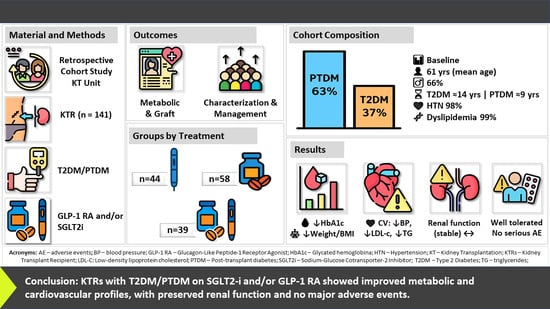
3.3. Study Participants
3.4. Clinical and Demographic Variables
3.5. Definitions
3.6. Metabolic Targets for Diabetes Mellitus Control
3.7. Outcome Measures
3.8. Statistical Analysis
4. Results
4.1. Sample Composition
4.2. Baseline Patient Characteristics
4.3. Cardiometabolic and Kidney Function Parameters
4.4. Pharmacologic Therapies
4.5. Diabetes-Related Complications
4.6. Adverse Effects
5. Discussion
Strengths and Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
Appendix A.1
| Cardiometabolic and Renal Parameters | Diabetes Mellitus | ||||||||
| All Patients (n = 141) | T2DM (n = 52) | PTDM (n = 89) | |||||||
| Baseline | Follow-Up | p * | Baseline | Follow-Up | p * | Baseline | Follow-Up | p * | |
| Glucose Metabolism and Lipid Profile | |||||||||
| Hb, g/dL | 13.15 ± 1.81 | 13.45 ± 1.77 | 0.65 | 13.29 ± 1.93 | 13.61 ± 1.69 | 0.06 | 13.07 ± 1.74 | 13.36 ± 1.81 | 0.02 |
| FPG, mg/dL (mean ± SD) | 122.22 ± 35.78 | 129.77 ± 36.64 | 0.68 | 139.77 ± 43.93 | 138.21 ± 44.52 | 0.46 | 112.62 ± 26.03 | 125.01 ± 30.60 | 0.03 |
| Hb A1c, % (mean ± SD) | 7.35 ± 1.20 | 7.04 ± 0.90 | 0.21 | 7.60 ± 1.20 | 7.10 ± 0.86 | <0.01 | 7.22 ± 1.18 | 7.01 ± 0.93 | 0.60 |
| TC, mg/dL (mean ± SD) | 175.87 ± 41.40 | 161.05 ± 40.41 | 0.02 | 176.47 ± 43.38 | 158.16 ± 39.24 | <0.01 | 175.53 ± 40.49 | 162.46 ± 41.14 | <0.01 |
| HDL-c, mg/dL (mean ± SD) | 49.10 ± 13.04 | 49.55 ± 12.20 | 0.17 | 49.82 ± 12.42 | 48.10 ± 10.95 | 0.13 | 48.69 ± 13.42 | 50.33 ± 12.84 | 0.07 |
| LDL-c, mg/dL (mean ± SD) | 89.68 ± 36.38 | 79.46 ± 33.33 | <0.001 | 94.04 ± 34.77 | 76.32 ± 29.68 | <0.001 | 87.23 ± 37.22 | 80.92 ± 35.00 | 0.02 |
| TG, mg/dL (mean ± SD) | 187.32 ± 99.86 | 175.09 ± 131.60 | 0.07 | 186.71 ± 92.98 | 166.08 ± 71.17 | 0.02 | 187.67 ± 104.03 | 179.72 ± 153.96 | 0.45 |
| Anthropometric Measurement | |||||||||
| Weight, kg (mean ± SD) | 75.36 ± 14.22 | 71.98 ± 12.81 | <0.001 | 77.89 ± 12.70 | 74.66 ± 10.73 | 0.70 | 73.95 ± 14.89 | 70.48 ± 13.67 | 0.03 |
| BMI, kg/m2 (mean ± SD) | 27.65 ± 4.63 | 26.37 ± 4.00 | <0.001 | 28.34 ± 4.14 | 27.14 ± 3.63 | <0.001 | 27.27 ± 4.86 | 25.95 ± 4.15 | <0.001 |
| Blood pressure status | |||||||||
| SBP, mmHg (mean ± SD) | 143.97 ± 17.69 | 137.85 ± 12.95 | <0.001 | 145.34 ± 18.43 | 139.06 ± 12.12 | <0.01 | 143.21 ± 17.31 | 137.18 ± 13.40 | <0.01 |
| DBP, mmHg (mean ± SD) | 76.06 ± 14.65 | 74.37 ± 10.02 | <0.001 | 75.68 ± 11.59 | 74.66 ± 9.39 | 0.58 | 76.27 ± 16.16 | 74.21 ± 10.41 | 0.08 |
| Renal function parameters | |||||||||
| Creatinine, mg/dL (mean ± SD) | 1.55 ± 0.55 | 1.58 ± 0.88 | 0.58 | 1.47 ± 0.49 | 1.61 ± 1.22 | 0.26 | 1.59 ± 0.58 | 1.57 ± 0.60 | 0.68 |
| Urea, mg/dL (mean ± SD) | 70.92 ± 27.81 | 69.45 ± 29.52 | 0.55 | 63.87 ± 24.22 | 66.24 ± 29.87 | 0.47 | 74.94 ± 29.03 | 71.19 ± 29.34 | 0.14 |
| eGFR, ml/min (mean ± SD) | 53.60 ± 21.71 | 54.38 ± 21.91 | 0.53 | 55.15 ± 23.70 | 53.57 ± 23.09 | 0.42 | 52.74 ± 20.61 | 54.81 ± 21.37 | 0.11 |
| * Significant p-values (p < 0.05) are highlighted in bold. Abbreviations: Hb, Hemoglobin; FPG, Fasting Plasma Glucose; Hb A1c, Glycated Hemoglobin; TC, Total Cholesterol; HDL-c, High-Density Lipoprotein Cholesterol; LDL-c, Low-Density Lipoprotein Cholesterol; TG, Triglycerides; BMI, Body Mass Index; SBP, Systolic Blood Pressure; DBP, Diastolic Blood Pressure; eGFR, Estimated Glomerular Filtration Rate. | |||||||||
Appendix A.2
| Pharmacological Treatment Distribution | Diabetes Mellitus | |||||
| All Patients (n = 141) | T2DM (n = 52) | PTDM (n = 89) | ||||
| Baseline | Follow-Up | Baseline | Follow-Up | Baseline | Follow-Up | |
| Antidiabetic Therapy * | ||||||
| BG (MTF), n (%) | 41/141 (29%) | 33/141(23%) | 12/52 (23%) | 9/52 (17%) | 29/89 (33%) | 24/89 (27%) |
| SU, n (%) | 12/141 (9%) | 9/141 (6%) | 2/52 (4%) | 0/52 (0%) | 10/89 (11%) | 9/89 (10%) |
| DPP-4i, n (%) | 50/141 (35%) | 38/141 (27%) | 15/52 (29%) | 13/52 (25%) | 35/89 (39%) | 25/89 (28%) |
| SGLT-2i **, n (%) | ------ | 97/141 (69%) | ------ | 32/52 (62%) | ------ | 65/89 (73%) |
| GLP-1 RA **, n (%) | ------ | 83/141 (59%) | ------ | 32/52 (62%) | ------ | 51/89 (57%) |
| TZD/AGI/MG, n (%) | NR | NR | NR | NR | NR | NR |
| Insulin Therapy ***, n (%) | 76/141 (54%) | 75/141 (53%) | 33/52 (63%) | 33/52 (63%) | 43/89 (48%) | 42/89 (47%) |
| Lipid-Lowering Agents | ||||||
| Statins | ||||||
| Atorvastatin (ATV) | 84/141 (60%) | 84/141 (60%) | 33/52 (63%) | 33/52 (63%) | 51/89 (57%) | 51/89 (57%) |
| Rosuvastatin (RSV) | 35/141 (25%) | 35/141 (25%) | 13/52 (25%) | 13/52 (25%) | 22/89 (25%) | 22/89 (25%) |
| Simvastatin (SV) | 22/141 (15%) | 22/141 (15%) | 6/52 (12%) | 6/52 (12%) | 16/89 (18%) | 16/89 (18%) |
| Pravastatin (PV) | NR | NR | NR | NR | NR | NR |
| Lovastatin (LV) | NR | NR | NR | NR | NR | NR |
| Fluvastatin (FV) | NR | NR | NR | NR | NR | NR |
| Pitavastatin (PTV) | NR | NR | NR | NR | NR | NR |
| Ezetimibe added to statin therapy | 58/141 (41%) | 58/141 (41%) | 21/52 (40%) | 21/52 (40%) | 37/89 (41%) | 37/89 (41%) |
| Antihypertensive Agents **** | ||||||
| ACEi, n (%) | 82/141 (58%) | 82/141 (58%) | 28/52 (54%) | 28/52 (54%) | 54/89 (61%) | 54/89 (61%) |
| ARB, n (%) | 28/141 (20%) | 28/141 (20%) | 8/52 (15%) | 8/52 (15%) | 20/89 (22%) | 20/89 (22%) |
| CCB, n (%) | 102/141 (72%) | 102/141 (72%) | 37/52 (71%) | 37/52 (71%) | 65/89 (73%) | 65/89 (73%) |
| BB, n (%) | 111/141 (79%) | 111/141 (79%) | 45/52 (87%) | 45/52 (87%) | 66/89 (74%) | 66/89 (74%) |
| Diuretics, n (%) | 55/141 (39%) | 55/141 (39%) | 24/52 (46%) | 24/52 (46%) | 31/89 (35%) | 31/89 (35%) |
| CAA, n (%) | 23/141 (16%) | 23/141 (16%) | 12/52 (23%) | 12/52 (23%) | 11/89 (12%) | 11/89 (12%) |
| * The classes of oral antidiabetic agents are not mutually exclusive and are often used in combination to achieve optimal glycemic control. ** SGLT2-i and GLP-1 RA were excluded from baseline analysis, as the study focuses on their introduction during treatment. *** Insulin can be used alone or combined with other antidiabetic drugs, tailored to the patient’s needs and glycemic control goals. **** The classes of antihypertensive agents are not mutually exclusive and are often used in combination to achieve optimal blood pressure control. NR: Not Reported. Abbreviations: ACEi: Angiotensin-Converting Enzyme Inhibitors; AGI: Alpha-Glucosidase Inhibitors; ARB: Angiotensin II Receptor Blockers; BB: Beta-Blockers; BG: Biguanides; CAA: Central Acting Agents; CCB: Calcium Channel Blockers; DPP-4i: Dipeptidyl Peptidase-4 Inhibitors; GLP-1 RA: Glucagon-Like Peptide-1 Receptor Agonists; MG: Meglitinides; SGLT-2i: Sodium-Glucose Cotransporter-2 Inhibitors; STAT: Statins (HMG-CoA Reductase Inhibitors); SU: Sulfonylureas; TZD: Thiazolidinediones. | ||||||
References
- Tonelli, M.; Wiebe, N.; Knoll, G.; Bello, A.; Browne, S.; Jadhav, D.; Klarenbach, S.; Gill, J. Systematic review: Kidney transplantation compared with dialysis in clinically relevant outcomes. Am. J. Transplant. 2011, 11, 2093–2109. [Google Scholar] [CrossRef]
- Cameron, J.I.; Whiteside, C.; Katz, J.; Devins, G.M. Differences in quality of life across renal replacement therapies: A meta-analytic comparison. Am. J. Kidney Dis. 2000, 35, 629–637. [Google Scholar] [CrossRef]
- McGill, R.L.; Ko, T.Y. Transplantation and the primary care physician. Adv. Chronic Kidney Dis. 2011, 18, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, R.A.; Ashby, V.B.; Milford, E.L.; Ojo, A.O.; Ettenger, R.E.; Agodoa, L.Y.C.; Held, P.J.; Port, F.K. Comparison of mortality in all patients on dialysis, patients on dialysis awaiting transplantation, and recipients of a first cadaveric transplant. N. Engl. J. Med. 1999, 341, 1725–1730. [Google Scholar] [CrossRef] [PubMed]
- Meier-Kriesche, H.-U.; Schold, J.D.; Kaplan, B. Long-term renal allograft survival: Have we made significant progress or is it time to rethink our analytic and therapeutic strategies? Am. J. Transplant. 2004, 4, 1289–1295. [Google Scholar] [CrossRef]
- Agrawal, A.; Ison, M.G.; Danziger-Isakov, L. Long-Term Infectious Complications of Kidney Transplantation. Clin. J. Am. Soc. Nephrol. 2022, 17, 286–295. [Google Scholar] [CrossRef]
- Khan, M.A.; Hanna, A.; Sridhara, S.; Chaudhari, H.; Me, H.M.; Attieh, R.M.; Abu Jawdeh, B.G. Maintenance Immunosuppression in Kidney Transplantation: A Review of the Current Status and Future Directions. J. Clin. Med. 2025, 14, 1821. [Google Scholar] [CrossRef]
- Kanbay, M.; Siriopol, D.; Guldan, M.; Ozbek, L.; Topcu, A.U.; Siriopol, I.; Tuttle, K. Prognostic impact of post-transplant diabetes mellitus in kidney allograft recipients: A meta-analysis. Nephrol. Dial. Transplant. 2024, 40, 554–576. [Google Scholar] [CrossRef]
- Malik, R.F.; Jia, Y.; Mansour, S.G.; Reese, P.P.; Hall, I.E.; Alasfar, S.; Doshi, M.D.; Akalin, E.; Bromberg, J.S.; Harhay, M.N.; et al. Post-transplant Diabetes Mellitus in Kidney Transplant Recipients: A Multicenter Study. Kidney360 2021, 2, 1296–1307. [Google Scholar] [CrossRef]
- Jeon, J.Y.; Han-Bit, S.; Park, B.H.; Lee, N.; Kim, H.J.; Kim, D.J.; Lee, K.-W.; Han, S.J. Impact of Post-Transplant Diabetes Mellitus on Survival and Cardiovascular Events in Kidney Transplant Recipients. Endocrinol. Metab. 2023, 38, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Rossi, M.R.; Mazzali, M.; de Sousa, M.V. Post-transplant diabetes mellitus: Risk factors and outcomes in a 5-year follow-up. Front. Clin. Diabetes Health 2024, 5, 1336896. [Google Scholar] [CrossRef]
- Shivaswamy, V.; Boerner, B.; Larsen, J. Post-Transplant Diabetes Mellitus: Causes, Treatment, and Impact on Outcomes. Endocr. Rev. 2015, 37, 37–61. [Google Scholar] [CrossRef]
- Kang, E.; Lee, J.; Kang, D.H.; Park, J.; Park, S.; Kim, Y.C.; Kim, D.K.; Joo, K.W.; Park, M.; Lim, Y.; et al. Clinical outcomes of posttransplantation diabetes mellitus in kidney transplantation recipients: A nationwide population-based cohort study in Korea. Sci. Rep. 2022, 12, 21632. [Google Scholar] [CrossRef] [PubMed]
- Zinman, B.; Wanner, C.; Lachin, J.M.; Fitchett, D.; Bluhmki, E.; Hantel, S.; Mattheus, M.; Devins, T.; Johansen, O.E.; Woerle, H.J.; et al. Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes. N. Engl. J. Med. 2015, 373, 2117–2128. [Google Scholar] [CrossRef]
- The EMPA-KIDNEY Collaborative Group; Herrington, W.G.; Staplin, N.; Wanner, C.; Green, J.B.; Hauske, S.J.; Emberson, J.R.; Preiss, D.; Judge, P.; Mayne, K.J.; et al. Empagliflozin in Patients with Chronic Kidney Disease. N. Engl. J. Med. 2023, 388, 117–127. [Google Scholar] [CrossRef]
- Wiviott, S.D.; Raz, I.; Bonaca, M.P.; Mosenzon, O.; Kato, E.T.; Cahn, A.; Silverman, M.G.; Zelniker, T.A.; Kuder, J.F.; Murphy, S.A.; et al. Dapagliflozin and Cardiovascular Outcomes in Type 2 Diabetes. N. Engl. J. Med. 2019, 380, 347–357. [Google Scholar] [CrossRef] [PubMed]
- Heerspink, H.J.L.; Stefánsson, B.V.; Correa-Rotter, R.; Chertow, G.M.; Greene, T.; Hou, F.-F.; Mann, J.F.E.; McMurray, J.J.V.; Lindberg, M.; Rossing, P.; et al. Dapagliflozin in Patients with Chronic Kidney Disease. N. Engl. J. Med. 2020, 383, 1436–1446. [Google Scholar] [CrossRef]
- Marso, S.P.; Daniels, G.H.; Brown-Frandsen, K.; Kristensen, P.; Mann, J.F.E.; Nauck, M.A.; Nissen, S.E.; Pocock, S.; Poulter, N.R.; Ravn, L.S.; et al. Liraglutide and Cardiovascular Outcomes in Type 2 Diabetes. N. Engl. J. Med. 2016, 375, 311–322. [Google Scholar] [CrossRef] [PubMed]
- Marso, S.P.; Bain, S.C.; Consoli, A.; Eliaschewitz, F.G.; Jódar, E.; Leiter, L.A.; Lingvay, I.; Rosenstock, J.; Seufert, J.; Warren, M.L.; et al. Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes. N. Engl. J. Med. 2016, 375, 1834–1844. [Google Scholar] [CrossRef]
- Gerstein, H.C.; Colhoun, H.M.; Dagenais, G.R.; Diaz, R.; Lakshmanan, M.; Pais, P.; Probstfield, J.; Riesmeyer, J.S.; Riddle, M.C.; Rydén, L.; et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): A double-blind, randomised placebo-controlled trial. Lancet 2019, 394, 121–130. [Google Scholar] [CrossRef]
- American Diabetes Association Professional Practice Committee; ElSayed, N.A.; Aleppo, G.; Bannuru, R.R.; Bruemmer, D.; Collins, B.S.; Ekhlaspour, L.; Gaglia, J.L.; Hilliard, M.E.; Johnson, E.L.; et al. 2. Diagnosis and classification of diabetes: Standards of care in diabetes—2024. Diabetes Care 2024, 47 (Suppl. 1), S20–S42. [Google Scholar] [CrossRef]
- Sharif, A.; Chakkera, H.; de Vries, A.P.J.; Eller, K.; Guthoff, M.; Haller, M.C.; Hornum, M.; Nordheim, E.; Kautzky-Willer, A.; Krebs, M.; et al. International consensus on post-transplantation diabetes mellitus. Nephrol. Dial. Transplant. 2024, 39, 531–549. [Google Scholar] [CrossRef]
- Stevens, P.E.; Ahmed, S.B.; Carrero, J.J.; Foster, B.; Francis, A.; Hall, R.K.; Herrington, W.G.; Hill, G.; Inker, L.A.; Kazancıoğlu, R.; et al. KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney Int. 2024, 105, S117–S314. [Google Scholar] [CrossRef]
- Halden, T.A.S.; Kvitne, K.E.; Midtvedt, K.; Rajakumar, L.; Robertsen, I.; Brox, J.; Bollerslev, J.; Hartmann, A.; Åsberg, A.; Jenssen, T. Efficacy and Safety of Empagliflozin in Renal Transplant Recipients with Posttransplant Diabetes Mellitus. Diabetes Care 2019, 42, 1067–1074. [Google Scholar] [CrossRef]
- Fructuoso, A.I.S.; Raba, A.B.; Deras, E.B.; Sánchez, L.A.V.; Cecilio, R.V.S.; Esteve, A.F.; Vega, L.C.; Martínez, E.G.; Garcia, M.E.G.; Coronado, P.S.; et al. Sodium-glucose cotransporter-2 inhibitor therapy in kidney transplant patients with type 2 or post-transplant diabetes: An observational multicentre study. Clin. Kidney J. 2023, 16, 1022–1034. [Google Scholar] [CrossRef]
- AlKindi, F.; Al-Omary, H.L.; Hussain, Q.; Al Hakim, M.; Chaaban, A.; Boobes, Y. Outcomes of SGLT2 Inhibitors Use in Diabetic Renal Transplant Patients. Transplant Proc. 2020, 52, 175–178. [Google Scholar] [CrossRef] [PubMed]
- Attallah, N.; Yassine, L. Use of Empagliflozin in Recipients of Kidney Transplant: A Report of 8 Cases. Transplant Proc. 2019, 51, 3275–3280. [Google Scholar] [CrossRef] [PubMed]
- Mahling, M.; Schork, A.; Nadalin, S.; Fritsche, A.; Heyne, N.; Guthoff, M. Sodium-Glucose Cotransporter 2 (SGLT2) Inhibition in Kidney Transplant Recipients with Diabetes Mellitus. Kidney Blood Press. Res. 2019, 44, 984–992. [Google Scholar] [CrossRef] [PubMed]
- Vigara, L.A.; Villanego, F.; Orellana, C.; Naranjo, J.; Torrado, J.; Garcia, T.; Mazuecos, A. Effectiveness and safety of glucagon-like peptide-1 receptor agonist in a cohort of kidney transplant recipients. Clin. Transplant. 2022, 36, e14633. [Google Scholar] [CrossRef]
- Mallik, R.; Ali, O.; Casabar, M.; Mukuba, D.; Byrne, C.; McCafferty, K.; Yaqoob, M.M.; Chowdhury, T.A. Glucagon-like peptide-1 receptor analogues in renal transplant recipients with diabetes: Medium term follow of patients from a single UK centre. Diabet. Med. 2023, 40, e15057. [Google Scholar] [CrossRef]
- Lemke, A.; Brokmeier, H.M.; Leung, S.B.; Mara, K.C.; Mour, G.K.; Wadei, H.M.; Hill, J.M.; Stegall, M.; Kudva, Y.C.; Shah, P.; et al. Sodium-glucose cotransporter 2 inhibitors for treatment of diabetes mellitus after kidney transplantation. Clin. Transplant. 2022, 36, e14718. [Google Scholar] [CrossRef] [PubMed]
- Kukla, A.; Hill, J.D.; Merzkani, M.; Bentall, A.; Lorenz, E.C.; Park, W.D.B.; D’cOsta, M.; Kudva, Y.C.; Stegall, M.D.; Shah, P. The Use of GLP1R Agonists for the Treatment of Type 2 Diabetes in Kidney Transplant Recipients. Transplant. Direct 2020, 6, e524. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Lee, J.; Jung, C.H.; Park, J.-Y.; Lee, W.J. Dulaglutide as an Effective Replacement for Prandial Insulin in Kidney Transplant Recipients with Type 2 Diabetes Mellitus: A Retrospective Review. Diabetes Metab. J. 2021, 45, 948–953. [Google Scholar] [CrossRef]
- Liou, J.-H.; Liu, Y.-M.; Chen, C.-H. Management of Diabetes Mellitus with Glucagonlike Peptide-1 Agonist Liraglutide in Renal Transplant Recipients: A Retrospective Study. Transplant. Proc. 2018, 50, 2502–2505. [Google Scholar] [CrossRef]
- González, A.Y.; Kanter, J.; Sancho, A.; Gavela, E.; Solá, E.; Ávila, A.; Pallardó, L.M. Institutional Experience with New Antidiabetic Drugs in Kidney Transplant. Transplant. Proc. 2021, 53, 2678–2680. [Google Scholar] [CrossRef] [PubMed]
- Schwaiger, E.; Burghart, L.; Signorini, L.; Ristl, R.; Kopecky, C.; Tura, A.; Pacini, G.; Wrba, T.; Antlanger, M.; Schmaldienst, S.; et al. Empagliflozin in posttransplantation diabetes mellitus: A prospective, interventional pilot study on glucose metabolism, fluid volume, and patient safety. Am. J. Transplant. 2018, 19, 907–919. [Google Scholar] [CrossRef]
- Rajasekeran, H.; Kim, S.J.; Cardella, C.J.; Schiff, J.; Cattral, M.; Cherney, D.Z.; Singh, S.K. Use of Canagliflozin in Kidney Transplant Recipients for the Treatment of Type 2 Diabetes: A Case Series. Diabetes Care 2017, 40, e75–e76. [Google Scholar] [CrossRef]
- Thomsen, R.W.; Mailhac, A.; Løhde, J.B.; Pottegård, A. Real-world evidence on the utilization, clinical and comparative effectiveness, and adverse effects of newer GLP-1RA-based weight-loss therapies. Diabetes Obes. Metab. 2025, 27 (Suppl. 2), 66–88. [Google Scholar] [CrossRef]


| Diabetes Mellitus | ||||
|---|---|---|---|---|
| Parameters | Total Cohort (n = 141) | T2DM Group (n = 52) | PTDM Group (n = 89) | p-Value * |
| Clinical Profile | ||||
| Recipient male gender, n (%) | 93 (66%) | 36 (69%) | 57 (64%) | 0.530 |
| Age, years (mean ± SD) | 61.20 ± 10.69 | 63.43 ± 9.96 | 59.95 ± 10.92 | 0.056 |
| Duration of DM, years (mean ± SD) | 10.94 ± 10.44 | 13.77 ± 14.50 | 9.29 ± 6.64 | 0.039 |
| Time since KT until study inclusion, years | 10.8 ± 6.2 | 6.12 ± 4.89 | 9.67 ± 6.28 | <0.001 |
| Family history of diabetes, n (%) | 83 (59%) | 35 (67%) | 48 (54%) | 0.140 |
| Renal replacement therapy ** | ||||
| Hemodialysis, n (%) | 112 (80%) | 44 (85%) | 68 (76%) | 0.350 |
| Peritoneal dialysis, n (%) | 20 (14%) | 6 (12%) | 14 (16%) | 0.440 |
| None, n (%) | 9 (6%) | 2 (4%) | 7 (8%) | 0.320 |
| Viral Serostatus | ||||
| Hepatitis C positive, n (%) | 5 (4%) | 3 (6%) | 2 (2%) | 0.280 |
| CMV antibody positive recipient, n (%) | 92 (65%) | 41 (79%) | 51 (57%) | <0.010 |
| Primary kidney disease | ||||
| Diabetic kidney disease (DKD), n (%) | 28 (20%) | 28 (54%) | NR | --- |
| Glomerular disease, n (%) | 30 (21%) | 6 (12%) | 24 (27%) | 0.05 |
| Polycystic disease, n (%) | 20 (14%) | 2 (4%) | 18 (20%) | <0.010 |
| Tubulointerstitial disease, n (%) | 12 (9%) | 3 (6%) | 9 (10%) | 0.420 |
| Other causes, n (%) | 14 (10%) | 3 (6%) | 11 (12%) | 0.250 |
| Unknown, n (%) | 37 (26%) | 10 (18%) | 27 (31%) | 0.720 |
| Transplantation information | ||||
| Deceased donor, n (%) | 114 (81%) | 45 (87%) | 69 (78%) | 0.190 |
| Graft number | ||||
| First, n (%) | 116 (82%) | 43 (83%) | 73 (82%) | 0.860 |
| Second, n (%) | 21 (15%) | 7 (13%) | 14 (16%) | 0.970 |
| More than two, n (%) | 4 (3%) | 2 (4%) | 2 (2%) | 0.730 |
| Immunosuppressive Therapy | ||||
| Prednisone (PDN) | 132 (94%) | 48 (92%) | 84 (94%) | 0.260 |
| Mycophenolate Mofetil (MMF) | 125 (89%) | 48 (92%) | 77 (87%) | 0.730 |
| Azathioprine (AZA) | 9 (6%) | 1 (2%) | 8 (9%) | 0.990 |
| Tacrolimus (TAC) | 119 (84%) | 44 (85%) | 75 (84%) | 0.300 |
| Cyclosporine (CsA) | 19 (13%) | 6 (12%) | 13 (15%) | 0.300 |
| mTOR Inhibitors (mTORi) | 33 (23%) | 14 (27%) | 19 (21%) | 0.250 |
| Treatment Exposure | ||||
| SGLT2 inhibitor use, n (%) | 58 (41.13%) | 20 (38.46%) | 38 (42.69%) | 0.612 |
| SGLT2i treatment duration, years | 0.412 | |||
| (mean ± SD) | 1.97 ± 2.36 | 1.73 ± 0.89 | 1.94 ± 1.37 | |
| median [IQR] | 1.59 [1.07–2.18] | 1.76 [1.24–2.18] | 1.59 [1.07–2.18] | |
| SGLT2i initiation year | 2022 [2018–2023] | 2022 [2019–2023] | 2022 [2018–2023] | |
| SGLT2i person-time, years | 114.2 | 55.4 | 120.3 | |
| GLP-1 RA use, n (%) | 44 (31.20%) | 20 (38.46%) | 24 (26.96%) | 0.154 |
| GLP-1 RA treatment duration, years | 0.487 | |||
| (mean ± SD) | 2.12 ± 1.35 | 1.97 ± 0.89 | 2.21 ± 1.53 | |
| median [IQR] | 1.85 [1.18–2.80] | 1.93 [1.35–2.55] | 1.85 [1.18–2.80] | |
| GLP-1 RA initiation year | 2022 [2013–2023] | 2022 [2019–2023] | 2022 [2013–2023] | |
| GLP-1 RA person-time, years | 93.3 | 39.4 | 53.04 | |
| Combination Therapy | ||||
| Concurrent use, n (%) | 39 (27.65%) | 12 (23.07%) | 27 (30.33%) | 0.358 |
| Combination therapy duration, years | 0.589 | |||
| (mean ± SD) | 1.95 ± 0.99 | 2.08 ± 1.07 | 1.90 ± 0.96 | |
| median [IQR] | 1.85 [1.18–2.55] | 1.93 [1.35–2.55] | 1.76 [1.18–2.55] | |
| Combination initiation year | 2022 [2019–2023] | 2022 [2019–2023] | 2022 [2019–2023] | |
| Combination therapy person-time, years | 76.05 | 24.96 | 51.3 | |
| Diabetes Mellitus Total Cohort (n = 141) | |||
|---|---|---|---|
| Parameters | Baseline | Follow-Up | p-Value * |
| Glucose Metabolism and Lipid Profile | |||
| Hb, g/dL | 13.15 ± 1.81 | 13.45 ± 1.77 | 0.65 |
| FPG, mg/dL (mean ± SD) | 122.22 ± 35.78 | 129.77 ± 36.64 | 0.68 |
| Hb A1c, % (mean ± SD) | 7.35 ± 1.20 | 7.04 ± 0.90 | 0.21 |
| TC, mg/dL (mean ± SD) | 175.87 ± 41.40 | 161.05 ± 40.41 | 0.02 |
| HDL-c, mg/dL (mean ± SD) | 49.10 ± 13.04 | 49.55 ± 12.20 | 0.17 |
| LDL-c, mg/dL(mean ± SD) | 89.68 ± 36.38 | 79.46 ± 33.33 | <0.001 |
| TG, mg/dL (mean ± SD) | 187.32 ± 99.86 | 175.09 ± 131.60 | 0.07 |
| Anthropometric Measurement | |||
| Weight, kg (mean ± SD) | 75.36 ± 14.22 | 71.98 ± 12.81 | <0.001 |
| BMI, kg/m2 (mean ± SD) | 27.65 ± 4.63 | 26.37 ± 4.00 | <0.001 |
| Blood pressure status | |||
| SBP, mmHg (mean ± SD) | 143.97 ± 17.69 | 137.85 ± 12.95 | <0.001 |
| DBP, mmHg (mean ± SD) | 76.06 ± 14.65 | 74.37 ± 10.02 | <0.001 |
| Renal function parameters | |||
| Creatinine, mg/dL (mean ± SD) | 1.55 ± 0.55 | 1.58 ± 0.88 | 0.58 |
| Urea, mg/dL (mean ± SD) | 70.92 ± 27.81 | 69.45 ± 29.52 | 0.55 |
| eGFR, ml/min (mean ± SD) | 53.60 ± 21.71 | 54.38 ± 21.91 | 0.53 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Navarrete, R.E.T.; Freitas, J.C.; Fonseca, I.; Cunha, A.; Sa, J.R.; Martins, L.S. Management of Type 2 and Post-Transplant Diabetes in Kidney Transplant Recipients: A Single-Center Clinical Experience with GLP-1 Receptor Agonists and SGLT-2 Inhibitors. Diabetology 2025, 6, 158. https://doi.org/10.3390/diabetology6120158
Navarrete RET, Freitas JC, Fonseca I, Cunha A, Sa JR, Martins LS. Management of Type 2 and Post-Transplant Diabetes in Kidney Transplant Recipients: A Single-Center Clinical Experience with GLP-1 Receptor Agonists and SGLT-2 Inhibitors. Diabetology. 2025; 6(12):158. https://doi.org/10.3390/diabetology6120158
Chicago/Turabian StyleNavarrete, Ricardo E. T., Joana C. Freitas, Isabel Fonseca, Ana Cunha, Joao Roberto Sa, and La Salete Martins. 2025. "Management of Type 2 and Post-Transplant Diabetes in Kidney Transplant Recipients: A Single-Center Clinical Experience with GLP-1 Receptor Agonists and SGLT-2 Inhibitors" Diabetology 6, no. 12: 158. https://doi.org/10.3390/diabetology6120158
APA StyleNavarrete, R. E. T., Freitas, J. C., Fonseca, I., Cunha, A., Sa, J. R., & Martins, L. S. (2025). Management of Type 2 and Post-Transplant Diabetes in Kidney Transplant Recipients: A Single-Center Clinical Experience with GLP-1 Receptor Agonists and SGLT-2 Inhibitors. Diabetology, 6(12), 158. https://doi.org/10.3390/diabetology6120158