Polysaccharide Vaccines: A Perspective on Non-Typhoidal Salmonella
Abstract
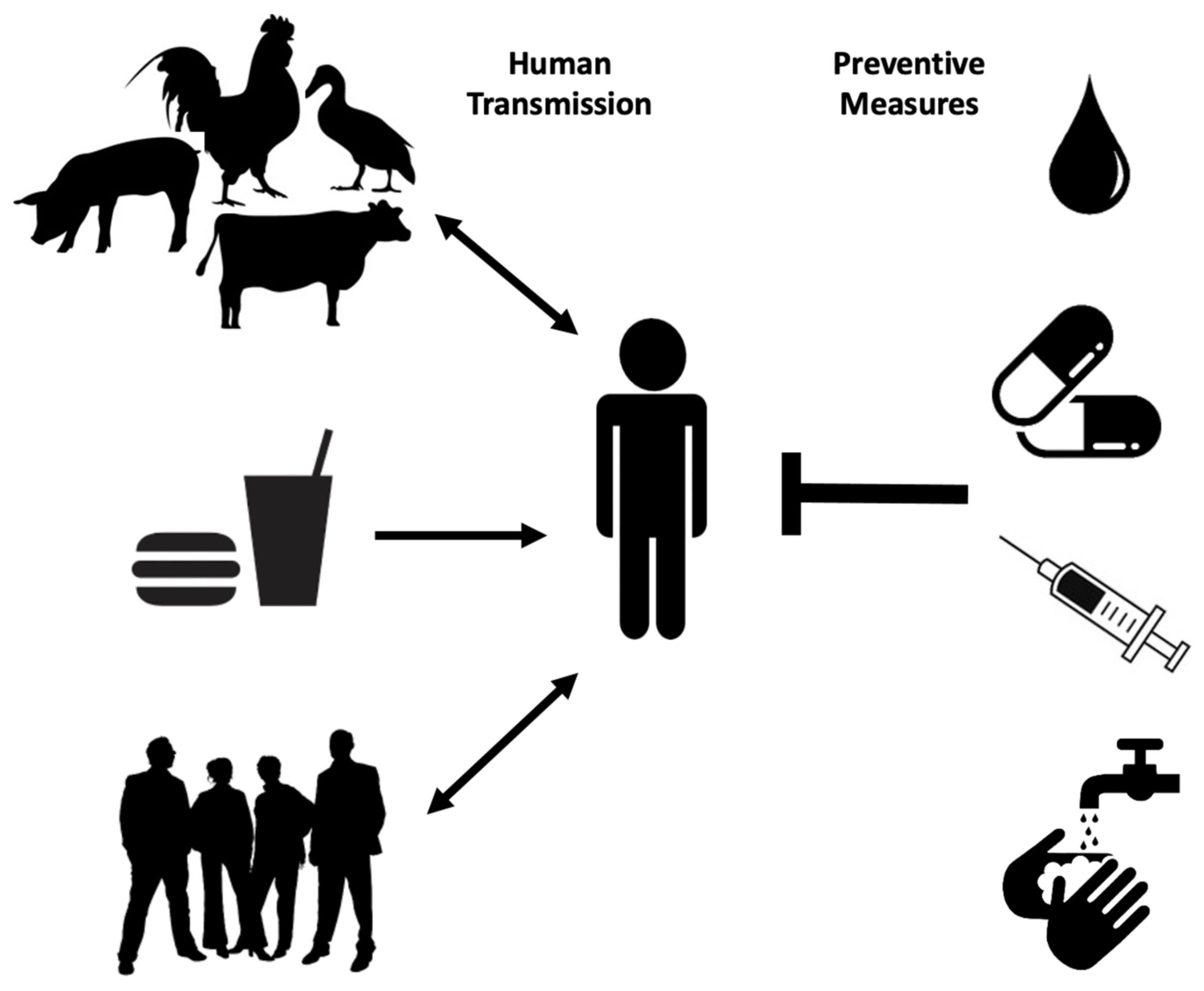
1. Salmonellosis
2. Capsular Polysaccharides
2.1. Capsular Polysaccharides—Classification
| Characteristic | Group 1 | Group 2 | Group 3 | Group 4 |
|---|---|---|---|---|
| Thermostability | Yes | No | No | Yes |
| Thermoregulated (expressed below 20 °C) | No | Yes | No | No |
| Co-expressed with colanic acid | No | Yes | Yes | Yes |
| Co-expressed with O serogroups | Limited | Many | Many | Mainly O8, O9. Can be none |
| Former K antigen group | 1A | II | I/II or III | IB (O-Antigen capsules) |
| Capsule assembly | Periplasmic face of the plasma membrane | Cytoplasmic face of the plasma membrane | Cytoplasmic face of the plasma membrane | Periplasmic face of the plasma membrane |
| Export system | Wzx pathway | ABC transporter | ABC transporter | Wzx pathway |
| Translocation proteins | Wza, Wzc | KpsD, KpsE (may be KpsF) | KpsD (may be KpsE) | Unknown |
| Polymerization system | Wzy dependent | Processive | Processive | Wzy dependent |
| Direction of polymer chain growth | Reducing terminus | Non-reducing terminus | Non-reducing terminus (hypothetical) | Reducing terminus |
| Terminal lipid moiety | Lipid A core in KLPS; unknown for capsular K antigen | α-Glycerophosphate | α-Glycerophosphate (hypothetical) | Lipid A core in KLPS; unknown for capsular K antigen |
| Elevated levels of CMP-Kdo synthetase | No | Yes | No | No |
| Positively regulated by the Rcs system | Yes | No | No | No |
2.2. Capsular Polysaccharides—Function
2.3. Capsular Polysaccharides—Regulation
2.4. Capsular Polysaccharides—Salmonella and Other Selected Bacterial Species
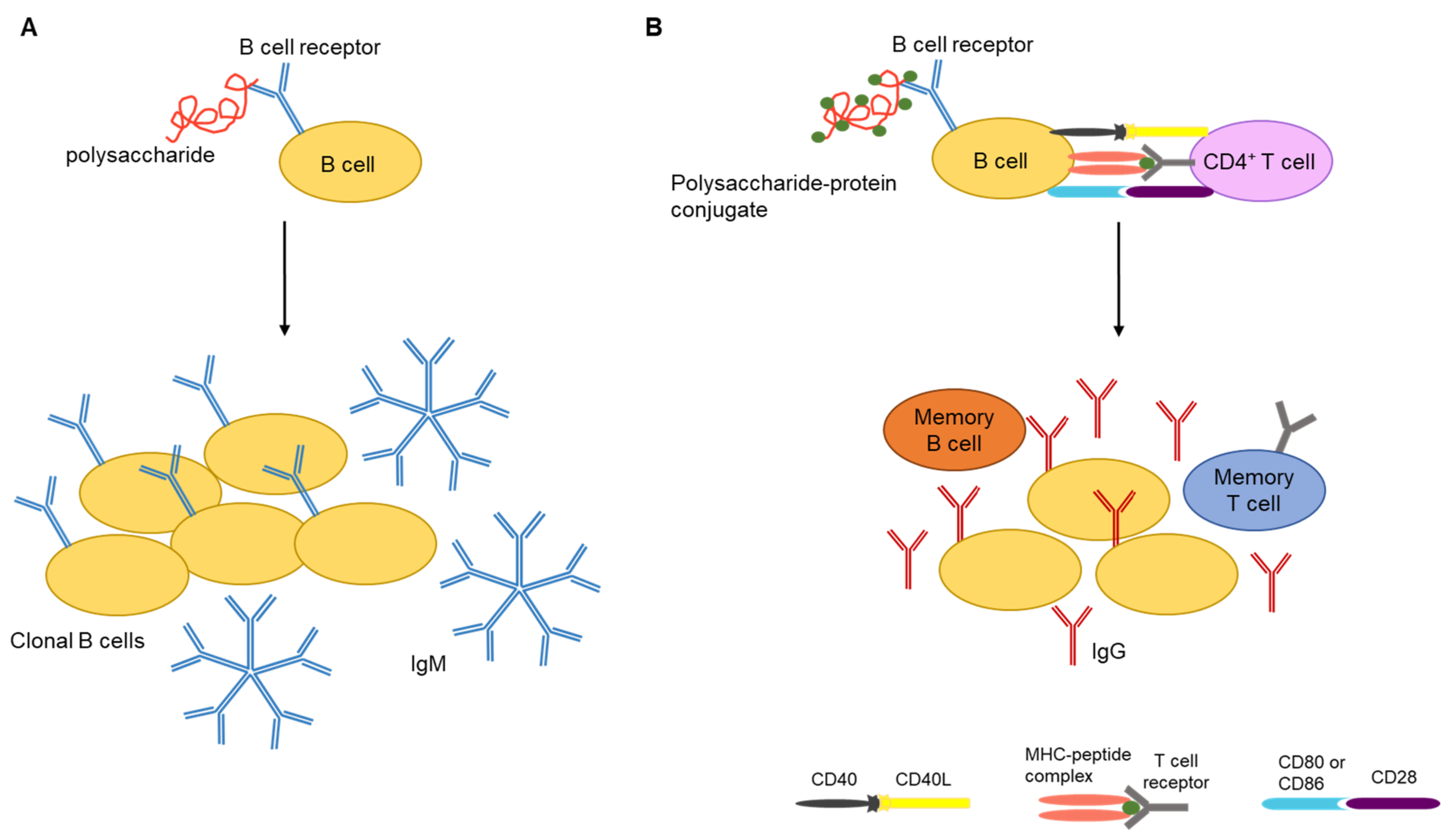
3. Polysaccharide Vaccines
3.1. Polysaccharide Vaccines in Use
3.1.1. Neisseria meningitidis
3.1.2. Streptococcus pneumonia
3.1.3. Haemophilus influenzae
3.1.4. Shigella
3.1.5. Staphylococcus aureus and Vibrio cholera
3.1.6. Typhoidal Salmonella Vaccines
4. Non-Typhoidal Salmonella Vaccines
4.1. Glycoconjugate Vaccines for NTS
4.2. Live-Attenuated Vaccines for NTS
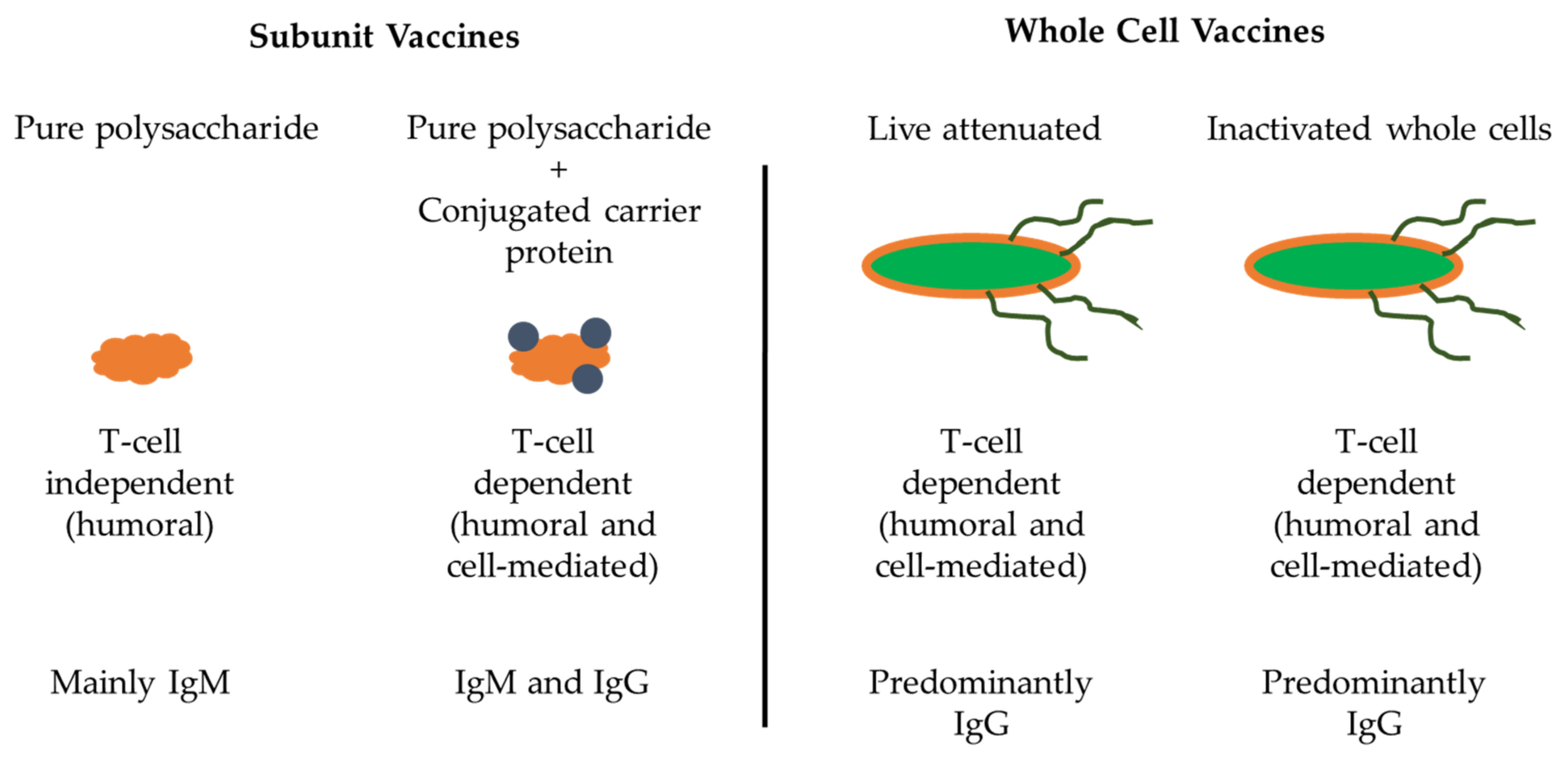
4.3. Subunit Vaccines
4.4. Other Vaccines Currently Being Developed for NTS
5. Looking Ahead
Author Contributions
Funding
Conflicts of Interest
References
- Hurley, D.; McCusker, M.P.; Fanning, S.; Martins, M. Salmonella-host interactions—Modulation of the host innate immune system. Front. Immunol. 2014, 5, 481. [Google Scholar] [CrossRef] [PubMed]
- WHO. Product Development for Vaccines Advisory Committee (PD-VAC) Meeting. 2015. Available online: http://www.who.int/immunization/research/meetings_workshops/pdvac/en/ (accessed on April 2021).
- Pathogen Safety Data Sheet: Infectious Substances—Salmonella Enterica Spp. Available online: http://www.phac-aspc.gc.ca/lab-bio/res/psds-ftss/salmonella-ent-eng.php (accessed on April 2021).
- FoodNet Canada Annual Report 2016. Available online: https://www.canada.ca/en/public-health/services/surveillance/foodnet-canada/publications/foodnet-canada-annual-report-2016.html (accessed on April 2021).
- De Jong, H.K.; Parry, C.M.; van der Poll, T.; Wiersinga, W.J. Host-pathogen interaction in invasive salmonellosis. PLoS Pathog. 2012, 8, e1002933. [Google Scholar] [CrossRef]
- Salmonella. Available online: https://www.cdc.gov/salmonella/index.html (accessed on April 2021).
- Salmonella Outbreak Investigations: Timeline for Reporting Cases. Available online: http://www.cdc.gov/salmonella/reportingtimeline.html (accessed on April 2021).
- Antibiotic Resistance Threats in the United States. Available online: https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&ved=2ahUKEwjiqq6U0sDwAhVH_54KHbTICiUQFjAOegQIEhAD&url=https%3A%2F%2Fwww.cdc.gov%2Fdrugresistance%2Fpdf%2Fthreats-report%2F2019-ar-threats-report-508.pdf&usg=AOvVaw1709sEktvf-ovR38riUreg (accessed on April 2021).
- Brisabois, A.; Cazin, I.; Breuil, J.; Collatz, E. Surveillance of antibiotic resistance in Salmonella. Eurosurveillance 1997, 2, 181. [Google Scholar] [CrossRef] [PubMed]
- Salmonella Heidelberg Ceftiofur-Related Resistance in Human and Retail Chicken Isolates. Available online: http://www.phac-aspc.gc.ca/cipars-picra/heidelberg/heidelberg-eng.php (accessed on April 2021).
- Upreti, R.K.; Kumar, M.; Shankar, V. Bacterial glycoproteins: Functions, biosynthesis and applications. Proteomics 2003, 3, 363–379. [Google Scholar] [CrossRef]
- Roberts, I.S. The biochemistry and genetics of capsular polysaccharide production in Bacteria. Annu. Rev. Microbiol. 1996, 50, 285–315. [Google Scholar] [CrossRef]
- Troy, F.A.; Frerman, F.E.; Heath, E.C. The biosynthesis of capsular polysaccharide in aerobacter aerogenes. J. Biol. Chem. 1971, 246, 118–133. [Google Scholar] [CrossRef]
- De Rezende, C.E.; Anriany, Y.; Carr, L.E.; Joseph, S.W.; Weiner, R.M. Capsular polysaccharide surrounds smooth and rugose types of Salmonella enterica serovar Typhimurium DT104. Appl. Environ. Microb. 2005, 71, 7345–7351. [Google Scholar] [CrossRef] [PubMed]
- Costerton, J.W.; Irvin, R.T.; Cheng, K.-J. The bacterial glycocalyx in nature and disease. Annu. Rev. Microbiol. 1981, 35, 299–324. [Google Scholar] [CrossRef]
- Mazmanian, S.K.; Kasper, D.L. The love-hate relationship between bacterial polysaccharides and the host immune system. Nat. Rev. Immunol. 2006, 6, 849–858. [Google Scholar] [CrossRef]
- Owens, T.W.; Taylor, R.J.; Pahil, K.S.; Bertani, B.R.; Ruiz, N.; Kruse, A.C.; Kahne, D. Structrual basis of unidirectional export of lipopolysaccharide to the cell surface. Nature 2019, 567, 550–553. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Orlando, B.J.; Liao, M. Structural basis of lipopolysaccharide extraction by the LptB2FGC complex. Nature 2019, 567, 486–490. [Google Scholar] [CrossRef]
- Whitfield, C. Biosynthesis and assembly of capsular polysaccharides in escherichia coli. Annu. Rev. Biochem. 2006, 75, 39–68. [Google Scholar] [CrossRef]
- Whitfield, C.; Amor, P.A.; Koplin, R. Modulation of the surface architecture of gram-negative bacteria by the action of surface polymer: Lipid a-core ligase and by determinants of polymer chain length. Mol. Microbiol. 1997, 23, 629–638. [Google Scholar] [CrossRef] [PubMed]
- Sukupolvi-Petty, S.; Grass, S.; StGeme, J.W., III. The haemophilus influenzae type B hcsA and hcsB gene products facilitate transport of capsular polysaccharide across the outer membrane and are essential for virulence. J. Bacteriol. 2006, 188, 3870–3877. [Google Scholar] [CrossRef]
- Weiss, D.S.; Brotcke, A.; Henry, T.; Margolis, J.J.; Chan, K.; Monack, D.M. In vivo negative selection screen identifies genes required for francisella virulence. Proc. Natl. Acad. Sci. USA 2007, 104, 6037–6042. [Google Scholar] [CrossRef] [PubMed]
- Gibson, D.L.; White, A.P.; Snyder, S.D.; Martin, S.; Heiss, C.; Azadi, P.; Surette, M.; Kay, W.W. Salmonella produces an o-antigen capsule regulated by agfd and important for environmental persistence. J. Bacteriol. 2006, 188, 7722–7730. [Google Scholar] [CrossRef]
- DeShazer, D.; Waag, D.M.; Fritz, D.; Woods, D.E. Identification of a Burkholderia mallei polysaccharide gene cluster by subtractive hybridization and demonstration that the encoded capsule is an essential virulence determinant. Microb. Pathog. 2001, 30, 253–269. [Google Scholar] [CrossRef] [PubMed]
- Laxalt, K.A.; Kozel, T.R. Chemotaxigenesis and activation of the alternative complement pathway by encapsulated and non-encapsulated cryptococcus neoformans. Infect. Immun. 1979, 26, 435–440. [Google Scholar] [CrossRef]
- Karlyshev, A.V.; Wren, B.W. Detection and initial characterization of novel capsular polysaccharide among diverse campylobacter jejuni strains using alcian blue dye. J. Clin. Microbiol. 2001, 39, 279–284. [Google Scholar] [CrossRef]
- Nelson, A.L.; Roche, A.M.; Gould, J.M.; Chim, K.; Ratner, A.J.; Weiser, J.N. Capsule enhances pneumococcal colonization by limiting mucus-mediated clearance. Infect. Immun. 2007, 75, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Fournier, J.-M.; Vann, W.F.; Karakawa, W.W. Purification and characterization of staphylococcus aureus type 8 capsular polysaccharide. Infect. Immun. 1984, 45, 87–93. [Google Scholar] [CrossRef]
- Orskov, F.; Orskov, I. Escherichia coli serotyping and disease in man and animals. Can. J. Microbiol. 1992, 38, 699–704. [Google Scholar] [CrossRef] [PubMed]
- Vann, W.F.; Soderstrom, T.; Egan, W.; Tsui, F.-P.; Schneerson, R.; Orskov, I.; Orskov, F. Serological, chemical, and structural analyses of the escherichia coli cross-reactive capsular polysaccharides K13, K20, and K23. Infect. Immun. 1983, 39, 623–629. [Google Scholar] [CrossRef]
- Goldman, R.C.; White, D.; Orskov, F.; Orskov, I.; Rick, P.D.; Lewis, M.S.; Bhattacharjee, A.K.; Leive, L. A surface polysaccharide of Escherichia coli O111 contains o-antigen and inhibits agglutination of cells by O-Antiserum. J. Bacteriol. 1982, 151, 1210–1221. [Google Scholar] [CrossRef]
- Amor, P.A.; Whitfield, C. Molecular and functional analysis of genes required for expression of group 1B K antigens in Escherichia coli: Characterization of the his-region containing gene clusters for multiple cell-surface polysaccharides. Mol. Microbiol. 1997, 26, 145–161. [Google Scholar] [CrossRef] [PubMed]
- Whitfield, C.; Roberts, I.S. Structure, assembly and regulation of expression of capsules in Escherichia coli. Mol. Microbiol. 1999, 31, 1307–1319. [Google Scholar] [CrossRef] [PubMed]
- Ophir, T.; Gutnick, D.L. A role for exopolysaccharides in the protection of microorganisms from desiccation. Appl. Environ. Microb. 1994, 60, 740–745. [Google Scholar] [CrossRef]
- Costerton, J.W.; Cheng, K.-J.; Geesey, G.G.; Ladd, T.I.; Nickel, J.C.; Dasgupta, M.; Marrie, T.J. Bacterial biofilms in nature and disease. Annu. Rev. Microbiol. 1987, 41, 435–464. [Google Scholar] [CrossRef]
- Ledeboer, N.A.; Jones, B.D. Exopolysaccharide sugars contribute to biofilm formation by Salmonella enterica serovar typhimurium on Hep-2 cells and chicken intestinal epithelium. J. Bacteriol. 2005, 187, 3214–3226. [Google Scholar] [CrossRef] [PubMed]
- Sood, R.K.; Fattom, A. Capsular polysaccharide-protein conjugate vaccines and intravenous immunoglobulins. Expert Opin. Investig. Drugs. 1998, 7, 333–347. [Google Scholar] [CrossRef] [PubMed]
- Pickard, D.; Li, J.; Roberts, M.; Maskell, D.; Hone, D.; Levine, M.; Dougan, G.; Chatfield, S. Characterization of defined ompR mutants of salmonella typhi: OmpR is involved in the regulation of vi polysaccharide expression. Infect. Immun. 1994, 62, 3984–3993. [Google Scholar] [CrossRef]
- Berry, A.; DeVault, J.D.; Chakrabarty, A.M. High osmolarity is a signal for enhanced algD transcription in mucoid and nonmucoid pseudomonas aeruginosa strains. J. Bacteriol. 1989, 171, 2312–2317. [Google Scholar] [CrossRef] [PubMed]
- Grados, O.; Ewing, W.H. Antigenic relationship between escherichia coli and neisseria meningitidis. J. Infect. Dis. 1970, 122, 100–103. [Google Scholar] [CrossRef] [PubMed]
- Liston, S.D.; Ovchinnikova, O.G.; Whitfield, C. Unique lipid anchor attaches vi antigen capsule to the surface of Salmonella enterica serovar Typhi. Proc. Natl. Acad. Sci. USA 2016, 113, 6719–6724. [Google Scholar] [CrossRef] [PubMed]
- Wangdi, T.; Lee, C.-Y.; Spees, A.M.; Yu, C.; Kingsbury, D.D.; Winter, S.E.; Hastey, C.J.; Wilson, R.P.; Heinrich, V.; Baumler, A.J. The vi capsular polysaccharide enables salmonella enterica serovar typhi to evade microbe-guided neutrophil chemotaxis. PLoS Pathog. 2014, 10, e1004306. [Google Scholar] [CrossRef] [PubMed]
- Wilson, R.P.; Winter, S.E.; Spees, A.M.; Winter, M.G.; Nishimori, J.H.; Sanchez, J.F.; Nuccio, S.-P.; Crawford, R.W.; Tukel, C.; Baumler, A.J. The vi capsular polysaccharide prevents complement receptor 3-mediated clearance of Salmonella enterica serotype typhi. Infect. Immun. 2011, 79, 830–837. [Google Scholar] [CrossRef] [PubMed]
- Raffatellu, M.; Chessa, D.; Wilson, R.P.; Tukel, C.; Akcelik, M.; Baumler, A.J. Capsule-mediated immune evasion: A new hypothesis explaining aspects of typhoid fever pathogenesis. Infect. Immun. 2006, 74, 19–27. [Google Scholar] [CrossRef]
- Hirose, K.; Ezaki, T.; Miyake, M.; Li, T.; Khan, A.Q.; Kawamura, Y.; Yokoyama, H.; Takami, T. Survival of vi-capsulated and vi-deleted salmonella typhi strains in cultured macrophage expressing different levels of CD14 antigen. FEMS Microbiol. Lett. 1997, 147, 259–265. [Google Scholar] [CrossRef]
- Quintero, E.; Weiner, R.M. Evidence for the adhesive function of the exopolysaccharide of hyphomonas strain MHS-3 in its attachment to surfaces. Appl. Environ. Microb. 1995, 61, 1897–1903. [Google Scholar] [CrossRef] [PubMed]
- Sokaribo, A.S.; Perera, S.R.; Sereggela, Z.; Krochak, R.; Balezantis, L.R.; Xing, X.; Lam, S.; Deck, W.; Attah-Poku, S.; Abbott, D.W.; et al. A GMMA-CPS-based vaccine for non-typhoidal salmonella. Vaccines 2021, 9, 165. [Google Scholar] [CrossRef]
- Kasper, D.L.; Onderdonk, A.B.; Crabb, J.; Bartlett, J.G. Protective efficacy of immunization with capsular antigen against experimental infection with bacteroides fragilis. J. Infect. Dis. 1979, 140, 724–731. [Google Scholar] [CrossRef] [PubMed]
- Kenzel, S.; Henneke, P. The innate immune system and its relevance to neonatal sepsis. Curr. Opin. Infect. Dis. 2006, 19, 264–270. [Google Scholar] [CrossRef]
- Wilder-Smith, A. Meningococcal vaccines: A neglected topic in travel medicine? Expert Rev. Vaccines 2009, 8, 1343–1350. [Google Scholar] [CrossRef]
- State Exemption Levels Low, National Vaccination Rates High. Available online: https://www.cdc.gov/media/releases/2015/p0827-vaccination-rates.html (accessed on April 2021).
- Pollard, A.J.; Perrett, K.P.; Beverley, P.C. Maintaining protection against invasive bacteria with protein-polysaccharide conjugate vaccines. Nat. Rev. Immunol. 2009, 9, 213–220. [Google Scholar] [CrossRef] [PubMed]
- MacLennan, C.A.; Martin, L.B.; Micoli, F. Vaccines against invasive salmonella disease. Hum. Vaccines Immunother. 2014, 10, 1478–1493. [Google Scholar] [CrossRef] [PubMed]
- Lesinski, G.B.; Westerink, M.A.J. Novel vaccine strategies to t-independent antigens. J. Microbiol. Methods. 2001, 47, 135–149. [Google Scholar] [CrossRef]
- Blanchard-Rohner, G.; Pollard, A. Long-term protection after immunization with protein-polysaccharide conjugate vaccines in infancy. Expert Rev. Vaccines 2011, 10, 673–684. [Google Scholar] [CrossRef]
- Kruetzmann, S.; Rosado, M.M.; Weber, H.; Germing, U.; Tournilhac, O.; Peter, H.-H.; Berner, R.; Peters, A.; Boehm, T.; Plebani, A.; et al. Human immunoglobulin M memory B cells controlling streptococcus pneumoniae infections are generated in the spleen. J. Exp. Med. 2003, 197, 939–945. [Google Scholar] [CrossRef] [PubMed]
- Kruschinski, C.; Zidan, M.; Debertin, A.S.; Von Horsten, S.; Pabst, R. Age-dependent development of the splenic marginal zone in human infants is associated with different causes of death. Hum. Pathol. 2004, 35, 113–121. [Google Scholar] [CrossRef]
- Jokhdar, H.; Borrow, R.; Sultan, A.; Adi, M.; Riley, C.; Fuller, E.; Baxter, D. Immunologic hyporesponsiveness to serogroup c but not serogroup a following repeated meningococcal a/c polysaccharide vaccination in Saudi Arabia. Clin. Diagn. Lab. Immun. 2004, 11, 83–88. [Google Scholar] [CrossRef]
- Kalka-Moll, W.M.; Tzianabos, A.O.; Bryant, P.W.; Niemeyer, M.; Ploegh, H.L.; Kasper, D.L. Zwitterionic polysaccharides stimulate T cells by MHC class II-dependent interactions. J. Immunol. 2002, 169, 6149–6153. [Google Scholar] [CrossRef]
- Makela, P.H.; Kayhty, H. Evolution of conjugate vaccines. Expert Rev. Vaccines 2002, 1, 399–410. [Google Scholar] [CrossRef] [PubMed]
- Ramsay, M.E.; McVernon, J.; Andrews, N.J.; Heath, P.T.; Slack, M.P. Estimating haemophilus influenzae type b vaccine effectiveness in England and wales by use of the screening method. J. Infect. Dis. 2003, 188, 481–485. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Heath, P.T.; McVernon, J. The UK hib vaccine experience. Arch. Dis. Child. 2002, 86, 396–399. [Google Scholar] [CrossRef] [PubMed]
- Ramsay, M.E.; Andrews, N.J.; Trotter, C.L.; Kaczmarski, E.B.; Miller, E. Herd immunity form meningococcal serogroup C conjugate vaccination in England: Database analysis. BMJ 2003, 326, 365–366. [Google Scholar] [CrossRef]
- Borrow, R.; Andrews, N.; Findlow, H.; Waight, P.; Southern, J.; Crowley-Luke, A.; Stapley, L.; England, A.; Findlow, J.; Miller, E. Kinetics of antibody persistence following administration of a combination meningococcal serogroup C and haemophilus influenzae type b conjugate vaccine in healthy infants in the United Kingdom primed with a monovalent meningococcal serogroup C vaccine. Clin. Vaccine Immunol. 2010, 17, 154–159. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Snape, M.D.; Kelly, D.F.; Salt, P.; Green, S.; Snowden, C.; Diggle, L.; Borkowski, A.; Yu, L.-M.; Moxon, R.; Pollard, A.J. Serogroup C meningococcal glycoconjugate vaccine in adolescents: Persistence of bactericidal antibodies and kinetics of the immune reponse to a booster vaccine more than 3 years after immunization. Clin. Infect. Dis. 2006, 43, 1387–1394. [Google Scholar] [CrossRef] [PubMed]
- Snape, M.D.; Kelley, D.F.; Lewis, S.; Banner, C.; Kibwana, L.; Moore, C.E.; Diggle, L.; John, T.; Yu, L.-M.; Borrow, R.; et al. Seroprotection against serogroup C meningococcal disease in adolescents in the United Kingdom: Observational study. BMJ 2008, 336, 1487–1491. [Google Scholar] [CrossRef] [PubMed]
- Auckland, C.; Gray, S.; Borrow, R.; Andrews, N.; Goldblatt, D.; Ramsay, M.; Miller, E. Clinical and immunologic risk factors for meningococcal c conjugate vaccine failure in the United Kingdom. J. Infect. Dis. 2006, 194, 1745–1752. [Google Scholar] [CrossRef] [PubMed]
- McVernon, J.; Johnson, P.D.R.; Pollard, A.J.; Slack, M.P.E.; Moxon, E.R. Immunologic memory in haemophilus influenzae type b conjugate vaccine failure. Arch. Dis. Child. 2003, 88, 379–383. [Google Scholar] [CrossRef] [PubMed]
- Blanchard-Rohner, G.; Snape, M.D.; Kelly, D.F.; John, T.; Morant, A.; Yu, L.-M.; Borkowski, A.; Ceddia, F.; Borrow, R.; Siegrist, C.-A.; et al. The magnitude of the antibody and memory b cell responses during priming with a protein-polysaccharide conjugate vaccine in human infants is associated with the persistence of antibody and the intensity of booster response. J. Immunol. 2008, 180, 2165–2173. [Google Scholar] [CrossRef]
- Plotkin, S.A. Vaccines, vaccination, and vaccinology. J. Infect. Dis. 2003, 187, 1349–1359. [Google Scholar] [CrossRef] [PubMed]
- Nair, M. Protein conjugate polysaccharide vaccines: Challenges in development and global implementation. Indian J. Community Med. 2012, 37, 79–82. [Google Scholar] [CrossRef] [PubMed]
- Richmond, P.; Kaczmarski, E.; Borrow, R.; Findlow, J.; Clark, S.; McCann, R.; Hill, J.; Barker, M.; Miller, E. Meningococcal C polysaccharide vaccine induces immunologic hyporesponsiveness in adults that is overcome by meningococcal C conjugate vaccine. J. Infect. Dis. 2000, 181, 761–764. [Google Scholar] [CrossRef] [PubMed]
- Snape, M.D.; Pollard, A.J. Meningococcal polysaccharide-protein conjugate vaccines. Lancet Infect. Dis. 2005, 5, 21–30. [Google Scholar] [CrossRef]
- Trotter, C.L.; Andrews, N.J.; Kaczmarski, E.B.; Miller, E.; Ramsay, M.E. Effectiveness of meningococcal serogroup C conjugate vaccine 4 years after introduction. Lancet 2004, 364, 365–367. [Google Scholar] [CrossRef]
- Shapiro, E.D.; Berg, A.T.; Austrian, R.; Schroeder, D.; Parcells, V.; Margolis, A.; Adair, R.K.; Clemens, J.D. the protective efficacy of polyvalent pneumococcal polysaccharide vaccine. N. Engl. J. Med. 1991, 325, 1453–1460. [Google Scholar] [CrossRef]
- Halloran, M.E.; Haber, M.; Longini, I.M., Jr.; Struchiner, C.J. Direct and indirect effects in vaccine efficacy and effectiveness. Am. J. Epidemiol. 1991, 133, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Robbins, J.B.; Chu, C.; Schneerson, R. Hypothesis for vaccine development: Protective immunity to enteric diseases caused by nontyphoidal salmonella and shigellae may be conferred by serum igG antibodies to the O-specific polysaccharide of their lipopolysaccharides. Clin. Infect. Dis. 1992, 15, 346–361. [Google Scholar] [CrossRef]
- Rodrigues, L.P.; Schneerson, R.; Robbins, J.B. Immunity to hemophilus influenzae type b. i. The isolation, and some physicochemical, serologic and biologic properties of the capsular polysaccharide of hemophilus influenzae type b. J. Immunol. 1971, 107, 1071–1080. [Google Scholar] [PubMed]
- Smith, D.H.; Peter, G.; Ingram, D.L.; Harding, A.L.; Anderson, P. Responses of children immunized with the capsular polysaccharide of hemophilus influenzae type B. Pediatrics 1973, 52, 637–644. [Google Scholar]
- Dagan, R.; Melamed, R.; Muallem, M.; Piglansky, L.; Greenberg, D.; Abramson, O.; Mendelman, P.M.; Bohidar, N.; Yagupsky, P. Reduction of nasopharyngeal carriage of pneumococci during the second year of life by a heptavalent conjugate pneumococcal vaccine. J. Infect. Dis. 1996, 174, 1271–1278. [Google Scholar] [CrossRef]
- Parke, J.C.; Schneerson, R.; Relmer, C.; Black, C.; Welfare, S.; Bryla, D.; Levi, L.; Pavliakova, D.; Cramton, T.; Schulz, D.; et al. Clinical and immunologic reponses to haemophilus influenzae type b-tetanus toxoid conjugate vaccine in infants injected at 3,5,7 and 18 months of age. J. Pediatr. 1991, 118, 184–190. [Google Scholar] [CrossRef]
- Claesson, B.A.; Trolfors, B.; Lagergard, T.; Taranger, J.; Bryla, D.; Otterman, G.; Cramton, T.; Yang, Y.; Reimer, C.B.; Robbins, J.B.; et al. Clinical and immunologic responses to capsular polysaccharide of haemophilus influenzae type b alone or conjugated to tetanus toxoid in 18- to 23-month-old children. J. Pediatr. 1988, 112, 695–702. [Google Scholar] [CrossRef]
- Adams, W.G.; Deaver, K.A.; Cochi, S.L.; Plikaytis, B.D.; Zell, E.R.; Broome, C.V.; Wenger, J.D. Decline of childhood haemophilus influenzae type b (Hib) disease in the Hib vaccine era. JAMA—J. Am. Med. Assoc. 1993, 269, 221–226. [Google Scholar] [CrossRef]
- Passwell, J.H.; Harlev, E.; Ashkenazi, S.; Chu, C.; Miron, D.; Ramon, R.; Farzan, N.; Shiloach, J.; Bryla, D.A.; Majadly, F.; et al. Safety and immunogenicity of improved shigella o-specific polysaccharide-protein conjugate vaccines in adults in Israel. Infect. Immun. 2001, 69, 1351–1357. [Google Scholar] [CrossRef]
- Ferreccio, C.; Prado, V.; Ojeda, A.; Cayyazo, M.; Abrego, P.; Guers, L.; Levine, M.M. Epidemiologic patterns of acute diarrhea and endemic shigella infections in children in a poor periurban setting in Santiago, Chile. Am. J. Epidemiol. 1991, 134, 614–627. [Google Scholar] [CrossRef] [PubMed]
- Cohen, D.; Orr, N.; Robin, G.; Slepon, R.; Ashkenazi, S.; Ashkenazi, I.; Shemer, J. Detection of antibodies to shigella lipopolysaccharide in urine after natural shigella infection or vaccination. Clin. Diagn. Lab. Immun. 1996, 3, 451–455. [Google Scholar] [CrossRef] [PubMed]
- Cohen, D.; Ashkenazi, S.; Green, M.S.; Gdalevich, M.; Robin, G.; Slepon, R.; Yavzori, M.; Orr, N.; Block, C.; Asshkenazi, I.; et al. Double-blinded vaccine-controlled randomised efficacy trial of an investigational shigella sonnei conjugate vaccine in young adults. Lancet 1997, 349, 155–159. [Google Scholar] [CrossRef]
- Fattom, A.I.; Sarwar, J.; Basham, L.; Ennifar, S.; Naso, R. Antigenic determinants of staphylococcus aureus type 5 and type 8 capsular polysaccharide vaccines. Infect. Immun. 1998, 66, 4588–4592. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.K.; Taylor, D.N.; Bryla, D.A.; Robbins, J.B.; Szu, S.C. Phase 1 evaluation of vibrio cholera O1, Serotype Inaba, polysaccharide-cholera toxin conjugates in adult volunteers. Infect. Immun. 1998, 66, 3095–3099. [Google Scholar] [CrossRef] [PubMed]
- Mastroeni, P.; Villarreal-Ramos, B.; Hormaeche, C.E. Adoptive transfer of immunity to oral challenge with virulent salmonellae in innately susceptible BALB/c mice requires both immune serum and T cells. Infect. Immun. 1993, 61, 3981–3984. [Google Scholar] [CrossRef] [PubMed]
- Salerno-Goncalves, R.; Pasetti, M.F.; Sztein, M.B. Characterization of CD8+ effector T cell responses in volunteers immunized with salmonella enterica serovar typhi strain Ty21a typhoid vaccine. J. Immunol. 2002, 169, 2196–2203. [Google Scholar] [CrossRef]
- Mastroeni, P.; Grant, A.; Restif, O.; Maskell, D. A dynamic view of the spread and intracellular distribution of Salmonella enterica. Nat. Rev. Microbiol. 2009, 7, 73–80. [Google Scholar] [CrossRef]
- Blanden, R.V.; Mackaness, G.B.; Collins, F.M. Mechanisms of acquired resistance in mouse typhoid. J. Exp. Med. 1966, 124, 585–600. [Google Scholar] [CrossRef]
- Mackaness, G.B.; Blanden, R.V.; Collins, F.M. Host-parasite relations in mouse typhoid. J. Exp. Med. 1966, 124, 573–583. [Google Scholar] [CrossRef] [PubMed]
- Sinha, K.; Mastroeni, P.; Harrison, J.; De Hormaeche, R.D.; Hormaeche, C.E. Salmonella typhimurium aroA, htrA, and aroD htrA mutants cause progressive infections in athymic (nu/nu) BALB/c mice. Infect. Immun. 1997, 65, 1566–1569. [Google Scholar] [CrossRef]
- McSorley, S.J.; Jenkins, M.K. Antibody is required for protection against virulent but not attenuated salmonella enterica serovar typhimurium. Infect. Immun. 2000, 68, 3344–3348. [Google Scholar] [CrossRef] [PubMed]
- Hawley, P.R.; Simmons, J.S. The effectiveness of vaccines used for the prevention of typhoid fever in the United States Army and Navy. Am. J. Public Health 1934, 24, 689–709. [Google Scholar] [CrossRef] [PubMed]
- Engels, E.A.; Falagas, M.E.; Lau, J.; Bennish, M.L. Typhoid fever vaccines: A meta-analysis of studies on efficacy and toxicity. BMJ 1998, 316, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Ivanoff, B.; Levine, M.M.; Lambert, P.H. Vaccination against typhoid fever: Present status. Bull. World Health Organ. 1994, 72, 957–971. [Google Scholar] [PubMed]
- Wahdan, M.H.; Sippel, J.E.; Mikhail, I.A.; Rahka, A.E.; Anderson, E.S.; Sparks, H.A.; Cvjetanovic, B. Controlled field trial of a typhoid vaccine prepared with a nonmotile mutant of salmonella typhi Ty2. Bull. World Health Organ. 1975, 52, 69–73. [Google Scholar]
- Nath, T.R.; Malaviya, A.N.; Kumar, R.; Balakrishnan, K.; Singh, B.P. A study of the efficacy of typhoid vaccine in inducing humoral and cell-mediated immune responses in human volunteers. Clin. Exp. Immunol. 1977, 30, 38–43. [Google Scholar] [PubMed]
- Rajagopalan, P.; Kumar, R.; Malaviya, A.N. a study of humoral and cell-mediated immune response following typhoid vaccination in human volunteers. Clin. Exp. Immunol. 1982, 47, 275–282. [Google Scholar] [PubMed]
- Germanier, R.; Furer, E. Isolation and characterization of Gal E mutant Ty 21a of Salmonella typhi: A candidate strain for a live, oral typhoid vaccine. J. Infect. Dis. 1975, 131, 553–558. [Google Scholar] [CrossRef]
- Fraser, A.; Paul, M.; Goldberg, E.; Acosta, C.J.; Leibovici, L. Typhoid fever vaccines: Systematic review and meta-analysis of randomised controlled trials. Vaccine 2007, 25, 7848–7857. [Google Scholar] [CrossRef] [PubMed]
- Ohtake, S.; Martin, R.; Saxena, A.; Pham, B.; Chiueh, G.; Osorio, M.; Kopecko, D.; Xu, D.Q.; Lechuga-Ballesteros, D.; Truong-Le, V. Room temperature stabilization of oral, live attenuated salmonella enterica serovar typhi-vectored vaccines. Vaccine 2011, 29, 2761–2771. [Google Scholar] [CrossRef] [PubMed]
- Cryz, S.J., Jr.; Vanprapar, N.; Thisyakorn, U.; Olanratmanee, T.; Losonsky, G.; Levine, M.M.; Chearskul, S. Safety and immunogenicity of Salmonella typhi Ty21a vaccine in young Thai children. Infect. Immun. 1993, 61, 1149–1151. [Google Scholar] [CrossRef] [PubMed]
- Pakkanen, S.H.; Kantele, J.M.; Kantele, A. Cross-reactive gut-directed immune response against salmonella enterica serovar paratyphi A and B in typhoid fever and after oral Ty21 a typhoid vaccination. Vaccine 2012, 30, 6047–6053. [Google Scholar] [CrossRef] [PubMed]
- Wahid, R.; Simon, R.; Zafar, S.J.; Levine, M.M.; Sztein, M.B. Live oral typhoid vaccine Ty21a induces cross-reactive humoral immune responses against Salmonella enterica serovar paratyphi A and S. Paratyphi B in humans. Clin. Vaccine Immunol. 2012, 19, 825–834. [Google Scholar] [CrossRef] [PubMed]
- Pakkanen, S.H.; Kantele, J.M.; Herzog, C.; Kantele, A. Cross-reactive immune response elicited by parenteral vi polysaccharide typhoid vaccine against non-typhoid Salmonellae. Vaccine 2014, 32, 544–551. [Google Scholar] [CrossRef] [PubMed]
- Kantele, A.; Pakkanen, S.H.; Siitonen, A.; Karttunen, R.; Kantele, J.M. Live oral typhoid salmonella typhi Ty21a—A surrogate vaccine against non-typhoid Salmonella? Vaccine 2010, 30, 7238–7245. [Google Scholar] [CrossRef] [PubMed]
- Tacket, C.O.; Pasetti, M.F.; Sztein, M.B.; Livio, S.; Levine, M.M. Immune responses to an oral typhoid vaccine strain that is modified to constitutively express vi capsular polysaccharide. J. Infect. Dis. 2004, 190, 565–570. [Google Scholar] [CrossRef] [PubMed]
- Lin, F.Y.C.; Ho, V.A.; Khiem, H.B.; Trach, D.D.; Bay, P.V.; Thanh, T.C.; Kossaczka, Z.; Bryla, D.A.; Shiloach, J.; Robbins, J.B.; et al. The efficacy of a Salmonella typhi Vi conjugate vaccine in two-to-five-year-old children. N. Engl. J. Med. 2001, 344, 1263–1269. [Google Scholar] [CrossRef] [PubMed]
- Thiem, V.D.; Lin, F.-Y.; Canh, D.G.; Son, N.H.; Anh, D.D.; Mao, N.D.; Chu, C.; Hunt, S.W.; Robbins, J.B.; Schneerson, R.; et al. The Vi conjugate typhoid vaccine is safe, elicits protective levels of igG anti-vi, and is compatible with routine infant vaccines. Clin. Vaccine Immunol. 2011, 18, 730–735. [Google Scholar] [CrossRef] [PubMed]
- MacLennan, C.A. Vaccines for low-income countries. Semin. Immunol. 2013, 25, 114–123. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Podda, A.; Saul, A.; Arora, R.; Bhutta, Z.; Sinha, A.; Gaind, R.; Singhal, T.; Saha, S.; Brooks, A.; Martin, L.B.; et al. Conjugated vaccines for enteric fever: Proceedings of a meeting organized in New Delphi, India in 2009. J. Infect. Dev. Ctries. 2010, 4, 404–411. [Google Scholar] [CrossRef]
- MacLennan, C.A.; Gondwe, E.N.; Msefula, C.L.; Kingsley, R.A.; Thomson, N.R.; White, S.A.; Goodall, M.; Pickard, D.J.; Graham, S.M.; Dougan, G.; et al. The neglected role of antibody in protection against bacteremia caused by nontyphoidal strains of Salmonella in African children. J. Clin. Investig. 2008, 118, 1553–1562. [Google Scholar] [CrossRef] [PubMed]
- Feasey, N.A.; Archer, B.N.; Heyderman, R.S.; Sooka, A.; Dennis, B.; Gordon, M.A.; Keddy, K.H. Typhoid fever and invasive nontyphoid Salmonellosis, Malawi and South Africa. Emerg. Infect. Dis. 2010, 16, 1448–1451. [Google Scholar] [CrossRef] [PubMed]
- Gondwe, E.N.; Molyneux, M.E.; Goodall, M.; Graham, S.M.; Mastroeni, P.; Drayson, M.T.; MacLennan, C.A. Importance of antibody and complement for oxidative burst and killing of invasive nontyphoidal Salmonella by blood cells in Africans. Proc. Natl. Acad. Sci. USA 2010, 107, 3070–3075. [Google Scholar] [CrossRef]
- Simon, R.; Tennant, S.M.; Wang, J.Y.; Schmidlein, P.J.; Lees, A.; Ernst, R.K.; Pasetti, M.F.; Galen, J.E.; Levine, M.M. Salmonella enterica serovar enteritidis core O polysaccharide conjugated to H:g,m flagellin as a candidate vaccine for protection against invasive infection with S. enteritidis. Infect. Immun. 2011, 79, 4240–4249. [Google Scholar] [CrossRef] [PubMed]
- Watson, D.C.; Robbins, J.B.; Szu, S.C. Protection of Mice against Salmonella typhimurium with an O-specific polysaccharide-protein conjugate vaccine. Infect. Immun. 1992, 60, 4679–4686. [Google Scholar] [CrossRef] [PubMed]
- Carlin, N.I.A.; Svenson, S.B.; Lindberg, A.A. Role of monoclonal O-antigen antibody epitope specificity and isotype in protection against experimental mouse typhoid. Microb. Pathog. 1987, 2, 171–183. [Google Scholar] [CrossRef]
- Singh, S.P.; Williams, Y.U.; Benjamin, W.H.; Klebba, P.E.; Boyd, D. Immunoprotection by monoclonal antibodies to the porins and lipopolysaccharide of Salmonella typhimurium. Microb. Pathog. 1996, 21, 249–263. [Google Scholar] [CrossRef]
- Rondini, S.; Lanzilao, L.; Necchi, F.; O’Shaughnessy, C.M.; Micoli, F.; Saul, A.; MacLennan, C.A. Invasive African salmonella typhimurium induces bactericidal antibodies against O-antigens. Microb. Pathog. 2013, 63, 19–23. [Google Scholar] [CrossRef] [PubMed]
- Gordon, M.A.; Banda, H.T.; Gondwe, M.; Gordon, S.B.; Boeree, M.J.; Walsh, A.L.; Corkill, J.E.; Hart, A.; Gilks, C.F.; Molyneux, M.E. Non-typhoidal Salmonella bacteraemia among HIV-infected Malawian adults: High mortality and frequent recrudescence. AIDS 2002, 16, 1633–1641. [Google Scholar] [CrossRef] [PubMed]
- Lazarus, G.M.; Neu, H.C. Agents responsible for infection in chronic granulomatous disease of childhood. J. Pediatr. 1975, 86, 415–417. [Google Scholar] [CrossRef]
- Svenson, S.B.; Lindberg, A.A. Artificial Salmonella vaccines: Salmonella typhimurium O-antigen-specific oligosaccharide-protein conjugates elicit protective antibodies in rabbits and mice. Infect. Immun. 1981, 32, 490–496. [Google Scholar] [CrossRef]
- Simon, R.; Levine, M.M. Glycoconjugate vaccine strategies for protection against invasive Salmonella infections. Hum. Vaccines Immunother. 2012, 8, 494–498. [Google Scholar] [CrossRef]
- Svenson, S.B.; Nurminen, M.; Lindberg, A.A. Artificial Salmonella vaccines: O-antigenic oligosaccharide-protein conjugates induce protection against infection with Salmonella typhimurium. Infect. Immun. 1979, 25, 863–872. [Google Scholar] [CrossRef] [PubMed]
- Eom, J.S.; Kim, J.S.; Jang, J.I.; Kim, B.-H.; Yoo, S.Y.; Choi, J.H.; Bang, L.-S.; Lee, I.S.; Park, Y.K. Enhancement of host immune responses by oral vaccination to Salmonella enterica serovar typhimurium harboring both FliC and FljB flagella. PLoS ONE 2013, 8, e74850. [Google Scholar] [CrossRef] [PubMed]
- Simon, R.; Wang, J.Y.; Boyd, M.A.; Tulapurkar, M.E.; Ramachandran, G.; Tennant, S.M.; Pasetti, M.; Galen, J.E.; Levine, M.M. Sustained protection in mice immunized with fractional doses of Salmonella enteritidis core and O polysaccharide-flagellin glycoconjugates. PLoS ONE 2013, 8, e64680. [Google Scholar] [CrossRef] [PubMed]
- Bobat, S.; Flores-Langarica, A.; Hitchcock, J.; Marshall, J.L.; Kingsley, R.A.; Goodall, M.; Gil-Cruz, C.; Serre, K.; Leyton, D.L.; Letran, S.E.; et al. Soluble flagellin, FliC, induces an Ag-specific Th2 response, yet promotes T-bet-regulated Th1 clearance of Salmonella typhimurium infection. J. Immunol. 2011, 41, 1606–1618. [Google Scholar] [CrossRef]
- Cunningham, A.F.; Khan, M.; Ball, J.; Toellner, K.-M.; Serre, K.; Mohr, E.; MacLennan, I.C.M. Responses to the soluble flagellar protein FliC are Th2, while those to FliC on Salmonella are Th2. J. Immunol. 2004, 34, 2986–2995. [Google Scholar]
- Kang, H.Y.; Srinivasan, J.; Curtiss, R., III. Immune responses to recombinant pneumococal PspA antigen delivered by live attenuated Salmonella enterica serovar typhimurium vaccine. Infect. Immun. 2002, 70, 1739–1749. [Google Scholar] [CrossRef] [PubMed]
- Su, H.; Liu, Q.; Bian, X.; Wang, S.; Curtiss, R., 3rd; Kong, Q. Synthesis and delivery of streptococcus pneumoniae capsular polysaccharides by recombinant attenuated Salmonella vaccines. Proc. Natl. Acad. Sci. USA 2021, 118, e2013350118. [Google Scholar] [CrossRef]
- Juarez-Rodriguez, M.D.; Arteaga-Cortes, L.T.; Kader, R.; Curtiss, R., 3rd. Live attenuated Salmonella vaccines against mycobacterium tuberculosis with antigen delivery via the type III secretion system. Infect. Immun. 2012, 80, 798–814. [Google Scholar] [CrossRef][Green Version]
- Li, Y.; Wang, S.; Scarpellini, G.; Gunn, B.; Xin, W.; Wanda, S.-Y.; Roland, K.L.; Curtis, R., 3rd. Evaluation of new generation of Salmonella enterica serovar typhimurium vaccines with regulated delayed attenuation to induce immune responses against PspA. Proc. Natl. Acad. Sci. USA 2009, 106, 593–598. [Google Scholar] [CrossRef]
- Wang, S.; Kong, Q.; Curtis, R., 3rd. New technologies in developing recombinant attenuated Salmonella vaccine vectors. Microb. Pathog. 2013, 58, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Li, Y.; Scarpellini, G.; Kong, W.; Shi, H.Y.; Baek, C.-H.; Gunn, B.; Wanda, S.-Y.; Roland, K.L.; Zhang, X.; et al. Salmonella vaccine vectors displaying delayed antigen synthesis in vivo to enhance immunogenicity. Infect. Immun. 2010, 78, 3969–3980. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.Y.; Curtis, R., 3rd. Immune responses dependent on antigen location in recombinant attenuated Salmonella typhimurium vaccines following oral immunization. FEMS Immunol. Med. Mic. 2003, 37, 99–104. [Google Scholar] [CrossRef]
- Gil-Cruz, C.; Bobat, S.; Marshall, J.L.; Kingsley, R.A.; Ross, E.A.; Henderson, I.R.; Leyton, D.L.; Coughlan, R.E.; Khan, M.; Jensen, K.T.; et al. The porin ompd from nontyphoidal salmonella is a key target for a protective B1b cell antibody response. Proc. Natl. Acad. Sci. USA 2009, 106, 9803–9808. [Google Scholar] [CrossRef]
- Secundino, I.; Lopez-Macias, C.; Cervantes-Barragan, L.; Gil-Cruz, C.; Rios-Sarabia, N.; Pastelin-Palacios, R.; Villasis-Keever, M.A.; Becker, I.; Puente, J.L.; Calva, E.; et al. Salmonella porins induce a sustained, lifelong specific bactericidal antibody memory response. Immunology 2005, 117, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Salazar-Gonzalez, R.M.; Maldonado-Bernal, C.; Ramirez-Cruz, N.E.; Rios-Sarabia, N.; Beltran-Nava, J.; Castanon-Gonzalez, J.; Castillo-Torres, N.; Palma-Aguirre, J.A.; Carrera-Camargo, M.; Lopez-Macias, C.; et al. Induction of cellular immune response and anti-Salmoneall enterica serovar typhi bactericidal antibodies in healthy volunteers by immunization with a vaccine candidate against typhoid fever. Immunol. Lett. 2004, 93, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Sette, A.; Rappuoli, R. Reverse vaccinology: Developing vaccines in the era of genomics. Immunity 2010, 33, 530–541. [Google Scholar] [CrossRef]
- Fiorino, F.; Pettini, E.; Koeberling, O.; Ciabattini, A.; Pozzi, G.; Martin, L.B.; Medaglini, D. Long-term anti-bacterial immunity against systemic infection by salmonella enterica serovar typhimurium elicited by a gmma-based vaccine. Vaccines 2021, 9, 495. [Google Scholar] [CrossRef] [PubMed]
- Micoli, F.; Bjarnarson, S.P.; Arcuri, M.; Pindi, A.A.A.; Magnusdottir, G.J.; Necchi, F.; Benedetto, R.D.; Carducci, M.; Schiavo, F.; Giannelli, C.; et al. Short vi-polysaccharide abroages t-independent immune response and hyporesponsiveness elicited by long Vi-CRM197 conjugate vaccine. Proc. Natl. Acad. Sci. USA 2020, 117, 24443–24449. [Google Scholar] [CrossRef]
- Micoli, F.; Alfini, R.; Di Benedetto, R.; Necchi, F.; Schiavo, F.; Mancini, F.; Carducci, M.; Palmieri, E.; Balocchi, C.; Gasperini, G.; et al. GMMA is a versatile platform to design effective multivalent combination vaccines. Vaccines 2020, 8, 540. [Google Scholar] [CrossRef]
| Description | Developer | Academic/Commercial | Phase |
|---|---|---|---|
| Vaccines Licensed for S. Typhi | |||
| Neotyf, Typhoral, Vivotif (Ty21a live attenuated) | Johnson & Johnson | Commercial | Marketed |
| Bio Typh (Vi capsular polysaccharide) | BioMed | Commercial | Marketed |
| Typhim Vi, Typhyvax (Vi capsular polysaccharide) | Sanofi (Pasteur Merieux) | Commercial | Marketed |
| Typherix (Vi capsular polysaccharide) | GlaxoSmithKline | Commercial | Marketed |
| Peda-Typh (conjugate) | BioMed | Commercial | Marketed |
| Typbar-TCV (conjugate) | Bharat Biotech | Commercial | Marketed |
| Typhoid vaccine | Microgen | Commercial | Marketed |
| Vi Polysaccharide typhoid vaccine | China National Pharmaceutical (Beijing Tiantan Biological) | Commercial | Marketed |
| Vi-rEPA (Vi conjugate) | Lanzhou Institute (China) | Commercial | Marketed |
| Tyvax VI plus | VHB Life Sciences | Commercial | Marketed |
| Typhoid vaccine | Zydus Cadila (Zydus Vaccicare) | Commercial | Marketed |
| Tyrix Vi | SK Holdings | Commercial | Marketed |
| Shantyph | Sanofi (Shantha Biotechnics) | Commercial | Marketed |
| Typhobox, Typhovax | Green Cross | Commercial | Marketed |
| Zerotyph | Boryung | Commercial | Marketed |
| vax-TyVi | Finlay Institute | Commercial | Marketed |
| Typhoid-Kovax | Sanofi | Commercial | Marketed |
| Hepatyrix | GlaxoSmithKline | Commercial | Marketed |
| VIVAXIM | Sanofi | Commercial | Marketed |
| Biovac Typhoid | Wockhardt | Commercial | Marketed |
| Typho-Vi | Bio-Med | Commercial | Marketed |
| Vaccines in Development in Recent Years for S. Typhi | |||
| Vi-DT typhoid conjugate vaccine | Bio Farma | Commercial | Phase I |
| Typhetec | Prokarium | Commercial | Phase I |
| OmpC and OmpF | Institute Mexicano del Seguro Social | Academic | Phase 1 in Mexico |
| Enteric fever vaccine | Prokarium | Commercial | Phase II |
| M01ZH09 (live attenuated) | Emergent BioSolutions | Commercial | Phase II |
| CVD 909 (live attenuated) | University of Maryland | Academic | Phase II |
| Ty800 (live attenuated) | Avant Immunotherapeutics | Commercial | Phase II |
| Vi-rEPA (Vi conjugate) | National Health Institute | Academic | Phase III |
| Vi-CRM (Vi conjugate) | Biological E | Commercial | Phase III |
| Vi conjugated to fusion protein PsaA-PdT | Harvard Medical School | Academic | Preclinical |
| O:9-DT (conjugate) | International Vaccine Institute (IVI) | Academic | Preclinical |
| Salmonella typhi + paratyphi vaccine | Prokarium | Commercial | Research/Preclinical |
| Salmonella Vaccine Project | Affinivax | Commercial | Research/Preclinical |
| Vi conjugated to fusion protein PsaA-PdT | Harvard Medical School | Academic | Research/Preclinical |
| Ty21a typhoid vaccine expressing Shigella LPS | Protein Potential LLC. | Commercial | Research/Preclinical |
| O:2,12-DT + Vi-DT | International Vaccine Institute (IVI) | Academic | Research/Preclinical |
| O:2,12-CRM197 + Vi-CRM197 | Biological E | Commercial | Research/Preclinical |
| Vaccines in Development in Recent Years for S. Paratyphi | |||
| CVD 1902 (live attenuated) | University of Maryland, Bharat Biotech | Commercial | Phase I |
| Paratyphoid A conjugate vaccine | Lanzhou Institute (China) | Academic | Phase II |
| O:2-TT (conjugate) | Technology transfer from NIH to Lanzhou Institute (China) | Commercial | Phase II |
| O:2,12-DT + Vi-DT | International Vaccine Institute (IVI) | Academic | Research/Preclinical |
| O:2,12-CRM197 + Vi-CRM197 | Biological E | Commercial | Research/Preclinical |
| O:2-TT (conjugate) | Technology transfer from NIH to Chengdu Institute (China) | Academic | Preclinical |
| O:2-TT (conjugate) | Changchun Institute of Biological Products | Commercial | Preclinical |
| NTS Vaccines in Development in Recent Years | |||
| WT05—aroC and ssaV mutants (live attenuated) | Microscience, Wokingham Berkshire | Academic | Phase 1 |
| NTS Vaccine | University of Maryland | Academic | Research/Preclinical |
| CVD 1921 and CVD 1941 NTS Project (live attenuated) | University of Maryland, Bharat Biotec, Wellcome Trust | Commercial | Research/Preclinical |
| Bivalent iNTS-GMMA | GlaxoSmithKline (Novartis Vaccine Institute for Global Health; NVGH) | Commercial | Research/Preclinical |
| Bivalent Conjugate (O:1,4[5],12-CRM197 + O:1,9,12-CRM197) | GlaxoSmithLine (Novartis Vaccine Institute for Global Health; NVGH NVGH) | Commercial | Research/Preclinical |
| OmpD | University of Birmingham | Academic | Research/Preclinical |
| O:4,5/O:9-flagellin (conjugate) | University of Maryland | Academic | Preclinical |
| O:4, 12-TT (conjugate) | National Institutes for Health NIH) | Academic | Preclinical |
| Os-po (O:4 porin conjugate) | National Bacteriology Laboratory, Stockholm | Preclinical | |
| S. Typhimurium ruvB mutant (live attenuated) | Seoul National University | Academic | Preclinical |
| Salmonella hfq deletion mutant (live attenuated) | Indian Institute of Science Bangalore | Academic | Preclinical |
| SA186 (live attenuated) | Istituto Superiore di Sanita Roma | Academic | Preclinical |
| MT13 (live attenuated) | KIIT University Odisha | Academic | Preclinical |
| DNA adenine methylase mutants (live attenuated) | University of California, Santa Barbara | Academic | Preclinical |
| live attenuated, regulated delayed attenuation | Arizona State University | Academic | Preclinical |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perera, S.R.; Sokaribo, A.S.; White, A.P. Polysaccharide Vaccines: A Perspective on Non-Typhoidal Salmonella. Polysaccharides 2021, 2, 691-714. https://doi.org/10.3390/polysaccharides2030042
Perera SR, Sokaribo AS, White AP. Polysaccharide Vaccines: A Perspective on Non-Typhoidal Salmonella. Polysaccharides. 2021; 2(3):691-714. https://doi.org/10.3390/polysaccharides2030042
Chicago/Turabian StylePerera, Sumudu R., Akosiererem S. Sokaribo, and Aaron P. White. 2021. "Polysaccharide Vaccines: A Perspective on Non-Typhoidal Salmonella" Polysaccharides 2, no. 3: 691-714. https://doi.org/10.3390/polysaccharides2030042
APA StylePerera, S. R., Sokaribo, A. S., & White, A. P. (2021). Polysaccharide Vaccines: A Perspective on Non-Typhoidal Salmonella. Polysaccharides, 2(3), 691-714. https://doi.org/10.3390/polysaccharides2030042






