Gut Microbiota and Obesity: The Chicken or the Egg?
Abstract
:1. Introduction
2. Materials and Methods
3. Results
4. Gut Microbiota-Derived Nutrients and Nutrient Absorption
4.1. Carbohydrates
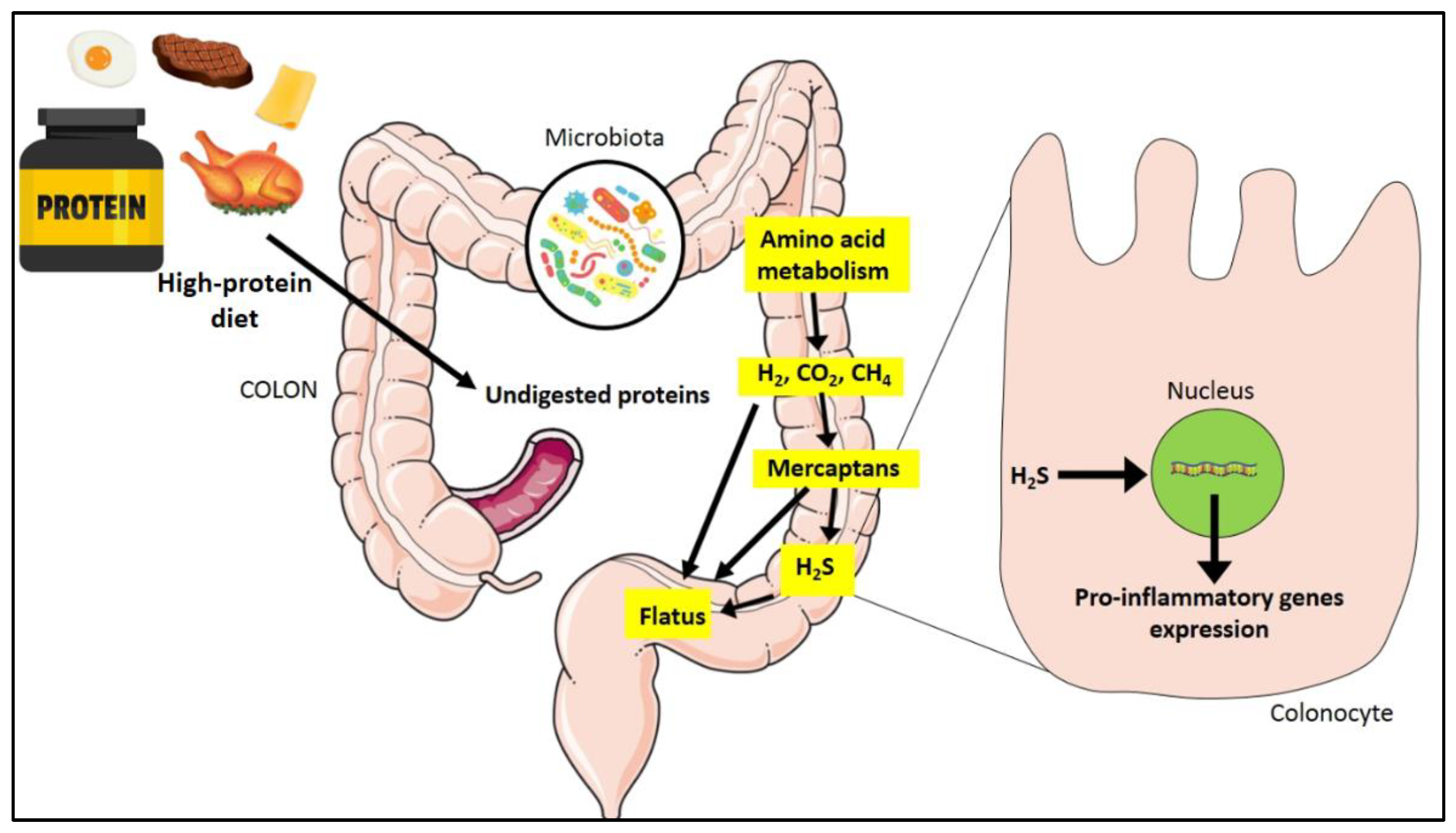
4.2. Protein
4.3. Lipids and SCFAs
4.4. Vitamins
4.5. Phytochemicals
5. Gut Microbiota-Derived Metabolites and Cardiovascular Disorders
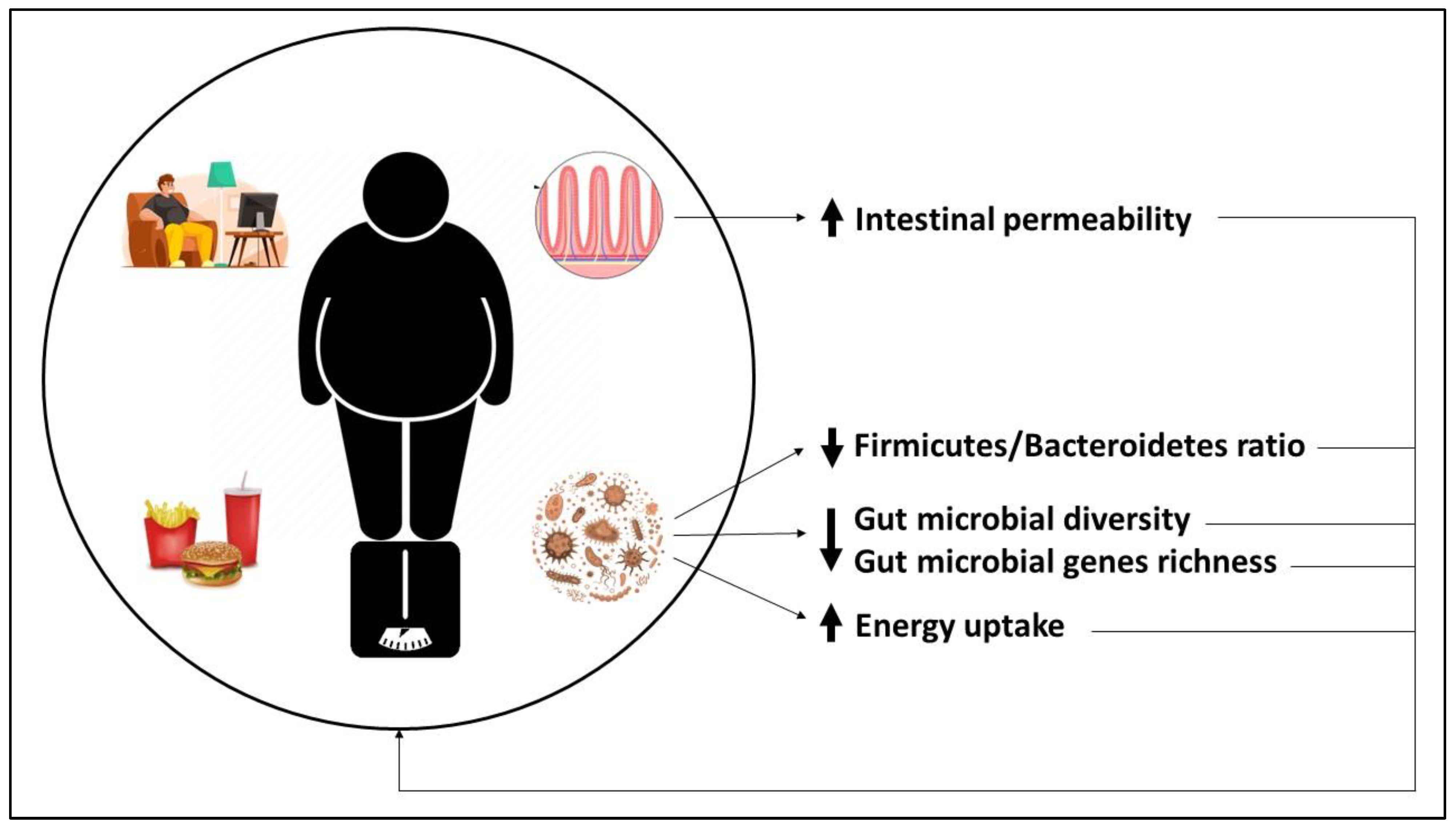
6. Gut Microbiota Composition in Obesity
7. Unhealthy Dietary Patterns
8. Healthy Dietary Patterns
9. Probiotics and Synbiotics in Weight Loss
10. Exercise
11. Microbiota Transplantation
12. Conclusions and Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Upadhyay, J.; Farr, O.; Perakakis, N.; Ghaly, W.; Mantzoros, C. Obesity as a Disease. Med. Clin. N. Am. 2018, 102, 13–33. [Google Scholar] [CrossRef]
- Fruh, S. Obesity: Risk factors, complications, and strategies for sustainable long-term weight management. J. Am. Assoc. Nurse Pract. 2019, 29, 3–14. [Google Scholar] [CrossRef]
- World Health Organization. Presents Technical Data and Information about Obesity in the World; World Health Organization: Geneva, Switzerland, 2021. [Google Scholar]
- Ward, Z.; Bleich, S.; Cradock, A.; Barrett, J.; Giles, C.; Flax, C.; Long, M.; Gortmaker, S. Projected U.S. State-Level Prevalence of Adult Obesity and Severe Obesity. N. Engl. J. Med. 2019, 381, 2440–2450. [Google Scholar] [CrossRef]
- Kelly, T.; Yang, W.; Chen, C.-S.; Reynolds, K.; He, J. Global burden of obesity in 2005 and projections to 2030. Int. J. Obes. 2008, 32, 1431–1437. [Google Scholar] [CrossRef]
- Wright, S.M.; Aronne, L.J. Causes of obesity. Abdom. Radiol. 2012, 37, 730–732. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Pedersen, O. Gut microbiota in human metabolic health and disease. Nat. Rev. Microbiol. 2021, 19, 55–71. [Google Scholar] [CrossRef] [PubMed]
- Greiner, T.; Bäckhed, F. Effects of the gut microbiota on obesity and glucose homeostasis. Trends Endocrinol. Metab. 2011, 22, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Tsai, F.; Coyle, W.J. The microbiome and obesity: Is obesity linked to our gut flora? Curr. Gastroenterol. Rep. 2009, 11, 307–313. [Google Scholar] [CrossRef]
- Cornejo-Pareja, I.; Muñoz-Garach, A.; Clemente-Postigo, M.; Tinahones, F.J. Importance of gut microbiota in obesity. Eur. J. Clin. Nutr. 2019, 72, 26–37. [Google Scholar] [CrossRef]
- Lynch, S.V.; Pedersen, O. The Human Intestinal Microbiome in Health and Disease. N. Engl. J. Med. 2016, 375, 2369–2379. [Google Scholar] [CrossRef]
- Kau, A.L.; Ahern, P.P.; Griffin, N.W.; Goodman, A.L.; Gordon, J.I. Human nutrition, the gut microbiome and the immune system. Nature 2011, 474, 327–336. [Google Scholar] [CrossRef]
- Deehan, E.C.; Yang, C.; Perez-Muñoz, M.E.; Nguyen, N.K.; Cheng, C.C.; Triador, L.; Zhang, Z.; Bakal, J.A.; Walter, J. Precision Microbiome Modulation with Discrete Dietary Fiber Structures Directs Short-Chain Fatty Acid Production. Cell Host Microbe 2020, 27, 389–404.e6. [Google Scholar] [CrossRef] [PubMed]
- Crovesy, L.; El-Bacha, T.; Rosado, E.L. Modulation of the gut microbiota by probiotics and symbiotics is associated with changes in serum metabolite profile related to a decrease in inflammation and overall benefits to metabolic health: A double-blind randomized controlled clinical trial in women with obesity. Food Funct. 2021, 12, 2161–2170. [Google Scholar] [CrossRef] [PubMed]
- Dong, T.S.; Luu, K.; Lagishetty, V.; Sedighian, F.; Woo, S.-L.; Dreskin, B.W.; Katzka, W.; Chang, C.; Zhou, Y.; Arias-Jayo, N.; et al. A High Protein Calorie Restriction Diet Alters the Gut Microbiome in Obesity. Nutrients 2020, 12, 3221. [Google Scholar] [CrossRef]
- Gøbel, R.J.; Larsen, N.; Jakobsen, M.; Mølgaard, C.; Michaelsen, K.F. Probiotics to Adolescents With Obesity: Effects on Inflammation and Metabolic Syndrome. J. Pediatr. Gastroenterol. Nutr. 2012, 55, 673–678. [Google Scholar] [CrossRef]
- Gomes, A.C.; Hoffmann, C.; Mota, J.F. Gut microbiota is associated with adiposity markers and probiotics may impact specific genera. Eur. J. Nutr. 2020, 59, 1751–1762. [Google Scholar] [CrossRef]
- Haro, C.; Rangel-Zúñiga, O.A.; Alcalá-Díaz, J.F.; Gómez-Delgado, F.; Pérez-Martínez, P.; Delgado-Lista, J.; Quintana-Navarro, G.M.; Landa, B.B.; Navas-Cortés, J.A.; Tena-Sempere, M.; et al. Intestinal Microbiota Is Influenced by Gender and Body Mass Index. PLoS ONE 2016, 11, e0154090. [Google Scholar] [CrossRef] [PubMed]
- Jian, C.; Luukkonen, P.; Sädevirta, S.; Yki-Järvinen, H.; Salonen, A. Impact of short-term overfeeding of saturated or unsaturated fat or sugars on the gut microbiota in relation to liver fat in obese and overweight adults. Clin. Nutr. 2021, 40, 207–216. [Google Scholar] [CrossRef]
- Kanazawa, A.; Aida, M.; Yoshida, Y.; Kaga, H.; Katahira, T.; Suzuki, L.; Tamaki, S.; Sato, J.; Goto, H.; Azuma, K.; et al. Effects of Synbiotic Supplementation on Chronic Inflammation and the Gut Microbiota in Obese Patients with Type 2 Diabetes Mellitus: A Randomized Controlled Study. Nutrients 2021, 13, 558. [Google Scholar] [CrossRef]
- Leber, B.; Tripolt, N.J.; Blattl, D.; Eder, M.; Wascher, T.C.; Pieber, T.R.; Stauber, R.; Sourij, H.; Oettl, K.; Stadlbauer, V. The influence of probiotic supplementation on gut permeability in patients with metabolic syndrome: An open label, randomized pilot study. Eur. J. Clin. Nutr. 2012, 66, 1110–1115. [Google Scholar] [CrossRef]
- Leong, K.S.W.; Jayasinghe, T.N.; Wilson, B.C.; Derraik, J.G.B.; Albert, B.B.; Chiavaroli, V.; Svirskis, D.M.; Beck, K.L.; Conlon, C.A.; Jiang, Y.; et al. Effects of Fecal Microbiome Transfer in Adolescents With Obesity: The Gut Bugs Randomized Controlled Trial. JAMA Netw. Open 2020, 3, e2030415. [Google Scholar] [CrossRef] [PubMed]
- Ley, R.E.; Turnbaugh, P.J.; Klein, S.; Gordon, J.I. Human gut microbes associated with obesity. Nature 2006, 444, 1022–1023. [Google Scholar] [CrossRef] [PubMed]
- Marungruang, N.; Tovar, J.; Björck, I.; Hållenius, F.F. Improvement in cardiometabolic risk markers following a multifunctional diet is associated with gut microbial taxa in healthy overweight and obese subjects. Eur. J. Nutr. 2018, 57, 2927–2936. [Google Scholar] [CrossRef] [PubMed]
- Meslier, V.; Laiola, M.; Roager, H.M.; De Filippis, F.; Roume, H.; Quinquis, B.; Giacco, R.; Mennella, I.; Ferracane, R.; Pons, N.; et al. Mediterranean diet intervention in overweight and obese subjects lowers plasma cholesterol and causes changes in the gut microbiome and metabolome independently of energy intake. Gut 2020, 69, 1258–1268. [Google Scholar] [CrossRef] [PubMed]
- Muralidharan, J.; Moreno-Indias, I.; Bulló, M.; Lopez, J.V.; Corella, D.; Castañer, O.; Vidal, J.; Atzeni, A.; Fernandez-García, J.C.; Torres-Collado, L.; et al. Effect on gut microbiota of a 1-y lifestyle intervention with Mediterranean diet compared with energy-reduced Mediterranean diet and physical activity promotion: PREDIMED-Plus Study. Am. J. Clin. Nutr. 2021, 114, 1148–1158. [Google Scholar] [CrossRef]
- Neyrinck, A.M.; Rodriguez, J.; Zhang, Z.; Seethaler, B.; Sánchez, C.R.; Roumain, M.; Hiel, S.; Bindels, L.B.; Cani, P.D.; Paquot, N.; et al. Prebiotic dietary fibre intervention improves fecal markers related to inflammation in obese patients: Results from the Food4Gut randomized placebo-controlled trial. Eur. J. Nutr. 2021, 60, 3159–3170. [Google Scholar] [CrossRef]
- Nicolucci, A.C.; Hume, M.P.; Martínez, I.; Mayengbam, S.; Walter, J.; Reimer, R.A. Prebiotics Reduce Body Fat and Alter Intestinal Microbiota in Children Who Are Overweight or With Obesity. Gastroenterology 2017, 153, 711–722. [Google Scholar] [CrossRef] [PubMed]
- Sergeev, I.N.; Aljutaily, T.; Walton, G.; Huarte, E. Effects of Synbiotic Supplement on Human Gut Microbiota, Body Composition and Weight Loss in Obesity. Nutrients 2020, 12, 222. [Google Scholar] [CrossRef]
- Van Son, J.; Serlie, M.J.; Ståhlman, M.; Bäckhed, F.; Nieuwdorp, M.; Aron-Wisnewsky, J. Plasma Imidazole Propionate Is Positively Correlated with Blood Pressure in Overweight and Obese Humans. Nutrients 2021, 13, 2706. [Google Scholar] [CrossRef]
- Vrieze, A.; Van Nood, E.; Holleman, F.; Salojärvi, J.; Kootte, R.S.; Bartelsman, J.F.W.M.; Dallinga–Thie, G.M.; Ackermans, M.T.; Serlie, M.J.; Oozeer, R.; et al. Transfer of Intestinal Microbiota From Lean Donors Increases Insulin Sensitivity in Individuals With Metabolic Syndrome. Gastroenterology 2012, 143, 913–916.e7. [Google Scholar] [CrossRef]
- Yu, E.W.; Gao, L.; Stastka, P.; Cheney, M.C.; Mahabamunuge, J.; Torres Soto, M.; Ford, C.B.; Bryant, J.A.; Henn, M.R.; Hohmann, E.L. Fecal microbiota transplantation for the improvement of metabolism in obesity: The FMT-TRIM double-blind placebo-controlled pilot trial. PLoS Med. 2020, 17, e1003051. [Google Scholar] [CrossRef]
- Bervoets, L.; Van Hoorenbeeck, K.; Kortleven, I.; Van Noten, C.; Hens, N.; Vael, C.; Goossens, H.; Desager, K.N.; Vankerckhoven, V. Differences in gut microbiota composition between obese and lean children: A cross-sectional study. Gut Pathog. 2013, 5, 10. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.Y. Lifestyle modifications result in alterations in the gut microbiota in obese children. BMC Microbiol. 2021, 21, 10. [Google Scholar] [CrossRef]
- Haro, C.; Montes-Borrego, M.; Rangel-Zúñiga, O.A.; Alcalá-Díaz, J.F.; Gómez-Delgado, F.; Pérez-Martínez, P.; Delgado-Lista, J.; Quintana-Navarro, G.M.; Tinahones, F.J.; Landa, B.B.; et al. Two Healthy Diets Modulate Gut Microbial Community Improving Insulin Sensitivity in a Human Obese Population. J. Clin. Endocrinol. Metab. 2016, 101, 233–242. [Google Scholar] [CrossRef] [PubMed]
- Jumpertz, R.; Le, D.S.; Turnbaugh, P.J.; Trinidad, C.; Bogardus, C.; Gordon, J.I.; Krakoff, J. Energy-balance studies reveal associations between gut microbes, caloric load, and nutrient absorption in humans. Am. J. Clin. Nutr. 2011, 94, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.-H.; Yun, K.E.; Kim, J.; Park, E.; Chang, Y.; Ryu, S.; Kim, H.-L.; Kim, H.-N. Gut microbiota and metabolic health among overweight and obese individuals. Sci. Rep. 2020, 10, 19417. [Google Scholar] [CrossRef] [PubMed]
- Kong, L.C.; Holmes, B.A.; Cotillard, A.; Habi-Rachedi, F.; Brazeilles, R.; Gougis, S.; Gausserès, N.; Cani, P.D.; Fellahi, S.; Bastard, J.-P.; et al. Dietary Patterns Differently Associate with Inflammation and Gut Microbiota in Overweight and Obese Subjects. PLoS ONE 2014, 9, e109434. [Google Scholar] [CrossRef] [PubMed]
- Menni, C.; Jackson, M.A.; Pallister, T.; Steves, C.J.; Spector, T.D.; Valdes, A.M. Gut microbiome diversity and high-fibre intake are related to lower long-term weight gain. Int. J. Obes. 2017, 41, 1099–1105. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Navarro, T.; Salazar, N.; Gutiérrez-Díaz, I.; De Los Reyes-Gavilán, C.; Gueimonde, M.; González, S. Different Intestinal Microbial Profile in Over-Weight and Obese Subjects Consuming a Diet with Low Content of Fiber and Antioxidants. Nutrients 2017, 9, 551. [Google Scholar] [CrossRef] [PubMed]
- Olivares, P.D.S.G.; Pacheco, A.B.F.; Aranha, L.N.; Oliveira, B.D.S.; Santos, A.A.; Santos, P.C.M.D.; Neto, J.F.N.; Rosa, G.; Oliveira, G.M.M. Gut microbiota of adults with different metabolic phenotypes. Nutrition 2021, 90, 111293. [Google Scholar] [CrossRef]
- Orsso, C.E.; Peng, Y.; Deehan, E.C.; Tan, Q.; Field, C.J.; Madsen, K.L.; Walter, J.; Prado, C.M.; Tun, H.M.; Haqq, A.M. Composition and Functions of the Gut Microbiome in Pediatric Obesity: Relationships with Markers of Insulin Resistance. Microorganisms 2021, 9, 1490. [Google Scholar] [CrossRef]
- Peters, B.A.; Shapiro, J.A.; Church, T.R.; Miller, G.; Trinh-Shevrin, C.; Yuen, E.; Friedlander, C.; Hayes, R.B.; Ahn, J. A taxonomic signature of obesity in a large study of American adults. Sci. Rep. 2018, 8, 9749. [Google Scholar] [CrossRef]
- Roland, B.C.; Lee, D.; Miller, L.S.; Vegesna, A.; Yolken, R.; Severance, E.; Prandovszky, E.; Zheng, X.E.; Mullin, G.E. Obesity increases the risk of small intestinal bacterial overgrowth (SIBO). Neurogastroenterol. Motil. 2018, 30, e13199. [Google Scholar] [CrossRef] [PubMed]
- Stefura, T.; Zapała, B.; Gosiewski, T.; Skomarovska, O.; Dudek, A.; Pędziwiatr, M.; Major, P. Differences in Compositions of Oral and Fecal Microbiota between Patients with Obesity and Controls. Medicina 2021, 57, 678. [Google Scholar] [CrossRef] [PubMed]
- Shen, N.; Caixàs, A.; Ahlers, M.; Patel, K.; Gao, Z.; Dutia, R.; Blaser, M.J.; Clemente, J.C.; Laferrère, B. Longitudinal changes of microbiome composition and microbial metabolomics after surgical weight loss in individuals with obesity. Surg. Obes. Relat. Dis. 2019, 15, 1367–1373. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, C.C.; Monteil, M.A.; Davis, E.M. Overweight and Obesity in Children Are Associated with an Abundance of Firmicutes and Reduction of Bifidobacterium in Their Gastrointestinal Microbiota. Child. Obes. 2020, 16, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Yun, Y.; Kim, H.-N.; Kim, S.E.; Heo, S.G.; Chang, Y.; Ryu, S.; Shin, H.; Kim, H.-L. Comparative analysis of gut microbiota associated with body mass index in a large Korean cohort. BMC Microbiol. 2017, 17, 151. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.; Chen, R.; McCormick, K.L.; Zhang, Y.; Lin, X.; Yang, X. The role of the gut microbiota on the metabolic status of obese children. Microb. Cell Factories 2021, 20, 53. [Google Scholar] [CrossRef]
- Zeng, Q.; Li, D.; He, Y.; Li, Y.; Yang, Z.; Zhao, X.; Liu, Y.; Wang, Y.; Sun, J.; Feng, X.; et al. Discrepant gut microbiota markers for the classification of obesity-related metabolic abnormalities. Sci. Rep. 2019, 9, 13424. [Google Scholar] [CrossRef]
- Zeng, Q.; Yang, Z.; Wang, F.; Li, D.; Liu, Y.; Wang, D.; Zhao, X.; Li, Y.; Wang, Y.; Feng, X.; et al. Association between metabolic status and gut microbiome in obese populations. Microb. Genom. 2021, 7, 000639. [Google Scholar] [CrossRef]
- Bo, T.; Wen, J.; Zhao, Y.; Tian, S.; Zhang, X.; Wang, D. Bifidobacterium pseudolongum reduces triglycerides by modulating gut microbiota in mice fed high-fat food. J. Steroid Biochem. Mol. Biol. 2020, 198, 105602. [Google Scholar] [CrossRef]
- Denou, E.; Marcinko, K.; Surette, M.G.; Steinberg, G.R.; Schertzer, J.D. High-intensity exercise training increases the diversity and metabolic capacity of the mouse distal gut microbiota during diet-induced obesity. Am. J. Physiol.-Endocrinol. Metab. 2016, 310, E982–E993. [Google Scholar] [CrossRef]
- Evans, C.C.; LePard, K.J.; Kwak, J.W.; Stancukas, M.C.; Laskowski, S.; Dougherty, J.; Moulton, L.; Glawe, A.; Wang, Y.; Leone, V.; et al. Exercise Prevents Weight Gain and Alters the Gut Microbiota in a Mouse Model of High Fat Diet-Induced Obesity. PLoS ONE 2014, 9, e92193. [Google Scholar] [CrossRef]
- Everard, A.; Lazarevic, V.; Gaïa, N.; Johansson, M.; Ståhlman, M.; Backhed, F.; Delzenne, N.M.; Schrenzel, J.; François, P.; Cani, P.D. Microbiome of prebiotic-treated mice reveals novel targets involved in host response during obesity. ISME J. 2014, 8, 2116–2130. [Google Scholar] [CrossRef]
- Fjære, E.; Myrmel, L.S.; Lützhøft, D.O.; Andersen, H.; Holm, J.B.; Kiilerich, P.; Hannisdal, R.; Liaset, B.; Kristiansen, K.; Madsen, L. Effects of exercise and dietary protein sources on adiposity and insulin sensitivity in obese mice. J. Nutr. Biochem. 2019, 66, 98–109. [Google Scholar] [CrossRef]
- Gu, Y.; Liu, C.; Zheng, N.; Jia, W.; Zhang, W.; Li, H. Metabolic and Gut Microbial Characterization of Obesity-Prone Mice under a High-Fat Diet. J. Proteome Res. 2019, 18, 1703–1714. [Google Scholar] [CrossRef] [PubMed]
- Guirro, M.; Costa, A.; Gual-Grau, A.; Herrero, P.; Torrell, H.; Canela, N.; Arola, L. Effects from diet-induced gut microbiota dysbiosis and obesity can be ameliorated by fecal microbiota transplantation: A multiomics approach. PLoS ONE 2019, 14, e0218143. [Google Scholar] [CrossRef] [PubMed]
- Hussain, A.; Kwon, M.H.; Kim, H.K.; Lee, H.S.; Cho, J.S.; Lee, Y.I. Anti-Obesity Effect of Lactobacillus plantarum LB818 Is Associated with Regulation of Gut Microbiota in High-Fat Diet-Fed Obese Mice. J. Med. Food 2020, 23, 750–759. [Google Scholar] [CrossRef] [PubMed]
- Ji, Y.; Ma, N.; Zhang, J.; Wang, H.; Tao, T.; Pei, F.; Hu, Q. Dietary intake of mixture coarse cereals prevents obesity by altering the gut microbiota in high-fat diet fed mice. Food Chem. Toxicol. 2021, 147, 111901. [Google Scholar] [CrossRef] [PubMed]
- Joung, H.; Chu, J.; Kim, B.-K.; Choi, I.-S.; Kim, W.; Park, T.-S. Probiotics ameliorate chronic low-grade inflammation and fat accumulation with gut microbiota composition change in diet-induced obese mice models. Appl. Microbiol. Biotechnol. 2021, 105, 1203–1213. [Google Scholar] [CrossRef] [PubMed]
- Ke, X.; Walker, A.; Haange, S.-B.; Lagkouvardos, I.; Liu, Y.; Schmitt-Kopplin, P.; Von Bergen, M.; Jehmlich, N.; He, X.; Clavel, T.; et al. Synbiotic-driven improvement of metabolic disturbances is associated with changes in the gut microbiome in diet-induced obese mice. Mol. Metab. 2019, 22, 96–109. [Google Scholar] [CrossRef] [PubMed]
- Kiilerich, P.; Myrmel, L.S.; Fjære, E.; Hao, Q.; Hugenholtz, F.; Sonne, S.B.; Derrien, M.; Pedersen, L.M.; Petersen, R.K.; Mortensen, A.; et al. Effect of a long-term high-protein diet on survival, obesity development, and gut microbiota in mice. Am. J. Physiol.-Endocrinol. Metab. 2016, 310, E886–E899. [Google Scholar] [CrossRef] [PubMed]
- Kübeck, R.; Bonet-Ripoll, C.; Hoffmann, C.; Walker, A.; Müller, V.M.; Schüppel, V.L.; Lagkouvardos, I.; Scholz, B.; Engel, K.-H.; Daniel, H.; et al. Dietary fat and gut microbiota interactions determine diet-induced obesity in mice. Mol. Metab. 2016, 5, 1162–1174. [Google Scholar] [CrossRef] [PubMed]
- Lai, Z.-L.; Tseng, C.-H.; Ho, H.J.; Cheung, C.K.Y.; Lin, J.-Y.; Chen, Y.-J.; Cheng, F.-C.; Hsu, Y.-C.; Lin, J.-T.; El-Omar, E.M.; et al. Fecal microbiota transplantation confers beneficial metabolic effects of diet and exercise on diet-induced obese mice. Sci. Rep. 2018, 8, 15625. [Google Scholar] [CrossRef]
- Li, S.; Yingyi, G.; Chen, L.; Lijuan, G.; Ou, S.; Peng, X. Lean rats gained more body weight from a high-fructooligosaccharide diet. Food Funct. 2015, 6, 2315–2321. [Google Scholar] [CrossRef]
- Lu, Y.; Fan, C.; Li, P.; Lu, Y.; Chang, X.; Qi, K. Short Chain Fatty Acids Prevent High-fat-diet-induced Obesity in Mice by Regulating G Protein-coupled Receptors and Gut Microbiota. Sci. Rep. 2016, 6, 37589. [Google Scholar] [CrossRef]
- Moreira Júnior, R.E.; De Carvalho, L.M.; Dos Reis, D.C.; Cassali, G.D.; Faria, A.M.C.; Maioli, T.U.; Brunialti-Godard, A.L. Diet-induced obesity leads to alterations in behavior and gut microbiota composition in mice. J. Nutr. Biochem. 2021, 92, 108622. [Google Scholar] [CrossRef]
- Moretti, C.H.; Schiffer, T.A.; Li, X.; Weitzberg, E.; Carlström, M.; Lundberg, J.O. Germ-free mice are not protected against diet-induced obesity and metabolic dysfunction. Acta Physiol. 2021, 231, e13581. [Google Scholar] [CrossRef]
- Oh, J.K.; Amoranto, M.B.C.; Oh, N.S.; Kim, S.; Lee, J.Y.; Oh, Y.N.; Shin, Y.K.; Yoon, Y.; Kang, D.-K. Synergistic effect of Lactobacillus gasseri and Cudrania tricuspidata on the modulation of body weight and gut microbiota structure in diet-induced obese mice. Appl. Microbiol. Biotechnol. 2020, 104, 6273–6285. [Google Scholar] [CrossRef]
- Ridaura, V.K.; Faith, J.J.; Rey, F.E.; Cheng, J.; Duncan, A.E.; Kau, A.L.; Griffin, N.W.; Lombard, V.; Henrissat, B.; Bain, J.R.; et al. Gut Microbiota from Twins Discordant for Obesity Modulate Metabolism in Mice. Science 2013, 341, 1241214. [Google Scholar] [CrossRef]
- Saiyasit, N.; Chunchai, T.; Prus, D.; Suparan, K.; Pittayapong, P.; Apaijai, N.; Pratchayasakul, W.; Sripetchwandee, J.; Chattipakorn, N.; Chattipakorn, S.C. Gut dysbiosis develops before metabolic disturbance and cognitive decline in high-fat diet–induced obese condition. Nutrition 2020, 69, 110576. [Google Scholar] [CrossRef]
- Shang, Y.; Khafipour, E.; Derakhshani, H.; Sarna, L.K.; Woo, C.W.; Siow, Y.L.; O, K. Short Term High Fat Diet Induces Obesity-Enhancing Changes in Mouse Gut Microbiota That are Partially Reversed by Cessation of the High Fat Diet. Lipids 2017, 52, 499–511. [Google Scholar] [CrossRef] [PubMed]
- Turnbaugh, P.J.; Ley, R.E.; Mahowald, M.A.; Magrini, V.; Mardis, E.R.; Gordon, J.I. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 2006, 444, 1027–1031. [Google Scholar] [CrossRef] [PubMed]
- Turnbaugh, P.J.; Ridaura, V.K.; Faith, J.J.; Rey, F.E.; Knight, R.; Gordon, J.I. The Effect of Diet on the Human Gut Microbiome: A Metagenomic Analysis in Humanized Gnotobiotic Mice. Sci. Transl. Med. 2009, 1, 6ra14. [Google Scholar] [CrossRef] [PubMed]
- Welly, R.J.; Liu, T.-W.; Zidon, T.M.; Rowles, J.L.; Park, Y.-M.; Smith, T.N.; Swanson, K.S.; Padilla, J.; Vieira-Potter, V.J. Comparison of Diet versus Exercise on Metabolic Function and Gut Microbiota in Obese Rats. Med. Sci. Sports Exerc. 2016, 48, 1688–1698. [Google Scholar] [CrossRef]
- Rowland, I.; Gibson, G.; Heinken, A.; Scott, K.; Swann, J.; Thiele, I.; Tuohy, K. Gut microbiota functions: Metabolism of nutrients and other food components. Eur. J. Nutr. 2018, 57, 1–24. [Google Scholar] [CrossRef]
- Musso, G.; Gambino, R.; Cassader, M. Interactions Between Gut Microbiota and Host Metabolism Predisposing to Obesity and Diabetes. Annu. Rev. Med. 2011, 62, 361–380. [Google Scholar] [CrossRef]
- Den Besten, G.; Van Eunen, K.; Groen, A.K.; Venema, K.; Reijngoud, D.-J.; Bakker, B.M. The role of short-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism. J. Lipid Res. 2013, 54, 2325–2340. [Google Scholar] [CrossRef]
- Devaraj, S.; Hemarajata, P.; Versalovic, J. The Human Gut Microbiome and Body Metabolism: Implications for Obesity and Diabetes. Clin. Chem. 2013, 59, 617–628. [Google Scholar] [CrossRef]
- Hamer, H.M.; Jonkers, D.; Venema, K.; Vanhoutvin, S.; Troost, F.J.; Brummer, R.-J. Review article: The role of butyrate on colonic function: REVIEW: ROLE OF BUTYRATE ON COLONIC FUNCTION. Aliment. Pharmacol. Ther. 2007, 27, 104–119. [Google Scholar] [CrossRef]
- Cummings, J.; Hill, M.; Bone, E.; Branch, W.; Jenkins, D.J.A. The effect of meat protein and dietary fiber on colonic function and metabolism II. Bacterial metabolites in feces and urine. Am. J. Clin. Nutr. 1979, 32, 2094–2101. [Google Scholar] [CrossRef]
- Nicholson, J.K.; Holmes, E.; Kinross, J.; Burcelin, R.; Gibson, G.; Jia, W.; Pettersson, S. Host-Gut Microbiota Metabolic Interactions. Science 2012, 336, 1262–1267. [Google Scholar] [CrossRef] [PubMed]
- Blachier, F.; Beaumont, M.; Portune, K.J.; Steuer, N.; Lan, A.; Audebert, M.; Khodorova, N.; Andriamihaja, M.; Airinei, G.; Benamouzig, R.; et al. High-protein diets for weight management: Interactions with the intestinal microbiota and consequences for gut health. A position paper by the my new gut study group. Clin. Nutr. 2019, 38, 1012–1022. [Google Scholar] [CrossRef] [PubMed]
- Yao, C.K.; Muir, J.G.; Gibson, P.R. Review article: Insights into colonic protein fermentation, its modulation and potential health implications. Aliment. Pharmacol. Ther. 2016, 43, 181–196. [Google Scholar] [CrossRef]
- Blachier, F.; Beaumont, M.; Andriamihaja, M.; Davila, A.-M.; Lan, A.; Grauso, M.; Armand, L.; Benamouzig, R.; Tomé, D. Changes in the Luminal Environment of the Colonic Epithelial Cells and Physiopathological Consequences. Am. J. Pathol. 2017, 187, 476–486. [Google Scholar] [CrossRef] [PubMed]
- Dallas, D.C.; Sanctuary, M.R.; Qu, Y.; Khajavi, S.H.; Van Zandt, A.E.; Dyandra, M.; Frese, S.A.; Barile, D.; German, J.B. Personalizing protein nourishment. Crit. Rev. Food Sci. Nutr. 2017, 57, 3313–3331. [Google Scholar] [CrossRef]
- Chiang, J.Y.L. Bile Acid Metabolism and Signaling. In Comprehensive Physiology, 1st ed.; Terjung, R., Ed.; Wiley: Hoboken, NJ, USA, 2013; pp. 1191–1212. ISBN 978-0-470-65071-4. [Google Scholar]
- Ridlon, J.M.; Kang, D.J.; Hylemon, P.B.; Bajaj, J.S. Bile acids and the gut microbiome. Curr. Opin. Gastroenterol. 2014, 30, 332–338. [Google Scholar] [CrossRef]
- Clinical and Research Information on Drug-Induced Liver Injury [Internet]; National Institute of Diabetes and Digestive and Kidney Diseases: Bethesda, MD, USA, 2012.
- Duszka, K. Versatile Triad Alliance: Bile Acid, Taurine and Microbiota. Cells 2022, 11, 2337. [Google Scholar] [CrossRef]
- Venema, K. Role of gut microbiota in the control of energy and carbohydrate metabolism. Curr. Opin. Clin. Nutr. Metab. Care 2010, 13, 432–438. [Google Scholar] [CrossRef]
- Van Winckel, M.; De Bruyne, R.; Van De Velde, S.; Van Biervliet, S. Vitamin K, an update for the paediatrician. Eur. J. Pediatr. 2009, 168, 127–134. [Google Scholar] [CrossRef]
- Dominguez-Bello, M.G.; Godoy-Vitorino, F.; Knight, R.; Blaser, M.J. Role of the microbiome in human development. Gut 2019, 68, 1108–1114. [Google Scholar] [CrossRef]
- Shearer, M.J. Vitamin K metabolism and nutriture. Blood Rev. 1992, 6, 92–104. [Google Scholar] [CrossRef] [PubMed]
- Pham, V.T.; Dold, S.; Rehman, A.; Bird, J.K.; Steinert, R.E. Vitamins, the gut microbiome and gastrointestinal health in humans. Nutr. Res. 2021, 95, 35–53. [Google Scholar] [CrossRef] [PubMed]
- Magnúsdóttir, S.; Ravcheev, D.; De Crécy-Lagard, V.; Thiele, I. Systematic genome assessment of B-vitamin biosynthesis suggests co-operation among gut microbes. Front. Genet. 2015, 6, 148. [Google Scholar] [CrossRef] [PubMed]
- Wan, M.L.Y.; Co, V.A.; El-Nezami, H. Dietary polyphenol impact on gut health and microbiota. Crit. Rev. Food Sci. Nutr. 2021, 61, 690–711. [Google Scholar] [CrossRef] [PubMed]
- Santos, H.O.; Genario, R.; Gomes, G.K.; Schoenfeld, B.J. Cherry intake as a dietary strategy in sport and diseases: A review of clinical applicability and mechanisms of action. Crit. Rev. Food Sci. Nutr. 2021, 61, 417–430. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [CrossRef]
- Braune, A.; Engst, W.; Blaut, M. Identification and functional expression of genes encoding flavonoid O- and C-glycosidases in intestinal bacteria: Bacterial flavonoid O- and C-glycosidase genes. Environ. Microbiol. 2016, 18, 2117–2129. [Google Scholar] [CrossRef]
- Rechner, A. Colonic metabolism of dietary polyphenols: Influence of structure on microbial fermentation products. Free Radic. Biol. Med. 2004, 36, 212–225. [Google Scholar] [CrossRef]
- Carreau, C.; Flouriot, G.; Bennetau-Pelissero, C.; Potier, M. Enterodiol and enterolactone, two major diet-derived polyphenol metabolites have different impact on ERα transcriptional activation in human breast cancer cells. J. Steroid Biochem. Mol. Biol. 2008, 110, 176–185. [Google Scholar] [CrossRef]
- Lampe, J.W. Is equol the key to the efficacy of soy foods? Am. J. Clin. Nutr. 2009, 89, 1664S–1667S. [Google Scholar] [CrossRef] [PubMed]
- Koh, A.; Molinaro, A.; Ståhlman, M.; Khan, M.T.; Schmidt, C.; Mannerås-Holm, L.; Wu, H.; Carreras, A.; Jeong, H.; Olofsson, L.E.; et al. Microbially Produced Imidazole Propionate Impairs Insulin Signaling through mTORC1. Cell 2018, 175, 947–961.e17. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Lu, Y.; Yuan, S.; Cai, X.; He, Y.; Chen, J.; Wu, Q.; He, D.; Fang, A.; Bo, Y.; et al. Gut microbiota–derived metabolite trimethylamine-N-oxide and multiple health outcomes: An umbrella review and updated meta-analysis. Am. J. Clin. Nutr. 2022, 116, 230–243. [Google Scholar] [CrossRef]
- Karusheva, Y.; Koessler, T.; Strassburger, K.; Markgraf, D.; Mastrototaro, L.; Jelenik, T.; Simon, M.-C.; Pesta, D.; Zaharia, O.-P.; Bódis, K.; et al. Short-term dietary reduction of branched-chain amino acids reduces meal-induced insulin secretion and modifies microbiome composition in type 2 diabetes: A randomized controlled crossover trial. Am. J. Clin. Nutr. 2019, 110, 1098–1107. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Li, Y.; Ivey, K.L.; Wang, D.D.; Wilkinson, J.E.; Franke, A.; Lee, K.H.; Chan, A.; Huttenhower, C.; Hu, F.B.; et al. Interplay between diet and gut microbiome, and circulating concentrations of trimethylamine N-oxide: Findings from a longitudinal cohort of US men. Gut 2022, 71, 724–733. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Dai, M. Trimethylamine N-Oxide Generated by the Gut Microbiota Is Associated with Vascular Inflammation: New Insights into Atherosclerosis. Mediat. Inflamm. 2020, 2020, 4634172. [Google Scholar] [CrossRef] [PubMed]
- Naghipour, S.; Cox, A.J.; Peart, J.N.; Du Toit, E.F.; Headrick, J.P. Trimethylamine N-oxide: Heart of the microbiota–CVD nexus? Nutr. Res. Rev. 2021, 34, 125–146. [Google Scholar] [CrossRef]
- Schiattarella, G.G.; Sannino, A.; Toscano, E.; Giugliano, G.; Gargiulo, G.; Franzone, A.; Trimarco, B.; Esposito, G.; Perrino, C. Gut microbe-generated metabolite trimethylamine-N-oxide as cardiovascular risk biomarker: A systematic review and dose-response meta-analysis. Eur. Heart J. 2017, 38, 2948–2956. [Google Scholar] [CrossRef]
- Farhana, A.; Khan, Y. Biochemistry, Lipopolysaccharide. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Schicho, R.; Marsche, G.; Storr, M. Cardiovascular Complications in Inflammatory Bowel Disease. Curr. Drug Targets 2015, 16, 181–188. [Google Scholar] [CrossRef]
- Al-Assal, K.; Martinez, A.C.; Torrinhas, R.S.; Cardinelli, C.; Waitzberg, D. Gut microbiota and obesity. Clin. Nutr. Exp. 2018, 20, 60–64. [Google Scholar] [CrossRef]
- Blaut, M. Gut microbiota and energy balance: Role in obesity. Proc. Nutr. Soc. 2015, 74, 227–234. [Google Scholar] [CrossRef]
- Adak, A.; Khan, M.R. An insight into gut microbiota and its functionalities. Cell. Mol. Life Sci. 2019, 76, 473–493. [Google Scholar] [CrossRef]
- MetaHIT Consortium (additional members); Arumugam, M.; Raes, J.; Pelletier, E.; Le Paslier, D.; Yamada, T.; Mende, D.R.; Fernandes, G.R.; Tap, J.; Bruls, T.; et al. Enterotypes of the human gut microbiome. Nature 2011, 473, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Crovesy, L.; Masterson, D.; Rosado, E.L. Profile of the gut microbiota of adults with obesity: A systematic review. Eur. J. Clin. Nutr. 2020, 74, 1251–1262. [Google Scholar] [CrossRef] [PubMed]
- Flint, H.J. Obesity and the Gut Microbiota. J. Clin. Gastroenterol. 2011, 45, S128–S132. [Google Scholar] [CrossRef]
- Delzenne, N.M.; Neyrinck, A.M.; Bäckhed, F.; Cani, P.D. Targeting gut microbiota in obesity: Effects of prebiotics and probiotics. Nat. Rev. Endocrinol. 2011, 7, 639–646. [Google Scholar] [CrossRef] [PubMed]
- Sanmiguel, C.; Gupta, A.; Mayer, E.A. Gut Microbiome and Obesity: A Plausible Explanation for Obesity. Curr. Obes. Rep. 2015, 4, 250–261. [Google Scholar] [CrossRef] [PubMed]
- Wolters, M.; Ahrens, J.; Romaní-Pérez, M.; Watkins, C.; Sanz, Y.; Benítez-Páez, A.; Stanton, C.; Günther, K. Dietary fat, the gut microbiota, and metabolic health—A systematic review conducted within the MyNewGut project. Clin. Nutr. 2019, 38, 2504–2520. [Google Scholar] [CrossRef]
- Redondo-Useros, N.; Nova, E.; González-Zancada, N.; Díaz, L.E.; Gómez-Martínez, S.; Marcos, A. Microbiota and Lifestyle: A Special Focus on Diet. Nutrients 2020, 12, 1776. [Google Scholar] [CrossRef]
- Patel, S.; Behara, R.; Swanson, G.; Forsyth, C.; Voigt, R.; Keshavarzian, A. Alcohol and the Intestine. Biomolecules 2015, 5, 2573–2588. [Google Scholar] [CrossRef]
- Lee, E.; Lee, J.-E. Impact of drinking alcohol on gut microbiota: Recent perspectives on ethanol and alcoholic beverage. Curr. Opin. Food Sci. 2021, 37, 91–97. [Google Scholar] [CrossRef]
- Bell, D.S.H. Changes seen in gut bacteria content and distribution with obesity: Causation or association? Postgrad. Med. 2015, 127, 863–868. [Google Scholar] [CrossRef] [PubMed]
- Martínez Leo, E.E.; Segura Campos, M.R. Effect of ultra-processed diet on gut microbiota and thus its role in neurodegenerative diseases. Nutrition 2020, 71, 110609. [Google Scholar] [CrossRef] [PubMed]
- Cuevas-Sierra, A.; Milagro, F.I.; Aranaz, P.; Martínez, J.A.; Riezu-Boj, J.I. Gut Microbiota Differences According to Ultra-Processed Food Consumption in a Spanish Population. Nutrients 2021, 13, 2710. [Google Scholar] [CrossRef] [PubMed]
- Hall, K.D.; Ayuketah, A.; Brychta, R.; Cai, H.; Cassimatis, T.; Chen, K.Y.; Chung, S.T.; Costa, E.; Courville, A.; Darcey, V.; et al. Ultra-Processed Diets Cause Excess Calorie Intake and Weight Gain: An Inpatient Randomized Controlled Trial of Ad Libitum Food Intake. Cell Metab. 2019, 30, 67–77.e3. [Google Scholar] [CrossRef]
- Seganfredo, F.B.; Blume, C.A.; Moehlecke, M.; Giongo, A.; Casagrande, D.S.; Spolidoro, J.V.N.; Padoin, A.V.; Schaan, B.D.; Mottin, C.C. Weight-loss interventions and gut microbiota changes in overweight and obese patients: A systematic review: Weight-loss impact on gut microbiota. Obes. Rev. 2017, 18, 832–851. [Google Scholar] [CrossRef]
- Koponen, K.K.; Salosensaari, A.; Ruuskanen, M.O.; Havulinna, A.S.; Männistö, S.; Jousilahti, P.; Palmu, J.; Salido, R.; Sanders, K.; Brennan, C.; et al. Associations of healthy food choices with gut microbiota profiles. Am. J. Clin. Nutr. 2021, 114, 605–616. [Google Scholar] [CrossRef]
- Cotillard, A.; Kennedy, S.P.; Kong, L.C.; Prifti, E.; Pons, N.; Le Chatelier, E.; Almeida, M.; Quinquis, B.; Levenez, F.; Galleron, N.; et al. Dietary intervention impact on gut microbial gene richness. Nature 2013, 500, 585–588. [Google Scholar] [CrossRef]
- Boysen, O.; Boysen-Urban, K.; Bradford, H.; Balié, J. Taxing highly processed foods: What could be the impacts on obesity and underweight in sub-Saharan Africa? World Dev. 2019, 119, 55–67. [Google Scholar] [CrossRef]
- La Fata, G.; Weber, P.; Mohajeri, M.H. Probiotics and the Gut Immune System: Indirect Regulation. Probiotics Antimicrob. Proteins 2018, 10, 11–21. [Google Scholar] [CrossRef]
- Kechagia, M.; Basoulis, D.; Konstantopoulou, S.; Dimitriadi, D.; Gyftopoulou, K.; Skarmoutsou, N.; Fakiri, E.M. Health Benefits of Probiotics: A Review. Int. Sch. Res. Not. 2013, 2013, 481651. [Google Scholar] [CrossRef] [PubMed]
- Gomes, A.C.; De Sousa, R.G.M.; Botelho, P.B.; Gomes, T.L.N.; Prada, P.O.; Mota, J.F. The additional effects of a probiotic mix on abdominal adiposity and antioxidant Status: A double-blind, randomized trial: Probiotic Mix and Abdominal Adiposity. Obesity 2017, 25, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Mohajeri, M.H.; Brummer, R.J.M.; Rastall, R.A.; Weersma, R.K.; Harmsen, H.J.M.; Faas, M.; Eggersdorfer, M. The role of the microbiome for human health: From basic science to clinical applications. Eur. J. Nutr. 2018, 57, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Swanson, K.S.; Gibson, G.R.; Hutkins, R.; Reimer, R.A.; Reid, G.; Verbeke, K.; Scott, K.P.; Holscher, H.D.; Azad, M.B.; Delzenne, N.M.; et al. The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of synbiotics. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 687–701. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Arraño, V.; Martín-Peláez, S. Effects of Probiotics and Synbiotics on Weight Loss in Subjects with Overweight or Obesity: A Systematic Review. Nutrients 2021, 13, 3627. [Google Scholar] [CrossRef]
- Borgeraas, H.; Johnson, L.K.; Skattebu, J.; Hertel, J.K.; Hjelmesaeth, J. Effects of probiotics on body weight, body mass index, fat mass and fat percentage in subjects with overweight or obesity: A systematic review and meta-analysis of randomized controlled trials: Effects of probiotics on anthropometrics. Obes. Rev. 2018, 19, 219–232. [Google Scholar] [CrossRef] [PubMed]
- Perna, S.; Ilyas, Z.; Giacosa, A.; Gasparri, C.; Peroni, G.; Faliva, M.A.; Rigon, C.; Naso, M.; Riva, A.; Petrangolini, G.; et al. Is Probiotic Supplementation Useful for the Management of Body Weight and Other Anthropometric Measures in Adults Affected by Overweight and Obesity with Metabolic Related Diseases? A Systematic Review and Meta-Analysis. Nutrients 2021, 13, 666. [Google Scholar] [CrossRef]
- Suzumura, E.A.; Bersch-Ferreira, Â.C.; Torreglosa, C.R.; Da Silva, J.T.; Coqueiro, A.Y.; Kuntz, M.G.F.; Chrispim, P.P.; Weber, B.; Cavalcanti, A.B. Effects of oral supplementation with probiotics or synbiotics in overweight and obese adults: A systematic review and meta-analyses of randomized trials. Nutr. Rev. 2019, 77, 430–450. [Google Scholar] [CrossRef]
- Pedersini, P.; Turroni, S.; Villafañe, J.H. Gut microbiota and physical activity: Is there an evidence-based link? Sci. Total Environ. 2020, 727, 138648. [Google Scholar] [CrossRef]
- Ortiz-Alvarez, L.; Xu, H.; Martinez-Tellez, B. Influence of Exercise on the Human Gut Microbiota of Healthy Adults: A Systematic Review. Clin. Transl. Gastroenterol. 2020, 11, e00126. [Google Scholar] [CrossRef]
- Allen, J.M.; Mailing, L.J.; Niemiro, G.M.; Moore, R.; Cook, M.D.; White, B.A.; Holscher, H.D.; Woods, J.A. Exercise Alters Gut Microbiota Composition and Function in Lean and Obese Humans. Med. Sci. Sports Exerc. 2018, 50, 747–757. [Google Scholar] [CrossRef]

| Author (Year) | Type of Study | Population | N | Intervention | Control | Time (Weeks) | Results |
|---|---|---|---|---|---|---|---|
| Crovesy, El-Bacha and Rosado [14] | Clinical trial, randomized, double-blind | Obese women | 32 | Hypocaloric diet + probiotic or symbiotic supplementation | Placebo supplementation | 8 | No differences in anthropometry between groups of intervention After the dietary intervention, all groups showed changes in the metabolic profile associated with the reduction in inflammation |
| Dong et al. [15] | Clinical trial, randomized | Overweight or obese adults and older adults | 80 | Hypoproteic diet, initially normocaloric and after with caloric reduction | Diet with normal content of PTN | 8 | No significant differences between weight loss in all groups Differences in microbiota composition between individuals according to higher or lower fiber consumption ↑ α diversity and abundance of 6 genera of bacteria in the intervention group |
| Gøbel et al. [16] | Clinical trial, randomized, double-blind | Obese adolescents | 50 | Probiotic supplementation with Lactobacillus salivarius Ls-33 | Placebo supplementation | 12 | No changes in inflammatory markers after intervention (fasting glucose, insulin, HOMA-IR, C-peptide) ↓ fasting insulin, HOMA-IR and C-peptide in the placebo group |
| Gomes, Hoffmann and Mota [17] | Clinical trial, randomized, double-blind | Overweight or obese women | 32 | Probiotic supplementation | Placebo supplementation | 12 | Best lipid profile showed ↑ Prevotella, Collinsella, Paraprevotella Enterococcus, Clostridiaceaee Veillonella, while the worst lipid profile showed ↑ phylum TM7, Lachnospiraceae and Roseburia Alterations in microbial composition in the intervention group: ↑ Firmicutes and ↓ Bacteroidetes |
| Haro, Borrego et al. [18] | Clinical trial, randomized | Obese men | 20 | Mediterranean diet | Low-lipid high-complex CHO diet | 48 | ↑ insulin sensitivity in all groups ↑ genera Prevotella and F. prausnitzii + ↓ Roseburia in low-lipid high-complex CHO diet ↑ genera Roseburia and Oscillospira in Mediterranean diet Both diets promoted changes in abundance of T2DM-related bacterial abundance, promoting a protective effect |
| Jian et al. [19] | Clinical trial, randomized | Overweight or obese individuals | 38 |
| - | 3 | ↑ phylum Proteobacteria in high-saturated-fat diet ↑ Lactococcus and Escherichia coli in a high-sugar diet ↑ butyrate producers in high-unsaturated-fat diet ↑ proportion of Firmicute to Bacteroidetes in non-alcoholic fatty liver disease ↑ BMI in all groups No differences between the richness of microbial genes and α diversity, comparing all groups |
| Kanazawa et al. [20] | Clinical trial, randomized | Obese and DM2 individuals | 88 | Symbiotic supplementation | No type of symbiotic, probiotic or prebiotic supplementation | 24 | ↑ fasting glucose and HbA1c in the symbiotic group, followed by normalization No differences in HbA1c, BMI, lipid profile and IL-6 between all groups at the end of the study ↑ Bifidobacterium, cluster Atopobium, total lactobacilli and Lactobacillus, Lacticaseibacillus and Limosilactobacillus in symbiotic group at the end of the study |
| Leber et al. [21] | Clinical trial, randomized | Individuals with metabolic syndrome or healthy | 38 | Supplementation with probiotic fermented milk (Lactobacillus casei Shirota) | No type of supplementation | 12 | Individuals with metabolic syndrome showed greater intestinal permeability in comparison to healthy individuals The probiotic showed no changes in the parameters tested in the study |
| Leong et al. [22] | Clinical trial, randomized, double-blind | Obese adolescents (14–18 years) | 87 | Fecal microbiota transplantation of eutrophic individuals by oral capsules | Placebo capsules | 26 | ↑ microbial diversity six weeks post-intervention in women. No differences were found in men ↓ android/gnoid fat ratio, particularly in women Resolution of metabolic syndrome in most individuals after intervention |
| Ley et al. [23] | Clinical trial, randomized | Obese individuals | 12 |
| - | 48 | Before intervention: ↑ Firmicutes and ↓ Bacteroidetes After intervention: ↑ Bacteroidetes and ↓ Firmicutes ↑ Bacteroidetes was associated with weight loss |
| Marungruang et al. [24] | Clinical trial, randomized | Older individuals (50–73 years) with BMI between 25 and 33 kg/m2 | 47 | Diet with biomarkers related to cardiometabolic risk (foods with anti-inflammatory potential, antioxidants and anti- anti-hypercholesterolemic, like omega-3, polyphenols, dietary fiber) | Conventional diet without biomarkers | 8 | Weight loss in both diets Improvement in lipid profile in the intervention group No differences in diversity α and taxonomic levels of phyla and genera in the microbiome between the groups ↑ ratio Prevotella/Bacteroides after intervention in multifunctional diet |
| Meslier et al. [25] | Clinical trial, randomized | Overweight or obese individuals | 82 | Mediterranean diet without energy restriction | Habitual diet | 8 | ↓ plasma cholesterol and HDL cholesterol ↓ fecal bile acids Changes in the composition of microbiota in the first week of intervention Greater microbial gene richnesses observed at low levels of PCR |
| Muralidharan et al. [26] | Clinical trial, randomized | Overweight or obese individuals | 343 | Mediterranean diet with energy restriction and physical activity promotion | Mediterranean diet without energy restriction | 48 | ↓ weight, ↑ Bacteroidetes and ↓ Firmicutes in the intervention group No significant differences in α and β diversity in all groups ↓ BMI, waist circumference, TG levels, glucose and HbA1c in the intervention group |
| Neyrinck et al. [27] | Clinical trial, randomized, double-blind | Obese individuals | 24 | Inulin prebiotic supplementation + hypocaloric diet | Placebo supplementation | 12 | No changes between the groups in zonulin ↓ marker for intestinal inflammation after intervention ↑ SCFAs in both groups, but not significant Modification in β diversity, ↑ Actinobacteria, families Bifidobacteriaceae and Lachnospiraceae, Lactobacillaceae and genera Bifidobacterium after intervention |
| Nicolucci et al. [28] | Clinical trial, randomized, double-blind | Overweight or obese children (7–12 years) | 38 | Prebiotic supplementation with inulin enriched with oligofructose | Placebo supplementation | 16 | ↓ weight gain and % body fat in the intervention group Four individuals with insulin resistance were no longer classified as such after prebiotic intervention ↑ fecal bile acids in the placebo group ↑ Bifidobacterium spp. in the intervention group |
| Sergeev et al. [29] | Clinical trial | Overweight or obese individual | 20 | Hypocaloric diet + symbiotic supplementation | Hypocaloric diet + placebo supplementation | 12 | No significant differences between the groups in body composition ↓ HbA1C, ↑ relative abundance of gut bacteria and ↓ microbial genera associated with inflammation in the intervention group |
| Van Son et al. [30] | Cohort | Overweight or obese men and post-menopause women | 107 | - | - | 284 | A positive correlation was found between PLm and diastolic BP No significant differences in Plm between insulin-resistant and -sensitive individuals |
| Vrieze et al. [31] | Clinical trial, randomized, double-blind | Adult men with metabolic syndrome | 18 | Fecal transplantation of microbiota by duodenal tube | Fecal transplantation of own feces collected and processed | 6 | No significant changes were found in energy expenditure at rest ↑ gut microbiota diversity ↓ fecal SCFAs ↑ peripheral insulin sensitivity Tendency to improve hepatic sensitivity |
| Yu et al. [32] | Clinical trial, randomized, double-blind | Obese adults with insulin resistance | 24 | Fecal transplantation by capsules | Placebo supplementation | 12 | Comparing the intervention group and the control group, no differences were found in HOMA-IR, weight, fasting lipids or energy expenditure at rest A modest reduction in HbA1c in the intervention group |
| Author (Year) | Type of Study | Population | n | Time (Weeks) | Results |
|---|---|---|---|---|---|
| Bervoets et al. [33] | Cross-sectional | Children and adolescents | 53 | - | ↑ Firmicutes/Bacteroidetes ratio in obese children compared to control ↑ Staphylococcus spp. was associated with ↑ energy consumption ↓ Bacteroides vulgatus in obese subjects Lactobacillus spp. concentrations were associated with CRP levels |
| Cho [34] | Cohort | Children and adolescents | 36 | 48 | Pre-dietary intervention: ↓ Bacteroidetes in the weight-gain group, in comparison to control ↓ richness of microbial genes Post-dietary intervention: ↑ Firmicutes, ↓ Bacteroidetes, ↓ richness of genes in the fat-loss group ↓ Firmicutes, ↑ Actinobacteria, ↓ class Clostridia in the weight-gain group Romboutsia, Ruminococcaeceae _UCG_013, Eubacterium coprostanollgenes-group and Parabacteroides are important to microbial changes in the weight-gain group Romboutsia genera, Eubacterium_halli_ group and Clostridium_sensu_stricto are important in microbial changes and interaction in the fat gain group |
| Haro, Borrego et al. [35] | Clinical trial, randomized | Obese men | 20 | 48 | ↑ insulin sensitivity in all groups ↑ genera Prevotella and F. prausnitzii + ↓ Roseburia in low-lipid high-complex CHO diet ↑ genera Roseburia and Oscillospira in Mediterranean diet Both diets promoted changes in abundance of T2DM-related bacterial abundance, promoting a protective effect |
| Haro, Zúñiga et al. [18] | Cohort | Adults | 75 | 240 | Microbiota composition seems to be different according to sex and seems to be influenced by BMI ↑ Firmicutes in women independent of BMI ↑ Firmicutes in men with BMI > 33 kg/m2 ↓ Bacteroides in men with a BMI of 33 kg/m2 |
| Jumpertz et al. [36] | Cohort | Lean or obese adults | 21 | - | Firmicutes → associated with increasing nutrient absorption Bacteroidetes → associated with a decrease in nutrient absorption (−150 kcal) No differences in caloric excretion in feces of eutrophic or obese with 2.400 kcal/d diet Eutrophic individuals lost less energy in feces with 3.400 kcal/d diet No differences in caloric excretion in feces of obese subjects between two diets |
| Kim et al. [37] | Cohort | Overweight or obese individuals | 747 | 16 | ↓ diversity α in MUH No differences in α diversity between the healthy control group and MH ↑ genera Oscillospira and Clostridium, ↑ family Coriobacteriaceae and Leuconostocaceae in MH ↑ Fusobacteria in MUH No differences in ratio Firmicutes/Bacteroidetes between MUH and MH |
| Kong et al. [38] | Cross-sectional | Lean, overweight or obese individuals | 45 | - | ↓ Clostridia leptum, Clostridia coccoides and Bacteroides/Prevotella in individuals that were overweight or obese ↑ richness and diversity of microbial genes in individuals with higher consumption of fruits, yogurts, soups and lower consumption of sugar and sugary drinks The worst food pattern was associated with alterations in lipid profile |
| Menni et al. [39] | Cross-sectional | Healthy women | 1.632 | - | ↓ α diversity in weight-gain group Dietary fiber intake was related to microbiota diversity and lower weight gain Firmicutes were related to a lower risk of weight gain Bacteroides was related to an increased risk of weight gain |
| Navarro et al. [40] | Cross-sectional | Adults, healthy | 68 | 336 | ↑ acetate concentrations, ↓ Bacteroides in obese subjects Lactobacillus was related as a risk factor for obesity |
| Olivares et al. [41] | Cross-sectional | Adults | 109 | - | ↓ microbial diversity and ↓ ratio Firmicutes/Bacteroidetes in ObMUH ↑ Bifidobacterium in eutrophic EuMH ↑ family Prevotellaceae and genera Eubacterium rectale and Faecalibacterium in people ObMH and OvMH compared to EuMH ↑ Coprococcus and Ruminococcus in OvMH |
| Orsso et al. [42] | Cross-sectional | Obese children | 21 | 80 | Increased HOMA-IR was associated with ↓ richness of microbial genes, ↓ species richness of Firmicutes and ↓ diversity of Proteobacteria ↓ α diversity was associated with ↑ PCR in obese subjects |
| Peters et al. [43] | Cross-sectional | Lean, overweight or obese individuals | 599 | - | ↓ richness of microbial genes in obese compared to eutrophic No differences in α diversity between overweight and eutrophic ↑ families Streptococcaceae, Lactobacillaceae, Veillonellaceae Gemellaceae and ↓ Christensenellaceae, Clostridiaceae, Dehalobacteriaceae in obese ↑ Lactobacillaceae, Streptococcaceae and ↓ Christensenellaceae, Clostridiaceae, Dehalobacteriaceae in overweight subjects |
| Roland et al. [44] | Cohort prospective | Individuals with suspicion of SIBO | 30 | 24 | Obese people showed a prevalence of SIBO ↑ small intestine transit time and ↑ gastric and small intestine pH in SIBO ↓ α diversity, ↓ genera Parabacteriodes, Oscillospira and families Bacteroidaceae, Lachnospiraceae in obese with SIBO compared to eutrophic with SIBO ↑ Firmicutes ↓ Bacteroidetes in obese |
| Stefura et al. [45] | Cohort prospective | Lean or grade III obese individuals | 96 | 48 | Eutrophic and obese showed phylum Firmucutes elevated compared Bacteroidetes ↑ genera Bacteroides, Odoribacter, Blautia in obese ↑ Ruminococcus, Christensenella, Faecalibacterium in eutrophic ↑ Romboutsia, Lactobacillus, Flavonifractor in BMI ≥ 50 kg/m2 |
| Shen et al. [46] | Cohort | Post-bariatric surgery individuals | 26 | 48 | No differences in ratio Firmicutes/Bacteroidetes pre- and post-surgery Post-bariatric surgery: ↑ α diversity, improvement in microbial metabolites and markers related to insulin resistance and DCV Several aspects of microbiota have been modified (composition, diversity) quickly (3–6 months) after the procedure. However, there was a reduction 12 m after surgery |
| Silva, Monteil and Davis [47] | Cohort | Children | 51 | - | ↓ family Bifidobacteriaceae and phylum Bifidobacterium, ↑ Lactobacillus and Firmicutes in overweight/obese children compared to eutrophic ↓ phylogenetic diversity in Ob/Ov |
| Van Son et al. [30] | Cohort | Overweight or obese men and post-menopause women | 107 | 284 | A positive correlation was found between PLm and diastolic BP No significant differences in Plm between insulin-resistant and -sensitive individuals |
| Yun et al. [48] | Cross-sectional | Adults | 1274 | 16 | ↓ α diversity in obese No differences in ratio Firmicutes/Bacteroidetes between obese, overweight and eutrophic Depletion in lipid metabolism, biodegradation of xenobiotics, ↑ gene-related to purine metabolism and oxidative phosphorylation, alterations in the immune response, ↓ metabolism of CHO, pyruvate and some amino acids in obese individuals In a taxonomic analysis separated by BMI, bacteria from obese individuals were not influenced by the dietary confounder |
| Yuan et al. [49] | Cohort | Obese children and adolescents | 86 | 28 | ↑ α and β diversity in ObMH and the control group ↑ genera Anaerostipes, Oscillospir, Odoribacter, Gemmiger, Parabacteroides, Alistipes in ObMH and the control group ↑ genera Bacteroides in ObMH ↑ Fusobacterium in ObMUH |
| Zeng et al. [50] | Cohort retrospective | Lean, overweight or obese adults | 1.914 | - | ↑ bacterial diversity in obese subjects without metabolic alterations compared to eutrophic Gradual changes in the microbiota with the aggravation of obesity |
| Zeng et al. [51] | Cohort | Obese individuals | 383 | - | ↑ microbial diversity and gene count, ↓ ratio Firmicutes/Bacteroidetes in ObMH compared to ObMUH ↑ Alistipes, Bifidobacterium, Eubacterium, Faecalibacterium, Ruminococcus, Subdoligranulum and ↓ phylum Fusobacteria in ObMH ↑ Escherichia, Clostridium, Fusobacterium and Megamonas in ObMUH ↑ microbial genes associated with LPS biosynthesis in ObMUH |
| Author (Year) | Type of Study | Population | n | Intervention | Control | Time (Weeks) | Results |
|---|---|---|---|---|---|---|---|
| Bo et al. [52] | EXP | C57BL/6J mice | 36 |
| Standard diet | 8 | ↓ glucose toleration and ↑ lipid profile markers in HFD ↓ visceral fat, ↑ Bacteroidetes, ↓ Firmicutes, ↑ Butyricimonas, Bifidobacterium and Odoribacter in obese mice using B. pseudolongum No differences between the groups in α and β diversity |
| Denou et al. [53] | EXP | Mice | 16 |
| Without physical activity | 12 | ↓ ratio Bacteroidetes/Firmicutes and ↓ α diversity in Ob/HFD ↑ α diversity, ↑ ratio Bacteroidetes/Firmicutes after HIIT in Ob/HFD Ob/HFD mice showed insulin and glucose intolerance No reduction in body mass or fasting glucose, but improved insulin sensitivity after HIIT in Ob/HFD |
| Evans et al. [54] | EXP | Male C57BL/6 mice | 48 |
| - | 14 | ↑ weight and body fat, change in glucose metabolism in group 2 ↓ Firmicules ↑ Bacteroidetes, ↑ families Lachnospiraceae Ruminococcaceae and S24-7, ↓ Lactobacillaceae and Turicibacteraceae in physical activity independent of diet ↑ Actinobacteria in group 1 ↑ families Clostridiaceae, Lachnospiraceae and Ruminococcaceae, ↓ Turicibacteraceae and S24-7, tendency ↑ Proteobacteria in HFD |
| Everard et al. [55] | EXP | C57BL/6J mice | 40 |
| Control diet | 8 | HFD + PREB: ↓ ratio Firmicutes/Bacteroidetes, ↓ proportion of Tenericutes, Cianobactérias and Verrucomicrobia, ↓ Bilophila, Butyrivibrio, LE30 and Oribacterium, ↑ Allobacullum and Prevotella, ↓ hepatic LBP, ↓ inflammatory markers ↑ SCFA and ↓ insulin resistance in using PREB in both diets PREB had a greater impact on HFD than the control diet |
| Fjære et al. [56] | EXP | Male C57BL/6J mice | 70 |
| - | 16 | No differences in α diversity between the groups ↑ phylum Verrucomicrobia and ↓ Proteobacteria, ↓ families Rikenellaceae, Desulfovibrionaceae and Clostridiaceae in LFD ↑ bacterial genes related to bile acids biosynthesis in sedentary animals in HFSD ↑ gene related to the transport of sugar in animals authorized to exercise voluntarily in HFSD |
| Gu et al. [57] | EXP | Male C57BL/6J mice | 22 | High-fat diet (HFD) | Standard diet | 8 | ↑ Firmicutes, Bacteroidetes and Proteobacteria in the control groups and obesity-resistant mice ↑ Bacteroidetes ↓ Firmicutes in obesity-prone mice Metabolic profile and gut microbiota profile were different between obesity-resistant and obesity-prone mice |
| Guirro et al. [58] | EXP | Male mice | 8 |
| - | 14 | ↑ ratio Bacteroidetes/Firmicutes in HFD compared to LFD Differences were identified between the families of microorganisms that colonize the microbiome in both diets In tests with antibiotics, the cecal microbial content was reduced. In a later test with fecal microbiota transplantation, the biodiversity of the microbiome was restored |
| Hussain et al. [59] | EXP | Male C57BL/6J mice with diet-induced obesity | 18 |
| Normal diet | 16 | ↓ body weight using LB818 ↓ body weight in group 2, compared to group 1 ↓ TG, LDL, fasting glucose and fat deposition in the liver, ↑ HDL in groups 2 and 3 ↑ Firmicutes in HFD compared to control ↑ species Akkermanasia and Bifidobacteria and ↓ Firmicutes using LB818 ↑ ratio Bacteroidetes/Firmicutes in groups 2 and 3 |
| Ji et al. [60] | EXP | Male C57BL/6 mice | 48 | High-fat diet (HFD) + coarse cereal mix (millet, corn, oats, soybeans and purple potatoes) | Feed + coarse cereal mix | 8 | ↓ weight gain and fat accumulation, ↑ SCFA in HFD + cereal mix ↑ glucose tolerance and improvement in lipid profile, ↑ diversity and microbial richness of microbiome, ↓ liberation of pro-inflammatory cytokines using cereal mix ↑ phylum Bacteroidetes and Actinobacterias, ↑ genera Bifidobacterium, Lactobacillus, Holdemanella, Barnesiella, Okibacterium and Streptophyta, ↓ ratio Firmicutes/Bacteroidetes using cereal mix |
| Joung et al. [61] | EXP | Male C57BL/6J mice | 40 |
| Normal diet | 12 | ↓ weight gain, fat accumulation, and slight improvement in intestinal permeability induced by HFD using LK50 ↓ TG, fasting glucose, ALT, AST, ↑ HDL, insulin improvement, ↓ ratio Firmicutes/Bacteroidetes, ↑ α and β diversity, ↓ liberation of pro-inflammatory cytokines using LK50 ↓ Actinobacteria and Erysipelotrichia, ↑ Lactobacillus using LK50 |
| Ke et al. [62] | EXP | Germ-free male C57BL/6J mice | 60 |
|
| 12 | PREB/symbiotic results after changes caused by HFD: ↓ weight gain, ↓ fasting insulin and cholesterol and improvement in HOMA-IR. PROB results: ↓ fasting insulin and slight weight reduction Symbiotic results: more efficient in ↓ fasting glucose ↑ microbial richness and ↑ SFCA using supplements ↓ bile acids and improvement in functional activities of the intestinal ecosystem from symbiotics |
| Kiilerich et al. [63] | EXP | Females C57BL/6JBomTac mice | 150 |
| Low-fat diet (LFD) | 72 | PTN and sucrose helped reduce weight gain, but HFPD showed greater weight gain ↓ survival of animals fed with HFSD Obesity was associated with mortality ↑ Lactobacillus in HFSD and HFPD ↓ ratio Bacteroidetes/Firmicutes according to animals’ age in HFPD and LFD |
| Kübeck et al. [64] | EXP | Germ-free mice (GFM) and pathogen-free male C57BL/6 mice (PFM) | 60 | In both types of animals:
| Control diet | 8 | GFM on diet 2 showed no weight gain, suggesting resistance to diet-induced obesity Reduced intestinal fat absorption and higher basal metabolic rate (↑ energy expenditure) in GFM on diet 2 PFM was obese compared to GFM, suggesting that microbial composition exerts some influence on the loss of lean phenotype ↑ Clostridiales spp. and Bacteroidales in HFD Dietary cholesterol may have a protective effect against diet-induced obesity |
| Lai et al. [65] | EXP | Male C57BL/6JNarl mice | 47 |
| - | 24 | Diet influenced more the composition of the microbiota and α diversity than exercise NFDE group microbiota transplantation transfers effects similar to physical exercise for weight loss and LIP on HFD diet ↑ genera Turicibacter, Sutterella, Prevotella, AF12 and Helicobacter in NFD and NFDE ↑ Odoribacter, AF12, Helicobacter and Akkermansia in HFDE and NFDE ↑ Odoribacter, Helicobacter and AF12 in the groups of microbiota transplantation Use of antibiotics that preceded transplantation ↑ obesity development risks |
| Li et al. [66] | EXP | Male Sprague Dawley mice | 20 |
| - | 19 | ↑ weight gain, ↑ Bacteroidetes, ↓ Proteobacteria, ↑ abundance of bacterial species in lean mice in group 1 ↑ ratio Firmicutes/Bacteroidetes in lean mice compared to obese mice ↓ ratio Firmicutes/Bacteroidetes after intervention in lean mice Weight gain was associated with increase in Bacteroidetes Few changes in microbiome community of obese animals: ↓ Desulfovibrionaceae and Lactobacillaceae, ↑ Ruminococcaceae |
| Lu et al. [67] | EXP | Male C57BL/6J mice | 60 |
| Low-fat diet (LFD) | 16 | The SCFA from groups 2, 3, 4 and 5 prevented weight gain, promoted a partial improvement in the composition of the microbiota and reduced the increase in TG and cholesterol caused by an HFD No differences between the groups in microbial diversity ↓ microbial richness, ↑ ratio Firmicutes/Bacteroidetes in HFD ↓ Firmicutes ↑ Bacteroidetes in groups 2 and 3 |
| Moreira Júnior et al. [68] | EXP | Pathogen-free C57BL/6 mice | 24 |
| - | 12 | ↑ weight and adiposity, development of hepatic steatosis, ↑ Firmicutes and Actinobacteria ↓ Bacteroidetes in group 2 ↑ relative abundance of Lachnoclostridium, Bifidobacterium, Parvibacter, Ruminiclostridium and Blautia in group 2 Diet of group 2 was associated with impulsivity and an anxiolytic effect |
| Moretti et al. [69] | EXP | Germ-free and conventional mice (normal microbiome) | 16 | Western diet | Regular diet | 16 | ↑ weight, ↑ fat mass, ↓ lean mass, ↑ fasting glucose, adipose tissue inflammation, development of obesity in both animal groups with a western diet |
| Oh et al. [70] | EXP | Male mice with diet-induced obesity | 36 |
| High-fat diet (HFD) | 10 | Less weight loss in group 4 ↑ microbial richness in group 2, but ↓ in group 3 ↑ microbial diversity in combined use or not of PROB and PREB ↑ ratio Firmicutes/Bacteroidetes in HFD and no changes using the supplement ↓ Proteobacteria and ↓ taxa associated with obesity using PROB and/or PREB Weight gain was positively associated with phylum Verrucomicrobia and negatively associated with Bacteroidetes and Firmicutes |
| Ridaura et al. [71] | EXP | Germ-free mice | 12 a 16 | Fecal microbiota transplantation from obese discordant human twins to germ-free mice | Mice transplanted with microbiota from lean twins | 1–4 | ↑ body mass in obese microbiota sample ↑ fermentation of butyrate and propionate, digestion of polysaccharides in the lean microbiota sample By housing an obese microbiota mouse with a lean one, the increase in adiposity in the obese animal was reduced, and similar characteristics to the lean animal were transferred |
| Saiyasit et al. [72] | EXP | Male Wistar mice | 140 |
| - | 40 | In HFD: cognitive impairment, ↑ weight, LPS, LDL, cholesterol, HOMA-IR, ↓ HDL ↑ ratio Firmicutes/Bacteroidetes and ↑ ratio Enterobacteriaceae/Eubacteria HFD promoted dysbiosis in animal microbiota from the first week of the study |
| Shang et al. [73] | EXP | Male C57BL/6J mice | 12 |
| Low-fat diet (LFD) | 7 | Higher α diversity in groups 1 and 2 compared to control Higher metabolism capacity of LIP, CH, starch and sucrose, ↓ S24-7, ↑ Lachnospiraceae in HFD ↑ ratio Bacteroidetes/Firmicutes, ↓ Proteobacteria in the control group LFD partially re-established diversity and composition of gut microbiota after HFD |
| Turnbaugh et al. [74] | EXP | Obese and lean mice | 22 | - | - | - | ↑ Firmicutes in Ob animals ↓ Firmicutes ↑ Bacteroidetes in lean animals ↑ final products of butyrate and acetate fermentation, ↓ residual energy of feces (compared to lean microbiome) in Ob mice In a fecal transplantation test of Ob and lean mice to germ-free mice, the characteristics of the obese microbiome were transmitted, promoting body fat gain |
| Turnbaugh et al. [75] | EXP | Germ-free male C57BL/6J mice | 15 | Fecal microbiota transplantation from human adults +
| Low-fat diet (LFD) and high content of plant polysaccharides | 12 | Fecal microbiota transplantation from human adults was successful Diet of group 1: ↑ Bacteroidetes Diet of group 2: ↑ Firmicutes (class Erysipelotrichi and Bacilli) and ↓ Bacteroidetes • In fecal microbiota transplantation from transplanted animals to germ-free mice, human gut microbiota were transmitted from generation to generation and hence maintained its diversity. However, the composition of the gut microbiome is directly influenced by the recipient’s diet The transplanted microbiota was similar to human microbiota after 7 days, while microbiome changes were observed by 1 day of the western diet |
| Welly et al. [76] | EXP | Obesity-prone male mice | 30 |
| HFD and sedentary | - | ↑ weight in the sedentary group No differences in α diversity, relative abundance of ratio Firmicutes/Bacteroidetes and phylum-level changes between groups ↓ Bacteroidetes in groups 2 and 3 compared to control Group 2: ↑ family Streptococcaceae and ↓ Rikenellaceae Group 3: ↓ Streptococcus compared to other groups |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tassoni, D.S.; Macedo, R.C.O.; Delpino, F.M.; Santos, H.O. Gut Microbiota and Obesity: The Chicken or the Egg? Obesities 2023, 3, 296-321. https://doi.org/10.3390/obesities3040024
Tassoni DS, Macedo RCO, Delpino FM, Santos HO. Gut Microbiota and Obesity: The Chicken or the Egg? Obesities. 2023; 3(4):296-321. https://doi.org/10.3390/obesities3040024
Chicago/Turabian StyleTassoni, Daniele S., Rodrigo C. O. Macedo, Felipe M. Delpino, and Heitor O. Santos. 2023. "Gut Microbiota and Obesity: The Chicken or the Egg?" Obesities 3, no. 4: 296-321. https://doi.org/10.3390/obesities3040024
APA StyleTassoni, D. S., Macedo, R. C. O., Delpino, F. M., & Santos, H. O. (2023). Gut Microbiota and Obesity: The Chicken or the Egg? Obesities, 3(4), 296-321. https://doi.org/10.3390/obesities3040024







