Acute and Chronic Effects of Low-Volume High-Intensity Interval Training Compared to Moderate-Intensity Continuous Training on Glycemic Control and Body Composition in Older Women with Type 2 Diabetes
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Protocol
2.2. Participants
2.3. Cardiopulmonary Exercise Testing
2.4. Anthropometrics and Body Composition
2.5. Cardiometabolic Profile
2.6. Continuous Glucose Monitoring
2.7. Macronutrient Intakes
2.8. Estimated Energy Expenditure and Physical Activity Levels
2.9. Training Capillary Blood Glucose Levels before and after Exercise
2.10. Exercise Intervention
2.11. Statistical Analysis
3. Results
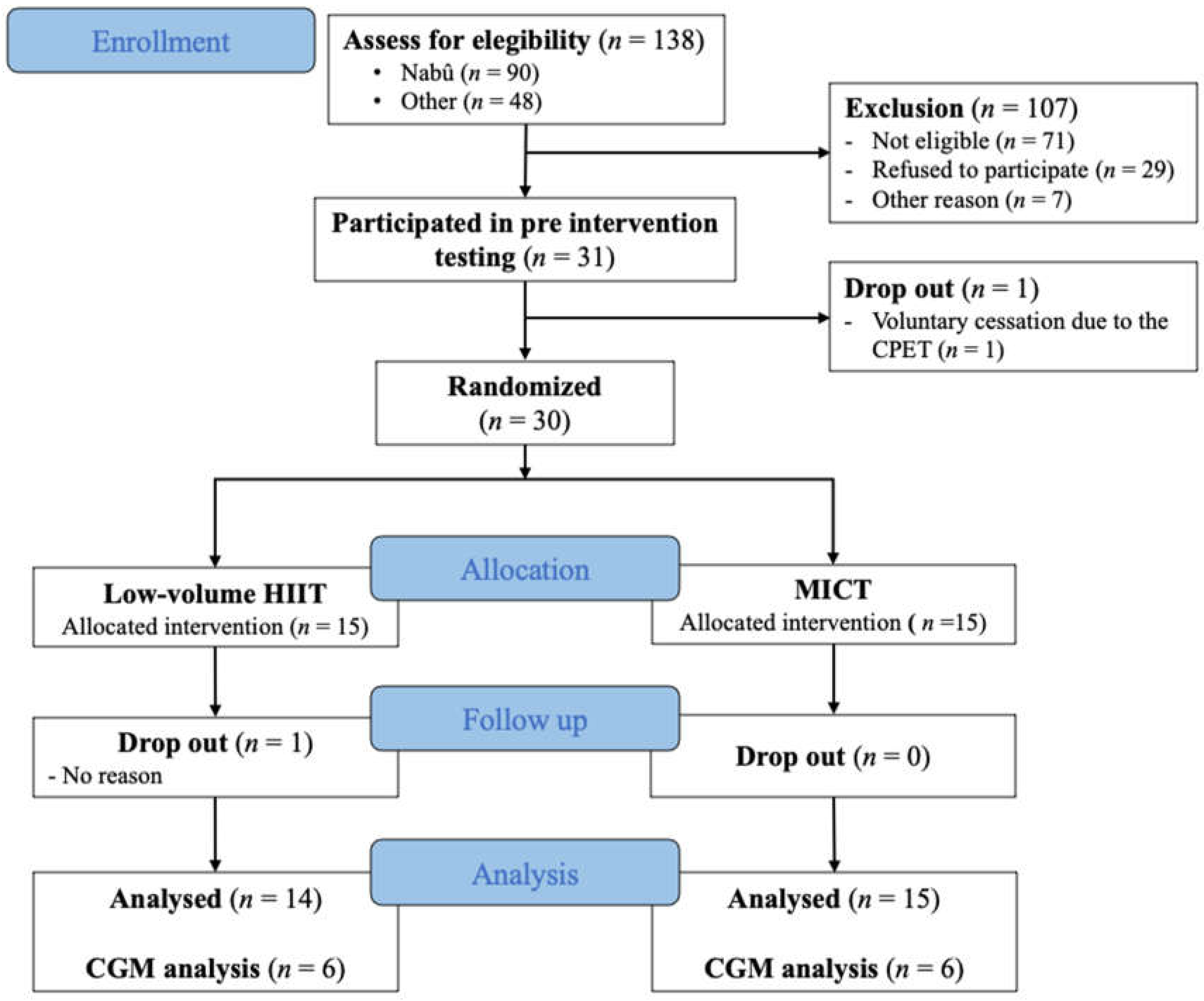
3.1. Recruitment and Participants Characteristics
3.2. Metabolic Profile and Glycemic Control
3.3. Anthropometrics and Body Composition
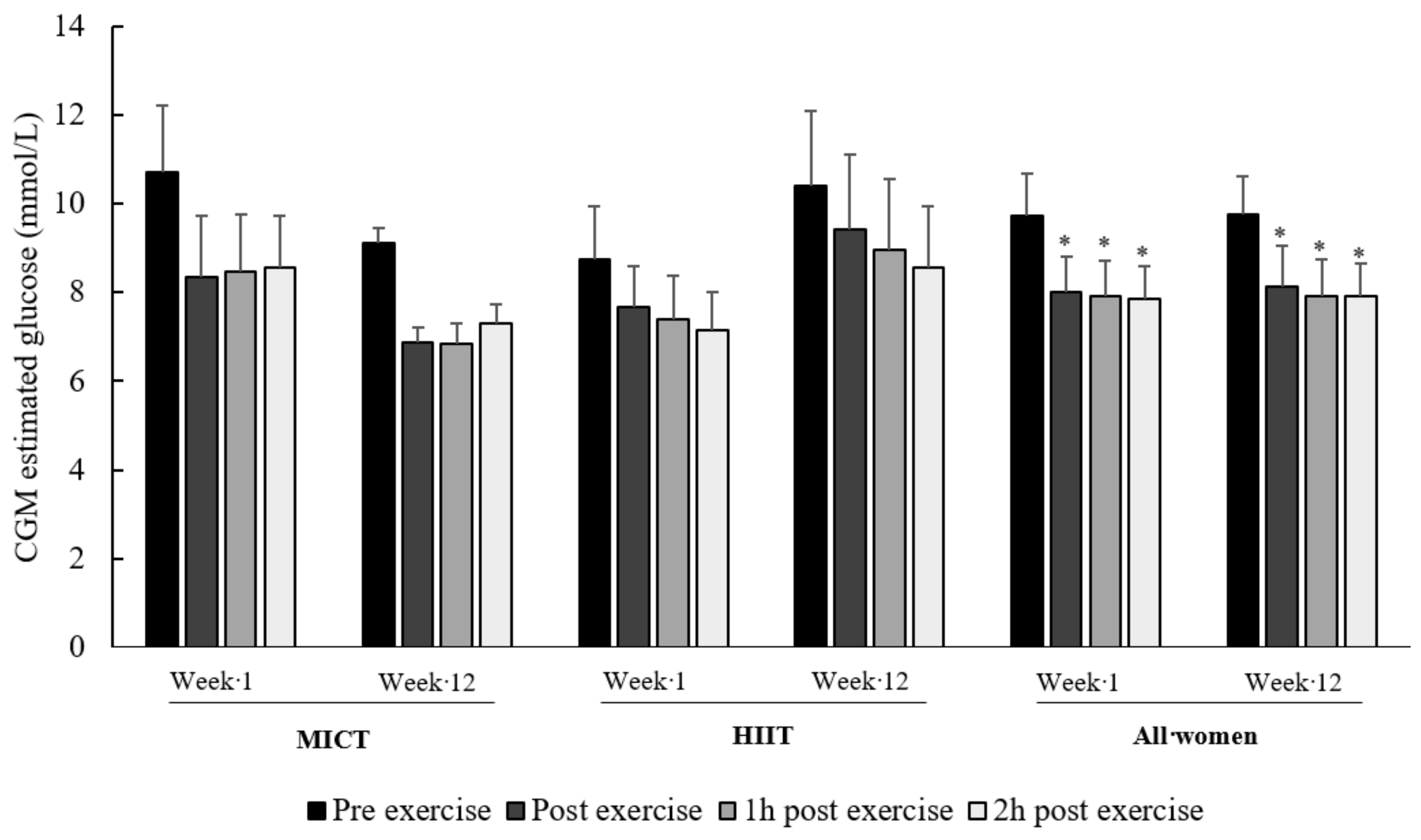
3.4. Acute Effect of Exercise on Continuous Glucose Monitoring
3.5. Capillary Glucose in Response to Exercise
3.6. Energy Intake, Energy Expenditure, and Energy Balance
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cho, N.H.; Shaw, J.E.; Karuranga, S.; Huang, Y.; da Rocha Fernandes, J.D.; Ohlrogge, A.W.; Malanda, B. IDF Diabetes Atlas: Global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Res. Clin. Pract. 2018, 138, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Mauvais-Jarvis, F. Gender differences in glucose homeostasis and diabetes. Physiol. Behav. 2018, 187, 20–23. [Google Scholar] [CrossRef] [PubMed]
- Wexler, D.J.; Grant, R.W.; Meigs, J.B.; Nathan, D.M.; Cagliero, E. Sex disparities in treatment of cardiac risk factors in patients with type 2 diabetes. Diabetes Care 2005, 28, 514–520. [Google Scholar] [CrossRef]
- Sigal, R.J.; Armstrong, M.J.; Bacon, S.L.; Boulé, N.G.; Dasgupta, K.; Kenny, G.P.; Riddell, M.C. Physical Activity and Diabetes. Can. J. Diabetes 2018, 42, S54–S63. [Google Scholar] [CrossRef]
- Association, A.D. Lifestyle management: Standards of medical care in Diabetesd. Diabetes Care 2018, 41, S38–S50. [Google Scholar] [CrossRef]
- Richter, E.A.; Hargreaves, M. Exercise, GLUT4, and skeletal muscle glucose uptake. Physiol. Rev. 2013, 93, 993–1017. [Google Scholar] [CrossRef]
- Holloszy, J.O.; Coyle, E.F. Adaptations of skeletal muscle to endurance exercise and their metabolic consequences. J. Appl. Physiol. Respir. Environ. Exerc. Physiol. 1984, 56, 831–838. [Google Scholar] [CrossRef]
- Pinkney, J.H.; Stehouwer, C.D.A.; Coppack, S.W.; Yudkin, J.S. Endothelial dysfunction: Cause of the insulin resistance syndrome. Diabetes 1997, 46, S9–S13. [Google Scholar] [CrossRef] [PubMed]
- Short, K.R.; Bigelow, M.L.; Kahl, J.; Singh, R.; Coenen-Schimke, J.; Raghavakaimal, S.; Nair, K.S. Decline in skeletal muscle mitochondrial function with aging in humans. Proc. Natl. Acad. Sci. USA 2005, 102, 5618–5623. [Google Scholar] [CrossRef]
- Jelleyman, C.; Yates, T.; O’Donovan, G.; Gray, L.J.; King, J.A.; Khunti, K.; Davies, M.J. The effects of high-intensity interval training on glucose regulation and insulin resistance: A meta-analysis. Obes. Rev. 2015, 16, 942–961. [Google Scholar] [CrossRef]
- De Nardi, A.T.; Tolves, T.; Lenzi, T.L.; Signori, L.U.; Silva, A.M.V. da High-intensity interval training versus continuous training on physiological and metabolic variables in prediabetes and type 2 diabetes: A meta-analysis. Diabetes Res. Clin. Pract. 2018, 137, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Lora-Pozo, I.; Lucena-Anton, D.; Salazar, A.; Galán-Mercant, A.; Moral-Munoz, J.A. Anthropometric, Cardiopulmonary and Metabolic Benefits of the High-Intensity Interval Training Versus Moderate, Low-Intensity or Control for Type 2 Diabetes: Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2019, 16, 4524. [Google Scholar] [CrossRef] [PubMed]
- Boulé, N.G.; Kenny, G.P.; Haddad, E.; Wells, G.A.; Sigal, R.J. Meta-analysis of the effect of structured exercise training on cardiorespiratory fitness in Type 2 diabetes mellitus. Diabetologia 2003, 46, 1071–1081. [Google Scholar]
- Liubaoerjijin, Y.; Terada, T.; Fletcher, K.; Boulé, N.G. Effect of aerobic exercise intensity on glycemic control in type 2 diabetes: A meta-analysis of head-to-head randomized trials. Acta Diabetol. 2016, 53, 769–781. [Google Scholar] [CrossRef] [PubMed]
- Leiter, L.A.; Ceriello, A.; Davidson, J.A.; Hanefeld, M.; Monnier, L.; Owens, D.R.; Tajima, N.; Tuomilehto, J. Postprandial glucose regulation: New data andnew implications. Clin. Ther. 2005, 27, S42–S56. [Google Scholar] [CrossRef] [PubMed]
- Nalysnyk, L.; Hernandez-Medina, M.; Krishnarajah, G. Glycaemic variability and complications in patients with diabetes mellitus: Evidence from a systematic review of the literature. Diabetes Obes. Metab. 2010, 12, 288–298. [Google Scholar] [CrossRef]
- Van Dijk, J.W.; Manders, R.J.F.; Tummers, K.; Bonomi, A.G.; Stehouwer, C.D.A.; Hartgens, F.; Van Loon, L.J.C. Both resistance- and endurance-type exercise reduce the prevalence of hyperglycaemia in individuals with impaired glucose tolerance and in insulin-treated and non-insulin-treated type 2 diabetic patients. Diabetologia 2012, 55, 1273–1282. [Google Scholar] [CrossRef]
- Karstoft, K.; Clark, M.A.; Jakobsen, I.; Müller, I.A.; Pedersen, B.K.; Solomon, T.P.J.; Ried-Larsen, M. The effects of 2 weeks of interval vs continuous walking training on glycaemic control and whole-body oxidative stress in individuals with type 2 diabetes: A controlled, randomised, crossover trial. Diabetologia 2017, 60, 508–517. [Google Scholar] [CrossRef] [PubMed]
- Stolar, M. Glycemic Control and Complications in Type 2 Diabetes Mellitus. Am. J. Med. 2010, 123, S3–S11. [Google Scholar] [CrossRef]
- Ross, R.; Aru, J.; Freeman, J.; Hudson, R.; Janssen, I. Abdominal adiposity and insulin resistance in obese men. Am. J. Physiol. Endocrinol. Metab. 2002, 282, 657–663. [Google Scholar] [CrossRef]
- Sievenpiper, J.L.; Chan, C.B.; Dworatzek, P.D.; Med, C.F.; Williams Med, S.L. 2018 Clinical Practice Guidelines Nutrition Therapy Diabetes Canada Clinical Practice Guidelines Expert Committee. Can. J. Diabetes 2018, 48, 896–902. [Google Scholar]
- Kaminsky, L.A.; Whaley, M.H. Evaluation of a New Standardized Ramp Protocol: The BSU/Bruc Ramp protocol. J. Cardiopulm. Rehabil. Prev. 1998, 18, 438–444. [Google Scholar] [CrossRef] [PubMed]
- Riebe, D.; Ehrman, J.K.; Liguori, G.; Magal, M. ACSM’s Guidelines for Exercise Testing and Prescription—American College of Sports Medicine, 10th ed.; Kluwer, W., Ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2018. [Google Scholar]
- Higgins, P.B.; Comuzzie, A.G. Measures of waist circumference. In Handbook of Anthropometry: Physical Measures of Human Form in Health and Disease; Springer: New York, NY, USA, 2012; pp. 881–891. ISBN 9781441917881. [Google Scholar]
- Levy, J.C.; Matthews, D.R.; Hermans, M.E. Correct homeostasis model assessment (HOMA) evaluation uses the computer program. Diabetes Care. 1998, 21, 2191–2192. [Google Scholar] [CrossRef]
- Battelino, T.; Danne, T.; Bergenstal, R.M.; Amiel, S.A.; Beck, R.; Biester, T.; Bosi, E.; Buckingham, B.A.; Cefalu, W.T.; Close, K.L.; et al. Clinical targets for continuous glucose monitoring data interpretation: Recommendations from the international consensus on time in range. Diabetes Care 2019, 42, 1593–1603. [Google Scholar] [CrossRef]
- Rodbard, D. Display of glucose distributions by date, time of day, and day of week: New and improved methods. J. Diabetes Sci. Technol. 2009, 3, 1388–1394. [Google Scholar] [CrossRef]
- Lührmann, P.M.; Herbert, B.M.; Gaster, C.; Neuhäuser-Berthold, M. Validation of a Self-Administered 3-Day Estimated Dietary Record for Use in the Elderly. Eur. J. Nutr. 1999, 38, 235–240. [Google Scholar] [PubMed]
- Hooker, S.P.; Feeney, A.; Hutto, B.; Pfeiffer, K.A.; McIver, K.; Heil, D.P.; Vena, J.E.; LaMonte, M.J.; Blair, S.N. Validation of the actical activity monitor in middle-aged and older adults. J. Phys. Act. Health 2011, 8, 372–381. [Google Scholar] [CrossRef][Green Version]
- Bouchard, C.; Tremblay, A.; Leblanc, C.; Lortie, G.; Savard, R.; Thériault, G. A method to assess energy expenditure in children and adults. Am. J. Clin. Nutr. 1983, 37, 461–467. [Google Scholar] [CrossRef]
- Washburn, R.A.; Smith, K.W.; Jette, A.M.; Janney, C.A. The physical activity scale for the elderly (PASE): Development and evaluation. J. Clin. Epidemiol. 1993, 46, 153–162. [Google Scholar] [CrossRef]
- Szanton, S.L.; Walker, R.K.; Roberts, L.; Thorpe, R.J.; Wolff, J.; Agree, E.; Roth, D.L.; Gitlin, L.N.; Seplaki, C. Older adults’ favorite activities are resoundingly active: Findings from the NHATS study. Geriatr. Nurs. 2015, 36, 131–135. [Google Scholar] [CrossRef]
- Mitranun, W.; Deerochanawong, C.; Tanaka, H.; Suksom, D. Continuous vs interval training on glycemic control and macro- and microvascular reactivity in type 2 diabetic patients. Scand. J. Med. Sci. Sports 2014, 24, e69–e76. [Google Scholar] [CrossRef]
- Terada, T.; Wilson, B.J.; Myette-Cóté, E.; Kuzik, N.; Bell, G.J.; McCargar, L.J.; Boulé, N.G. Targeting specific interstitial glycemic parameters with high-intensity interval exercise and fasted-state exercise in type 2 diabetes. Metabolism 2016, 65, 599–608. [Google Scholar] [CrossRef] [PubMed]
- Rosethal, R.; Cooper, H.; Hedges, L. Parametric measures of effect size. The Handbook of Research Synthesis 1994, 621, 232–244. [Google Scholar]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences. Academic press 2013. [Google Scholar]
- Curran-Everett, D. Multiple comparisons: Philosophies and illustrations. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2000, 279, R1–R8. [Google Scholar] [CrossRef]
- Wasserstein, R.L.; Schirm, A.L.; Lazar, N.A. Moving to a World Beyond “p < 0.05”. Am. Stat. 2019, 73, 1–19. [Google Scholar]
- Houmard, J.A.; Tanner, C.J.; Slentz, C.A.; Duscha, B.D.; McCartney, J.S.; Kraus, W.E. Effect of the volume and intensity of exercise training on insulin sensitivity. J. Appl. Physiol. 2004, 96, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Winding, K.M.; Munch, G.W.; Iepsen, U.W.; Van Hall, G.; Pedersen, B.K.; Mortensen, S.P. The effect on glycaemic control of low-volume high-intensity interval training versus endurance training in individuals with type 2 diabetes. Diabetes Obes. Metab. 2018, 20, 1131–1139. [Google Scholar] [CrossRef] [PubMed]
- Wewege, M.; van den Berg, R.; Ward, R.E.; Keech, A. The effects of high-intensity interval training vs. moderate-intensity continuous training on body composition in overweight and obese adults: A systematic review and meta-analysis. Obes. Rev. 2017, 18, 635–646. [Google Scholar] [CrossRef]
- Maillard, F.; Pereira, B.; Boisseau, N. Effect of High-Intensity Interval Training on Total, Abdominal and Visceral Fat Mass: A Meta-Analysis. Sport. Med. 2018, 48, 269–288. [Google Scholar] [CrossRef]
- Boukabous, I.; Marcotte-Chénard, A.; Amamou, T.; Boulay, P.; Brochu, M.; Tessier, D.; Dionne, I.; Riesco, E. Low-volume high-intensity interval training versus moderate-intensity continuous training on body composition, cardiometabolic profile, and physical capacity in older women. J. Aging Phys. Act. 2019, 27, 879–889. [Google Scholar] [CrossRef] [PubMed]
- Rees, J.L.; Chang, C.R.; François, M.E.; Marcotte-Chénard, A.; Fontvieille, A.; Klaprat, N.D.; Dyck, R.A.; Funk, D.R.; Snydmiller, G.; Bastell, K.; et al. Minimal effect of walking before dinner on glycemic responses in type 2 diabetes: Outcomes from the multi-site E-PAraDiGM study. Acta Diabetol. 2019, 56, 755–765. [Google Scholar] [CrossRef] [PubMed]
- Haxhi, J.; Leto, G.; Palumbo, A.; Sbriccoli, P.; Guidetti, L.; Fantini, C.; Buzzetti, R.; Caporossi, D.; Luigi, L.; Sacchetti, M.; et al. Exercise at lunchtime: Effect on glycemic control and oxidative stress in middle-aged men with type 2 diabetes. Eur. J. Appl. Physiol. 2016, 116, 573–582. [Google Scholar] [CrossRef]
- Karstoft, K.; Christensen, C.S.; Pedersen, B.K.; Solomon, T.P.J. The acute effects of interval- Vs continuous-walking exercise on glycemic control in subjects with type 2 diabetes: A crossover, controlled study. J. Clin. Endocrinol. Metab. 2014, 99, 3334–3342. [Google Scholar] [CrossRef] [PubMed]
- Godkin, F.E.; Jenkins, E.M.; Little, J.P.; Nazarali, Z.; Percival, M.E.; Gibala, M.J. The effect of brief intermittent stair climbing on glycemic control in people with type 2 diabetes: A pilot study. Appl. Physiol. Nutr. Metab. 2018, 43, 969–972. [Google Scholar] [CrossRef] [PubMed]
- Figueira, F.R.; Umpierre, D.; Casali, K.R.; Tetelbom, P.S.; Henn, N.T.; Ribeiro, J.P.; Schaan, B.D. Aerobic and combined exercise sessions reduce glucose variability in type 2 diabetes: Crossover randomized trial. PLoS ONE 2013, 8, e57733. [Google Scholar] [CrossRef]
- Munan, M.; Oliveira, C.L.P.; Marcotte-Chénard, A.; Rees, J.L.; Prado, C.M.; Riesco, E.; Boulé, N.G. Acute and Chronic Effects of Exercise on Continuous Glucose Monitoring Outcomes in Type 2 Diabetes: A Meta-Analysis. Front. Endocrinol. 2020, 11, 495. [Google Scholar] [CrossRef] [PubMed]
- Merino, J.; Leong, A.; Posner, D.C.; Porneala, B.; Masana, L.; Dupuis, J.; Florez, J.C. Genetically driven hyperglycemia increases risk of coronary artery disease separately from type 2 diabetes. Diabetes Care. 2017, 40, 687–693. [Google Scholar] [CrossRef]
- Monnier, L.; Colette, C.; Rabasa-Lhoret, R.; Lapinski, H.; Caubel, C.; Avignon, A.; Boniface, H. Morning hyperglycemic excursions: A constant failure in the metabolic control of non-insulin-using patients with type 2 diabetes. Diabetes Care. 2002, 25, 737–741. [Google Scholar] [CrossRef]
- Hanefeld, M.; Fischer, S.; Julius, U.; Schulze, J.; Schwanebeck, U.; Schmechel, H.; Ziegelasch, H.J.; Lindner, J. Risk factors for myocardial infarction and death in newly detected NIDDM: The Diabetes Intervention Study, 11-year follow-up. Diabetologia 1996, 39, 1577–1583. [Google Scholar] [CrossRef]
- Sievers, M.L.; Bennett, P.H.; Nelson, R.G. Effect of glycemia on mortality in Pima Indians with type 2 diabetes. Diabetes 1999, 48, 896–902. [Google Scholar] [CrossRef] [PubMed]
- Ottinger, M.A. A Comparative Approach to Metabolic Aspects of Aging: Conserved Mechanisms and Effects of Calorie Restriction and Environment. Prog. Mol. Biol. Transl. Sci. 2018, 155, 109–127. [Google Scholar] [PubMed]
- Gibala, M.J.; Gillen, J.B.; Percival, M.E. Physiological and Health-Related Adaptations to Low-Volume Interval Training: Influences of Nutrition and Sex. Sport. Med. 2014, 44, 127–137. [Google Scholar] [CrossRef] [PubMed]

| Variables | Total Sample | CGM Sample | ||
|---|---|---|---|---|
| HIIT (n = 14) | MICT (n = 15) | HIIT (n = 6) | MICT (n = 6) | |
| Age (years) | 67.1 (63.0–70.6) | 68.1 (64.1–70.4) | 66.2 (63.0–70.3) | 63.9 (62.55–69.55) |
| Duration of T2D (years) | 10.0 (5.1–15.1) | 10.0 (3.5–12.0) | 7.5 (2.4–17.5) | 10.0 (3.0–11.5) |
| BMI (kg/m2) | 31.4 (28.0–37.0) | 32.4 (27.9–40.2) | 31.2 (28.2–44.5) | 35.0 (28.0–44.0) |
| Waist circumference (cm) | 105.3 (99.3–109.9) | 109.8 (96.4–119.3) | 99.1 (91.7–120.1) | 109.7 (98.5–122.5) |
| Fasting glucose (mmol/L) | 6.8 (5.9–8.0) | 8.2 (5.9–9.3) | 6.7 (5.7–7.7) | 8.8 (7.5–11.9) |
| A1c (%) | 6.5 (6.1–7.2) | 6.9 (6.4–7.5) † | 6.3 (5.9–7.7) | 7.5 (6.9–8.2) † |
| Systolic BP (mmHg) | 128 (114–134) | 130 (123–136) | 122 (114–139) | 125 (111–144) |
| Diastolic BP (mmHg) | 77 (71–78) | 78 (73–82) | 77 (74–79) | 80 (74–83) |
| VO2 peak (mL/kg/min) | 18.0 (15.5–20.6) | 17.8 (15.5–21.2) | 20.6 (15.3–21.8) | 16.9 (13.1–22.0) |
| Medication | 5.5 (4.0–8.5) | 6.0 (4.0–7.0) | 6.5 (4.0–9.8) | 6.5 (4.0–7.25) |
| Glucose lowering medication (n (%)) | ||||
| Metformin | 10 (71) | 10 (67) | 4 (66) | 4 (66) |
| Sulfonylurea | 2 (14) | 4 (27) | 1 (17) | 2 (33) |
| DPP-4 inhibitor | 6 (43) | 6 (40) | 2 (33) | 3 (50) |
| SGLT2 inhibitor | 2 (14) | 1 (7) | 0 (0) | 1 (17) |
| Thiazolidinediones | 1 (7) | 0 (0) | 0 (0) | 0 (0) |
| Hypotensive medication n (%) | ||||
| Calcium channel blocker | 2 (14) | 4 (27) | 2 (33) | 2 (33) |
| ACE inhibitor | 11 (79) | 13 (87) | 5 (83) | 6 (100) |
| Diuretic | 7 (47) | 6 (43) | 4 (67) | 3 (50) |
| Beta blocker | 4 (29) | 2 (13) | 3 (50) | 2 (33) |
| Lipid lowering medication n (%) | ||||
| Statin | 12 (86) | 10 (67) | 4 (66) | 5 (83) |
| Variables | Before | After | Time Effect p-value | Effect Size (r) | |
|---|---|---|---|---|---|
| A1c (%) | HIIT | 6.5 (6.0–7.5) | 6.3 (5.8–7.4) | 0.411 | 0.15 |
| MICT | 7.0 (6.4–7.5) | 6.7 (5.9–7.2) | 0.014 | 0.45 | |
| Fasting glucose (mmol/L) | HIIT | 7.0 (5.8–8.2) | 7.1 (5.5–7.7) | 0.373 | 0.16 |
| MICT | 8.2 (5.9–9.5) | 6.7 (5.0–8.2) | 0.001 | 0.59 | |
| Fasting insulin (pmol/L) | HIIT | 81.0 (46.0–176.5) | 62.0 (43.5–197.5) | 0.328 | 0.18 |
| MICT | 95.0 (68.0–139.5) | 67.0 (47.5–116.0) | 0.116 | 0.29 | |
| HOMA2-IR | HIIT | 1.54 (0.91–1.96) | 1.28 (0.91–3.91) | 0.695 | 0.07 |
| MICT | 1.98 (1.21–2.37) | 1.40 (1.12–2.27) | 0.116 | 0.29 |
| Variables | Before | After | p-value Time | p-value Time x Group | |
|---|---|---|---|---|---|
| Weight (kg) | HIIT | 78.7 (69.4–90.7) | 74.9 (70.2–90.5) | 0.025 | 0.403 |
| MICT | 77.1 (66.2–97.5) | 74.8 (65.5–94.8) | |||
| BMI (kg/m2) | HIIT | 31.4 (28.0–37.0) | 30.6 (27.5–36.9) | 0.057 | 0.263 |
| MICT | 32.4 (27.9–40.2) | 29.7 (27.7–39.7) | |||
| WC (cm) | HIIT | 105.3 (99.3–109.9) | 101.0 (97.9–109.1) | 0.150 | 0.431 |
| MICT | 109.8 (96.4–119.3) | 109.5 (96.8–122.0) | |||
| Total FM (kg) | HIIT | 32.1 (26.9–42.3) | 29.4 (26.1–41.0) | 0.007 | 0.799 |
| MICT | 33.6 (26.4–43.6) | 31.6 (26.2–44.0) | |||
| Visceral FM (kg) | HIIT | 1.66 (1.38–2.04) | 1.49 (1.07–1.96) | 0.299 | 0.686 |
| MICT | 1.77 (1.42–2.29) | 1.55 (1.38–2.39) | |||
| Total LBM (kg) | HIIT | 42.6 (38.0–45.2) | 42.4 (38.9–44.7) | 0.756 | 0.886 |
| MICT | 41.0 (36.6–45.2) | 41.3 (36.7–46.0) | |||
| App. LBM (kg) | HIIT | 19.9 (18.1–21.3) | 20.0 (18.0–21.1) | 0.035 | 0.510 |
| MICT | 18.8 (16.4–21.4) | 19.1 (17.0–21.4) |
| Week 1 | Week 12 | ||||
|---|---|---|---|---|---|
| MICT | HIIT | MICT | HIIT | ||
| Mean 24-h glucose (mmol/L) | CONT (Day 1) | 8.7 (6.9–10.3) | 6.7 (6.2–9.2) | 8.1 (7.2–8.7) | 6.7 (6.4–11.2) |
| EX (Day 2) | 8.4 (7.4–9.5) | 6.6 (5.7–8.9) | 8.3 (7.2–9.0) | 6.6 (5.6–10.8) * | |
| Post-EX (Day 3) | 8.6 (6.9–9.1) | 7.1 (5.3–9.3) | 8.4 (7.5–8.5) | 7.5 (5.8–11.5) | |
| Peak glucose (mmol/L) | CONT (Day 1) | 12.4 (9.7–15.9) | 9.6 (8.6–14.0) | 11.5 (9.6–12.6) | 9.8 (8.9–16.4) |
| EX (Day 2) | 11.6 (9.8–14.6) | 9.1 (8.3–14.1) | 11.3 (10.3–12.6) | 9.3 (7.9–16.1) * | |
| Post-EX (Day 3) | 12.9 (10.5–14.1) | 10.3 (9.9–14.5) | 11.4 (10.0–11.9) | 10.0 (8.6–16.9) | |
| Mild-Hyperglycemia (> 8.3 mmol/L; min) | CONT (Day 1) | 813 (204–1237) | 143 (19–776) | 548 (244–749) | 125 (54–1432) |
| EX (Day 2) | 577 (368–980) | 68 (19–591) | 623 (325–859) | 70 (28–1168) * | |
| Post-EX (Day 3) | 640 (187–911) | 490 (65–963) | 580 (394–859) | 305 (25–1358) | |
| Hyperglycemia (>10 mmol/L; min) | CONT (Day 1) | 225 (0–670) | 25 (0–545) | 160 (0–289) | 0 (0–1036) |
| EX (Day 2) | 170 (4–520) | 0 (0–428) | 173 (39–494) | 0 (0–888) | |
| Post-EX (Day 3) | 235 (41–346) | 23 (0–441) | 95 (45–178) | 10 (0–973) | |
| SD (mmol/L) | CONT (Day 1) | 1.31 (0.98–2.08) | 1.16 (0.71–1.84) | 1.21 (0.79–1.77) | 1.07 (0.71–1.51) |
| EX (Day 2) | 1.31 (0.99–2.06) | 0.81 (0.57–1.76) | 1.39 (1.12–1.88) | 1.58 (0.78–2.51) | |
| Post-EX (Day 3) | 1.35 (1.17–1.63) | 1.35 (0.93–1.93) | 1.05 (0.95–1.44) | 0.94 (0.75–2.14) | |
| CONGA1 (mmol/L) | CONT (Day 1) | 8.02 (6.25–9.31) | 6.06 (5.41–8.81) | 7.50 (6.53–7.90) | 6.20 (5.88–10.16) |
| EX (Day 2) | 7.70 (6.66–8.81) | 6.18 (5.34–8.16) | 7.72 (6.70–8.18) | 6.14 (5.13–9.85) | |
| Post-EX (Day 3) | 7.81 (6.17–8.20) | 6.50 (4.46–8.43) | 7.39 (6.69–7.55) | 6.79 (5.30–10.72) | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marcotte-Chénard, A.; Tremblay, D.; Mony, M.-M.; Boulay, P.; Brochu, M.; Morais, J.A.; Dionne, I.J.; Langlois, M.-F.; Mampuya, W.M.; Tessier, D.M.; et al. Acute and Chronic Effects of Low-Volume High-Intensity Interval Training Compared to Moderate-Intensity Continuous Training on Glycemic Control and Body Composition in Older Women with Type 2 Diabetes. Obesities 2021, 1, 72-87. https://doi.org/10.3390/Obesities1020007
Marcotte-Chénard A, Tremblay D, Mony M-M, Boulay P, Brochu M, Morais JA, Dionne IJ, Langlois M-F, Mampuya WM, Tessier DM, et al. Acute and Chronic Effects of Low-Volume High-Intensity Interval Training Compared to Moderate-Intensity Continuous Training on Glycemic Control and Body Composition in Older Women with Type 2 Diabetes. Obesities. 2021; 1(2):72-87. https://doi.org/10.3390/Obesities1020007
Chicago/Turabian StyleMarcotte-Chénard, Alexis, Dominic Tremblay, Marie-Michelle Mony, Pierre Boulay, Martin Brochu, José A. Morais, Isabelle J. Dionne, Marie-France Langlois, Warner M. Mampuya, Daniel M. Tessier, and et al. 2021. "Acute and Chronic Effects of Low-Volume High-Intensity Interval Training Compared to Moderate-Intensity Continuous Training on Glycemic Control and Body Composition in Older Women with Type 2 Diabetes" Obesities 1, no. 2: 72-87. https://doi.org/10.3390/Obesities1020007
APA StyleMarcotte-Chénard, A., Tremblay, D., Mony, M.-M., Boulay, P., Brochu, M., Morais, J. A., Dionne, I. J., Langlois, M.-F., Mampuya, W. M., Tessier, D. M., Boulé, N. G., & Riesco, E. (2021). Acute and Chronic Effects of Low-Volume High-Intensity Interval Training Compared to Moderate-Intensity Continuous Training on Glycemic Control and Body Composition in Older Women with Type 2 Diabetes. Obesities, 1(2), 72-87. https://doi.org/10.3390/Obesities1020007






