Assessment of Groundwater Contamination in the Southeastern Coast of Brazil: A Potential Threat to Human Health in Marica Municipality
Abstract
:1. Introduction
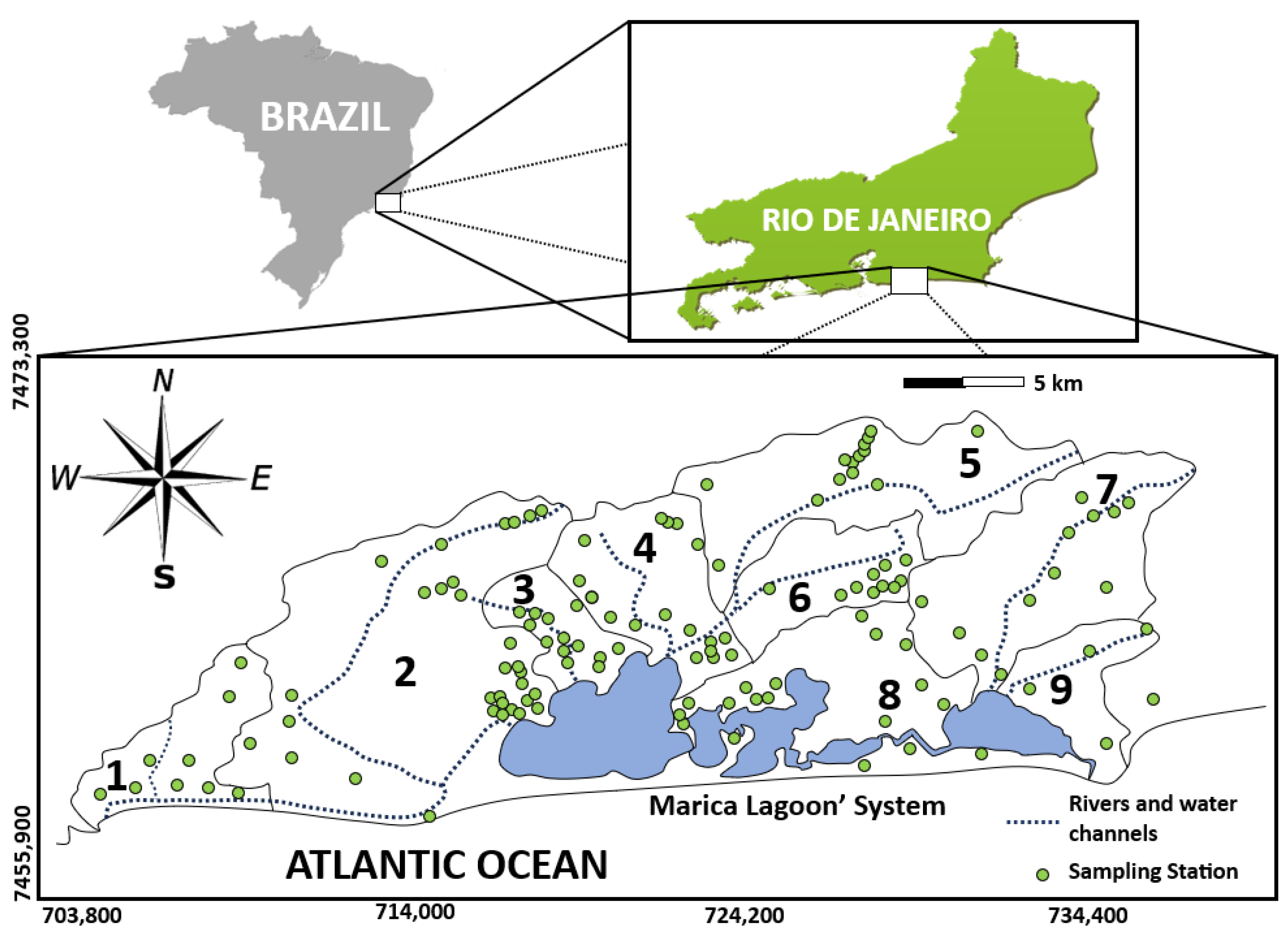
2. Study Site
3. Methodology
4. Results and Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ullah, Z.; Rashid, A.; Ghani, J.; Nawab, J.; Zeng, X.-C.; Shah, M.; Alrefaei, A.F.; Kamel, M.; Aleya, L.; Abdel-Daim, M.M.; et al. Groundwater Contamination through Potentially Harmful Metals and Its Implications in Groundwater Management. Front. Environ. Sci. 2022, 10, 1021596. [Google Scholar] [CrossRef]
- International Association of Hydrogeologists. Groundwater—More about the Hidden Resource; International Association of Hydrogeologists: London, UK, 2020. [Google Scholar]
- Li, P.; Tian, R.; Xue, C.; Wu, J. Progress, Opportunities, and Key Fields for Groundwater Quality Research under the Impacts of Human Activities in China with a Special Focus on Western China. Environ. Sci. Pollut. Res. 2017, 24, 13224–13234. [Google Scholar] [CrossRef]
- Velis, M.; Conti, K.I.; Biermann, F. Groundwater and Human Development: Synergies and Trade-Offs within the Context of the Sustainable Development Goals. Sustain. Sci. 2017, 12, 1007–1017. [Google Scholar] [CrossRef]
- Mazhar, S.; Pellegrini, E.; Contin, M.; Bravo, C.; De Nobili, M. Impacts of Salinization Caused by Sea Level Rise on the Biological Processes of Coastal Soils—A Review. Front. Environ. Sci. 2022, 10, 909415. [Google Scholar] [CrossRef]
- Saalidong, B.M.; Aram, S.A.; Otu, S.; Lartey, P.O. Examining the Dynamics of the Relationship between Water PH and Other Water Quality Parameters in Ground and Surface Water Systems. PLoS ONE 2022, 17, e0262117. [Google Scholar] [CrossRef]
- Li, P. To Make the Water Safer. Expo. Health 2020, 12, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Wu, J. Sustainable Living with Risks: Meeting the Challenges. Human. Ecol. Risk Assess. Int. J. 2019, 25, 1–10. [Google Scholar] [CrossRef]
- Ugochukwu, U.C.; Ochonogor, A. Groundwater Contamination by Polycyclic Aromatic Hydrocarbon Due to Diesel Spill from a Telecom Base Station in a Nigerian City: Assessment of Human Health Risk Exposure. Environ. Monit. Assess. 2018, 190, 249. [Google Scholar] [CrossRef] [PubMed]
- Marić, N.; Štrbački, J.; Mrazovac Kurilić, S.; Beškoski, V.P.; Nikić, Z.; Ignjatović, S.; Malbašić, J. Hydrochemistry of Groundwater Contaminated by Petroleum Hydrocarbons: The Impact of Biodegradation (Vitanovac, Serbia). Environ. Geochem. Health 2020, 42, 1921–1935. [Google Scholar] [CrossRef]
- Nivetha, C.; Deepika, T.; Arjunan, A.; Sivalingam, P.; Revathi, N.; Muthuselvam, M. Antimicrobial and Antioxidant Activities of Streptomyces Sps Isolated from Muthupettai Mangrove Soil. J. Pharm. Res. Int. 2021, 33, 210–234. [Google Scholar] [CrossRef]
- Li, P.; Karunanidhi, D.; Subramani, T.; Srinivasamoorthy, K. Sources and Consequences of Groundwater Contamination. Arch. Environ. Contam. Toxicol. 2021, 80, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Monteiro da Fonseca, E.; Machado Publio, M.C.; de Freitas Delgado, J. Segurança Hídrica. Sist. Gestão 2023, 18, 2. [Google Scholar] [CrossRef]
- Basu, A.; Saha, D.; Saha, R.; Ghosh, T.; Saha, B. A Review on Sources, Toxicity and Remediation Technologies for Removing Arsenic from Drinking Water. Res. Chem. Intermed. 2014, 40, 447–485. [Google Scholar] [CrossRef]
- Pandey, H.K.; Duggal, S.K.; Jamatia, A. Fluoride Contamination of Groundwater and It’s Hydrogeological Evolution in District Sonbhadra (U.P.) India. Proc. Natl. Acad. Sci. India Sect. A Phys. Sci. 2016, 86, 81–93. [Google Scholar] [CrossRef]
- Subba Rao, N.; Ravindra, B.; Wu, J. Geochemical and Health Risk Evaluation of Fluoride Rich Groundwater in Sattenapalle Region, Guntur District, Andhra Pradesh, India. Human. Ecol. Risk Assess. Int. J. 2020, 26, 2316–2348. [Google Scholar] [CrossRef]
- Clement, M.; Meunie, A. Is Inequality Harmful for the Environment? An Empirical Analysis Applied to Developing and Transition Countries. Rev. Soc. Econ. 2010, 68, 413–445. [Google Scholar] [CrossRef]
- Hayashi, A.; Akimoto, K.; Tomoda, T.; Kii, M. Global Evaluation of the Effects of Agriculture and Water Management Adaptations on the Water-Stressed Population. Mitig. Adapt. Strateg. Glob. Change 2013, 18, 591–618. [Google Scholar] [CrossRef]
- Lam, S.; Nguyen-Viet, H.; Tuyet-Hanh, T.T.; Nguyen-Mai, H.; Harper, S. Evidence for Public Health Risks of Wastewater and Excreta Management Practices in Southeast Asia: A Scoping Review. Int. J. Environ. Res. Public Health 2015, 12, 12863–12885. [Google Scholar] [CrossRef]
- Bertrand, G.; Hirata, R.; Pauwels, H.; Cary, L.; Petelet-Giraud, E.; Chatton, E.; Aquilina, L.; Labasque, T.; Martins, V.; Montenegro, S.; et al. Groundwater Contamination in Coastal Urban Areas: Anthropogenic Pressure and Natural Attenuation Processes. Example of Recife (PE State, NE Brazil). J. Contam. Hydrol. 2016, 192, 165–180. [Google Scholar] [CrossRef]
- Gomes, O.V.O.; Marques, E.D.; Kütter, V.T.; Aires, J.R.; Travi, Y.; Silva-Filho, E.V. Origin of Salinity and Hydrogeochemical Features of Porous Aquifers from Northeastern Guanabara Bay, Rio de Janeiro, SE, Brazil. J. Hydrol. Reg. Stud. 2019, 22, 100601. [Google Scholar] [CrossRef]
- Nicolodi, J.L.; Pettermann, R.M. Vulnerability of the Brazilian Coastal Zone in Its Environmental, Social, and Technological Aspects. J. Coast. Res. 2011, 64, 1372–1379. [Google Scholar]
- Filgueira, J.M.; Pereira Júnior, A.O.; de Araújo, R.S.B.; da Silva, N.F. Economic and Social Impacts of the Oil Industry on the Brazilian Onshore. Energies 2020, 13, 1922. [Google Scholar] [CrossRef]
- Da Silva, A.L.C.; da Silva, M.A.M.; Gambôa, L.A.P.; Rodrigues, A.R. Sedimentary Architecture and Depositional Evolution of the Quaternary Coastal Plain of Maricá, Rio de Janeiro, Brazil. Braz. J. Geol. 2014, 44, 191–206. [Google Scholar] [CrossRef]
- Cruz, A.; da Silva, G.C., Jr.; de Almeida, G.M. Modelagem Hidrogeoquímica Do Aquífero Freático Da Restinga de Piratininga, Niterói-RJ. In XIV Congresso Brasileiro de Águas Subterrâneas; ABAS: Curitiba, Brazil, 2006; pp. 1–19. [Google Scholar]
- GB/T 14848-2017; Standard for Groundwater Quality. Standardization Administration of the PRC: Beijing, China, 2017.
- Van Hall, C.E.; Safranko, J.; Stenger, V.A. Rapid Combustion Method for the Determination of Organic Substances in Aqueous Solutions. Anal. Chem. 1963, 35, 315–319. [Google Scholar] [CrossRef]
- Sharp, J.H.; Peltzer, E.T.; Alperin, M.J.; Cauwet, G.; Farrington, J.W.; Fry, B.; Karl, D.M.; Martin, J.H.; Spitzy, A.; Tugrul, S.; et al. Procedures Subgroup Report. Mar. Chem. 1993, 41, 37–49. [Google Scholar] [CrossRef]
- EPA. Method 1603: Escherichia Coli (E. Coli) in Water by Membrane Filtration Using Modified Membrane-Thermotolerant Escherichia Coli Agar (Modified MTEC); EPA: Washington, DC, USA, 2014. [Google Scholar]
- Srivastav, A.L.; Ranjan, M. Inorganic Water Pollutants. In Inorganic Pollutants in Water; Elsevier: Amsterdam, The Netherlands, 2020; pp. 1–15. [Google Scholar] [CrossRef]
- Block, S.S. Desinfection, Sterilization, and Preservation, 5th ed.; Lippincot Willians & Wilkins: Philadelphia, PA, USA, 2001. [Google Scholar]
- Puri, A.; Kumar, M. A Review of Permissible Limits of Drinking Water. Indian. J. Occup. Environ. Med. 2012, 16, 40–44. [Google Scholar] [CrossRef]
- Banks, D. An Introduction to Thermogeology: Ground Source Heating and Cooling, 2nd ed.; John Wiley & Sons, Ltd: Oxford, UK, 2008. [Google Scholar]
- Bonte, M.; Stuyfzand, P.J.; Hulsmann, A.; Van Beelen, P. Underground Thermal Energy Storage: Environmental Risks and Policy Developments in the Netherlands and European Union. Ecol. Soc. 2011, 16, 1–22. [Google Scholar] [CrossRef]
- Griffioen, J.; Appelo, A.J. Nature and Extent of Carbonate Precipitation during Aquifer Thermal Energy Storage. Appl. Geochem. 1993, 8, 161–176. [Google Scholar] [CrossRef]
- Bonte, M.; Van Breukelen, B.M.; Stuyfzand, P.J. Environmental Impacts of Aquifer Thermal Energy Storage Investigated by Field and Laboratory Experiments. J. Water Clim. Change 2013, 4, 77–89. [Google Scholar] [CrossRef]
- Bonte, M.; van Breukelen, B.M.; Stuyfzand, P.J. Temperature-Induced Impacts on Groundwater Quality and Arsenic Mobility in Anoxic Aquifer Sediments Used for Both Drinking Water and Shallow Geothermal Energy Production. Water Res. 2013, 47, 5088–5100. [Google Scholar] [CrossRef]
- Bonte, M.; Röling, W.F.M.; Zaura, E.; van der Wielen, P.W.J.J.; Stuyfzand, P.J.; van Breukelen, B.M. Impacts of Shallow Geothermal Energy Production on Redox Processes and Microbial Communities. Environ. Sci. Technol. 2013, 47, 14476–14484. [Google Scholar] [CrossRef] [PubMed]
- Jesußek, A.; Köber, R.; Grandel, S.; Dahmke, A. Aquifer Heat Storage: Sulphate Reduction with Acetate at Increased Temperatures. Environ. Earth Sci. 2013, 69, 1763–1771. [Google Scholar] [CrossRef]
- Fraser, D.W.; Tsai, T.R.; Orenstein, W.; Parkin, W.E.; Beecham, H.J.; Sharrar, R.G.; Harris, J.; Mallison, G.F.; Martin, S.M.; McDade, J.E.; et al. Legionnaires’ Disease. N. Engl. J. Med. 1977, 297, 1189–1197. [Google Scholar] [CrossRef] [PubMed]
- Shands, K.N. Potable Water as a Source of Legionnaires’ Disease. JAMA J. Am. Med. Assoc. 1985, 253, 1412. [Google Scholar] [CrossRef]
- WHO. WHO Housing and Health Guidelines; WHO: Geneva, Switzerland, 2018. [Google Scholar]
- Weiner, E.R. Applications of Environmental Aquatic Chemistry, 3rd ed.; CRC Press: Boca Raton, FL, USA, 2013. [Google Scholar]
- Hayton, J. Industrial Water Treatment Process Technology; Butterworth-Heinemann: Oxford, UK, 2017. [Google Scholar]
- Rose, S.; Long, A. Monitoring Dissolved Oxygen in Ground Water: Some Basic Considerations. Groundw. Monit. Remediat. 1988, 8, 93–97. [Google Scholar] [CrossRef]
- Zhang, Z.; Xiao, C.; Adeyeye, O.; Yang, W.; Liang, X. Source and Mobilization Mechanism of Iron, Manganese and Arsenic in Groundwater of Shuangliao City, Northeast China. Water 2020, 12, 534. [Google Scholar] [CrossRef]
- Jakobsen, R.; Postma, D. In Situ Rates of Sulfate Reduction in an Aquifer (Romo Denmark) and Implications for the Reactivity of Organic Matter. Geology 1994, 22, 1103–1106. [Google Scholar] [CrossRef]
- Harvey, R.W.; Barber, L.B. Associations of Free-Living Bacteria and Dissolved Organic Compounds in a Plume of Contaminated Groundwater. J. Contam. Hydrol. 1992, 9, 91–103. [Google Scholar] [CrossRef]
- Judd, K.E.; Crump, B.C.; Kling, G.W. Variation in Dissolved Organic Matter Controls Bacterial Production and Community Composition. Ecology 2006, 87, 2068–2079. [Google Scholar] [CrossRef]
- Romera-Castillo, C.; Pinto, M.; Langer, T.M.; Álvarez-Salgado, X.A.; Herndl, G.J. Dissolved Organic Carbon Leaching from Plastics Stimulates Microbial Activity in the Ocean. Nat. Commun. 2018, 9, 1430. [Google Scholar] [CrossRef]
- Richards, L.A.; Lapworth, D.J.; Magnone, D.; Gooddy, D.C.; Chambers, L.; Williams, P.J.; van Dongen, B.E.; Polya, D.A. Dissolved Organic Matter Tracers Reveal Contrasting Characteristics across High Arsenic Aquifers in Cambodia: A Fluorescence Spectroscopy Study. Geosci. Front. 2019, 10, 1653–1667. [Google Scholar] [CrossRef]
- Baker, M.A.; Valett, H.M.; Dahm, C.N. Organic Carbon Supply and Metabolism in a Shallow Groundwater Ecosystem. Ecology 2000, 81, 3133–3148. [Google Scholar] [CrossRef]
- Shen, Y.; Chapelle, F.H.; Strom, E.W.; Benner, R. Origins and Bioavailability of Dissolved Organic Matter in Groundwater. Biogeochemistry 2015, 122, 61–78. [Google Scholar] [CrossRef]
- Longnecker, K.; Kujawinski, E.B. Composition of Dissolved Organic Matter in Groundwater. Geochim. Cosmochim. Acta 2011, 75, 2752–2761. [Google Scholar] [CrossRef]
- Batiot-Guilhe, C.; Emblanch, C.; Blavoux, B. Total Organic Carbon (TOC) and Magnésium: Two Complementary Tracers of Residence Time in Karstic Systems. C. R. Geosci. 2003, 335, 205–214. [Google Scholar]
- Barcelona, M.J. TOC Determinations in Ground Water. Ground Water 1984, 22, 18–24. [Google Scholar] [CrossRef]
- Pernthaler, J. Predation on Prokaryotes in the Water Column and Its Ecological Implications. Nat. Rev. Microbiol. 2005, 3, 537–546. [Google Scholar] [CrossRef]
- Goldscheider, N.; Hunkeler, D.; Rossi, P. Review: Microbial Biocenoses in Pristine Aquifers and an Assessment of Investigative Methods. Hydrogeol. J. 2006, 926–941. [Google Scholar] [CrossRef]
- Gopal, K.; Tripathy, S.S.; Bersillon, J.L.; Dubey, S.P. Chlorination Byproducts, Their Toxicodynamics and Removal from Drinking Water. J. Hazard. Mater. 2007, 140, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Chomycia, J.C.; Hernes, P.J.; Harter, T.; Bergamaschi, B.A. Land Management Impacts on Dairy-Derived Dissolved Organic Carbon in Ground Water. J. Environ. Qual. 2008, 37, 333–343. [Google Scholar] [CrossRef]
- Rajendran, A.; Shimizu, G.K.H.; Woo, T.K. The Challenge of Water Competition in Physical Adsorption of CO2 by Porous Solids for Carbon Capture Applications—A Short Perspective. Adv. Mater. 2023. [Google Scholar] [CrossRef] [PubMed]
- Gruau, G.; Dia, A.; Olivié-Lauquet, G.; Davranche, M.; Pinay, G. Controls on the Distribution of Rare Earth Elements in Shallow Groundwaters. Water Res. 2004, 38, 3576–3586. [Google Scholar] [CrossRef] [PubMed]
- Makehelwala, M.; Wei, Y.; Weragoda, S.K.; Weerasooriya, R.; Zheng, L. Characterization of Dissolved Organic Carbon in Shallow Groundwater of Chronic Kidney Disease Affected Regions in Sri Lanka. Sci. Total Environ. 2019, 660, 865–875. [Google Scholar] [CrossRef]
- Ahmed, A.U.; Hoddinott, J.F.; Md Shaiful Islam, K.; Mahbubur Rahman Khan, A.; Abedin, N.; Hossain, N.Z.; Ghostlaw, J.; Parvin, A.; Quabili, W.; Tahsin Rahaman, S.; et al. Impacts of Bt Brinjal (Eggplant) Impacts In Bangladesh; International Food Policy Research Institute: Washington, DC, USA, 2019. [Google Scholar]
- McDonough, L.K.; Rutlidge, H.; O’Carroll, D.M.; Andersen, M.S.; Meredith, K.; Behnke, M.I.; Spencer, R.G.M.; McKenna, A.M.; Marjo, C.E.; Oudone, P.; et al. Characterisation of Shallow Groundwater Dissolved Organic Matter in Aeolian, Alluvial and Fractured Rock Aquifers. Geochim. Cosmochim. Acta 2020, 273, 163–176. [Google Scholar] [CrossRef]
- Anawar, H.M.; Akai, J.; Komaki, K.; Terao, H.; Yoshioka, T.; Ishizuka, T.; Safiullah, S.; Kato, K. Geochemical Occurrence of Arsenic in Groundwater of Bangladesh: Sources and Mobilization Processes. J. Geochem. Explor. 2003, 77, 109–131. [Google Scholar] [CrossRef]
- McDonough, L.K.; Andersen, M.S.; Behnke, M.I.; Rutlidge, H.; Oudone, P.; Meredith, K.; O’Carroll, D.M.; Santos, I.R.; Marjo, C.E.; Spencer, R.G.M.; et al. A New Conceptual Framework for the Transformation of Groundwater Dissolved Organic Matter. Nat. Commun. 2022, 13, 1–11. [Google Scholar] [CrossRef]
- MacDonald, A.M.; Dochartaigh, B.É.Ó.; Kinniburgh, D.G.; Darling, W.G. Baseline Scotland: Groundwater Chemistry of Southern Scotland; British Geological Survey: Nottingham, UK, 2008. [Google Scholar]
- Edwards, A.M.C.; Thornes, J.B. Annual Cycle in River Water Quality: A Time Series Approach. Water Resour. Res. 1973, 9, 1286–1295. [Google Scholar] [CrossRef]
- Cun, C.; Vilagines, R. Time Series Analysis on Chlorides, Nitrates, Ammonium and Dissolved Oxygen Concentrations in the Seine River near Paris. Sci. Total Environ. 1997, 208, 59–69. [Google Scholar] [CrossRef]
- Hunt, M.; Herron, E.; Green, L. Chlorides in Fresh Water; University of Rhode Island: Kingston, RI, USA, 2012. [Google Scholar]
- Hong, Y.; Zhu, Z.; Liao, W.; Yan, Z.; Feng, C.; Xu, D. Freshwater Water-Quality Criteria for Chloride and Guidance for the Revision of the Water-Quality Standard in China. Int. J. Environ. Res. Public. Health 2023, 20, 2875. [Google Scholar] [CrossRef]
- Chen, J.; Wu, H.; Qian, H. Groundwater Nitrate Contamination and Associated Health Risk for the Rural Communities in an Agricultural Area of Ningxia, Northwest China. Expo. Health 2016, 8, 349–359. [Google Scholar] [CrossRef]
- Majumdar, D. The Blue Baby Syndrome. Resonance 2003, 8, 20–30. [Google Scholar] [CrossRef]
- Schijven, J.F.; Imůnek, J.S. Kinetic Modeling of Virus Transport at the Field Scale. J. Contam. Hydrol. 2002, 55, 113–135. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, M.E.; Shoaib, S.M.A.; Salam, M.A.; Elsayed, S.M. Efficient and Fast Removal of Total and Fecal Coliform, BOD, COD and Ammonia from Raw Water by Microwave Heating Technique. Groundw. Sustain. Dev. 2022, 19, 100847. [Google Scholar] [CrossRef]
- Dayanti, M.P.; Fachrul, M.F.; Wijayanti, A. Escherichia Coli as Bioindicator of the Groundwater Quality in Palmerah District, West Jakarta, Indonesia. IOP Conf. Ser. Earth Environ. Sci. 2018, 106, 012081. [Google Scholar] [CrossRef]
- Rosen, B.H. Waterborne Pathogens in Agricultural Watersheds; NRAES: New York, NY, USA, 2000. [Google Scholar]
- Tyrrel, S.F.; Quinton, J.N. Overland Flow Transport of Pathogens from Agricultural Land Receiving Faecal Wastes. J. Appl. Microbiol. 2003, 94, 87–93. [Google Scholar] [CrossRef]
- Unc, A.; Goss, M.J. Movement Of Faecal Bacteria Through The Vadose Zone. Water Air Soil. Pollut. 2003, 149, 327–337. [Google Scholar] [CrossRef]
- Cotruvo, J.A.; Dufour, A.; Rees, G.; Bartram, J.; Carr, R.; Cliver, D.O.; Craun, G.F.; Fayer, R.; Gannon, V.P.J.; Dufour, A.; et al. Waterborne Zoonoses; World Health Organization: London, UK, 2004. [Google Scholar]
- Fong, T.-T.; Lipp, E.K. Enteric Viruses of Humans and Animals in Aquatic Environments: Health Risks, Detection, and Potential Water Quality Assessment Tools. Microbiol. Mol. Biol. Rev. 2005, 69, 357–371. [Google Scholar] [CrossRef]
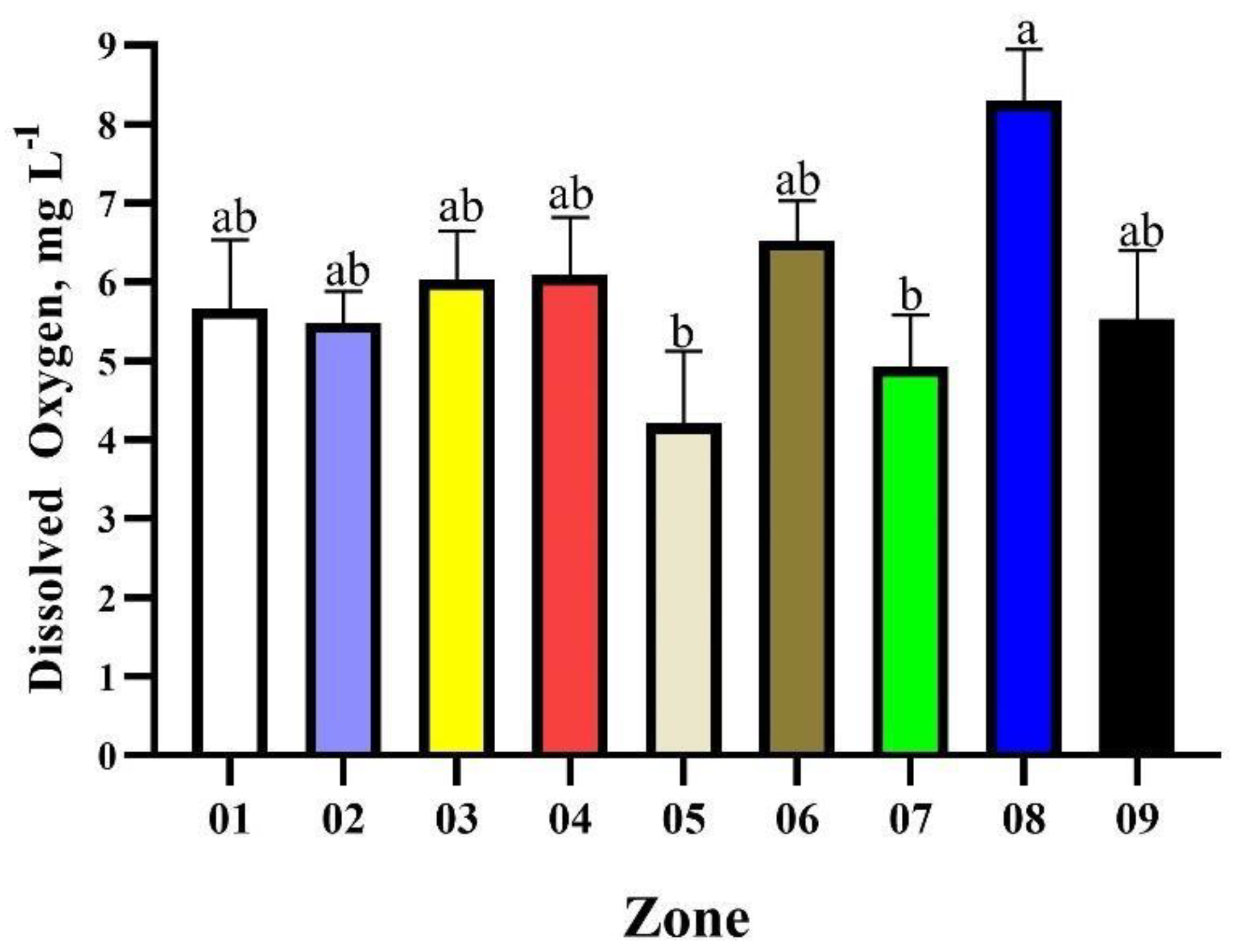
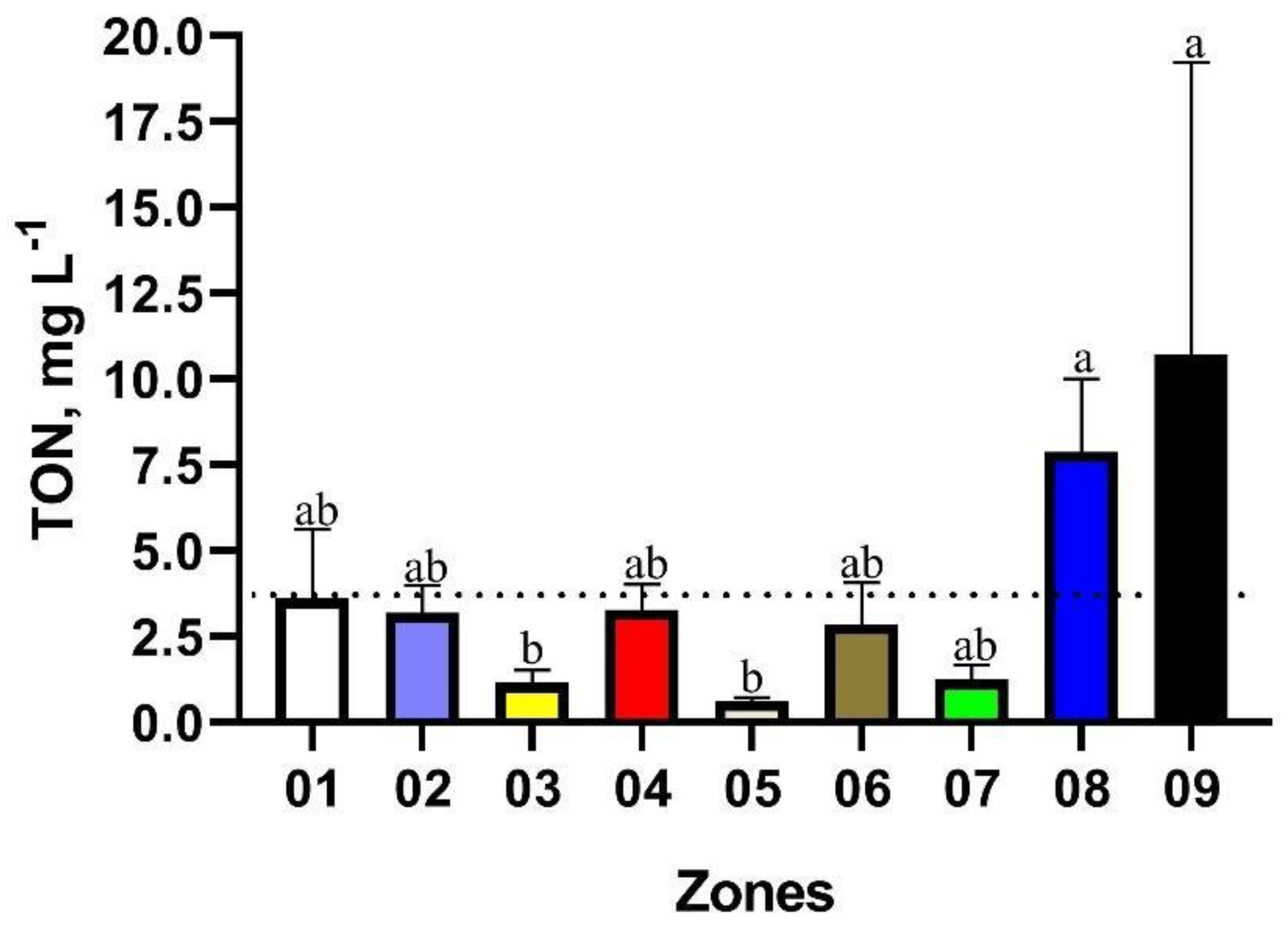
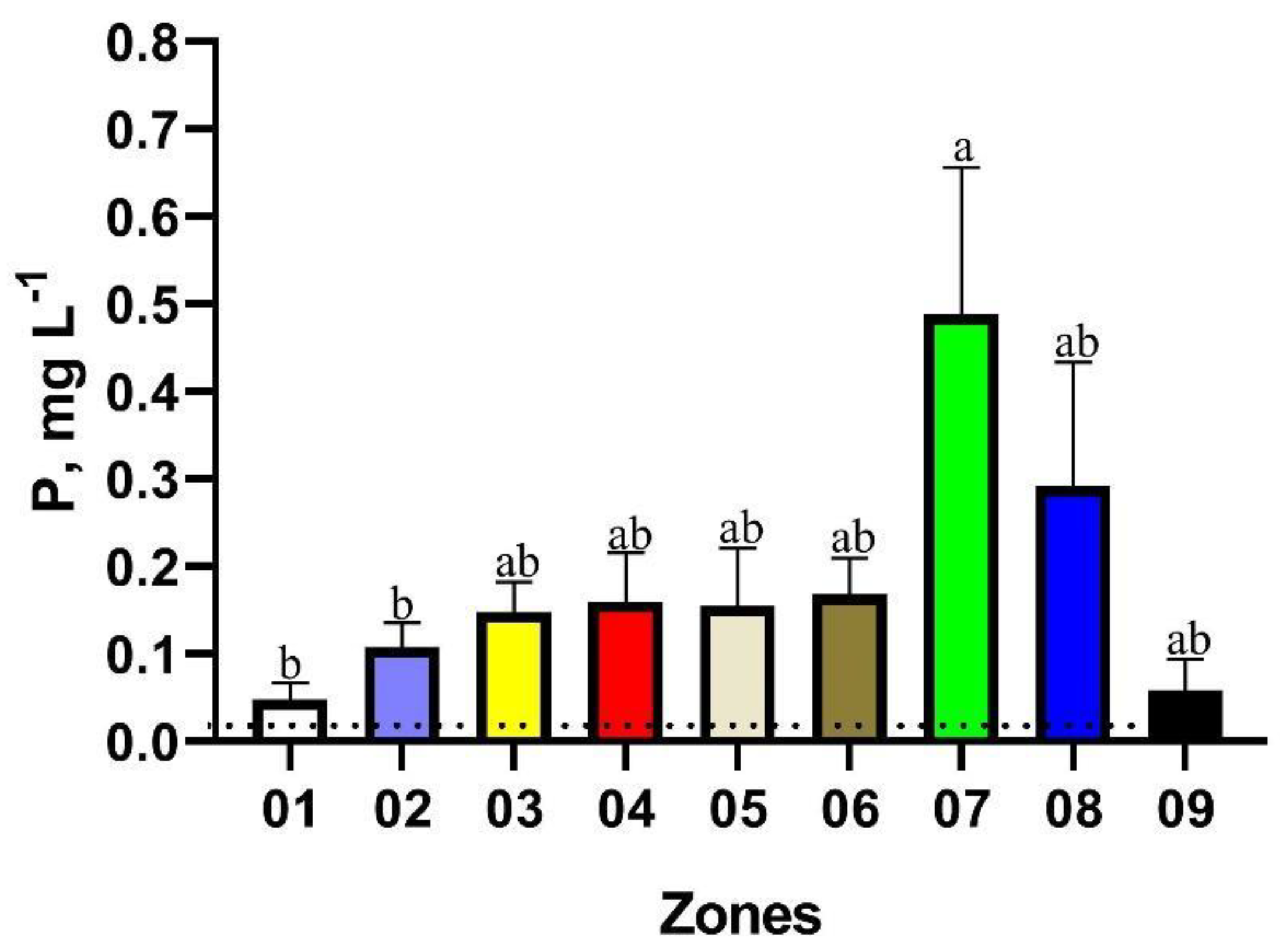

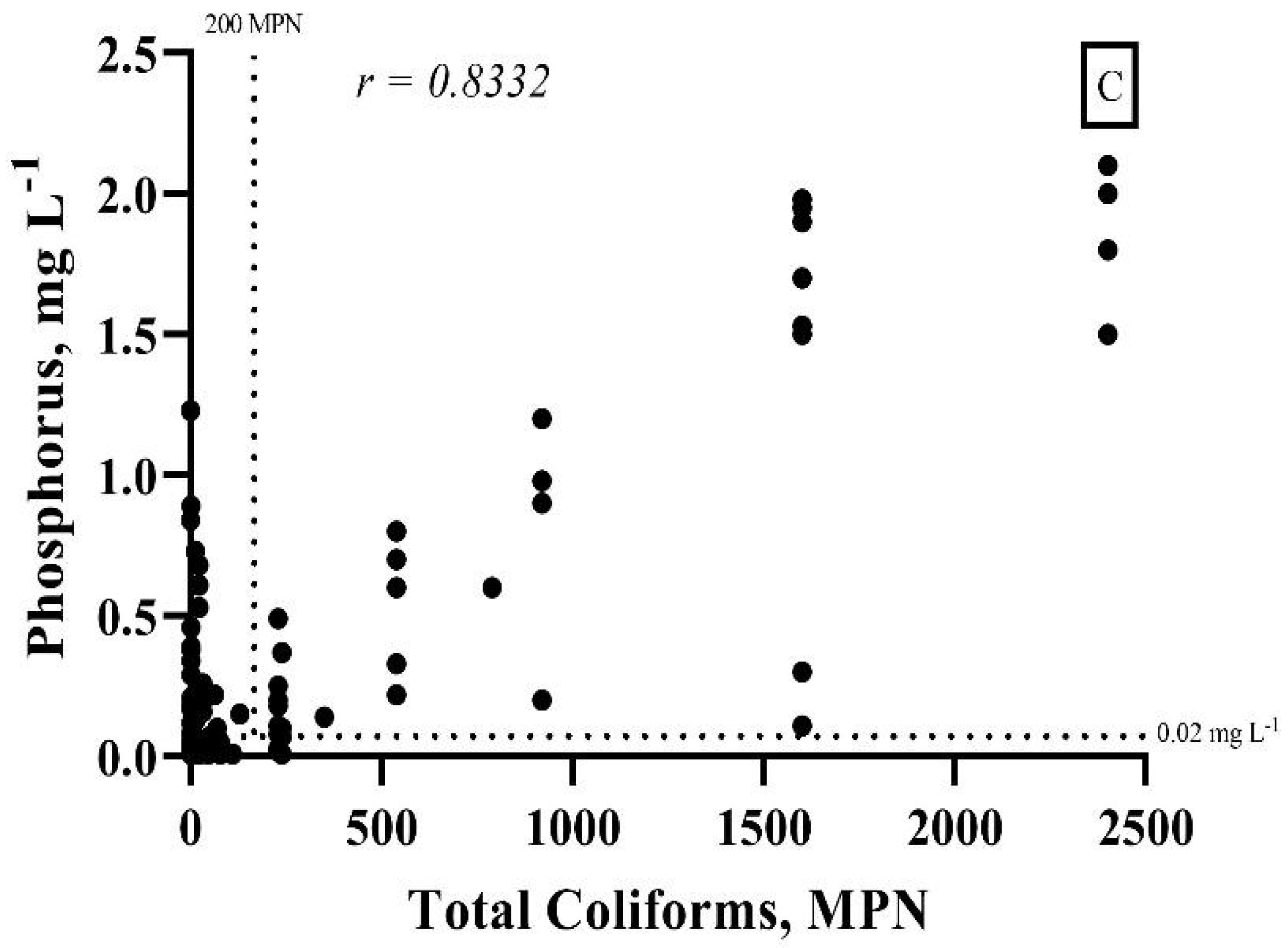
| Zone | Mean | Minimum Value | Maximum Value | Brazilian Legislation Limits | N° of Points in Disagreement with Brazilian Legislation | Proportion of Points in Disagreement with Brazilian Legislation, % |
|---|---|---|---|---|---|---|
| 01 | 5.92 | 5.01 | 6.88 | 6.00 to 9.00 | 06 | 66.66 |
| 02 | 5.83 | 3.11 | 6.97 | 10 | 34.48 | |
| 03 | 6.52 | 4.92 | 7.54 | 02 | 11.76 | |
| 04 | 6.18 | 4.95 | 7.91 | 07 | 43.75 | |
| 05 | 6.17 | 5.23 | 7.20 | 03 | 50.00 | |
| 06 | 6.23 | 5.43 | 7.09 | 06 | 50.00 | |
| 07 | 6.25 | 5.69 | 6.97 | 03 | 30.00 | |
| 08 | 5.95 | 3.45 | 7.25 | 07 | 38.88 | |
| 09 | 6,04 | 4,88 | 6,79 | 03 | 42,85 |
| Zone | Mean | Minimum Value | Maximum Value | Brazilian Legislation Limits | N° of Points in Disagreement with Brazilian Legislation | Proportion of Points in Disagreement with Brazilian Legislation, % |
|---|---|---|---|---|---|---|
| 01 | 8.01 | 1.90 | 35.30 | 10 mg·L−1 | 02 | 22.22 |
| 02 | 5.40 | 0.50 | 29.20 | 03 | 44.82 | |
| 03 | 11.56 | 1.20 | 49.10 | 08 | 47.05 | |
| 04 | 7.97 | 0.00 | 28.00 | 05 | 31.25 | |
| 05 | 7.77 | 0.90 | 18.40 | 04 | 66.66 | |
| 06 | 9.91 | 0.60 | 35.00 | 06 | 50.00 | |
| 07 | 6.41 | 0.50 | 15.10 | 05 | 50.00 | |
| 08 | 9.36 | 0.50 | 37.00 | 07 | 38.88 | |
| 09 | 6.37 | 0.50 | 12.00 | 03 | 42.85 |
| Site | Reference | Max DOC (mg·L−1) | Min DOC (mg·L−1) | Average DOC (mg·L−1) |
|---|---|---|---|---|
| Maricá, Brazil | Present study | 49.10 | <0.5 | 8.06 |
| Puducherry, India | [61] | 29 | 0.5 | 3.6 |
| Petit Hermitage, France | [62] | - | - | 4.4 |
| Sri Lanka | [63] | 2.08 | 1.35 | 1.69 |
| Bangladesh | [64] | 20.39 | 1.29 | 4.90 |
| Anna Bay, Australia | [65] | 15.1 | 1.0 | 8.0 |
| Bangladesh | [66] | 5.9 | 0.20 | 2.79 |
| Macquarie River, Australia | [67] | - | - | 8.26 |
| Bell River, Australia | [67] | - | - | 1.87 |
| Elfin Crossing, Australia | [67] | - | - | 1.34 |
| Silurian S, Scotland | [68] | 3.05 | 0.45 | - |
| Zone | Mean | Minimum Value | Maximum Value | Brazilian Legislation Limits | N° of Points in Disagreement with Brazilian Legislation | Proportion of Points in Disagreement with Brazilian Legislation, % |
|---|---|---|---|---|---|---|
| 01 | 170.4 | 32.0 | 853.6 | 250 mg·L−1 | 01 | 11.11 |
| 02 | 157.9 | 12.2 | 1259.6 | 04 | 13.79 | |
| 03 | 185.8 | 32.5 | 483.2 | 05 | 29.41 | |
| 04 | 93.0 | 21.0 | 317.0 | 01 | 6.25 | |
| 05 | 327.5 | 16.4 | 1472.4 | 02 | 33.30 | |
| 06 | 58.8 | 11.5 | 178.3 | 00 | 0.00 | |
| 07 | 14.0 | 77.1 | 1.04 | 00 | 0.00 | |
| 08 | 148.9 | 739.8 | 27.54 | 01 | 5.55 | |
| 09 | 382.8 | 29.5 | 1466.2 | 03 | 42.85 |
| Zone | Mean | Minimum Value | Maximum Value | Brazilian Legislation Limits | N° of points in Disagreement with Brazilian Legislation | Proportion of Points in Disagreement with Brazilian Legislation, % |
|---|---|---|---|---|---|---|
| 01 | 4.15 | 0.00 | 16.89 | 10 mg·L−1 | 01 | 11.11 |
| 02 | 3.37 | 0.05 | 15.04 | 02 | 6.89 | |
| 03 | 1.89 | 0.05 | 15.84 | 01 | 5.88 | |
| 04 | 2.66 | 0.00 | 7.00 | 00 | 0.00 | |
| 05 | 0.16 | 0.05 | 0.41 | 00 | 0.00 | |
| 06 | 2.23 | 0.05 | 11.47 | 01 | 8.33 | |
| 07 | 0.28 | 0.05 | 1.04 | 00 | 0.00 | |
| 08 | 4.86 | 0.05 | 27.54 | 03 | 16.66 | |
| 09 | 4.51 | 0.08 | 27.98 | 01 | 14.28 |
| Zone | Mean | Minimum Value | Maximum Value | Brazilian Legislation Limits | N° of Points in Disagreement with Brazilian Legislation | Proportion of Points in Disagreement with Brazilian Legislation, % |
|---|---|---|---|---|---|---|
| 01 | 3.61 | 0.30 | 13.90 | 3.7 mg·L−1 | 03 | 33.33 |
| 02 | 3.20 | 0.10 | 17.00 | 10 | 34.48 | |
| 03 | 1.16 | 0.10 | 5.50 | 02 | 11.76 | |
| 04 | 3.28 | 0.00 | 8.70 | 07 | 43.75 | |
| 05 | 0.67 | 0.30 | 0.90 | 00 | 00.00 | |
| 06 | 2.85 | 0.30 | 14.30 | 02 | 16.66 | |
| 07 | 1.24 | 0.10 | 4.20 | 01 | 10.00 | |
| 08 | 7.87 | 1.40 | 30.60 | 09 | 50.00 | |
| 09 | 10.70 | 0.10 | 36.23 | 01 | 14.28 |
| Zone | Mean | Minimum Value | Maximum | Brazilian Legislation Limits | N° of Points in Disagreement with Brazilian Legislation | Proportion of Points in Disagreement with Brazilian Legislation, % |
|---|---|---|---|---|---|---|
| Value | ||||||
| 01 | 0.05 | 0.01 | 0.19 | 0.02 mg·L−1 | 04 | 44.44 |
| 02 | 0.11 | 0.01 | 0.61 | 16 | 55.17 | |
| 03 | 0.15 | 0.01 | 0.44 | 13 | 76.47 | |
| 04 | 0.16 | 0.00 | 0.89 | 14 | 87.5 | |
| 05 | 0.16 | 0.02 | 0.46 | 05 | 83.33 | |
| 06 | 0.17 | 0.01 | 0.49 | 11 | 91.66 | |
| 07 | 0.35 | 0.01 | 1.23 | 07 | 70.00 | |
| 08 | 0.26 | 0.01 | 2.32 | 12 | 66.66 | |
| 09 | 0.06 | 0.01 | 0.26 | 03 | 42.85 |
| Zone | Mean | Minimum Value | Maximum | Brazilian Legislation Limits | N° of Points in Disagreement with Brazilian Legislation | Proportion of Points in Disagreement with Brazilian Legislation, % |
|---|---|---|---|---|---|---|
| Value | ||||||
| 01 | 370 | 0 | 2400 | 200 MPN | 04 | 44.44 |
| 02 | 218 | 2 | 2400 | 04 | 13.79 | |
| 03 | 286 | 2 | 1600 | 06 | 35.29 | |
| 04 | 433 | 0 | 1600 | 06 | 37.5 | |
| 05 | 18 | 2 | 49 | 00 | 0.00 | |
| 06 | 1455 | 2 | 1600 | 10 | 83.33 | |
| 07 | 934 | 0 | 920 | 04 | 40.00 | |
| 08 | 962 | 2 | 1600 | 09 | 50.00 | |
| 09 | 291 | 0 | 1400 | 03 | 42.85 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Publio, M.C.M.; Delgado, J.F.; Pierri, B.S.; Lima, L.d.S.; Gaylarde, C.C.; Baptista Neto, J.A.; Neves, C.V.; Fonseca, E.M. Assessment of Groundwater Contamination in the Southeastern Coast of Brazil: A Potential Threat to Human Health in Marica Municipality. Eng 2023, 4, 2640-2655. https://doi.org/10.3390/eng4040151
Publio MCM, Delgado JF, Pierri BS, Lima LdS, Gaylarde CC, Baptista Neto JA, Neves CV, Fonseca EM. Assessment of Groundwater Contamination in the Southeastern Coast of Brazil: A Potential Threat to Human Health in Marica Municipality. Eng. 2023; 4(4):2640-2655. https://doi.org/10.3390/eng4040151
Chicago/Turabian StylePublio, Maria Cristina M., Jessica F. Delgado, Bruno S. Pierri, Leonardo da S. Lima, Christine C. Gaylarde, José Antônio Baptista Neto, Charles V. Neves, and Estefan M. Fonseca. 2023. "Assessment of Groundwater Contamination in the Southeastern Coast of Brazil: A Potential Threat to Human Health in Marica Municipality" Eng 4, no. 4: 2640-2655. https://doi.org/10.3390/eng4040151
APA StylePublio, M. C. M., Delgado, J. F., Pierri, B. S., Lima, L. d. S., Gaylarde, C. C., Baptista Neto, J. A., Neves, C. V., & Fonseca, E. M. (2023). Assessment of Groundwater Contamination in the Southeastern Coast of Brazil: A Potential Threat to Human Health in Marica Municipality. Eng, 4(4), 2640-2655. https://doi.org/10.3390/eng4040151






