Abstract
Background/Objectives: Chronic rhinosinusitis (CRS) is a prevalent inflammatory condition of the nasal cavity and paranasal sinuses that significantly impact patients’ quality of life and imposes a substantial burden on healthcare systems and society, including considerable costs associated with surgical management when required. This review aimed to provide a comprehensive overview of the burden of CRS, encompassing its impact on patients, society, and the economy, while also highlighting the latest advancements in diagnosis and treatment. Methods: A systematic review using PubMed, Embase and Google Scholar databases identified recent literature published in English, adhering to PRISMA guidelines. Search terms included “chronic rhinosinusitis”, “burden”, “quality of life”, “economic impact”, “diagnosis”, “treatment”, and “endotypes”. Original research, reviews, and meta-analyses were included, while case reports, surgical-only studies, and non-English articles were excluded. Results: Characterized by persistent symptoms such as nasal congestion, facial pressure, and discharge, CRS often leads to decreased productivity, missed workdays, and impaired sleep. Recent advances in understanding the pathophysiology of CRS have led to the identification of distinct endotypes, including type 2 inflammation characterized by eosinophilic infiltration, and type 1 inflammation with a neutrophilic predominance. Conclusions: This understanding has paved the way for targeted therapies, including biologic agents that have shown promising results in managing CRS, particularly in patients with type 2 inflammation.
1. Introduction
CRS is a persistent inflammatory disorder that affects the nasal cavity and paranasal sinuses, resulting in a range of bothersome symptoms and a significant impairment in patients’ quality of life [1]. The condition is characterized by two or more cardinal symptoms, including nasal obstruction, nasal discharge (anterior and/or posterior nasal drip), facial pressure, and a reduction in or the loss of sense of smell. CRS affects a substantial portion of the population, with prevalence estimates ranging from 5% to 12%, although these figures may be underreported due to misdiagnosis or underdiagnosis [2].
The impact of CRS extends far beyond the individual patient, affecting families, workplaces, and healthcare systems [3]. The chronic nature of the disease, with its persistent symptoms and frequent exacerbations, can lead to decreased productivity, missed workdays, and impaired sleep, resulting in substantial economic and social burdens. Furthermore, when surgical intervention becomes necessary, it adds another layer to these societal costs, encompassing direct medical expenses and indirect losses due to recovery and potential complications. Moreover, CRS is often associated with other comorbidities, such as asthma, allergies, and sleep disorders, further complicating its management and contributing to its overall burden [4].
Recent advances in understanding the pathophysiology of CRS have led to the identification of distinct endotypes, each characterized by unique inflammatory patterns and clinical presentations [5]. This has paved the way for more targeted therapies, including biologic agents that have shown promising results in managing CRS, particularly in patients with type 2 inflammation. These advancements offer hope for improved disease control and a better quality of life for patients with CRS [6].
This review aimed to provide a comprehensive overview of the burden of CRS, encompassing its impact on patients, society, and the economy, while also highlighting the latest advancements in diagnosis and treatment [7]. By shedding light on the multifaceted challenges posed by CRS, this review seeks to promote greater awareness of the disease and encourage further research into its causes, prevention, and management [8].
2. Materials and Methods
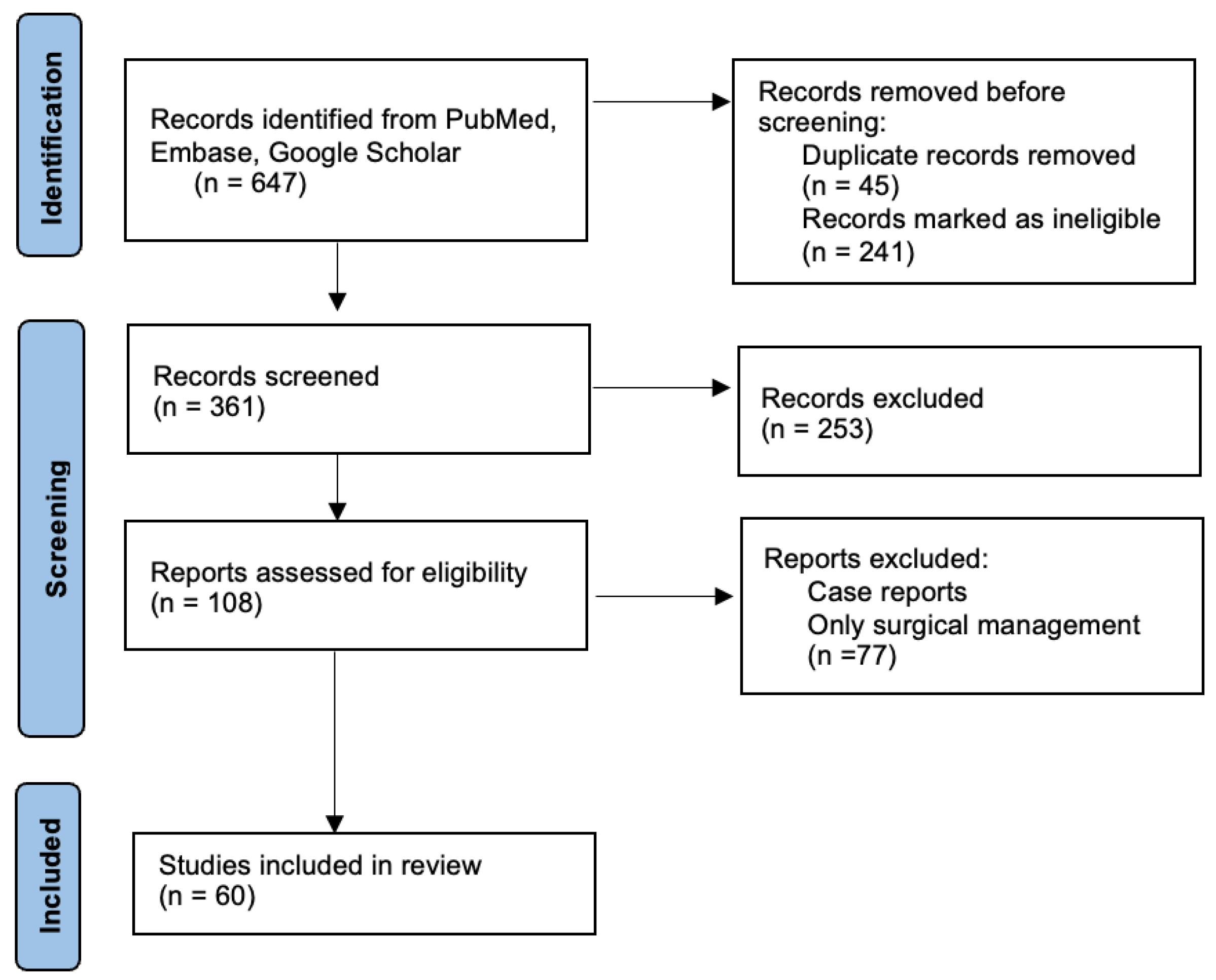
This review was conducted by systematically searching the PubMed, Embase, and Google Scholar databases for relevant articles, with a focus on recent advancements in understanding the burden of CRS. The MeSH terms included “chronic rhinosinusitis,” “burden,” “quality of life,” “economic impact,” “diagnosis,” “treatment,” and “endotypes,” and the search was limited to articles published in English. In addition, reference lists of relevant articles were reviewed to identify other potentially relevant studies. Articles were included if they were original research articles, review articles, or meta-analyses that specifically addressed the burden of CRS, including its impact on patients, society, and the economy, and that discussed the latest advancements in diagnosis and treatment of CRS. Case reports and case series, studies that focused solely on the surgical management of CRS, and studies that were not published in English were excluded. The literature review is presented in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. A flow chart showing the literature review process is shown in Figure 1.
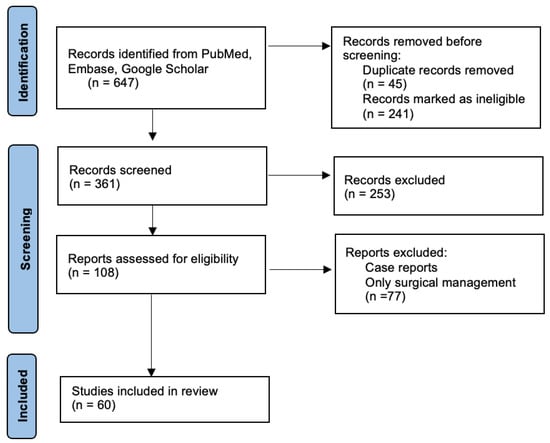
Figure 1.
Flow chart of the literature review process.
3. Results
The initial search yielded 647 articles, and after screening the titles and abstracts, 137 articles were selected for a full-text review. Of these, 60 articles met the inclusion criteria and were included in this review. The selected articles were critically appraised for their quality and relevance to the topic, and the information extracted from these articles was synthesized and presented, focusing on the key themes and findings related to the burden of CRS. To synthesize and compare the main key findings from studies, we have centralized this information in Table 1, which provides a structured overview of the existing literature on this topic.

Table 1.
Summary of the identified studies.
3.1. Epidemiology and Risk Factors
CRS is a global health concern, affecting individuals across various ages, ethnicities, and socioeconomic backgrounds. While its prevalence varies across different regions and populations, it is estimated that CRS affects approximately 12% of the adult population in Western countries. This substantial prevalence translates to a significant burden on healthcare systems and society, with CRS accounting for numerous physician visits, diagnostic tests, and treatment costs [37].
Several risk factors contribute to the development and persistence of CRS, broadly categorized into host factors, environmental factors, and triggering factors. Host factors encompass individual characteristics and predispositions that increase the likelihood of developing CRS. These include genetic predisposition, with a family history of CRS and certain genetic variations linked to an increased risk. Individuals with impaired immune function, such as those with allergies, asthma, or immunodeficiency disorders, are more susceptible to CRS. Structural variations in the nasal cavity and sinuses, like a deviated septum or nasal polyps, can disrupt normal sinus drainage and ventilation, predisposing individuals to CRS [38].
Environmental factors play a crucial role in the development and exacerbation of CRS. Exposure to air pollutants, such as particulate matter, ozone, and cigarette smoke, can trigger inflammation and impair mucociliary clearance, contributing to CRS. Certain occupational settings involving exposure to dust, fumes, or chemicals can increase the risk of CRS. Changes in temperature and humidity can affect the nasal mucosa and mucociliary function, potentially influencing the development of CRS.
Triggering factors can initiate or worsen CRS symptoms in individuals predisposed to the disease. Viral upper respiratory infections are a common trigger for acute rhinosinusitis, which can sometimes progress to CRS. Exposure to allergens, such as pollen, dust mites, and pet dander, can trigger allergic inflammation in the nasal passages, contributing to CRS. Irritants, such as strong odors, perfumes, and cleaning products, can also trigger inflammation and exacerbate CRS symptoms [39].
The complex interplay of these various risk factors contributes to the development and persistence of CRS, highlighting the multifaceted nature of the disease. Understanding these risk factors is crucial for developing effective prevention and management strategies [40].
3.2. Pathophysiology and Endotypes
CRS is a complex disorder characterized by chronic inflammation of the sinonasal mucosa. The exact pathophysiology of CRS remains incompletely understood, but it is believed to involve a complex interplay of factors, including genetic predisposition, environmental triggers, and the dysregulation of the immune system [41].
One of the key features of CRS is the disruption of mucociliary clearance, a critical mechanism for removing debris and pathogens from the sinuses. The sinonasal mucosa is lined with cilia, microscopic hair-like projections that beat in a coordinated manner to propel mucus and trapped particles towards the nasal cavity. In CRS, this process is impaired due to various factors, including inflammation, infection, and structural abnormalities [42].
Inflammation plays a central role in the pathophysiology of CRS. The inflammatory response in CRS is characterized by the infiltration of various immune cells, including neutrophils, eosinophils, and lymphocytes, into the sinonasal mucosa. These cells release inflammatory mediators, such as cytokines and chemokines, which contribute to the persistent inflammation and tissue remodeling observed in CRS [43].
Recent advances in CRS research have led to the identification of distinct endotypes, each characterized by unique inflammatory patterns and clinical presentations. The most well-characterized endotypes are Type 2 CRS and non-Type 2 CRS. Type 2 CRS is characterized by a predominance of eosinophilic inflammation and is often associated with nasal polyps, asthma, and aspirin sensitivity. It is driven by a T helper 2 (Th2) immune response, with increased levels of cytokines such as interleukin (IL)-4, IL-5, and IL-13. Non-Type 2 CRS encompasses a heterogeneous group of patients who do not exhibit the characteristic features of Type 2 inflammation. Non-Type 2 CRS may be associated with neutrophilic inflammation, and the underlying immune mechanisms are less well defined [44].
The identification of these distinct endotypes has important implications for the diagnosis and management of CRS. Recognizing the specific endotype can help guide treatment decisions and improve patient outcomes. Patients with Type 2 CRS may benefit from targeted therapies that block the activity of Th2 cytokines, such as biologic agents [45].
There is growing recognition of other potential endotypes, such as those characterized by specific infectious agents or genetic variations. Further research is needed to fully elucidate the complex pathophysiology of CRS and identify additional endotypes that may have clinical relevance [46].
3.3. Clinical Presentation and Diagnosis
CRS typically manifests with a combination of nasal symptoms, including nasal congestion or obstruction, nasal discharge (which can be anterior, posterior, or both), and facial pressure or pain. Patients may also experience a reduced or loss of sense of smell, headaches, ear fullness, fatigue, and cough. The specific symptoms and their severity can vary depending on the underlying cause of CRS, the presence or absence of nasal polyps, and individual factors [46].
Diagnosing CRS involves a comprehensive evaluation that includes detailed medical history, physical examination, and often additional diagnostic tests. Medical history should focus on the duration and nature of symptoms, any previous treatments, and the presence of any comorbidities, such as allergies or asthma [20]. Physical examination typically involves anterior rhinoscopy or nasal endoscopy to visualize the nasal cavity and assess for signs of inflammation, polyps, or anatomical abnormalities [47].
In cases where the diagnosis is unclear or to further evaluate the extent of disease, imaging studies may be necessary. Computed tomography (CT) scans are commonly used to visualize the paranasal sinuses and assess for mucosal thickening, sinus opacification, or other abnormalities. While CT scans provide detailed anatomical information, they do not provide information about the underlying inflammatory process [21].
Other diagnostic tests may be considered depending on the individual patient’s presentation. These may include allergy testing to identify potential allergic triggers, nasal cytology to assess the type of inflammation, and olfactory testing to evaluate the sense of smell [22].
The accurate diagnosis and identification of the underlying endotype are crucial for guiding treatment decisions and improving patient outcomes. A personalized approach to diagnosis and management is essential, considering the individual’s specific symptoms, disease severity, and preferences [23].
3.4. Quality of Life and Disease Burden
CRS significantly diminishes patients’ quality of life, imposing a substantial burden on individuals, families, and society. The chronic and often debilitating nature of the disease can lead to physical, emotional, and social consequences, affecting various aspects of daily life [63].
One of the most significant impacts of CRS is on patients’ physical health. Persistent nasal congestion, facial pain, and a reduced sense of smell can significantly impair daily activities, such as sleep, work, and social interactions. Patients with CRS often report experiencing fatigue, difficulty concentrating, and reduced productivity, with one study finding that over 60% of patients reported fatigue and difficulty concentrating [18]. The chronic inflammation and nasal obstruction can also lead to sleep disturbances, including snoring and obstructive sleep apnea, further contributing to fatigue and reduced quality of life. Studies have shown that up to 70% of patients with CRS experience sleep disturbances [24].
CRS can also lead to headaches due to pressure and inflammation in the sinuses. It can cause ear infections and hearing loss due to blockage of the Eustachian tube [25]. In rare cases, CRS can even lead to vision problems due to the proximity of the sinuses to the eyes [26]. Additionally, CRS can cause a reduced or lost sense of smell (hyposmia or anosmia) and taste, significantly impacting quality of life [27]. Studies have shown that up to 80% of patients with CRS experience some degree of smell loss. The inflammation from CRS can sometimes affect the teeth and gums, leading to pain and discomfort [28].
CRS also takes a toll on patients’ emotional well-being. The chronic nature of the disease, with its persistent symptoms and unpredictable exacerbations, can lead to frustration, anxiety, and depression [48]. Patients with CRS may experience social isolation and decreased self-esteem due to their symptoms. One study found that over 40% of patients with CRS reported symptoms of depression [49]. The impact on mental health can be particularly pronounced in patients with severe CRS or those who have undergone multiple surgeries without achieving complete relief [50].
CRS can also have a significant social impact on patients. The chronic symptoms, such as nasal congestion, facial pain, and reduced sense of smell, can make it difficult to participate in social activities and maintain relationships. Patients may avoid social situations due to embarrassment about their symptoms or fear of experiencing an exacerbation. This social isolation can further contribute to feelings of depression and anxiety [29].
The economic burden of CRS is substantial, both for individuals and society. Patients with CRS often miss work or school due to their symptoms, resulting in lost productivity and income [30]. One study estimated that CRS costs the US economy over USD 22 billion annually in direct healthcare costs and lost productivity [51]. A breakdown of these costs includes direct costs, such as medical consultations, diagnostic tests, medications, and surgeries [16]. Specifically, surgical interventions like Functional Endoscopic Sinus Surgery (FESS), the most common procedure for CRS, represent a significant component of these direct costs. In the United States, for example, the direct cost of an outpatient FESS procedure can range from approximately USD 8000 to USD 15,000 per case, potentially increasing with complications or the need for inpatient care [16,51]. These surgical costs typically cover surgeon and hospital fees, anesthesia, operative medications, and immediate postoperative care. Patients with Chronic Rhinosinusitis with Nasal Polyps (CRSwNP) undergoing FESS often incur some of the highest direct healthcare expenditures within this patient group [15].
The average annual direct cost per patient can range from USD 500 to over USD 5000, depending on the severity of the disease and the types of treatments required [15]. Indirect costs include lost productivity due to missed workdays and reduced efficiency at work. It is estimated that CRS leads to an average of 7.5 lost workdays per year per patient [13]. Surgical treatment for CRS, while aiming for long-term symptom relief, also contributes significantly to these indirect costs. The postoperative recovery period necessitates time off work, leading to lost wages for patients and reduced productivity for employers. Studies have shown that overall indirect costs related to CRS, including productivity losses from absenteeism and presenteeism (working while sick but at reduced capacity), can amount to billions of dollars annually in a large economy like the U.S. [11]. For instance, some analyses indicate that patients with refractory CRSwNP, who are often surgical candidates, can experience annual productivity losses valued at several thousand dollars [9,14].
Beyond the immediate economic costs, the management of CRS, especially with surgical interventions, introduces additional considerations related to treatment failure and potential complications. While Functional Endoscopic Sinus Surgery (FESS) is a common procedure for CRS, the rate of revision surgery can be considerable, particularly in patients with Chronic Rhinosinusitis with Nasal Polyps (CRSwNP), leading to increased long-term costs and demand on healthcare resources. Despite the initial financial outlay for surgery being high, some economic evaluations suggest that for patients with refractory CRS who have failed maximal medical therapy, FESS can be a cost-effective strategy in the long term, potentially offsetting initial surgical expenditure through improved quality of life, reduced medication reliance, and fewer physician visits. However, the need for proficient surgeons and optimized training programs for complex procedures like FESS is crucial for improving overall healthcare costs and patient outcomes. Post-operative follow-up visits for debridement and monitoring are also significant components of healthcare resource utilization following surgery.
The societal impact is compounded when considering caregiver burden, as family members may also need to take time off work to support the patient during the recovery phase. Indirect costs also encompass the burden on caregivers, who may need to take time off work or incur expenses related to caring for the patient with CRS [52].
In addition to these economic costs, there are also intangible costs associated with CRS, such as reduced quality of life, pain and suffering, and emotional distress [53].
The burden of CRS extends beyond the individual patient, affecting families and caregivers. Family members may experience stress and anxiety due to their loved one’s condition, and they may need to provide additional support and care. The financial burden of CRS can also place a significant strain on families, particularly those with limited resources [54].
The overall burden of CRS is substantial, affecting individuals, families, and society. Recognizing the multifaceted impact of CRS is crucial for healthcare providers, policymakers, and patients. Efforts to improve the awareness, diagnosis, and management of CRS are essential to reduce the burden of this prevalent and often debilitating disease [31].
The burden of CRS can vary depending on the severity of the disease, the presence of comorbidities, and individual factors. Patients with severe CRS or those who have not responded well to treatment may experience a particularly high burden of disease. There is a need for more research to fully understand the burden of CRS and to develop effective strategies to reduce its impact [17].
Beyond immediate surgical costs and lost productivity, the surgical management of CRS, particularly for recurrent or complex cases like CRSwNP, leads to substantial healthcare resource utilization. This includes multiple pre-operative consultations, diagnostic imaging such as CT scans, frequent post-operative follow-up visits for debridement and monitoring, and, significantly, the potential need for revision surgeries. The challenge of providing effective and accessible surgical training, particularly for FESS, has led to the exploration of alternative anatomical models, addressing both the need for proficient surgeons and the economic considerations in medical training [64].
Studies on surgical training for FESS emphasize the role of objective performance assessment. One such study [65] utilized accelerometer-based sensors to analyze hand movements on a sheep’s head model. Such objective data is crucial for tailoring training programs, potentially optimizing the development of surgical skills and improving the efficiency of preparing surgeons for complex procedures like FESS, which can have implications for overall healthcare costs associated with treating conditions such as rhinosinusitis [65].
The rate of revision surgery can be considerable, especially in CRSwNP patients, further escalating long-term costs and the demand on healthcare resources [12,19].
While the initial financial outlay for surgical treatment is high, some economic evaluations suggest that for patients with refractory CRS who have failed maximal medical therapy, FESS can be a cost-effective strategy in the long term compared to ongoing medical management alone. The long-term improvements in quality of life, reduced reliance on medications, and fewer physician visits can offset the initial surgical expenditure over several years [10].
3.5. Treatment Strategies
The management of CRS aims to alleviate symptoms, improve quality of life, and prevent disease progression. Treatment strategies are tailored to the individual patient, considering factors such as disease severity, endotype, presence of comorbidities, and patient preferences. Treatment options include medical therapies, surgical interventions, and complementary approaches [32]. Medical therapies form the cornerstone of CRS management, aiming to reduce inflammation, improve mucociliary clearance, and control infection [55]. Commonly used medical therapies include intranasal corticosteroids, which effectively reduce inflammation in the sinonasal mucosa and are available in various formulations, typically administered once or twice daily. Nasal saline irrigation helps rinse away mucus, allergens, and irritants, promoting mucociliary clearance and improving nasal symptoms [56].
Oral and topical decongestants can provide temporary relief from nasal congestion but should be used cautiously due to the risk of rebound congestion with prolonged use. Antihistamines can be helpful in patients with allergic CRS to reduce allergic inflammation. Antibiotics are typically reserved for patients with acute bacterial rhinosinusitis or those with evidence of bacterial infection complicating CRS [33].
Surgical interventions may be considered for patients with CRS who have not responded adequately to medical therapy or those with anatomical abnormalities that contribute to disease persistence. The most common surgical procedure for CRS is FESS, which involves minimally invasive techniques to open the sinus ostia and improve sinus ventilation and drainage [57]. As discussed, this intervention carries significant direct and indirect societal costs, although its long-term cost-effectiveness for appropriately selected patients with refractory disease is also an important consideration in treatment planning [10].
Beyond acute bacterial infections, the low-dose, long-term administration of 14-membered macrolide antibiotics, often referred to as macrolide therapy, has been shown to favorably modify the clinical condition of non-Type 2 CRS. This approach, using drugs such as roxithromycin and clarithromycin, works through immunomodulatory and anti-inflammatory effects rather than direct antimicrobial action, making it a distinct pharmacological strategy for managing chronic inflammation in specific CRS endotypes [17].
Complementary approaches, such as acupuncture, herbal remedies, and dietary modifications, are sometimes used by patients with CRS to manage their symptoms. While some individuals may find these approaches helpful, evidence supporting their efficacy is limited [34]. Recent advances in CRS research have led to the development of novel therapies that target specific inflammatory pathways. Biologic agents, such as dupilumab and omalizumab, have shown promising results in patients with CRS, particularly those with Type 2 inflammation. These medications block the activity of key cytokines involved in the inflammatory process, leading to significant improvements in symptoms and quality of life [35].
Recent advances in CRS research have led to the development of novel therapies that target specific inflammatory pathways [35]. Biologic agents, such as dupilumab and omalizumab, have shown promising results, particularly in patients with Type 2 inflammation [35]. These medications work by blocking the activity of key cytokines involved in the inflammatory process, leading to significant improvements in symptoms and quality of life [35]. The use of biologics in CRS treatment is a relatively new approach that has shown promising results, especially in patients with Type 2 inflammation or those with nasal polyps who have not responded well to other treatments [36]. Biologics are genetically engineered proteins that target specific inflammatory pathways, effectively reducing inflammation and improving symptoms. Examples include dupilumab, which inhibits IL-4 and IL-13, and omalizumab, which targets IgE. Studies have shown that biologics can significantly reduce nasal polyp size, improve nasal airflow, and decrease the need for additional surgery [58,59].
The optimal treatment strategy for CRS is individualized, considering the patient’s specific needs and preferences. Shared decision-making between the patient and healthcare provider is crucial to ensure that the chosen treatment plan aligns with the patient’s goals and values [60,66].
3.6. Limitations and Future Goals for CRS Medications
Despite advancements in understanding and treating CRS, current pharmacological approaches still face limitations, highlighting areas for future research and development. While medical therapies form the cornerstone of management, including intranasal corticosteroids, nasal saline irrigation, and decongestants, their efficacy can be limited in severe or refractory cases, particularly those with Type 2 inflammation or significant nasal polyps. Antihistamines are useful for allergic CRS, and antibiotics are generally reserved for bacterial infections, emphasizing the need for targeted treatments for the broader inflammatory spectrum of CRS [2,67].
Even with the promising introduction of biologic agents like dupilumab and omalizumab, which target specific inflammatory pathways and have shown significant improvements in symptoms and quality of life for Type 2 CRS [35,36,58,59], access, long-term efficacy, and potential side effects remain considerations. There is also a need to better understand their optimal duration of use and the potential for resistance or reduced responsiveness over time. Furthermore, non-Type 2 CRS, characterized by neutrophilic inflammation, has less well-defined underlying immune mechanisms, presenting a significant challenge for targeted pharmacological intervention [41,42].
Future goals for CRS medications include the development of more personalized medicine approaches, where treatments are tailored to the individual patient’s specific endotype, disease severity, and genetic predispositions [61,67,68]. This will involve leveraging advances in genomic and proteomic technologies to identify novel biomarkers for precise diagnosis and to develop therapies that more effectively target the diverse inflammatory mechanisms across all CRS endotypes [41,43]. Research should also focus on developing effective strategies to prevent the progression from acute rhinosinusitis to chronic CRS and identify modifiable risk factors that can be addressed through pharmacological interventions [33,51]. Ultimately, the aim is to develop medications that not only alleviate symptoms but also modify the disease course, reduce recurrence rates, and minimize the overall burden of CRS.
4. Conclusions
Chronic rhinosinusitis (CRS) represents a significant and multifaceted burden on individuals and healthcare systems globally, impacting physical health, emotional well-being, and social engagement, while incurring substantial economic costs, particularly through the necessity of surgical interventions for many patients. The profound effects, including fatigue, sleep disturbances, smell loss, and psychological distress, underscore the critical need for effective management strategies.
Advancements in elucidating CRS pathophysiology, particularly the identification of distinct endotypes such as Type 2 and non-Type 2 inflammation, have been pivotal. This deeper understanding has directly informed the evolution of more targeted diagnostic approaches and therapeutic interventions. The emergence of biologic agents, specifically for Type 2 inflammation, stands out as a promising development, offering significant improvements in symptoms and quality of life for previously refractory cases.
However, the journey towards comprehensive management is ongoing. Challenges persist in fully understanding the complex interplay of genetic, environmental, and immunological factors contributing to CRS across all endotypes, and in developing universally effective preventive strategies. Future research must prioritize personalized medicine approaches, leveraging advancements in genomic and proteomic technologies for precise diagnosis and the development of highly targeted therapies. Furthermore, continued investigation into modifiable risk factors and interventions to prevent disease progression from acute to chronic forms remains crucial. By fostering collaborative research and sustained innovation, we can strive to mitigate the extensive burden of CRS and markedly improve the quality of life for affected individuals.
Author Contributions
Conceptualization, D.G.R., V.B. and A.A.M.; methodology, D.G.R. and A.A.M.; software, D.G.R.; validation, D.G.R., V.B., C.S., O.D.P. and A.A.M.; formal analysis, D.G.R.; investigation, D.G.R. and A.A.M.; resources, D.G.R.; data curation, D.G.R.; writing—original draft preparation, D.G.R. and A.A.M.; writing—review and editing, V.B., C.S. and O.D.P.; visualization, D.G.R.; supervision, V.B. and O.D.P.; project administration, C.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| CRS | Chronic Rhinosinusitis |
| CRSwNP | Chronic Rhinosinusitis With Nasal Polyps |
| Th2 | T helper 2 |
| IL | Interleukin |
| CT | Computed Tomography |
| FESS | Functional Endoscopic Sinus Surgery |
References
- Tănase, M.I.; Tanase, M.; Cosgarea, M.; Radeanu, G.D.; Pop, S.S.; Maniu, A.A. Biologic Treatments for Chronic Rhinosinusitis with Nasal Polyps (CRSwNP): A Comparative Review of Efficiency and Risks. Cureus 2025, 17, e77804. [Google Scholar] [CrossRef] [PubMed]
- Fokkens, W.J.; Lund, V.J.; Hopkins, C.; Hellings, P.W.; Kern, R.; Reitsma, S.; Toppila-Salmi, S.; Bernal-Sprekelsen, M.; Mullol, J.; Alobid, I.; et al. European Position Paper on Rhinosinusitis and Nasal Polyps 2020. Rhinology 2020, 58 (Suppl. S29), 1–464. [Google Scholar] [CrossRef] [PubMed]
- Mora, T.; Sánchez-Collado, I.; Muñoz-Cano, R.; Ribó, P.; Mullol, J.; Valero, A. Differential Healthcare Direct Costs of Asthma and Chronic Rhinosinusitis with Nasal Polyps in Catalonia (Spain). Rhinology 2024, 62, 590–596. [Google Scholar] [CrossRef] [PubMed]
- Wallace, D.V. Treatment Options for Chronic Rhinosinusitis with Nasal Polyps. Allergy Asthma Proc. 2021, 42, 450–460. [Google Scholar] [CrossRef]
- Kim, H.A.J.; Lee, D.J.; Shin, D.; Horton, G.; Gignac, M.; Lee, J.M.; Chan, Y. Assessing the Use of Patient-Reported Outcome Measures in the Routine Clinical Care of Chronic Rhinosinusitis Patients: A Canadian Perspective. J. Otolaryngol.-Head Neck Surg. 2024, 53, 19160216241288806. [Google Scholar] [CrossRef]
- Duggal, C.; Chan, Y.; Rotenberg, B.W. What is Considered Adequate Surgery for Chronic Rhinosinusitis with Nasal Polyps? Laryngoscope 2025, 135, 1856–1857. [Google Scholar] [CrossRef]
- Hur, K.; Gao, J.; Adili, A.; Tam, B.; Herrera, K.; Rice, D.; Wrobel, B.; Wu, S. Treatment Decision-Making Among Chinese Americans with Chronic Rhinosinusitis. Laryngoscope 2025, 135, 1920–1927. [Google Scholar] [CrossRef]
- Biadsee, A.; Abu Amsha, N.; Sowerby, L.J.; Boldes, T.; Kassem, F. Validation of the Arabic Chronic Rhinosinusitis Patient-Reported Outcome (CRS-PRO): Translation and Cultural Adaptation. Healthcare 2025, 13, 206. [Google Scholar] [CrossRef]
- Bhattacharyya, N. The Economic Burden and Symptom Manifestations of Chronic Rhinosinusitis. Am. J. Rhinol. 2003, 17, 27–32. [Google Scholar] [CrossRef]
- Rudmik, L.; Soler, Z.M.; Mace, J.C.; Schlosser, R.J.; Smith, T.L. Economic evaluation of endoscopic sinus surgery versus continued medical therapy for refractory chronic rhinosinusitis. Laryngoscope 2015, 125, 25–32. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rudmik, L. Economics of Chronic Rhinosinusitis. Curr. Allergy Asthma Rep. 2017, 17, 20. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, N.; Villeneuve, S.; Joish, V.N.; Amand, C.; Mannent, L.; Amin, N.; Rowe, P.; Maroni, J.; Eckert, L.; Yang, T.; et al. Cost burden and resource utilization in patients with chronic rhinosinusitis and nasal polyps. Laryngoscope 2019, 129, 1969–1975. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wahid, N.; Smith, R.; Clark, A.; Salam, M.; Philpott, C. The Socioeconomic Cost of Chronic Rhinosinusitis Study. Rhinology 2020, 58, 112–125. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Zhou, A.; Emmanuel, B.; Garcia, D.; Rosta, E. Systematic literature review of humanistic and economic burdens of chronic rhinosinusitis with nasal polyposis. Curr. Med. Res. Opin. 2020, 36, 1913–1926. [Google Scholar] [CrossRef]
- Bachert, C.; Bhattacharyya, N.; Desrosiers, M.; Khan, A.H. Burden of Disease in Chronic Rhinosinusitis with Nasal Polyps. J. Asthma Allergy 2021, 14, 127–134. [Google Scholar] [CrossRef]
- Zhao, Q.; Yu, L.; Jin, P.; Ma, W.; Duan, S.; Luo, H. A Comprehensive Investigation of the Demographics, Treatments, Comorbidities, and Disease Burden of Chronic Rhinosinusitis with Nasal Polyposis Patients: A Descriptive Analysis. Ann. Transl. Med. 2022, 10, 150. [Google Scholar] [CrossRef]
- Parasher, A.K.; Gliksman, M.; Segarra, D.; Lin, T.; Rudmik, L.; Quast, T. Economic Evaluation of Dupilumab Versus Endoscopic Sinus Surgery for the Treatment of Chronic Rhinosinusitis with Nasal Polyps. Int. Forum Allergy Rhinol. 2022, 12, 813–820. [Google Scholar] [CrossRef]
- Tumlin, P.; Al-Asadi, Z.; Turner, M.; Ramadan, H.H.; Makary, C.A. Gender Differences in Quality-of-Life Outcome in Patients Undergoing Endoscopic Endonasal Skull Base Surgery. J. Neurol. Surg. B Skull Base 2023, 85, e80–e85. [Google Scholar] [CrossRef]
- Hoffman, V.; Mulder, K.; Topuria, I.; Gawlicka, A.K.; Kallman, J.E. Reduction in healthcare resource use through 24 months following sinus surgery with steroid-eluting implants in chronic rhinosinusitis patients with and without nasal polyps: A real-world study. Curr. Med. Res. Opin. 2023, 39, 1613–1619. [Google Scholar] [CrossRef]
- Hildenbrand, T.; Milger-Kneidinger, K.; Baumann, I.; Weber, R. The Diagnosis and Treatment of Chronic Rhinosinusitis. Dtsch. Arztebl. Int. 2024, 121, 643–653. [Google Scholar] [CrossRef]
- Liu, P.; Liu, M.; Sun, Y.; Yu, M.; Lei, W.; Xu, Y. Osteopontin as a Novel Biomarker in Distinguishing Chronic Rhinosinusitis with Nasal Polyp Endotypes and Predicting Disease Severity. Int. Arch. Allergy Immunol. 2024, 186, 473–483. [Google Scholar] [CrossRef] [PubMed]
- Otten, J.J.; Fokkens, W.J.; Reitsma, S. Olfactory Dysfunction in Chronic Rhinosinusitis with Nasal Polyps: Effect of Treatment with Emphasis on Biological Therapy. Curr. Allergy Asthma Rep. 2024, 25, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Gomes, P.M.; Cabral, D.C.; Barreto, J.; Carção, A.A.; Duarte, D.; Penêda, J.F. Chronic Rhinosinusitis with Nasal Polyps: Predictors of Recurrence 5 Years After Surgery. Acta Oto-Laryngol. 2024, 144, 635–640. [Google Scholar] [CrossRef] [PubMed]
- Garvey, E.; Duffy, A.; Tekumalla, S.; Naimi, B.; Kahn, C.; Yang, A.; Urdang, Z.; Farquhar, D.; Rosen, M.; Nyquist, G.G.; et al. Obstructive Sleep Apnea and Chronic Rhinosinusitis: Understanding the Impact of OSA on CRS Disease Burden. Otolaryngol. Head Neck Surg. 2024, 171, 1879–1886. [Google Scholar] [CrossRef]
- Peric, A.; Djeric, D. Immunological Mechanisms of Sensorineural Hearing Impairment in Patients with Different Clinical Phenotypes of Chronic Rhinosinusitis: A Narrative Review. Immunol. Investig. 2025, 54, 396–411. [Google Scholar] [CrossRef]
- Maniaci, A.; Gagliano, C.; Lavalle, S.; van der Poel, N.; La Via, L.; Longo, A.; Russo, A.; Zeppieri, M. Ocular Manifestations of Pediatric Rhinosinusitis: A Comprehensive Review. Diseases 2024, 12, 239. [Google Scholar] [CrossRef]
- Rehman, A.; Maqbool, S.; Maaz, M. Does Managing Patients with Chronic Rhinosinusitis Improve Their Depression Score? Prospective Study. J. Otolaryngol.-Head Neck Surg. 2024, 53, 19160216241282607. [Google Scholar] [CrossRef]
- Choi, A.; Xu, S.; Luong, A.U.; Wise, S.K. Current Review of Comorbidities in Chronic Rhinosinusitis. Curr. Allergy Asthma Rep. 2024, 25, 1–13. [Google Scholar] [CrossRef]
- Han, J.K.; Silver, J.; Dhangar, I.; Veeranki, P.; Deb, A. Quantifying Corticosteroid Burden in Chronic Rhinosinusitis with Nasal Polyps: A Retrospective US Database Study. Ann. Allergy Asthma Immunol. 2024, 134, 685–693.e5. [Google Scholar] [CrossRef]
- Bhat, A.M.; Soler, Z.M.; Rathi, V.K.; Schlosser, R.J. Spending, Utilization, and Coverage for Chronic Rhinosinusitis with Nasal Polyposis Therapies Among Medicare Advantage Beneficiaries. Int. Forum Allergy Rhinol. 2024, 14, 1643–1646. [Google Scholar] [CrossRef]
- Zhang, J.; Sima, Y.; Shao, S.; Zheng, M.; Zhao, Y.; Gao, F.; Wang, X.; Zhang, L. Cost-Utility and Clinical Outcome Analysis of Surgery or Oral Corticosteroids Treatment in Patients with Chronic Rhinosinusitis with Nasal Polyps. Asia Pac. Allergy 2024, 14, 174–182. [Google Scholar] [CrossRef]
- Blauwblomme, M.; Gevaert, P.; Van Nevel, S.; Riemann, S.; Vandewalle, E.; Holtappels, G.; De Ruyck, N.; Derycke, L.; Eeckels, A.-S.; Vanhee, S.; et al. Case Report: Dual Monoclonal Antibody Therapy in Chronic Rhinosinusitis with Nasal Polyps and Severe Eosinophilic Asthma—A Proteome Analysis. Front. Allergy 2024, 5, 1484931. [Google Scholar] [CrossRef]
- Scheire, S.; Germonpré, S.; Mehuys, E.; Van Tongelen, I.; De Sutter, A.; Steurbaut, S.; Van Hees, T.; Demarche, S.; Lahousse, L.; Gevaert, P.; et al. Rhinitis Control and Medication Use in a Real-World Sample of Patients with Persistent Rhinitis or Rhinosinusitis: A Community Pharmacy Study. J. Allergy Clin. Immunol. Pract. 2024, 12, 1865–1876.e6. [Google Scholar] [CrossRef] [PubMed]
- Umadevi, K.; Bharathi, E.V.; Mooventhan, A.; Nivethitha, L. Eight Months of Integrated Yoga, Hydrotherapy and Acupuncture on Serum IgE Levels and Symptoms Severity of a Patient with Chronic Allergic Rhinosinusitis: A Case Report. J. Bodyw. Mov. Ther. 2024, 39, 541–543. [Google Scholar] [CrossRef]
- Huang, Z.; Xuan, X.; Liu, S.; Lin, J.; Qian, Z.; Chen, L.; Qiu, R.; Cai, Y. A Systematic Review and Meta-Analysis of the Efficacy and Safety of Traditional Chinese Medicine in the Treatment of Rhinosinusitis. Medicine 2024, 103, e40192. [Google Scholar] [CrossRef] [PubMed]
- Giombi, F.; Mercante, G.; Nappi, E.; Marzio, V.; Malvezzi, L. Tailoring the Treatment of Type-2 Related Chronic Rhinosinusitis with or Without Nasal Polyps: A Comparative Analysis of Medical and Surgical Approaches. Curr. Opin. Allergy Clin. Immunol. 2024. [Google Scholar] [CrossRef]
- Talugula, S.; Nyenhuis, S.M.; Eldeirawi, K.; Lee, V.S. Proximity to Traffic Is Associated with Worse Symptom Severity in Patients with Chronic Rhinosinusitis with Nasal Polyps. Am. J. Otolaryngol. 2025, 46, 104580. [Google Scholar] [CrossRef]
- Singh, A.; Zeig-Owens, R.; Webber, M.P.; Mueller, A.K.; Prezant, D.J. Self-Reported Chronic Rhinosinusitis Diagnoses and Symptoms in World Trade Center-Exposed and Non-World Trade Center-Exposed United States Firefighters. Prev. Med. 2025, 191, 108216. [Google Scholar] [CrossRef]
- Thornton, C.S.; Radu, L.; Boechler, N.; Clark, J.; Somayaji, R. Sex and Gender in Rhinosinusitis: A Review. Future Microbiol. 2025, 20, 259–264. [Google Scholar] [CrossRef]
- Tie, K.; Aboueisha, M.A.; Wang, M.; Caradonna, D.S.; Brook, C.D. The Impact of Primary Immunodeficiency on the Severity of Chronic Rhinosinusitis. Am. J. Otolaryngol. 2025, 46, 104541. [Google Scholar] [CrossRef]
- Long, S.; Wang, H.; Li, W.; Hu, L.; Yu, H.; Liu, Z.; Du, K.; Sun, X.; Wang, D.; Li, H. Advancements and Future Directions in Chronic Rhinosinusitis: Understanding Inflammatory Mechanisms (2000–2023). Eur. Arch. Oto-Rhino-Laryngol. 2025, 282, 2899–2909. [Google Scholar] [CrossRef]
- Okano, M.; Yamada, M.; Oka, A. Personalized Medicine in Chronic Rhinosinusitis: Treatable Traits Using Biologics for Unmet Needs. Allergy Asthma Immunol. Res. 2025, 17, 8–21. [Google Scholar] [CrossRef] [PubMed]
- Park, A.C.; Nguyen, T.V.; Chou, L.; Dilley, K.K.; Zhu, Z.; Lal, A.; Nair, P.; Chen, Z.; Kuan, E.C.; Wong, B.J. Variations in Ciliary Beat Frequency Based on Chronic Rhinosinusitis Endotype and Phenotype. Ann. Allergy Asthma Immunol. 2025, 134, 442–447. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.E.; Olonisakin, T.F.; Moore, J.A.; Chiang, S.; Lee, S.E. The Association of TSLP and IL-4 with Patient-Reported Outcome Measures in Chronic Rhinosinusitis with Nasal Polyps. Am. J. Rhinol. Allergy 2025, 39, 118–127. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Simpson, C.A.; Srivastava, M.; Bera, A.; Cappelletti, M.; Suh, J.D.; Wang, M.B.; Beswick, D.M.; Maxim, T.; Basak, S.K.; et al. Biodiversity of the Bacterial and Fungal Microbiome and Associated Inflammatory Cytokine Profile in Chronic Rhinosinusitis. Int. Forum Allergy Rhinol. 2025, 15, 502–512. [Google Scholar] [CrossRef]
- Voruz, F.; Neofytos, D.; Van Delden, C.; Lobrinus, J.; De Vito, C.; Macario, S.; Daskalou, D.; Hsieh, J.W.; Becker, M.; Landis, B.N. The Importance of MRI in the Early Diagnosis of Acute Invasive Fungal Rhinosinusitis. Diagnostics 2025, 15, 311. [Google Scholar] [CrossRef]
- Kallenberger, E.M.; Briggs, E.E.; Nguyen, S.A.; Ripp, A.T.; Duffy, A.N.; Schafer, I.V.; Soler, Z.M.; Mace, J.C.; Smith, T.L.; Schlosser, R.J. Impact of Sinus Surgery on Nasal Discharge in Chronic Rhinosinusitis: A Systematic Review and Meta-Analysis. Int. Forum Allergy Rhinol. 2025, 15, 174–184. [Google Scholar] [CrossRef]
- Peng, L.; Wang, H.; Wang, D.; Wen, Y.; Zhong, H.; Wen, W.; Li, J. Screening for Psychiatric Disorders in Chronic Rhinosinusitis Patients Waiting for Surgery: A Prospective Cross-Sectional Study. Clin. Otolaryngol. 2025, 50, 88–97. [Google Scholar] [CrossRef]
- Cheah, X.Y.; Tan, C.J.W.; Yeo, B.S.Y.; Lim, N.E.; Tan, Q.W.; Teoh, M.Z.X.; Tan, B.K.J.; Rawtaer, I.; Teo, N.W.Y.; Charn, T.C. The Impact of Sinus Surgery for Chronic Rhinosinusitis on Concomitant Depression and Anxiety Symptoms: A Systematic Review and Meta-Analysis. Int. Forum Allergy Rhinol. 2025, 15, 577–584. [Google Scholar] [CrossRef]
- Min, H.K.; Lee, S.; Kim, S.; Son, Y.; Park, J.; Kim, H.J.; Lee, J.; Lee, H.; Smith, L.; Rahmati, M.; et al. Global Incidence and Prevalence of Chronic Rhinosinusitis: A Systematic Review. Clin. Exp. Allergy 2025, 55, 52–66. [Google Scholar] [CrossRef]
- Klooster, B.; Chatterton, K.; Ibrahim, N.; Bernstein, M.C.; Shields, A.L.; Allen, V. Patient-Centered Outcomes for Clinical Trials in Chronic Rhinosinusitis with or Without Nasal Polyps and Allergic Fungal Rhinosinusitis. J. Patient-Rep. Outcomes 2025, 9, 11. [Google Scholar] [CrossRef] [PubMed]
- Chu, M.M.; Garcia, J.T.; Sedaghat, A.R.; Scangas, G.A.; Phillips, K.M. A Cost Utility Analysis for the Management of Acute Exacerbations of Chronic Rhinosinusitis. Int. Forum Allergy Rhinol. 2025, 15, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Soler, Z.M.; Patel, Z.M.; Mullol, J.; Mattos, J.; Nash, S.; Xia, C.; Wang, Z.; Borsos, K.; Corbett, M.; Jacob-Nara, J.A.; et al. Association Between Smell Loss, Disease Burden, and Dupilumab Efficacy in Chronic Rhinosinusitis with Nasal Polyps. Am. J. Rhinol. Allergy 2025, 39, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Almarri, F.K.; Algahtani, S.; Alokby, G.; Alanazi, M.; Alsaleh, S. Practice Patterns of Biologics Prescriptions and Surgery in Chronic Rhinosinusitis with Nasal Polyps. Am. J. Rhinol. Allergy 2024, 39, 49–57. [Google Scholar] [CrossRef]
- Margarita, P.-V.A.; Roberto, C.-M.A. Exercise and Sinonasal Diseases: Key Interactions and Management Pearls. Immunol. Allergy Clin. N. Am. 2025, 45, 1–11. [Google Scholar] [CrossRef]
- Lima, A.D.; Giffoni, R.B.; Arguelles-Hernandez, J.; Santos, G.; Sena, V.L.; Aguiar, R.S.; Cruz, M.L.; Dalmaschio, M.E.; Nakanishi, M. Effectiveness of Hypertonic Saline Irrigation Following Functional Endoscopic Sinus Surgery: A Systematic Review and Meta-Analysis. Braz. J. Otorhinolaryngol. 2025, 91, 101517. [Google Scholar] [CrossRef]
- Dhamija, R.; Das, N.; Ding, P. Complication Rates Following Endoscopic Sinus Surgery for Chronic Sinusitis. Am. J. Rhinol. Allergy 2025, 39, 197–204. [Google Scholar] [CrossRef]
- Russo, P.; Bassano, E.; Menichetti, M.; Lucidi, D.; Minniti, R.M.; Cigarini, E.; Menabue, S.; Marchioni, D.; Perano, D.; Ghidini, A. Long-Term Effectiveness of Dupilumab in Severe Uncontrolled Chronic Rhinosinusitis with Nasal Polyps: A Multicenter Retrospective Study. Am. J. Rhinol. Allergy 2025, 39, 175–180. [Google Scholar] [CrossRef]
- Jin, Z.; Yan, B.; Zhang, L.; Wang, C. Biological Therapy in Chronic Rhinosinusitis with Nasal Polyps. Expert Rev. Clin. Immunol. 2025, 21, 473–492. [Google Scholar] [CrossRef]
- Bakhshaee, M.; Hosseini, S.S.; Zanghaei, A.; Najafi, M.N.; Bavi, F.; Roshanzamir, I. The Auxiliary Effect of Vitamin D in the Treatment of Chronic Rhinosinusitis with Nasal Polyposis, a Clinical Trial. Acta Oto-Laryngol. 2025, 145, 313–318. [Google Scholar] [CrossRef]
- Lommatzsch, M.; Blumchen, K.; Beck, L.A.; Bousquet, J.; Brusselle, G.G.; Fokkens, W.J.; Hamelmann, E.; Lau, S.; Ott, H.; Pfaar, O.; et al. Roads to Remission: Evolving Treatment Concepts in Type 2 Inflammatory Diseases. eClinicalMedicine 2025, 80, 103050. [Google Scholar] [CrossRef] [PubMed]
- Levi, L.; Ye, K.; Fieux, M.; Renteria, A.; Lin, S.; Xing, L.; Ayoub, N.F.; Patel, Z.M.; Nayak, J.V.; Hwang, P.H.; et al. Machine Learning of Endoscopy Images to Identify, Classify, and Segment Sinonasal Masses. Int. Forum Allergy Rhinol. 2025, 15, 524–535. [Google Scholar] [CrossRef] [PubMed]
- Koo, M.-S.; Moon, S.; Rha, M.-S. Mucosal Inflammatory Memory in Chronic Rhinosinusitis. Cells 2024, 13, 1947. [Google Scholar] [CrossRef]
- Stan, C.; Blebea, C.; Tănase, M.I.; Ujvary, P.L.; Tănase, M.; Pop, S.S.; Maniu, A.A.; Cosgarea, M.; Rădeanu, D.G. Exploring the Ovine Anatomy: A Comprehensive Study of the Sheep’s Head for Basic Training in Functional Endoscopic Sinus Surgery. Cureus 2024, 16, e53529. [Google Scholar] [CrossRef]
- Stan, C.; Ujvary, P.L.; Blebea, C.; Tănase, M.I.; Tănase, M.; Pop, S.S.; Maniu, A.A.; Cosgarea, M.; Rădeanu, D.G. Hand Motion Analysis Using Accelerometer-Based Sensors and Sheep’s Head Model for Basic Training in Functional Endoscopic Sinus Surgery. Cureus 2024, 16, e59725. [Google Scholar] [CrossRef] [PubMed]
- Edalati, S.; Sharma, S.; Guda, R.; Vasan, V.; Mohamed, S.; Gidumal, S.; Govindaraj, S.; Iloreta, A.M. Assessing Adult Sinusitis Guidelines: A Comparative Analysis of AAO-HNS and AI Chatbots. Am. J. Otolaryngol. 2025, 46, 104563. [Google Scholar] [CrossRef]
- Orlandi, R.R.; Kingdom, T.T.; Smith, T.L.; Bleier, B.; DeCondeD, A.; Luong, A.U.; Poetker, D.M.; Soler, Z.; Welch, K.C.; Wise, S.K.; et al. International consensus statement on allergy and rhinology: Rhinosinusitis 2021. Int. Forum Allergy Rhinol. 2021, 11, 213–739, Erratum in Int. Forum Allergy Rhinol. 2022, 12, 974. https://doi.org/10.1002/alr.22987.. [Google Scholar] [CrossRef]
- Abdelwahab, S.I.; Taha, M.M.E.; Jerah, A.A.; Farasani, A.; Abdullah, S.M.; Aljahdali, I.A.; Ibrahim, R.; Oraibi, O.; Oraibi, B.; Alfaifi, H.A.; et al. Artificial Intelligence and Microbiome Research: Evolution of Hotspots, Research Trends, and Thematic-Based Narrative Review. Cell. Mol. Biol. 2024, 70, 182–192. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).