Anti-Dyslipidemic and Anti-Diabetic Properties of Corosolic Acid: A Narrative Review
Abstract
:1. Introduction
2. Molecular Mechanisms
2.1. Pharmacokinetics
2.2. Biological Properties
3. Effect of Corosolic Acid: Preclinical Evidence
3.1. Anti-Inflammatory and Anti-Oxidant Effects
3.2. Anti-Diabetic Activity
3.3. Anti-Tumor Activity
3.4. Neuroprotective Properties
3.5. Effects on Dyslipidemia and Hepatic Steatosis
4. Effect of Corosolic Acid: Clinical Evidence
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. Noncommunicable Diseases: Risk Factors. The Global Health Observatory. 2021. Available online: https://www.who.int/data/gho/data/themes/topics/topic-details/GHO/ncd-risk-factors (accessed on 15 July 2023).
- Global Health Data Exchange. GBD Results Tool. Institute for Health Metrics and Evaluation. 2021. Available online: http://ghdx.healthdata.org/gbd-results-tool (accessed on 15 July 2023).
- Younossi, Z.M.; Koenig, A.B.; Abdelatif, D.; Fazel, Y.; Henry, L.; Wymer, M. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 2016, 64, 73–84. [Google Scholar] [CrossRef] [PubMed]
- Saklayen, M.G. The Global Epidemic of the Metabolic Syndrome. Curr. Hypertens. Rep. 2018, 20, 12. [Google Scholar] [CrossRef]
- Available online: https://www.niddk.nih.gov/health-information/health-communication-programs/ndep/health-care-professionals/game-plan/facts-statistics/Pages/index.aspx (accessed on 15 July 2023).
- Glovaci, D.; Fan, W.; Wong, N.D. Epidemiology of Diabetes Mellitus and Cardiovascular Disease. Curr. Cardiol. Rep. 2019, 21, 21. [Google Scholar] [CrossRef] [PubMed]
- Berberich, A.J.; Hegele, R.A. A Modern Approach to Dyslipidemia. Endocr. Rev. 2022, 43, 611–653. [Google Scholar] [CrossRef]
- McCracken, E.; Monaghan, M.; Sreenivasan, S. Pathophysiology of the metabolic syndrome. Clin. Dermatol. 2018, 36, 14–20. [Google Scholar] [CrossRef]
- Ahmad, E.; Lim, S.; Lamptey, R.; Webb, D.R.; Davies, M.J. Type 2 diabetes. Lancet 2022, 400, 1803–1820. [Google Scholar] [CrossRef] [PubMed]
- Colonna, S.; Fulk, G.; Marangoni, F. The food for the health. In The Health Foods; Springer: Milan, Italy, 2013; pp. 211–220. [Google Scholar]
- Cicero, A.F.G.; Colletti, A. Role of phytochemicals in the management of metabolic syndrome. Phytomedicine 2016, 23, 1134–1144. [Google Scholar] [CrossRef] [PubMed]
- Serafini, M.; Peluso, I.; Raguzzini, A. Flavonoids as anti-inflammatory agents. Proc. Nutr. Soc. 2010, 69, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. Omega-3 Fatty Acids in Inflammation and Autoimmune Diseases. J. Am. Coll. Nutr. 2002, 21, 495–505. [Google Scholar] [CrossRef]
- Silva, E.P., Jr.; Borges, L.S.; Mendes-Da-Silva, C.; Hirabara, S.M.; Lambertucci, R.H. l-Arginine supplementation improves rats’ antioxidant system and exercise performance. Free Radic. Res. 2017, 51, 281–293. [Google Scholar] [CrossRef]
- Leh, H.E.; Lee, L.K. Lycopene: A Potent Antioxidant for the Amelioration of Type II Diabetes Mellitus. Molecules 2022, 27, 2335. [Google Scholar] [CrossRef]
- Zhou, D.; Luo, M.; Huang, S.; Saimaiti, A.; Shang, A.; Gan, R.; Li, H. Effects and Mechanisms of Resveratrol on Aging and Age-Related Diseases. Oxidative Med. Cell. Longev. 2021, 2021, 9932218. [Google Scholar] [CrossRef]
- Borlinghaus, J.; Albrecht, F.; Gruhlke, M.C.H.; Nwachukwu, I.D.; Slusarenko, A.J. Allicin: Chemistry and Biological Properties. Molecules 2014, 19, 12591–12618. [Google Scholar] [CrossRef]
- Xiong, Z.; Cao, X.; Wen, Q.; Chen, Z.; Cheng, Z.; Huang, X.; Zhang, Y.; Long, C.; Zhang, Y.; Huang, Z. An overview of the bioactivity of monacolin K/lovastatin. Food Chem. Toxicol. 2019, 131, 110585. [Google Scholar] [CrossRef]
- Bevilacqua, A.; Bizzarri, M. Inositols in Insulin Signaling and Glucose Metabolism. Int. J. Endocrinol. 2018, 2018, 1968450. [Google Scholar] [CrossRef]
- Song, D.; Hao, J.; Fan, D. Biological properties and clinical applications of berberine. Front. Med. 2020, 14, 564–582. [Google Scholar] [CrossRef]
- Ciecierska, A.; Drywień, M.E.; Hamulka, J.; Sadkowski, T. Nutraceutical functions of beta-glucans in human nutrition. Rocz. Państwowego Zakładu Hig. 2019, 70, 315–324. [Google Scholar] [CrossRef]
- Khan, N.; Mukhtar, H. Tea Polyphenols in Promotion of Human Health. Nutrients 2018, 11, 39. [Google Scholar] [CrossRef] [PubMed]
- Nattagh-Eshtivani, E.; Barghchi, H.; Pahlavani, N.; Barati, M.; Amiri, Y.; Fadel, A.; Khosravi, M.; Talebi, S.; Arzhang, P.; Ziaei, R.; et al. Biological and pharmacological effects and nutritional impact of phytosterols: A comprehensive review. Phytother. Res. 2022, 36, 299–322. [Google Scholar] [CrossRef] [PubMed]
- Vahabzadeh, M.; Amiri, N.; Karimi, G. Effects of silymarin on metabolic syndrome: A review. J. Sci. Food Agric. 2018, 98, 4816–4823. [Google Scholar] [CrossRef]
- Miyazawa, T.; Burdeos, G.C.; Itaya, M.; Nakagawa, K.; Miyazawa, T. Vitamin E: Regulatory Redox Interactions. IUBMB Life 2019, 71, 430–441. [Google Scholar] [CrossRef]
- Chang, M.X.; Xiong, F. Astaxanthin and its Effects in Inflammatory Responses and Inflammation-Associated Diseases: Recent Advances and Future Directions. Molecules 2020, 25, 5342. [Google Scholar] [CrossRef]
- Peng, Y.; Ao, M.; Dong, B.; Jiang, Y.; Yu, L.; Chen, Z.; Hu, C.; Xu, R. Anti-Inflammatory Effects of Curcumin in the Inflammatory Diseases: Status, Limitations and Countermeasures. Drug Des. Dev. Ther. 2021, 15, 4503–4525. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Xu, M.; Xu, M.; Wang, Y.; Zou, Q.; Xie, S.; Wang, L. Effects of betaine on non-alcoholic liver disease. Nutr. Res. Rev. 2022, 35, 28–38. [Google Scholar] [CrossRef]
- Zhu, R.; Liu, H.; Liu, C.; Wang, L.; Ma, R.; Chen, B.; Li, L.; Niu, J.; Fu, M.; Zhang, D.; et al. Cinnamaldehyde in diabetes: A review of pharmacology, pharmacokinetics and safety. Pharmacol. Res. 2017, 122, 78–89. [Google Scholar] [CrossRef]
- Zhao, J.; Zhou, H.; An, Y.; Shen, K.; Yu, L. Biological effects of corosolic acid as an anti-inflammatory, anti-metabolic syndrome and anti-neoplasic natural compound. Oncol. Lett. 2021, 21, 84. [Google Scholar] [CrossRef]
- Dwyer, J.T.; Coates, P.M.; Smith, M.J. Dietary Supplements: Regulatory Challenges and Research Resources. Nutrients 2018, 10, 41. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, T.; Maruyama, H.; Kasai, R.; Hattori, K.; Takasuga, S.; Hazeki, O.; Yamasaki, K.; Tanaka, T. Ellagitannins from Lagerstroemia speciosa as Activators of Glucose Transport in Fat Cells. Planta Medica 2002, 68, 173–175. [Google Scholar] [CrossRef]
- Qian, X.-P.; Zhang, X.-H.; Sun, L.-N.; Xing, W.-F.; Wang, Y.; Sun, S.-Y.; Ma, M.-Y.; Cheng, Z.-P.; Wu, Z.-D.; Xing, C.; et al. Corosolic acid and its structural analogs: A systematic review of their biological activities and underlying mechanism of action. Phytomedicine 2021, 91, 153696. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Kim, J.-K.; Li, Y.; Liu, X.-Q.; Li, J.; Chen, X. An Extract of Lagerstroemia speciosa L. Has Insulin-Like Glucose Uptake–Stimulatory and Adipocyte Differentiation–Inhibitory Activities in 3T3-L1 Cells. J. Nutr. 2001, 131, 2242–2247. [Google Scholar] [CrossRef]
- Yamada, K.; Hosokawa, M.; Yamada, C.; Watanabe, R.; Fujimoto, S.; Fujiwara, H.; Kunitomo, M.; Miura, T.; Kaneko, T.; Tsuda, K.; et al. Dietary Corosolic Acid Ameliorates Obesity and Hepatic Steatosis in KK-Ay Mice. Biol. Pharm. Bull. 2008, 31, 651–655. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Lu, Y.; Li, P.; Wen, X.; Yang, J. Study on the absorption of corosolic acid in the gastrointestinal tract and its metabolites in rats. Toxicol. Appl. Pharmacol. 2019, 378, 114600. [Google Scholar] [CrossRef]
- Li, J.-J.; Li, Y.; Bai, M.; Tan, J.-F.; Wang, Q.; Yang, J. Simultaneous determination of corosolic acid and euscaphic acid in the plasma of normal and diabetic rat after oral administration of extract of Potentilla discolor Bunge by high-performance liquid chromatography/electrospray ionization mass spectrometry. Biomed. Chromatogr. 2014, 28, 717–724. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, G.R. Drug Metabolism and Variability among Patients in Drug Response. N. Engl. J. Med. 2005, 352, 2211–2221. [Google Scholar] [CrossRef]
- Pinto, N.; Dolan, M.E. Clinically Relevant Genetic Variations in Drug Metabolizing Enzymes. Curr. Drug Metab. 2011, 12, 487–497. [Google Scholar] [CrossRef] [PubMed]
- Stohs, S.J.; Miller, H.; Kaats, G.R. A Review of the Efficacy and Safety of Banaba (Lagerstroemia speciosa L.) and Corosolic Acid. Phytother. Res. 2012, 26, 317–324. [Google Scholar] [CrossRef] [PubMed]
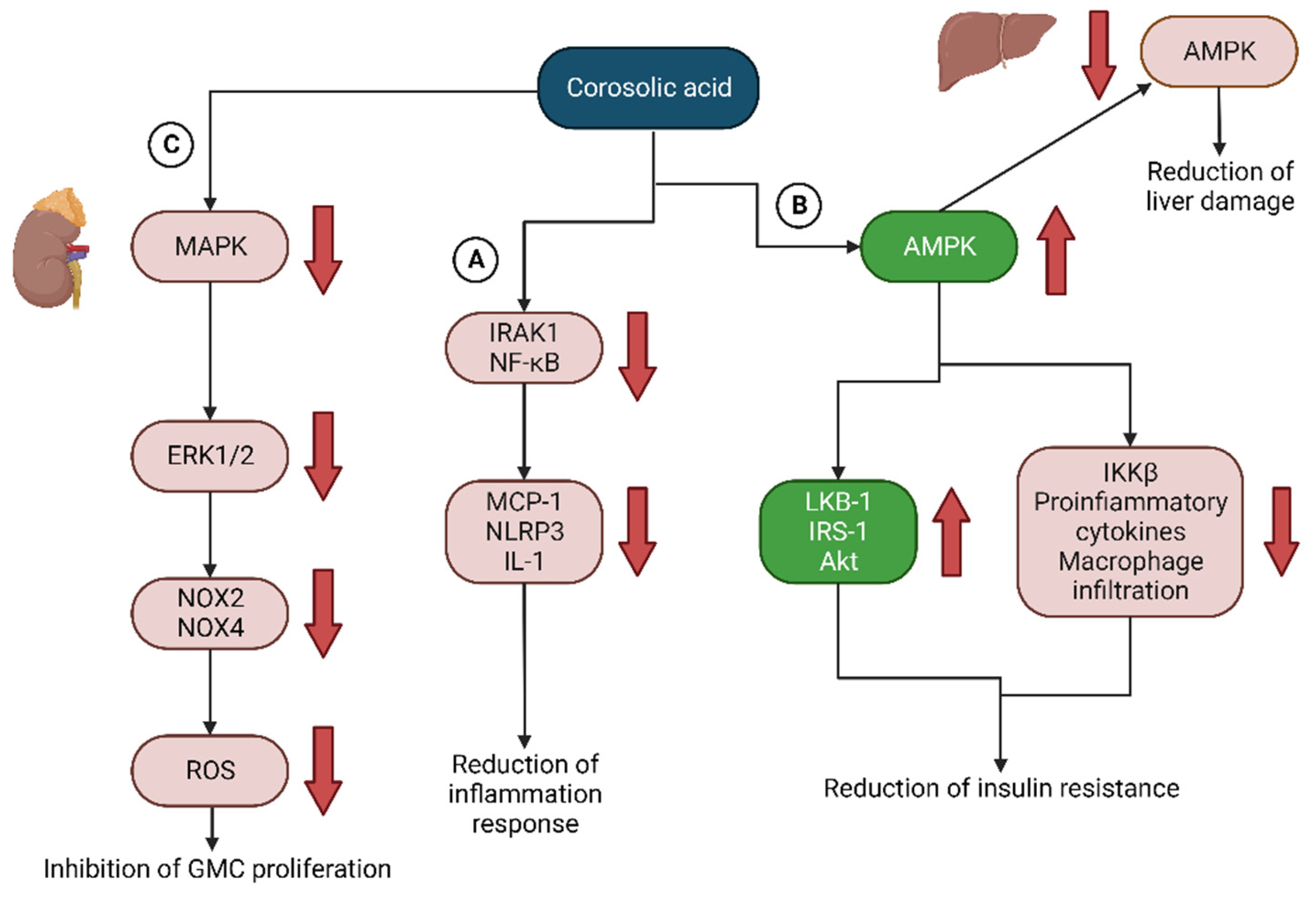
- Kim, S.-J.; Cha, J.-Y.; Kang, H.S.; Lee, J.-H.; Lee, J.Y.; Park, J.-H.; Bae, J.-H.; Song, D.-K.; Im, S.-S. Corosolic acid ameliorates acute inflammation through inhibition of IRAK-1 phosphorylation in macrophages. BMB Rep. 2016, 49, 276–281. [Google Scholar] [CrossRef]
- Lin, K.-M.; Hu, W.; Troutman, T.D.; Jennings, M.; Brewer, T.; Li, X.; Nanda, S.; Cohen, P.; Thomas, J.A.; Pasare, C. IRAK-1 bypasses priming and directly links TLRs to rapid NLRP3 inflammasome activation. Proc. Natl. Acad. Sci. USA 2014, 111, 775–780. [Google Scholar] [CrossRef] [PubMed]
- Li, X.-Q.; Tian, W.; Liu, X.-X.; Zhang, K.; Huo, J.-C.; Liu, W.-J.; Li, P.; Xiao, X.; Zhao, M.-G.; Cao, W. Corosolic acid inhibits the proliferation of glomerular mesangial cells and protects against diabetic renal damage. Sci. Rep. 2016, 6, 26854. [Google Scholar] [CrossRef]
- Guo, X.; Cui, R.; Zhao, J.; Mo, R.; Peng, L.; Yan, M. Corosolic acid protects hepatocytes against ethanol-induced damage by modulating mitogen-activated protein kinases and activating autophagy. Eur. J. Pharmacol. 2016, 791, 578–588. [Google Scholar] [CrossRef]
- Yamaguchi, Y.; Yamada, K.; Yoshikawa, N.; Nakamura, K.; Haginaka, J.; Kunitomo, M. Corosolic acid prevents oxidative stress, inflammation and hypertension in SHR/NDmcr-cp rats, a model of metabolic syndrome. Life Sci. 2006, 79, 2474–2479. [Google Scholar] [CrossRef] [PubMed]
- Alkholifi, F.K.; Devi, S.; Yusufoglu, H.S.; Alam, A. The Cardioprotective Effect of Corosolic Acid in the Diabetic Rats: A Possible Mechanism of the PPAR-γ Pathway. Molecules 2023, 28, 929. [Google Scholar] [CrossRef]
- Takagi, S.; Miura, T.; Ishibashi, C.; Kawata, T.; Ishihara, E.; Gu, Y.; Ishida, T. Effect of Corosolic Acid on the Hydrolysis of Disaccharides. J. Nutr. Sci. Vitaminol. 2008, 54, 266–268. [Google Scholar] [CrossRef]
- Ni, M.; Pan, J.; Hu, X.; Gong, D.; Zhang, G. Inhibitory effect of corosolic acid on α -glucosidase: Kinetics, interaction mechanism, and molecular simulation. J. Sci. Food Agric. 2019, 99, 5881–5889. [Google Scholar] [CrossRef]
- Zhang, B.-W.; Xing, Y.; Wen, C.; Yu, X.-X.; Sun, W.-L.; Xiu, Z.-L.; Dong, Y.-S. Pentacyclic triterpenes as α-glucosidase and α-amylase inhibitors: Structure-activity relationships and the synergism with acarbose. Bioorg. Med. Chem. Lett. 2017, 27, 5065–5070. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Zhang, W.; Zhou, Y.-Y.; Zhang, Y.-N.; Li, J.-Y.; Hu, L.-H.; Li, J. Corosolic acid stimulates glucose uptake via enhancing insulin receptor phosphorylation. Eur. J. Pharmacol. 2008, 584, 21–29. [Google Scholar] [CrossRef]
- White, M.F. The insulin signalling system and the IRS proteins. Diabetologia 1997, 40, S2–S17. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, P.R.; Withers, D.J.; Siddle, K. Phosphoinositide 3-kinase: The key switch mechanism in insulin signalling. Biochem. J. 1998, 333 Pt 3, 471–490. [Google Scholar] [CrossRef]
- Miura, T.; Itoh, Y.; Kaneko, T.; Ueda, N.; Ishida, T.; Fukushima, M.; Matsuyama, F.; Seino, Y. Corosolic Acid Induces GLUT4 Translocation in Genetically Type 2 Diabetic Mice. Biol. Pharm. Bull. 2004, 27, 1103–1105. [Google Scholar] [CrossRef] [PubMed]
- Dubois, M.-J.; Bergeron, S.; Kim, H.-J.; Dombrowski, L.; Perreault, M.; Fournès, B.; Faure, R.; Olivier, M.; Beauchemin, N.; Shulman, G.I.; et al. The SHP-1 protein tyrosine phosphatase negatively modulates glucose homeostasis. Nat. Med. 2006, 12, 549–556. [Google Scholar] [CrossRef]
- Ahmad, F.; Considine, R.V.; Bauer, T.L.; Ohannesian, J.P.; Marco, C.C.; Goldstein, B.J. Improved sensitivity to insulin in obese subjects following weight loss is accompanied by reduced protein-tyrosine phosphatases in adipose tissue. Metabolism 1997, 46, 1140–1145. [Google Scholar] [CrossRef]
- Asante-Appiah, E.; Kennedy, B.P. Protein tyrosine phosphatases: The quest for negative regulators of insulin action. Am. J. Physiol. Endocrinol. Metab. 2003, 284, E663–E670. [Google Scholar] [CrossRef]
- Okar, D.A.; Lange, A.J. Fructose-2,6-bisphosphate and control of carbohydrate metabolism in eukaryotes. BioFactors 1999, 10, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.; Hosokawa, M.; Fujimoto, S.; Fujiwara, H.; Fujita, Y.; Harada, N.; Yamada, C.; Fukushima, M.; Ueda, N.; Kaneko, T.; et al. Effect of corosolic acid on gluconeogenesis in rat liver. Diabetes Res. Clin. Pract. 2008, 80, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Davies, G.F.; Khandelwal, R.L.; Wu, L.; Juurlink, B.H.; Roesler, W.J. Inhibition of phosphoenolpyruvate carboxykinase (PEPCK) gene expression by troglitazone: A peroxisome proliferator-activated receptor-γ (PPARγ)-independent, antioxidant-related mechanism. Biochem. Pharmacol. 2001, 62, 1071–1079. [Google Scholar] [CrossRef]
- Xu, S.; Wang, G.; Peng, W.; Xu, Y.; Zhang, Y.; Ge, Y.; Jing, Y.; Gong, Z. Corosolic acid isolated from Eriobotrya japonica leaves reduces glucose level in human hepatocellular carcinoma cells, zebrafish and rats. Sci. Rep. 2019, 9, 4388. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Leng, J.; Li, J.-J.; Tang, J.-F.; Li, Y.; Liu, B.-L.; Wen, X.-D. Corosolic acid inhibits adipose tissue inflammation and ameliorates insulin resistance via AMPK activation in high-fat fed mice. Phytomedicine 2016, 23, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.S.; Cha, E.Y.; Thuong, P.T.; Kim, J.Y.; Ahn, M.S.; Sul, J.Y. Down-Regulation of Human Epidermal Growth Factor Receptor 2/neu Oncogene by Corosolic Acid Induces Cell Cycle Arrest and Apoptosis in NCI-N87 Human Gastric Cancer Cells. Biol. Pharm. Bull. 2010, 33, 931–937. [Google Scholar] [CrossRef]
- Yang, J.; Wu, R.; Li, W.; Gao, L.; Yang, Y.; Li, P.; Kong, A.-N. The triterpenoid corosolic acid blocks transformation and epigenetically reactivates Nrf2 in TRAMP-C1 prostate cells. Mol. Carcinog. 2018, 57, 512–521. [Google Scholar] [CrossRef]
- Nho, K.J.; Chun, J.M.; Kim, H.K. Corosolic acid induces apoptotic cell death in human lung adenocarcinoma A549 cells in vitro. Food Chem. Toxicol. 2013, 56, 8–17. [Google Scholar] [CrossRef]
- Zhang, L.; Sui, S.; Wang, S.; Sun, J. Neuroprotective Effect of Corosolic Acid Against Cerebral Ischemia-Reperfusion Injury in Experimental Rats. J. Oleo Sci. 2022, 71, 1501–1510. [Google Scholar] [CrossRef]
- Takagi, S.; Miura, T.; Ishihara, E.; Ishida, T.; Chinzei, Y. Effect of corosolic acid on dietary hypercholesterolemia and hepatic steatosis in KK-Ay diabetic mice. Biomed. Res. 2010, 31, 213–218. [Google Scholar] [CrossRef] [PubMed]
- Chou, C.J.; Haluzik, M.; Gregory, C.; Dietz, K.R.; Vinson, C.; Gavrilova, O.; Reitman, M.L. WY14,643, a Peroxisome Proliferator-activated Receptor α (PPARα) Agonist, Improves Hepatic and Muscle Steatosis and Reverses Insulin Resistance in Lipoatrophic A-ZIP/F-1 Mice. J. Biol. Chem. 2002, 277, 24484–24489. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, T.; Kamon, J.; Waki, H.; Murakami, K.; Motojima, K.; Komeda, K.; Ide, T.; Kubota, N.; Terauchi, Y.; Tobe, K.; et al. The Mechanisms by Which Both Heterozygous Peroxisome Proliferator-activated Receptor γ (PPARγ) Deficiency and PPARγ Agonist Improve Insulin Resistance. J. Biol. Chem. 2001, 276, 41245–41254. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Cui, Z.; Gao, X.; Liu, H.; Wang, L.; Gong, J.; Wang, A.; Zhang, J.; Ma, Q.; Huang, Y.; et al. Corosolic acid ameliorates non-alcoholic steatohepatitis induced by high-fat diet and carbon tetrachloride by regulating TGF-β1/Smad2, NF-κB, and AMPK signaling pathways. Phytother. Res. 2021, 35, 5214–5226. [Google Scholar] [CrossRef] [PubMed]
- Singh, T.R.; Ezhilarasan, D. Lagerstroemia speciosa (L.) Pers., ethanolic leaves extract attenuates dapsone-induced liver inflammation in rats. Drug Chem. Toxicol. 2022, 45, 2361–2370. [Google Scholar] [CrossRef] [PubMed]
- Judy, W.V.; Hari, S.P.; Stogsdill, W.W.; Judy, J.S.; Naguib, Y.M.; Passwater, R. Antidiabetic activity of a standardized extract (Glucosol™) from Lagerstroemia speciosa leaves in Type II diabetics: A dose-dependence study. J. Ethnopharmacol. 2003, 87, 115–117. [Google Scholar] [CrossRef]
- Tsuchibe, S.; Kataumi, S.; Mori, M.H.; Mori, H. An inhibitory effect on the increase in the postprandial glucose by banaba extract capsule enriched corosolic acid. J. Integr. Study Diet. Habits 2006, 17, 255–259. [Google Scholar] [CrossRef]
- Miura, T.; Takagi, S.; Ishida, T. Management of Diabetes and Its Complications with Banaba (Lagerstroemia speciosa L.) and Corosolic Acid. Evid.-Based Complement. Altern. Med. 2012, 2012, 871495. [Google Scholar] [CrossRef]
- Fukushima, M.; Matsuyama, F.; Ueda, N.; Egawa, K.; Takemoto, J.; Kajimoto, Y.; Yonaha, N.; Miura, T.; Kaneko, T.; Nishi, Y.; et al. Effect of corosolic acid on postchallenge plasma glucose levels. Diabetes Res. Clin. Pract. 2006, 73, 174–177. [Google Scholar] [CrossRef]
- Choi, M.-S.; Ryu, R.; Seo, Y.R.; Jeong, T.-S.; Shin, D.-H.; Park, Y.B.; Kim, S.R.; Jung, U.J. The beneficial effect of soybean (Glycine max (L.) Merr.) leaf extracts in adults with prediabetes: A randomized placebo controlled trial. Food Funct. 2014, 5, 1621–1630. [Google Scholar] [CrossRef] [PubMed]

| Nutraceutical Compounds | Effects | References |
|---|---|---|
| Flavonoids | ↑ NO availability; ↓ ROS formation; ET-1 inhibition; ↓ ACE activity; anti-inflammatory activity; ↑ insulin sensitivity | Serafini et al., 2010 [12] |
| Poly-unsaturated Fatty Acids (PUFAs) | Anti-inflammatory activity; ↑ PG vasodilators; ↑ NO synthase; ↓ insulin resistance | Simopoulos, 2002 [13] |
| L-Arginine | ↑ NO availability; anti-oxidant action | Silva et al., 2017 [14] |
| Lycopene | Anti-oxidant action; free radical scavenger | Leh and Lee, 2022 [15] |
| Resveratrol | Anti-oxidant action; anti-inflammatory activity; ↑ NO availability | Zhou et al., 2021 [16] |
| Allicin | ↑ NO availability; ↓ ACE activity; ↑ insulin sensitivity; ↑ HDL-C | Borlinghaus et al., 2014 [17] |
| Monacolines | HMG-CoA-reductase inhibition; ↓ LDL-C; ↑ HDL-C | Xiong et al., 2019 [18] |
| Inositols | ↓ LDL-C; ↑ insulin sensitivity | Bevilacqua and Bizzarri, 2018 [19] |
| Berberine | ↑ hepatic LDL-C receptors; ↑ insulin receptors | Song et al., 2020 [20] |
| Beta-glucans | ↓ intestinal absorption of cholesterol; ↑ insulin sensitivity | Ciecierska et al., 2019 [21] |
| Polyphenols | ↓ intestinal absorption of glucose; protective action on β cells; ↓ hepatic glucose production; direct action on GLUT4 | Khan and Mukhtar, 2018 [22] |
| Phytosterols | ↓ intestinal absorption of cholesterol; ↓ LDL-C | Nattagh-Eshtivani et al., 2022 [23] |
| Silymarin | ↑ insulin sensitivity; ↓ hepatic inflammation; ↑ liver protein synthesis | Vahabzadeh et al., 2018 [24] |
| Vitamin E | Anti-oxidant properties; ↑ cell renewal | Miyazawa et al., 2019 [25] |
| Astaxanthin | ↓ lipogenesis; ↓ insulin resistance; ↓ hepatic inflammation | Chang and Xiong, 2020 [26] |
| Curcumin | ↑ insulin sensitivity; anti-oxidant action; anti-inflammatory activity | Peng et al., 2021 [27] |
| Betaine | Hepatoprotection; ↓ homocysteinemia | Chen et al., 2022 [28] |
| Cinnamaldehyde | Hypoglycemic, cholesterol-lowering, and anti-hypertensive activity | Zhu et al., 2017 [29] |
| Corosolic Acid | ↑ insulin sensitivity; ↓ body weight; ↓ LDL-C | Zhao et al., 2021 [30] |
| Authors and Year of Publication | Main Findings |
|---|---|
| Hypercholesterolemia and Hepatic Steatosis | |
| Takagi et al., 2010 [66] | CA reduces hypercholesterolemia and hepatic steatosis caused by dietary cholesterol in T2DM mice and may inhibit the activity of cholesterol acyltransferase in the small intestine |
| Yamada et al., 2008 [58] | CA reduces hepatic steatosis in obese mice by increasing PPAR-α expression in liver and PPAR-γ expression in WAT and ameliorates insulin sensitivity by increasing plasma adiponectin levels and AdipoR1 in WAT |
| Liu et al., 2021 [69] | CA reduces inflammation and fibrosis in NASH by regulating AMPK signaling pathways, NF-κB, and TGF-β1/Smad2 |
| Singh and Ezhilarasan, 2022 [70] | EBLE containing CA and silymarin reduces hepatotoxicity through their anti-inflammatory and anti-oxidant effect |
| Lin et al., 2014 [42] | CA regulates IRAK1 phosphorylation via an NF-κB-independent pathway and plays a role in the inhibitory effect on acute inflammation |
| Insulin resistance, metabolic syndrome, and prediabetes | |
| Yang et al., 2016 [61] | CA suppresses phosphorylation of IKKβ and reduces gene expression of pro-inflammatory cytokines and enhances insulin signal transduction through modification of Ser/Thr phosphorylation of IRS-1 and Akt and stimulating the AMPK signaling pathway in adipose tissue |
| Yamaguchi et al., 2006 [45] | CA ameliorates hypertension, oxidative stress, and the inflammatory state in mice with metabolic syndrome |
| Diabetes | |
| Ni et al., 2019 [48] | CA could exert an inhibitory effect on α-glucosidase in a non-competitive and reversible manner through binding to the enzyme and causing a conformational change that interferes with its catalytic action |
| Zhang et al., 2017 [49] | CA in combination with acarbose inhibits α-amylase and α-glucosidase in a non-competitive manner |
| Xu et al., 2019 [60] | CA ameliorates hyperglycemia, hyperlipidemia, and insulin resistance in T2DM models through decreasing the expression of PEPCK and other genes involved in glucose metabolism, oxidative stress, and inflammation related to T2DM |
| Cardioprotective effects in diabetes | |
| Alkholifi et al., 2023 [46] | CA through the PPAR-γ pathway exerts cardioprotective and anti-oxidant effects on myocardial tissue in diabetic mice with acute myocardial infarction |
| Kidney protection in diabetes | |
| Li et al., 2016 [43] | CA inhibits the proliferation of diabetic glomerular mesangial cells via NADPH/ERK1/2 and p38 MAPK signaling pathways and prevents renal damage in diabetic animals |
| Authors and Year of Publication | Main Findings |
|---|---|
| Judy et al., 2003 [71] | Banaba extract containing 1% CA results in a 30% reduction in blood glucose levels after 2 weeks of treatment in patients with T2DM |
| Tsuchibe et al., 2006 [72] | 10 mg of CA from Banaba extract reduces fasting and postprandial 1 h blood glucose by 12 percent in addition to resulting in a reduction in body weight of about 3 kg after 2 weeks of treatment in patients with impaired fasting blood glucose |
| Xu et al., 2008 (unpublished) | 10 mg of CA reduced fasting and postprandial 2 h blood glucose levels by 10% after one month of treatment in patients with T2DM. In addition, a reduction in symptoms associated with diabetes was observed |
| Fukushima et al., 2006 [74] | 10 mg of CA before a 75 g oral glucose tolerance test reduces blood glucose levels at 60, 90, and 120 min in patients with prediabetes or diabetes |
| Choi et al., 2014 [75] | Banaba extract containing 0.3% CA reduces fasting blood glucose, HbA1c, HOMA-IR, AST, ALT, and blood triglyceride levels in overweight and prediabetes patients |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cannarella, R.; Garofalo, V.; Calogero, A.E. Anti-Dyslipidemic and Anti-Diabetic Properties of Corosolic Acid: A Narrative Review. Endocrines 2023, 4, 616-629. https://doi.org/10.3390/endocrines4030044
Cannarella R, Garofalo V, Calogero AE. Anti-Dyslipidemic and Anti-Diabetic Properties of Corosolic Acid: A Narrative Review. Endocrines. 2023; 4(3):616-629. https://doi.org/10.3390/endocrines4030044
Chicago/Turabian StyleCannarella, Rossella, Vincenzo Garofalo, and Aldo E. Calogero. 2023. "Anti-Dyslipidemic and Anti-Diabetic Properties of Corosolic Acid: A Narrative Review" Endocrines 4, no. 3: 616-629. https://doi.org/10.3390/endocrines4030044
APA StyleCannarella, R., Garofalo, V., & Calogero, A. E. (2023). Anti-Dyslipidemic and Anti-Diabetic Properties of Corosolic Acid: A Narrative Review. Endocrines, 4(3), 616-629. https://doi.org/10.3390/endocrines4030044








