PRDX4 Potentially Predicts the Postoperative Outcome in Advanced Papillary Thyroid Carcinoma
Abstract
1. Introduction
2. Methods
2.1. Patients and Pathological Specimens
2.2. Dispensing/Immunostaining for PRDX4 and Secondary Antibody
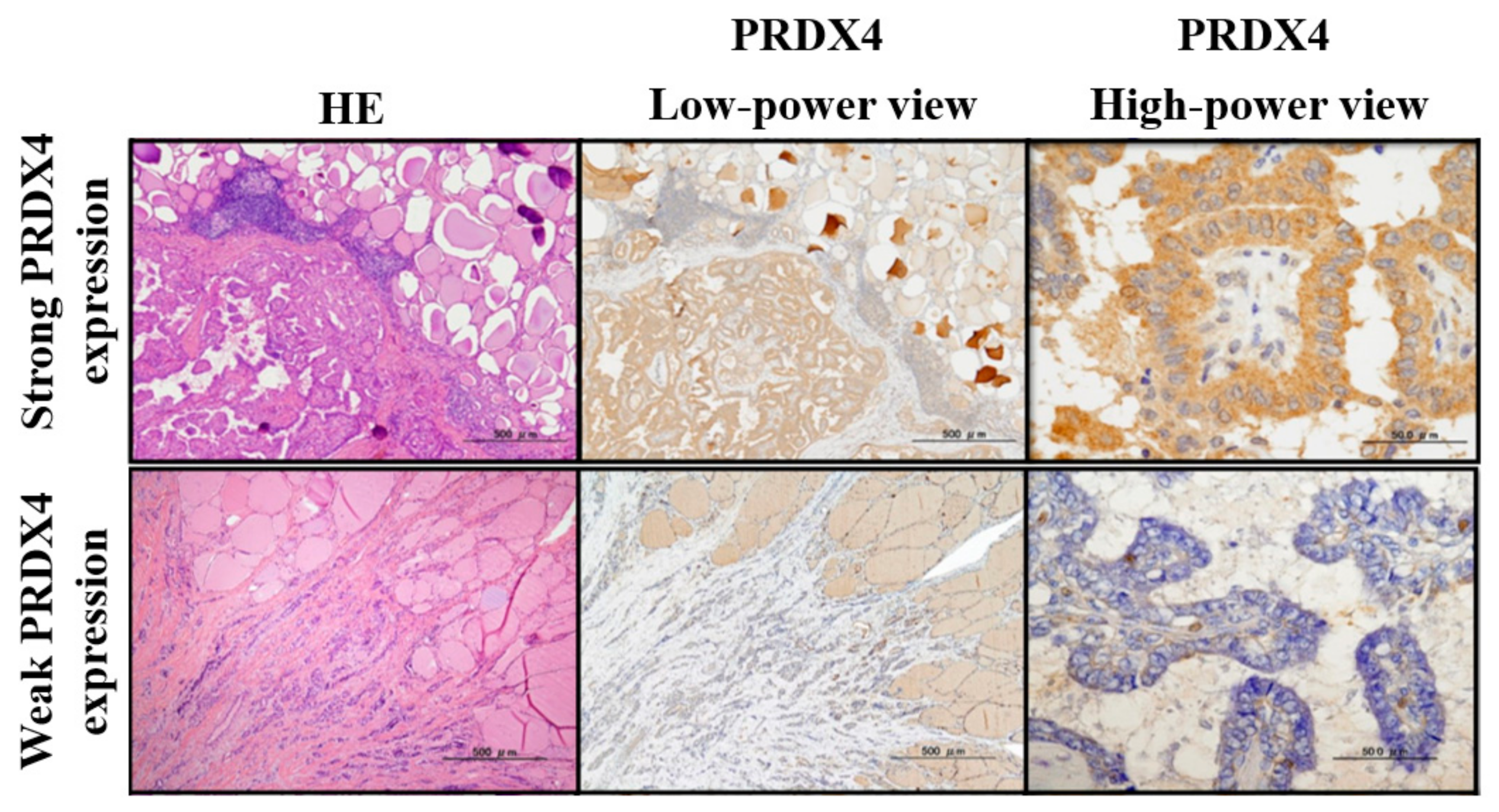
2.3. Intensity of Immunostaining
2.4. Cell Culture
2.5. Cell Transfection
2.6. Western Blotting
2.7. Cell Proliferation Assay
2.8. Statistical Analysis
3. Results
3.1. Patient Characteristics
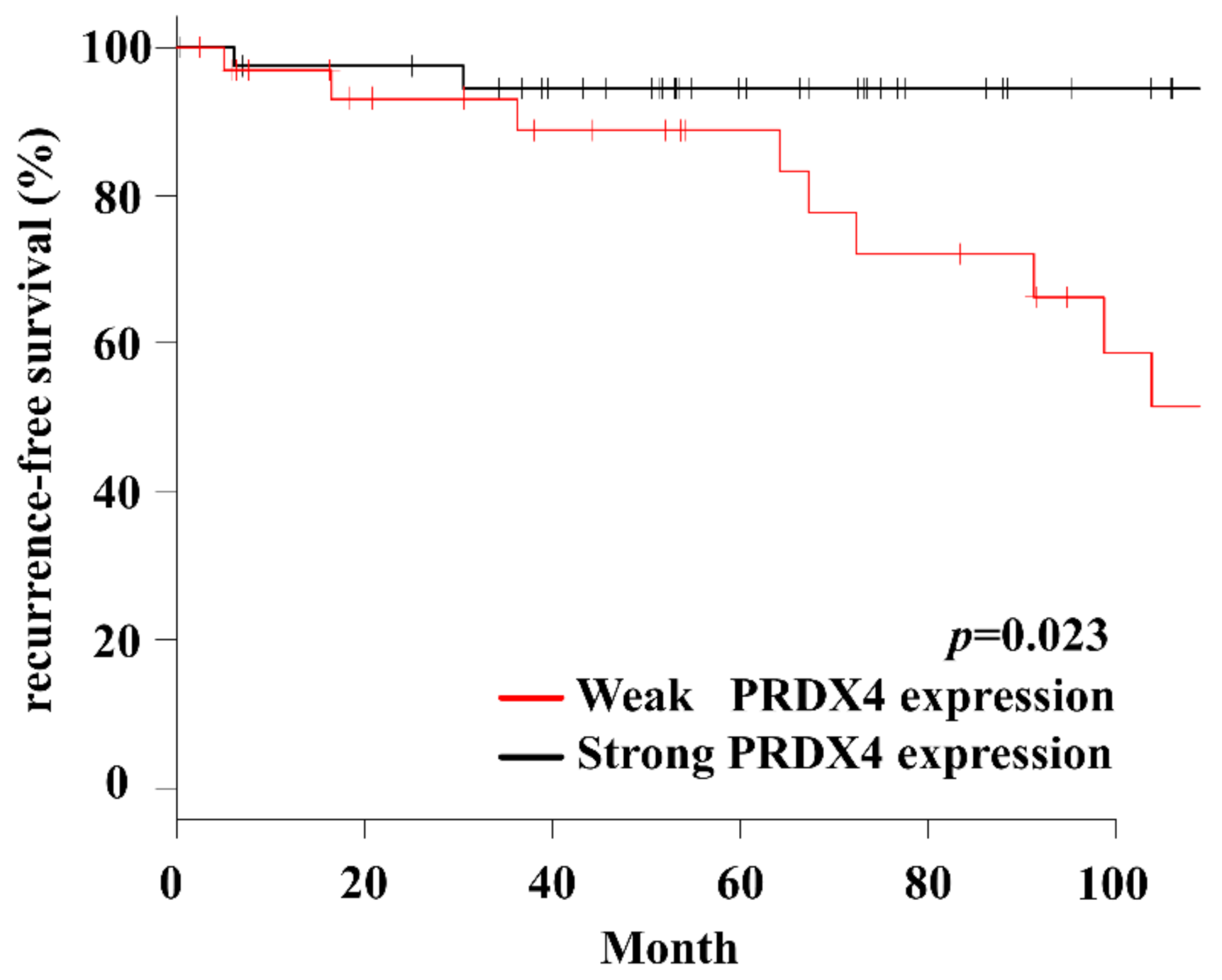
3.2. Relationship between the PRDX4 Expression Rate and Clinicopathological Variables and Disease-Free Recurrence Period
3.3. Cox Proportional Hazards Regression Analysis
| Univariate | Multivariate | |||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p-Value | HR | 95% CI | p-Value | |
| PRDX4 < 30% | 4.88 | 1.04–22.8 | 0.04 | 7.52 | 1.24–42.6 | 0.02 |
| Vascular invasion | 1.72 | 0.52–5.67 | 0.36 | 9.00 | 0.23–358.8 | 0.24 |
| Lymphatic invasion | 1.26 | 0.38–4.12 | 0.71 | 0.14 | 0.003–5.94 | 0.31 |
| Tumor diameter > 2 cm | 2.95 | 0.90–9.68 | 0.07 | 6.96 | 0.90–53.7 | 0.06 |
| Extrathyroid > 1 | 1.21 | 0.32–4.56 | 0.78 | 0.10 | 0.009–1.17 | 0.07 |
| Regional metastasis | 1.48 | 0.39–5.66 | 0.57 | 2.18 | 0.39–12.2 | 0.38 |
| Distant metastasis | 5.85 | 1.39–24.7 | 0.02 | 4.04 | 0.73–22.3 | 0.11 |
| Multifocality | 1.29 | 0.38–4.30 | 0.69 | 0.57 | 0.12–2.66 | 0.48 |
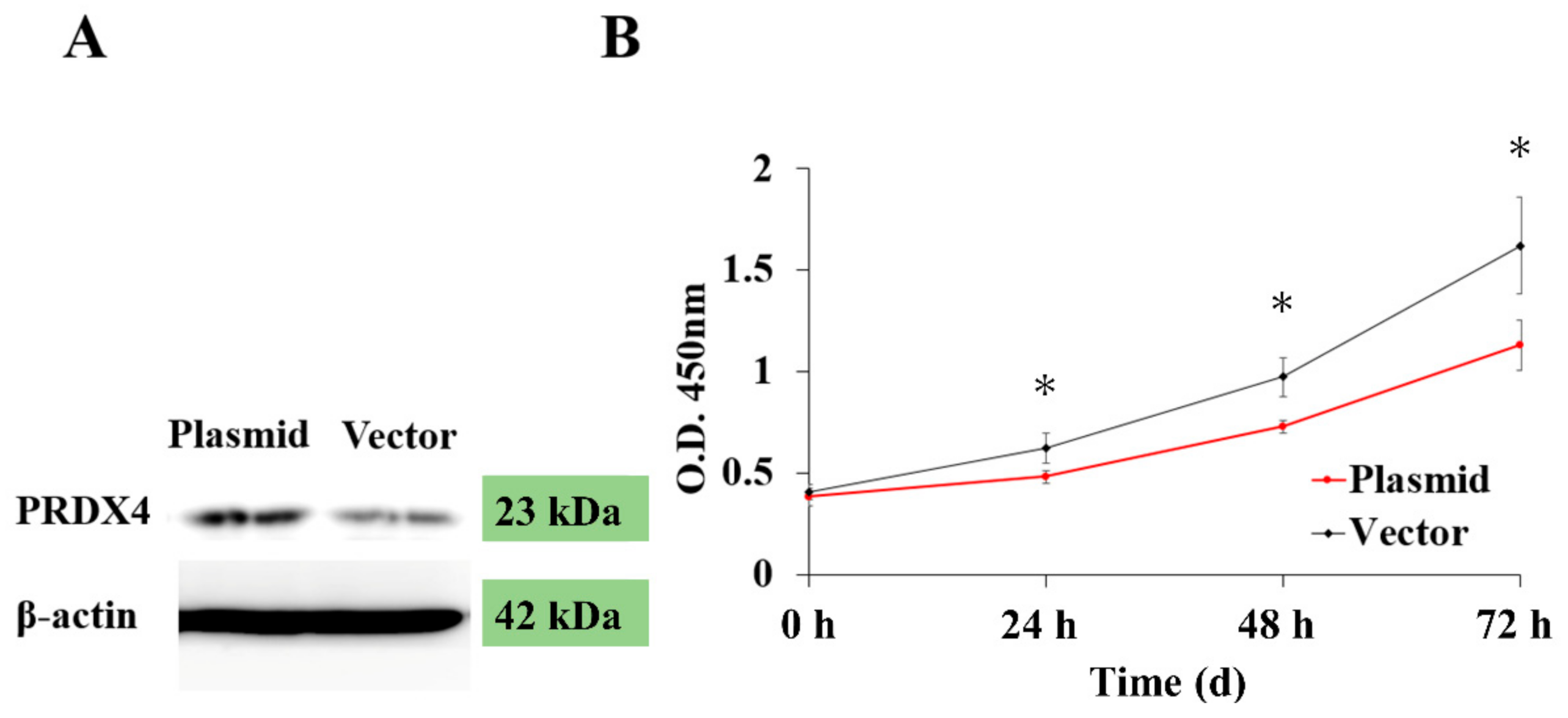
3.4. The Expression of PRDX4 and Cell Proliferation Ability
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Matsuzu, K.; Sugino, K.; Masudo, K.; Nagahama, M.; Kitagawa, W.; Shibuya, H.; Ohkuwa, K.; Uruno, T.; Suzuki, A.; Magoshi, S.; et al. Thyroid Lobectomy for Papillary Thyroid Cancer: Long-term Follow-up Study of 1,088 Cases. World J. Surg. 2014, 38, 68–79. [Google Scholar] [CrossRef] [PubMed]
- Adam, M.A.; Pura, J.; Goffredo, P.; Dinan, M.A.; Hyslop, T.; Reed, S.D.; Scheri, R.P.; Roman, S.A.; Sosa, J.A. Impact of Extent of Surgery on Survival for Papillary Thyroid Cancer Patients Younger Than 45 Years. J. Clin. Endocrinol. Metab. 2015, 100, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Ebina, A.; Sugitani, I.; Fujimoto, Y.; Yamada, K. Risk-adapted management of papillary thyroid carcinoma according to our own risk group classification system: Is thyroid lobectomy the treatment of choice for low-risk patients? Surgery 2014, 156, 1579–1589. [Google Scholar] [CrossRef] [PubMed]
- Mendelsohn, A.H.; Elashoff, D.A.; Abemayor, E.; John, M.A.S. Surgery for Papillary Thyroid Carcinoma. Arch. Otolaryngol.-Head Neck Surg. 2010, 136, 1055–1061. [Google Scholar] [CrossRef] [PubMed]
- Verburg, F.A.; Mäder, U.; Luster, M.; Reiners, C. The effects of the Union for International Cancer Control/American Joint Committee on Cancer Tumour, Node, Metastasis system version 8 on staging of differentiated thyroid cancer: A comparison to version 7. Clin. Endocrinol. 2018, 88, 950–956. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, T.; Ito, Y.; Onoda, N. Thyroid Tumor Practice Guidelines 2018. Off. J. Jpn. Assoc. Endocr. Surg. Jpn. Soc. Thyroid Surg. 2018, 35. [Google Scholar]
- Haugen, B.R.; Alexander, E.K.; Bible, K.C.; Doherty, G.M.; Mandel, S.J.; Nikiforov, Y.E.; Pacini, F.; Randolph, G.W.; Sawka, A.M.; Schlumberger, M.; et al. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 2016, 26, 1–133. [Google Scholar] [CrossRef]
- Fruehauf, J.P.; Meyskens, F.L., Jr. Reactive Oxygen Species: A Breath of Life or Death? Clin. Cancer Res. 2007, 13, 789–794. [Google Scholar] [CrossRef] [PubMed]
- Reuter, S.; Gupta, S.C.; Chaturvedi, M.M.; Aggarwal, B.B. Oxidative stress, inflammation, and cancer: How are they linked? Free Radic. Biol. Med. 2010, 49, 1603–1616. [Google Scholar] [CrossRef] [PubMed]
- El Hassani, R.A.; Buffet, C.; Leboulleux, S.; Dupuy, C. Oxidative stress in thyroid carcinomas: Biological and clinical significance. Endocr.-Relat. Cancer 2019, 26, R131–R143. [Google Scholar] [CrossRef]
- Wang, N.; Feng, J.-F.; Zeng, P.; Yang, Y.-H.; Luo, J.; Yang, Y.-W. Total oxidant/antioxidant status in sera of patients with thyroid cancers. Endocr.-Relat. Cancer 2011, 18, 773–782. [Google Scholar] [CrossRef] [PubMed]
- Metere, A.; Chiesa, C.; Di Cosimo, C.; Fierro, G.; Giacomelli, L.; Pietraforte, D. A novel approach to study oxidative stress in thyroid diseases: A preliminary study. Eur. Rev. Med. Pharmacol. Sci. 2012, 16, 646–652. [Google Scholar]
- Rovcanin, B.R.; Gopcevic, K.R.; Kekic, D.; Zivaljevic, V.R.; Diklic, A.D.; Paunovic, I.R. Papillary Thyroid Carcinoma: A Malignant Tumor with Increased Antioxidant Defense Capacity. Tohoku J. Exp. Med. 2016, 240, 101–111. [Google Scholar] [CrossRef] [PubMed]
- Tabur, S.; Aksoy, S.N.; Korkmaz, H.; Özkaya, M.; Aksoy, N.; Akarsu, E. Investigation of the role of 8-OHdG and oxidative stress in papillary thyroid carcinoma. Tumor Biol. 2014, 36, 2667–2674. [Google Scholar] [CrossRef] [PubMed]
- Nicolussi, A.; D’Inzeo, S.; Capalbo, C.; Giannini, G.; Coppa, A. The role of peroxiredoxins in cancer. Mol. Clin. Oncol. 2017, 6, 139–153. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Kim, Y.S.; Lee, H.L.; Shim, J.Y.; Lee, K.S.; Oh, Y.J.; Shin, S.S.; Choi, Y.H.; Park, K.J.; Park, R.W.; et al. Expression of peroxiredoxin and thioredoxin in human lung cancer and paired normal lung. Respirology 2006, 11, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Goncalves, K.; Sullivan, K.; Phelan, S. Differential Expression and Function of Peroxiredoxin 1 and Peroxiredoxin 6 in Cancerous MCF-7 and Noncancerous MCF-10A Breast Epithelial Cells. Cancer Investig. 2012, 30, 38–47. [Google Scholar] [CrossRef] [PubMed]
- Hoshino, I.; Matsubara, H.; Akutsu, Y.; Nishimori, T.; Yoneyama, Y.; Murakami, K.; Sakata, H.; Matsushita, K.; Ochiai, T. Tumor suppressor Prdx1 is a prognostic factor in esophageal squamous cell carcinoma patients. Oncol. Rep. 2007, 18, 867–871. [Google Scholar] [CrossRef]
- Rolfs, F.; Huber, M.; Gruber, F.; Böhm, F.; Pfister, H.J.; Bochkov, V.N.; Tschachler, E.; Dummer, R.; Hohl, D.; Schäfer, M.; et al. Dual Role of the Antioxidant Enzyme Peroxiredoxin 6 in Skin Carcinogenesis. Cancer Res. 2013, 73, 3460–3469. [Google Scholar] [CrossRef] [PubMed]
- Yanagawa, T.; Ishikawa, T.; Ishii, T.; Tabuchi, K.; Iwasa, S.; Bannai, S.; Omura, K.; Suzuki, H.; Yoshida, H. Peroxiredoxin I expression in human thyroid tumors. Cancer Lett. 1999, 145, 127–132. [Google Scholar] [CrossRef]
- Du, Z.-X.; Yan, Y.; Zhang, H.-Y.; Liu, B.-Q.; Gao, Y.-Y.; Niu, X.-F.; Guan, Y.; Meng, X.; Wang, H.-Q. Suppression of MG132-mediated cell death by peroxiredoxin 1 through influence on ASK1 activation in human thyroid cancer cells. Endocr.-Relat. Cancer 2010, 17, 553–560. [Google Scholar] [CrossRef] [PubMed]
- Nicolussi, A.; D’Inzeo, S.; Mincione, G.; Buffone, A.; di Marcantonio, M.C.; Cotellese, R.; Cichella, A.; Capalbo, C.; Di Gioia, C.; Nardi, F.; et al. PRDX1 and PRDX6 are repressed in papillary thyroid carcinomas via BRAF V600E-dependent and -independent mechanisms. Int. J. Oncol. 2014, 44, 548–556. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gérard, A.-C.; Many, M.-C.; Daumerie, C.; Knoops, B.; Colin, I. Peroxiredoxin 5 Expression in the Human Thyroid Gland. Thyroid 2005, 15, 205–209. [Google Scholar] [CrossRef] [PubMed]
- Nabeshima, A.; Yamada, S.; Guo, X.; Tanimoto, A.; Wang, K.-Y.; Shimajiri, S.; Kimura, S.; Tasaki, T.; Noguchi, H.; Kitada, S.; et al. Peroxiredoxin 4 Protects Against Nonalcoholic Steatohepatitis and Type 2 Diabetes in a Nongenetic Mouse Model. Antioxid. Redox Signal. 2013, 19, 1983–1998. [Google Scholar] [CrossRef] [PubMed]
- Yamada, S.; Guo, X. Peroxiredoxin 4 (PRDX4): Its critical in vivo roles in animal models of metabolic syndrome ranging from atherosclerosis to nonalcoholic fatty liver disease. Pathol. Int. 2018, 68, 91–101. [Google Scholar] [CrossRef]
- Guo, X.; Yamada, S.; Tanimoto, A.; Ding, Y.; Wang, K.-Y.; Shimajiri, S.; Murata, Y.; Kimura, S.; Tasaki, T.; Nabeshima, A.; et al. Overexpression of Peroxiredoxin 4 Attenuates Atherosclerosis in Apolipoprotein E Knockout Mice. Antioxid. Redox Signal. 2012, 17, 1362–1375. [Google Scholar] [CrossRef]
- Ummanni, R.; Barreto, F.; Venz, S.; Scharf, C.; Barett, C.; Mannsperger, H.A.; Brase, J.C.; Kuner, R.; Schlomm, T.; Sauter, G.; et al. Peroxiredoxins 3 and 4 Are Overexpressed in Prostate Cancer Tissue and Affect the Proliferation of Prostate Cancer Cells in Vitro. J. Proteome Res. 2012, 11, 2452–2466. [Google Scholar] [CrossRef]
- Chang, K.-P.; Yu, J.-S.; Chien, K.-Y.; Lee, C.-W.; Liang, Y.; Liao, C.-T.; Yen, T.-C.; Lee, L.-Y.; Huang, L.-L.; Liu, S.-C.; et al. Identification of PRDX4 and P4HA2 as Metastasis-Associated Proteins in Oral Cavity Squamous Cell Carcinoma by Comparative Tissue Proteomics of Microdissected Specimens Using iTRAQ Technology. J. Proteome Res. 2011, 10, 4935–4947. [Google Scholar] [CrossRef]
- Zheng, J.; Guo, X.; Nakamura, Y.; Zhou, X.; Yamaguchi, R.; Zhang, J.; Ishigaki, Y.; Uramoto, H.; Yamada, S. Overexpression of PRDX4 Modulates Tumor Microenvironment and Promotes Urethane-Induced Lung Tumorigenesis. Oxidative Med. Cell. Longev. 2020, 2020, 8262730. [Google Scholar] [CrossRef]
- Wang, W.; Shen, X.-B.; Huang, D.-B.; Jia, W.; Liu, W.-B.; He, Y.-F. Peroxiredoxin 4 suppresses anoikis and augments growth and metastasis of hepatocellular carcinoma cells through the beta-catenin/ID2 pathway. Cell. Oncol. 2019, 42, 769–781. [Google Scholar] [CrossRef]
- Rafiei, S.; Tiedemann, K.; Tabariès, S.; Siegel, P.M.; Komarova, S.V. Peroxiredoxin 4: A novel secreted mediator of cancer induced osteoclastogenesis. Cancer Lett. 2015, 361, 262–270. [Google Scholar] [CrossRef] [PubMed]
- Greene, F.L.; Page, D.L.; Fleming, I.D.; Fritz, A.; Balch, C.M.; Haller, D.G.; Morrow, M. AJCC Cancer Staging Manual, 6th ed.; Springer: Berlin, Germany, 2002. [Google Scholar]
- Brierley, J.D.; Gospodarowicz, M.K.; Wittekind, C.; Union for International Cancer Control. TNM Classification of Malignant Tumours, 8th ed.; John Wiley & Sons: Hoboken, NJ, USA, 2017. [Google Scholar]
- Lloyd, R.V.; Osamura, R.Y.; Kloppel, G. WHO Classification of Tumors of Endocrine Organs, 4th ed.; World Health Organization: Geneva, Switzerland, 2018. [Google Scholar]
- Köhrle, J. Selenium and the thyroid. Curr. Opin. Endocrinol. Diabetes Obes. 2015, 22, 392–401. [Google Scholar] [CrossRef]
- Mizutani, K.; Guo, X.; Shioya, A.; Zhang, J.; Zheng, J.; Kurose, N.; Ishibashi, H.; Motono, N.; Uramoto, H.; Yamada, S. The impact of PRDX4 and the EGFR mutation status on cellular proliferation in lung adenocarcinoma. Int. J. Med. Sci. 2019, 16, 1199–1206. [Google Scholar] [CrossRef] [PubMed]
- Shioya, A.; Guo, X.; Motono, N.; Mizuguchi, S.; Kurose, N.; Nakada, S.; Aikawa, A.; Ikeda, Y.; Uramoto, H.; Yamada, S. The Combination of Weak Expression of PRDX4 and Very High MIB-1 Labelling Index Independently Predicts Shorter Disease-free Survival in Stage I Lung Adenocarcinoma. Int. J. Med. Sci. 2018, 15, 1025–1034. [Google Scholar] [CrossRef]
- Zheng, J.; Guo, X.; Shioya, A.; Yoshioka, T.; Matsumoto, K.; Hiraki, T.; Kusano, H.; Oyama, T.; Kurose, N.; Yamaguchi, R.; et al. Peroxiredoxin 4 promotes embryonal hepatoblastoma cell migration but induces fetal cell differentiation. Am. J. Transl. Res. 2020, 12, 2726–2737. [Google Scholar]
- Guo, X.; Noguchi, H.; Ishii, N.; Homma, T.; Hamada, T.; Hiraki, T.; Zhang, J.; Matsuo, K.; Yokoyama, S.; Ishibashi, H.; et al. The Association of Peroxiredoxin 4 with the Initiation and Progression of Hepatocellular Carcinoma. Antioxid. Redox Signal. 2019, 30, 1271–1284. [Google Scholar] [CrossRef]
- Ito, Y.; Masuoka, H.; Fukushima, M.; Inoue, H.; Kihara, M.; Tomoda, C.; Higashiyama, T.; Takamura, Y.; Kobayashi, K.; Miya, A.; et al. Excellent Prognosis of Patients with Solitary T1N0M0 Papillary Thyroid Carcinoma Who Underwent Thyroidectomy and Elective Lymph Node Dissection Without Radioiodine Therapy. World J. Surg. 2010, 34, 1285–1290. [Google Scholar] [CrossRef]
- Schlumberger, M.; Catargi, B.; Borget, I.; Deandreis, D.; Zerdoud, S.; Bridji, B.; Bardet, S.; Leenhardt, L.; Bastie, D.; Schvartz, C.; et al. Tumeurs de la Thyroïde Refractaires Network for the Essai Stimulation Ablation Equivalence Trial: Strategies of radioiodine ablation in patients with low-risk thyroid cancer. N. Engl. J. Med. 2012, 366, 1663–1673. [Google Scholar] [CrossRef]
- Giuliano, S.; Mirabelli, M.; Chiefari, E.; Vergine, M.; Gervasi, R.; Brunetti, F.; Innaro, N.; Donato, G.; Aversa, A.; Brunetti, A. Malignancy Analyses of Thyroid Nodules in Patients Subjected to Surgery with Cytological- and Ultrasound-Based Risk Stratification Systems. Endocrines 2020, 1, 102–118. [Google Scholar] [CrossRef]

| Characteristic | Patients (n = 70) |
|---|---|
| Age | |
| Median | 66 |
| Range | 55–80 |
| Sex | |
| Male | 13 |
| Female | 57 |
| Clinical Stage | |
| II | 48 |
| III | 17 |
| IV | 5 |
| Distant metastasis | 8 |
| Regional metastasis | 40 |
| Operative Method | |
| Lobectomy | 24 |
| Subtotal | 3 |
| Thyroidectomy | 43 |
| Recurrence | 11 |
| Local | 0 |
| Regional | 11 |
| Distant | 2 |
| Pathological | |
| Tumor size, mm | |
| Median | 19.5 |
| Range | 1.2–100 |
| Vascular invasion | 32 |
| Lymphatic invasion | 35 |
| Extension | |
| 0 | 7 |
| 1 | 45 |
| 2 | 18 |
| Multifocality | 27 |
| Variable, n (%) | Weak Expression (n = 32) | Strong Expression (n = 38) | p |
|---|---|---|---|
| Gender | |||
| Male | 7 (22) | 6 (16) | 0.55 |
| Female | 25 (78) | 32 (84) | |
| Tumor size | |||
| >20 mm | 22 (69) | 28 (74) | 0.79 |
| ≤2 mm | 10 (31) | 10 (26) | |
| Vascular invasion | |||
| Positive | 11 (34) | 21 (55) | 0.1 |
| Negative | 21 (66) | 17 (45) | |
| Lymphatic invasion | |||
| Positive | 13 (41) | 22 (58) | 0.23 |
| Negative | 19 (59) | 16 (42) | |
| Extension | |||
| Positive | 3 (9) | 4 (11) | 1 |
| Negative | 29 (91) | 34 (89) | |
| Multifocality | |||
| Positive | 13 (41) | 10 (26) | 0.31 |
| Negative | 19 (59) | 28 (74) | |
| Regional metastasis | |||
| Positive | 13 (41) | 18 (47) | 0.63 |
| Negative | 19 (59) | 20 (53) | |
| Distant metastasis | |||
| Positive | 4 (14) | 4 (8) | 1 |
| Negative | 28 (86) | 34 (92) | |
| Recurrence | |||
| Positive | 9 (18) | 2 (5) | 0.02 |
| Negative | 23 (72) | 36 (95) | |
| Stage | |||
| II | 29 (76) | 19 (59) | 0.20 |
| III, IV | 9 (24) | 13 (41) | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Takaoka, Y.; Guo, X.; Shioya, A.; Han, J.; Shimode, Y.; Kobayasi, Y.; Kitamura, M.; Tsuji, H.; Yamada, S. PRDX4 Potentially Predicts the Postoperative Outcome in Advanced Papillary Thyroid Carcinoma. Endocrines 2022, 3, 139-149. https://doi.org/10.3390/endocrines3010013
Takaoka Y, Guo X, Shioya A, Han J, Shimode Y, Kobayasi Y, Kitamura M, Tsuji H, Yamada S. PRDX4 Potentially Predicts the Postoperative Outcome in Advanced Papillary Thyroid Carcinoma. Endocrines. 2022; 3(1):139-149. https://doi.org/10.3390/endocrines3010013
Chicago/Turabian StyleTakaoka, Yuki, Xin Guo, Akihiro Shioya, Jia Han, Yuzo Shimode, Yoshiaki Kobayasi, Morimasa Kitamura, Hiroyuki Tsuji, and Sohsuke Yamada. 2022. "PRDX4 Potentially Predicts the Postoperative Outcome in Advanced Papillary Thyroid Carcinoma" Endocrines 3, no. 1: 139-149. https://doi.org/10.3390/endocrines3010013
APA StyleTakaoka, Y., Guo, X., Shioya, A., Han, J., Shimode, Y., Kobayasi, Y., Kitamura, M., Tsuji, H., & Yamada, S. (2022). PRDX4 Potentially Predicts the Postoperative Outcome in Advanced Papillary Thyroid Carcinoma. Endocrines, 3(1), 139-149. https://doi.org/10.3390/endocrines3010013






