Experimental Manipulation of Psychological Control Scenarios: Implications for Exercise and Memory Research
Abstract
:1. Introduction
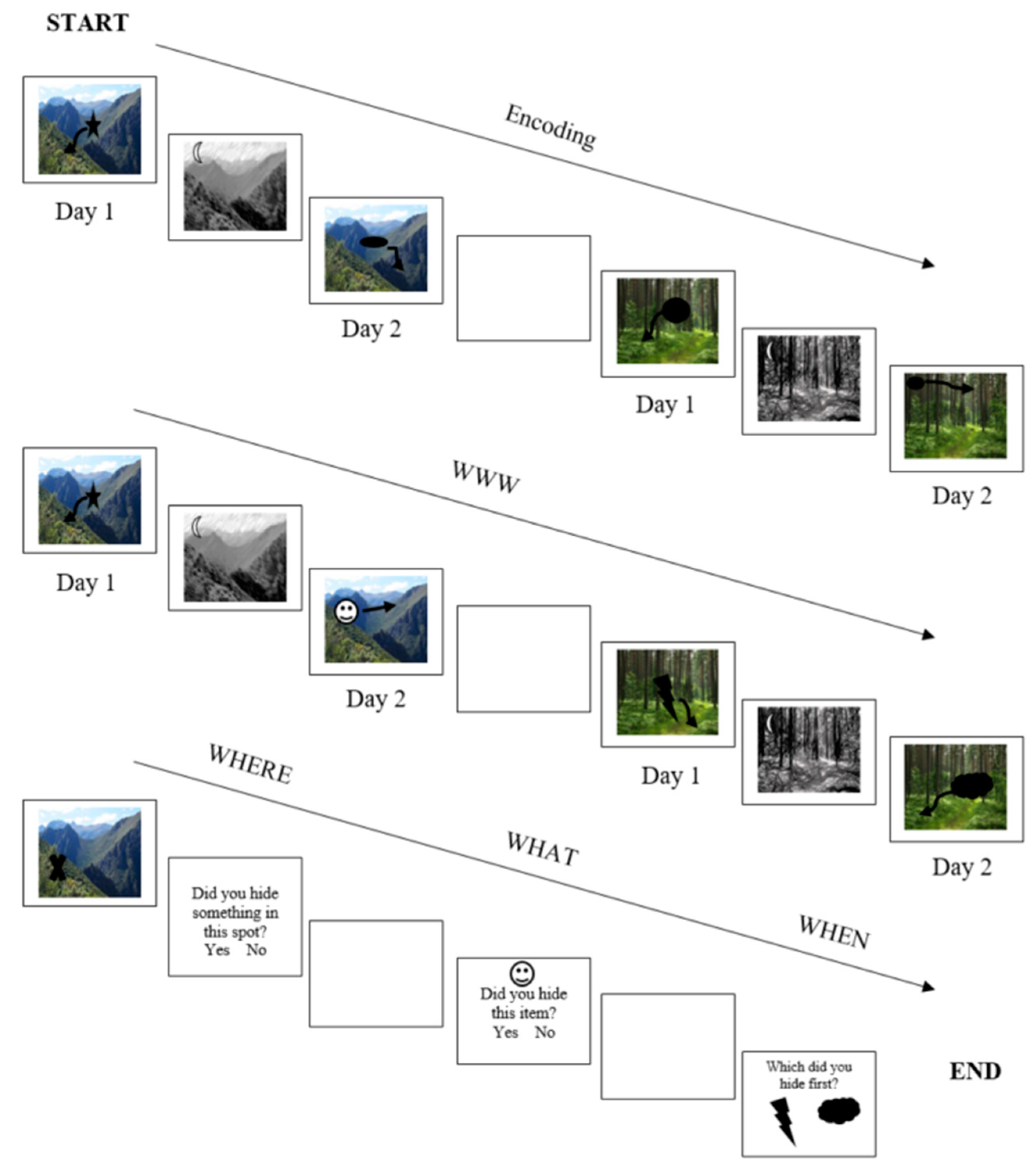
2. Methods
Study Design and Participants
3. Measures
4. Statistical Analysis
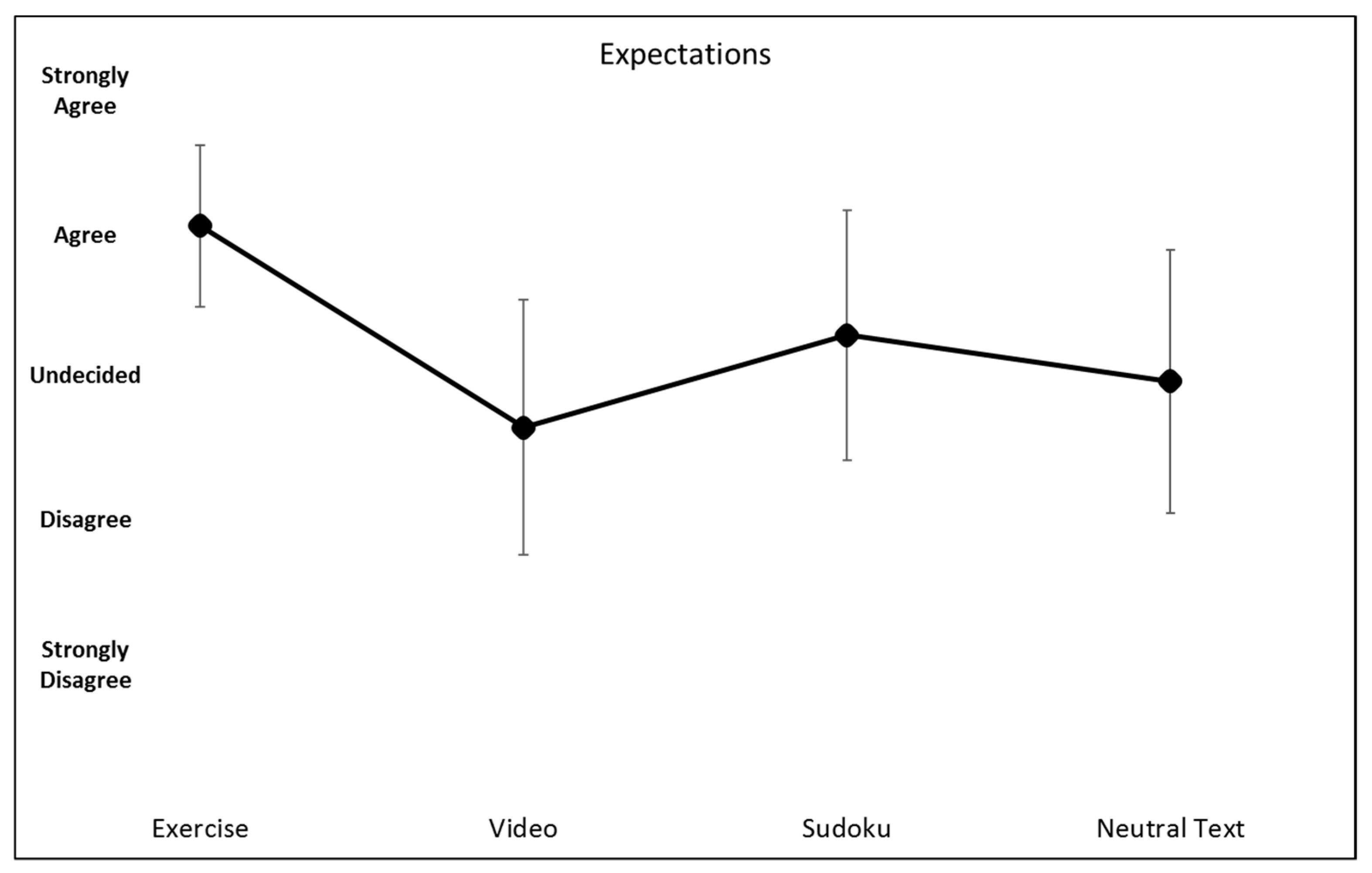
5. Results
6. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Roig, M.; Nordbrandt, S.; Geertsen, S.S.; Nielsen, J.B. The effects of cardiovascular exercise on human memory: A review with meta-analysis. Neurosci. Biobehav. Rev. 2013, 37, 1645–1666. [Google Scholar] [CrossRef]
- Loprinzi, P.D.; Frith, E.; Edwards, M.K.; Sng, E.; Ashpole, N. The effects of exercise on memory function among young to middle-aged adults: Systematic review and recommendations for future research. Am. J. Health Promot. 2018, 32, 691–704. [Google Scholar] [CrossRef] [PubMed]
- Pontifex, M.B.; McGowan, A.L.; Chandler, M.C.; Gwizdala, K.L.; Parks, A.C.; Fenn, K.; Kamijo, K. A primer on investigating the after effects of acute bouts of physical activity on cognition. Psychol. Sport Exerc. 2018, 40, 1–22. [Google Scholar] [CrossRef]
- Boot, W.R.; Simons, D.J.; Stothart, C.; Stutts, C. The pervasive problem with placebos in psychology: Why active control groups are not sufficient to rule out placebo effects. Perspect. Psychol. Sci. 2013, 8, 445–454. [Google Scholar] [CrossRef] [PubMed]
- Kremer, E.F. The truly random control procedure: Conditioning to the static cues. J. Comp. Physiol. Psychol. 1974, 86, 700. [Google Scholar] [CrossRef] [PubMed]
- Kremer, E.F. Truly random and traditional control procedures in CER conditioning in the rat. J. Comp. Physiol. Psychol. 1971, 76, 441. [Google Scholar] [CrossRef]
- Dudai, Y. Memory from A to Z: Keywords, Concepts, and Beyond; Oxford University Press: New York, NY, USA, 2004. [Google Scholar]
- Loprinzi, P.D.; Blough, J.; Crawford, L.; Ryu, S.; Zou, L.; Li, H. The temporal effects of acute exercise on episodic memory function: Systematic review with meta-analysis. Brain Sci. 2019, 9, 87. [Google Scholar] [CrossRef] [PubMed]
- Crush, E.A.; Loprinzi, P.D. Dose-response effects of exercise duration and recovery on cognitive functioning. Percept. Mot. Ski. 2017, 124, 1164–1193. [Google Scholar] [CrossRef]
- Siddiqui, A.; Loprinzi, P. Experimental Investigation of the Time Course Effects of Acute Exercise on False Episodic Memory. J. Clin. Med. 2018, 7, 157. [Google Scholar] [CrossRef] [PubMed]
- Sng, E.; Frith, E.; Loprinzi, P.D. Temporal effects of acute walking exercise on learning and memory function. Am. J. Health Promot. 2018, 32, 1518–1525. [Google Scholar] [CrossRef] [PubMed]
- McNerney, M.W.; Radvansky, G.A. Mind racing: The influence of exercise on long-term memory consolidation. Memory 2015, 23, 1140–1151. [Google Scholar] [CrossRef] [PubMed]
- Labban, J.D.; Etnier, J.L. The Effect of Acute Exercise on Encoding and Consolidation of Long-Term Memory. J. Sport Exerc. Psychol. 2018, 40, 336–342. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.L.J.; Graham, S.; Zorawski, M. Enhanced selective memory consolidation following post-learning pleasant and aversive arousal. Neurobiol. Learn. Mem. 2008, 89, 36–46. [Google Scholar] [CrossRef]
- Wingate, S.; Crawford, L.; Frith, E.; Loprinzi, P.D. Experimental investigation of the effects of acute exercise on memory interference. Health Promot. Perspect. 2018, 8, 208. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, H.C.; Rijpkema, M.; Fernandez, G.; Kessels, R.P. The effects of valence and arousal on associative working memory and long-term memory. PLoS ONE 2012, 7, e52616. [Google Scholar] [CrossRef]
- Jubelt, L.E.; Barr, R.S.; Goff, D.C.; Logvinenko, T.; Weiss, A.P.; Evins, A.E. Effects of transdermal nicotine on episodic memory in non-smokers with and without schizophrenia. Psychopharmacology 2008, 199, 89–98. [Google Scholar] [CrossRef] [Green Version]
- Klaming, R.; Annese, J.; Veltman, D.J.; Comijs, H.C. Episodic memory function is affected by lifestyle factors: A 14-year follow-up study in an elderly population. Aging Neuropsychol. Cognit. 2017, 24, 528–542. [Google Scholar] [CrossRef]
- Henry, J.D.; Rendell, P.G. A review of the impact of pregnancy on memory function. J. Clin. Exp. Neuropsychol. 2007, 29, 793–803. [Google Scholar] [CrossRef] [PubMed]
- Wammes, J.D.; Good, T.J.; Fernandes, M.A. Autobiographical and episodic memory deficits in mild traumatic brain injury. Brain Cognit. 2017, 111, 112–126. [Google Scholar] [CrossRef] [PubMed]
- Hindocha, C.; Freeman, T.; Xia, J.; Shaban, N.; Curran, H. Acute memory and psychotomimetic effects of cannabis and tobacco both ‘joint’ and individually: A placebo-controlled trial. Psychol. Med. 2017, 47, 2708–2719. [Google Scholar] [CrossRef] [PubMed]
- Le Berre, A.P.; Fama, R.; Sullivan, E.V. Executive functions, memory, and social cognitive deficits and recovery in chronic alcoholism: A critical review to inform future research. Alcohol. Clin. Exp. Res. 2017, 41, 1432–1443. [Google Scholar] [CrossRef] [PubMed]
- Sherman, S.M.; Buckley, T.P.; Baena, E.; Ryan, L. Caffeine enhances memory performance in young adults during their non-optimal time of day. Front. Psychol. 2016, 7, 1764. [Google Scholar] [CrossRef] [PubMed]
- Labban, J.D.; Etnier, J.L. Effects of acute exercise on long-term memory. Res. Q. Exerc. Sport 2011, 82, 712–721. [Google Scholar] [CrossRef]
- Singer, J.A.; Salovey, P. Mood and memory: Evaluating the network theory of affect. Clin. Psychol. Rev. 1988, 8, 211–251. [Google Scholar] [CrossRef]
- Watson, D.; Clark, L.A.; Tellegen, A. Development and validation of brief measures of positive and negative affect: The PANAS scales. J. Personal. Soc. Psychol. 1988, 54, 1063. [Google Scholar] [CrossRef]
- Ball, T.J.; Joy, E.A.; Gren, L.H.; Cunningham, R.; Shaw, J.M. Predictive validity of an adult physical activity “Vital Sign” recorded in electronic health records. J. Phys. Act. Health 2016, 13, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Ball, T.J.; Joy, E.A.; Gren, L.H.; Shaw, J.M. Peer Reviewed: Concurrent Validity of a Self-Reported Physical Activity “Vital Sign” Questionnaire With Adult Primary Care Patients. Prev. Chronic Dis. 2016, 13, E16. [Google Scholar] [CrossRef] [PubMed]
- Ball, T.J.; Joy, E.A.; Goh, T.L.; Hannon, J.C.; Gren, L.H.; Shaw, J.M. Validity of two brief primary care physical activity questionnaires with accelerometry in clinic staff. Prim. Health Care Res. Dev. 2015, 16, 100–108. [Google Scholar] [CrossRef]
- Garber, C.E.; Blissmer, B.; Deschenes, M.R.; Franklin, B.A.; Lamonte, M.J.; Lee, I.M.; Nieman, D.C.; Swain, D.P. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: Guidance for prescribing exercise. Med. Sci. Sports Exerc. 2011, 43, 1334–1359. [Google Scholar] [CrossRef]
- Wilson, M. MRC psycholinguistic database: Machine-usable dictionary, version 2.00. Behav. Res. Methods Instrum. Comput. 1988, 20, 6–10. [Google Scholar] [CrossRef] [Green Version]
- Geffen, G.M.; Butterworth, P.; Geffen, L.B. Test-retest reliability of a new form of the auditory verbal learning test (AVLT). Arch. Clin. Neuropsychol. 1994, 9, 303–316. [Google Scholar] [CrossRef] [PubMed]
- Cheke, L.G. What–where–when memory and encoding strategies in healthy aging. Learn. Mem. 2016, 23, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Cheke, L.G.; Simons, J.S.; Clayton, N.S. Higher body mass index is associated with episodic memory deficits in young adults. Q. J. Exp. Psychol. 2016, 69, 2305–2316. [Google Scholar] [CrossRef] [PubMed]
- Krikorian, R.; Bartok, J.; Gay, N. Tower of London procedure: A standard method and developmental data. J. Clin. Exp. Neuropsychol. 1994, 16, 840–850. [Google Scholar] [CrossRef]
- Schnirman, G.M.; Welsh, M.C.; Retzlaff, P.D. Development of the Tower of London-revised. Assessment 1998, 5, 355–360. [Google Scholar] [CrossRef] [PubMed]
- Shallice, T. Specific impairments of planning. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 1982, 298, 199–209. [Google Scholar] [CrossRef]
- Bull, R.; Espy, K.A.; Senn, T.E. A comparison of performance on the Towers of London and Hanoi in young children. J. Child Psychol. Psychiatry 2004, 45, 743–754. [Google Scholar] [CrossRef]
- Köstering, L.; Schmidt, C.S.; Egger, K.; Amtage, F.; Peter, J.; Klöppel, S.; Beume, L.A.; Hoeren, M.; Weiller, C.; Kaller, C.P. Assessment of planning performance in clinical samples: Reliability and validity of the Tower of London task (TOL-F). Neuropsychologia 2015, 75, 646–655. [Google Scholar] [CrossRef]
- Köstering, L.; Nitschke, K.; Schumacher, F.K.; Weiller, C.; Kaller, C.P. Test–retest reliability of the Tower of London Planning Task (TOL-F). Psychol. Assess. 2015, 27, 925. [Google Scholar] [CrossRef]
- Lee, S.-Y.; Song, X.-Y. Evaluation of the Bayesian and maximum likelihood approaches in analyzing structural equation models with small sample sizes. Multivar. Behav. Res. 2004, 39, 653–686. [Google Scholar] [CrossRef]
- Hox, J.J.; van de Schoot, R.; Matthijsse, S. How few countries will do? Comparative survey analysis from a Bayesian perspective. Surv. Res. Methods 2012, 6, 87–93. [Google Scholar]
- Jarosz, A.F.; Wiley, J. What are the odds? A practical guide to computing and reporting Bayes factors. J. Probl. Solving 2014, 7, 2. [Google Scholar] [CrossRef]
- Loprinzi, P.D. Intensity-specific effects of acute exercise on human memory function: Considerations for the timing of exercise and the type of memory. Health Promot. Perspect. 2018, 8, 255. [Google Scholar] [CrossRef]
- Jaffery, A.; Edwards, M.K.; Loprinzi, P.D. The effects of acute exercise on cognitive function: Solomon experimental design. J. Prim. Prev. 2018, 39, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Loprinzi, P.; Ponce, P.; Frith, E. Hypothesized mechanisms through which acute exercise influences episodic memory. Physiol. Int. 2018, 105, 285–297. [Google Scholar] [CrossRef] [Green Version]
- Loprinzi, P.D.; Edwards, M.K.; Frith, E. Potential avenues for exercise to activate episodic memory-related pathways: A narrative review. Eur. J. Neurosci. 2017, 46, 2067–2077. [Google Scholar] [CrossRef]
- Haynes, J.T., IV; Frith, E.; Sng, E.; Loprinzi, P.D. Experimental effects of acute exercise on episodic memory function: Considerations for the timing of exercise. Psychol. Rep. 2018, 0033294118786688. [Google Scholar]
- Frith, E.; Sng, E.; Loprinzi, P.D. Randomized controlled trial evaluating the temporal effects of high-intensity exercise on learning, short-term and long-term memory, and prospective memory. Eur. J. Neurosci. 2017, 46, 2557–2564. [Google Scholar] [CrossRef] [PubMed]
| Variable | Point Estimate |
|---|---|
| Age (mean years [sd]) | 21.04 (1.12) |
| Sex (% female) | 66.7 |
| Race-Ethnicity, % | |
| Non-Hispanic White (n = 19) | 79.2 |
| Other/Multi-race (n = 3) | 12.5 |
| Non-Hispanic Black (n = 2) | 8.3 |
| Taking Medication to Regulate Mood/Emotion (% Yes) | 4.2 |
| Currently diagnosed with ADD/ADHD (% Yes) | 4.2 |
| BMI (mean [sd]) | 26.58 (6.28) |
| MVPA (mean min per week [sd]) | 175.4 (116.8) |
| Variable | Exercise Visit | Video Visit | Sudoku Visit | Neutral Text Visit | Test Statistic |
|---|---|---|---|---|---|
| What-Where-When (WWW) (mean % [sd]) | 63.28 (21.6) | 69.53 (21.91) | 65.63 (23.89) | 61.2 (22.04) | F3,23 = 0.989, P = 0.403, η2p = 0.041, BF10 = 0.167 |
| What Loop (mean % [sd]) | 99.48 (1.77) | 99.22 (2.8) | 99.22 (2.11) | 99.48 (1.77) | F3,23 = 0.113, P = 0.952, η2p = 0.005, BF10 = 0.065 |
| Where Loop (mean % [sd]) | 87.03 (10.25) | 87.24 (15.03) | 90.89 (9.03) | 87.24 (13.6) | F3,23 = 0.571, P = 0.636, η2p = 0.024, BF10 = 0.113 |
| When Loop (mean % [sd]) | 84.38 (15.09) | 91.19 (7.87) | 90.16 (11.49) | 93.49 (7.01) | F3,23 = 4.455, P = 0.006, η2p = 0.162, BF10 = 7.596 |
| Tower of London (mean score [sd]) | 33.25 (2.72) | 33.58 (2.640 | 33.42 (2.28) | 33.83 (2.3) | F3,23 = 0.277, P = 0.842, η2p = 0.012, BF10 = 0.079 |
| Immediate Recall (mean score [sd]) | 7.54 (2.34) | 8.49 (2.4) | 7.67 (2.18) | 7.58 (2.28) | F(Visit) = 1.289, P = 0.285, η2p = 0.053, F(Time) = 125.995, P < 0.001, η2p = 0.846, F(Interaction) = 1.635, P = 0.189, η2p = 0.066 |
| Delayed Recall (mean score [sd]) | 4.67 (1.93) | 5.29 (2.9) | 4.42 (2.83 | 5.29 (2.65) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Blough, J.; Loprinzi, P.D. Experimental Manipulation of Psychological Control Scenarios: Implications for Exercise and Memory Research. Psych 2019, 1, 279-289. https://doi.org/10.3390/psych1010019
Blough J, Loprinzi PD. Experimental Manipulation of Psychological Control Scenarios: Implications for Exercise and Memory Research. Psych. 2019; 1(1):279-289. https://doi.org/10.3390/psych1010019
Chicago/Turabian StyleBlough, Jeremiah, and Paul D. Loprinzi. 2019. "Experimental Manipulation of Psychological Control Scenarios: Implications for Exercise and Memory Research" Psych 1, no. 1: 279-289. https://doi.org/10.3390/psych1010019
APA StyleBlough, J., & Loprinzi, P. D. (2019). Experimental Manipulation of Psychological Control Scenarios: Implications for Exercise and Memory Research. Psych, 1(1), 279-289. https://doi.org/10.3390/psych1010019





