Silane-Based Coatings Containing TiO2 for Corrosion Protection of 316L Stainless Steel
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Surface Functionalization of TiO2 Nanoparticles
2.3. Application of Coatings
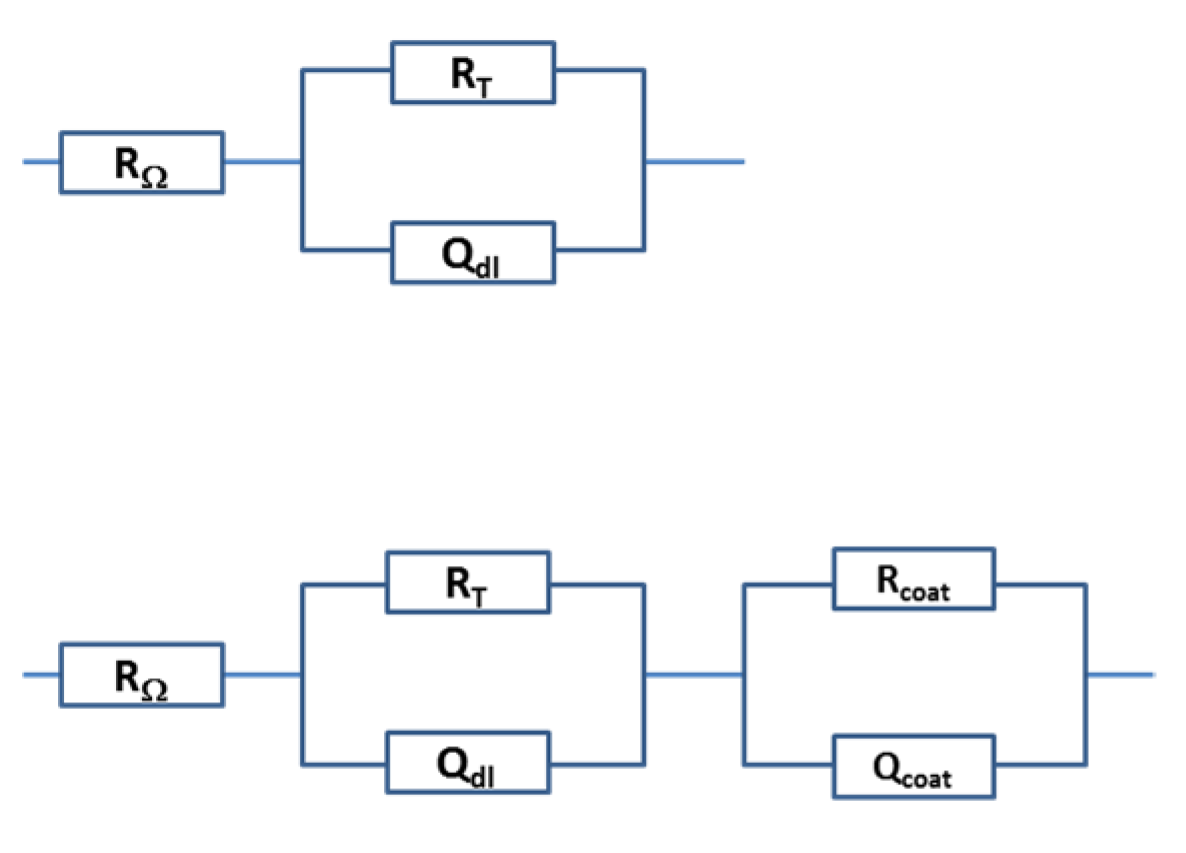
2.4. Characterization of the Coatings
- The open-circuit potential (OCP) of the samples was stabilized for 1 h.
- The impedance experiments were conducted over a frequency range from 105 Hz to 10 mHz by superimposing a 10 mV AC amplitude over the DC bias of the potentiostat that was the OCP. Seven points were registered per decade.
- Finally, the anodic polarization curves were obtained at a scan rate of 1 mV/s, starting at the OCP. The scan direction was reversed at J = 10 mA/cm2 to observe the possible repassivation ability of the system.
- The corrosion potential (Ecorr), in this case equivalent to OCP, is related to the thermodynamic corrosion tendency of the sample.
- The breakdown potential (Eb) and the passivation range (Eb-Ecorr) were estimated and correlated with the pitting resistance of the silane coating.
- The corrosion current density (Jcorr) is related to the corrosion rate at the corrosion potential. The value of this parameter was obtained from the intersection of the anodic Tafel branch and the corrosion potential value [13].
3. Results and Discussion
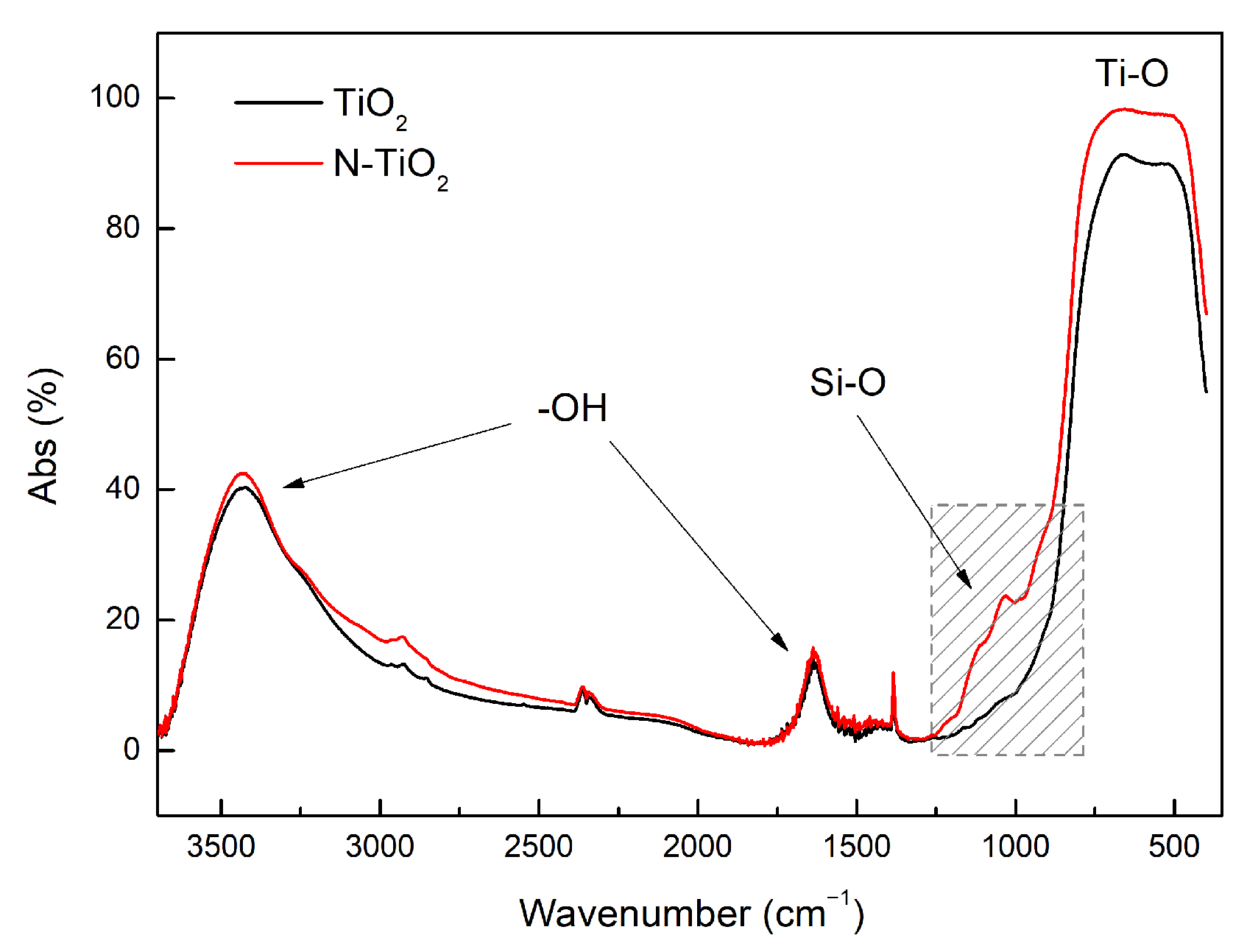
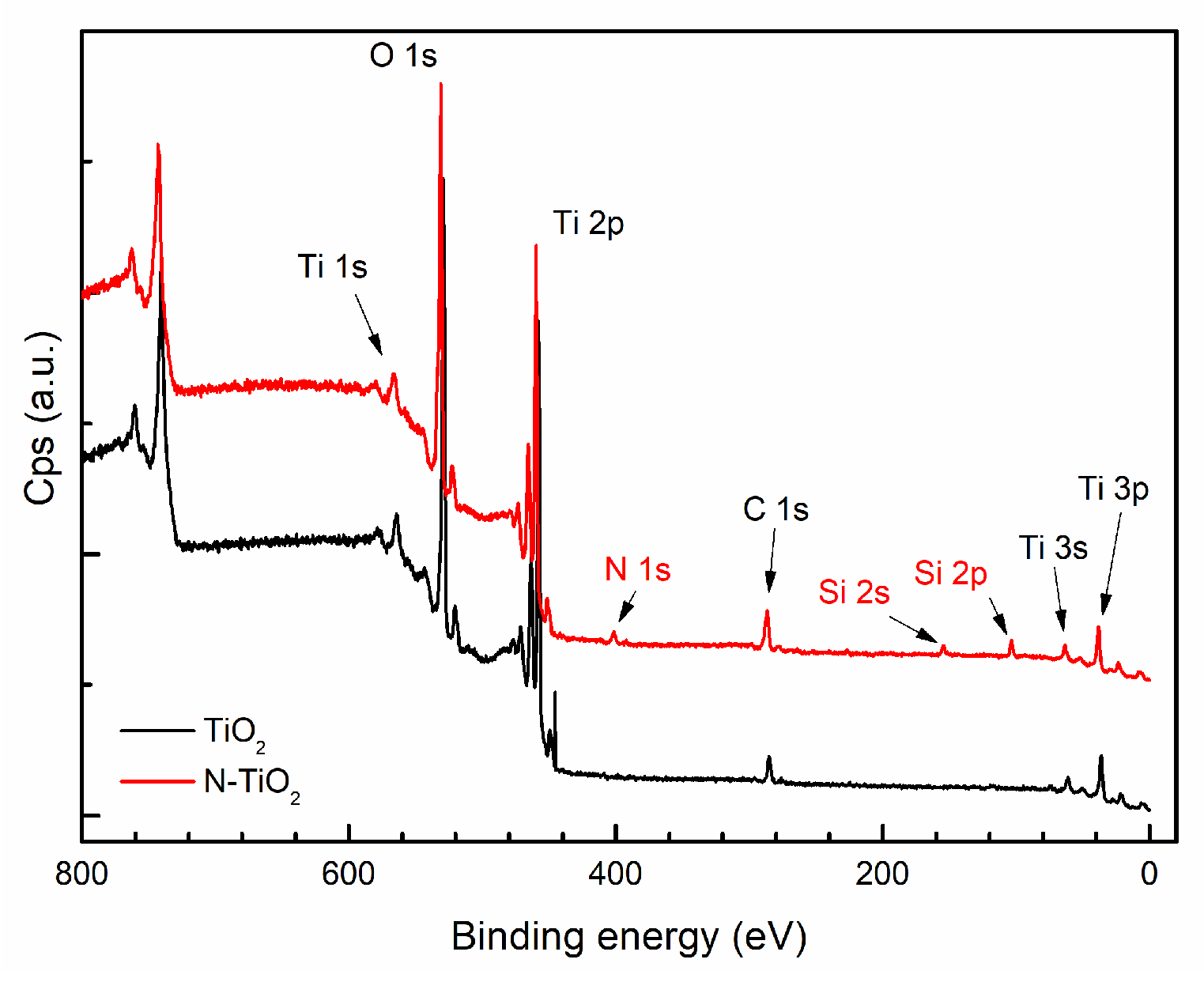
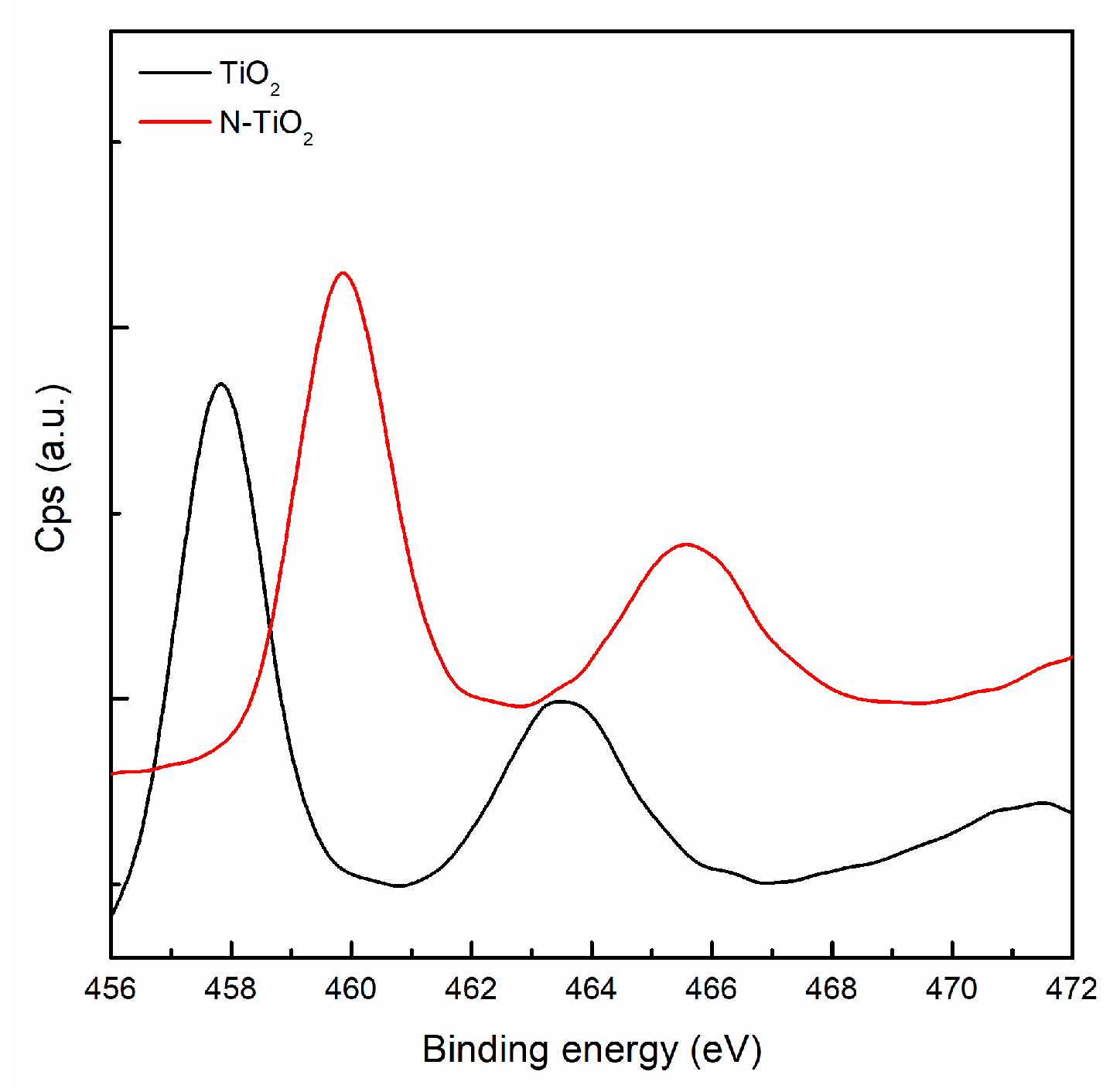
3.1. Functionalization of Particles
3.2. Characterization of the Coatings
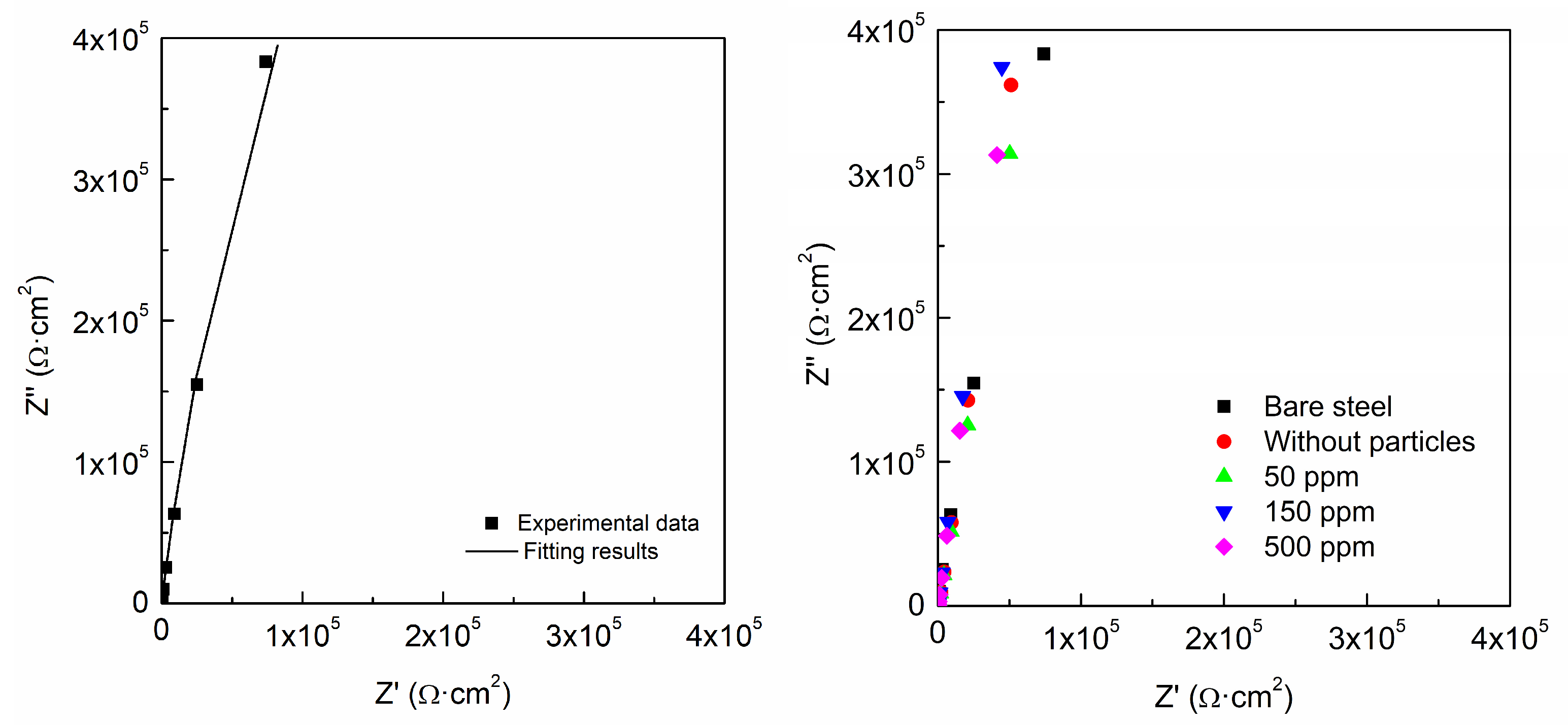
3.3. Protection Performance
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| TEOS | Tetraethyl orthosilicate |
| TMES | Trimethylethoxysilane |
| APS | 3-aminopropyl trimethoxysilane |
| 316L SS | 316L stainless steel |
| PSBF | Phosphate containing simulated body fluid |
References
- Rosenbloom, S.N.; Corbett, R.A. An Assessment of ASTM F 2129 Electrochemical Testing of Small Medical Implants—Lessons Learned. In Proceedings of the CORROSION 2007, Nashville, TN, USA, 11–15 March 2007; NACE International: Houston, TX, USA, 2007; pp. 1–10. [Google Scholar] [CrossRef]
- Xu, W.; Yu, F.; Yang, L.; Zhang, B.; Hou, B.; Li, Y. Accelerated Corrosion of 316L Stainless Steel in Simulated Body Fluids in the Presence of H2O2 and Albumin. Mater. Sci. Eng. C 2018, 92, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Talha, M.; Behera, C.K.; Sinha, O.P. Potentiodynamic Polarization Study of Type 316L and 316LVM Stainless Steels for Surgical Implants in Simulated Body Fluids. J. Chem. Pharm. Res. 2012, 4, 203–208. [Google Scholar]
- Xu, Y.; Li, Y.; Chen, T.; Dong, C.; Zhang, K.; Bao, X. A Short Review of Medical-Grade Stainless Steel: Corrosion Resistance and Novel Techniques. J. Mater. Res. Technol. 2024, 29, 2788–2798. [Google Scholar] [CrossRef]
- Barrino, F. Hybrid Organic–Inorganic Materials Prepared by Sol–Gel and Sol–Gel-Coating Method for Biomedical Use: Study and Synthetic Review of Synthesis and Properties. Coatings 2024, 14, 425. [Google Scholar] [CrossRef]
- Bollino, F.; Catauro, M. Sol-Gel Technology to Prepare Advanced Coatings. In Photoenergy and Thin Film Materials; Wiley: Hoboken, NJ, USA, 2019; pp. 321–378. [Google Scholar] [CrossRef]
- Rafiaei, F.; Naderi, R.; Dehghanian, C. Impact of Curing on the Corrosion Performance of an Eco-Friendly Silane Sol-Gel Coating on 304L Stainless Steel. RSC Adv. 2015, 5, 43225–43233. [Google Scholar] [CrossRef]
- Somasundaram, S. Silane Coatings of Metallic Biomaterials for Biomedical Implants: A Preliminary Review. J. Biomed. Mater. Res. B Appl. Biomater. 2018, 106, 2901–2918. [Google Scholar] [CrossRef] [PubMed]
- Kierat, O.; Dudek, A. Sol–Gel-Derived Vinyltrimethoxysilane (VTMS)/Tetraethoxysilane (TEOS) Hybrid Coatings on Titanium Materials for Use in Medical Applications. Materials 2025, 18, 2273. [Google Scholar] [CrossRef] [PubMed]
- Altube, A.; García-Lecina, E.; Merino, L.; Díez, J.A.; Grande, H. Development and Characterization of Conductive Sol-Gel Coatings for Corrosion Protection of 316L Steel. In Proceedings of the Solgel 2017, SG2017-336, Liège, Belgium, 3–8 September 2017. [Google Scholar]
- Kulich, P.; Marvanová, S.; Skoupý, R.; Škorič, M.; Vysloužil, J.; Šerý, O.; Mikuška, P.; Alexa, L.; Coufalík, P.; Křůmal, K.; et al. Subchronic Inhalation of TiO2 Nanoparticles Leads to Deposition in the Lung and Alterations in Erythrocyte Morphology in Mice. J. Appl. Toxicol. 2025, 45, 1004–1018. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Yakovlev, N.L. Adsorption and Interaction of Organosilanes on TiO2 Nanoparticles. Appl. Surf. Sci. 2010, 257, 1395–1400. [Google Scholar] [CrossRef]
- Kuratani, K.; Fukami, K.; Tsuchiya, H.; Usui, H.; Chiku, M.; Yamazaki, S. Electrochemical Polarization Part 1: Fundamentals and Corrosion. Electrochemistry 2022, 90, 22–66085. [Google Scholar] [CrossRef]
- Kazemi, M.; Navarchian, A.H.; Ahangaran, F. Effects of Silica Surface Modification with Silane and Poly(Ethylene Glycol) on Flexural Strength, Protein-Repellent, and Antibacterial Properties of Acrylic Dental Nanocomposites. Dent. Mater. 2023, 39, 863–871. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Fang, S.; Zheng, Y.; Sun, J.; Lv, K.; Yu, J.C.; Ho, W.K. Thiourea-Modified TiO2 Nanorods with Enhanced Photocatalytic Activity. Molecules 2016, 21, 181. [Google Scholar] [CrossRef] [PubMed]
- Kusiak-Nejman, E.; Moszyński, D.; Kapica-Kozar, J.; Wanag, A.; Morawski, A.W. Assessment of the Suitability of the One-Step Hydrothermal Method for Preparation of Non-Covalently/Covalently Bonded TiO2/Graphene-Based Hybrids. Nanomaterials 2018, 8, 647. [Google Scholar] [CrossRef] [PubMed]
- Cheng, F.; Sajedin, S.M.; Kelly, S.M.; Lee, A.F.; Kornherr, A. UV-Stable Paper Coated with APTES-Modified P25 TiO2 Nanoparticles. Carbohydr. Polym. 2014, 114, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Foo, C.; Li, Y.; Lebedev, K.; Chen, T.; Day, S.; Tang, C.; Tsang, S.C.E. Characterisation of Oxygen Defects and Nitrogen Impurities in TiO2 Photocatalysts Using Variable-Temperature X-Ray Powder Diffraction. Nat. Commun. 2021, 12, 661. [Google Scholar] [CrossRef] [PubMed]
- Martins, N.C.T.; Ângelo, J.; Girão, A.V.; Trindade, T.; Andrade, L.; Mendes, A. N-Doped Carbon Quantum Dots/TiO2 Composite with Improved Photocatalytic Activity. Appl. Catal. B 2016, 193, 67–74. [Google Scholar] [CrossRef]
- Ozgowicz, W.; Kurc-Lisiecka, A.; Grajcar, A. Corrosion Behaviour Corrosion Behaviour of Cold-Deformed Austenitic Alloys. In Environmental and Industrial Corrosion—Practical and Theoretical Aspects; InTech: London, UK, 2012. [Google Scholar] [CrossRef]
- Kuffner, B.H.B.; Capellato, P.; Ribeiro, L.M.S.; Sachs, D.; Silva, G. Production and Characterization of a 316l Stainless Steel/β-Tcp Biocomposite Using the Functionally Graded Materials (Fgms) Technique for Dental and Orthopedic Applications. Metals 2021, 11, 1923. [Google Scholar] [CrossRef]
- Bou-Saleh, Z.; Shahryari, A.; Omanovic, S. Enhancement of Corrosion Resistance of a Biomedical Grade 316LVM Stainless Steel by Potentiodynamic Cyclic Polarization. Thin Solid Films 2007, 515, 4727–4737. [Google Scholar] [CrossRef]
- Altube, A.; Pierna, A.R.; Marzo, F.F. Hydrogen Evolution Reaction with Finemet–Co Amorphous Alloys in Acid Solution. J. Non Cryst. Solids 2001, 287, 297–301. [Google Scholar] [CrossRef]
- Ahmed, R.; Vourlias, G.; Algoburi, A.; Vogiatzis, C.; Chaliampalias, D.; Skolianos, S.; Berger, L.M.; Paul, S.; Faisal, N.H.; Toma, F.L.; et al. Comparative Study of Corrosion Performance of HVOF-Sprayed Coatings Produced Using Conventional and Suspension WC-Co Feedstock. J. Therm. Spray Technol. 2018, 27, 1579–1593. [Google Scholar] [CrossRef]












| Cr | Ni | Mo | Mn | Si | C | P | S | Others |
|---|---|---|---|---|---|---|---|---|
| 16.5–18 | 10–13 | 2–2.5 | 2 | 0.75 | 0.07 | 0.045 | 0.015 | 0.10 |
| Composition | Content |
|---|---|
| NaCl | 8 g/L |
| CaCl2 | 0.15 g/L |
| KCl | 0.40 g/L |
| MgCl2·6H2O | 0.10 g/L |
| NaHCO3 | 0.35 g/L |
| NaH2PO4·2H2O | 0.06 g/L |
| KH2PO4 | 0.06 g/L |
| MgSO4·7H2O | 0.06 g/L |
| Glucose | 1 g/L |
| Uncoated | Without Particles | 50 ppm | 150 ppm | 500 ppm | |
|---|---|---|---|---|---|
| WCA [°] | 99 ± 2 | 86 ± 5 | 84 ± 4 | 87 ± 2 | 86 ± 3 |
| OCP mV vs. Ag/AgCl | Jcorr ×10−6 mA/cm2 | Eb mV vs. Ag/AgCl | Eb-Ecorr mV | |
|---|---|---|---|---|
| Uncoated | 85 ± 5 | 7 ± 1 | 430 ± 10 | 350 ± 20 |
| Without particles | −6 ± 5 | 6 ± 1 | 890 ± 20 | 900 ± 25 |
| 50 ppm | −75 ± 5 | 8 ± 1 | 930 ± 20 | 1000 ± 25 |
| 150 ppm | 5 ± 5 | 5 ± 1 | 920 ± 20 | 920 ± 25 |
| 500 ppm | −33 ± 5 | 6 ± 1 | 1250 ± 20 | 1290 ± 25 |
| |Z| | Double Layer (2 Time Constant) | Double Layer (1 Time Constant) | |||
|---|---|---|---|---|---|
| Ω·cm2 (±10%) | Qdl (F/cm2) (±10%) | n | Qdl (F/cm2) (±10%) | n | |
| Uncoated | 383,000 | 1 × 10−5 | 0.93 | ||
| Without particles | 362,000 | 1.37 × 10−5 | 0.84 | 1.97 × 10−5 | 0.98 |
| 50 ppm | 314,000 | 1.56 × 10−5 | 0.84 | 1.55 × 10−5 | 0.89 |
| 150 ppm | 374,000 | 1.37 × 10−5 | 0.89 | 1.37 × 10−5 | 0.92 |
| 500 ppm | 313,000 | 1.65 × 10−5 | 0.89 | 1.60 × 10−5 | 0.92 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Altube, A.; Rodríguez-Cambero, E.; Viñuales, A.I.; García-Lecina, E.; Díez, J.A.; Grande, H.J. Silane-Based Coatings Containing TiO2 for Corrosion Protection of 316L Stainless Steel. Corros. Mater. Degrad. 2026, 7, 10. https://doi.org/10.3390/cmd7010010
Altube A, Rodríguez-Cambero E, Viñuales AI, García-Lecina E, Díez JA, Grande HJ. Silane-Based Coatings Containing TiO2 for Corrosion Protection of 316L Stainless Steel. Corrosion and Materials Degradation. 2026; 7(1):10. https://doi.org/10.3390/cmd7010010
Chicago/Turabian StyleAltube, Ainhoa, Estibaliz Rodríguez-Cambero, Ana I. Viñuales, Eva García-Lecina, José Antonio Díez, and Hans Jürgen Grande. 2026. "Silane-Based Coatings Containing TiO2 for Corrosion Protection of 316L Stainless Steel" Corrosion and Materials Degradation 7, no. 1: 10. https://doi.org/10.3390/cmd7010010
APA StyleAltube, A., Rodríguez-Cambero, E., Viñuales, A. I., García-Lecina, E., Díez, J. A., & Grande, H. J. (2026). Silane-Based Coatings Containing TiO2 for Corrosion Protection of 316L Stainless Steel. Corrosion and Materials Degradation, 7(1), 10. https://doi.org/10.3390/cmd7010010







