Adsorption of Lanthanide Atoms on a Graphene Cluster Model Incorporating Stone–Wales Defect
Abstract
1. Introduction
2. Computational Methodology
3. Results and Discussion
- (1)
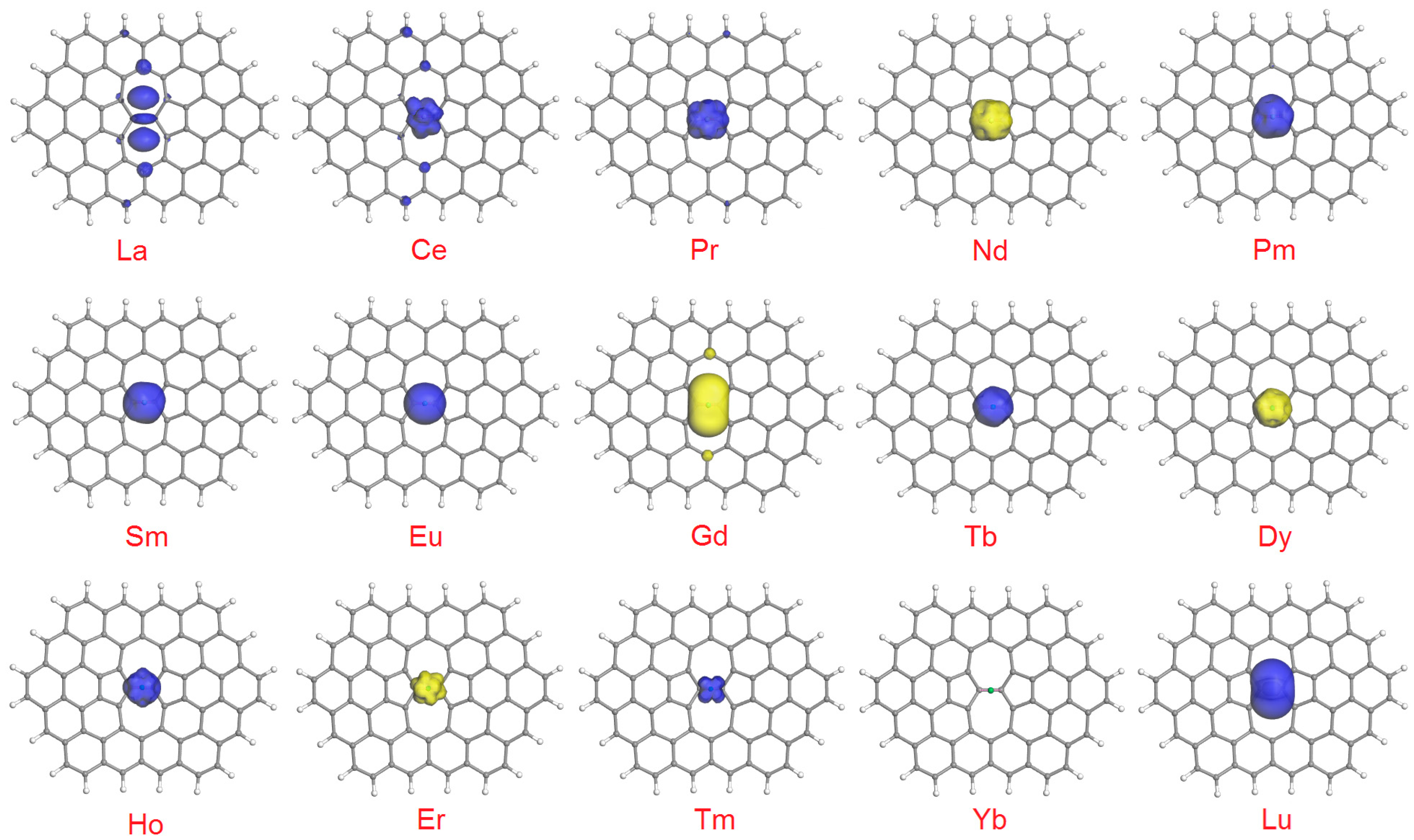
- Both HOMO and LUMO are found exclusively on the Ln atom: the cases of Sm and Tm;
- (2)
- Both HOMO and LUMO are found on the Ln atom, but LUMO also extends to the carbon atoms of SWG: this is observed for Tb, Dy, and Ho;
- (3)
- Both HOMO and LUMO are found on the Ln atom, plus minor lobes can be found on the C atoms (usually in proximity to the adsorption site): the cases of Ce, Nd, Eu, Er, Yb, and Lu;
- (4)
- HOMO is found exclusively (Pr and Pm) or mostly (La) on the Ln atom, but LUMO extends to the carbon atoms only;
- (5)
- LUMO is found solely on the lanthanide, and HOMO on Ln plus on (mainly adjacent) carbon atoms.
4. Conclusions
5. Outlook
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Izquierdo-García, P.; Fernández-García, J.M.; Martín, N. Twenty years of graphene: From pristine to chemically engineered nano-sized flakes. J. Am. Chem. Soc. 2024, 146, 32222–32234. [Google Scholar] [CrossRef]
- Dutta, T.; Chaturvedi, P.; Thakur, A.; Mishra, S.K. Review and outlook of graphene-based catalysis: Revolutionizing water splitting for sustainable hydrogen production. Energy Fuels 2025, 39, 8827–8870. [Google Scholar] [CrossRef]
- Elizalde-Herrera, F.J.; Flores-Soto, P.A.; Mora-Cortes, L.F.; González, F.J.; Soria-Arguello, G.; Avalos-Belmontes, F.; Narro-Céspedes, R.I.; Hoyos, M. Recent development of graphene-based composites for electronics, energy storage, and biomedical applications: A review. J. Compos. Sci. 2024, 8, 481. [Google Scholar] [CrossRef]
- Hashimoto, A.; Suenaga, K.; Gloter, A.; Urita, K.; Iijima, S. Direct evidence for atomic defects in graphene layers. Nature 2004, 430, 870–873. [Google Scholar] [CrossRef]
- Rutter, G.M.; Crain, J.N.; Guisinger, N.P.; Li, T.; First, P.N.; Stroscio, J.A. Scattering and interference in epitaxial graphene. Science 2007, 317, 219–222. [Google Scholar] [CrossRef]
- Gass, M.H.; Bangert, U.; Bleloch, A.L.; Wang, P.; Nair, R.R.; Geim, A.K. Free-standing graphene at atomic resolution. Nat. Nanotechnol. 2008, 3, 676–681. [Google Scholar] [CrossRef] [PubMed]
- Meyer, J.C.; Kisielowski, C.; Erni, R.; Rossell, M.D.; Crommie, M.F.; Zettl, A. Direct imaging of lattice atoms and topological defects in graphene membranes. Nano Lett. 2008, 8, 3582–3586. [Google Scholar] [CrossRef] [PubMed]
- Banhart, F.; Kotakoski, J.; Krasheninnikov, A.V. Structural defects in graphene. ACS Nano 2011, 5, 26–41. [Google Scholar] [CrossRef] [PubMed]
- Robertson, A.W.; Allen, C.S.; Wu, Y.A.; He, K.; Olivier, J.; Neethling, J.; Kirkland, A.I.; Warner, J.H. Spatial control of defect creation in graphene at the nanoscale. Nat. Commun. 2012, 3, 1144. [Google Scholar] [CrossRef]
- Wei, Y.; Wu, J.; Yin, H.; Shi, X.; Yang, R.; Dresselhaus, M. The nature of strength enhancement and weakening by pentagon–heptagon defects in graphene. Nat. Mater. 2012, 11, 759–763. [Google Scholar] [CrossRef]
- Lee, G.-D.; Yoon, E.; He, K.; Robertson, A.W.; Warner, J.H. Detailed formation processes of stable dislocations in graphene. Nanoscale 2014, 6, 14836–14844. [Google Scholar] [CrossRef]
- Bhatt, M.D.; Kim, H.; Kim, G. Various defects in graphene: A review. RSC Adv. 2022, 12, 21520–21547. [Google Scholar] [CrossRef]
- Thrower, P.A. The study of defects in graphite by transmission electron microscopy. Chem. Phys. Carbon 1969, 5, 217–319. [Google Scholar]
- Stone, A.J.; Wales, D.J. Theoretical studies of icosahedral C60 and some related structures. Chem. Phys. Lett. 1986, 128, 501–503. [Google Scholar]
- Tiwari, S.K.; Pandey, S.K.; Pandey, R.; Wang, N.; Bystrzejewski, M.; Mishra, Y.K.; Zhu, Y. Stone–Wales defect in graphene. Small 2023, 19, 2303340. [Google Scholar] [CrossRef]
- Chen, M.; Han, X.; Tang, K. Topological regulations of Stone–Wales graphene. Carbon 2024, 226, 119163. [Google Scholar] [CrossRef]
- Yang, F.O.; Huang, B.; Li, Z.; Xiao, J.; Wang, H.; Xu, H. Chemical functionalization of graphene nanoribbons by carboxyl groups on Stone–Wales defects. J. Phys. Chem. C 2008, 112, 12003–12007. [Google Scholar] [CrossRef]
- Chen, L.; Ouyang, Y.; Wang, Y.; Sun, Y.; Pan, H. The influence of Stone–Wales defects on magnetic properties in graphene. Phys. E Low-Dimens. Syst. Nanostruct. 2010, 43, 593–597. [Google Scholar] [CrossRef]
- Qin, X.; Meng, Q.; Zhao, W. Effects of Stone–Wales defect upon adsorption of formaldehyde on graphene sheet with or without Al dopant: A first principle study. Surf. Sci. 2011, 605, 930–933. [Google Scholar] [CrossRef]
- Chen, L.; Li, J.; Li, D.; Wei, M.; Wang, X. Chemical functionalization of graphene by H adsorption on Stone-Thrower-Wales defects. Solid State Commun. 2012, 152, 1985–1989. [Google Scholar] [CrossRef]
- Guang, H.; Aoki, M.; Tanaka, S.; Kohyama, M. Hole doping by adsorption of oxygen on a Stone–Thrower–Wales defect in graphene. Solid State Commun. 2013, 174, 10–15. [Google Scholar] [CrossRef]
- Zhou, Q.; Wang, C.; Fu, Z.; Tang, Y.; Zhang, H. Adsorption of formaldehyde molecule on Stone–Wales defected graphene doped with Cr, Mn, and Co: A theoretical study. Comp. Mater. Sci. 2014, 83, 398–402. [Google Scholar] [CrossRef]
- Harman, A.J.; Raelynn, L. Density functional theory study of metal adatoms at or near a Stone–Wales defect in graphene. Procedia Eng. 2014, 93, 2–7. [Google Scholar] [CrossRef]
- Kim, D.; Lee, S.; Hwang, Y.; Yun, K.-H.; Chung, Y.-C. Hydrogen storage in Li dispersed graphene with Stone–Wales defects: A first-principles study. Int. J. Hydrog. Energy 2014, 39, 13189–13194. [Google Scholar] [CrossRef]
- Yildirim, H.; Kinaci, A.; Zhao, Z.-J.; Chan, M.K.Y.; Greeley, J.P. First-principles analysis of defect-mediated Li adsorption on graphene. ACS Appl. Mater. Interfaces 2014, 6, 21141–21150. [Google Scholar] [CrossRef]
- Long, X.J.; Zhao, F.P.; Liu, H.K.; Huang, J.Y.; Lin, Y.; Zhu, J.; Luo, S.N. Anisotropic shock response of Stone–Wales defects in graphene. J. Phys. Chem. C 2015, 119, 7453–7460. [Google Scholar] [CrossRef]
- Ebrahimi, S. Influence of Stone–Wales defects orientations on stability of graphene nanoribbons under a uniaxial compression strain. Solid State Commun. 2015, 220, 17–20. [Google Scholar] [CrossRef]
- Zhou, Q.; Fu, Z.; Wang, C.; Tang, Y.; Zhang, H.; Yuan, L.; Yang, X. The electronic and magnetic properties of B-doping Stone–Wales defected graphene decorated with transition-metal atoms. Phys. E Low-Dimens. Syst. Nanostruct. 2015, 73, 257–261. [Google Scholar] [CrossRef]
- Fu, Y.; Ragab, T.; Basaran, C. The effect of Stone–Wales defects on the mechanical behavior of graphene nano-ribbons. Comp. Mater. Sci. 2016, 124, 142–150. [Google Scholar] [CrossRef]
- Zhang, X.; Tang, C.; Jiang, Q. Electric field induced enhancement of hydrogen storage capacity for Li atom decorated graphene with Stone–Wales defects. Int. J. Hydrog. Energy 2016, 41, 10776–10785. [Google Scholar] [CrossRef]
- Wang, Y.; Sun, X.; He, F.; Li, K.; Wu, Z. Haeckelite and N-doped haeckelite as catalysts for oxygen reduction reaction: Theoretical studies. J. Phys. Chem. C 2017, 121, 28339–28347. [Google Scholar] [CrossRef]
- Zaminpayma, E.; Razavi, M.E.; Nayebi, P. Electronic properties of graphene with single vacancy and Stone–Wales defects. Appl. Surf. Sci. 2017, 414, 101–106. [Google Scholar] [CrossRef]
- Rudi, S.G.; Faez, R.; Moravvej-Farshi, M.K.; Saghafi, K. Effect of Stone–Wales defect on an armchair graphene nanoribbon-based photodetector. Superlattices Microstruct. 2019, 130, 127–138. [Google Scholar] [CrossRef]
- Basiuk, V.A.; Kakazey, M.; Vlasova, M.; Basiuk, E.V. Effect of structural defects on the strength of adsorption of La and Lu species on graphene. Diam. Relat. Mater. 2019, 100, 107597. [Google Scholar] [CrossRef]
- Sadeghzadeh, S.; Ghojavand, M.; Mahmoudi, J. Influence of Stone–Wales defects on the mechanical properties of graphene-like polyaniline (PANI) C3N nanosheets. Diam. Relat. Mater. 2020, 101, 107555. [Google Scholar] [CrossRef]
- Kumar, J.; Ansh; Shrivastava, M. Stone–Wales defect and vacancy-assisted enhanced atomic orbital interactions between graphene and ambient gases: A first-principles insight. ACS Omega 2020, 5, 31281–31288. [Google Scholar] [CrossRef] [PubMed]
- Arokiyanathan, A.L.; Panjulingam, N.; Lakshmipathi, S. Chemical properties of lithium cluster (Lix, x = 2–8) on Stone–Wales defect graphene sheet: A DFT study. J. Phys. Chem. C 2020, 124, 7229–7237. [Google Scholar] [CrossRef]
- Yang, S.; Qian, X.; Xu, H.; Xiong, J.; Wang, Z.; Gu, H. The improved CO adsorption/sensing performance of Stone–Wales defected graphene doped with Fe: A DFT study. Phys. E Low-Dimens. Syst. Nanostruct. 2021, 128, 114603. [Google Scholar] [CrossRef]
- Mendoza-Domínguez, C.U.; Basiuk, V.A. Distortion and bonding strength of phthalocyanine molecules adsorbed on topological defects in graphene. Mater. Chem. Phys. 2021, 271, 124963. [Google Scholar] [CrossRef]
- Mendoza-Domínguez, C.U.; Basiuk, V.A. Adsorption of yttrium bisphthalocyanine on pristine and defect-contaning graphene models: A DFT study. Diam. Relat. Mater. 2022, 126, 109051. [Google Scholar] [CrossRef]
- Rudi, S.G.; Soleimani-Amiri, S. Bilayer armchair graphene nanoribbon photodetector with Stone–Wales defect: A computational study. Mater. Sci. Semicond. Proc. 2022, 150, 106918. [Google Scholar] [CrossRef]
- Mendoza-Domínguez, C.U.; Bolivar-Pineda, L.M.; Basiuk, V.A. Effect of structural defects in graphene on the geometry and electronic properties of adsorbed lanthanide bisphthalocyanines: A DFT analysis. Comp. Theor. Chem. 2023, 1225, 114152. [Google Scholar] [CrossRef]
- Oliveira, T.A.; Silva, P.V.; Meunier, V.; Girão, E.C. Tuning the carrier mobility and electronic structure of graphene nanoribbons using Stone–Wales defects. Carbon 2023, 201, 222–233. [Google Scholar] [CrossRef]
- Jovanović, A.Z.; Dobrota, A.S.; Skorodumova, N.V.; Pašti, I.A. Reactivity of Stone–Wales defect in graphene lattice—DFT study. FlatChem 2023, 42, 100573. [Google Scholar] [CrossRef]
- Wang, S.F.; Xue, D.; Liang, J.; Chen, L.Y.; Xie, Y.; Zhang, J.M. The synergistic regulation effect on the structure and electronic properties of graphene by methane plasma, Stone–Wales defect and equibiaxial strain. Diam. Relat. Mater. 2023, 140, 110426. [Google Scholar] [CrossRef]
- An, Y. Barrier height breakpoint in employing the Arrhenius equation to proton isotope permeation through Stone–Wales graphene. J. Phys. Chem. Lett. 2023, 14, 10779–10783. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, M.; Chowdhury, S.; Majumdar, A.; Jana, D. Stone–Wales decorated phagraphene: A potential candidate for supercapacitor electrodes and thermal transport. ACS Appl. Electr. Mater. 2023, 5, 6725–6737. [Google Scholar] [CrossRef]
- Yuan, P.; Li, C.; Zhang, J.; Wang, F.; Chen, X. The special activity of Stone–Wales defect graphene for the oxygen reduction reaction: A comparison study between the charge-neutral model and the constant potential model calculated by density functional theory. J. Phys. Chem. C 2024, 128, 3250–3259. [Google Scholar] [CrossRef]
- Basiuk, V.A.; Wu, Y.; Prezhdo, O.V.; Basiuk, E.V. Lanthanide atoms induce strong graphene sheet distortion when adsorbed on Stone–Wales defects. J. Phys. Chem. Lett. 2024, 15, 9706–9713. [Google Scholar] [CrossRef]
- Kotakoski, J.; Meyer, J.C.; Kurasch, S.; Santos-Cottin, D.; Kaiser, U.; Krasheninnikov, A.V. Stone-Wales-type transformations in carbon nanostructures driven by electron irradiation. Phys. Rev. B 2011, 83, 245420. [Google Scholar] [CrossRef]
- Bandyopadhyay, D.; Sun, Y.; Griffin, R.; Lee, L.Y. Advancing graphene imaging for clear identification of lattice defects: The application of revolve sphere levelling to scanning tunnelling microscopy images. ACS Omega 2024, 9, 43447–43452. [Google Scholar] [CrossRef]
- Basiuk, V.A.; Basiuk, E.V. The coordination of lanthanide atoms with Stone–Wales defects on graphene: A cluster DFT analysis using ECP pseudopotentials. Surfaces 2025, 8, 32. [Google Scholar] [CrossRef]
- Basiuk, V.A.; Prezhdo, O.V.; Basiuk, E.V. Adsorption of lanthanide atoms on graphene: Similar, yet different. J. Phys. Chem. Lett. 2022, 13, 6042–6047. [Google Scholar] [CrossRef] [PubMed]
- Basiuk, E.V.; Prezhdo, O.V.; Basiuk, V.A. Strong bending distortion of supercoronene graphene model upon adsorption of lanthanide atoms. J. Phys. Chem. Lett. 2023, 14, 2910–2916. [Google Scholar] [CrossRef]
- Basiuk, V.A.; Acevedo-Guzmán, D.A.; Meza-Laguna, V.; Álvarez-Zauco, E.; Huerta, L.; Serrano, M.; Kakazey, M.; Basiuk, E.V. High-energy ball-milling preparation and characterization of Ln2O3−graphite nanocomposites. Mater. Today Commun. 2021, 26, 102030. [Google Scholar] [CrossRef]
- Delley, B. An all-electron numerical method for solving the local density functional for polyatomic molecules. J. Chem. Phys. 1990, 92, 508–517. [Google Scholar] [CrossRef]
- Delley, B. Fast calculation of electrostatics in crystals and large molecules. J. Phys. Chem. 1996, 100, 6107–6110. [Google Scholar] [CrossRef]
- Delley, B. From molecules to solids with the DMol3 approach. J. Chem. Phys. 2000, 113, 7756–7764. [Google Scholar] [CrossRef]
- Delley, B.; Ellis, D.; Freeman, A.; Baerends, E.; Post, D. Binding energy and electronic structure of small copper particles. Phys. Rev. B 1983, 27, 2132–2144. [Google Scholar] [CrossRef]
- Perdew, J.; Burke, K.; Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 1996, 77, 3865–3868. [Google Scholar] [CrossRef]
- Grimme, S. Semiempirical GGA-type density functional constructed with a long-range dispersion correction. J. Comput. Chem. 2006, 27, 1787–1799. [Google Scholar] [CrossRef]
- Basiuk, V.A. Interaction of tetraaza[14]annulenes with single-walled carbon nanotubes: A DFT study. J. Phys. Chem. B 2004, 108, 19990–19994. [Google Scholar] [CrossRef]
- Dolg, M.; Wedig, U.; Stoll, H.; Preuss, H. Energy-adjusted ab initio pseudopotentials for the first row transition elements. J. Chem. Phys. 1987, 86, 866. [Google Scholar] [CrossRef]
- Bergner, A.; Dolg, M.; Kuechle, W.; Stoll, H.; Preuss, H. Ab initio energy-adjusted pseudopotentials for elements of groups 13–17. Mol. Phys. 1993, 80, 1431–1441. [Google Scholar] [CrossRef]
- Basiuk, V.A. DFT calculations on single lanthanide atoms: Is the task as simple as it seems? Comp. Theor. Chem. 2025, 1248, 115170. [Google Scholar] [CrossRef]
- Basiuk, V.A.; Henao-Holguín, L.V. Effects of orbital cutoff in DMol3 DFT calculations: A case study of meso-tetraphenylporphine−C60 complex. J. Comput. Theor. Nanosci. 2013, 10, 1266–1272. [Google Scholar] [CrossRef]
- Chávez-Colorado, E.; Basiuk, V.A. Noncovalent interactions of free-base phthalocyanine with elongated fullerenes as carbon nanotube models. Struct. Chem. 2017, 28, 1765–1773. [Google Scholar] [CrossRef]
- Basiuk, V.A.; Chávez-Colorado, E. Adsorption of free-base phthalocyanine on Stone-Wales defect-containing carbon nanotubes: A DFT study. Diam. Relat. Mater. 2019, 97, 107443. [Google Scholar] [CrossRef]
- Basiuk, V.A.; Prezhdo, O.V.; Basiuk, E.V. Thermal smearing in DFT calculations: How small is really small? A case of La and Lu atoms adsorbed on graphene. Mater. Today Commun. 2020, 25, 101595. [Google Scholar] [CrossRef]
- Basiuk, V.A. Electron smearing in DFT calculations: A case study of doxorubicin interaction with single-walled carbon nanotubes. Int. J. Quantum Chem. 2011, 111, 4197–4205. [Google Scholar] [CrossRef]
- Martínez-Flores, C.; Bolivar-Pineda, L.M.; Basiuk, V.A. Lanthanide bisphthalocyanine single-molecule magnets: A DFT survey of their geometries and electronic properties from lanthanum to lutetium. Mater. Chem. Phys. 2022, 287, 126271. [Google Scholar] [CrossRef]
- Slanina, Z.; Uhlík, F.; Akasaka, T.; Lu, X.; Adamowicz, L. Computational modeling of the Ce@C82 metallofullerene isomeric composition. ECS J. Solid State Sci. Technol. 2019, 8, M118–M121. [Google Scholar] [CrossRef]
- Slanina, Z.; Uhlík, F.; Akasaka, T.; Lu, X.; Adamowicz, L. Calculated relative thermodynamic stabilities of the Gd@C82 isomers. ECS J. Solid State Sci. Technol. 2021, 10, 071013. [Google Scholar] [CrossRef]
- Manz, T.A. Apples to apples comparison of standardized to unstandardized principal component analysis of methods that assign partial atomic charges in molecules. RSC Adv. 2022, 12, 31617. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Wang, C.Z.; Hupalo, M.; Yao, Y.X.; Tringides, M.C.; Lu, W.C.; Ho, K.M. Adsorption and growth morphology of rare-earth metals on graphene studied by ab initio calculations and scanning tunneling microscopy. Phys. Rev. B 2010, 82, 245408. [Google Scholar] [CrossRef]
- Luo, H.; Xu, K.; Gong, Z.; Li, N.; Zhang, K.; Wu, W. NH3, PH3, AsH3 adsorption and sensing on rare earth metal doped graphene: DFT insights. Appl. Surf. Sci. 2021, 566, 150390. [Google Scholar] [CrossRef]
- Bissett, M.A.; Konabe, S.; Okada, S.; Tsuji, M.; Ago, H. Enhanced chemical reactivity of graphene induced by mechanical strain. ACS Nano 2013, 7, 10335–10343. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Wu, Y.; Hao, Y.; Geng, J.; Charlton, M.; Chen, S.; Ren, Y.; Ji, H.; Li, H.; Boukhvalov, D.W.; et al. Selective surface functionalization at regions of high local curvature in graphene. Chem. Commun. 2013, 49, 677–679. [Google Scholar] [CrossRef] [PubMed]
- Qu, Y.; Ke, Y.; Shao, Y.; Chen, W.; Kwok, C.T.; Shi, X.; Pan, H. Effect of curvature on the hydrogen evolution reaction of graphene. J. Phys. Chem. C 2018, 122, 25331–25338. [Google Scholar] [CrossRef]
- Basiuk, V.A.; Bolivar-Pineda, L.M.; Meza-Laguna, V.; Rybak-Akimova, E.V.; Basiuk, E.V. Carbon nanotubes and graphene promote pyrolysis of free-base phthalocyanine. J. Phys. Chem. Lett. 2018, 9, 4420–4427. [Google Scholar] [CrossRef]
- Kosmala, T.; Baby, A.; Lunardon, M.; Perilli, D.; Liu, H.; Durante, C.; Di Valentin, C.; Agnoli, S.; Granozzi, G. Operando Visualization of the hydrogen evolution reaction with atomic-scale precision at different metal–graphene interfaces. Nat. Catal. 2021, 4, 850–859. [Google Scholar] [CrossRef]
- Banerjee, S.; Rappe, A.M. Mechanochemical molecular migration on graphene. J. Am. Chem. Soc. 2022, 144, 7181–7188. [Google Scholar] [CrossRef]
- Hawthorne, N.; Banerjee, S.; Moore, Q.; Rappe, A.M.; Batteas, J.D. Studies of the reactivity of graphene driven by mechanical distortions. J. Phys. Chem. C 2022, 126, 17569–17578. [Google Scholar] [CrossRef]
- Kim, C.-E.; Lee, J.; Walsh, A.; Lordi, V.; Bahr, D.F. Role of ripples in altering the electronic and chemical properties of graphene. J. Chem. Phys. 2022, 156, 054708. [Google Scholar] [CrossRef]
- Sun, P.Z.; Xiong, W.Q.; Bera, A.; Timokhin, I.; Wu, Z.F.; Mishchenko, A.; Sellers, M.C.; Liu, B.L.; Cheng, H.M.; Janzen, E.; et al. Unexpected catalytic activity of nanorippled graphene. Proc. Natl. Acad. Sci. USA 2023, 120, e2300481120. [Google Scholar] [CrossRef]
- Rodríguez-Galván, A.; Rivera, M.; García-López, P.; Medina, L.A.; Basiuk, V.A. Gadolinium-containing carbon nanomaterials for magnetic resonance imaging: Trends and challenges. J. Cell. Mol. Med. 2020, 24, 3779–3794. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Flores, C.; Basiuk, V.A. Ln@C60 endohedral fullerenes: A DFT analysis for the complete series from lanthanum to lutetium. Comp. Theor. Chem. 2022, 1217, 113878. [Google Scholar] [CrossRef]
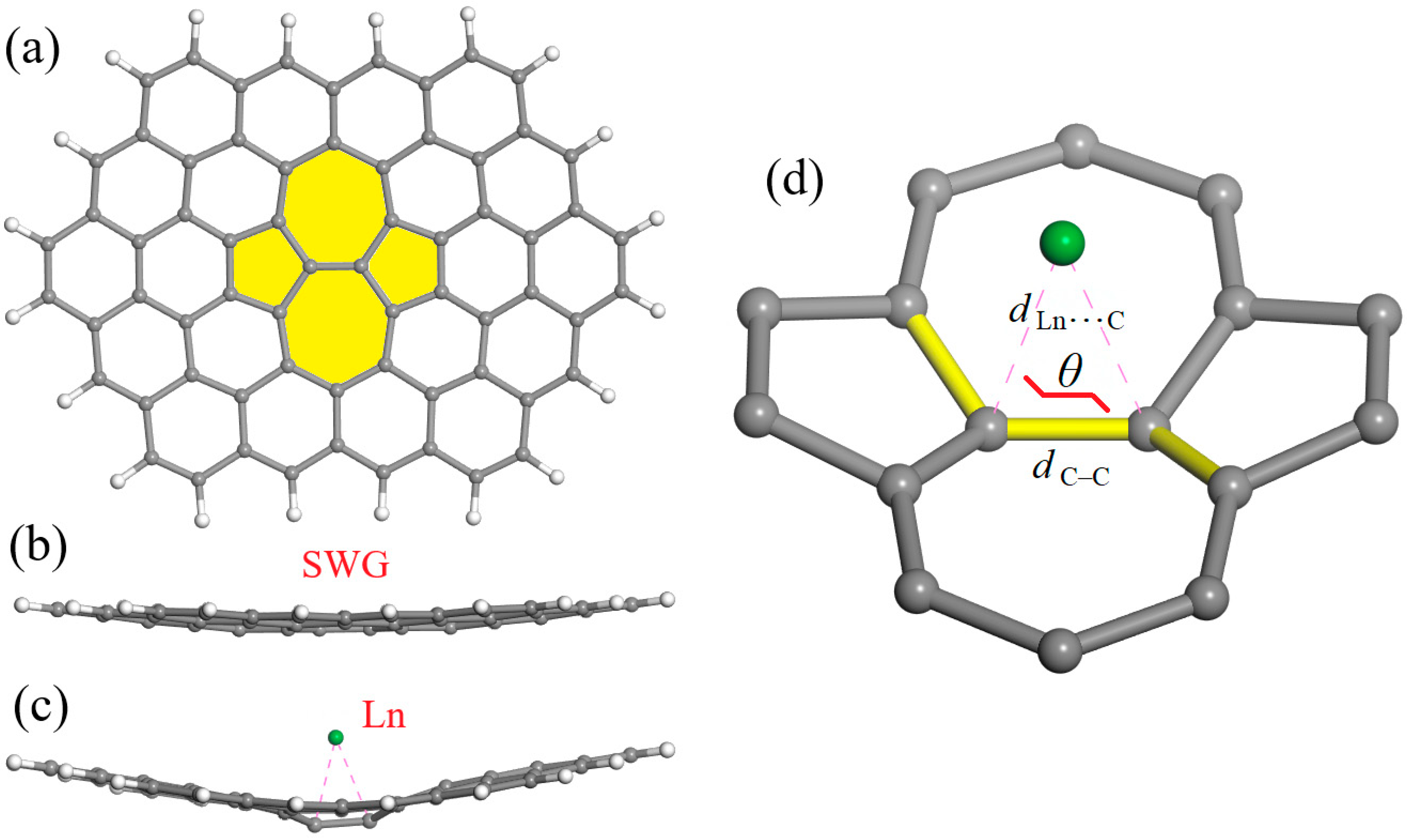
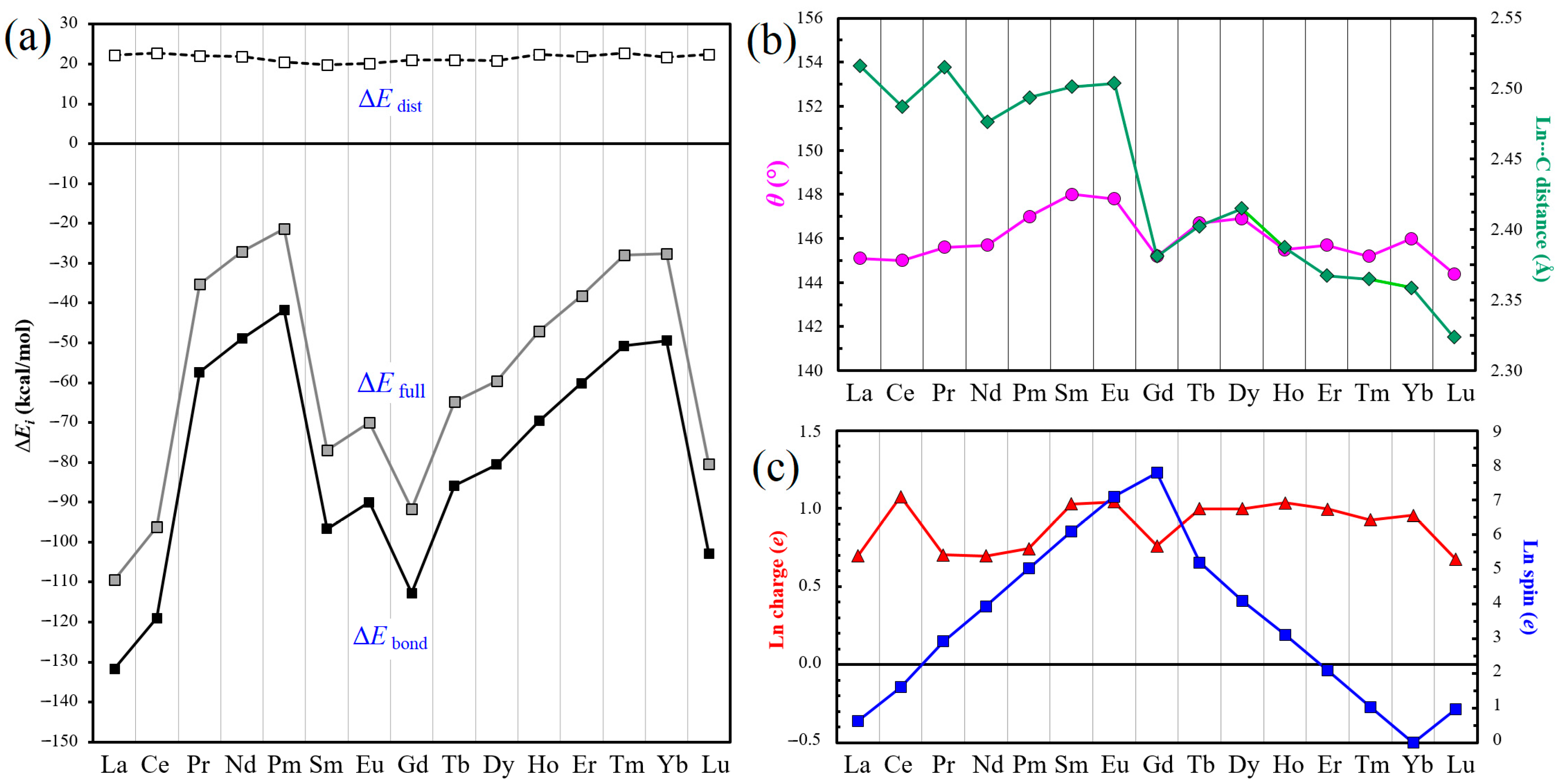

| Atomic Number | Ln | ΔEfull (kcal/mol) | ΔEdist (kcal/mol) | ΔEbond (kcal/mol) | HOMO (eV) | LUMO (eV) | HOMO–LUMO Gap (eV) |
|---|---|---|---|---|---|---|---|
| 57 | La | −109.4 | 22.3 | −131.7 | −3.387 | −3.134 | 0.253 |
| 58 | Ce | −96.2 | 22.8 | −119.0 | −3.287 | −3.110 | 0.177 |
| 59 | Pr | −35.2 | 22.1 | −57.3 | −3.290 | −3.106 | 0.184 |
| 60 | Nd | −27.0 | 21.9 | −48.9 | −3.431 | −3.107 | 0.324 |
| 61 | Pm | −21.3 | 20.5 | −41.8 | −3.279 | −3.101 | 0.178 |
| 62 | Sm | −76.9 | 19.8 | −96.7 | −3.574 | −3.310 | 0.264 |
| 63 | Eu | −69.9 | 20.1 | −90.0 | −4.102 | −3.115 | 0.987 |
| 64 | Gd | −91.7 | 21.0 | −112.7 | −3.687 | −3.659 | 0.028 |
| 65 | Tb | −64.8 | 21.0 | −85.8 | −3.483 | −3.200 | 0.283 |
| 66 | Dy | −59.6 | 20.9 | −80.5 | −3.750 | −3.211 | 0.539 |
| 67 | Ho | −47.1 | 22.5 | −69.6 | −3.623 | −3.273 | 0.350 |
| 68 | Er | −38.2 | 21.9 | −60.1 | −3.482 | −3.297 | 0.185 |
| 69 | Tm | −27.9 | 22.8 | −50.7 | −3.718 | −3.333 | 0.385 |
| 70 | Yb | −27.6 | 21.8 | −49.4 | −4.054 | −3.387 | 0.667 |
| 71 | Lu | −80.4 | 22.4 | −102.8 | −4.249 | −3.568 | 0.681 |
| Atomic Number | Ln | dLn…C (Å) | dC–C (Å) | θ (°) | Ln Charge (e) | Ln Spin (e) |
|---|---|---|---|---|---|---|
| 57 | La | 2.473, 2.480 | 1.419 | 145.1 | 0.699 | 0.634 |
| 58 | Ce | 2.450, 2.460 | 1.420 | 145.0 | 1.077 | 1.601 |
| 59 | Pr | 2.472, 2.484 | 1.423 | 145.6 | 0.703 | 2.923 |
| 60 | Nd | 2.441, 2.458 | 1.422 | 145.7 | 0.696 | −3.933 |
| 61 | Pm | 2.455, 2.469 | 1.421 | 147.0 | 0.743 | 5.019 |
| 62 | Sm | 2.461, 2.464 | 1.421 | 148.0 | 1.029 | 6.093 |
| 63 | Eu | 2.463, 2.465 | 1.422 | 147.8 | 1.043 | 7.093 |
| 64 | Gd | 2.365, 2.366 | 1.414 | 145.2 | 0.760 | −7.786 |
| 65 | Tb | 2.382, 2.382 | 1.422 | 146.7 | 0.999 | 5.191 |
| 66 | Dy | 2.392, 2.392 | 1.423 | 146.9 | 0.998 | −4.096 |
| 67 | Ho | 2.370, 2.379 | 1.424 | 145.5 | 1.037 | 3.115 |
| 68 | Er | 2.354, 2.370 | 1.423 | 145.7 | 0.997 | −2.080 |
| 69 | Tm | 2.352, 2.354 | 1.425 | 145.2 | 0.928 | 1.039 |
| 70 | Yb | 2.347, 2.349 | 1.424 | 146.0 | 0.956 | −0.002 |
| 71 | Lu | 2.319, 2.322 | 1.420 | 144.4 | 0.674 | 0.970 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Basiuk, V.A.; Basiuk, E.V. Adsorption of Lanthanide Atoms on a Graphene Cluster Model Incorporating Stone–Wales Defect. Surfaces 2025, 8, 63. https://doi.org/10.3390/surfaces8030063
Basiuk VA, Basiuk EV. Adsorption of Lanthanide Atoms on a Graphene Cluster Model Incorporating Stone–Wales Defect. Surfaces. 2025; 8(3):63. https://doi.org/10.3390/surfaces8030063
Chicago/Turabian StyleBasiuk, Vladimir A., and Elena V. Basiuk. 2025. "Adsorption of Lanthanide Atoms on a Graphene Cluster Model Incorporating Stone–Wales Defect" Surfaces 8, no. 3: 63. https://doi.org/10.3390/surfaces8030063
APA StyleBasiuk, V. A., & Basiuk, E. V. (2025). Adsorption of Lanthanide Atoms on a Graphene Cluster Model Incorporating Stone–Wales Defect. Surfaces, 8(3), 63. https://doi.org/10.3390/surfaces8030063







