Fe2O3 Blocking Layer Produced by Cyclic Voltammetry Leads to Improved Photoelectrochemical Performance of Hematite Nanorods
Abstract
:1. Introduction
2. Materials and Methods
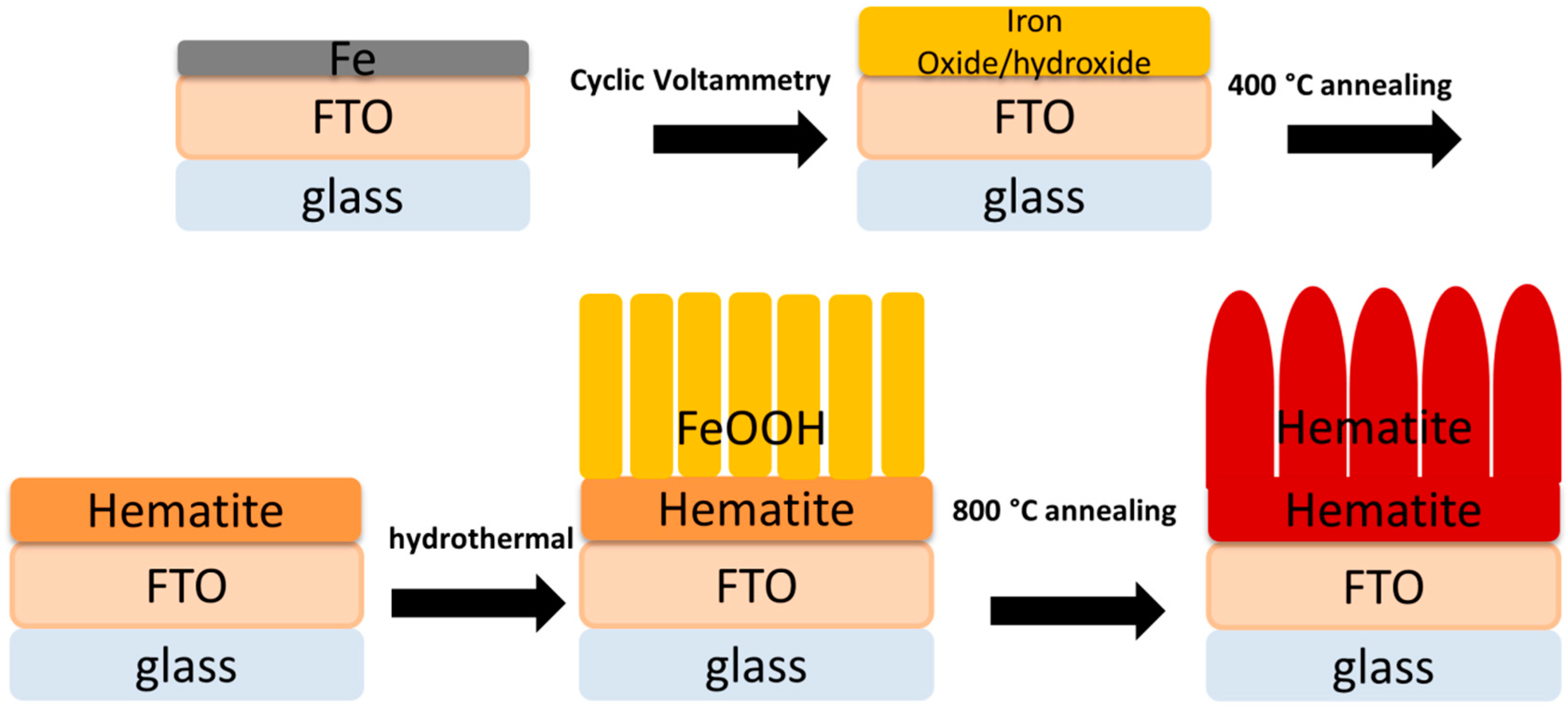
2.1. Materials Fabrication
2.2. Zn-Co LDH Treated Samples
2.3. Morphological and Structural Characterization
2.4. Photoelectrochemical (PEC) Measurements
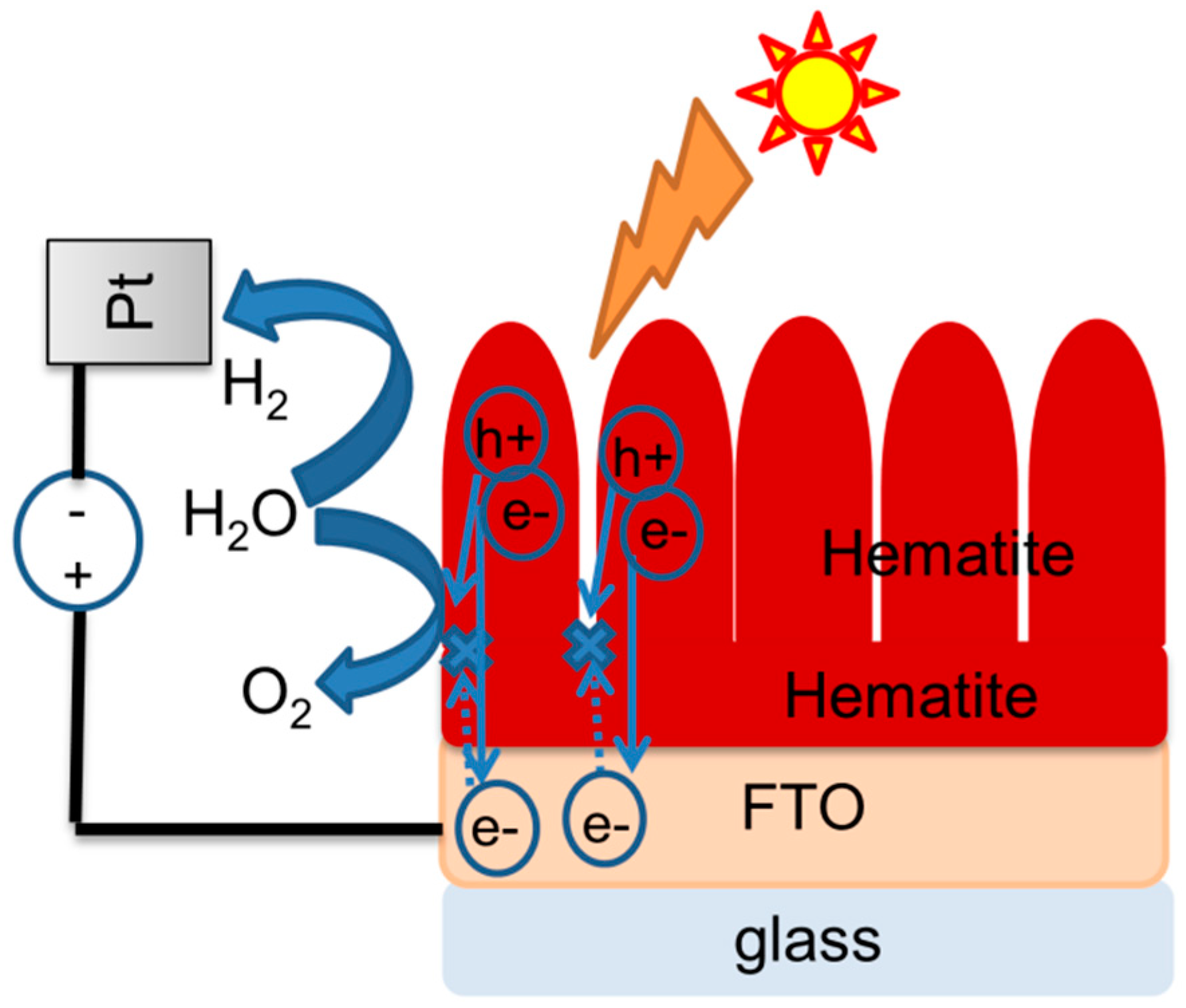
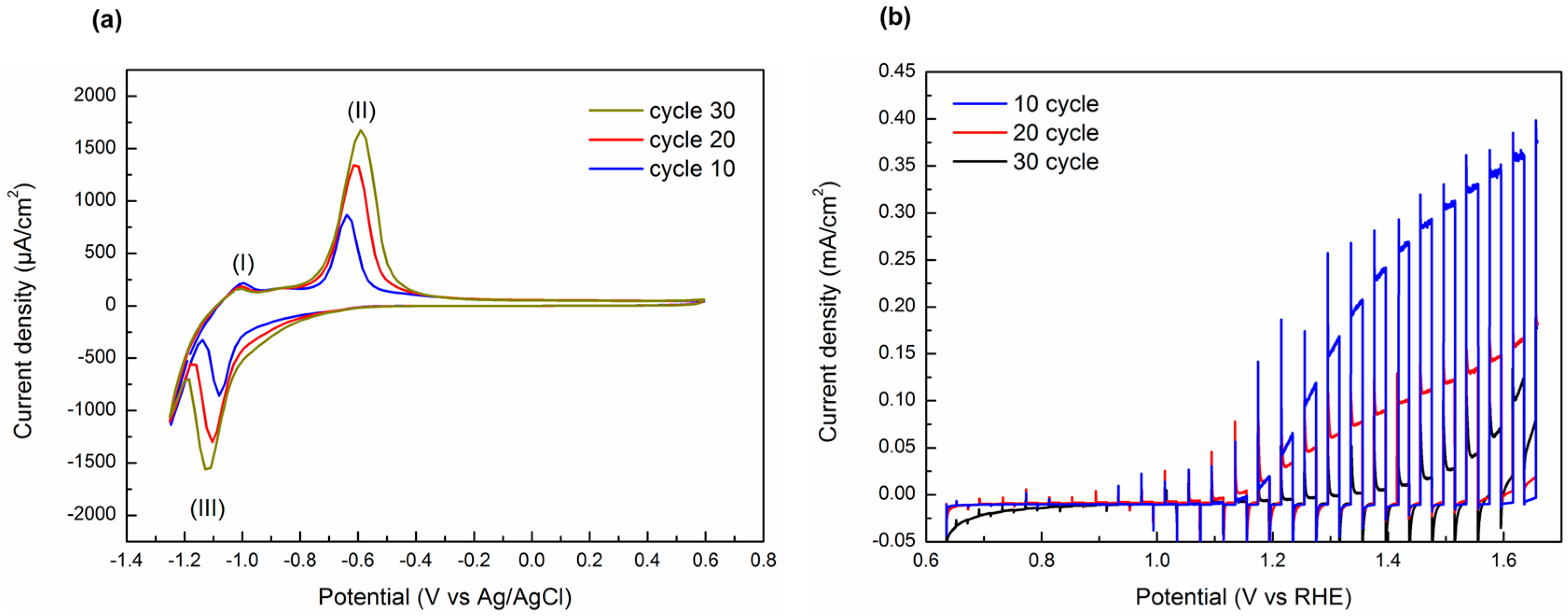
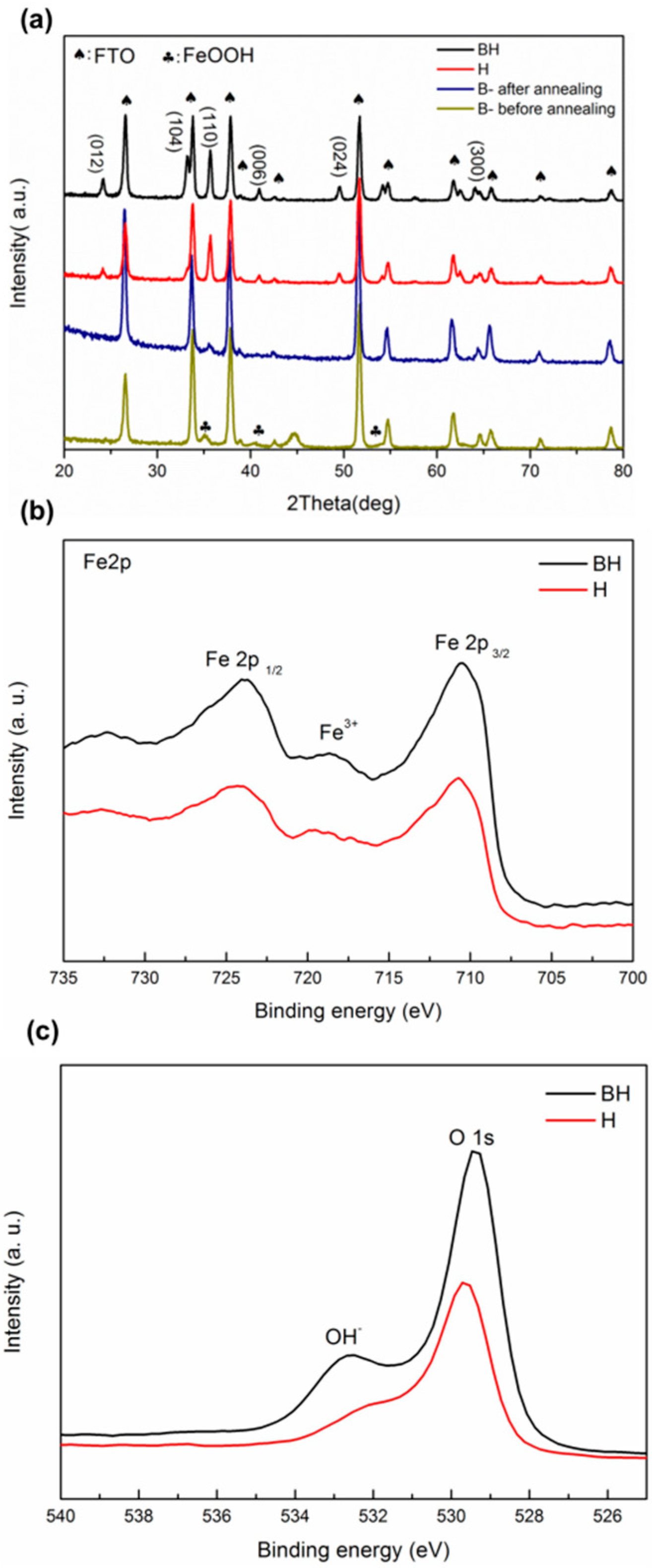
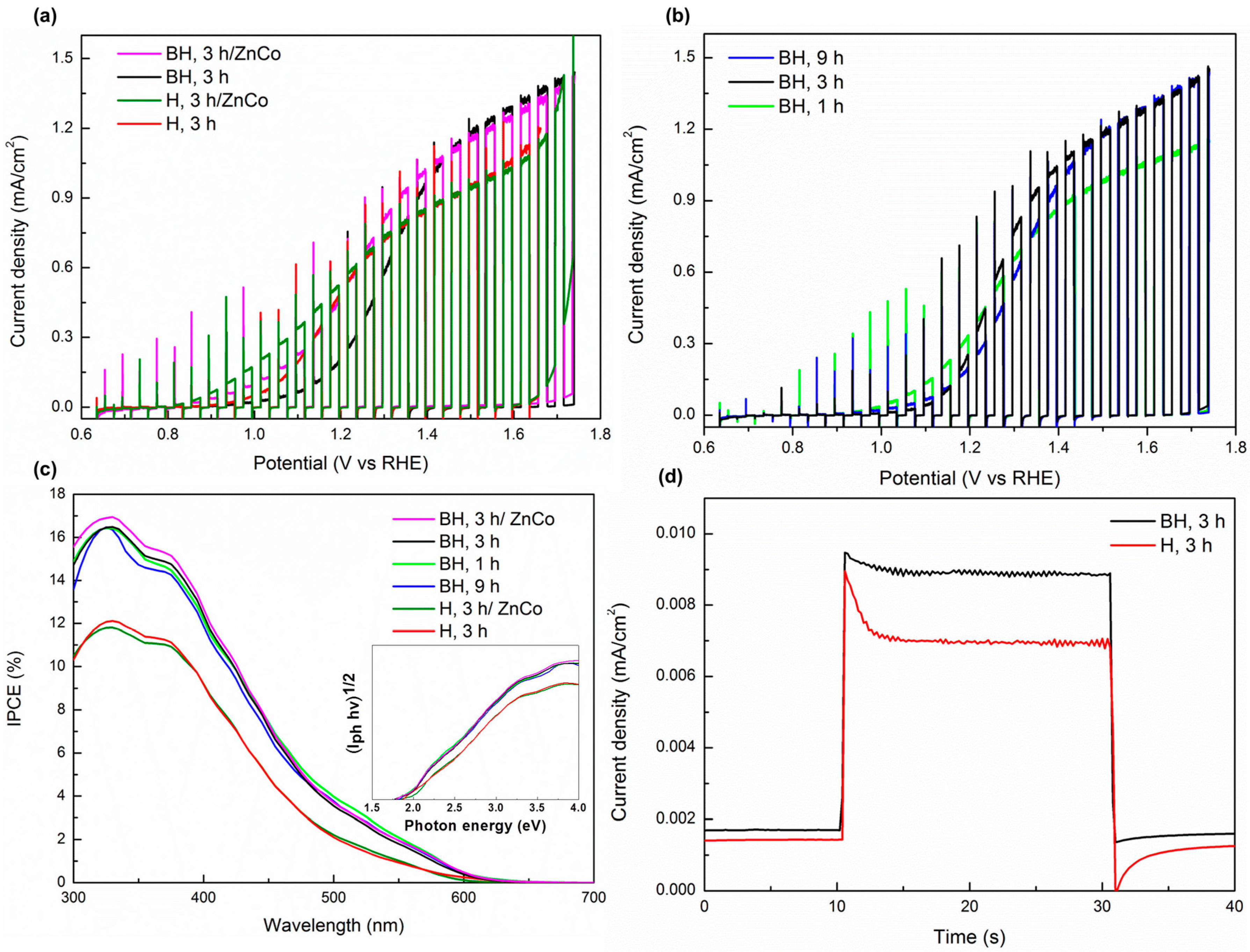
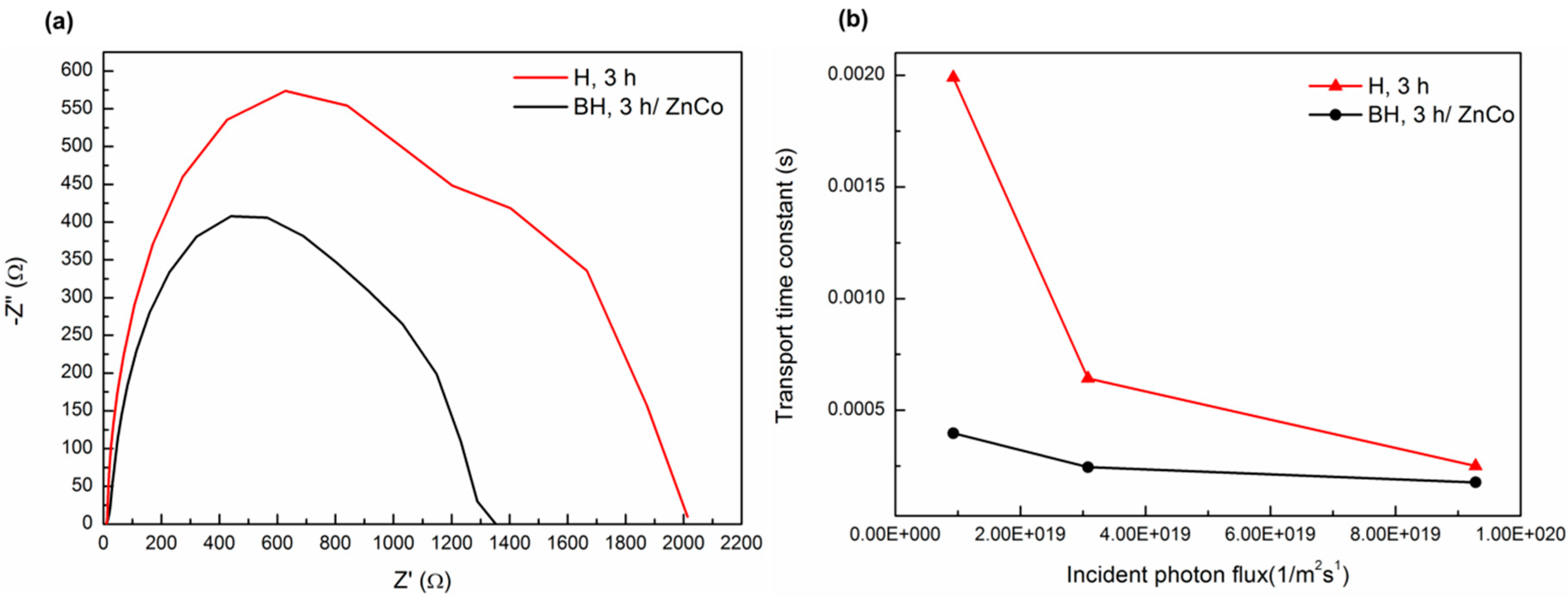
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sivula, K.; Le Formal, F.; Grätzel, M. Solar water splitting: progress using hematite (α-Fe2O3) photoelectrodes. ChemSusChem 2011, 4, 432–499. [Google Scholar] [CrossRef] [PubMed]
- Roy, P.; Berger, S.; Schmuki, P. TiO2 nanotubes: Synthesis and applications. Angew. Chem. Int. Ed. 2011, 50, 2904–2939. [Google Scholar] [CrossRef] [PubMed]
- Grätzel, M. Photoelectrochemical Cells. Nature 2006, 414, 338–344. [Google Scholar] [CrossRef] [PubMed]
- Kin, J.Y.; Youn, D.H.; Kim, J.H.; Kim, H.G.; Lee, J.S. Nanostructures-preserved hematite thin film for efficient solar water splitting. ACS Appl. Mater. Interfaces 2015, 7, 14123–14129. [Google Scholar] [CrossRef]
- Khan, M.E.; Khan, M.M.; Cho, M.H. Fabrication of WO3 nanorods on graphene nanosheets for improved visible light-induced photocapacitive and photocatalytic performance. RSC Adv. 2016, 6, 20824–20833. [Google Scholar] [CrossRef]
- Khan, M.E.; Khan, M.M.; Min, B.K.; Cho, M.H. Microbial fuel cell assisted band gap narrowed TiO2 for visible light-induced photocatalytic activities and power generation. Sci. Rep. 2018, 8, 1723–1735. [Google Scholar] [CrossRef] [PubMed]
- Grigorescu, S.; Lee, C.Y.; Lee, K.; Albu, S.; Paramasivam, I.; Demetrescu, I. Thermal air oxidation of Fe: Rapid hematite nanowire growth and photoelectrochemical water splitting performance. Electrochem. Commun. 2012, 23, 59–62. [Google Scholar] [CrossRef]
- Gajda-Schrantz, K.; Tymen, S.; Boudoire, F.; Toth, R.; Bora, D.K.; Calvet, W.; Gratzel, M.; Constable, E.C.; Braun, A. Formation of an electron hole doped film in the α-Fe2O3 photoanode upon electrochemical oxidation. Phys. Chem. Chem. Phys. 2013, 15, 1443–1451. [Google Scholar] [CrossRef]
- Sivula, K.; Formal, F.L.; Grätzel, M. WO3–Fe2O3 Photoanodes for water splitting: A host scaffold, guest absorber approach. Chem. Mater. 2009, 21, 2862–2867. [Google Scholar] [CrossRef]
- Peerakiatkhajohn, P.; Yun, J.H.; Chen, H.; Lyu, M.; Butburee, T.; Wang, L. Stable hematite nanosheet photoanodes for enhanced photoelectrochemical water splitting. Adv. Mater. 2016, 28, 6405–6410. [Google Scholar] [CrossRef]
- Kay, A.; Cesar, I.; Grätzel, M. New benchmark for water photooxidation by nanostructured α-Fe2O3 films. J. Am. Chem. Soc. 2006, 128, 15714–15721. [Google Scholar] [CrossRef] [PubMed]
- Rangaraju, R.R.; Panday, A.; Raja, K.S.; Misra, M. Nanostructured anodic iron oxide film as photoanode for water oxidation. J. Phys. D Appl. Phys. 2009, 42, 1–10. [Google Scholar] [CrossRef]
- Barroso, M.; Mesa, C.A.; Pendlebury, S.H.; Cowan, A.J.; Hisatomi, T.; Sivula, K.; Grätzel, M.; Klug, D.R.; Durrant, J.R. Dynamics of photogenerated holes in surface modified α-Fe2O3 photoanodes for solar water splitting. PANS 2012, 109, 15640–15645. [Google Scholar] [CrossRef] [PubMed]
- Yue, G.; Ni, H.W.; Chen, R.S.; Li, Y.W.; Li, J.H. Time-dependent growth of hematiete (α-Fe2O3) nanotube arrays produced by iron anodizing in ethylene glycol solution. Adv. Mater. Res. 2012, 599, 145–150. [Google Scholar] [CrossRef]
- Zhang, Z.; Hossain, M.F.; Takahashi, T. Self-assembled hematite (α-Fe2O3) nanotube arrays for photoelectrocatalytic degradation of azo dye under simulated solar light irradiation. Appl. Catal. B Environ. 2010, 95, 423–429. [Google Scholar] [CrossRef]
- Liu, T.; Ling, Y.; Yang, Y.; Finn, L.; Collazo, E.; Zhai, T.; Tong, Y.; Li, Y. Investigation of hematite nanorod–nanoflake morphological transformation and the application of ultrathin nanoflakes for electrochemical devices. Nano Energy 2015, 12, 169–177. [Google Scholar] [CrossRef]
- Wang, L.; Lee, C.Y.; Kirchgeorg, R.; Hildebrand, H.; Müller, J.; Spiecker, E.; Schmuki, P. A significant cathodic shift in the onset potential of photoelectrochemical water splitting for hematite nanostructures grown from Fe-Si alloys. Mater. Horizons 2014, 1, 344–347. [Google Scholar] [CrossRef]
- Wen, X.; Wang, S.; Ding, Y.; Wang, Z.L.; Yang, S. Controlled growth of large-area, uniform, vertically aligned arrays of α-Fe2O3 nanobelts and nanowires. J. Phys. Chem. B 2005, 109, 215–220. [Google Scholar] [CrossRef]
- Vincent, T.; Gross, M.; Dotan, H.; Rothschild, A. Thermally oxidized iron oxide nanoarchitectures for hydrogen production by solar-induced water splitting. Int. J. Hydrogen Energy 2012, 37, 8102–8109. [Google Scholar] [CrossRef]
- Li, S.; Cai, J.; Mei, Y.; Ren, Y.; Qin, G. Thermal Oxidation Preparation of Doped Hematite Thin Films for Photoelectrochemical Water Splitting. Int. J. Photoenergy 2014, 1–6. [Google Scholar] [CrossRef]
- Shinde, P.S.; Lee, H.H.; Lee, S.Y.; Lee, Y.M.; Jang, J.S. PRED treatment mediated stable and efficient water oxidation performance of the Fe2O3 nano-coral structure. Nanoscale 2015, 7, 14906–14913. [Google Scholar] [CrossRef] [PubMed]
- Chernomordik, B.D.; Russell, H.B.; Cvelbar, U.; Jasinski, J.B.; Kumar, V.; Deutsch, T.; Sunkara, M.K. Photoelectrochemical activity of as-grown, α-Fe2O3 nanowire array electrodes for water splitting. Nanotechnology 2012, 23, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, D.A.; Wang, G.; Ling, Y.; Li, Y.; Zhang, J.Z. Nanostructured hematite: synthesis, characterization, charge carrier dynamics, and photoelectrochemical properties. Energy Environ. Sci. 2012, 5, 6682–6702. [Google Scholar] [CrossRef]
- Rangaraju, R.R.; Raja, K.S.; Panday, A.; Misra, M. An investigation on room temperature synthesis of vertically oriented arrays of iron oxide nanotubes by anodization of iron. Electrochim. Acta 2010, 55, 785–793. [Google Scholar] [CrossRef]
- Albu, S.P.; Ghicov, A.; Schmuki, P. High aspect ratio, self-ordered iron oxide nanopores formed by anodization of Fe in ethylene glycol/NH4F electrolytes. Phys. Status Solidi RRL 2009, 3, 64–66. [Google Scholar] [CrossRef]
- Lucas-Granados, B.; Sánchez-Tovar, R.; Fernández-Domene, R.M.; García-Antón, J. Controlled hydrodynamic conditions on the formation of iron oxide nanostructures synthesized by electrochemical anodization: Effect of the electrode rotation speed. Appl. Surf. Sci. 2017, 392, 503–513. [Google Scholar] [CrossRef]
- Lee, C.Y.; Wang, L.; Kado, Y.; Killian, M.S.; Schmuki, P. Anodic nanotubular/porous hematite photoanode for solar water splitting: substantial effect of iron substrate purity. ChemSusChem 2014, 7, 934–940. [Google Scholar] [CrossRef]
- Wang, L.; Lee, C.Y.; Schmuki, P. Improved photoelectrochemical water splitting of hematite nanorods thermally grown on Fe-Ti alloys. Electrochem. Commun. 2014, 44, 49–53. [Google Scholar] [CrossRef]
- Hiralal, P.; Unalan, H.E.; Wijayantha, K.G.U.; Kursumovic, A.; Jefferson, D.; MacManus-Driscoll, J.L.; Amaratunga, G.A.J. Growth and process conditions of aligned and patternable films of iron (III) oxide nanowires by thermal oxidation of iron. Nanotechnology 2008, 19, 1–7. [Google Scholar] [CrossRef]
- Li, M.; Deng, J.; Pu, A.; Zhang, P.; Zhang, H.; Gao, J.; Hao, Y.; Zhong, J.; Sun, X. Hydrogen-treated hematite nanostructures with low onset potential for highly efficient solar water oxidation. J. Mater. Chem. A 2014, 2, 6727–6733. [Google Scholar] [CrossRef]
- Yilmaz, C.; Unal, U. Single step synthesis of (α-Fe2O3) hematite films by hydrothermal electrochemical deposition. RSC Adv. 2015, 5, 16082–16088. [Google Scholar] [CrossRef]
- Annamalai, A.; Shinde, P.S.; Jeon, T.H.; Lee, H.H.; Kim, H.G.; Choi, W.; Jang, J.S. Fabrication of superior α-Fe2O3 nanorod photoanodes through ex-situ Sn-doping for solar water splitting. Sol. Energy Mater. Sol. Cells 2016, 144, 247–255. [Google Scholar] [CrossRef]
- Bohn, C.D.; Agrawal, A.K.; Walter, E.C.; Vaudin, M.D.; Herzing, A.A.; Haney, P.M.; Talin, A.A.; Szalai, V.A. Effect of Tin Doping on α-Fe2O3 Photoanodes for Water Splitting. J. Phys. Chem. C 2012, 116, 15290–15296. [Google Scholar] [CrossRef]
- Fu, Z.; Jiang, T.; Zhang, L.; Liu, B.; Wang, D.; Wang, L.; Xie, T. Surface treatment with Al3+ on a Ti-doped α-Fe2O3 nanorod array photoanode for efficient photoelectrochemical water splitting. J. Mater. Chem. A 2014, 2, 13705–13712. [Google Scholar] [CrossRef]
- Ahn, H.J.; Goswami, A.; Riboni, F.; Kment, S.; Naldoni, A.; Mohajernia, S.; Zboril, R.; Schmuki, P. Hematite photoanode with complex nanoarchitecture providing tunable gradient doping and low onset potential for photoelectrochemical water splitting. ChemSusChem 2018, 11, 1873–1879. [Google Scholar] [CrossRef]
- Cho, I.S.; Han, H.S.; Logar, M.; Park, J.; Zheng, X. Enhancing Low-Bias performance of hematite photoanodes for solar water splitting by simultaneous reduction of Bulk, interface, and surface recombination pathways. Adv. Energy Mater. 2016, 6, 1–9. [Google Scholar] [CrossRef]
- Cowan, A.J.; Barnett, C.J.; Pendlebury, S.R.; Barroso, M.; Sivula, K.; Grätzel, M.; Durrant, J.R.; Klug, D.R. Activation energies for the rate-limiting step in water photooxidation by nanostructured α-Fe2O3 and TiO2. J. Am. Chem. Soc. 2013, 133, 10134–10140. [Google Scholar] [CrossRef] [PubMed]
- Steier, L.; Herraiz-Cardona, I.; Gimenez, S.; Fabregat-Santiago, F.; Bisquert, J.; Tilley, S.D.; Grätzel, M. Understanding the role of underlayers and overlayers in thin film hematite photoanodes. Adv. Funct. Mater. 2014, 24, 7681–7688. [Google Scholar] [CrossRef]
- Cao, D.; Luo, W.; Feng, J.; Zhao, X.; Li, Z.; Zou, Z. Cathodic shift of onset potential for water oxidation on a Ti4+ doped Fe2O3 photoanode by suppressing the back reaction. Energy Environ. Sci. 2014, 7, 752–759. [Google Scholar] [CrossRef]
- Hisatomi, T.; Dotan, H.; Stefik, M.; Sivula, K.; Rothschild, A.; Grätzel, M.; Mathews, N. Enhancement in the performance of ultrathin hematite photoanode for water splitting by an oxide underlayer. Adv. Mater. 2012, 24, 2699–2702. [Google Scholar] [CrossRef] [PubMed]
- Abel, A.J.; Garcia-Torregrosa, I.; Patel, A.M.; Opasanont, B.; Baxter, J.B. SILAR-deposited hematite films for photoelectrochemical water splitting: Effects of Sn, Ti, thickness, and nanostructuring. J. Phys. Chem. C 2015, 119, 4454–4465. [Google Scholar] [CrossRef]
- Bouhjar, F.; Bessaïs, B.; Marí, B. Ultrathin-layer α-Fe2O3 deposited under hematite for solar water splitting. J. Solid State Electrochem. 2018, 22, 2347–2356. [Google Scholar] [CrossRef]
- Liang, Y.; Enache, C.S.; van de Krol, R. Photoelectrochemical characterization of sprayed 𝛼-Fe2O3 thin films: Influence of Si doping and SnO2 interfacial layer. Int. J. Photoenergy 2008, 2008, 739864. [Google Scholar] [CrossRef]
- Hisatomi, T.; Brillet, J.; Cornuz, M.; Le Formal, F.; Tétreault, N.; Sivula, K.; Grätzel, M. A Ga2O3 underlayer as an isomorphic template for ultrathin hematite films toward efficient photoelectrochemical water splitting. Faraday Discuss. 2012, 155, 223–232. [Google Scholar] [CrossRef]
- Schmuki, P.; Büchler, M.; Virtanen, S.; Isaacs, H.S.; Ryan, M.P.; Böhni, H. Passivity of iron in alkaline solutions studied by in situ XANES and a laser reflection technique. J. Electrochem. Soc. 1999, 146, 2097–2102. [Google Scholar] [CrossRef]
- Díaz, B.; Freire, L.; Montemor, M.F.; Nóvoa, X.R. Oxide film growth by CSV on AISI 316L: A combined electrochemical and analytical characterization. J. Braz. Chem. Soc. 2013, 24, 1246–1258. [Google Scholar] [CrossRef]
- Amaral, S.T.; Martini, E.M.A.; Müller, I.L. An attempt of experimental separation of the potentiodynamic anodic peaks of iron in alkaline solutions and application of the ohmic model for passive film growth. Corros. Sci. 2001, 43, 853–879. [Google Scholar] [CrossRef]
- Büchler, M. Alternating current corrosion of cathodically protected pipelines: Discussion of the involved processes and their consequences on the critical interference values. Mater. Corros. 2012, 63, 1181–1187. [Google Scholar] [CrossRef]
- Duschek, K.; Uhlemann, M.; Schlörb, H.; Nielsch, K.; Leistner, K. Electrochemical and in situ magnetic study of iron/iron oxide films oxidized and reduced in KOH solution for magneto-ionic switching. Electrochem. Commun. 2016, 72, 153–156. [Google Scholar] [CrossRef]
- Xi, L.; Tran, P.D.; Chiam, S.Y.; Bassi, P.S.; Mak, W.F.; Mulmudi, H.K.; Batabyal, S.K.; Barber, J.; Loo, J.S.C.; Wong, L.H. Co3O4-decorated hematite nanorods as an effective photoanode for solar water oxidation. J. Phys. Chem. C 2012, 116, 13884–13889. [Google Scholar] [CrossRef]
- Ling, Y.; Wang, G.; Wheeler, D.A.; Zhang, J.Z.; Li, Y. Sn-doped hematite nanostructures for photoelectrochemical water splitting. Nano Lett. 2011, 11, 2119–2125. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.H.; Guo, W.R.; Mishra, M.; Huang, Y.C.; Chang, J.K.; Lee, T.C. Combinatorial studies on wet-chemical synthesized Ti-doped α-Fe2O3: How does Ti4+ improve photoelectrochemical activity? ACS Appl. Nano Mater. 2018, 1, 3145–3154. [Google Scholar] [CrossRef]
- Annamalai, A.; Sandström, R.; Gracia-Espino, E.; Boulanger, N.; Boily, J.F.; Mühlbacher, I.; Shchukarev, A.; Wågberg, T. Influence of Sb5+ as a double donor on hematite (Fe3+) photoanodes for surface-enhanced photoelectrochemical water oxidation. ACS Appl. Mater. Interfaces 2018, 10, 16467–16473. [Google Scholar] [CrossRef] [PubMed]
- Qiu, P.; Yang, H.; Yang, L.; Wang, Q.; Ge, L. Solar water splitting with nanostructured hematite: The role of annealing-temperature. Electrochim. Acta 2018, 266, 431–440. [Google Scholar] [CrossRef]
- McDonald, K.J.; Choi, K.S. Photodeposition of co-based oxygen evolution catalysts on α-Fe2O3 photoanodes. Chem. Mater. 2011, 23, 1686–1693. [Google Scholar] [CrossRef]
- Tang, D.; Han, Y.; Ji, W.; Qiao, S.; Zhou, X.; Liu, R.; Han, X.; Huang, H.; Liu, Y.; Kang, Z. A high-performance reduced graphene oxide/ZnCo layered double hydroxide electrocatalyst for efficient water oxidation. Dalton Trans. 2014, 43, 15119–15125. [Google Scholar] [CrossRef] [PubMed]
- Segev, G.; Dotan, H.; Malviya, K.D.; Kay, A.; Mayer, M.T.; Grätzel, M.; Rothschild, A. High solar flux concentration water splitting with hematite (α-Fe2O3) photoanodes. Adv. Energy Mater. 2016, 6, 1–7. [Google Scholar] [CrossRef]
- Li, M.; Yang, Y.; Ling, Y.; Qiu, W.; Wang, F.; Liu, T.; Song, Y.; Liu, X.; Fang, P.; Tong, Y.; et al. Morphology and doping engineering of Sn-doped hematite nanowire photoanodes. Nano Lett. 2017, 17, 2490–2495. [Google Scholar] [CrossRef]
- Guo, X.; Wang, L.; Tan, Y. Hematite nanorods Co-doped with Ru cations with different valence states as high performance photoanodes for water splitting. Nano Energy 2015, 16, 320–328. [Google Scholar] [CrossRef]
- Tamirat, A.G.; Su, W.N.; Dubale, A.A.; Chen, H.M.; Hwang, B.J. Photoelectrochemical water splitting at low applied potential using a NiOOH coated codoped (Sn, Zr) α-Fe2O3 photoanode. J. Mater. Chem. A 2015, 3, 5949–5961. [Google Scholar] [CrossRef]
- Wang, L.; Zhou, X.; Nguyen, N.T.; Schmuki, P. Plasmon-Enhanced Photoelectrochemical Water Splitting Using Au Nanoparticles Decorated on Hematite Nanoflake Arrays. ChemSusChem 2015, 8, 618–622. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Zhang, X.T.; Sun, P.P.; Lu, S.; Wang, L.L.; Wei, Y.A.; Liu, Y.C. Enhanced photoelectrochemical water splitting on hematite thin film with layer-by-layer deposited ultrathin TiO2 underlayer. Int. J. Hydrogen Energy 2014, 39, 16212–16219. [Google Scholar] [CrossRef]
- Deng, J.; Zhang, Q.; Feng, K.; Lan, H.; Zhong, J.; Chaker, M.; Ma, D. Efficient Photoelectrochemical Water Oxidation on Hematite with Fluorine-Doped FeOOH and FeNiOOH as Dual Cocatalysts. ChemSusChem 2018, 11, 3783–3789. [Google Scholar] [CrossRef] [PubMed]
- Qin, D.D.; Tao, C.L.; In, S.I.; Yang, Z.Y.; Mallouk, T.E.; Bao, N.; Grimes, C.A. Facile solvothermal method for fabricating arrays of vertically oriented α-Fe2O3 nanowires and their application in photoelectrochemical water oxidation. Energy Fuels 2011, 25, 5257–5263. [Google Scholar] [CrossRef]
- Yang, T.Y.; Kang, H.Y.; Jin, K.; Park, S.; Lee, J.H.; Sim, U.; Jeong, H.Y.; Joo, Y.C.; Nam, K.T. An iron oxide photoanode with hierarchical nanostructure for efficient water oxidation. J. Mater. Chem. A 2014, 2, 2297–2305. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poornajar, M.; Nguyen, N.T.; Ahn, H.-J.; Büchler, M.; Liu, N.; Kment, S.; Zboril, R.; Yoo, J.E.; Schmuki, P. Fe2O3 Blocking Layer Produced by Cyclic Voltammetry Leads to Improved Photoelectrochemical Performance of Hematite Nanorods. Surfaces 2019, 2, 131-144. https://doi.org/10.3390/surfaces2010011
Poornajar M, Nguyen NT, Ahn H-J, Büchler M, Liu N, Kment S, Zboril R, Yoo JE, Schmuki P. Fe2O3 Blocking Layer Produced by Cyclic Voltammetry Leads to Improved Photoelectrochemical Performance of Hematite Nanorods. Surfaces. 2019; 2(1):131-144. https://doi.org/10.3390/surfaces2010011
Chicago/Turabian StylePoornajar, Mahshid, Nhat Truong Nguyen, Hyo-Jin Ahn, Markus Büchler, Ning Liu, Stepan Kment, Radek Zboril, Jeong Eun Yoo, and Patrik Schmuki. 2019. "Fe2O3 Blocking Layer Produced by Cyclic Voltammetry Leads to Improved Photoelectrochemical Performance of Hematite Nanorods" Surfaces 2, no. 1: 131-144. https://doi.org/10.3390/surfaces2010011
APA StylePoornajar, M., Nguyen, N. T., Ahn, H.-J., Büchler, M., Liu, N., Kment, S., Zboril, R., Yoo, J. E., & Schmuki, P. (2019). Fe2O3 Blocking Layer Produced by Cyclic Voltammetry Leads to Improved Photoelectrochemical Performance of Hematite Nanorods. Surfaces, 2(1), 131-144. https://doi.org/10.3390/surfaces2010011





