Organic Remains in Early Christian Egyptian Metal Vessels: Investigation with Fourier Transform Infrared Spectroscopy and Gas Chromatography–Mass Spectrometry
Abstract
1. Introduction
2. Experimental Procedures
2.1. Fourier Transform Infrared Spectroscopy
2.2. FTIR Analysis of Powder Samples in KBr Discs
2.3. Solvent Extraction for FTIR Analysis
2.4. Gas Chromatography–Mass Spectrometry
2.4.1. Sample Preparation for GC-MS
2.4.2. GC-MS Analysis
3. Results and Discussion
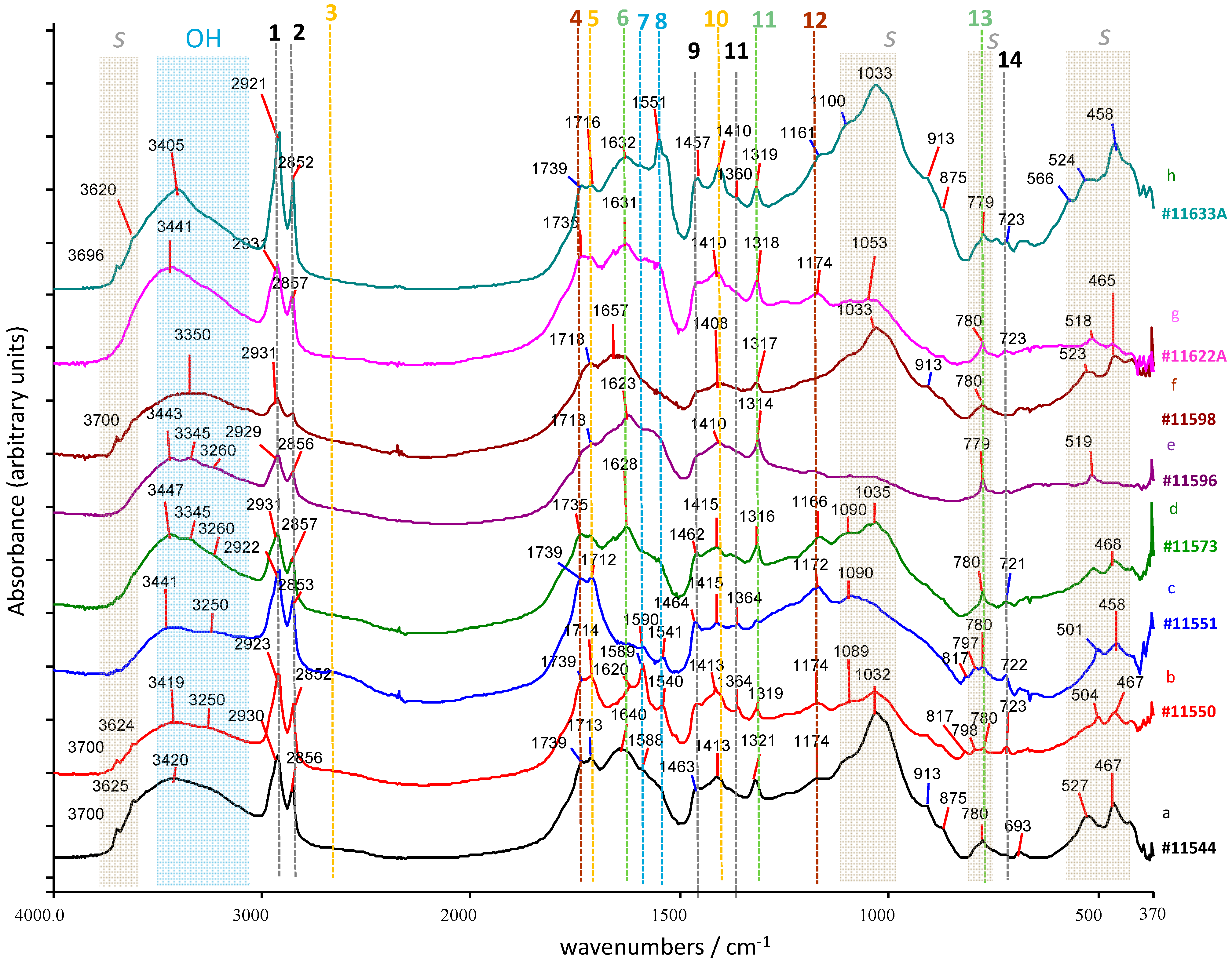
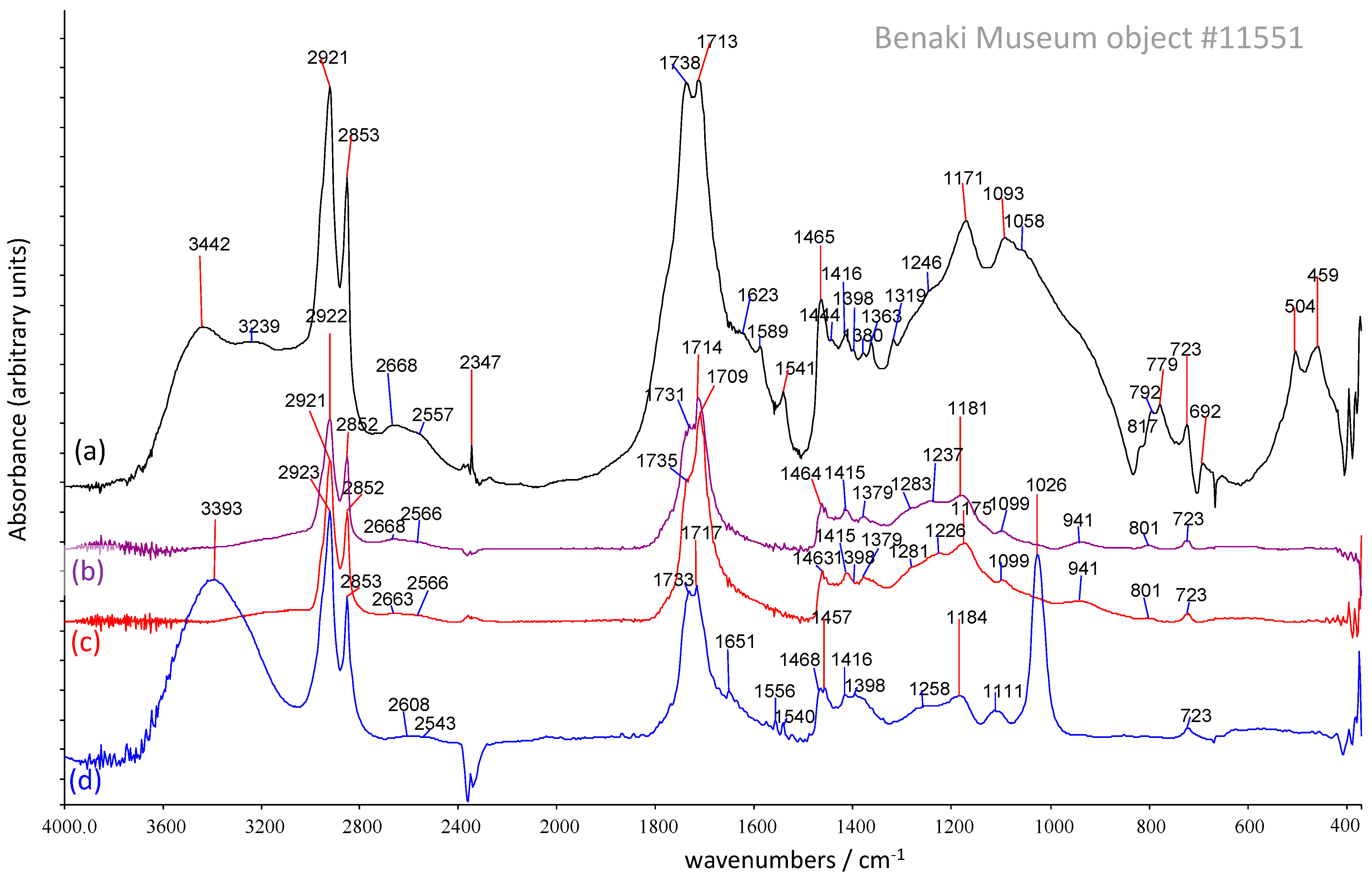
3.1. Fourier Transform Infrared Spectroscopy
3.1.1. Powder Samples
3.1.2. Solvent-Extracted FTIR Results
3.2. Solvent-Extraction-μFTIR
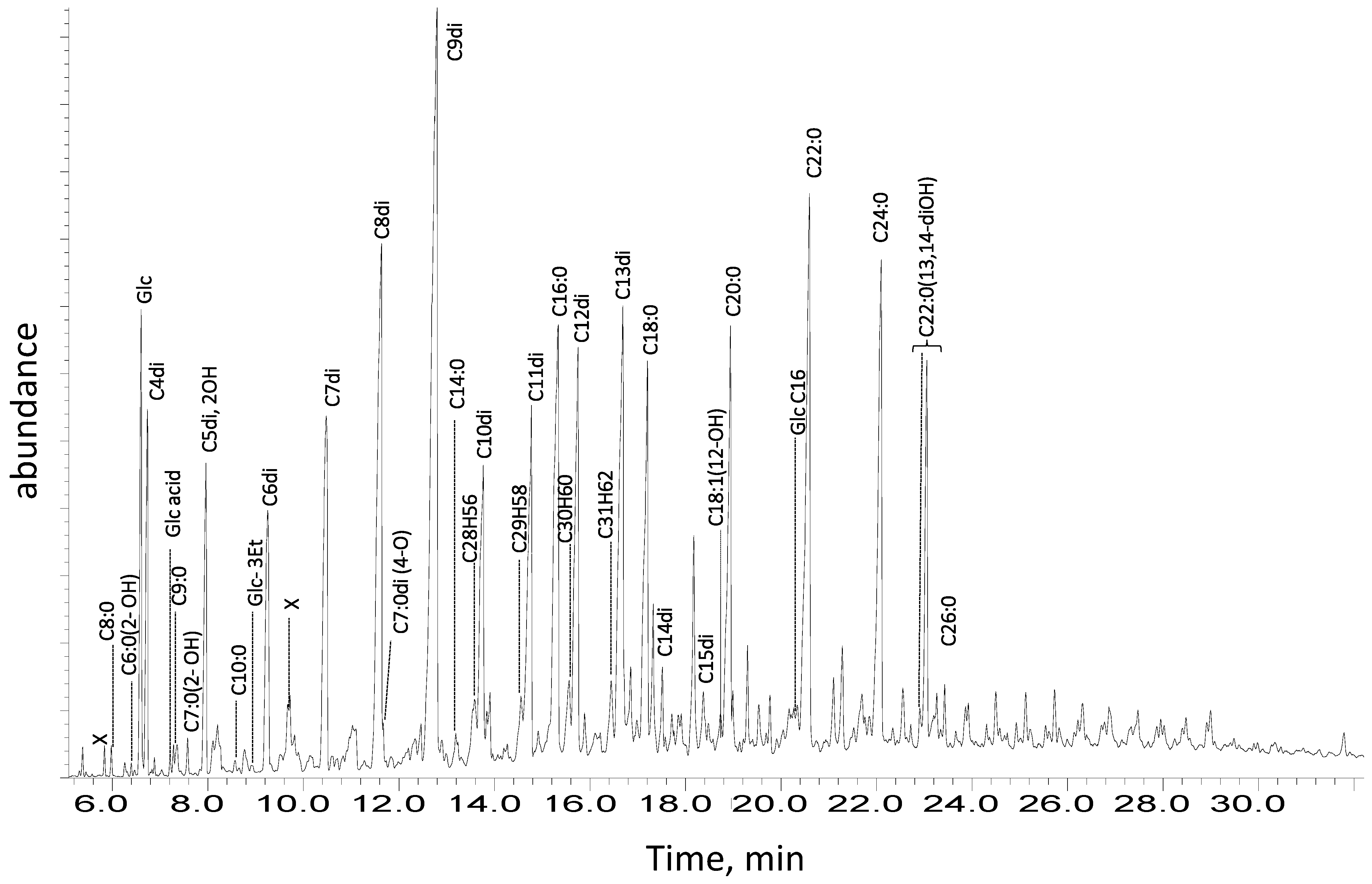
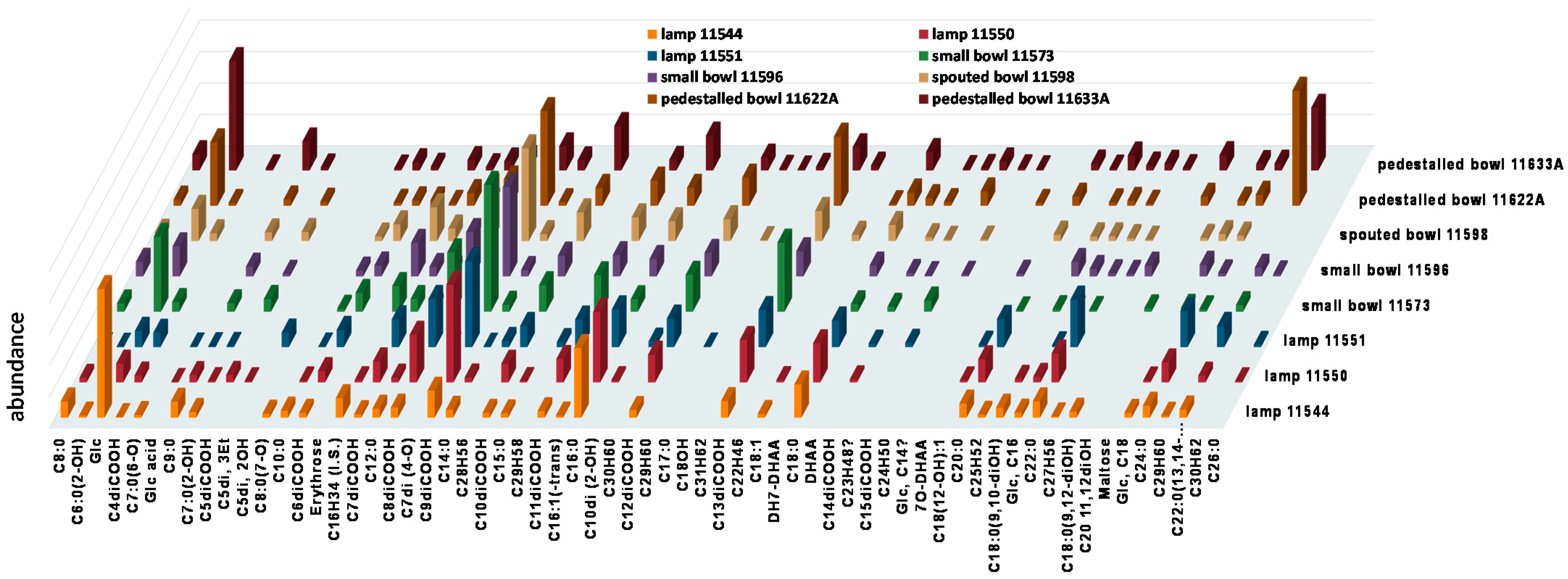
3.3. Gas Chromatography–Mass Spectrometry
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MAG | Mono-fatty Acyl Glycerols |
| DAG | Di-fatty Acyl Glycerols |
| TAG | Tri-fatty Acyl Glycerols |
| DiFA | diacids (dicarboxylic acids) |
| FA | Fatty Acids |
| FAMS | Fatty Acids Metal Salts |
| FTIR | Fourier Transform Infra-Red spectroscopy |
| GC-MS | gas chromatography–mass spectrometry |
| SE-FTIR | Solvent-Extracted FTIR |
| SE-μFTIR | Solvent-Extracted microscopy-FTIR |
References
- Evershed, R.P. Experimental approaches to the interpretation of absorbed organic residues in archaeological ceramics. World Archaeol. 2008, 40, 26–47. [Google Scholar] [CrossRef]
- Roffet-Salque, M.; Dunne, J.; Altoft, D.T.; Casanova, E.; Cramp, L.J.; Smyth, J.; Whelton, H.L.; Evershed, R.P. From the inside out: Upscaling organic residue analyses of archaeological ceramics. J. Archaeol. Sci. Rep. 2017, 16, 627–640. [Google Scholar] [CrossRef]
- Lambert, J.B. Archaeological chemistry. Acc. Chem. Res. 2002, 35, 583–584. [Google Scholar] [CrossRef]
- Orna, M.V.; Lambert, J.B. New directions in archaeological chemistry. In Archaeological Chemistry Organic, Inorganic, and Biochemical Analysis; American Chemical Society: Washington, DC, USA, 1996; pp. 1–9. [Google Scholar] [CrossRef]
- Pollard, A.M.; Batt, C.M.; Stern, B.; Young, S.M.M. Analytical Chemistry in Archaeology (Cambridge Manuals in Archaeology); Cambridge University Press: Cambridge, UK; New York, NY, USA; Melbourne, Australia; Madrid, Spain; Cape Town, South Africa; Singapore; São Paulo, Brazil, 2007. [Google Scholar]
- Evershed, R.P. Organic Residue Analysis in Archaeology: The Archaeological Biomarker Revolution. Archaeometry 2008, 50, 895–924. [Google Scholar] [CrossRef]
- Evershed, R.P. Biomolecular archaeology and lipids. World Archaeol. 1993, 25, 74–93. [Google Scholar] [CrossRef] [PubMed]
- Colombini, M.P.; Modugno, F.; Ribechini, E. Organic mass spectrometry in archaeology: Evidence for Brassicaceae seed oil in Egyptian ceramic lamps. J. Mass Spectrom. 2005, 40, 890–898. [Google Scholar] [CrossRef]
- Pellegrini, D.; Duce, C.; Bonaduce, I.; Biagi, S.; Ghezzi, L.; Colombini, M.P.; Tinè, M.R.; Bramanti, E. Fourier transform infrared spectroscopic study of rabbit glue/inorganic pigments mixtures in fresh and aged reference paint reconstructions. Microchem. J. 2016, 124, 31–35. [Google Scholar] [CrossRef]
- Degano, I.; La Nasa, J.; Ghelardi, E.; Modugno, F.; Colombini, M.P. Model study of modern oil-based paint media by triacylglycerol profiling in positive and negative ionization modes. Talanta 2016, 161, 62–70. [Google Scholar] [CrossRef]
- Garnier, N.; Rolando, C.; Høtje, J.M.; Tokarski, C. Analysis of archaeological triacylglycerols by high resolution nanoESI, FT-ICR MS and IRMPD MS/MS: Application to 5th century BC–4th century AD oil lamps from Olbia (Ukraine). Int. J. Mass Spectrom. 2009, 284, 47–56. [Google Scholar] [CrossRef]
- Gregg, M.W.; Slater, G.F. A new method for extraction, isolation and transesterification of free fatty acids from archaeological pottery. Archaeometry 2010, 52, 833–854. [Google Scholar] [CrossRef]
- Regert, M.; Garnier, N.; Decavallas, O.; Cren-Oliv, C.C.; Rolando, C. Structural characterization of lipid constituents from natural substances preserved in archaeological environments. Meas. Sci. Technol. 2003, 14, 1620–1630. [Google Scholar] [CrossRef]
- Ioakimoglou, E.; Boyatzis, S.; Argitis, P.; Fostiridou, A.; Papapanagiotou, K.; Yannovits, N. Thin-Film Study on the Oxidation of Linseed Oil in the Presence of Selected Copper Pigments. Chem. Mater. 1999, 11, 2013–2022. [Google Scholar] [CrossRef]
- Frankel, E.N. Lipid Oxidation, 2nd ed.; Lipid Library Series; Woodhead Publishing/Oily Press: Cambridge, UK, 2005. [Google Scholar]
- Colombini, M.P.; Modugno, F. Organic Mass Spectrometry in Art and Archaeology, 1st ed.; John Wiley & Sons, Ltd.: Chichester, UK, 2009. [Google Scholar]
- Andreotti, A.; Bonaduce, I.; Colombini, M.P.; Gautier, G.; Modugno, A.F.; Ribechini, E. Combined GC/MS analytical procedure for the characterization of glycerolipid, waxy, resinous, and proteinaceous materials in a unique paint microsample. Anal. Chem. 2006, 78, 4490–4500. [Google Scholar] [CrossRef] [PubMed]
- Stern, B.; Heron, C.; Tellefsen, T.; Serpico, M. New investigations into the Uluburun resin cargo. J. Archaeol. Sci. 2008, 35, 2188–2203. [Google Scholar] [CrossRef]
- Stern, B.; Heron, C.; Serpico, M.; Bourriau, J. A comparison of methods for establishing fatty acid concentration gradients across potsherds: A case study using Late Bronze Age Canaanite amphorae. Archaeometry 2000, 42, 399–414. [Google Scholar] [CrossRef]
- Pitonzo, R.; Armetta, F.; Saladino, M.L.; Oliveri, F.; Tusa, S.; Caponetti, E. Application of Gas Chromatography coupled with Mass Spectroscopy (GC/MS) to the analysis of archaeological ceramic amphorae belonging to the Carthaginian fleet that was defeated in the Egadi battle (241 B.C.). Acta IMEKO 2017, 6, 67–70. [Google Scholar] [CrossRef][Green Version]
- Blanco-Zubiaguirre, L.; Ribechini, E.; Degano, I.; La Nasa, J.; Carrero, J.A.; Iñañez, J.; Olivares, M.; Castro, K. GC–MS and HPLC-ESI-QToF characterization of organic lipid residues from ceramic vessels used by Basque whalers from 16th to 17th centuries. Microchem. J. 2018, 137, 190–203. [Google Scholar] [CrossRef]
- Craig, O.E.; Saul, H.; Spiteri, C. Residue Analysis. In Archaeological Science; Richards, M., Britton, K., Eds.; Cambridge University Press: Cambridge, UK, 2019; pp. 70–98. [Google Scholar]
- Evershed, R.P.; Berstan, R.; Grew, F.; Copley, M.S.; Charmant, A.J.H.; Barham, E.; Mottram, H.R.; Brown, G. Formulation of a Roman cosmetic. Nature 2004, 432, 35–36. [Google Scholar] [CrossRef] [PubMed]
- Frith, J.; Appleby, R.; Stacey, R.; Heron, C. Sweetness and Light: Chemical Evidence of Beeswax and Tallow Candles at Fountains Abbey, North Yorkshire. Mediev. Archaeol. 2004, 48, 220–227. [Google Scholar] [CrossRef]
- RRibechini, E.; Modugno, F.; Baraldi, C.; Baraldi, P.; Colombini, M.P. An integrated analytical approach for characterizing an organic residue from an archaeological glass bottle recovered in Pompeii (Naples, Italy). Talanta 2008, 74, 555–561. [Google Scholar] [CrossRef]
- Griffiths, P.R.; Haseth, J.A. Fourier Transform Infrared Spectrometry, 2nd ed.; Wiley-Interscience: Hoboken, NJ, USA, 2007. [Google Scholar]
- Nakamoto, K. Infrared and Raman Spectra of Inorganic and Coordination Compounds, Part A: Theory and Applications in Inorganic Chemistry; Wiley: Hoboken, NJ, USA, 1978. [Google Scholar]
- Mayo, D.; Miller, F.; Hannah, R. Course Notes on the Interpretation of Infrared and Raman Spectra; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2004. [Google Scholar]
- Derrick, M.R.; Stulik, D.; Landry, J.M. Infrared Spectroscopy in Conservation Science; Getty Conservation Institute: Los Angeles, CA, USA, 1999. [Google Scholar]
- Stuart, B.H. Infrared Spectroscopy: Fundamentals and Applications; John Wiley & Sons, Ltd.: Chichester, UK, 2004. [Google Scholar]
- Shillito, L.M.; Almond, M.J.; Wicks, K.; Marshall, L.-J.R.; Matthews, W. The use of FT-IR as a screening technique for organic residue analysis of archaeological samples. Spectrochim Acta Part A Mol. Biomol. Spectrosc. 2009, 72, 120–125. [Google Scholar] [CrossRef] [PubMed]
- Thickett, D.; Pretzel, B. FTIR surface analysis for conservation. Herit. Sci. 2020, 8, 5. [Google Scholar] [CrossRef]
- Bell, J.; Nel, P.; Stuart, B. Non-invasive identification of polymers in cultural heritage collections: Evaluation, optimisation and application of portable FTIR (ATR and external reflectance) spectroscopy to three-dimensional polymer-based objects. Herit. Sci. 2019, 7, 95. [Google Scholar] [CrossRef]
- Saviello, D.; Toniolo, L.; Goidanich, S.; Casadio, F. Non-invasive identification of plastic materials in museum collections with portable FTIR reflectance spectroscopy: Reference database and practical applications. Microchem. J. 2016, 124, 868–877. [Google Scholar] [CrossRef]
- Invernizzi, C.; Rovetta, T.; Licchelli, M.; Malagodi, M. Mid and near-infrared reflection spectral database of natural organic materials in the cultural heritage field. Int. J. Anal. Chem. 2018, 2018, 7823248. [Google Scholar] [CrossRef]
- Tasiouli, N.; Boyatzis, S.; Karatzani, A.; Karydas, A.G. Study and Conservation of a 19th Century Printed Silk Scarf from the Collection of the National Historical Museum of Greece. Archaeology 2021, 9, 56–67. [Google Scholar] [CrossRef]
- Sommer, A.J. Mid-Infrared Transmission Microspectroscopy. In Handbook of Vibrational Spectroscopy; Griffiths, P.R., Ed.; John Wiley & Sons, Ltd.: Chichester, UK, 2006. [Google Scholar]
- Pilc, J.; White, R. The Application of FTIR-Microscopy to the Analysis of Paint Binders in Easel Paintings. Natl. Gallery Tech. Bull. 1995, 16, 73–84. [Google Scholar]
- Rizzo, A. Progress in the application of ATR-FTIR microscopy to the study of multi-layered cross-sections from works of art. Anal. Bioanal. Chem. 2008, 392, 47–55. [Google Scholar] [CrossRef]
- Vetter, W.; Schreiner, M. Characterization of pigment-binding media systems: Comparison of non-invasive in-situ reflection FTIR with transmission FTIR microscopy. e-Preserv. Sci. 2011, 8, 10–22. [Google Scholar]
- Rosi, F.; Legan, L.; Miliani, C.; Ropret, P. Micro transflection on a metallic stick: An innovative approach of reflection infrared spectroscopy for minimally invasive investigation of painting varnishes. Anal. Bioanal. Chem. 2017, 409, 3187–3197. [Google Scholar] [CrossRef]
- Ajò, D.; Casellato, U.; Fiorin, E.; Vigato, P. Ciro Ferri’s frescoes: A study of painting materials and technique by SEM-EDS microscopy, X-ray diffraction, micro FT-IR and photoluminescence spectroscopy. J. Cult. Herit. 2004, 5, 333–348. [Google Scholar] [CrossRef]
- Sciutto, G.; Oliveri, P.; Prati, S.; Catelli, E.; Bonacini, I.; Mazzeo, R. A Multivariate Methodological Workflow for the Analysis of FTIR Chemical Mapping Applied on Historic Paint Stratigraphies. Int. J. Anal. Chem. 2017, 2017, 4938145. [Google Scholar] [CrossRef] [PubMed]
- La Russa, M.F.; Ruffolo, S.A.; Crisci, G.M.; Barone, G.; Mazzoleni, P.; Pezzino, A. The Use of FTIR and Micro-FTIR Spectroscopy: An Example of Application to Cultural Heritage. Int. J. Spectrosc. 2009, 2009, 893528. [Google Scholar] [CrossRef]
- Kotzamani, D.; Phoca, A.; Karydi, G.; Zacharia, M.; Kantarelou, V.; Karatasios, J.; Boyatzis, S.C.; Perdikatsis, V. The Metallurgical Investigation of Copper-Alloys Metalwork of The Benaki Museum Dated in the 4–7th Centuries A.D. In Proceedings of the International Symposium on History, Technology and Conservation of Ancient Metal, Glasses and Enamels, Athens, Greece, 16–19 November 2011. [Google Scholar]
- Boyatzis, S.C.; Kotzamani, D.; Phoca, A.; Karydi, G.; Zacharia, M.; Kantarelou, V. Characterization of organic remains found in copper alloy vessels of the Benaki museum collection with Fourrier-transform infrared spectroscopy. In Proceedings of the 3rd ARCH_RNT, Archaeological Research and New Technologies, Kalamata, Greece, 3–5 October 2012; Zacharias, N., Ed.; University of the Peloponnese: Tripoli, Greece, 2014; pp. 95–102. [Google Scholar]
- Drandaki, A. Late Antique Metalware, Bibliothèque de L’Antiquité, Tardive; Brepole Publishers: Turnhout, Belgium, 2020. [Google Scholar]
- Arya, R.K. Drying induced phase separation in multicomponent polymeric coatings—Simulation study. Int. J. Sci. Technol. Res. 2012, 1, 48–52. [Google Scholar]
- Mitchell, D.S. Solvent Extraction Process; OSTI: Oak Ridge, TN, USA, 1980.
- THolcombe, T.C.; Bress, D.F.; Casparian, R.E. Process for Drying and Solvent-Extraction of Solids and Sludges. International Patent WO1992022367A1, 23 December 1992. [Google Scholar]
- Orata, F. Derivatization reactions and reagents for gas chromatography analysis. In Advanced Gas Chromatography—Progress in Agricultural, Biomedical and Industrial Applications; InTech: Rijeka, Croatia, 2012; pp. 83–156. [Google Scholar] [CrossRef]
- Husek, P. Derivatization. In Encyclopedia of Separation Science; Academic Press: Cambridge, MA, USA, 2000; pp. 434–443. [Google Scholar]
- Colombini, M.P.; Modugno, F.; Giacomelli, M.; Francesconi, S. Characterisation of proteinaceous binders and drying oils in wall painting samples by gas chromatography-mass spectrometry. J. Chromatogr. A 1999, 846, 113–124. [Google Scholar] [CrossRef]
- Weiner, S. Microarchaeology, 1st ed.; Cambridge University Press: Cambridge, UK, 2010. [Google Scholar]
- Nevin, A.; Melia, J.L.; Osticioli, I.; Gautier, G.; Colombini, M.P. The identification of copper oxalates in a 16th century Cypriot exterior wall painting using micro FTIR, micro Raman spectroscopy and Gas Chromatography-Mass Spectrometry. J. Cult. Herit. 2008, 9, 154–161. [Google Scholar] [CrossRef]
- Purvis, O.W. The Occurrence of Copper Oxalate in Lichens Growing on Copper Sulphide-Bearing Rocks in Scandinavia. Lichenologist 1984, 16, 197–204. [Google Scholar] [CrossRef]
- Bellamy, L.J. The Infra-Red Spectra of Complex Molecules; Springer: Dordrecht, The Netherlands, 1975. [Google Scholar]
- Larkin, P. Infrared and Raman Spectroscopy: Principles and Spectral Interpretation; Elsevier: Amsterdam, The Netherlands, 2011. [Google Scholar]
- Coates, J. Interpretation of Infrared Spectra, A Practical Approach. In Encyclopedia of Analytical Chemistry; John Wiley & Sons, Ltd.: Chichester, UK, 2006; pp. 10815–10837. [Google Scholar]
- Lambert, J.B.; Shurvell, H.F.; Cooks, R.G. Introduction to Organic Spectroscopy, 1st ed.; Macmillan: New York, NY, USA, 1987. [Google Scholar]
- Nakamoto, K. Infrared and Raman Spectra of Inorganic and Coordination Compounds, Part B, Applications in Coordination, Organometallic, and Bioinorganic Chemistry, 6th ed.; John Wiley & Sons: Hoboken, NJ, USA, 2009. [Google Scholar]
- Baeten, J.; Romanus, K.; Degryse, P.; De Clercq, W.; Poelman, H.; Verbeke, K.; Luypaerts, A.; Walton, M.; Jacobs, P.; De Vos, D.; et al. Application of a multi-analytical toolset to a 16th century ointment: Identification as lead plaster mixed with beeswax. Microchem. J. 2010, 95, 227–234. [Google Scholar] [CrossRef]
- Hammann, S.; Scurr, D.J.; Alexander, M.R.; Cramp, L.J.E. Mechanisms of lipid preservation in archaeological clay ceramics revealed by mass spectrometry imaging. Proc. Natl. Acad. Sci. USA 2020, 117, 14688–14693. [Google Scholar] [CrossRef]
- Anna Filopoulou Vlachou, S.; Boyatzis, S.C. Fatty Acids and Their Metal Salts: A Review of Their Infrared Spectra in Light of Their Presence in Cultural Heritage (accepted for publication). Molecules 2021, in press. [Google Scholar]
- Lynch, M.L.; Pan, A.Y.; Laughlin, R.G. Spectroscopic and Thermal Characterization of 1:2 Sodium Soap/Fatty Acid Acid—Soap Crystals. J. Phys. Chem. 1996, 100, 357–361. [Google Scholar] [CrossRef]
- Otero, V.; Sanches, D.; Montagner, C.; Vilarigues, M.; Carlyle, L.; Lopes, J.A.; Melo, M.J. Characterisation of metal carboxylates by Raman and infrared spectroscopy in works of art. J. Raman Spectrosc. 2014, 45, 1197–1206. [Google Scholar] [CrossRef]
- Baij, L.; Hermans, J.J.; Keune, K.; Iedema, P. Time-Dependent ATR-FTIR Spectroscopic Studies on Fatty Acid Diffusion and the Formation of Metal Soaps in Oil Paint Model Systems. Angew. Chem. Int. Ed. 2018, 57, 7351–7354. [Google Scholar] [CrossRef] [PubMed]
- Kazarian, S.; Chan, K. Applications of ATR-FTIR spectroscopic imaging to biomedical samples. Biochim. Biophys. Acta Biomembr. 2006, 1758, 858–867. [Google Scholar] [CrossRef] [PubMed]
- Hermans, J.J. Metal Soaps in Oil Paint: Structure, Mechanisms and Dynamics; University of Amsterdam: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Catalano, J.; Yao, Y.; Murphy, A.; Zumbulyadis, N.; Centeno, S.A.; Dybowski, C. Analysis of Lead Carboxylates and Lead-Containing Pigments in Oil Paintings by Solid- State Nuclear Magnetic Resonance. MRS Proc. 2014, 1656, 149–156. [Google Scholar] [CrossRef]
- Hermans, J.J.; Keune, K.; van Loon, A.; Iedema, P.D. The crystallization of metal soaps and fatty acids in oil paint model systems. Phys. Chem. Chem. Phys. 2016, 18, 10896–10905. [Google Scholar] [CrossRef] [PubMed]
- McGovern, P.E.; Hall, G.R. Charting a Future Course for Organic Residue Analysis in Archaeology. J. Archaeol. Method Theory 2015, 23, 592–622. [Google Scholar] [CrossRef]
- Chapman, D. Infrared spectroscopy of lipids. J. Am. Oil Chem. Soc. 1965, 42, 353–371. [Google Scholar] [CrossRef]
- Jackson, M.; Haris, P.I.; Chapman, D. Fourier transform infrared spectroscopic studies of lipids, polypeptides and proteins. J. Mol. Struct. 1989, 214, 329–355. [Google Scholar] [CrossRef]
- Chapman, D. Infrared spectra and the polymorphism of glycerides. Part II. 1:3-Diglycerides and saturated triglycerides. J. Chem. Soc. 1956, 2522–2528. [Google Scholar] [CrossRef]
- Boyatzis, S.C.; Douvas, A.M.; Argyropoulos, V.; Siatou, A.; Vlachopoulou, M. Characterization of a water-dispersible metal protective coating with fourier transform infrared spectroscopy, modulated differential scanning calorimetry, and ellipsometry. Appl. Spectrosc. 2012, 66, 580–590. [Google Scholar] [CrossRef]
- Jones, R.N. The Effects of Chain Length on the Infrared Spectra of Fatty Acids and Methyl Esters. Can. J. Chem. 1962, 40, 321–333. [Google Scholar] [CrossRef]
- Meiklejohn, R.A.; Meyer, R.J.; Aronovic, S.M.; Schuette, H.A.; Meloche, V.W. Characterization of Long-Chain Fatty Acids by Infrared Spectroscopy. Anal. Chem. 1957, 29, 329–334. [Google Scholar] [CrossRef]
- De Ruig, W.G. Infrared Spectra of Monoacid Triglycerides with Some Applications to Fat Analysis; Center for Agricultural Publishing and Documentation: Wageningen, The Netherlands, 1971. [Google Scholar]
- Tiwari, R.D.; Sharma, J.P.; Belcher, R. The Determination of Carboxylic Functional Groups: Monographs in Organic Chemistry; Elsevier: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Frankel, E.N. Lipid oxidation: Mechanisms, products and biological significance. J. Am. Oil Chem. Soc. 1984, 61, 1908–1917. [Google Scholar] [CrossRef]
- Corish, P.J.; Chapman, D. The infrared spectra of some monocarboxylic acids. J. Chem. Soc. 1957, 18, 1746–1751. [Google Scholar] [CrossRef]
- O’Connor, R.T.; DuPre, E.F.; Feuge, R.O. The infrared spectra of mono-, di-, and triglycerides. J. Am. Oil Chem. Soc. 1955, 32, 88–93. [Google Scholar] [CrossRef]
- Chapman, D. The Polymorphism of Glycerides. Chem. Rev. 1962, 62, 433–456. [Google Scholar] [CrossRef]
- Chapman, D. The 720 cm–1 band in the infrared spectra of crystalline long-chain compounds. J. Chem. Soc. 1957, 18, 4489–4491. [Google Scholar] [CrossRef]
- Shurvell, H.F. Spectra- Structure Correlations in the Mid- and Far-Infrared. In Handbook of Vibrational Spectroscopy; Chalmers, J.M., Ed.; John Wiley & Sons, Ltd.: Chichester, UK, 2006. [Google Scholar]
- Lewis, R.N.A.H.; McElhaney, R.N. Vibrational Spectroscopy of Lipids. In Handbook of Vibrational Spectroscopy; Griffiths, P.R., Ed.; John Wiley & Sons, Ltd.: Chichester, UK, 2006. [Google Scholar]
- Regert, M. Investigating the history of prehistoric glues by gas chromatography—Mass spectrometry Original Paper. J. Sep. Sci. 2004, 27, 244–254. [Google Scholar] [CrossRef]
- Boyatzis, S.; Ioakimoglou, E.; Argitis, P. UV exposure and temperature effects on curing mechanisms in thin linseed oil films: Spectroscopic and chromatographic studies. J. Appl. Polym. Sci. 2002, 84, 936–949. [Google Scholar] [CrossRef]
- Copley, M.S.; Bland, H.A.; Rose, P.; Horton, M.; Evershed, R.P. Gas chromatographic, mass spectrometric and stable carbon isotopic investigations of organic residues of plant oils and animal fats employed as illuminants in archaeological lamps from Egypt. Analyst 2005, 130, 860–871. [Google Scholar] [CrossRef]
- Romanus, K.; Van Neer, W.; Marinova, E.; Verbeke, K.; Luypaerts, A.; Accardo, S.; Hermans, I.; Jacobs, P.; De Vos, D.; Waelkens, M. Brassicaceae seed oil identified as illuminant in Nilotic shells from a first millennium AD Coptic church in Bawit, Egypt. Anal. Bioanal. Chem. 2007, 390, 783–793. [Google Scholar] [CrossRef] [PubMed]
- Lucas, A.; Harris, J.R. Ancient Egyptian Materials and Industries; Histories and Mysteries of Man, Ltd.: London, UK, 1962. [Google Scholar]
- Serpico, M.; White, R. Oil, fat and wax. In Ancient Egyptian Materials and Technology; Nicholson, P.T., Shaw, I., Eds.; Cambridge University Press: Cambridge, UK, 2000; pp. 390–429. [Google Scholar]
- La Nasa, J.; Modugno, F.; Aloisi, M.; Lluveras-Tenorio, A.; Bonaduce, I. Development of a GC/MS method for the qualitative and quantitative analysis of mixtures of free fatty acids and metal soaps in paint samples. Anal. Chim. Acta 2018, 1001, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Regert, M.; Bland, H.A.; Dudd, S.N.; Van Bergen, P.F.; Evershed, R.P. Free and bound fatty acid oxidation products in archaeological ceramic vessels. Proc. R. Soc. B Boil. Sci. 1998, 265, 2027–2032. [Google Scholar] [CrossRef]
- Colombini, M.P.; Modugno, F.; Ribechini, E. Direct exposure electron ionization mass spectrometry and gas chromatography/mass spectrometry techniques to study organic coatings on archaeological amphorae. J. Mass Spectrom. 2005, 40, 675–687. [Google Scholar] [CrossRef] [PubMed]


| Sample Number; Description | KBr-FTIR Results | Organics through Solvent-Extracted FTIR Results | Organics through GC-MS |
|---|---|---|---|
| 11544 Remain from interior | Inorganics a: Calcite (l.a.), silicates, SiO2 (r); oxalates. Organics b: carboxylic acids, metal soaps (l.a.), esters. | Methanol: glyceryl mono- and diesters, fatty monoacids, diacids, and carboxylates (metal soaps). Acetone: acylglycerols (l.a.), saturated fatty monoacids (r.), anhydrides/lactones. Dichloromethane: acylglycerols, short/mid-chain fatty monoacids (r.) | FAs C16:0 (r.) and C18:0; diacids C9di and C13di; Even-numbered sat. FAs C8:0–C24:0;Odd-numbered sat. FAs: C9:0 and C15:0; Unsat. FAs: C16:1 and C18:1.Oxo- and hydroxy-FAs C6, C7, C8. Dihydroxy-FAs C18(diOH) and C22(diOH). Diacid series (C4di–C14di). C16- and C18-MAG (l.a.). Beta-sitosterol (l.a.). Glycerol (r.). |
| 11550 Remain from interior | Inorganics a: Silicates, SiO2; calcium oxalate (whewhellite), copper oxalate (moolooite) (r.). Organics b: carboxylic acids, metal soaps, esters. | Methanol: glyceryl mono- and diesters, fatty monoacids, diacids, and carboxylates (metal soaps). Acetone: acylglycerols, fatty monoacids (r.). Dichloromethane: acylglycerols (l.a.), short/mid-chain fatty monoacids (r.). | FAs: C16:0 (r.) and C18:0; diacids C8di, C9di (r.) and C13di. Even-numbered sat. FAs: C8:0–C26:0. Odd-numbered sat. FAs: C9:0 and C15:0. Unsat. FAs: C18:1. Full range of diacids: C4di–C14di. Glycerol (l.a.), MAG C16-G and C18-G; glyceric acid (tr). |
| 11551 Remain from interior | Inorganics a: Silicates, SiO2, copper oxalate (moolooite). Organics b: carboxylic acids, metal soaps, esters. | Methanol: Glycerol and/or acylglycerols (r.), fatty acids (r.), diacids, carboxylates (metal soaps). Acetone: acylglycerols (r.), fatty monoacids (r.), diacids, anhydrides/lactones (l.a.). Dichloromethane: acylglycerols (r.), short/mid-chain fatty monoacids (r.). | C16:0 (r.), C18:0, C22:0 and C24:0 FAs; diacids C8di (r.), C9di and C13di. Even-numbered sat. FAs (l.a.): C8:0–C28:0 Odd-numbered sat. FA: C9:0 Glycerol (l.a.), C16- and C18-MAG; glyceric acid (tr). HCs: sat. C43–C49 and unsat. C28–C31. [meaning?] |
| 11573 Remain from connection area | Inorganics a: calcite, silicates, calcium oxalate (whewhellite) (r.) Organics b: carboxylic acids (l.a.), esters (l.a.). | Methanol: acylglycerols, carboxylates (metal soaps). Acetone: acylglycerols, fatty monoacids (r.). Dichloromethane: acylglycerols, short/mid-chain fatty monoacids. | Diacids C8di, C9di (r.), C11di and C13di (r.). Glycerol (r.). Even-numbered sat. FAs C8:0–C24:0. Odd-numbered sat. FA: C9:0 MAG C16-G and C18-G. Diacids: C4di–C15di. |
| 11596 Remain from interior | Inorganics a: calcium oxalate (whewhellite) (r.) Organics: carboxylic acids (l.a.), esters (l.a.). | Methanol: Glycerol, carboxylates (metal soaps). Acetone: unspecified trace amounts. Dichloromethane: short/mid-chain fatty monoacids (l.a.). | Diacids C8di and C9di (r.). Glycerol and MAG C16-G and C18-G. Even-numbered sat. FAs C8:0–C24:0. Odd-numbered sat. FA: C9:0 MAG C16-G and C18-G. Diacids: C4di–C14di. HCs: sat. C23, C24, C27, C30. |
| 11598 Remain from interior | Inorganics a: calcite (l.a.), silicates (r), oxalates. Organics b: carboxylic acids, carboxylate salts (tr.), esters. | Methanol: acylglycerols, fatty acids, carboxylates (metal soaps). Acetone: acylglycerols. Dichloromethane: short/mid-chain acylglycerols (r.), fatty monoacids (tr.). | Diacids C8di and C9di (r.). Glycerol and MAG C16-G and C18-G. Even-numbered sat. FAs C8:0–C24:0. Odd-numbered sat. FA: C9:0 and C17:0 unsat. FAs: C18:1. MAG C16-G and C18-G. Diacids: C4di–C14di. HCs: sat. C23, C24, C27, C30. Hypoxanthine, maltose (possible contaminants). |
| 11622A Remain from cavities in connection area | Inorganics a: silicates (l.a.), calcium oxalate (whewhellite) (r.) Organics b: carboxylic acids (l.a.), carboxylate salts (tr.), esters. | Methanol: acylglycerols, carboxylates (metal soaps). Acetone: acylglycerols. Dichloromethane: acylglycerols, short/mid-chain fatty monoacids. | Dihydroxy FA: 13,14-dihydroxy C22:0 FA (r.); Diacids C8di, C9di (r.) and C11di (r.); Glycerol (r.). Even-numbered sat. FAs C8:0–C24:0. Odd-numbered sat. FA: C9:0 MAG: C16-G and C18-G Diacids: C4di–C15di. Dehydroabietic acid and 7-oxo-dehydroabietic acid. |
| 11622B Powder from addition on vessel body | Inorganics a: Gypsum (r.), calcite, silicates, SiO2 (r). Organics b: n.d. | No organics found. | n.a. |
| 11633A Remain from connection area | Inorganics a: Silicates, SiO2, (r), calcium oxalate (whewhellite) (r.), copper oxalate (moolooite) (tr.). Organics b: carboxylic acids (l.a.), metal soaps (r.), esters (l.a.). | Methanol: acylglycerols, carboxylates (metal soaps). Acetone: acylglycerols. Dichloromethane: acylglycerols, short/mid-chain fatty monoacids (r.). | Glycerol (r.); C22(diOH) (r.). Even-numbered sat. FAs C8:0–C24:0. Odd-numbered sat. FA: C9:0 and C17:0. MAG: C16-G and C18-G. Diacids: C4di–C15di. |
| 11633B Powder from interior remains | Inorganics a: Gypsum (r.), calcite, silicates, SiO2. Organics: n.d. | No organics found. | n.a. |
| Maximum, Wavenumber (cm−1) | Assignment 1 | Type(s) of Compounds |
|---|---|---|
| 3700–3696 | vOH | Hydroxides or crystalline water in silicates (clay) |
| 3624–3620 | vOH | Hydroxides or crystalline water in silicates (clay) |
| 3440–3400 (br) | vOH | Hydroxyl in acylglycerols, hydroxy-acids Adsorbed water in silicates |
| 3300–3200 | vOH | Carboxyl in free FA |
| 2960–2950 | vasCH3 | FA and FAE |
| 2951–2937 | vasCH2 | Diacids |
| 2940–2935 | C4–C6 | |
| 2930–2920 | C7–C12 | |
| 2918–2910 | C13–C18 | |
| 1740–1730 | vC=O | Fatty esters (acylglycerols) |
| 1718–1712 | Short-chain fatty monoacids (C4–C9) | |
| 1711–1709 | Mid-chain fatty monoacids (C10–C12) | |
| 1708–1701 | Long-chain fatty monoacids (>C12) | |
| 1700–1695 | Diacids | |
| 1650–1630 (br) | δOH | Adsorbed water in silicates |
| 1640 | vasCOO- | Copper oxalate (moolooite) |
| 1623 | vasCOO- | Calcium oxalate (whewhellite) |
| 1590 1551 1541 | vasCOO- | Fatty acid metal salts |
| 1470 | δCH2 | Alkyl chains in diacids and fatty esters (acylglycerols) |
| 1458 | δCH2 | Alkyl chains in monocarboxylic acids |
| 1425 | δipC–O–H | Diacids |
| 1412 | δCH2 | Alkyl chains in diacids |
| 1414–1408 | δipC–O–H | Fatty monoacids |
| 1393 | δipC–O–H | Alcohol in glycerol or glyceryl group |
| 1380–1364 | δsCH3 | Terminal methyl groups in fatty monoacids and their glycerol esters; relatively stronger as compared to δCH2 in short-chain monoacids |
| 1317 | vsCOO- | Calcium oxalate (whewhellite) Copper oxalate (moolooite) |
| 1180 | vC–O–C=O | Ester links (acylglycerols) |
| 1105 | vC–O–C=O | Ester links (acylglycerols) |
| 1090 | vSi–O | Silicates: quartz sand |
| 1026 | vSi–O | Silicates: clay |
| vC–O–H | Glycerol or glyceryl group | |
| 817 | δOCO | In carboxylates: copper oxalate (moolooite) |
| 780 | δOCO | In carboxylates: calcium oxalate (whewhellite) |
| 798/779 | vasSi–O–Si | Silicates: quartz sand |
| 722, 691 | ρCH2 | Long alkyl chain of fatty acids and salts |
| 527 | δOSiO | Silicates |
| 467 | δOSiO | Silicates |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koupadi, K.; Boyatzis, S.C.; Roumpou, M.; Kalogeropoulos, N.; Kotzamani, D. Organic Remains in Early Christian Egyptian Metal Vessels: Investigation with Fourier Transform Infrared Spectroscopy and Gas Chromatography–Mass Spectrometry. Heritage 2021, 4, 3611-3629. https://doi.org/10.3390/heritage4040199
Koupadi K, Boyatzis SC, Roumpou M, Kalogeropoulos N, Kotzamani D. Organic Remains in Early Christian Egyptian Metal Vessels: Investigation with Fourier Transform Infrared Spectroscopy and Gas Chromatography–Mass Spectrometry. Heritage. 2021; 4(4):3611-3629. https://doi.org/10.3390/heritage4040199
Chicago/Turabian StyleKoupadi, Kyriaki, Stamatis C. Boyatzis, Maria Roumpou, Nick Kalogeropoulos, and Despoina Kotzamani. 2021. "Organic Remains in Early Christian Egyptian Metal Vessels: Investigation with Fourier Transform Infrared Spectroscopy and Gas Chromatography–Mass Spectrometry" Heritage 4, no. 4: 3611-3629. https://doi.org/10.3390/heritage4040199
APA StyleKoupadi, K., Boyatzis, S. C., Roumpou, M., Kalogeropoulos, N., & Kotzamani, D. (2021). Organic Remains in Early Christian Egyptian Metal Vessels: Investigation with Fourier Transform Infrared Spectroscopy and Gas Chromatography–Mass Spectrometry. Heritage, 4(4), 3611-3629. https://doi.org/10.3390/heritage4040199







