3.1. Physical Oceanography of the Wreck Site in the Sea of Marmara
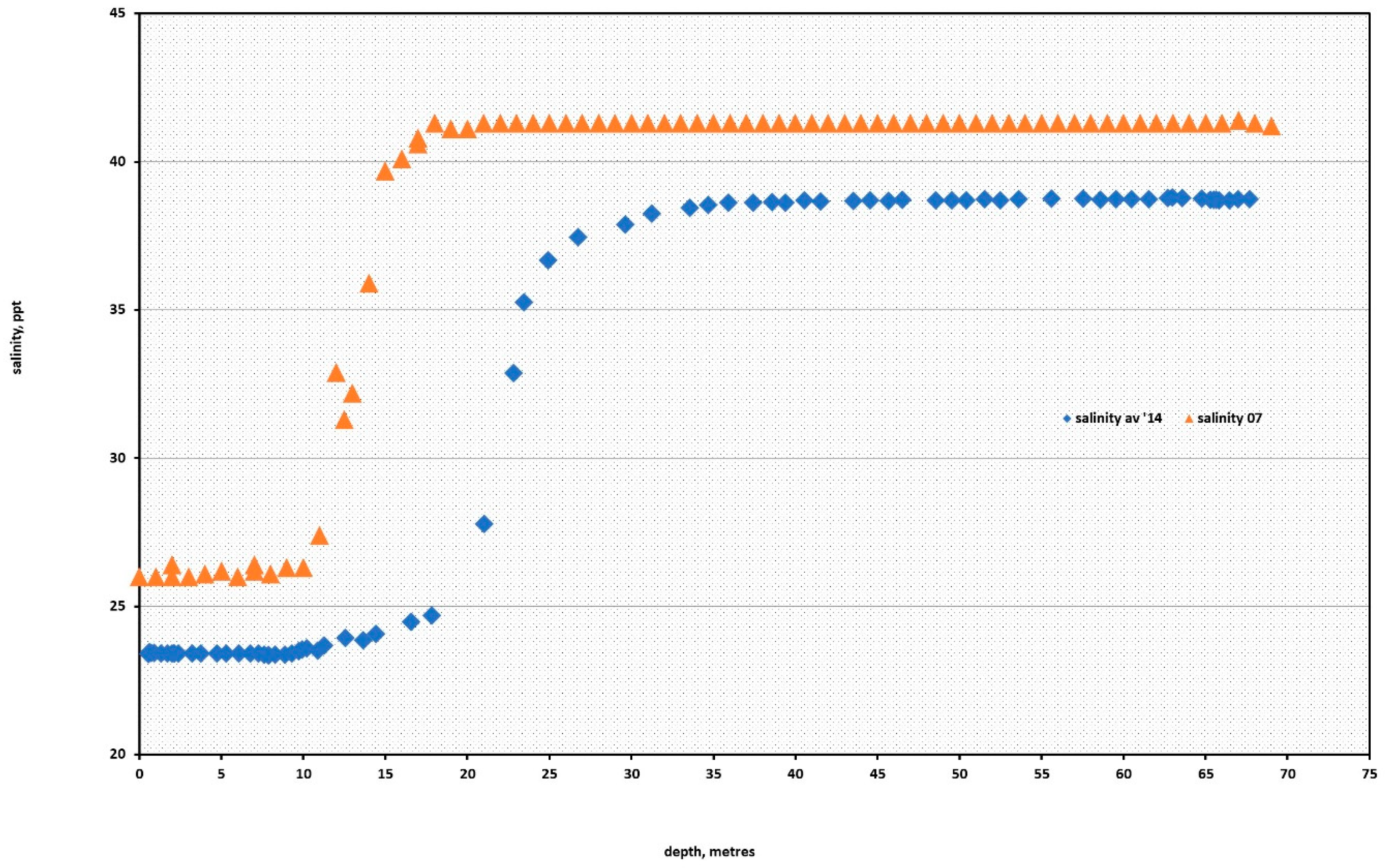
During the 2004 assessment, the water column above the wreck was measured for salinity, dissolved oxygen (YSI probe) and temperature at 0.5-m intervals, using a TPS 90DC meter that logged the data, to a maximum depth of 73 m. The first 13 m had a mean salinity of 26.1‰ ± 0.1‰, which reflects evaporative concentration of the 18‰ water inflowing from the Black Sea. Over the next eight metres, the salinity rapidly increased to 41.3‰ ± 0.8‰ at the bottom of the halocline. The higher salinity is typical of the hypersaline Aegean Sea, which enters the Sea of Marmara via the Dardanelles. Captain Stoker had used this counter current to help him penetrate the mined straits. At the 2004 halocline there was a corresponding thermocline (change in temperature) where the temperature fell from 26 to 18 °C and a second thermocline, at 40 m, saw the temperature fall from 18 to 16 °C, which was due to the heat capacity of the higher salinity water (
Figure 1).
During the 2014 expedition, a more sophisticated Sonde recorded the dissolved oxygen, temperature, salinity and redox potential every 20 s. Graphical comparative analysis of the data from the two times of the summer and spring, seven years apart, showed similar trends. This data recorded the greatest fall in temperature in the lower end of the less salty water, between 10–20 m, and above this depth the temperature was relatively constant at 15 ± 0.9 °C between 20 and 72 m, the depth of the wreck.
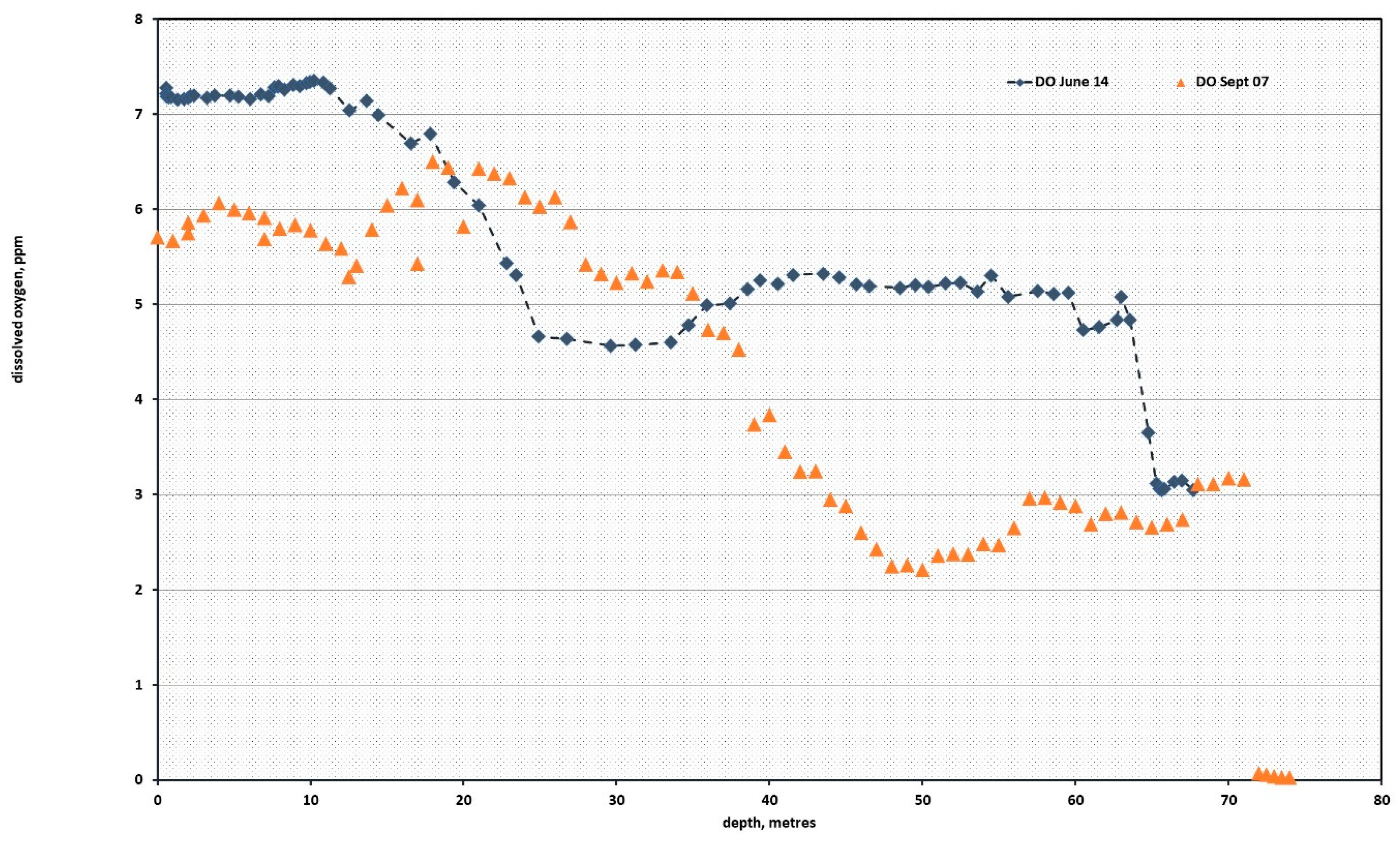
The surface waters in the Sea of Marmara were well oxygenated between 7 and 5.5 ppm, as shown in the 2007 and 2014 profiles shown in
Figure 2. The data from 2007 shows the dissolved oxygen falling from 5 to 3 ppm as the depth increases from 35 to 50 m. At the upper sections of the submarine, the dissolved oxygen was recorded at 2.8 ± 0.2 ppm, which represents 36% saturation of the seawater for a salinity of 41.3‰ at 16 °C [
6].
The data from June 2014 showed a dissolved oxygen that fell with increasing salinity, to reach a minimum of 4.5 ppm, and then increased in response to currents flowing in the hypersaline waters at depths until the submarine environment was reached, at 67 m, where it fell to the 3-ppm level seen in 2007.
3.2. Corrosion of the Submarine in the Sea of Marmara
The submarine lies on the bottom of the Sea of Marmara with a silt mound coming half-way up the hull. Images of the wreck at
www.submarineinstitute.com show that a combination of corrosion and trawler net damage has holed the thin plate sections at the bow and on the combing (structure that covers a range of fittings to streamline the water flow) behind the conning tower, leaving the structural frames exposed (
Figure 3 and
Figure 4). The drop camera used in 2007 showed that the non-ferrous metal fixtures on the interior of the conning tower were covered in a thin layer of concretion from the galvanic protection afforded by the surrounding iron objects [
7]. The interior of the conning tower was made of bronze to make sure that the magnetic compass would not be affected by the proximity of ferrous metals. The hatch cover, ladder, control valves and steering gear were all made of brass or bronze. Owing to the confined nature of the seawater inside the vessel it is likely that the massive banks of batteries will be involved in long-range or proximity corrosion that will have affected the electric motors and the diesel engines used for recharging the battery banks [
8].
Proximity corrosion refers to long-distance galvanic coupling found on historic shipwreck sites and on submerged off-shore modern structures, such as production gas and oil platforms. When dissimilar metals are covered with a concretion layer, they lose their electrical isolation, and the more reactive metals corrode while the more noble alloys are protected.
Based on the drop camera study, it was deemed likely that the hatch cover could be opened, since cathodic protection from the hull will have prevented major corrosion attack on the hinge mechanism. The opening of the main hatch to allow the ROV to enter the submarine was delayed underwater when divers found that there was 25–35-mm-thick concretion on the hatch mechanism, some 15 times thicker than expected. Because the higher salinity environments have higher total mineral content, the alkalization of the hinge resulted in a massive concretion deposit (
Figure 5). The bond around the spindle and hinge brackets was broken using a combination of de-concreting tools and a hydraulic jack. A chain block was needed to gently ease the hatch open for the first time in over 100 years. Cathodic current from reduction of oxygen on the non-ferrous metals makes the surface more alkaline (Equation (1)). In the case of the
AE2 the outcome was the precipitation of massive calcareous deposits.
By consuming the soluble bicarbonate ion present in seawater, the hydroxide ions produced in Equation (1) increase the carbonate concentration and so the calcareous deposits are formed (Equation (2)).
A barnacle from the base of the concretion was a deep charcoal grey to black, which is likely to be associated with magnetite, the corrosion product associated with iron corrosion in a low oxygen environment.
3.4. Concretion and Site History
The accidental impact of a two-tonne concrete anchor during an intense storm removed a small section (50 cm
2) of concretion from the
AE2 hull, which facilitated direct measurement of the hull plate thickness. Samples of the concretion were examined by materials and corrosion scientists. An ROV inspection showed that the bare metal had been covered by a new concretion layer within three years [
10,
11]. The concretion sample showed a typical anaerobic concretion, with a very dense milieu of sharp shell debris and black iron corrosion products, which consisted of magnetite (Fe
3O
4 determined by petrographic analysis) and a number of iron sulphides, which included FeS, based on atomic ratios from the Energy Dispersive X-Ray Spectrum (EDS) analysis from the electron microscope. As expected, there were several other species, containing iron, sulphur, chloride and oxygen, present [
12]. Samples of the priming coat of red lead oxide, Pb
3O
4, were found at the original metal surface, which was covered by a primary concretion layer ≈1.6 mm thick, a secondary 8.8 mm layer, and the outer layer was 3.9 mm thick. Sections of concretion showed pitting corrosion beneath the original surface, to a depth of 3 mm.
The differences in colonization of the conning tower and the upper sections of the combing indicates that the sediment levels have not always been the same as those found when the site was inspected in 2007. The concretion model indicates an initial four–five years of relatively intense corrosion, as paint layers are penetrated and a steady corrosion rate is established [
13]. The primary corrosion layer in the concretion appears to correspond to the first 13 ± two years of immersion (i.e., from 1915 to 1928). Changing conditions due to massive storms disturbing the site resulted in the second and third layers.
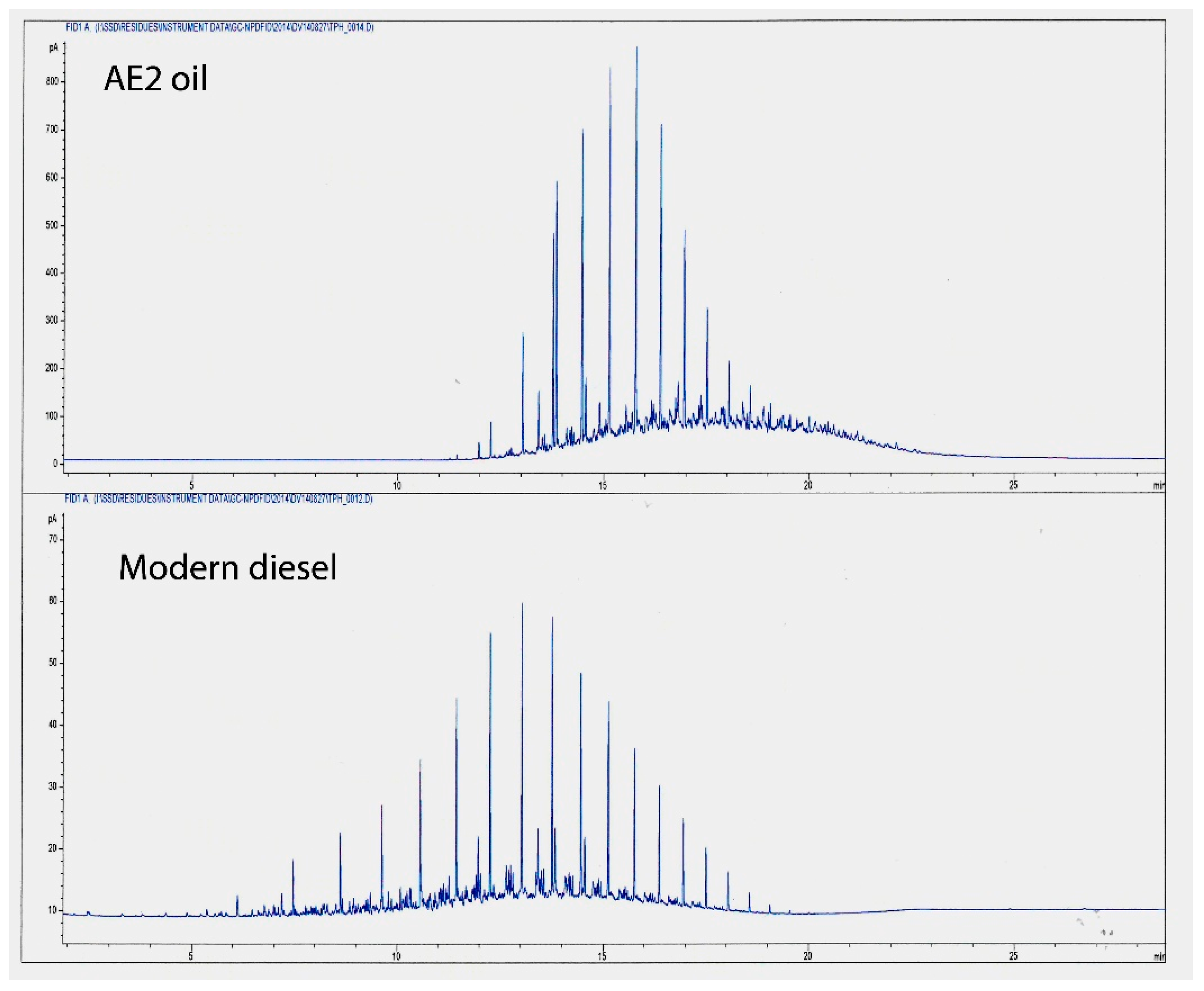
Samples of oil from inside the boat were collected from the surface of the ROV following its successful navigation of the internal spaces of the engine room. Viscosity data and gas chromatographic analysis of the volatile organic compounds coming from the oily residue indicated that it was diesel fuel, which had been leaking from the forward tanks. The analysis showed that the oil was biologically fractionated through removal of the sour and acidic smelling thiols and thiophenes. The bacteria preferentially consumed much of the long chain alkanes of the general formula C
nH
2n+2 (
Figure 6). The biodegradation of the AE2 oil mixture was indicated by the presence of the branched alkanes, norpristane (C
18H
38), pristane (C
19H
40) and phytane (C
20H
42), which are much more stable than the branched hydrocarbons. It is interesting that pristane is derived from shark liver oil and so this may be one of the principal components of lubricating oils and diesel fuel for the generators that was recovered from the surface of the ROV. The video inside the submarine has evidenced movement of large black mats of biological growth, which are the by-products of the microbiological attack on the diesel fuel.
3.5. Assessment of the Corrosion Rate on AE2
The hand-held readings from the Cygnus
® ultrasonic metal thickness gauge gave 4.6 ± 0.9 mm for the bare metal of the ballast tank, which was originally 6.35 mm thick. The apparent loss of 1.55 mm of metal over the 92.4 years of immersion equates to a corrosion rate equivalent to 0.017 ± 0.003 mm/year. Comparative data from the
Monitor showed that the interior of the turret, which was in an anaerobic microenvironment like the
AE2, had the same corrosion rate of 0.016 mm/year [
14]. It should be recalled that the E
corr values of the two wrecks were the same. The corrosion rate for iron shipwrecks in open-ocean waters can be calculated using an empirical equation (Equation (3)) relating the log of the corrosion rate, measured in mm/year as
dg or depth of graphitization of cast iron objects on the wreck to water depth
d in metres [
3] viz.,
With a depth of 71 m, Equation (3) predicts a corrosion rate of 0.018(3) mm/year, which is the same as the observed rate of 0.017 ± 0.003 mm/year. The silty microenvironment of the submarine would naturally result in an overall lower corrosion rate than that for wrecks lying proud of the seabed in well oxygenated conditions. More simply, the lower sections of the submarine may be in a better state of preservation, but testing would be required to assess the impact of microbially-induced corrosion (MIC) in the lower section of the pressure hull.
3.7. Modelling Corrosion Processes on AE2
It has been previously reported that the concretion sample was used to study how fast the corrosion reaction of steel plate equilibrates in seawater [
15]. The concretion sample was attached to a section from an 1890’s paddle steamer boiler, which had openable spaces to allow monitoring of both the pH and the chloride ions, and an insulated copper wire permitted monitoring of the E
corr of the metal. During the 11-month experiment there was a systematic increase in the acidity and the chlorinity of the solutions trapped underneath the concretion. The pH of the seawater fell from 7.9 to 4.3, while the chloride increased from 16,300 to 34,600 ppm. The increased acidity was due to hydrolysis reactions of the primary corrosion product FeCl
2 with water (Equation (5)).
The corrosion cell took 10 months to reach equilibrium. On this basis, it is reasonable to assume that recorded in-situ values of the pH and the E
corr, of this data accurately reflect the dynamic equilibrium between the wreck and the local marine environment. Plots of the concentration of chloride and pH best fitted square root of time plots, which support the diffusion-controlled nature of the corrosion process. The
AE2 concretion corrosion cell showed that the chloride ion concentration increased by a factor of 2.1 times after sitting for 11 months in an unstirred solution. By comparison, the increase in chlorinity with the high energy surging environment of the
Zuytdorp (1712) wreck, off the Western Australian coast, was 2.9 times after 270 years [
3]. Monitoring of the
cell E
corr value showed that it reached the on-site value in just eight months, but it took another few months of measurements to confirm that the plateau had been reached.
3.8. Cathodic Protection System
Given that relocation of the submarine was not a possibility, owing to the combination of the high risk of detonation of an unexpended torpedo and the prohibitively expensive creation of a dry-dock and museum conservation facility, an alternative approach to the conservation management had to be developed. In the 18th century, Volta found that the difference in reactivity of various elements could be harvested through the development of a voltaic pile, consisting of alternating layers of copper and zinc sheets. These studies were the first major experiments in differential corrosion rates of metals. The use of sacrificial reactions of a more reactive metal to provide a cathodic (negative) current, to stop the removal of electrons from a more noble metal, has been the subject of electrochemical experiments since the time of Sir Humphrey Davy, on Royal Navy copper-sheathed wooden vessels in 1761. In the case of AE2, the difference in reactivity of zinc and iron provides the driving force of the cathodic protection system for the wreck. There is approximately 400 mV difference between the Ecorr of the iron on the AE2 and the zinc anodes, and this is enough to stop corrosion of the submarine. As cathodic current flows through the insulated copper cables connecting the anode pods to the boat, the change of polarity causes outward migration of chloride ions and consumption of the acidity that had resulted from the accumulation of acid during the hydrolysis reactions of the corrosion products.
Design calculations considered a 5–10-year protection program and it was understood that the rate of anode consumption would be initially much higher than the long-term rate, as readily consumed hydrogen ions had built up to a moderate level underneath the concretion. Another consideration impacting on the design life was the high cost of mounting sea-borne diving operations. The final configuration consisted of three anode pods (
Figure 9). The cathodic protection of the submarine is the largest in-situ conservation project ever attempted on an historic iron shipwreck. Previous work by the author has resulted in successful in-situ treatment of guns, anchors and a marine steam engine, which resulted in a more rapid processing of the artefacts from recovery to exhibition status [
2,
3,
16].
The anode pods were assembled on a 2.6 m sided square base, which was filled with concrete to prevent them sinking into the silt. The supporting structure weighed 0.5 tonnes, giving an aggregate weight of around 1.5 tonnes to each pod. Each anode (150 × 7.5 × 7.5 cm) had a central iron core, to which insulated current-carrying cables were attached (
Figure 10). The distal end of the cable was attached to the submarine using an industrial three bolt system, as used in the cathodic protection of gas and oil production facilities. The 17 anodes on each pod had a geometric surface area of 7.84 m
2. In order to achieve a good spread of protection through the length of the boat, one pod was attached aft near the rear hydroplane, one amidships at the conning tower and one up forward on the windlass (
Figure 10). With an estimated geometric surface area of approximately 660 m
2 for the submarine and with the area of the anodes at 23.5 m
2, the industry standard ratio of approximately 30 times object to anode ratio was achieved. This ratio helps to avoid damage to the submarine if it was over-protected, as this could produce too much hydrogen pressure, which would blast off the protective concretion and lead to excessive consumption of anodes.
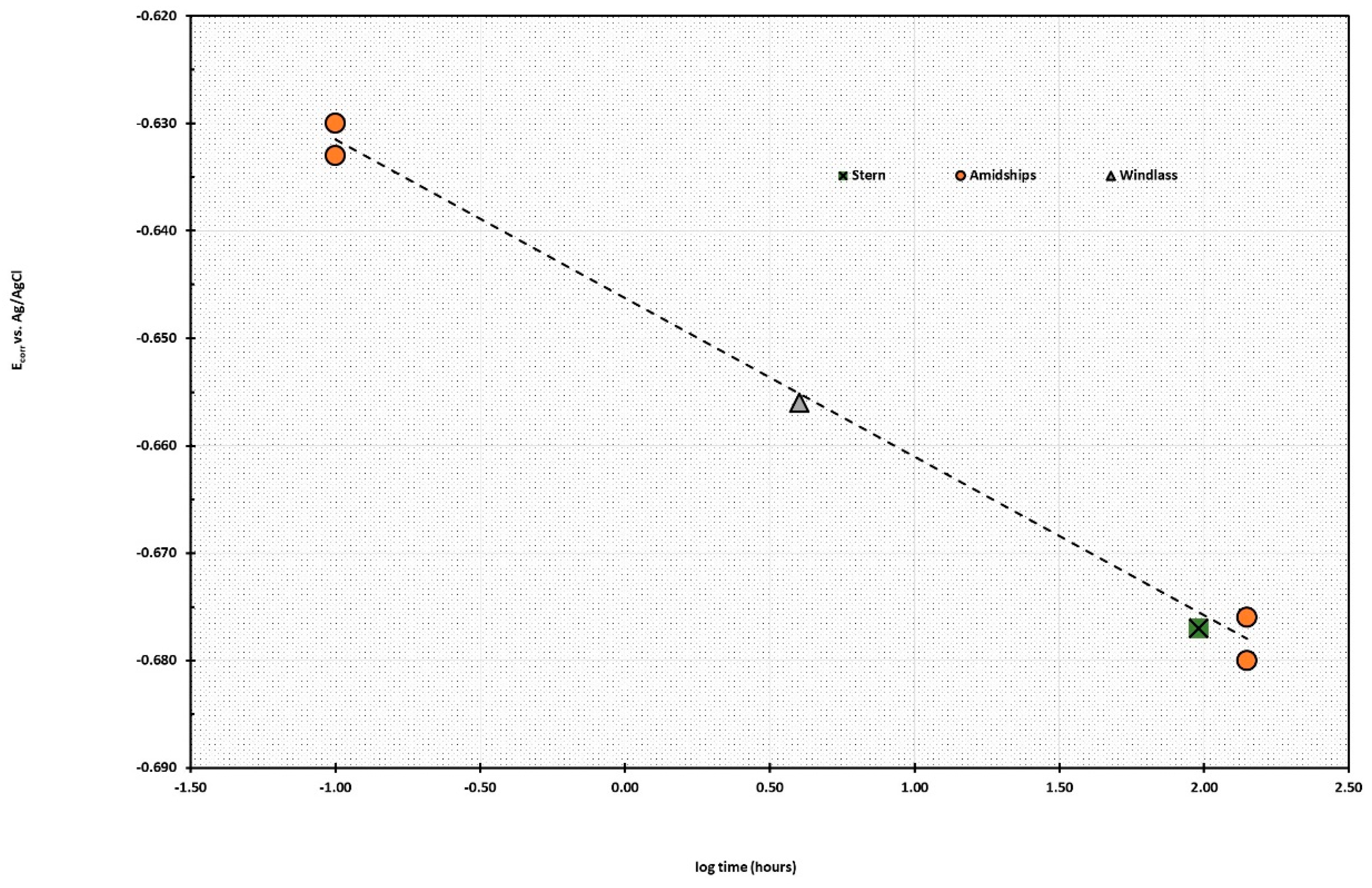
This ratio was developed by John McCoy of McCoy and Associates to comply with industry standards. Each of the locations was selected on the basis that there was significant residual metal present in that part of the wreck, to enable the safe and long-lasting attachment of the cables from the anode pods onto the submarine. The data listed in
Table 1 reports the variability of the E
corr measurements, which were reproducible to within a few millivolts.
Prior to attaching the anode cables with clamps to the submarine, the surfaces were hydro-blasted to remove small areas of concretion from the metal surface to get ohmic connections. The elapsed time at each attachment point was measured from the time at which the pods were attached and the time when the E
corr measurements were taken. The observed voltages were dependent on the logarithm of the elapsed time between anode attachment and voltage measurement. The linear regression analysis gives Equation (6), which had an R
2 of 0.9648 with an associated equation,
The numbers in parentheses are the errors associated with the intercept and the rate at which the submarine was being cathodically polarised. Since all the measurements fall along the same line, this indicated that the submarine was acting as an interconnected electrical unit, which was an unexpected bonus as it indicated that the overall condition of the submarine was structurally sound.
The data in
Table 1 shows that the greatest voltage drop between the natural cathodic protected E
corr of the conning tower was 50 mV after 141 h or close to eight days. This voltage drop is the equivalent of a 42% drop in the corrosion rate of the submarine, which is remarkable given the size of the boat! The E
corr values are diagrammatically illustrated in
Figure 11. This calculation is based on a combination of theoretical and practical determination of the rate at which the corrosion changes for concreted materials. For electrochemically controlled reactions, such as the in-situ treatment of the
AE2 submarine, the log of the corrosion rate can be calculated according to the changes in E
corr values, with a drop of approximately 330 mV needed for a ten-fold decrease in the rate of corrosion.
3.8.1. Chemical Environment in the Water Column and Inside AE2
During diving operations on the 2014 expedition, the ROV and the attached Sonde recording the in-situ data became stuck inside the submarine for just over a day, recording information every 20 s. A total of nearly 657,900 sets of 12 variables were recorded before the ROV was recovered, but before the data could be analysed it had to be averaged over every two minutes, as there was insufficient memory on the museum computers to resolve nearly eight million data points. One of the first outcomes of this analysis was that the apparent halocline observed in 2007, with the drop camera, was an artefact due to poor lighting and specular reflection from suspended material inside the vessel.
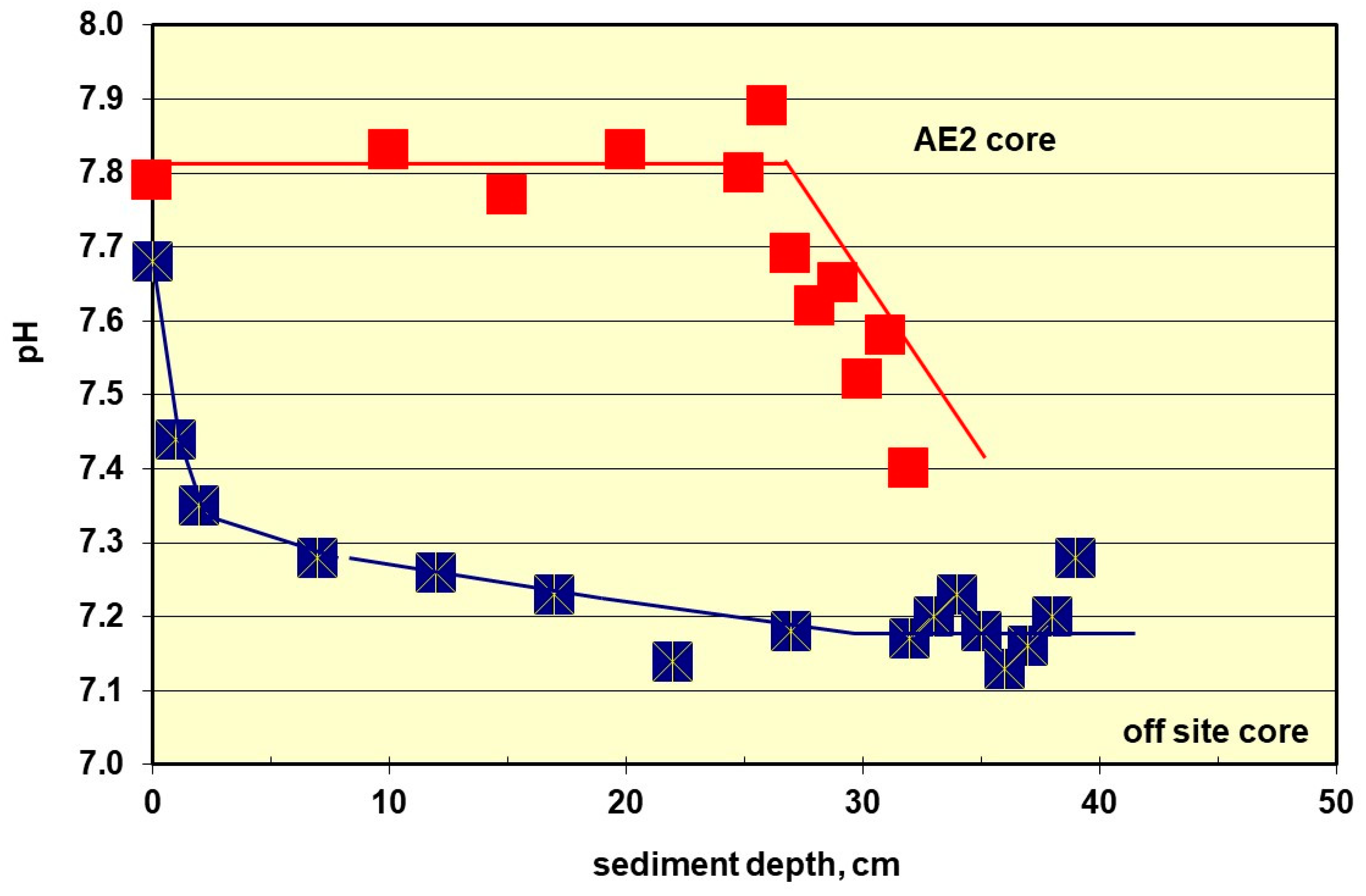
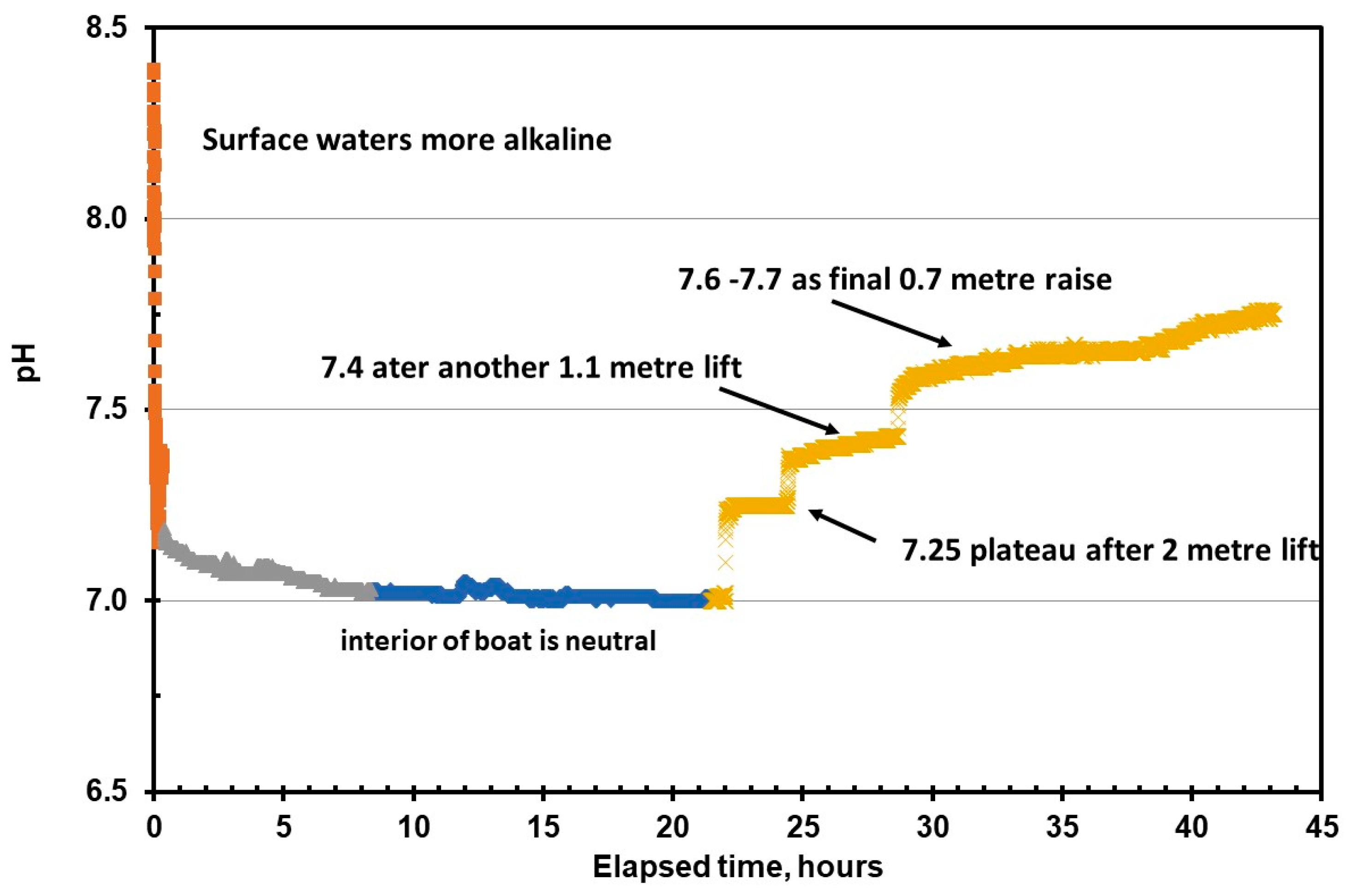
The pH data recorded by the Sonde is shown versus time inside the conning tower (
Figure 12). The red-brown line shows a fall from the surface value that is within the normal range of pH for the Sea of Marmara, which has an average pH of 8.26 ± 0.18. The green line represents the period when the Sonde was inside the submarine and operating as normal, which shows an average pH of 7.15 ± 0.03, which is typical of marine sediments that have bacteria active in them.
The extended blue line represents the period when the oxygen and salinity sensors were “poisoned” by sulphur species in the stirred-up sediment. When the divers began to recover the Sonde, the purple line and the plateau value of 7.2 represents the solution value in the water column above the sediment. As the instrument recovery operations increased, the plateau readings of 7.4 and 7.6 represent the different chemical microenvironments inside the submarine. The value of 7.4 is for the water 3.1 m up from the floor of the control room and 7.6 is for water at 3.8 m. The latter pH corresponds to the mean pH of the sediment cores from outside the submarine. The values between 7.2 and 7.4, those closer to the internal spaces of the lower control room, correspond with the mean pH (7.26 ± 0.08) from the external core adjacent to the submarine. The equivalent value at the first plateau region of the pH with the sediment core close to the submarine is a strong indication that the chemical processes of diagenesis inside the submarine were the same as those externally, but near the hull.
3.8.2. Analysis of the Dissolved Oxygen profiles inside AE2
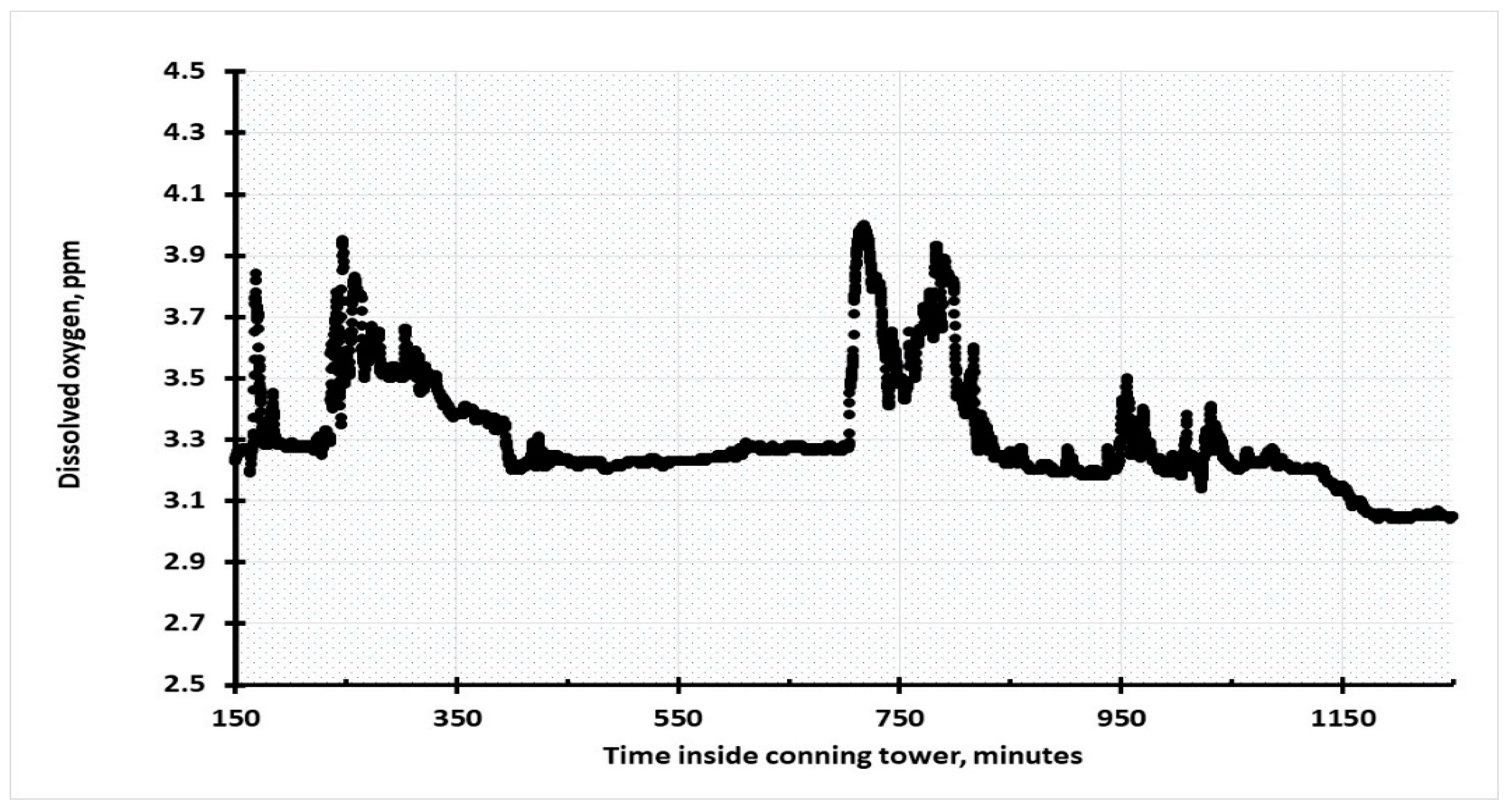
The dissolved oxygen inside the conning tower was generally around 3.2 ± 0.1 ppm but there were a series of rapidly rising and falling spikes, which saw an upward excursion to approximately 4.0 ± 0.2 ppm. At times these elevated levels were restored to their former values with a sigmoidal decrease in the concentration of oxygen. This is probably due to bacteria that consumed oxygen. More generally, there was a rapid return to the steady state value, as shown in
Figure 13. These phenomena indicate that there is mixing of different bodies of water inside the conning tower and that there is a significant level of microbiological activity inside the submarine.
The specific time intervals have no meaning other that providing an insight into the fluid dynamics inside the submarine. There was an interval between 400 and 700 min when there was no apparent mixing of the layers of solution, but the time between oxygen spikes was typically 75 ± five minutes. The varying speeds at which the elevated dissolved oxygen levels resumed their stasis values were due to which of the three processes were dominant: Firstly, there is natural diffusion from a high to a low concentration of oxygen, the second is bacterial consumption, and the third method is water movement around the sensing heads of the Sonde. Imaging recovered from the ROV showed large black algal matts. The simplest explanation of the oscillating oxygen levels is that the Sonde was positioned at a point where there was movement of different bodies of water inside the submarine, and that, at times, a more oxygenated solution flowed into the boat from an external source. Inspection of the dissolved oxygen vs. depth plots, in
Figure 2, show that just above the submarine the oxygen concentration was around 4 ppm, and so mixing of these waters with the internally equilibrated solutions would result in the fluctuations. Observations from the ROV operators of the cameras inside the submarine confirm that there was a small but measurable flow of water inside the control room and the waterborne sediments fall from suspension when the current meets the bulkhead. At this point the changed direction of water flow caused the sediment to collect in that area of the control room. The entrapment of the Sonde inside the submarine was extremely fortunate for it provided an extended period of passive observation of how the currents were changing the chemical environment inside the submarine. The data from the Sonde indicates that there are a range of environments inside the submarine that range from low oxygen to anaerobic, and that these different environments will have varying effects on the corrosion of artefacts that are naturally retained within the confines of the hull of
AE2.