Study of the Influence of Different Yeast Strains on Red Wine Fermentation with NIR Spectroscopy and Principal Component Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Grape Characteristics and Winemaking
2.2. FT-NIR Instrument and Chemometric Analysis
2.3. Sensory Analysis
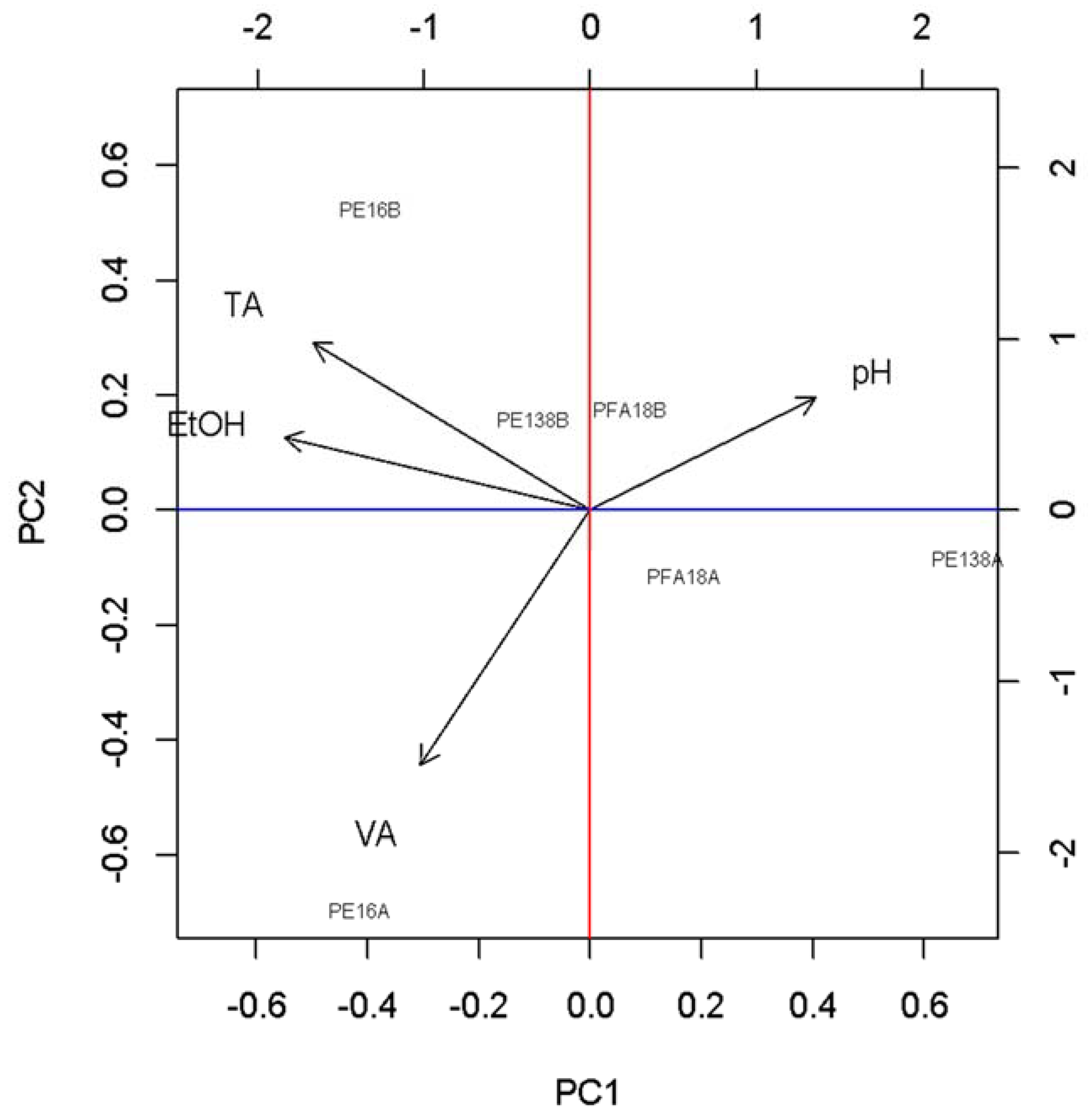
3. Results
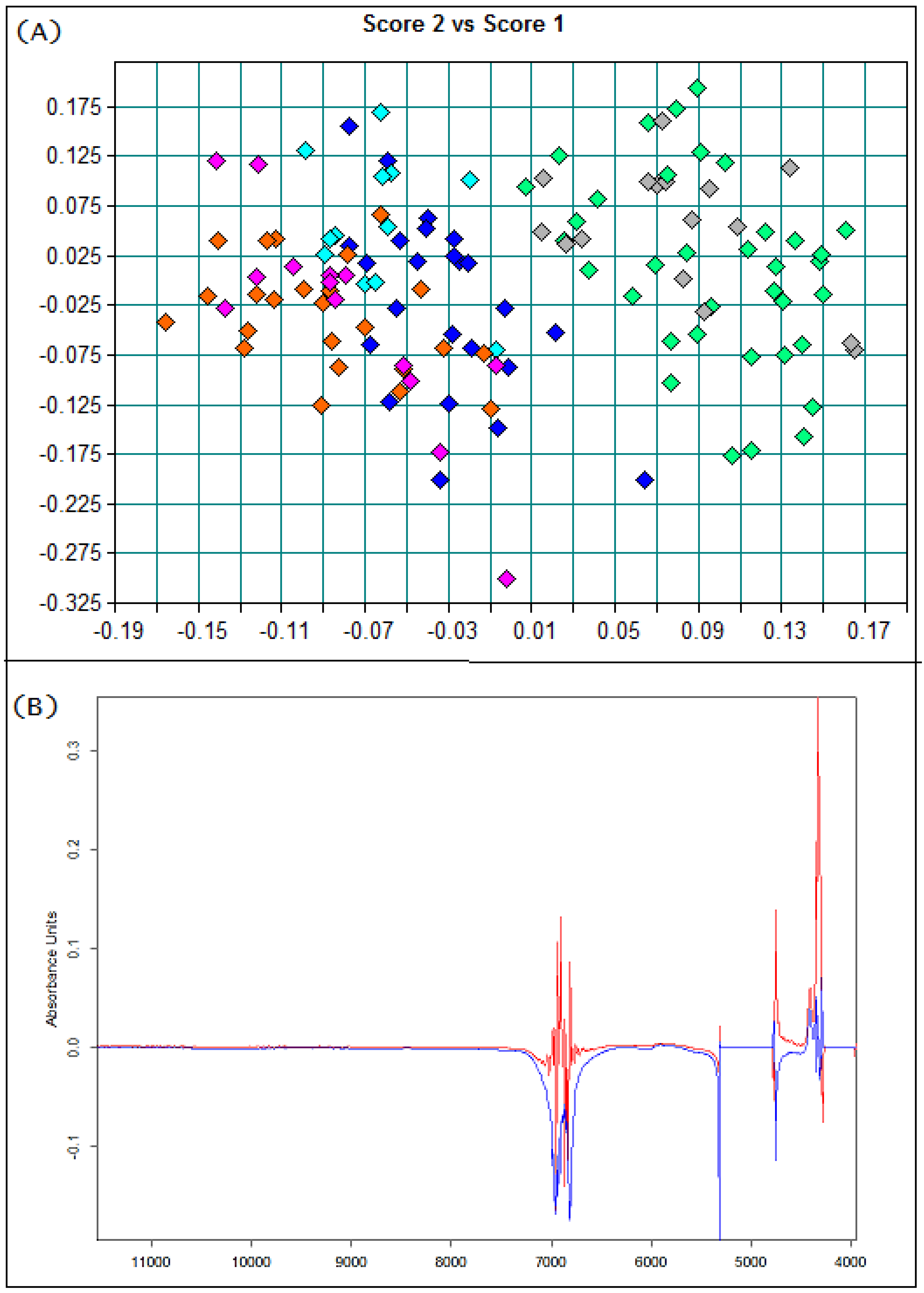
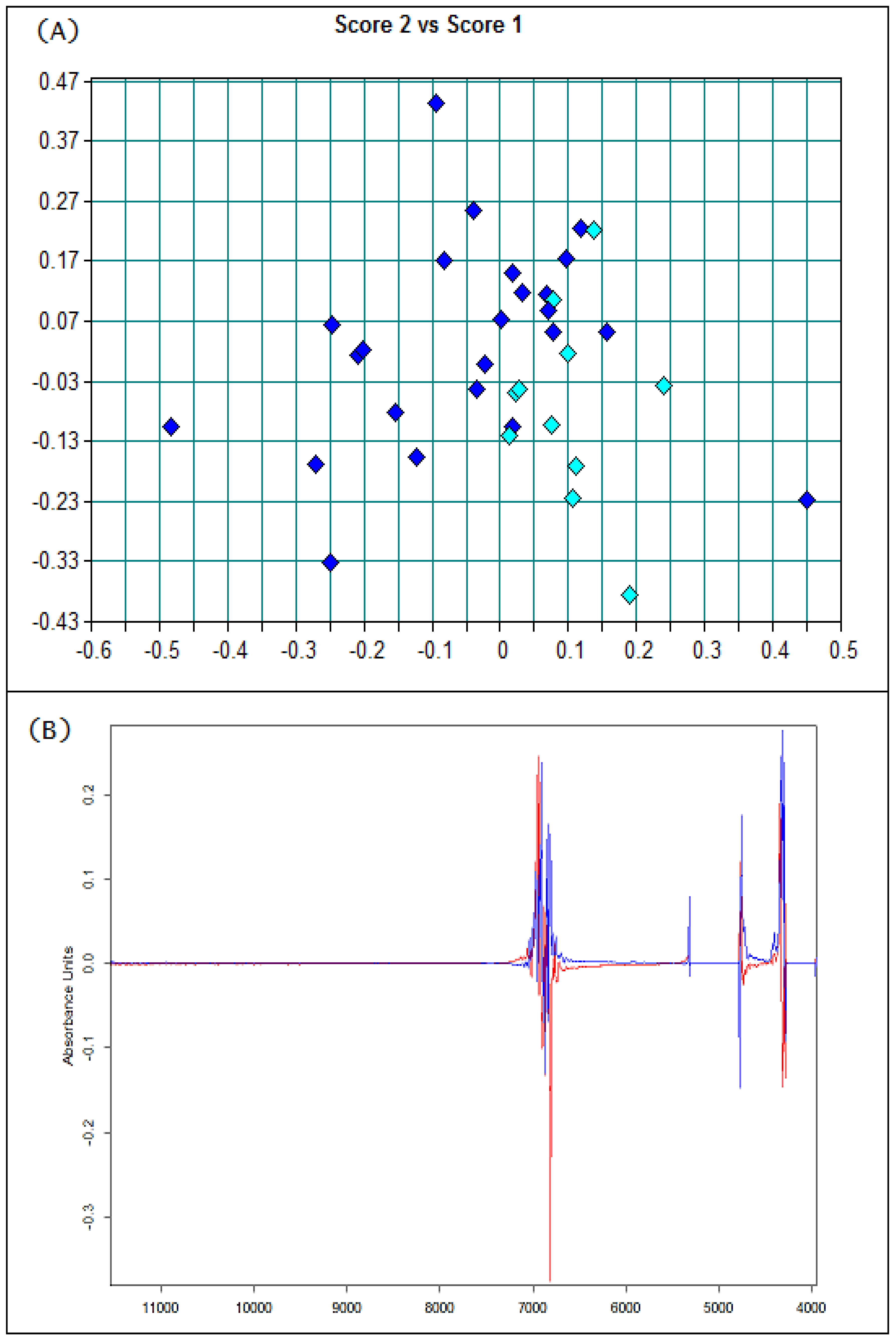
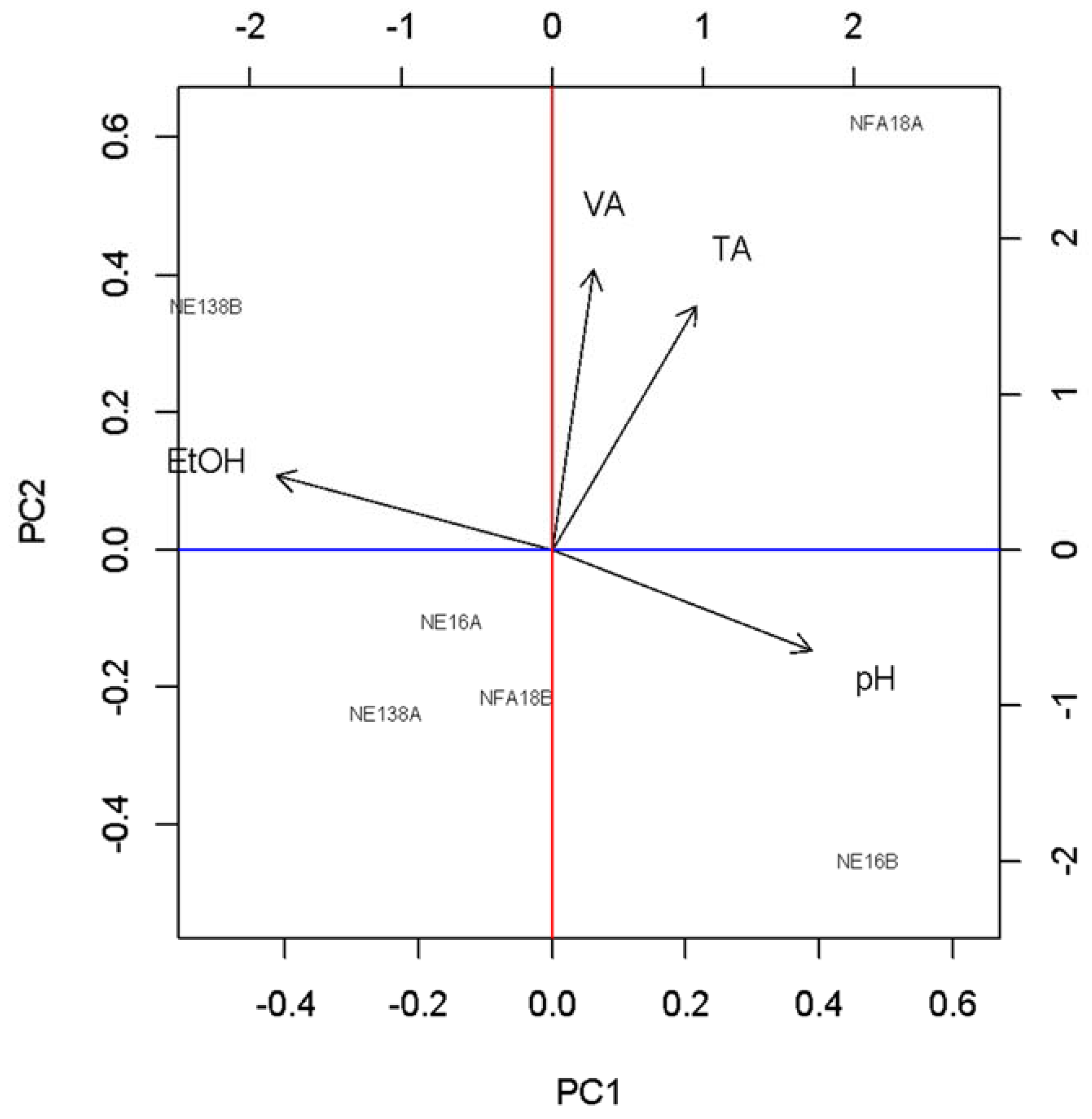
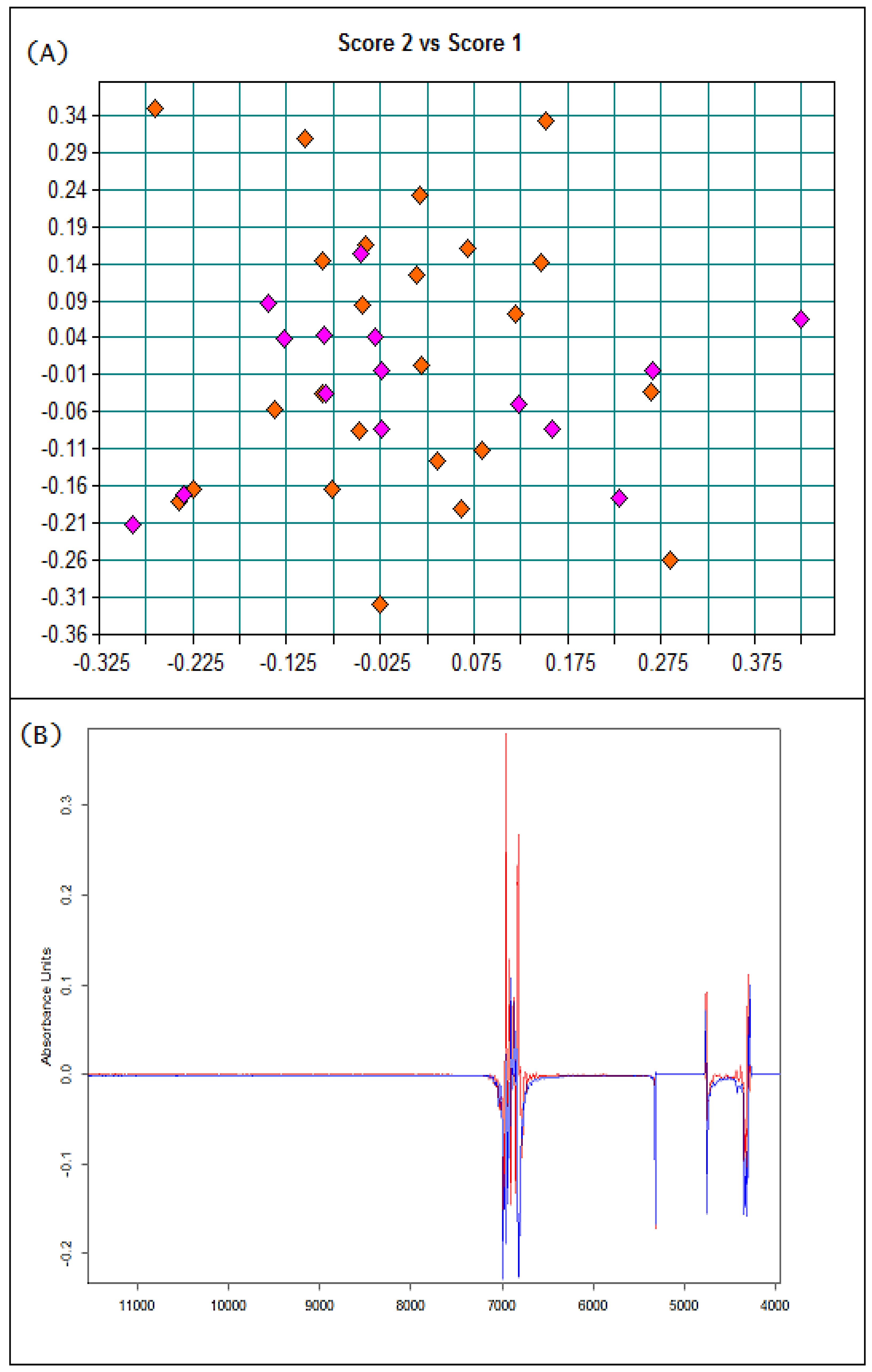
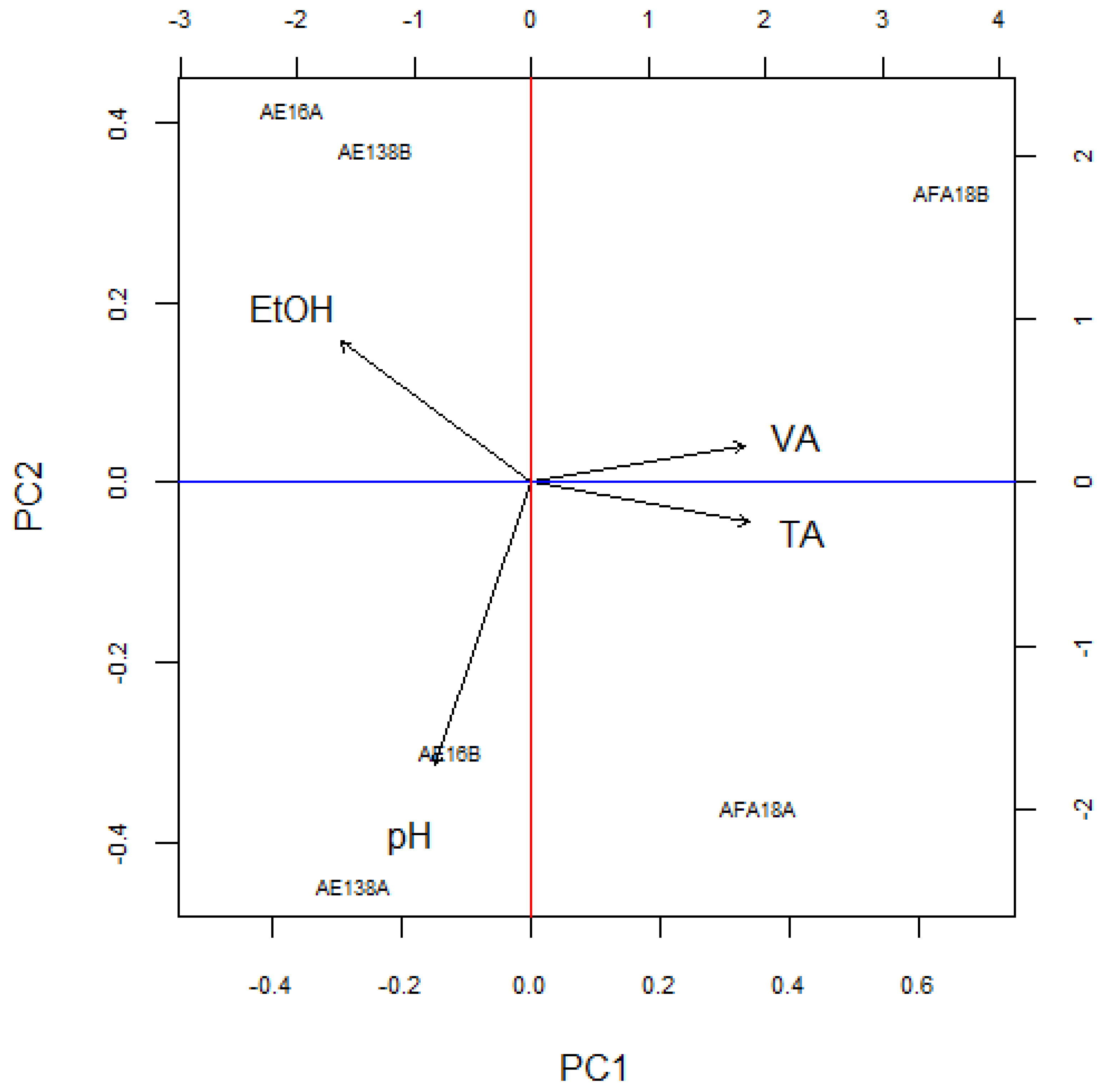
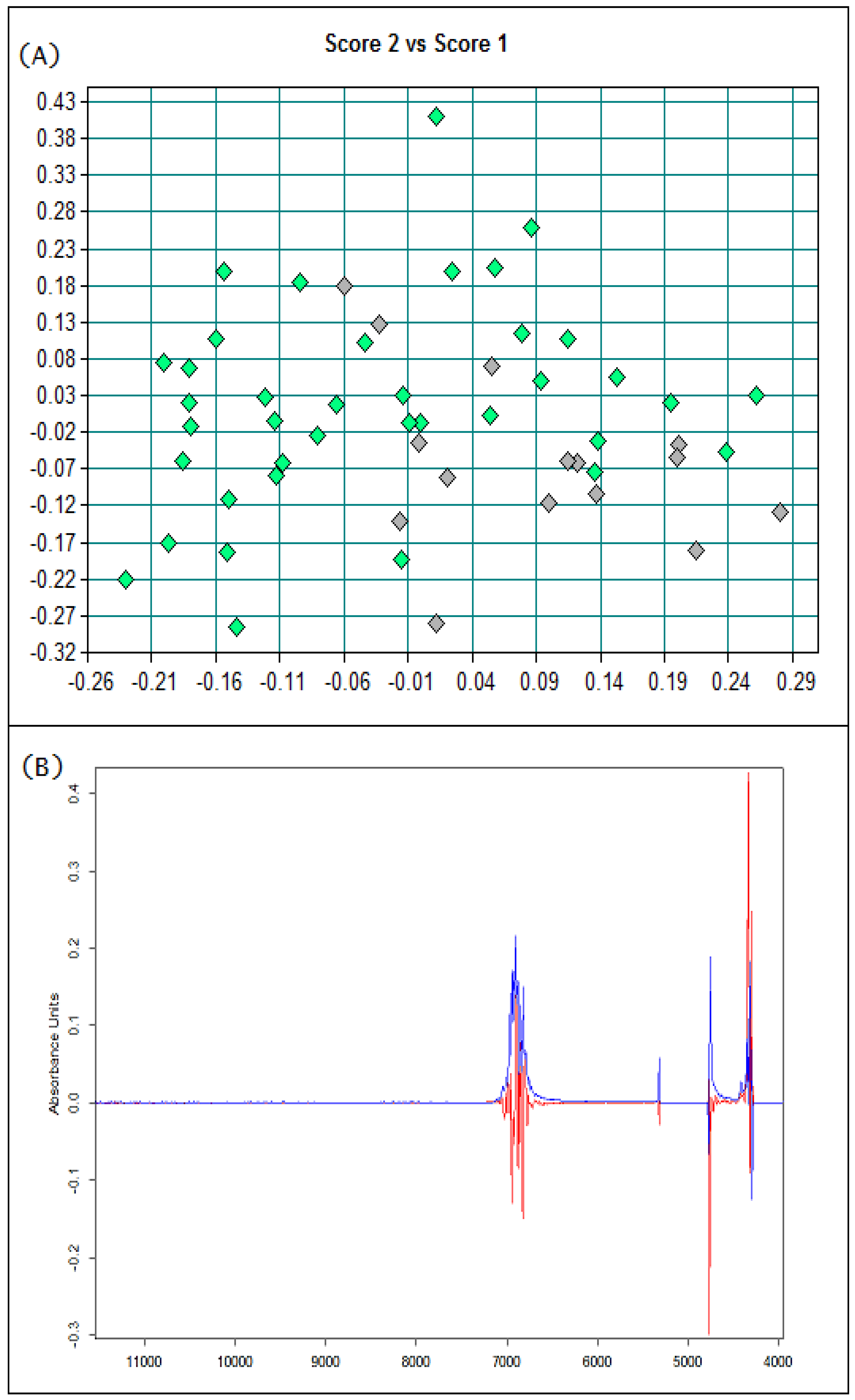
3.1. Basic Parameters and NIR Analysis
3.2. Sensory Analysis
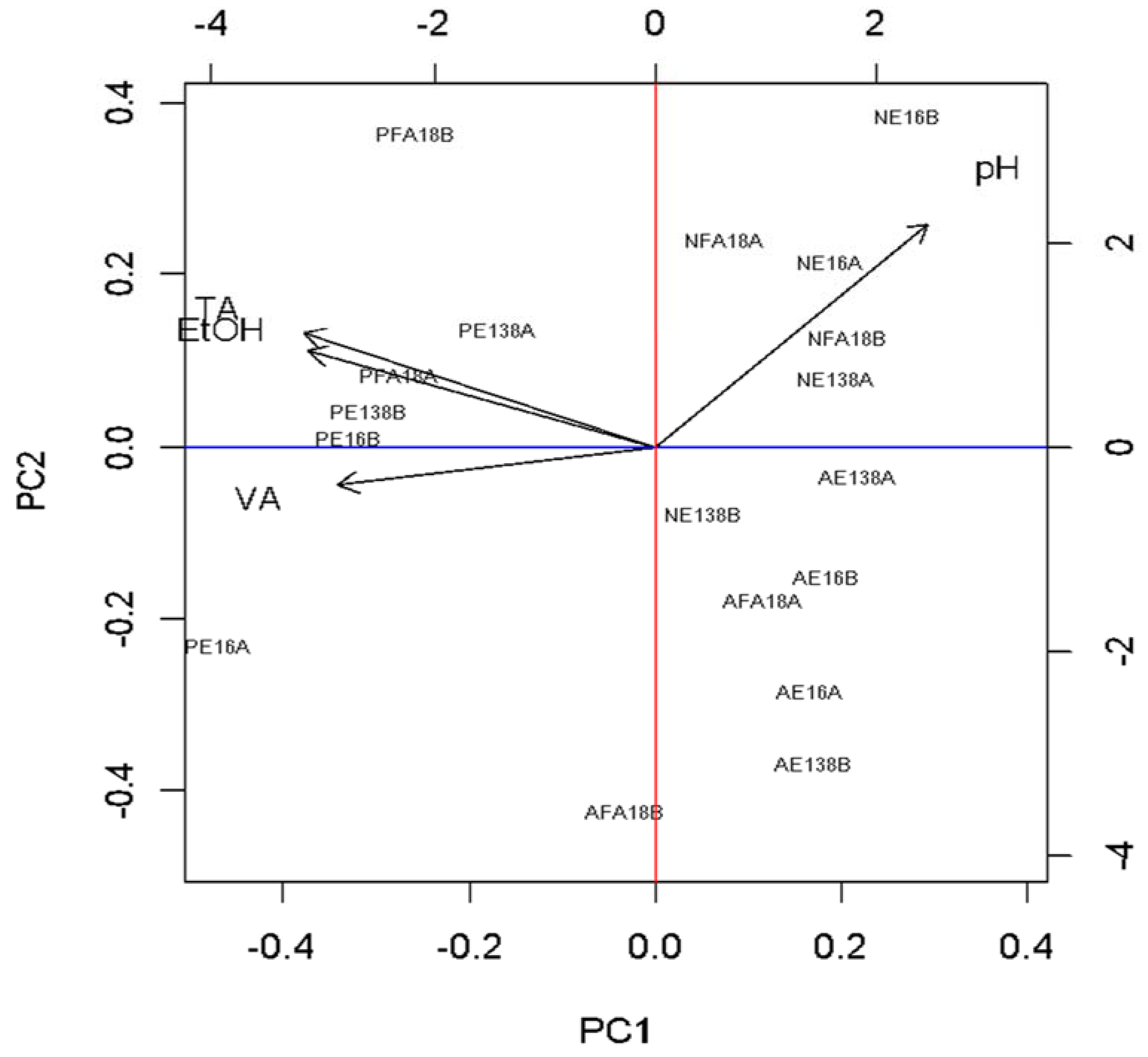
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Vitiviniculture Situation. OIV Statistical Report on World Vitiviniculture. 2017. Available online: http://www.oiv.int/public/medias/5479/oiv-en-bilan-2017.pdf (accessed on 3 September 2018).
- Fleet, G.H. Yeast interactions and wine flavour. Int. J. Food Microb. 2003, 86, 11–22. [Google Scholar] [CrossRef]
- Ciani, M.; Capece, A.; Comitini, F.; Canonico, L.; Siesto, G.; Romano, P. Yeast interactions in inoculated wine fermentation. Front. Microbiol. 2016, 7, 555. [Google Scholar] [CrossRef] [PubMed]
- Comitini, F.; Gobbi, M.; Domizio, P.; Romani, C.; Lencioni, L.; Mannazzu, I.; Ciani, M. Selected non-Saccharomyces wine yeasts in controlled multistarter fermentations with Saccharomyces cerevisiae. Food Microbiol. 2011, 28, 873–882. [Google Scholar] [CrossRef] [PubMed]
- Tofalo, R.; Schirone, M.; Torriani, S.; Rantsiou, K.; Cocolin, L.; Perpetuini, G.; Suzzi, G. Diversity of Candida zemplinina strains from grapes and Italian wines. Food Microbiol. 2012, 29, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Magyar, I.; Tóth, T. Comparative evaluation of some oenological properties in wine strains of Candida stellata, Candida zemplinina, Saccharomyces uvarum and Saccharomyces cerevisiae. Food Microbiol. 2011, 28, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Englezos, V.; Rantsiou, K.; Cravero, F.; Torchio, F.; Ortiz-Julien, A.; Gerbi, V.; Rolle, L.; Cocolin, L. Starmerella bacillaris and Saccharomyces cerevisiae mixed fermentations to reduce ethanol content in wine. Appl. Microbiol. Biotechnol. 2016, 100, 5515–5526. [Google Scholar] [CrossRef] [PubMed]
- Englezos, V.; Torchio, F.; Cravero, F.; Marengo, F.; Giacosa, S.; Gerrbi, V.; Rantsiou, K.; Rolle, L.; Cocolin, L. Aroma profile and composition of Barbera wines obtained by mixed fermentations of Starmerella bacillaris (synonym Candida zemplinina) and Saccharomyces cervisiae. LWT Food Sci. Technol. 2016, 73, 567–575. [Google Scholar] [CrossRef]
- Martelo-Vidal, M.J.; Vázquez, M. Evaluation of Ultraviolet, Visible, and Near Infrared Spectroscopy for the Analysis of Wine Compounds. Czech J. Food Sci. 2014, 32, 37–47. [Google Scholar] [CrossRef]
- Gishen, M.; Dambergs, R.G.; Cozzolino, D. Grape and wine analysis—Enhancing the power of spectroscopy with chemometrics. A review of some applications in the Australian wine industry. Aust. J. Grape Wine Res. 2005, 11, 296–305. [Google Scholar] [CrossRef]
- Cozzolino, D.; Dambergs, R.G.; Janik, L.; Cynkar, W.U.; Gishen, M. Analysis of grapes and wine by near infrared spectroscopy. J. Near Infrared Spectrosc. 2006, 14, 279–289. [Google Scholar] [CrossRef]
- López, M.G.; Sarahí García-González, A.; Franco-Robles, E. Carbohydrate Analysis by NIRS-Chemometrics. In Developments in Near-Infrared Spectroscopy; Kyprianidis, K., Ed.; IntechOpen: London, UK, 2017; pp. 81–95. Available online: https://www.intechopen.com/books/developments-in-near-infrared-spectroscopy/carbohydrate-analysis-by-nirs-chemometrics (accessed on 3 September 2018).
- Garcia-Jares, C.M.; Médina, B. Application of multivariate calibration to the simultaneous routine determination of ethanol, glycerol, fructose, glucose and total residual sugars in botrytized-grape sweet wines by means of near-infrared reflectance spectroscopy. Fresenius J. Anal. Chem. 1997, 357, 86–91. [Google Scholar] [CrossRef]
- Di Egidio, V.; Sinelli, N.; Giovanelli, G.; Moles, A.; Casiraghi, E. NIR and MIR spectroscopy as rapid methods to monitor red wine fermentation. Eur. Food Res. Technol. 2010, 230, 947–955. [Google Scholar] [CrossRef]
- ISO 3591. Sensory Analysis—Apparatus—Wine-Tasting Glass; International Organization for Standardization: Geneva, Switzerland, 1977. [Google Scholar]
- ISO 6658. Sensory Analysis—Methodology—General Guidance; International Organization for Standardization: Geneva, Switzerland, 2005. [Google Scholar]
- ISO 8598. Sensory Analysis—General Guidance for the Design of Test Rooms; International Organization for Standardization: Geneva, Switzerland, 2007. [Google Scholar]
- ISO 4121. Sensory Analysis—Guidelines for the Use of Quantitative Response Scales; International Organization for Standardization: Geneva, Switzerland, 2003. [Google Scholar]
- Meilgaard, M.; Civille, G.V.; Carr, B.T. Sensory Evaluation Techniques, 3rd ed.; CRC Press: Boca Raton, FL, USA, 1999; ISBN 0849302765. [Google Scholar]
- Conzen, J.P. Multivariate Calibration. A Practical Guide for Developing Methods in the Quantitative Analytical Chemistry, 3rd ed.; Bruker Optik GmbH: Ettlingen, Germany, 2014; ISBN 978-3-929431-13-1. [Google Scholar]
- Workman, J., Jr.; Weyer, L. Chapter 6 Water. In Practical Guide and Spectral Atlas for Interpretive Near-Infrared Spectroscopy, 2nd ed.; CRC Press Taylor and Francis Group: Boca Raton, FL, USA, 2012; pp. 55–63. ISBN 9781439875254. [Google Scholar]
- Queji, M.D.; Wosiacki, G.; Cordeiro, G.A.; Peralta-Zamora, P.G.; Nagata, N. Determination of simple sugars, malic acid and total phenolic compounds in apple pomace by infrared spectroscopy and PLSR. Int. J. Food Sci. Technol. 2010, 45, 602–609. [Google Scholar] [CrossRef] [Green Version]
- Masneuf-Pomarede, I.; Juquin, E.; Miot-Sertier, C.; Renault, P.; Laizet, Y.; Salin, F.; Alexandre, H.; Capozzi, V.; Cocolin, L.; Colonna-Ceccaldi, B.; et al. The yeast Starmerella bacillaris (synonym Candida zemplinina) shows high genetic diversity in winemaking environments. FEMS Yeast Res. 2015, 15, fov045. [Google Scholar] [CrossRef] [PubMed]
- Englezos, V.; Giacosa, S.; Rantsiou, K.; Rolle, L.; Cocolin, L. Starmerella bacillaris in winemaking: Opportunities and risks. Curr. Opin. Food Sci. 2017, 17, 30–35. [Google Scholar] [CrossRef]
- Lawless, H.T.; Heymann, H. Sensory Evaluation of Food: Principles and Practices, 2nd ed.; Springer: New York, NY, USA, 2010. [Google Scholar]
- Varela, P.; Ares, G. Sensory profiling, the blurred line between sensory and consumer science. A review of novel methods for product characterization. Food Res. Int. 2012, 48, 893–908. [Google Scholar] [CrossRef]
| Samples | Alcohol (%Vol) | Volatile Acidity † (g/L) | pH | Titratable Acidity ‡ (g/L) |
|---|---|---|---|---|
| Primitivo | ||||
| E16A | 15.4 | 0.54 ± 0.01 | 3.10 ± 0.01 | 7.71 ± 0.03 |
| E16B | 15.6 | 0.42 ± 0.01 | 3.14 ± 0.02 | 7.90 ± 0.02 |
| E138A | 14.4 | 0.41 ± 0.00 | 3.23 ± 0.02 | 7.5 ± 0.01 |
| E138B | 15.3 | 0.44 ± 0.00 | 3.17 ± 0.02 | 7.75 ± 0.02 |
| FA18A | 15.1 | 0.45 ± 0.00 | 3.20 ± 0.02 | 7.60 ± 0.02 |
| FA18B | 15.3 | 0.47 ± 0.00 | 3.29 ± 0.01 | 7.73 ± 0.05 |
| Negroamaro | ||||
| E16A | 12.5 | 0.36 ± 0.00 | 3.36 ± 0.01 | 6.10 ± 0.02 |
| E16B | 12.1 | 0.35 ± 0.00 | 3.43 ± 0.01 | 6.02 ± 0.03 |
| E138A | 12.4 | 0.33 ± 0.00 | 3.31 ± 0.01 | 6.06 ± 0.03 |
| E138B | 12.6 | 0.42 ± 0.01 | 3.27 ± 0.01 | 6.12 ± 0.05 |
| FA18A | 12.2 | 0.44 ± 0.01 | 3.37 ± 0.01 | 6.62 ± 0.03 |
| FA18B | 12.3 | 0.32 ± 0.00 | 3.32 ± 0.01 | 6.19 ± 0.00 |
| Aleatico nero | ||||
| E16A | 12.1 | 0.35 ± 0.00 | 3.22 ± 0.01 | 5.59 ± 0.06 |
| E16B | 11.8 | 0.38 ± 0.01 | 3.28 ± 0.01 | 5.61 ± 0.03 |
| E138A | 11.9 | 0.36 ± 0.00 | 3.31 ± 0.01 | 5.67 ± 0.05 |
| E138B | 11.9 | 0.35 ± 0.00 | 3.20 ± 0.00 | 5.50 ± 0.02 |
| FA18A | 11.6 | 0.39 ± 0.01 | 3.25 ± 0.01 | 6.15 ± 0.06 |
| FA18B | 11.7 | 0.47 ± 0.02 | 3.18 ± 0.02 | 6.21 ± 0.03 |
| Attributes | FA18A | E138B | E16B | E16A | E138A | FA18B | Significance |
|---|---|---|---|---|---|---|---|
| Colour Intensity | 6 | 7 | 6 | 4 | 6 | 6 | n.s. |
| Fruity (Fresh) | 1b | 4ab | 4ab | 6ab | 4ab | 0b | ** |
| Fruity (Mature) | 1ab | 3ab | 2a | 3ab | 3a | 0b | * |
| Floral | 1 | 2 | 2 | 2 | 1 | 0 | n.s. |
| Herbal | 1 | 2 | 1 | 2 | 1 | 0 | n.s. |
| Spicy | 1 | 4 | 3 | 2 | 2 | 0 | n.s. |
| Acidity | 7 | 7 | 7 | 6 | 6 | 7 | n.s. |
| Astringency | 7 | 5 | 4 | 4 | 5 | 3 | n.s. |
| Body | 4 | 4 | 4 | 5 | 5 | 2 | n.s. |
| Alcohol | 6 | 6 | 6 | 6 | 7 | 4 | n.s. |
| Persistency | 4 | 5 | 4 | 5 | 4 | 4 | n.s. |
| Appreciation | 2bc | 5a | 4ab | 6a | 4a | 0c | *** |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marsico, A.D.; Perniola, R.; Cardone, M.F.; Velenosi, M.; Antonacci, D.; Alba, V.; Basile, T. Study of the Influence of Different Yeast Strains on Red Wine Fermentation with NIR Spectroscopy and Principal Component Analysis. J 2018, 1, 133-147. https://doi.org/10.3390/j1010013
Marsico AD, Perniola R, Cardone MF, Velenosi M, Antonacci D, Alba V, Basile T. Study of the Influence of Different Yeast Strains on Red Wine Fermentation with NIR Spectroscopy and Principal Component Analysis. J. 2018; 1(1):133-147. https://doi.org/10.3390/j1010013
Chicago/Turabian StyleMarsico, Antonio Domenico, Rocco Perniola, Maria Francesca Cardone, Matteo Velenosi, Donato Antonacci, Vittorio Alba, and Teodora Basile. 2018. "Study of the Influence of Different Yeast Strains on Red Wine Fermentation with NIR Spectroscopy and Principal Component Analysis" J 1, no. 1: 133-147. https://doi.org/10.3390/j1010013
APA StyleMarsico, A. D., Perniola, R., Cardone, M. F., Velenosi, M., Antonacci, D., Alba, V., & Basile, T. (2018). Study of the Influence of Different Yeast Strains on Red Wine Fermentation with NIR Spectroscopy and Principal Component Analysis. J, 1(1), 133-147. https://doi.org/10.3390/j1010013