CO2-Selective Capture from Light Hydrocarbon Mixtures by Metal-Organic Frameworks: A Review
Abstract
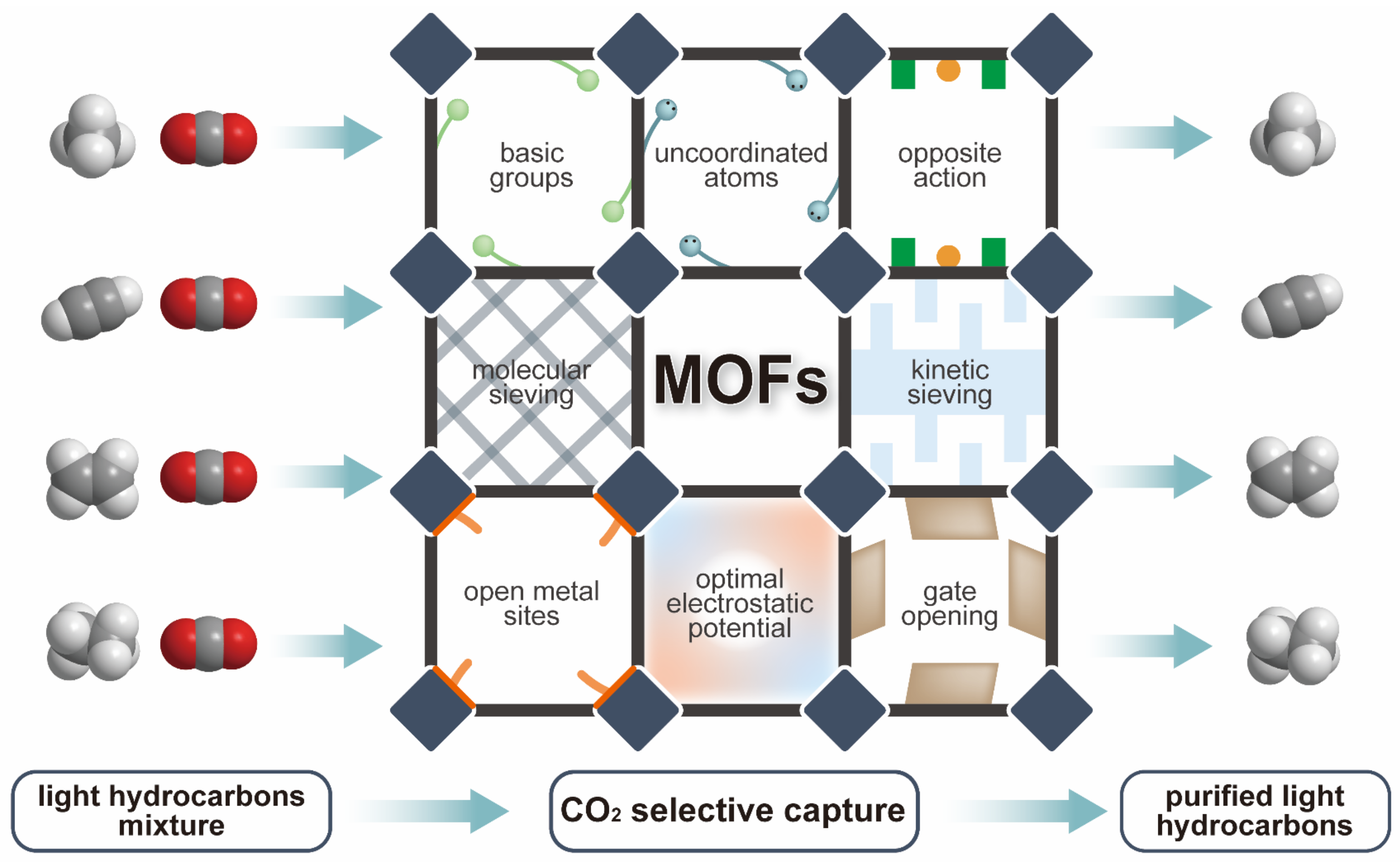
1. Introduction
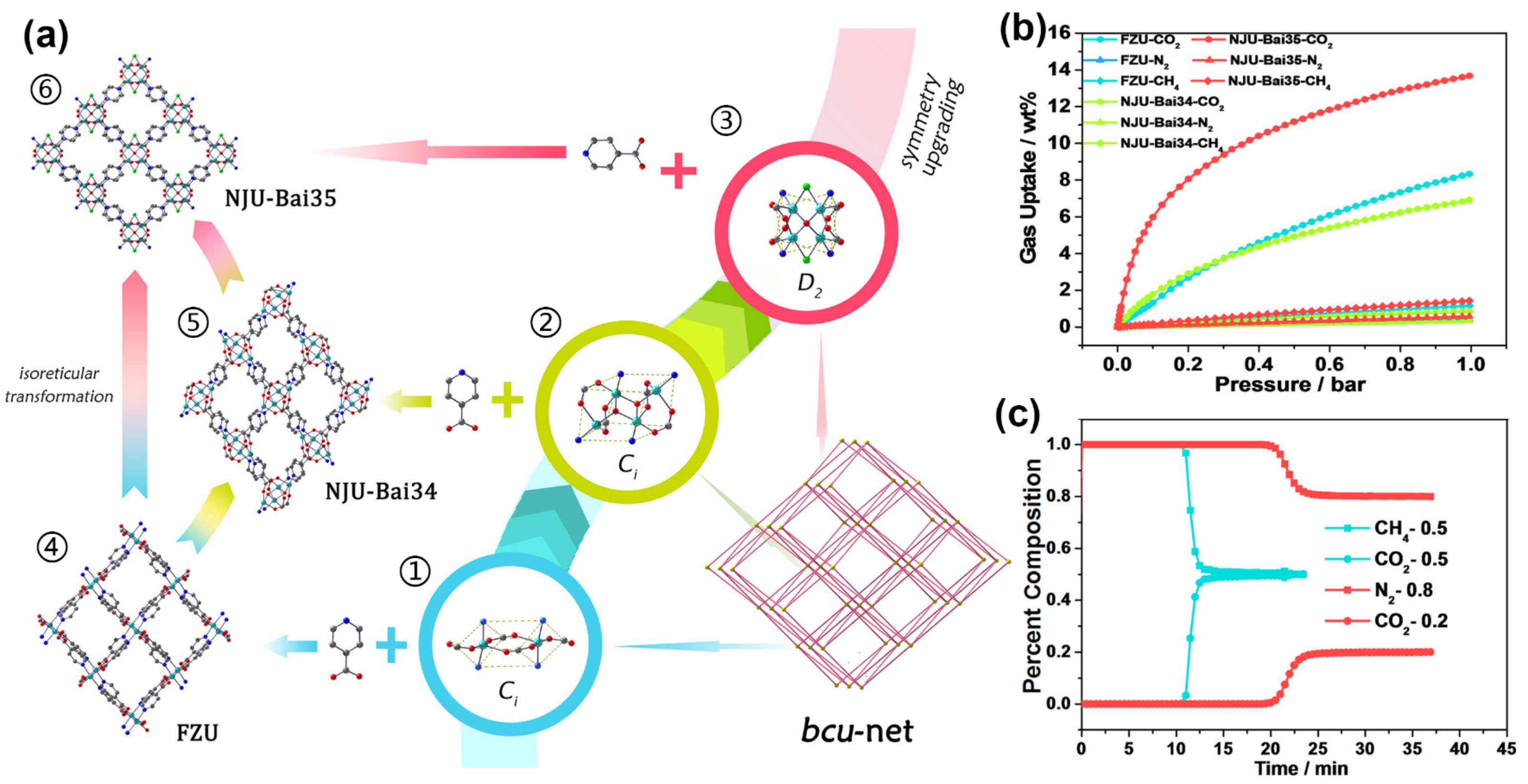
2. CO2-Selective Capture from CO2/CH4 Mixture
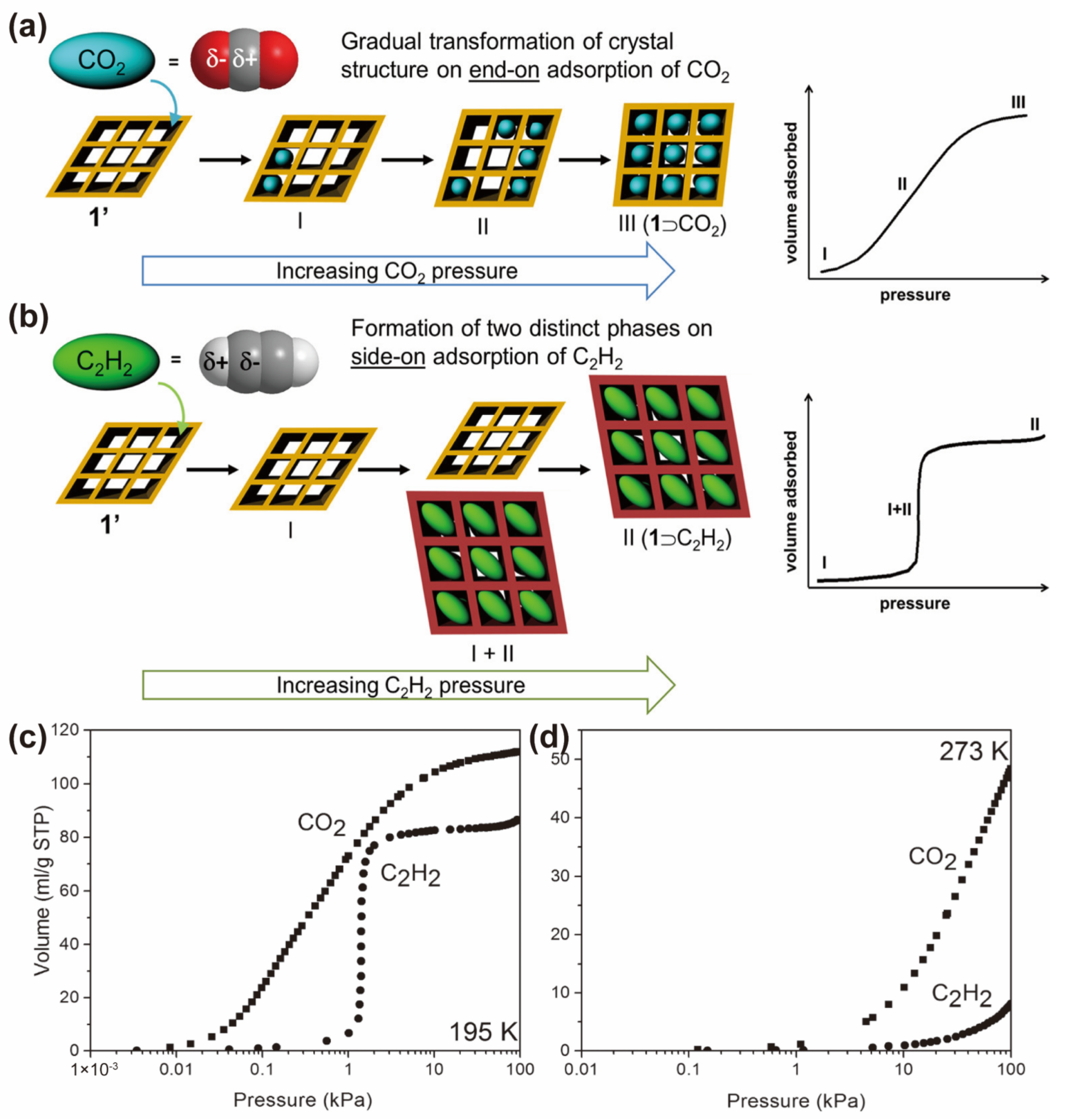
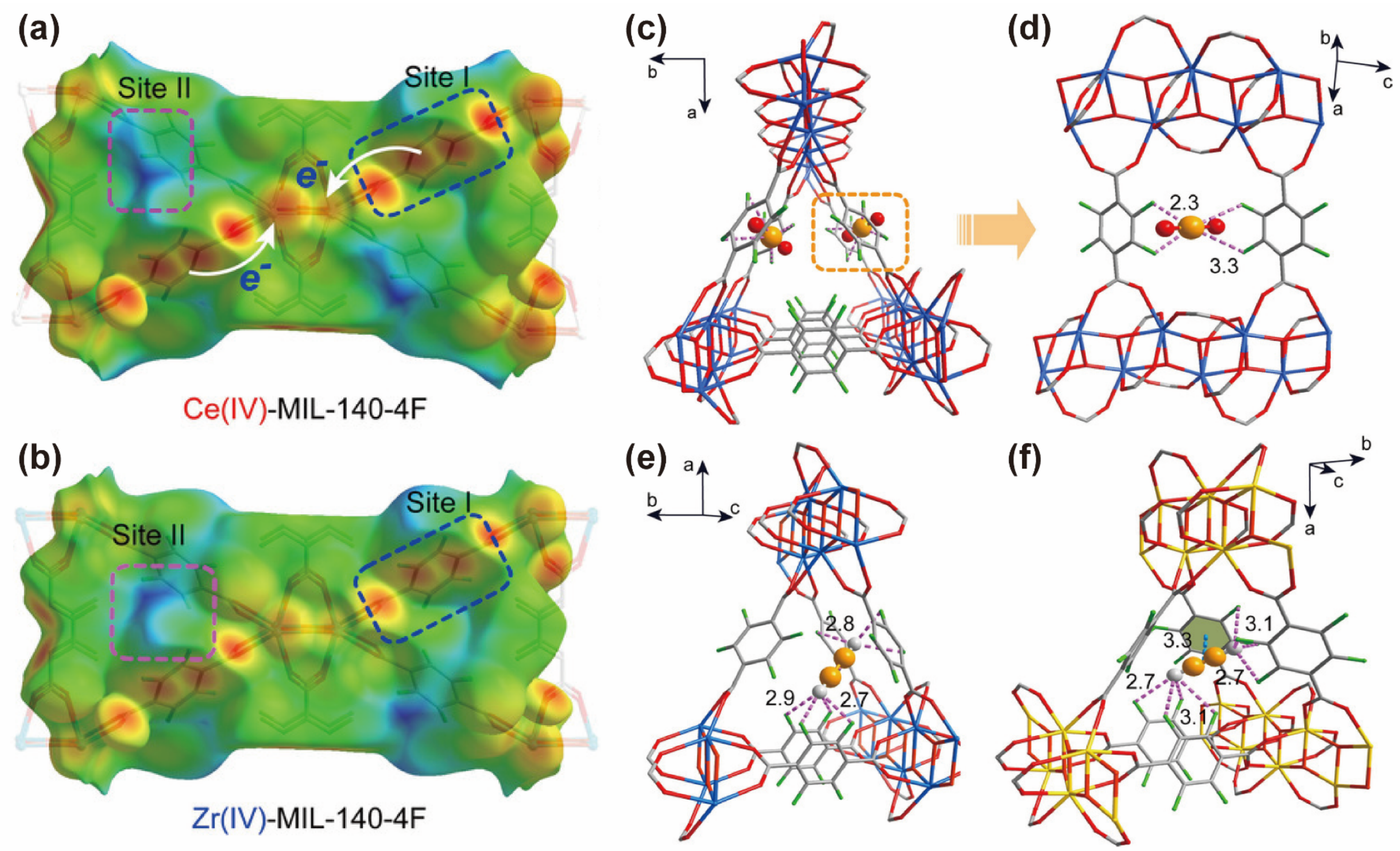
3. CO2-Selective Capture from CO2/C2H2 Mixture
4. CO2-Selective Capture from Other Binary Light Hydrocarbon Mixtures
5. CO2-Selective Capture from Multicomponent Light Hydrocarbon Mixtures
6. Outlook
- (1)
- Elimination of the trade-off between CO2 uptake capacity and selectivity. This problem is more evident in CO2 capture from CO2/C2H2 mixtures, as they are extremely close in nature. To develop optimal MOFs combining high CO2 selectivity and high capacity, in the future, researchers should focus on (a) designing flexible MOFs that can transform structures to accommodate target gas to substantially improve selectivity; (b) hyperfine control of pore size, shape and environment through reticular chemistry or crystal engineering strategies; and (c) developing MOF composites or grading combination strategies to compensate for the shortcomings of individual pristine MOFs.
- (2)
- CO2-selective capture at trace concentrations. The CO2/hydrocarbon ratios used for experiments in most studies are 1/1, 1/2 or 1/9, whereas the initial concentration of CO2 in real hydrocarbon mixtures is typically much lower (<5%). It is extremely challenging to maintain superior selective adsorption for CO2 at such low concentrations. In this regard, flexible MOFs with gate-opening effects for CO2 at low pressure are highly promising.
- (3)
- CO2 selective adsorption from multicomponent hydrocarbon mixtures. Porous materials capable of recognizing CO2 in more complex systems with high selectivity and specificity are extremely challenging but sought-after for material design. A grading combination of multiple MOFs or integration of pores with multiple properties into one MOF platform may be effective approaches.
- (4)
- Combination of desirable separation performance with broader performance for practical applications, such as high thermal/water/mechanical stability and low regeneration energy. Compared to conventional porous materials, MOF materials have more suitable ultra-microporous-level channels for CO2 molecules, high designability and the potential to capture CO2 from more complex systems. However, many MOFs are not as stable as porous carbon materials in terms of moisture stability and thermal stability. The introduction of open metal sites should be avoided, owing to their typically high activation temperature, regeneration energy consumption and poor water stability. An increasing number of researchers are employing strategies such as the construction of robust coordination geometries to build stable MOF materials.
- (5)
- Further reduction in economic and energy costs (from precursors, solvents, synthesis temperatures, activation conditions, etc.). Unaffordable raw materials and severe synthesis or activation conditions have become one of the most challenging issues in scaling MOFs up for practical applications.
- (6)
- Large-scale synthesis and industrialization. Taking technology from the laboratory to industrialization is always a challenge. A good adsorbent material should be synthesized on a large scale with high purity; however, this is rarely achieved in MOF studies. Making use of non-toxic metal ions, low-cost sustainable organic linkers and solvent-recoverable or solvent-free reactions may be beneficial in industrialization.
- (7)
- Introducing the design strategy of MOFs into other porous materials and other gas separation applications. For instance, in reticular chemistry, COFs and HOFs feature organic frameworks similar to that of MOFs and can therefore be developed by introducing design strategies for MOFs. On the other hand, air is a complex gas mixture with a CO2 concentration of only 0.04%. If MOFs for CO2 capture from light hydrocarbons can be extended to direct CO2 capture from air, considerable ecological and economic benefits can be realized.
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shi, X.; Xiao, H.; Azarabadi, H.; Song, J.; Wu, X.; Chen, X.; Lackner, K.S. Sorbents for the Direct Capture of CO2 from Ambient Air. Angew. Chem. Int. Ed. 2020, 59, 6984–7006. [Google Scholar] [CrossRef] [PubMed]
- Creamer, A.E.; Gao, B. Carbon-Based Adsorbents for Postcombustion CO2 Capture: A Critical Review. Environ. Sci. Technol. 2016, 50, 7276–7289. [Google Scholar] [CrossRef] [PubMed]
- Project, G.C. Annual CO2 Emissions. Available online: http://www.globalcarbonatlas.org/en/CO2-emissions (accessed on 15 September 2022).
- Pachauri, R.K.; Allen, M.R.; Barros, V.R.; Broome, J.; Cramer, W.; Christ, R.; Church, J.A.; Clarke, L.; Dahe, Q.; Dasgupta, P. Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; IPCC: Geneva, Switzerland, 2014. [Google Scholar]
- Bui, M.; Adjiman, C.S.; Bardow, A.; Anthony, E.J.; Boston, A.; Brown, S.; Fennell, P.S.; Fuss, S.; Galindo, A.; Hackett, L.A.; et al. Carbon capture and storage (CCS): The way forward. Energy Environ. Sci. 2018, 11, 1062–1176. [Google Scholar] [CrossRef]
- Yan, J.; Zhang, Z. Carbon Capture, Utilization and Storage (CCUS). Appl. Energy 2019, 235, 1289–1299. [Google Scholar] [CrossRef]
- Sumida, K.; Rogow, D.L.; Mason, J.A.; McDonald, T.M.; Bloch, E.D.; Herm, Z.R.; Bae, T.-H.; Long, J.R. Carbon Dioxide Capture in Metal-Organic Frameworks. Chem. Rev. 2012, 112, 724–781. [Google Scholar] [CrossRef]
- Sun, Y.; Tang, J.; Li, G.; Hua, Y.; Sun, Y.; Hu, S.; Wen, X. Adsorption, separation and regeneration of cation-exchanged X zeolites for LNG purification: Li+, K+, Mg2+ and Ca2+. Microporous Mesoporous Mater. 2022, 340, 112032. [Google Scholar] [CrossRef]
- Wang, S.; Li, X.; Wu, H.; Tian, Z.; Xin, Q.; He, G.; Peng, D.; Chen, S.; Yin, Y.; Jiang, Z.; et al. Advances in high permeability polymer-based membrane materials for CO2 separations. Energy Environ. Sci. 2016, 9, 1863–1890. [Google Scholar] [CrossRef]
- Pässler, P.; Hefner, W.; Buckl, K.; Meinass, H.; Meiswinkel, A.; Wernicke, H.-J.; Ebersberg, G.; Müller, R.; Bässler, J.; Behringer, H.; et al. Acetylene. In Ullmann’s Encyclopedia of Industrial Chemistry; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2011. [Google Scholar] [CrossRef]
- Kitagawa, S. Porous Materials and the Age of Gas. Angew. Chem. Int. Ed. 2015, 54, 10686–10687. [Google Scholar] [CrossRef]
- Göttlicher, G.; Pruschek, R. Comparison of CO2 removal systems for fossil-fuelled power plant processes. Energy Convers. Manage. 1997, 38, S173–S178. [Google Scholar] [CrossRef]
- Takht Ravanchi, M.; Sahebdelfar, S. Carbon dioxide capture and utilization in petrochemical industry: Potentials and challenges. Appl. Petrochem. Res. 2014, 4, 63–77. [Google Scholar] [CrossRef]
- Rochelle, G.T. Amine Scrubbing for CO2 Capture. Science 2009, 325, 1652–1654. [Google Scholar] [CrossRef]
- Yang, R.T. Gas Separation by Adsorption Processes; Imperial College Press; World Scientific Publishing Co.: London, UK, 1997. [Google Scholar]
- Zeng, H.; Qu, X.; Xu, D.; Luo, Y. Porous Adsorption Materials for Carbon Dioxide Capture in Industrial Flue Gas. Front. Chem. 2022, 10, 939701. [Google Scholar] [CrossRef]
- Jahandar Lashaki, M.; Khiavi, S.; Sayari, A. Stability of amine-functionalized CO2 adsorbents: A multifaceted puzzle. Chem. Soc. Rev. 2019, 48, 3320–3405. [Google Scholar] [CrossRef]
- Sreenivasulu, B.; Sreedhar, I.; Suresh, P.; Raghavan, K.V. Development Trends in Porous Adsorbents for Carbon Capture. Environ. Sci. Technol. 2015, 49, 12641–12661. [Google Scholar] [CrossRef]
- Silva, F.A.D.; Rodrigues, A.E. Propylene/propane separation by vacuum swing adsorption using 13X zeolite. AlChE J. 2001, 47, 341–357. [Google Scholar] [CrossRef]
- Grande, C.A.; Poplow, F.; Rodrigues, A.E. Vacuum Pressure Swing Adsorption to Produce Polymer-Grade Propylene. Sep. Sci. Technol. 2010, 45, 1252–1259. [Google Scholar] [CrossRef]
- Krishna, R. Methodologies for evaluation of metal-organic frameworks in separation applications. RSC Adv. 2015, 5, 52269–52295. [Google Scholar] [CrossRef]
- Mersmann, A.; Fill, B.; Hartmann, R.; Maurer, S. The Potential of Energy Saving by Gas-Phase Adsorption Processes. Chem. Eng. Technol. 2000, 23, 937–944. [Google Scholar] [CrossRef]
- Li, L.; Lin, R.-B.; Krishna, R.; Li, H.; Xiang, S.; Wu, H.; Li, J.; Zhou, W.; Chen, B. Ethane/ethylene separation in a metal-organic framework with iron-peroxo sites. Science 2018, 362, 443–446. [Google Scholar] [CrossRef]
- Yang, L.; Qian, S.; Wang, X.; Cui, X.; Chen, B.; Xing, H. Energy-efficient separation alternatives: Metal-organic frameworks and membranes for hydrocarbon separation. Chem. Soc. Rev. 2020, 49, 5359–5406. [Google Scholar] [CrossRef]
- Yang, S.-Q.; Hu, T.-L. Reverse-selective metal–organic framework materials for the efficient separation and purification of light hydrocarbons. Coord. Chem. Rev. 2022, 468, 214628. [Google Scholar] [CrossRef]
- Burns, T.D.; Pai, K.N.; Subraveti, S.G.; Collins, S.P.; Krykunov, M.; Rajendran, A.; Woo, T.K. Prediction of MOF Performance in Vacuum Swing Adsorption Systems for Postcombustion CO2 Capture Based on Integrated Molecular Simulations, Process Optimizations, and Machine Learning Models. Environ. Sci. Technol. 2020, 54, 4536–4544. [Google Scholar] [CrossRef] [PubMed]
- Usman, M.; Iqbal, N.; Noor, T.; Zaman, N.; Asghar, A.; Abdelnaby, M.M.; Galadima, A.; Helal, A. Advanced Strategies in Metal-Organic Frameworks for CO2 Capture and Separation. Chem. Rec. 2022, 22, e202100230. [Google Scholar] [CrossRef] [PubMed]
- Liang, W.; Bhatt, P.M.; Shkurenko, A.; Adil, K.; Mouchaham, G.; Aggarwal, H.; Mallick, A.; Jamal, A.; Belmabkhout, Y.; Eddaoudi, M. A Tailor-Made Interpenetrated MOF with Exceptional Carbon-Capture Performance from Flue Gas. Chem 2019, 5, 950–963. [Google Scholar] [CrossRef]
- Schneemann, A.; Bon, V.; Schwedler, I.; Senkovska, I.; Kaskel, S.; Fischer, R.A. Flexible metal-organic frameworks. Chem. Soc. Rev. 2014, 43, 6062–6096. [Google Scholar] [CrossRef]
- Wang, Q.; Luo, J.; Zhong, Z.; Borgna, A. CO2 capture by solid adsorbents and their applications: Current status and new trends. Energy Environ. Sci. 2011, 4, 42–55. [Google Scholar] [CrossRef]
- Ding, M.; Flaig, R.W.; Jiang, H.-L.; Yaghi, O.M. Carbon capture and conversion using metal-organic frameworks and MOF-based materials. Chem. Soc. Rev. 2019, 48, 2783–2828. [Google Scholar] [CrossRef]
- Sahoo, R.; Mondal, S.; Mukherjee, D.; Das, M.C. Metal-Organic Frameworks for CO2 Separation from Flue and Biogas Mixtures. Adv. Funct. Mater. 2022, 32, 2207197. [Google Scholar] [CrossRef]
- Bastin, L.; Bárcia, P.S.; Hurtado, E.J.; Silva, J.A.C.; Rodrigues, A.E.; Chen, B. A Microporous Metal−Organic Framework for Separation of CO2/N2 and CO2/CH4 by Fixed-Bed Adsorption. J. Phys. Chem. C 2008, 112, 1575–1581. [Google Scholar] [CrossRef]
- Bárcia, P.S.; Bastin, L.; Hurtado, E.J.; Silva, J.A.C.; Rodrigues, A.E.; Chen, B. Single and Multicomponent Sorption of CO2, CH4 and N2 in a Microporous Metal-Organic Framework. Sep. Sci. Technol. 2008, 43, 3494–3521. [Google Scholar] [CrossRef]
- Banerjee, R.; Furukawa, H.; Britt, D.; Knobler, C.; O’Keeffe, M.; Yaghi, O.M. Control of Pore Size and Functionality in Isoreticular Zeolitic Imidazolate Frameworks and their Carbon Dioxide Selective Capture Properties. J. Am. Chem. Soc. 2009, 131, 3875–3877. [Google Scholar] [CrossRef]
- Bao, Z.; Yu, L.; Ren, Q.; Lu, X.; Deng, S. Adsorption of CO2 and CH4 on a magnesium-based metal organic framework. J. Colloid Interface Sci. 2011, 353, 549–556. [Google Scholar] [CrossRef]
- Lin, R.-B.; Chen, D.; Lin, Y.-Y.; Zhang, J.-P.; Chen, X.-M. A Zeolite-Like Zinc Triazolate Framework with High Gas Adsorption and Separation Performance. Inorg. Chem. 2012, 51, 9950–9955. [Google Scholar] [CrossRef]
- Burd, S.D.; Ma, S.; Perman, J.A.; Sikora, B.J.; Snurr, R.Q.; Thallapally, P.K.; Tian, J.; Wojtas, L.; Zaworotko, M.J. Highly Selective Carbon Dioxide Uptake by [Cu(bpy-n)2(SiF6)] (bpy-1 = 4,4′-Bipyridine; bpy-2 = 1,2-Bis(4-pyridyl)ethene). J. Am. Chem. Soc. 2012, 134, 3663–3666. [Google Scholar] [CrossRef]
- Nugent, P.; Belmabkhout, Y.; Burd, S.D.; Cairns, A.J.; Luebke, R.; Forrest, K.; Pham, T.; Ma, S.; Space, B.; Wojtas, L.; et al. Porous materials with optimal adsorption thermodynamics and kinetics for CO2 separation. Nature 2013, 495, 80–84. [Google Scholar] [CrossRef]
- Du, L.; Lu, Z.; Zheng, K.; Wang, J.; Zheng, X.; Pan, Y.; You, X.; Bai, J. Fine-Tuning Pore Size by Shifting Coordination Sites of Ligands and Surface Polarization of Metal-Organic Frameworks To Sharply Enhance the Selectivity for CO2. J. Am. Chem. Soc. 2013, 135, 562–565. [Google Scholar] [CrossRef]
- Yan, Q.; Lin, Y.; Kong, C.; Chen, L. Remarkable CO2/CH4 selectivity and CO2 adsorption capacity exhibited by polyamine-decorated metal–organic framework adsorbents. Chem. Commun. 2013, 49, 6873–6875. [Google Scholar] [CrossRef]
- Chen, K.-J.; Madden, D.G.; Pham, T.; Forrest, K.A.; Kumar, A.; Yang, Q.-Y.; Xue, W.; Space, B.; Perry Iv, J.J.; Zhang, J.-P.; et al. Tuning Pore Size in Square-Lattice Coordination Networks for Size-Selective Sieving of CO2. Angew. Chem. Int. Ed. 2016, 55, 10268–10272. [Google Scholar] [CrossRef]
- Jiang, M.; Li, B.; Cui, X.; Yang, Q.; Bao, Z.; Yang, Y.; Wu, H.; Zhou, W.; Chen, B.; Xing, H. Controlling Pore Shape and Size of Interpenetrated Anion-Pillared Ultramicroporous Materials Enables Molecular Sieving of CO2 Combined with Ultrahigh Uptake Capacity. ACS Appl. Mater. Interfaces 2018, 10, 16628–16635. [Google Scholar] [CrossRef]
- Jiang, J.; Lu, Z.; Zhang, M.; Duan, J.; Zhang, W.; Pan, Y.; Bai, J. Higher Symmetry Multinuclear Clusters of Metal-Organic Frameworks for Highly Selective CO2 Capture. J. Am. Chem. Soc. 2018, 140, 17825–17829. [Google Scholar] [CrossRef]
- Madden, D.G.; O’Nolan, D.; Chen, K.-J.; Hua, C.; Kumar, A.; Pham, T.; Forrest, K.A.; Space, B.; Perry, J.J.; Khraisheh, M.; et al. Highly selective CO2 removal for one-step liquefied natural gas processing by physisorbents. Chem. Commun. 2019, 55, 3219–3222. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Cui, X.; Zhang, Y.; Wang, Q.; Zhang, Z.; Suo, X.; Xing, H. Anion Pillared Metal-Organic Framework Embedded with Molecular Rotors for Size-Selective Capture of CO2 from CH4 and N2. ACS Sustain. Chem. Eng. 2019, 7, 3138–3144. [Google Scholar] [CrossRef]
- Mohan, M.; Essalhi, M.; Durette, D.; Rana, L.K.; Ayevide, F.K.; Maris, T.; Duong, A. A Rational Design of Microporous Nitrogen-Rich Lanthanide Metal-Organic Frameworks for CO2/CH4 Separation. ACS Appl. Mater. Interfaces 2020, 12, 50619–50627. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.-M.; Qi, H.-F.; Qadir, S.; Sun, T.-J.; Wei, R.; Zhao, S.-S.; Liu, X.-W.; Lai, Z.; Wang, S.-D. A Two-Dimensional Stacked Metal-Organic Framework for Ultra Highly-Efficient CO2 Sieving. Chem. Eng. J. 2022, 449, 137768. [Google Scholar] [CrossRef]
- Lin, R.-B.; Li, L.; Alsalme, A.; Chen, B. An Ultramicroporous Metal–Organic Framework for Sieving Separation of Carbon Dioxide from Methane. Small Struct. 2020, 1, 2000022. [Google Scholar] [CrossRef]
- Hu, P.; Wang, H.; Xiong, C.; Liu, H.; Han, J.; Zhou, J.; Zhao, Z.; Wang, Y.; Ji, H. Probing the Node Chemistry of a Metal–Organic Framework to Achieve Ultrahigh Hydrophobicity and Highly Efficient CO2/CH4 Separation. ACS Sustain. Chem. Eng. 2021, 9, 15897–15907. [Google Scholar] [CrossRef]
- Qazvini, O.T.; Babarao, R.; Telfer, S.G. Selective capture of carbon dioxide from hydrocarbons using a metal-organic framework. Nat. Commun. 2021, 12, 197. [Google Scholar] [CrossRef]
- Shi, Y.; Xie, Y.; Cui, H.; Alothman, Z.A.; Alduhaish, O.; Lin, R.-B.; Chen, B. An ultramicroporous metal–organic framework with dual functionalities for high sieving separation of CO2 from CH4 and N2. Chem. Eng. J. 2022, 446, 137101. [Google Scholar] [CrossRef]
- Li, L.; He, J.; Xu, W.; Zhang, K.; Xing, T.; Li, Z.; Zhen, D.; Xiong, B.; Ge, Z.; Zhang, X.; et al. High CO2 separation performance on a metal–organic framework composed of nano-cages lined with an ultra-high density of dual-side open metal sites. Mater. Adv. 2022, 3, 493–497. [Google Scholar] [CrossRef]
- Ma, L.-N.; Wang, G.-D.; Hou, L.; Zhu, Z.; Wang, Y.-Y. Efficient One-Step Purification of C1 and C2 Hydrocarbons over CO2 in a New CO2-Selective MOF with a Gate-Opening Effect. ACS Appl. Mater. Interfaces 2022, 14, 26858–26865. [Google Scholar] [CrossRef]
- Lin, R.-B.; Li, L.; Zhou, H.-L.; Wu, H.; He, C.; Li, S.; Krishna, R.; Li, J.; Zhou, W.; Chen, B. Molecular sieving of ethylene from ethane using a rigid metal–organic framework. Nat. Mater. 2018, 17, 1128–1133. [Google Scholar] [CrossRef]
- Navarro, J.A.R.; Barea, E.; Rodríguez-Diéguez, A.; Salas, J.M.; Ania, C.O.; Parra, J.B.; Masciocchi, N.; Galli, S.; Sironi, A. Guest-Induced Modification of a Magnetically Active Ultramicroporous, Gismondine-like, Copper(II) Coordination Network. J. Am. Chem. Soc. 2008, 130, 3978–3984. [Google Scholar] [CrossRef]
- Ebadi Amooghin, A.; Sanaeepur, H.; Luque, R.; Garcia, H.; Chen, B. Fluorinated metal-organic frameworks for gas separation. Chem. Soc. Rev. 2022, 51, 7427–7508. [Google Scholar] [CrossRef]
- Scott, H.S.; Shivanna, M.; Bajpai, A.; Madden, D.G.; Chen, K.-J.; Pham, T.; Forrest, K.A.; Hogan, A.; Space, B.; Perry Iv, J.J.; et al. Highly Selective Separation of C2H2 from CO2 by a New Dichromate-Based Hybrid Ultramicroporous Material. ACS Appl. Mater. Interfaces 2017, 9, 33395–33400. [Google Scholar] [CrossRef]
- Gao, J.; Qian, X.; Lin, R.-B.; Krishna, R.; Wu, H.; Zhou, W.; Chen, B. Mixed Metal–Organic Framework with Multiple Binding Sites for Efficient C2H2/CO2 Separation. Angew. Chem. Int. Ed. 2020, 59, 4396–4400. [Google Scholar] [CrossRef]
- Pei, J.; Shao, K.; Wang, J.-X.; Wen, H.-M.; Yang, Y.; Cui, Y.; Krishna, R.; Li, B.; Qian, G. A Chemically Stable Hofmann-Type Metal−Organic Framework with Sandwich-Like Binding Sites for Benchmark Acetylene Capture. Adv. Mater. 2020, 32, 1908275. [Google Scholar] [CrossRef]
- Yang, H.; Trieu, T.X.; Zhao, X.; Wang, Y.; Wang, Y.; Feng, P.; Bu, X. Lock-and-Key and Shape-Memory Effects in an Unconventional Synthetic Path to Magnesium Metal-Organic Frameworks. Angew. Chem. Int. Ed. 2019, 58, 11757–11762. [Google Scholar] [CrossRef]
- Niu, Z.; Cui, X.; Pham, T.; Verma, G.; Lan, P.C.; Shan, C.; Xing, H.; Forrest, K.A.; Suepaul, S.; Space, B.; et al. A MOF-based Ultra-Strong Acetylene Nano-trap for Highly Efficient C2H2/CO2 Separation. Angew. Chem. Int. Ed. 2021, 60, 5283–5288. [Google Scholar] [CrossRef]
- Ye, Y.; Ma, Z.; Lin, R.-B.; Krishna, R.; Zhou, W.; Lin, Q.; Zhang, Z.; Xiang, S.; Chen, B. Pore Space Partition within a Metal–Organic Framework for Highly Efficient C2H2/CO2 Separation. J. Am. Chem. Soc. 2019, 141, 4130–4136. [Google Scholar] [CrossRef]
- Peng, Y.-L.; Pham, T.; Li, P.; Wang, T.; Chen, Y.; Chen, K.-J.; Forrest, K.A.; Space, B.; Cheng, P.; Zaworotko, M.J.; et al. Robust Ultramicroporous Metal-Organic Frameworks with Benchmark Affinity for Acetylene. Angew. Chem. Int. Ed. 2018, 57, 10971–10975. [Google Scholar] [CrossRef]
- Lee, J.; Chuah, C.Y.; Kim, J.; Kim, Y.; Ko, N.; Seo, Y.; Kim, K.; Bae, T.H.; Lee, E. Separation of Acetylene from Carbon Dioxide and Ethylene by a Water-Stable Microporous Metal–Organic Framework with Aligned Imidazolium Groups inside the Channels. Angew. Chem. Int. Ed. 2018, 57, 7869–7873. [Google Scholar] [CrossRef] [PubMed]
- Luo, F.; Yan, C.; Dang, L.; Krishna, R.; Zhou, W.; Wu, H.; Dong, X.; Han, Y.; Hu, T.-L.; O’Keeffe, M.; et al. UTSA-74: A MOF-74 Isomer with Two Accessible Binding Sites per Metal Center for Highly Selective Gas Separation. J. Am. Chem. Soc. 2016, 138, 5678–5684. [Google Scholar] [CrossRef] [PubMed]
- Eguchi, R.; Uchida, S.; Mizuno, N. Inverse and High CO2/C2H2 Sorption Selectivity in Flexible Organic–Inorganic Ionic Crystals. Angew. Chem. Int. Ed. 2012, 51, 1635–1639. [Google Scholar] [CrossRef] [PubMed]
- Ma, B.; Li, D.; Zhu, Q.; Li, Y.; Ueda, W.; Zhang, Z. A Zeolitic Octahedral Metal Oxide with Ultra-Microporosity for Inverse CO2/C2H2 Separation at High Temperature and Humidity. Angew. Chem. Int. Ed. 2022, 61, e202209121. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Davies, A.J.; Lin, X.; Suyetin, M.; Matsuda, R.; Blake, A.J.; Wilson, C.; Lewis, W.; Parker, J.E.; Tang, C.C.; et al. Selective CO2 uptake and inverse CO2/C2H2 selectivity in a dynamic bifunctional metal–organic framework. Chem. Sci. 2012, 3, 2993–2999. [Google Scholar] [CrossRef]
- Foo, M.L.; Matsuda, R.; Hijikata, Y.; Krishna, R.; Sato, H.; Horike, S.; Hori, A.; Duan, J.; Sato, Y.; Kubota, Y.; et al. An Adsorbate Discriminatory Gate Effect in a Flexible Porous Coordination Polymer for Selective Adsorption of CO2 over C2H2. J. Am. Chem. Soc. 2016, 138, 3022–3030. [Google Scholar] [CrossRef]
- Chen, K.-J.; Scott, H.S.; Madden, D.G.; Pham, T.; Kumar, A.; Bajpai, A.; Lusi, M.; Forrest, K.A.; Space, B.; Perry, J.J.; et al. Benchmark C2H2/CO2 and CO2/C2H2 Separation by Two Closely Related Hybrid Ultramicroporous Materials. Chem 2016, 1, 753–765. [Google Scholar] [CrossRef]
- Li, L.; Wang, J.; Zhang, Z.; Yang, Q.; Yang, Y.; Su, B.; Bao, Z.; Ren, Q. Inverse Adsorption Separation of CO2/C2H2 Mixture in Cyclodextrin-Based Metal-Organic Frameworks. ACS Appl. Mater. Interfaces 2019, 11, 2543–2550. [Google Scholar] [CrossRef]
- Ma, D.; Li, Z.; Zhu, J.; Zhou, Y.; Chen, L.; Mai, X.; Liufu, M.; Wu, Y.; Li, Y. Inverse and highly selective separation of CO2/C2H2 on a thulium–organic framework. J. Mater. Chem. A 2020, 8, 11933–11937. [Google Scholar] [CrossRef]
- Gu, Y.; Zheng, J.-J.; Otake, K.-i.; Shivanna, M.; Sakaki, S.; Yoshino, H.; Ohba, M.; Kawaguchi, S.; Wang, Y.; Li, F.; et al. Host–Guest Interaction Modulation in Porous Coordination Polymers for Inverse Selective CO2/C2H2 Separation. Angew. Chem. Int. Ed. 2021, 60, 11688–11694. [Google Scholar] [CrossRef]
- Xie, Y.; Cui, H.; Wu, H.; Lin, R.-B.; Zhou, W.; Chen, B. Electrostatically Driven Selective Adsorption of Carbon Dioxide over Acetylene in an Ultramicroporous Material. Angew. Chem. Int. Ed. 2021, 60, 9604–9609. [Google Scholar] [CrossRef]
- Zhang, Z.; Peh, S.B.; Krishna, R.; Kang, C.; Chai, K.; Wang, Y.; Shi, D.; Zhao, D. Optimal Pore Chemistry in an Ultramicroporous Metal-Organic Framework for Benchmark Inverse CO2/C2H2 Separation. Angew. Chem. Int. Ed. 2021, 60, 17198–17204. [Google Scholar] [CrossRef]
- Shi, Y.; Xie, Y.; Cui, H.; Ye, Y.; Wu, H.; Zhou, W.; Arman, H.; Lin, R.-B.; Chen, B. Highly Selective Adsorption of Carbon Dioxide over Acetylene in an Ultramicroporous Metal–Organic Framework. Adv. Mater. 2021, 33, 2105880. [Google Scholar] [CrossRef]
- Li, X.-Y.; Song, Y.; Zhang, C.-X.; Zhao, C.-X.; He, C. Inverse CO2/C2H2 separation in a pillared-layer framework featuring a chlorine-modified channel by quadrupole-moment sieving. Sep. Purif. Technol. 2021, 279, 119608. [Google Scholar] [CrossRef]
- Cai, L.-Z.; Yao, Z.-Z.; Lin, S.-J.; Wang, M.-S.; Guo, G.-C. Photoinduced Electron-Transfer (PIET) Strategy for Selective Adsorption of CO2 over C2H2 in a MOF. Angew. Chem. Int. Ed. 2021, 60, 18223–18230. [Google Scholar] [CrossRef]
- Choi, D.S.; Kim, D.W.; Kang, D.W.; Kang, M.; Chae, Y.S.; Hong, C.S. Highly selective CO2 separation from a CO2/C2H2 mixture using a diamine-appended metal–organic framework. J. Mater. Chem. A 2021, 9, 21424–21428. [Google Scholar] [CrossRef]
- Cui, J.; Qiu, Z.; Yang, L.; Zhang, Z.; Cui, X.; Xing, H. Kinetic-Sieving of Carbon Dioxide from Acetylene through a Novel Sulfonic Ultramicroporous Material. Angew. Chem. Int. Ed. 2022, 61, e202208756. [Google Scholar] [CrossRef]
- Hao, C.; Ren, H.; Zhu, H.; Chi, Y.; Zhao, W.; Liu, X.; Guo, W. CO2-favored metal-organic frameworks SU-101(M) (M = Bi, In, Ga, and Al) with inverse and high selectivity of CO2 from C2H2 and C2H4. Sep. Purif. Technol. 2022, 290, 120804. [Google Scholar] [CrossRef]
- Gücüyener, C.; Van den Bergh, J.; Gascon, J.; Kapteijn, F. Ethane/Ethene Separation Turned on Its Head: Selective Ethane Adsorption on the Metal−Organic Framework ZIF-7 through a Gate-Opening Mechanism. J. Am. Chem. Soc. 2010, 132, 17704–17706. [Google Scholar] [CrossRef]
- Böhme, U.; Barth, B.; Paula, C.; Kuhnt, A.; Schwieger, W.; Mundstock, A.; Caro, J.; Hartmann, M. Ethene/Ethane and Propene/Propane Separation via the Olefin and Paraffin Selective Metal-Organic Framework Adsorbents CPO-27 and ZIF-8. Langmuir 2013, 29, 8592–8600. [Google Scholar] [CrossRef]
- Krause, S.; Hosono, N.; Kitagawa, S. Chemistry of Soft Porous Crystals: Structural Dynamics and Gas Adsorption Properties. Angew. Chem. Int. Ed. 2020, 59, 15325–15341. [Google Scholar] [CrossRef] [PubMed]
- Gu, C.; Hosono, N.; Zheng, J.-J.; Sato, Y.; Kusaka, S.; Sakaki, S.; Kitagawa, S. Design and control of gas diffusion process in a nanoporous soft crystal. Science 2019, 363, 387–391. [Google Scholar] [CrossRef] [PubMed]
- Behera, N.; Duan, J.; Jin, W.; Kitagawa, S. The chemistry and applications of flexible porous coordination polymers. EnergyChem 2021, 3, 100067. [Google Scholar] [CrossRef]
- Duan, J.; Higuchi, M.; Foo, M.L.; Horike, S.; Rao, K.P.; Kitagawa, S. A Family of Rare Earth Porous Coordination Polymers with Different Flexibility for CO2/C2H4 and CO2/C2H6 Separation. Inorg. Chem. 2013, 52, 8244–8249. [Google Scholar] [CrossRef]
- Kelman, S.; Lin, H.; Sanders, E.S.; Freeman, B.D. CO2/C2H6 separation using solubility selective membranes. J. Membr. Sci. 2007, 305, 57–68. [Google Scholar] [CrossRef]
- Horike, S.; Kishida, K.; Watanabe, Y.; Inubushi, Y.; Umeyama, D.; Sugimoto, M.; Fukushima, T.; Inukai, M.; Kitagawa, S. Dense Coordination Network Capable of Selective CO2 Capture from C1 and C2 Hydrocarbons. J. Am. Chem. Soc. 2012, 134, 9852–9855. [Google Scholar] [CrossRef]
- He, T.; Xiao, Y.; Zhao, Q.; Zhou, M.; He, G. Ultramicroporous Metal–Organic Framework Qc-5-Cu for Highly Selective Adsorption of CO2 from C2H4 Stream. Ind. Eng. Chem. Res. 2020, 59, 3153–3161. [Google Scholar] [CrossRef]
- Li, S.-Y.; Zhang, F.-F.; Wang, X.; He, M.-H.; Ding, Z.; Chen, M.; Hou, X.-Y.; Chen, X.-L.; Tang, L.; Yue, E.-L.; et al. Flexible ligands-dependent formation of a new column layered MOF possess 1D channel and effective separation performance for CO2. J. Solid State Chem. 2021, 294, 121896. [Google Scholar] [CrossRef]



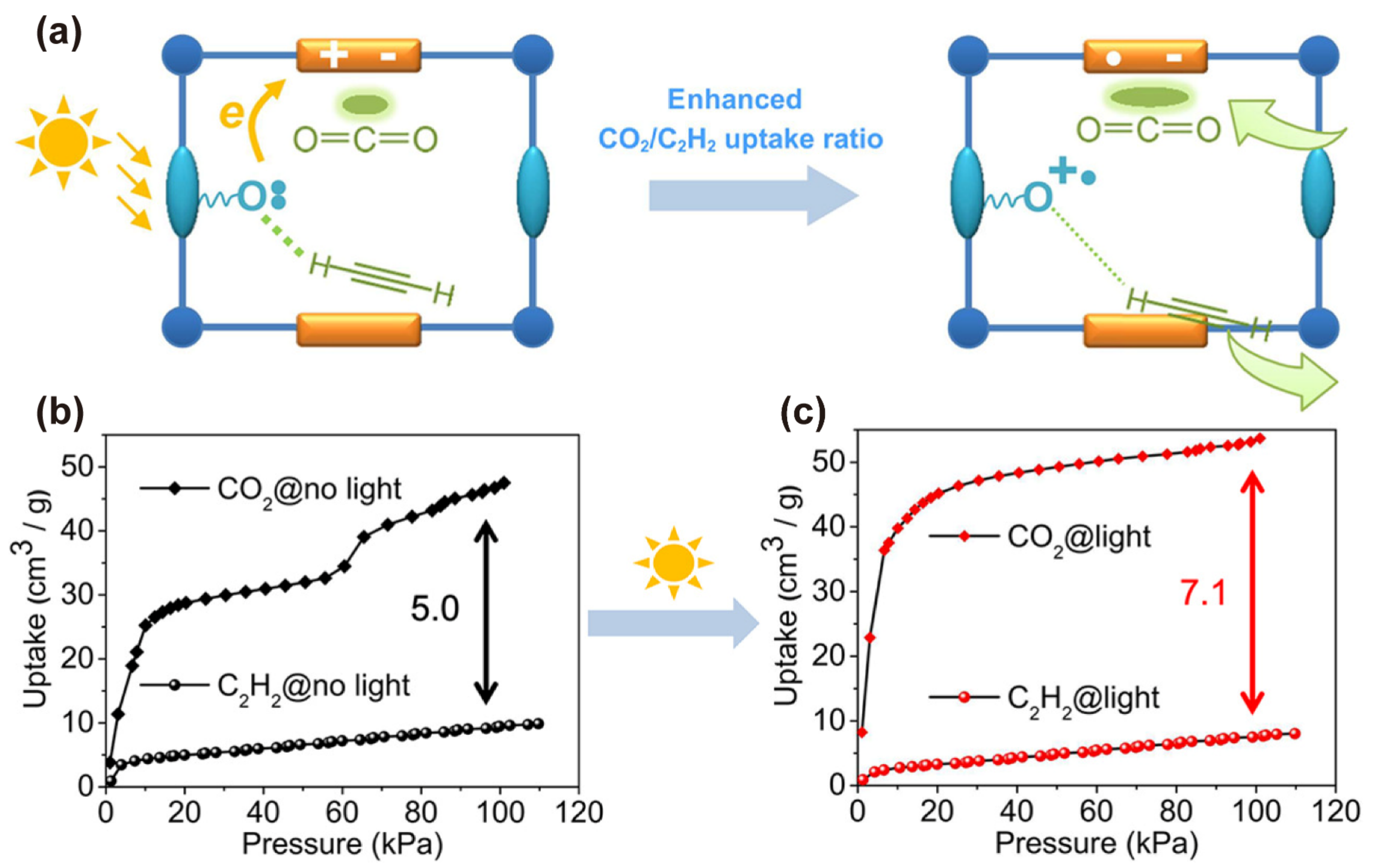
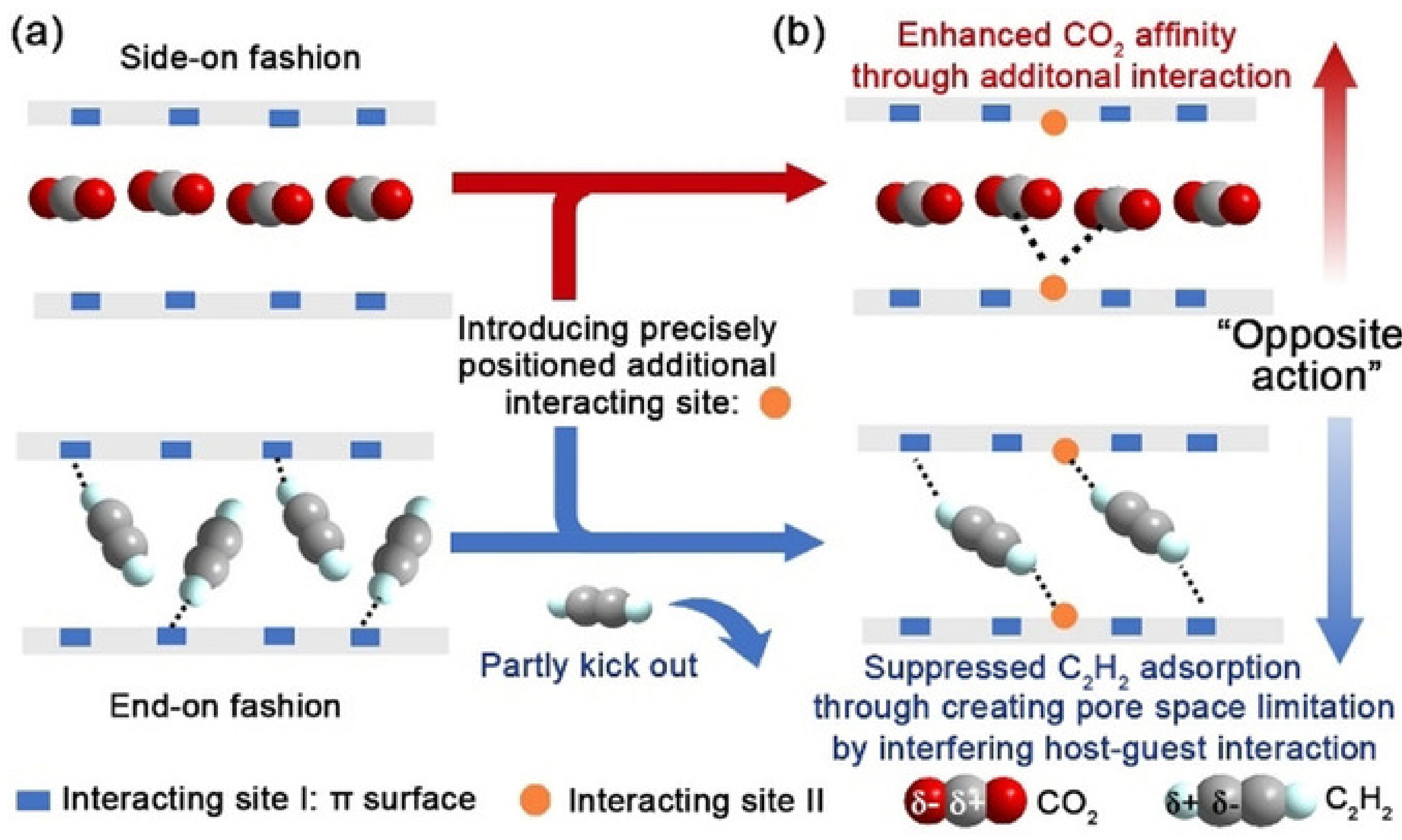
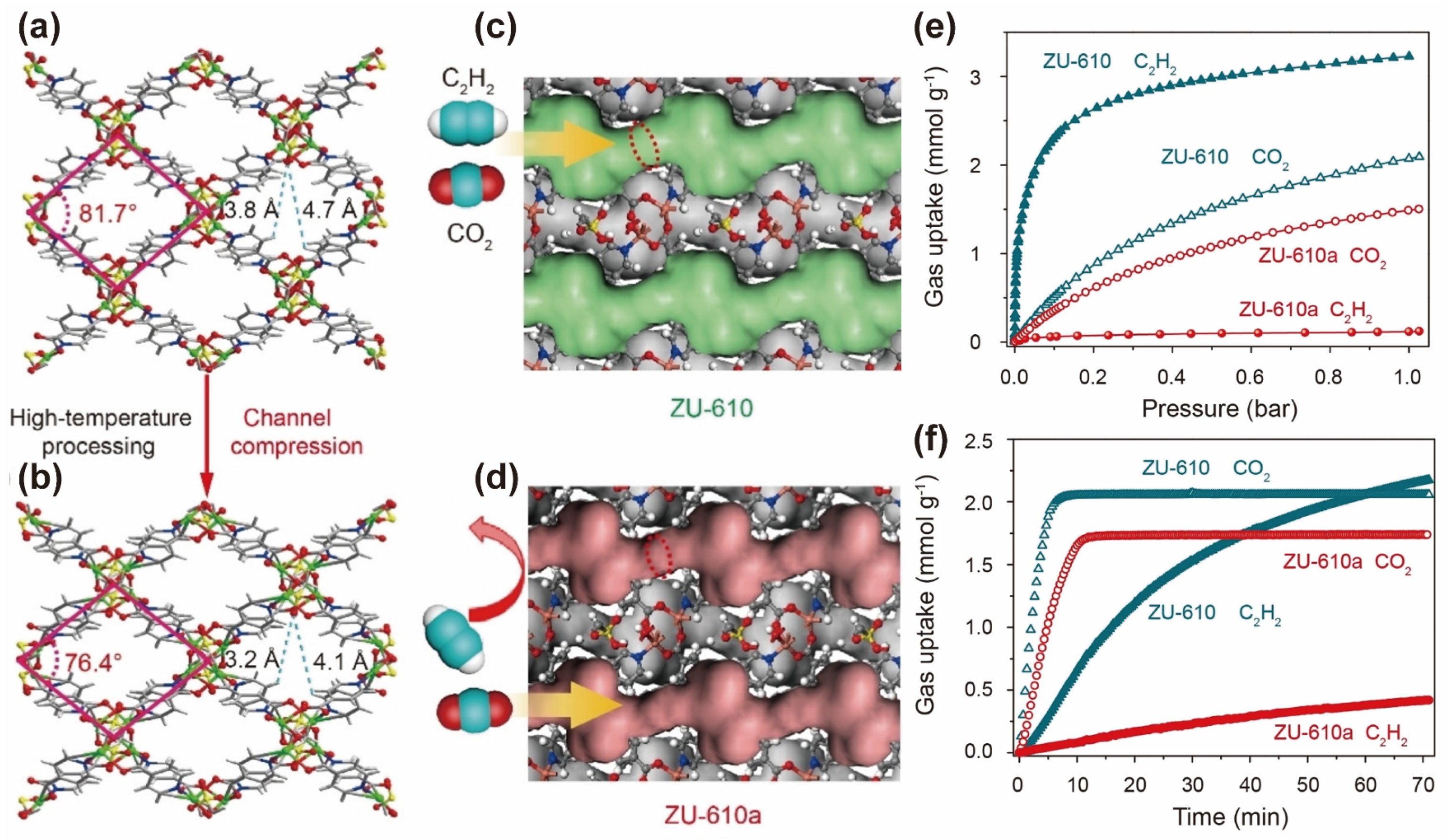
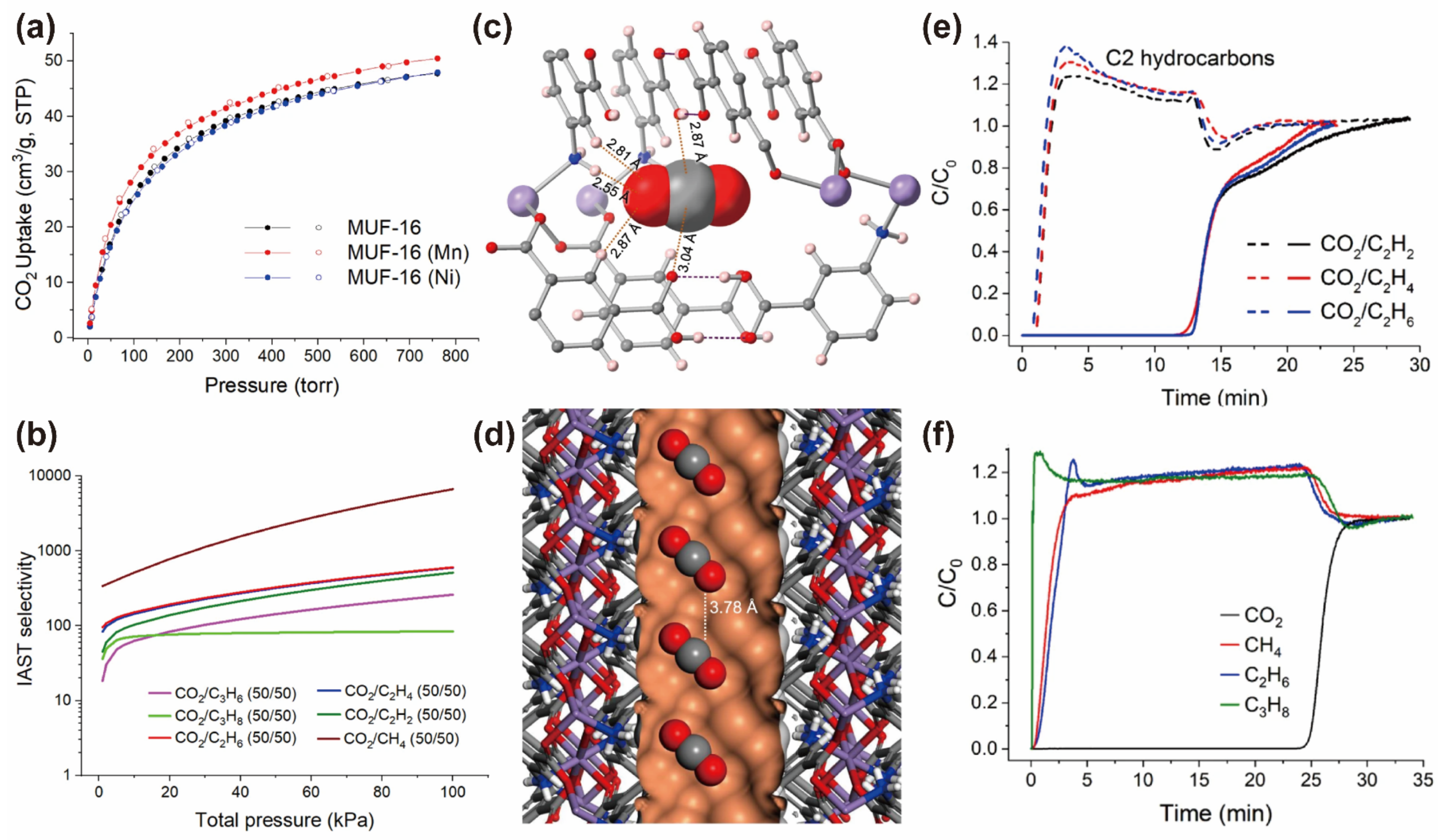
| Property | CO2 | CH4 | C2H2 | C2H4 | C2H6 |
|---|---|---|---|---|---|
| Kinetic diameter (Å) | 3.30 | 3.76 | 3.30 | 4.16 | 4.44 |
| Boiling point (K) | 194.7 | 111.6 | 189.3 | 169.4 | 184.5 |
| Polarizability (×10−25 cm3) | 29.11 | 26.0 | 33.3–39.3 | 42.52 | 44.3–44.7 |
| Quadrupole moment (×1026 esu cm2) | −4.30 | 0 | 7.50 | 1.5 | 0.65 |
| MOF | Functional Site | CO2 Capacity (mmol/g) | CO2/CH4 Selectivity | Qst for CO2 (kJ/mol) | Ref. |
|---|---|---|---|---|---|
| MOF-508b | quadrupole interactions | 1.78 a | 3–6 a | 14.9 | [33,34] |
| ZIF-78 | Dipole–quadrupole interactions | 2.32 | 10.6 b | —— | [35] |
| Mg-MOF-74 | open metal sites | 8.61 | 8 | 73 | [36] |
| MAF-66 | uncoordinated N atoms | 4.41 | 5.8 | 26 | [37] |
| SIFSIX-1-Cu | SiF62− | 5.2 | 10.5 | 27 | [38] |
| SIFSIX-2-Cu-i | SiF62− | 5.4 | 33 | 31.9 | [39] |
| SIFSIX-3-Zn | SiF62− | 2.54 | 231 | 45 | [39] |
| SYSU | narrow channels | 3.11 | 4.7 | 28.2 | [40] |
| NJU-Bai7 | narrow channels | 2.91 | 14.1 c | 40.5 | [40] |
| NJU-Bai8 | uncoordinated N atoms | 2.57 | 40.8 c | 37.7 | [40] |
| PEI-incorporated amine-MIL-101(Cr) | amine groups | 3.6 | 931 | —— | [41] |
| Qc-5-Cu-sql-β | molecular sieving | 2.16 d | 3300 | 36 | [42] |
| SIFSIX-14-Cu-i | molecular sieving | 4.71 | 46.7 e | 37.7 | [43] |
| NJU-Bai35 | molecular sieving | 3.125 | 11.6 | 33.37 | [44] |
| dptz-CuTiF6 | TiF62− | 4.52 | —— | 33.3 | [28] |
| dptz-CuSiF6 | SiF62− | 4.04 | —— | 38.2 | [28] |
| TIFSIX-3-Ni | TiF62− | 2.213 | 158 | 50.0 | [45] |
| NbOFFIVE-1-Ni | NbOF52− | 2.308 | 366 | 54.0 | [45] |
| TIFSIX-2-Cu-i | TiF62− | 4.229 | 16 | 35.8 | [45] |
| ZU-66 | molecular sieving | 4.56 | 136 | 35 | [46] |
| IRH-3 | uncoordinated N atoms | 2.7 | 27 | —— | [47] |
| In(aip)2 | molecular sieving and –NH2 groups | 1.27 | 1808 | 34.3 | [48] |
| UTSA-280 | molecular sieving | 3.00 | molecular sieving | 42.9 | [49] |
| UiO-66(N10%-Zr) | uncoordinated N atoms and kinetic effect | 2.1 | 326 | 35.7 | [50] |
| MUF-16 | N-H···O and C-H··O | 2.13 d | 6690 d | 32.3 | [51] |
| MUF-16 (Mn) | N-H···O and C-H··O | 2.25 d | 470 d | 36.6 | [51] |
| MUF-16 (Ni) | N-H···O and C-H··O | 2.13 d | 1220 d | 37.3 | [51] |
| Cu-F-pymo | molecular sieving | 1.61 f | >107 | 29.1 | [52] |
| [Cu3(μ3-OH)(PCA)3] | open metal sites | 2.93 | 15.9 | 31.5 | [53] |
| [Zn(odip)0.5(bpe)0.5] | gate opening | 5.3 | 376.0 | 42.3 | [54] |
| MOF | Functional Site | CO2 Capacity (mmol/g) | CO2/C2H2 Selectivity | Qst for CO2 (kJ/mol) | Ref. |
|---|---|---|---|---|---|
| Co(HLdc) | gate opening | 10.69 a | 1.7 a,b | —— | [69] |
| [Mn(bdc)(dpe)] | gate opening | 2.17 c | 8.8 c | 29.5 | [70] |
| SIFSIX-3-Ni | SiF62− | 2.7 | 7.69 d | 50.9 | [71] |
| CD-MOF-1 | uncoordinated primary hydroxyl groups | 2.87 | 6.6 d | 41.0 | [72] |
| CD-MOF-2 | uncoordinated primary hydroxyl groups | 2.65 | 16 d | 67.2 | [72] |
| [Tm2(OH-bdc)] (1a) | OH groups | 5.83 | 17.5 d | 45.2 | [73] |
| [Tm2(OH-bdc)] (1a′) | OH groups | 6.21 | 1.65 d | 32.7 | [73] |
| PCP-NH2-bdc | amino group | 3.03 | 4.4 | 34.57 | [74] |
| PCP-NH2-ipa | amino group | 3.21 | 6.4 | 36.6 | [74] |
| Cd-NP | electrostatic potential | 2.59 | 85 | 27.7 | [75] |
| CeIV-MIL-140-4F | electrostatic potential | 2.24 | 9.5 | 39.5 | [76] |
| Cu-F-pymo | electrostatic potential | 1.19 | 105 | 28.8 | [77] |
| [Zn(atz)(BDC-Cl4)0.5]n | electrostatic potential | 0.94 f,g | 2.4 f | 32.7 | [78] |
| PMOF-1(irra) | electrostatic potential | 2.38 c | 694 c | —— | [79] |
| MUF-16 | electrostatic potential | 2.13 e | 510 e | 32.3 | [51] |
| MUF-16(Mn) | electrostatic potential | 2.25 e | 31 e | 36.6 | [51] |
| MUF-16(Ni) | electrostatic potential | 2.13 e | 46 e | 37.3 | [51] |
| en-MOF | amine groups | 4.8 | —— | 71.2 | [80] |
| nmen-MOF | amine groups | 4.55 | —— | 62.3 | [80] |
| een-MOF | amine groups | 4.9 | —— | 68.8 | [80] |
| ZU-610a | kinetic sieving | 1.51 | 207 | 27.3 | [81] |
| SU-101(Bi) | carbonyl oxygen atoms | 2.4 | 5.5 | 30.5 | [82] |
| SU-101(Al) | carbonyl oxygen atoms | 2.37 | 15.5 | 31.3 | [82] |
| SU-101(In) | carbonyl oxygen atoms | 2.46 | 6.2 | 28.3 | [82] |
| SU-101(Ga) | carbonyl oxygen atoms | 1.79 | 11.1 | 27.7 | [82] |
| [Zn(odip)0.5(bpe)0.5] | Gate Opening | 5.3 | 13.2 | 42.3 | [54] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, H.; Wang, L.; Zhang, X.; Zhao, H.; Gu, Y. CO2-Selective Capture from Light Hydrocarbon Mixtures by Metal-Organic Frameworks: A Review. Clean Technol. 2023, 5, 1-24. https://doi.org/10.3390/cleantechnol5010001
Huang H, Wang L, Zhang X, Zhao H, Gu Y. CO2-Selective Capture from Light Hydrocarbon Mixtures by Metal-Organic Frameworks: A Review. Clean Technologies. 2023; 5(1):1-24. https://doi.org/10.3390/cleantechnol5010001
Chicago/Turabian StyleHuang, Hengcong, Luyao Wang, Xiaoyu Zhang, Hongshuo Zhao, and Yifan Gu. 2023. "CO2-Selective Capture from Light Hydrocarbon Mixtures by Metal-Organic Frameworks: A Review" Clean Technologies 5, no. 1: 1-24. https://doi.org/10.3390/cleantechnol5010001
APA StyleHuang, H., Wang, L., Zhang, X., Zhao, H., & Gu, Y. (2023). CO2-Selective Capture from Light Hydrocarbon Mixtures by Metal-Organic Frameworks: A Review. Clean Technologies, 5(1), 1-24. https://doi.org/10.3390/cleantechnol5010001






