Tooth-Derived Matrix Granules for Enhanced Bone Healing: Chemical Composition, Morphological Aspects, and Clinical Outcomes
Abstract
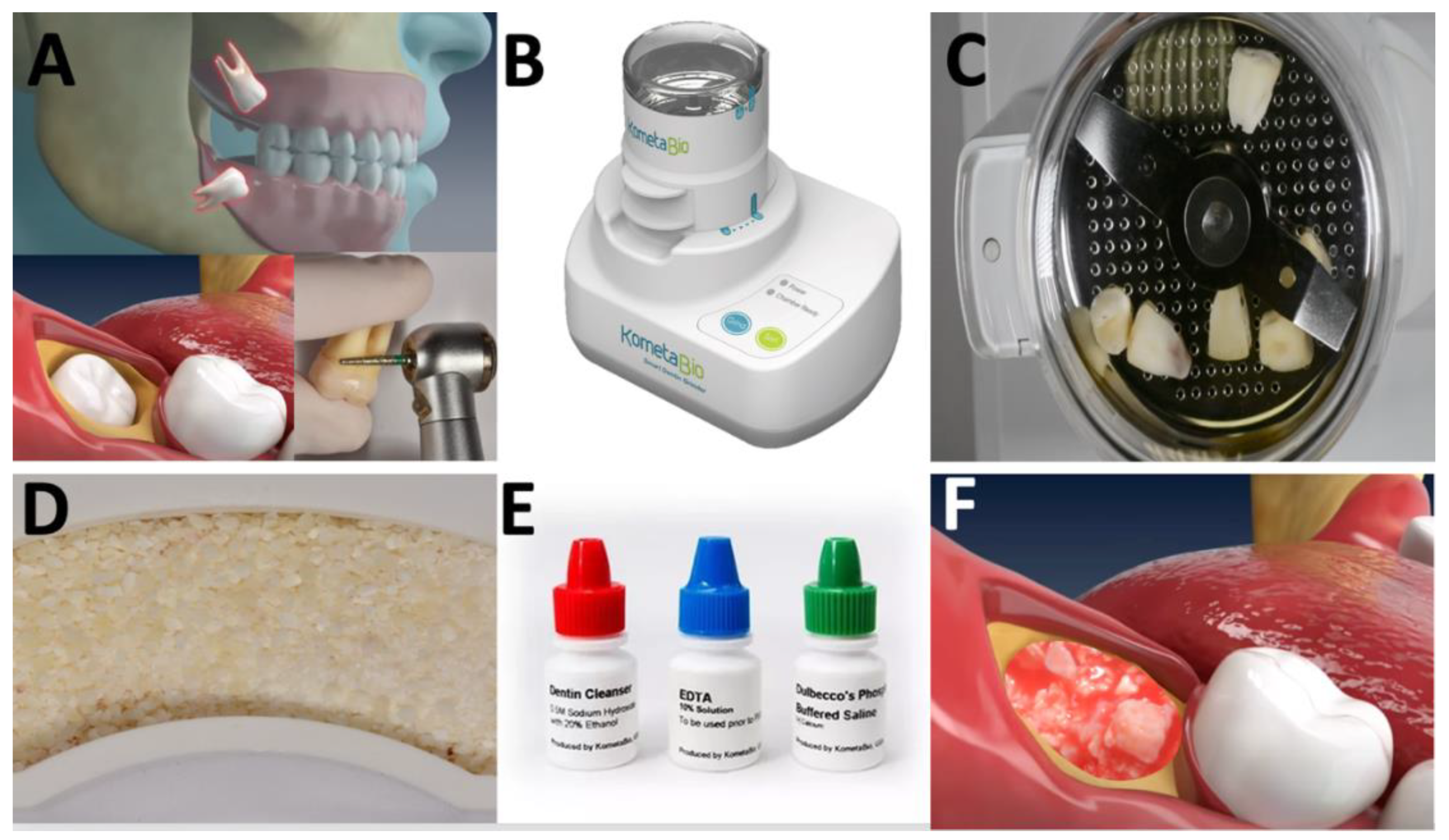
1. Introduction
2. Method
3. Results and Discussion
3.1. Chemical Composition and Morphological Aspects of Dentin-Derived Matrix
3.2. In Vivo Evidence of Bone Healing
4. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Paolone, M.G.; Kaitsas, R. Orthodontic-periodontal interactions: Orthodontic extrusion in interdisciplinary regenerative treatments. Int. Orthod. 2018, 16, 217–245. [Google Scholar] [CrossRef] [PubMed]
- Gideon Hallel, I.B. A Novel Procedure to Process Extracted Teeth for Immediate Grafting of Autogenous Dentin. JBR J. Interdiscip. Med. Dent. Sci. 2014, 2, 2–6. [Google Scholar] [CrossRef]
- Park, S.-M.; Um, I.-W.; Kim, Y.-K.; Kim, K.-W. Clinical application of auto-tooth bone graft material. J. Korean Assoc. Oral Maxillofac. Surg. 2012, 38, 2. [Google Scholar] [CrossRef]
- Becker, W.; Urist, M.; Becker, B.E.; Jackson, W.; Parry, D.A.; Bartold, M.; Vincenzzi, G.; De Georges, D.; Niederwanger, M. Clinical and histologic observations of sites implanted with intraoral autologous bone grafts or allografts. 15 human case reports. J. Periodontol. 1996, 67, 1025–1033. [Google Scholar] [CrossRef]
- Hämmerle, C.H.F.; Araújo, M.G.; Simion, M. Evidence-based knowledge on the biology and treatment of extraction sockets. Clin. Oral Implant. Res. 2012, 23, 80–82. [Google Scholar] [CrossRef]
- Artzi, Z.; Tal, H.; Dayan, D. Porous bovine bone mineral in healing of human extraction sockets. Part 1: Histomorphometric evaluations at 9 months. J. Periodontol. 2000, 71, 1015–1023. [Google Scholar] [CrossRef]
- Valen, M.; Ganz, S.D. A synthetic bioactive resorbable graft for predictable implant reconstruction: Part one. J. Oral. Implantol. 2002, 28, 167–177. [Google Scholar] [CrossRef]
- Kim, Y.-K.; Lee, J.; Yun, J.-Y.; Yun, P.-Y.; Um, I.-W. Comparison of autogenous tooth bone graft and synthetic bone graft materials used for bone resorption around implants after crestal approach sinus lifting: A retrospective study. J. Periodontal Implant. Sci. 2014, 44, 216–221. [Google Scholar] [CrossRef]
- Urban, I.A.; Jovanovic, S.A.; Lozada, J.L. Vertical ridge augmentation using guided bone regeneration (GBR) in three clinical scenarios prior to implant placement: A retrospective study of 35 patients 12 to 72 months after loading. Int. J. Oral Maxillofac. Implant. 2009, 24, 502–510. [Google Scholar]
- Dłucik, R.; Orzechowska-Wylęgala, B.; Dłucik, D.; Puzzolo, D.; Micali, A. Socket preservation or guided bone regeneration—A case report. Pol. Merkur. Lek. 2021, 49, 153–157. [Google Scholar]
- Minetti, E.; Giacometti, E.; Gambardella, U.; Contessi, M.; Ballini, A.; Marenzi, G.; Celko, M.; Mastrangelo, F. Alveolar Socket Preservation with Different Autologous Graft Materials: Preliminary Results of a Multicenter Pilot Study in Human. Materials 2020, 13, 1153. [Google Scholar] [CrossRef] [PubMed]
- Mendoza-Azpur, G.; de la Fuente, A.; Chavez, E.; Valdivia, E.; Khouly, I. Horizontal ridge augmentation with guided bone regeneration using particulate xenogenic bone substitutes with or without autogenous block grafts: A randomized controlled trial. Clin. Implant. Dent. Relat. Res. 2019, 21, 521–530. [Google Scholar] [CrossRef]
- von Arx, T.; Cochran, D.L.; Hermann, J.S.; Schenk, R.K.; Buser, D. Lateral ridge augmentation using different bone fillers and barrier membrane application. A histologic and histomorphometric pilot study in the canine mandible. Clin. Oral Implant. Res. 2001, 12, 260–269. [Google Scholar] [CrossRef]
- Pohl, S.; Binderman, I.; Tomac, J. Maintenance of Alveolar Ridge Dimensions Utilizing an Extracted Tooth Dentin Particulate Autograft and PlateletRich Fibrin: A Retrospective Radiographic ConeBeam Computed Tomography Study. Materials 2020, 13, 1083. [Google Scholar] [CrossRef] [PubMed]
- Araujo, M.G.; Lindhe, J. Dimensional ridge alterations following tooth extraction. An experimental study in the dog. J. Clin. Periodontol. 2005, 32, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Mezzomo, L.A.; Shinkai, R.S.; Mardas, N.; Donos, N. Alveolar ridge preservation after dental extraction and before implant placement: A literature review. Rev. Odonto Ciênc. 2011, 26, 77–83. [Google Scholar] [CrossRef]
- Esposito, M.; Maghaireh, H.; Grusovin, M.G.; Ziounas, I.; Worthington, H.V. Interventions for replacing missing teeth: Management of soft tissues for dental implants. Cochrane Database Syst. Rev. 2012, CD006697. [Google Scholar] [CrossRef]
- Giesenhagen, B.; Martin, N.; Donkiewicz, P.; Perić Kačarević, Ž.; Smeets, R.; Jung, O.; Schnettler, R.; Barbeck, M. Vertical bone augmentation in a single-tooth gap with an allogenic bone ring: Clinical considerations. J. Esthet. Restor. Dent. 2018, 30, 480–483. [Google Scholar] [CrossRef]
- Maeda, D.; Lima, F.; Meza, J.; Ciotti, D.L.; Mizutani, F.S.; Doyle, H.; Faveri, M. Alveolar Ridge Regeneration of Damaged Extraction Sockets using a Bovine-derived Bone Graft in Association with a Titanium Foil: Prospective Case Series. J. Int. Acad. Periodontol. 2020, 22, 109–116. [Google Scholar]
- Qiu, L.; Yu, H. Onlay grafting with bovine bone mineral block for horizontal reconstruction of severely atrophic alveolar ridges in anterior maxillae: A 6-year prospective study. J. Craniomaxillofac. Surg. 2018, 46, 1199–1204. [Google Scholar] [CrossRef]
- Cardaropoli, D.; Nevins, M.; Schupbach, P. New Bone Formation Using an Extracted Tooth as a Biomaterial: A Case Report with Histologic Evidence. Int. J. Periodontics Restor. Dent. 2019, 39, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Andersson, L.; Ramzi, A.; Joseph, B. Studies on dentin grafts to bone defects in rabbit tibia and mandible; development of an experimental model. Dent. Traumatol. Off. Publ. Int. Assoc. Dent. Traumatol. 2009, 25, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Bormann, K.H.; Suarez-Cunqueiro, M.M.; Sinikovic, B.; Kampmann, A.; von See, C.; Tavassol, F.; Binger, T.; Winkler, M.; Gellrich, N.C.; Rücker, M. Dentin as a suitable bone substitute comparable to ß-TCP--an experimental study in mice. Microvasc. Res. 2012, 84, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Brudevold, F.; Steadman, L.T.; Smith, F.A. Inorganic and Organic Components of Tooth Structure. Ann. N. Y. Acad. Sci. 1960, 85, 110–132. [Google Scholar] [CrossRef] [PubMed]
- Leonhardt, Å.; Dahlén, G.; Renvert, S. Five-Year Clinical, Microbiological, and Radiological Outcome Following Treatment of Peri-Implantitis in Man. J. Periodontol. 2005, 74, 1415–1422. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, F.; Becker, J.; Hazar, D.; Becker, K.; Sader, R.; Becker, J. Efficacy of autogenous tooth roots for lateral alveolar ridge augmentation and staged implant placement. A prospective controlled clinical study. J. Clin. Periodontol. 2018, 45, 996–1004. [Google Scholar] [CrossRef]
- Dragoo, M.R.; Kaldahl, W.B. Clinical and histological evaluation of alloplasts and allografts in regenerative periodontal surgery in humans. Int. J. Periodontics Restor. Dent. 1983, 3, 8–29. [Google Scholar]
- Cenicante, J.; Botelho, J.; Machado, V.; Mendes, J.J.; Mascarenhas, P.; Alcoforado, G.; Santos, A. The use of autogenous teeth for alveolar ridge preservation: A literature review. Appl. Sci. 2021, 11, 1853. [Google Scholar] [CrossRef]
- Hussain, I.; Moharamzadeh, K.; Brook, I.M.; José de Oliveira Neto, P.; Salata, L.A. Evaluation of osteoconductive and osteogenic potential of a dentin-based bone substitute using a calvarial defect model. Int. J. Dent. 2012, 2012, 396316. [Google Scholar] [CrossRef]
- Yüceer-Çetiner, E.; Özkan, N.; Önger, M.E. Effect of Autogenous Dentin Graft on New Bone Formation. J. Craniofac. Surg. 2021, 32, 1354–1360. [Google Scholar] [CrossRef] [PubMed]
- Graziano, A.; d’Aquino, R.; Laino, G.; Papaccio, G. Dental pulp stem cells: A promising tool for bone regeneration. Stem Cell Rev. 2008, 4, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Calvo-Guirado, J.L.; Maté-Sánchez de Val, J.E.; Ramos-Oltra, M.L.; Pérez-Albacete Martínez, C.; Ramírez-Fernández, M.P.; Maiquez-Gosálvez, M.; Gehrke, S.A.; Fernández-Domínguez, M.; Romanos, G.E.; Delgado-Ruiz, R.A. The Use of Tooth Particles as a Biomaterial in Post-Extraction Sockets. Experimental Study in Dogs. Dent. J. 2018, 6, 12. [Google Scholar] [CrossRef]
- Calvo-Guirado, J.L.; Ballester Montilla, A.; De Aza, P.N.; Fernández-Domínguez, M.; Gehrke, S.A.; Cegarra-Del Pino, P.; Mahesh, L.; Pelegrine, A.A.; Aragoneses, J.M.; Maté-Sánchez de Val, J.E. Particulated, Extracted Human Teeth Characterization by SEM−EDX Evaluation as a Biomaterial for Socket Preservation: An in vitro Study. Materials 2019, 12, 380. [Google Scholar] [CrossRef] [PubMed]
- Calvo-Guirado, J.; Garcés-Villalá, M.; Mahesh, L.; De Carlos-Villafranca, F. Effectiveness of chemical disinfection in discarding pathogenic bacteria of human particulate tooth graft: An In vitro study. Indian J. Dent. Sci. 2021, 13, 277. [Google Scholar] [CrossRef]
- Santos, A.; Botelho, J.; Machado, V.; Borrecho, G.; Proença, L.; Mendes, J.J.; Mascarenhas, P.; Alcoforado, G. Autogenous Mineralized Dentin versus Xenograft granules in Ridge Preservation for Delayed Implantation in Post-extraction Sites: A Randomized controlled clinical trial with an 18 months follow-up. Clin. Oral Implant. Res. 2021, 32, 905–915. [Google Scholar] [CrossRef]
- Mazzucchi, G.; Lollobrigida, M.; Lamazza, L.; Serafini, G.; Di Nardo, D.; Testarelli, L.; De Biase, A. Autologous Dentin Graft after Impacted Mandibular Third Molar Extraction to Prevent Periodontal Pocket Formation-A Split-Mouth Pilot Study. Materials 2022, 15, 1431. [Google Scholar] [CrossRef]
- Catanzaro-Guimarães, S.A.; Catanzaro Guimarães, B.P.; Garcia, R.B.; Alle, N. Osteogenic potential of autogenic demineralized dentin implanted in bony defects in dogs. Int. J. Oral Maxillofac. Surg. 1986, 15, 160–169. [Google Scholar] [CrossRef]
- Radoczy-Drajko, Z.; Windisch, P.; Svidro, E.; Tajti, P.; Molnar, B.; Gerber, G. Clinical, radiographical and histological evaluation of alveolar ridge preservation with an autogenous tooth derived particulate graft in EDS class 3-4 defects. BMC Oral Health 2021, 21, 63. [Google Scholar] [CrossRef]
- Li, P.; Zhu, H.; Huang, D. Autogenous DDM versus Bio-Oss granules in GBR for immediate implantation in periodontal postextraction sites: A prospective clinical study. Clin. Implant Dent. Relat. Res. 2018, 20, 923–928. [Google Scholar] [CrossRef]
- Tanwatana, S.; Kiewjurat, A.; Suttapreyasri, S. Chemical and thermal deproteinization of human demineralized tooth matrix: Physicochemical characterization and osteoblast cell biocompatibility. J. Biomater. Appl. 2019, 34, 651–663. [Google Scholar] [CrossRef]
- Bono, N.; Tarsini, P.; Candiani, G. Demineralized dentin and enamel matrices as suitable substrates for bone regeneration. J. Appl. Biomater. Funct. Mater. 2017, 15, e236–e243. [Google Scholar] [CrossRef] [PubMed]
- Lamont, T.; Worthington, H.V.; Clarkson, J.E.; Beirne, P.V. Routine scale and polish for periodontal health in adults. Cochrane Database Syst. Rev. 2018, 12, CD004625. [Google Scholar] [CrossRef] [PubMed]
- Muduroglu, R.; Ionescu, A.C.; Del Fabbro, M.; Scolavino, S.; Brambilla, E. Distribution of adhesive layer in class II composite resin restorations before/after interproximal matrix application. J. Dent. 2020, 103, 103494. [Google Scholar] [CrossRef] [PubMed]
- Murata, M.; Okubo, N.; Shakya, M.; Arafat Kabir, M.; Yokozeki, K.; Zhu, B.; Ishikawa, M.; Kitamura, R.; Akazawa, T. Dentin Materials as Biological Scaffolds for Tissue Engineering. In Biomaterial-Supported Tissue Reconstruction or Regeneration; IntechOpen: London, UK, 2019; p. 13. [Google Scholar]
- Farzad, P.; Lundgren, T.; Al-Asfour, A.; Andersson, L.; Dahlin, C. Integration of Dental Implants in Conjunction with EDTA-Conditioned Dentin Grafts: An Experimental Study. Dent. J. 2021, 9, 63. [Google Scholar] [CrossRef] [PubMed]
- Minamizato, T.; Koga, T.; Takashi, I.; Nakatani, Y.; Umebayashi, M.; Sumita, Y.; Ikeda, T.; Asahina, I. Clinical application of autogenous partially demineralized dentin matrix prepared immediately after extraction for alveolar bone regeneration in implant dentistry: A pilot study. Int. J. Oral Maxillofac. Surg. 2018, 47, 125–132. [Google Scholar] [CrossRef]
- Koga, T.; Minamizato, T.; Kawai, Y.; Miura, K.I.; Takashi, I.; Nakatani, Y.; Sumita, Y.; Asahina, I. Bone regeneration using dentin matrix depends on the degree of demineralization and particle size. PLoS ONE 2016, 11, e0147235. [Google Scholar] [CrossRef]
- Um, I.-W.; Lee, J.-K.; Kim, J.-Y.; Kim, Y.-M.; Bakhshalian, N.; Jeong, Y.K.; Ku, J.-K. Allogeneic Dentin Graft: A Review on Its Osteoinductivity and Antigenicity. Materials 2021, 14, 1713. [Google Scholar] [CrossRef]
- Pai, G.P.; Dayakar, M.M.; Sreedhar, A.S. Dentin Graft—The Hidden Treasure. Int. J. Res. Eng. Sci. Manag. 2021, 4, 19–21. [Google Scholar]
- Pashley, D.H. Dentin: A dynamic substrate—A review. Scanning Microsc. 1989, 3, 161–176. [Google Scholar]
- Feng, S.; Li, R.; Wang, Z. Experimental study on the biocompatibility and osteogenesis induction ability of PLLA/DDM scaffolds. Odontology 2022, 110, 508–522. [Google Scholar] [CrossRef]
- Goldberg, M.; Kulkarni, A.B.; Young, M.; Boskey, A. Dentin: Structure, composition and mineralization. Front. Biosci. Elite 2011, 3, 711–735. [Google Scholar] [CrossRef] [PubMed]
- Brennan, C.M.; Eichholz, K.F.; Hoey, D.A. The effect of pore size within fibrous scaffolds fabricated using melt electrowriting on human bone marrow stem cell osteogenesis. Biomed. Mater. 2019, 14, 065016. [Google Scholar] [CrossRef] [PubMed]
- Reddi, A.H.; Huggins, C.B. Influence of Geometry of Transplanted Tooth and Bone on Transformation of Fibroblasts. Exp. Biol. Med. 1973, 143, 634–637. [Google Scholar] [CrossRef] [PubMed]
- Palma, P.J.; Matos, S.; Ramos, J.; Guerra, F.; Figueiredo, M.H.; Kauser, J. New formulations for space provision and bone regeneration. Biodental Eng. I 2010, 1, 71–76. [Google Scholar]
- Noronha Oliveira, M.; Varela, H.A.; Caramês, J.; Silva, F.; Henriques, B.; Teughels, W.; Quirynen, M.; Souza, J.C.M. Synergistic Benefits on Combining Injectable Platelet-Rich Fibrin and Bone Graft Porous Particulate Materials. Biomed. Mater. Devices 2022. [Google Scholar] [CrossRef]
- Cortellini, S.; Castro, A.B.; Temmerman, A.; Van Dessel, J.; Pinto, N.; Jacobs, R.; Quirynen, M. Leucocyte- and platelet-rich fibrin block for bone augmentation procedure: A proof-of-concept study. J. Clin. Periodontol. 2018, 45, 624–634. [Google Scholar] [CrossRef]
- Matsuzawa, Y.; Okubo, N.; Tanaka, S.; Kashiwazaki, H.; Kitagawa, Y.; Ohiro, Y.; Mikoya, T.; Akazawa, T.; Murata, M. Primary Teeth-Derived Demineralized Dentin Matrix Autograft for Unilateral Maxillary Alveolar Cleft during Mixed Dentition. J. Funct. Biomater. 2022, 13, 153. [Google Scholar] [CrossRef]
- Ramanauskaite, A.; Sahin, D.; Sader, R.; Becker, J.; Schwarz, F. Efficacy of autogenous teeth for the reconstruction of alveolar ridge deficiencies: A systematic review. Clin. Oral Investig. 2019, 23, 4263–4287. [Google Scholar] [CrossRef]
- Andrade, C.; Camino, J.; Nally, M.; Quirynen, M.; Martínez, B.; Pinto, N. Combining autologous particulate dentin, L-PRF, and fibrinogen to create a matrix for predictable ridge preservation: A pilot clinical study. Clin. Oral Investig. 2020, 24, 1151–1160. [Google Scholar] [CrossRef]
- Um, I.-W.; Ku, J.-K.; Kim, Y.-K.; Lee, B.-K.; Leem, D.H. Histological Review of Demineralized Dentin Matrix as a Carrier of rhBMP-2. Tissue Eng. Part B Rev. 2020, 26, 284–293. [Google Scholar] [CrossRef]
- Kim, Y.K.; Lee, J.; Um, I.W.; Kim, K.W.; Murata, M.; Akazawa, T.; Mitsugi, M. Tooth-derived bone graft material. J. Korean Assoc. Oral Maxillofac. Surg. 2013, 39, 103–111. [Google Scholar] [CrossRef]
- Kim, S.Y.; Kim, Y.K.; Park, Y.H.; Park, J.C.; Ku, J.K.; Um, I.W.; Kim, J.Y. Evaluation of the Healing Potential of Demineralized Dentin Matrix Fixed with Recombinant Human Bone Morphogenetic Protein-2 in Bone Grafts. Materials 2017, 10, 1049. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-K.; Pang, K.-M.; Yun, P.-Y.; Leem, D.-H.; Um, I.-W. Long-term follow-up of autogenous tooth bone graft blocks with dental implants. Clin. Case Rep. 2017, 5, 108–118. [Google Scholar] [CrossRef] [PubMed]
- Jin, S.-C.; Kim, S.-G.; Oh, J.-S.; Lee, S.-Y.; Jang, E.-S.; Piao, Z.-G.; Lim, S.-C.; Jeong, M.-A.; Kim, J.-S.; You, J.-S.; et al. A comparative study of bone formation following grafting with different ratios of particle dentin and tricalcium phosphate combinations. J. Biomed. Nanotechnol. 2013, 9, 475–478. [Google Scholar] [CrossRef] [PubMed]
- Gharpure, A.S.; Bhatavadekar, N.B. Clinical Efficacy of Tooth-Bone Graft: A Systematic Review and Risk of Bias Analysis of Randomized Control Trials and Observational Studies. Implant Dent. 2018, 27, 119–134. [Google Scholar] [CrossRef]
- Al-Asfour, A.; Andersson, L.; Kamal, M.; Joseph, B. New bone formation around xenogenic dentin grafts to rabbit tibia marrow. Dent. Traumatol. Off. Publ. Int. Assoc. Dent. Traumatol. 2013, 29, 455–460. [Google Scholar] [CrossRef]
- Kim, Y.-K.; Lee, J.-H.; Um, I.-W.; Cho, W.-J. Guided Bone Regeneration Using Demineralized Dentin Matrix: Long-Term Follow-Up. J. Oral Maxillofac. Surg. 2016, 74, 515.e1–515.e9. [Google Scholar] [CrossRef]
- Cervera-Maillo, J.M.; Morales-Schwarz, D.; Morales-Melendez, H.; Mahesh, L.; Calvo-Guirado, J.L. Autologous Tooth Dentin Graft: A Retrospective Study in Humans. Medicina 2021, 58, 56. [Google Scholar] [CrossRef]
- Valdec, S.; Pasic, P.; Soltermann, A.; Thoma, D.; Stadlinger, B.; Rücker, M. Alveolar ridge preservation with autologous particulated dentin-a case series. Int. J. Implant Dent. 2017, 3, 12. [Google Scholar] [CrossRef]
- Jeong, K.I.; Kim, S.G.; Kim, Y.K.; Oh, J.S.; Jeong, M.A.; Park, J.J. Clinical study of graft materials using autogenous teeth in maxillary sinus augmentation. Implant Dent. 2011, 20, 471–475. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Souza, J.C.M.; Escobar, M.; Pimentel, I.S.; Caramês, J.; Teughels, W.; Silva, F.; Henriques, B. Tooth-Derived Matrix Granules for Enhanced Bone Healing: Chemical Composition, Morphological Aspects, and Clinical Outcomes. Ceramics 2022, 5, 981-990. https://doi.org/10.3390/ceramics5040070
Souza JCM, Escobar M, Pimentel IS, Caramês J, Teughels W, Silva F, Henriques B. Tooth-Derived Matrix Granules for Enhanced Bone Healing: Chemical Composition, Morphological Aspects, and Clinical Outcomes. Ceramics. 2022; 5(4):981-990. https://doi.org/10.3390/ceramics5040070
Chicago/Turabian StyleSouza, Júlio C. M., Mario Escobar, Inês Sofia Pimentel, João Caramês, Wim Teughels, Filipe Silva, and Bruno Henriques. 2022. "Tooth-Derived Matrix Granules for Enhanced Bone Healing: Chemical Composition, Morphological Aspects, and Clinical Outcomes" Ceramics 5, no. 4: 981-990. https://doi.org/10.3390/ceramics5040070
APA StyleSouza, J. C. M., Escobar, M., Pimentel, I. S., Caramês, J., Teughels, W., Silva, F., & Henriques, B. (2022). Tooth-Derived Matrix Granules for Enhanced Bone Healing: Chemical Composition, Morphological Aspects, and Clinical Outcomes. Ceramics, 5(4), 981-990. https://doi.org/10.3390/ceramics5040070










