Flash Sintering Samaria-Doped Ceria–Carbon Nanotube Composites
Abstract
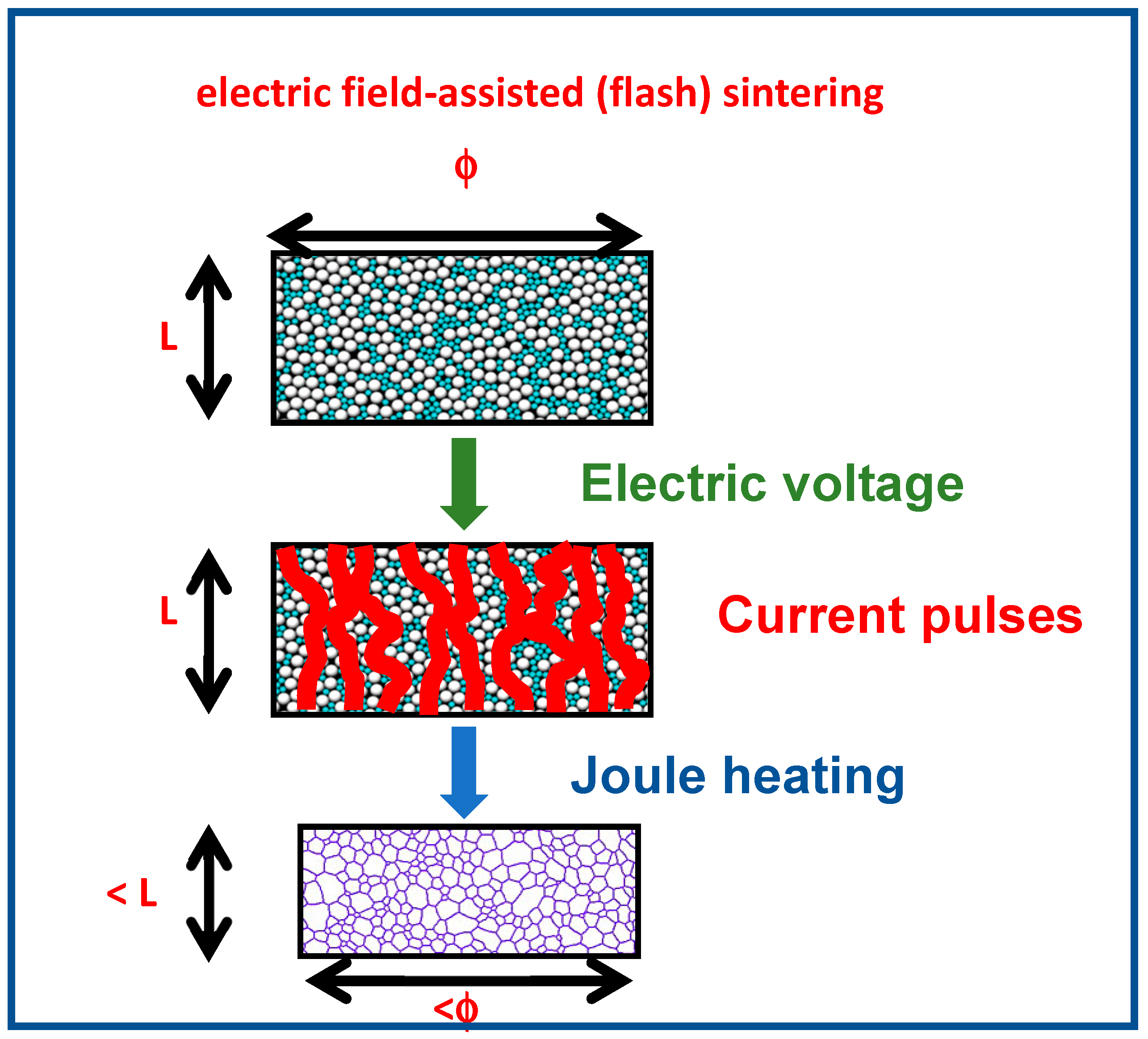
1. Introduction
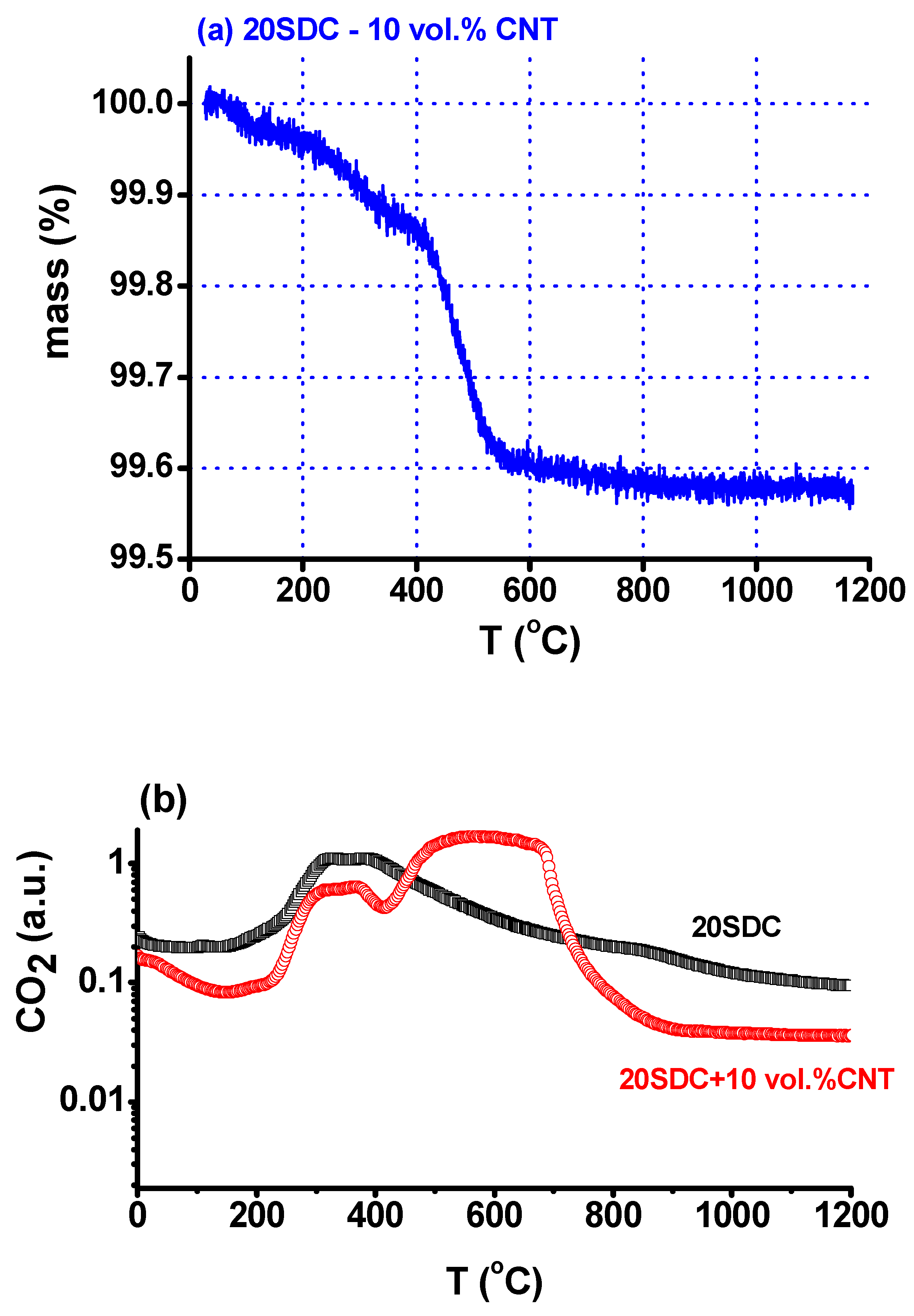
2. Materials and Methods
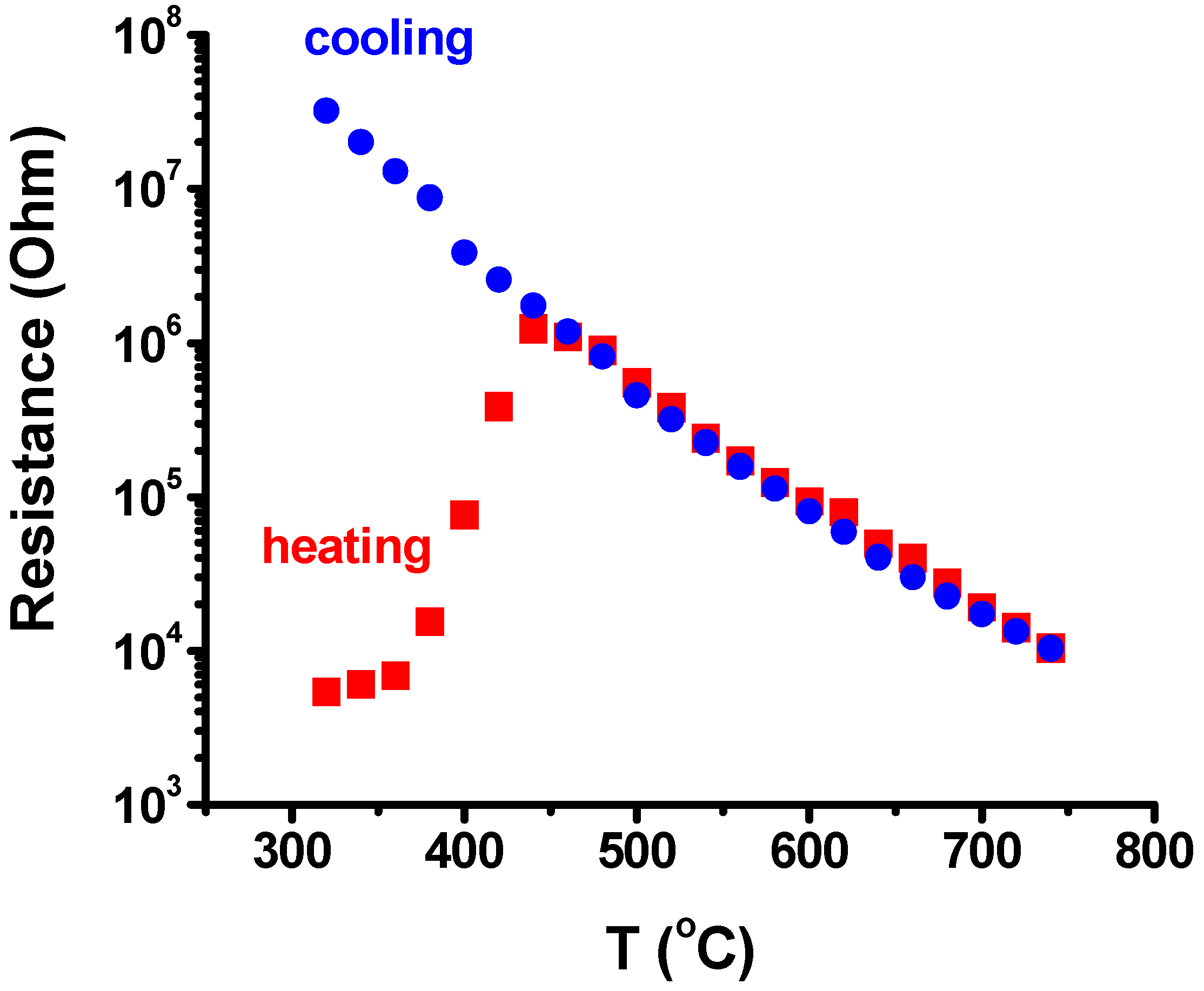
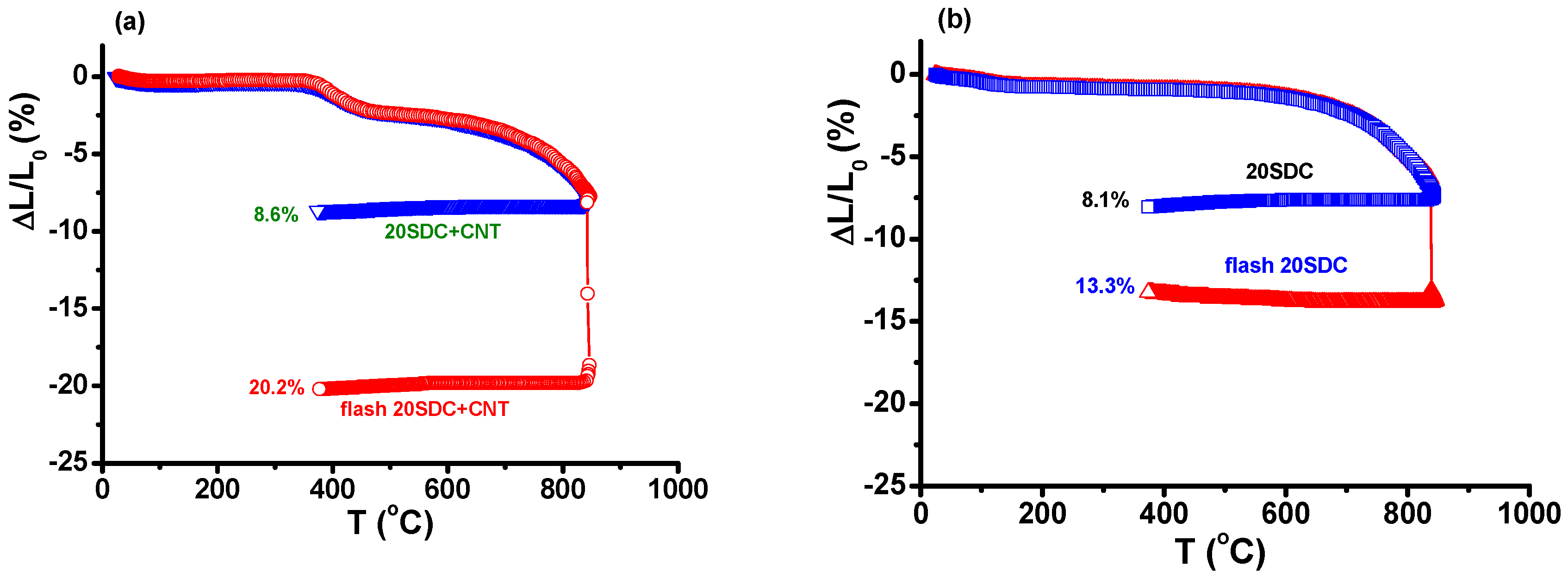
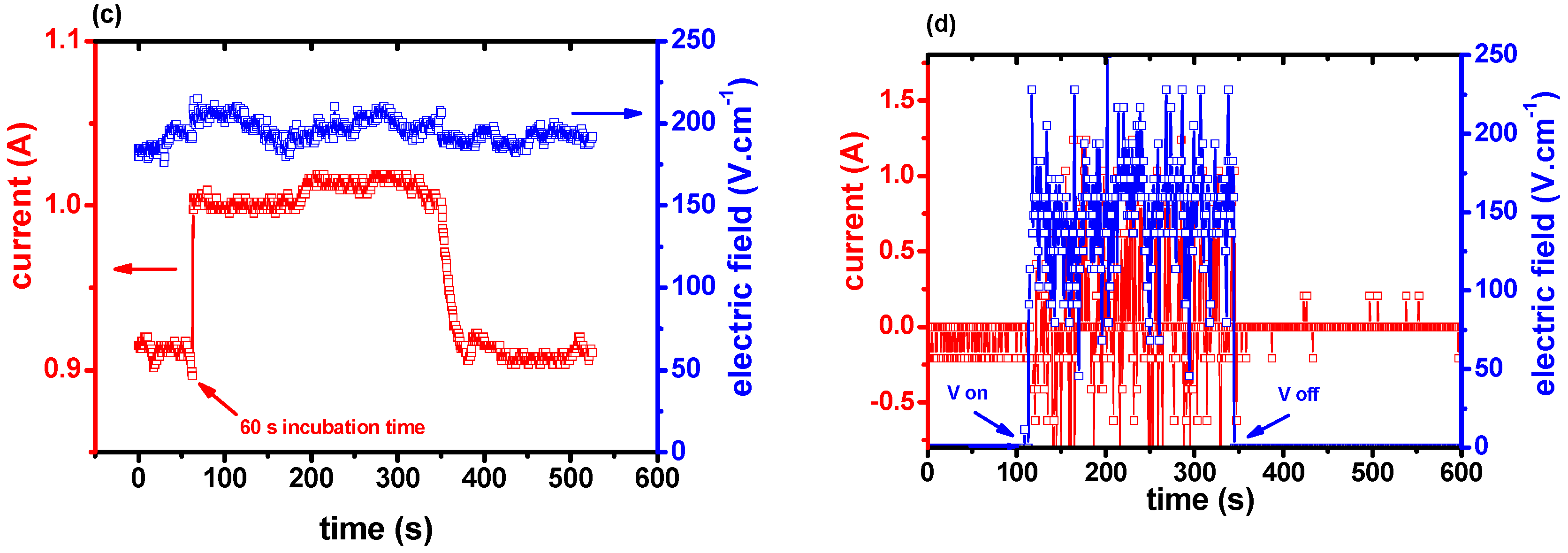
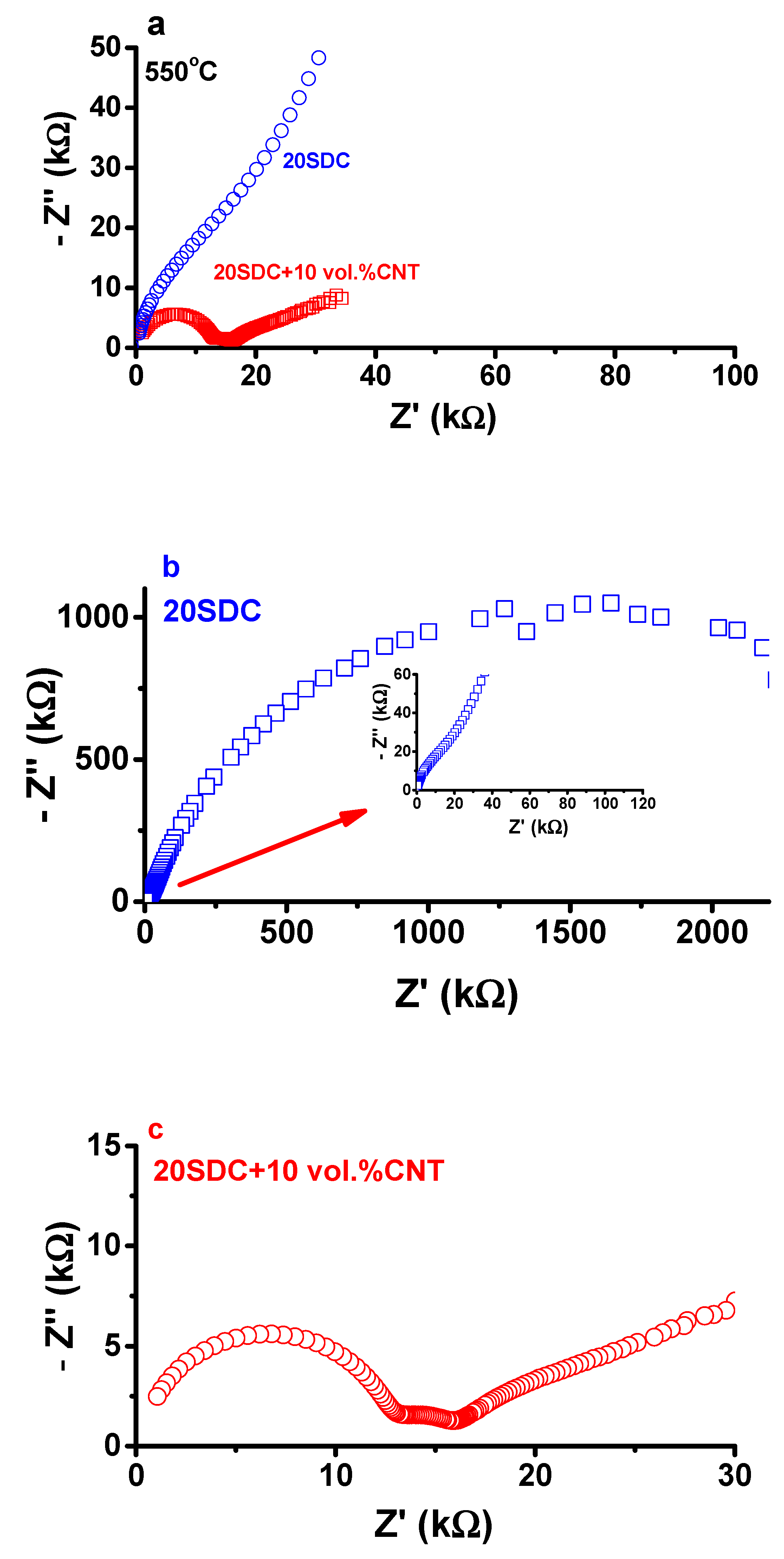
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cologna, M.; Rashkova, B.; Raj, R. Flash sintering of nanograin zirconia in <5 s at 850 °C. J. Am. Ceram. Soc. 2010, 93, 3556–3559. [Google Scholar] [CrossRef]
- Cologna, M.; Raj, R. Surface diffusion-controlled neck growth kinetics in early stage sintering of zirconia, with and without applied DC electrical field. J. Am. Ceram. Soc. 2011, 94, 391–395. [Google Scholar] [CrossRef]
- Obare, J.; Griffin, W.D.; Conrad, H. Effects of heating rate and DC electric field during sintering on the grain size distribution in fully sintered tetragonal zirconia polycrystals stabilized with 3% molar yttria (3Y-TZP). J. Mater. Sci. 2012, 47, 5141–5147. [Google Scholar] [CrossRef]
- Ji, W.; Parker, B.; Falco, S.; Zhang, J.Y.; Fu, Z.Y.; Todd, R.I. Ultra-fast firing: Effect of heating rate on sintering of 3YSZ, with and without an electric field. J. Eur. Ceram. Soc. 2017, 37, 2547–2551. [Google Scholar] [CrossRef]
- Conrad, H. Space charge and grain boundary energy in zirconia (3Y-TZP). J. Am. Ceram. Soc. 2011, 94, 3641–3642. [Google Scholar] [CrossRef]
- Carvalho, S.G.M.; Muccillo, E.N.S.; Muccillo, R. Electrical behavior and microstructural features of electric field-assisted and conventionally sintered 3 mol% yttria-stabilized zirconia. Ceramics 2018, 1, 2. [Google Scholar] [CrossRef]
- Muccillo, R.; Kleitz, M.; Muccillo, E.N.S. Flash grain welding in yttria stabilized zirconia. J. Eur. Ceram. Soc. 2011, 31, 1517–1521. [Google Scholar] [CrossRef]
- Muccillo, R.; Muccillo, E.N.S. An experimental setup for shrinkage evaluation during electric field-assisted flash sintering: Application to yttria-stabilized zirconia. J. Eur. Ceram. Soc. 2013, 33, 515–520. [Google Scholar] [CrossRef]
- Downs, J.A.; Sglavo, V.M. Electric field assisted sintering of cubic zirconia at 390 °C. J. Am. Ceram. Soc. 2013, 96, 1342–1344. [Google Scholar] [CrossRef]
- Steil, M.C.; Marinha, D.; Aman, Y.; Gomes, J.R.C.; Kleitz, M. From conventional ac flash-sintering of YSZ to hyper-flash and double flash. J. Eur. Ceram. Soc. 2012, 33, 2093–2101. [Google Scholar] [CrossRef]
- Muccillo, R.; Muccillo, E.N.S. Shrinkage control of yttria-stabilized zirconia during ac electric field-assisted sintering. J. Eur. Ceram. Soc. 2014, 34, 3871–3877. [Google Scholar] [CrossRef]
- Muccillo, E.N.S.; Carvalho, S.G.M.; Muccillo, R. Electric field-assisted pressureless sintering of zirconia–scandia–ceria solid electrolytes. J. Mater. Sci. 2018, 53, 1658–1671. [Google Scholar] [CrossRef]
- Hao, X.; Liu, Y.; Wang, Z.; Qiao, J.; Sun, K. A novel sintering method to obtain fully dense gadolinia doped ceria by applying a direct current. J. Power Sources 2012, 210, 86–91. [Google Scholar] [CrossRef]
- Valdebenito, J.U.; Akbari-Fakhrabadi, A.; Viswanathan, M.R. Effect of flash sintering on microstructure of Ce0.9Gd0.1O1.95 electrolyte fabricated by tape-casting. Mater. Lett. 2017, 209, 291–294. [Google Scholar] [CrossRef]
- Biesuz, M.; Del’Agli, G.; Spiridigliozzi, L.; Ferone, C.; Sglavo, V.M. Conventional and field-assisted sintering of nanosized Gd-doped ceria synthesized by co-precipitation. Ceram. Int. 2016, 42, 11766–11771. [Google Scholar] [CrossRef]
- Spiridigliozzi, L.; Biesuz, M.; Dell’Agli, G.; Di Bartolomeo, E.; Zurlo, F.; Sglavo, V.M. Microstructural and electrical investigation of flash-sintered Gd/Sm-doped ceria. J. Mater. Sci. 2017, 52, 7479–7488. [Google Scholar] [CrossRef]
- Jiang, T.; Wang, Z.; Zhang, J.; Hao, X.; Rooney, D.; Liu, Y.; Sun, W.; Qiao, J.; Sun, K.; Jia, T. Understanding the flash sintering of rare-earth-doped ceria for solid oxide fuel cell. J. Am. Ceram. Soc. 2015, 98, 1717–1723. [Google Scholar] [CrossRef]
- Li, J.; Guan, L.; Zhang, W.; Luo, M.; Song, J.; Song, X.; An, S. Sintering behavior of samarium doped ceria under DC electrical field. Ceram. Int. 2018, 44, 2470–2477. [Google Scholar] [CrossRef]
- Muccillo, R.; Muccillo, E.N.S.; Kleitz, M. Densification and enhancement of the grain boundary conductivity of gadolinium-doped barium cerate by ultra fast flash grain welding. J. Eur. Ceram. Soc. 2012, 32, 2311–2316. [Google Scholar] [CrossRef]
- Muccillo, R.; Esposito, V.; de Florio, D.Z.; Muccillo, E.N.S. Electric field-assisted pressureless sintering gadolinium-, yttrium- and samarium-doped barium cerate. Scr. Mater. 2018, 156, 6–9. [Google Scholar] [CrossRef]
- M’Peko, J.-C.; Francis, J.S.C.; Raj, R. Field-assisted sintering of undoped BaTiO3: Microstructure evolution and dielectric permittivity. J. Eur. Ceram. Soc. 2014, 34, 3655–3660. [Google Scholar] [CrossRef]
- Uehashi, A.; Yoshida, H.; Tokunaga, T.; Sasaki, K.; Yamamoto, T. Enhancement of sintering rates in BaTiO3 by controlling of DC electric current. J. Ceram. Soc. Japan 2015, 123, 465–468. [Google Scholar] [CrossRef]
- Nakagawa, Y.; Yoshida, H.; Uehashi, A.; Tokunaga, T.; Sasaki, K.; Yamamoto, T. Electric current-controlled synthesis of BaTiO3. J. Am. Ceram Soc. 2017, 100, 3843–3850. [Google Scholar] [CrossRef]
- Perez-Maqueda, L.A.; Gil-Gonzalez, E.; Perejon, A.; Lebrun, J.-M.; Sanchez-Jimenez, P.E.; Raj, R. Flash sintering of highly insulating nanostructured phase-pure BiFeO3. J. Am. Ceram. Soc. 2017, 100, 3365–3369. [Google Scholar] [CrossRef]
- Jesus, L.M.; Silva, R.S.; Raj, R.; M’Peko, J.-C. Electric field-assisted flash sintering of CaCu3Ti4O12: Microstructure characteristics and dielectric properties. J. Alloys Compd. 2016, 682, 753–758. [Google Scholar] [CrossRef]
- Zhang, Y.; Luo, J. Promoting the flash sintering of ZnO in reduced atmospheres to achieve nearly full densities at furnace temperatures of <120 °C. Scr. Mater. 2015, 106, 26–29. [Google Scholar] [CrossRef]
- Zhang, Y.; Jung, J.-I.; Luo, J. Thermal runaway, flash sintering and asymmetrical microstructural development of ZnO and ZnO-Bi2O3 under direct currents. Acta Mater. 2015, 94, 87–100. [Google Scholar] [CrossRef]
- Gao, H.; Asel, T.J.; Cox, J.W.; Zhang, Y.; Luo, J.; Brillson, L.J. Native point defect formation in flash sintered ZnO studied by depth-resolved cathodoluminescence spectroscopy. J. Appl. Phys. 2016, 120, 105302. [Google Scholar] [CrossRef]
- Muccillo, E.N.S.; Muccillo, R. Electric field-assisted sintering of tin dioxide with manganese dioxide addition. J. Eur. Ceram. Soc. 2014, 34, 3699–3706. [Google Scholar] [CrossRef]
- Muccillo, R.; Muccillo, E.N.S. Electric field-assisted flash sintering of tin dioxide. J. Eur. Ceram. Soc. 2014, 34, 915–923. [Google Scholar] [CrossRef]
- Gonzalez-Julian, J.; Guillon, O. Effect of electric field/current on liquid phase sintering. J. Am. Ceram. Soc. 2015, 98, 2018–2027. [Google Scholar] [CrossRef]
- Biesuz, M.; Sglavo, V.M. Current-induced abnormal and oriented grain growth in corundum upon flash sintering. Scr. Mater. 2018, 150, 82–86. [Google Scholar] [CrossRef]
- Lemke, F.; Rheinheimer, W.; Hoffmann, M.J. A comparison of power controlled flash sintering and conventional sintering of strontium titanate. Scr. Mater. 2017, 130, 187–190. [Google Scholar] [CrossRef]
- Shomrat, N.; Baltisnski, S.; Don, E.; Tsur, Y. The influence of doping on flash sintering conditions is SrTi1−xFexO3-δ. J. Eur. Ceram. Soc. 2017, 37, 179–188. [Google Scholar] [CrossRef]
- Straka, W.; Amoah, S.; Schwartz, J. Densification of thoria through flash sintering. MRS Commun. 2017, 7, 677–682. [Google Scholar] [CrossRef]
- Raftery, A.M.; da Silva, J.G.P.; Byler, D.D.; Andersson, D.A.; Uberuaga, B.P.; Stanek, C.R.; McClellan, K.J. Onset conditions for flash sintering of UO2. J. Nucl. Mater. 2017, 493, 264–270. [Google Scholar] [CrossRef]
- Valdez, J.A.; Byler, D.D.; Kardoulaki, E.; Francis, J.S.C.; McClellan, K.J. Flash sintering of stoichiometric and hyper-stoichiometric urania. J. Nucl. Mater. 2018, 505, 37–40. [Google Scholar] [CrossRef]
- Charalambous, H.; Jha, S.K.; Wang, H.; Phuah, X.L.; Wang, H.; Tsakalakos, T. Inhomogeneous reduction and its relation to grain growth of titania during flash sintering. Scr. Mater. 2018, 155, 37–40. [Google Scholar] [CrossRef]
- Candelario, V.M.; Moreno, R.; Todd, R.I.; Ortiz, A.L. Liquid-phase assisted flash sintering of SiC from powder mixtures prepared by aqueous colloidal processing. J. Eur. Ceram. Soc. 2017, 37, 485–498. [Google Scholar] [CrossRef]
- Yoshida, H.; Morita, K.; Nam, B.N.; Sakka, Y.; Yamamoto, T. Reduction in sintering temperature for flash-sintering of yttria by nickel cation-doping. Acta Mater. 2016, 106, 344–352. [Google Scholar] [CrossRef]
- Muccillo, R.; Muccillo, E.N.S. Electric field assisted sintering of electroceramics and in situ analysis by impedance spectroscopy. J. Electroceram. 2016, 38, 24–42. [Google Scholar] [CrossRef]
- Dancer, C.E.J. Flash sintering of ceramic materials. Res. Express 2016, 3, 102001. [Google Scholar] [CrossRef]
- Yu, M.; Grasso, S.; McKinnon, R.; Saunders, T.; Reece, M.J. Review of flash sintering: Materials, mechanisms and modelling. Adv. Appl. Ceram. 2016, 116, 24–60. [Google Scholar] [CrossRef]
- Todd, R.I.; Zapata-Solvas, E.; Bonilla, R.S.; Sneddon, T.; Wilshaw, P.R. Electrical characteristics of flash sintering: Thermal runaway of Joule heating. J. Eur. Ceram. Soc. 2015, 35, 1865–1877. [Google Scholar] [CrossRef]
- Hewitt, I.J.; Lacey, A.A.; Todd, R.I. A mathematical model for flash sintering. Math. Model. Nat. Phenom. 2015, 10, 77–89. [Google Scholar] [CrossRef]
- Jha, S.K.; Terauds, K.; Lebrun, J.; Raj, R. Beyond flash sintering in 3 mol% yttria stabilized zirconia. J. Ceram. Soc. Japan 2016, 124, 283–288. [Google Scholar] [CrossRef]
- Narayan, J. Grain growth model for electric field-assisted processing and flash sintering of materials. Scr. Mater. 2013, 68, 785–788. [Google Scholar] [CrossRef]
- Raj, R. Joule heating during flash-sintering. J. Eur. Ceram. Soc. 2012, 32, 2293–2301. [Google Scholar] [CrossRef]
- Naik, K.S.; Sglavo, V.M.; Raj, R. Flash sintering as a nucleation phenomenon and a model thereof. J. Eur. Ceram. Soc. 2014, 34, 4063–4067. [Google Scholar] [CrossRef]
- Qin, W.; Majidi, H.; Yun, J.; van Benthem, K. Electrode effects on microstructure formation during flash sintering of yttrium stabilized zirconia. J. Am. Ceram. Soc. 2016, 99, 2253–2259. [Google Scholar] [CrossRef]
- Cologna, M.; Francis, J.S.C.; Raj, R. Field assisted and flash sintering of alumina and its relationship to conductivity and MgO doping. J. Eur. Ceram. Soc. 2011, 31, 2827–2837. [Google Scholar] [CrossRef]
- Francis, J.S.C.; Cologna, M.; Montinaro, D.; Raj, R. Flash sintering of anode-electrolyte multilayers for SOFC applications. J. Am. Ceram. Soc. 2013, 96, 1352–1354. [Google Scholar] [CrossRef]
- Jha, S.K.; Lebrun, J.M.; Raj, R. Phase transformation in the alumina–titania system during flash sintering experiments. J. Eur. Ceram. Soc. 2016, 36, 733–739. [Google Scholar] [CrossRef]
- Zhang, X.S.; Yang, L.W.; Liu, H.T. High temperature conduction behavior of carbon nanotube fiber from 25 °C to 1100 °C. Appl. Phys. Lett. 2018, 112, 164103. [Google Scholar] [CrossRef]
- Arabacı, A.; Serin, Ö.; Sarıboga, V.; Öksüzömer, M.F. Characterization of Sm and Nd co-doped ceria-based electrolyte materials. Acta Phys. Polonica A 2016, 129, 524–527. [Google Scholar] [CrossRef]
- Iijima, S. Helical microtubules of graphitic carbon. Nature 1991, 354, 56–58. [Google Scholar] [CrossRef]
- Volder, M.F.L.; Tawfick, S.H.; Baughman, R.H.; Hart, A.J. Carbon nanotubes: Present and future commercial applications. Science 2013, 339, 535–539. [Google Scholar] [CrossRef]
- Halder, R.; Sarkar, S.; Bandyopadhyay, S.; Chakraborti, P.C. Sintering and tribomechanical properties of gel-combustion-derived nano-alumina and its composites with carbon nanotubes. J. Mater. Sci. 2018, 53, 8989–9001. [Google Scholar] [CrossRef]
- Available online: https://fuelcellmaterials.com/products/powders/electrolyte-powders/samarium-doped-ceria-20-sm-nanopowder/ (accessed on 10 January 2019).
- Da Cunha, T.H.R.; de Oliveira, S.; Martins, I.L.; Geraldo, V.; Miquita, D.; Ramos, S.L.M.; Lacerda, R.G.; Ladeira, L.O.; Ferlauto, A.S. High-yield synthesis of bundles of double- and triple-walled carbon nanotubes on aluminum flakes. Carbon 2018, 133, 53–61. [Google Scholar] [CrossRef]
- Available online: https://www.tosoh.com/our-products/advanced-materials/zirconia-grinding--dispersion-media (accessed on 10 January 2019).
- Kleitz, M.; Kennedy, J.H. Resolution of multicomponent impedance diagrams. In Fastion Transport in Solids; Mundy, J.N., Shenoy, G.K., Vashishta, P., Eds.; Elsevier North Holland, Inc.: New York, NY, USA, 1979; pp. 185–188. [Google Scholar]
- Guan, L.; Li, J.; Song, X. Graphite assisted flash sintering of Sm2O3 doped CeO2 ceramics at the onset temperature of 25 °C. Scr. Mater. 2019, 159, 72–75. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muccillo, R.; S. Ferlauto, A.; N.S. Muccillo, E. Flash Sintering Samaria-Doped Ceria–Carbon Nanotube Composites. Ceramics 2019, 2, 64-73. https://doi.org/10.3390/ceramics2010006
Muccillo R, S. Ferlauto A, N.S. Muccillo E. Flash Sintering Samaria-Doped Ceria–Carbon Nanotube Composites. Ceramics. 2019; 2(1):64-73. https://doi.org/10.3390/ceramics2010006
Chicago/Turabian StyleMuccillo, Reginaldo, André S. Ferlauto, and Eliana N.S. Muccillo. 2019. "Flash Sintering Samaria-Doped Ceria–Carbon Nanotube Composites" Ceramics 2, no. 1: 64-73. https://doi.org/10.3390/ceramics2010006
APA StyleMuccillo, R., S. Ferlauto, A., & N.S. Muccillo, E. (2019). Flash Sintering Samaria-Doped Ceria–Carbon Nanotube Composites. Ceramics, 2(1), 64-73. https://doi.org/10.3390/ceramics2010006




