Optimization of Operating Conditions for Electrochemical Decolorization of Methylene Blue with Ti/α-PbO2/β-PbO2 Composite Electrode
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Materials
2.2. Preparation of Electrodes
2.3. Characterization
2.4. Electrochemical Decolorization
3. Results and Discussion
3.1. Characterization of Electrodes
3.1.1. XRD Analysis
3.1.2. SEM Study
3.1.3. XPS Analysis
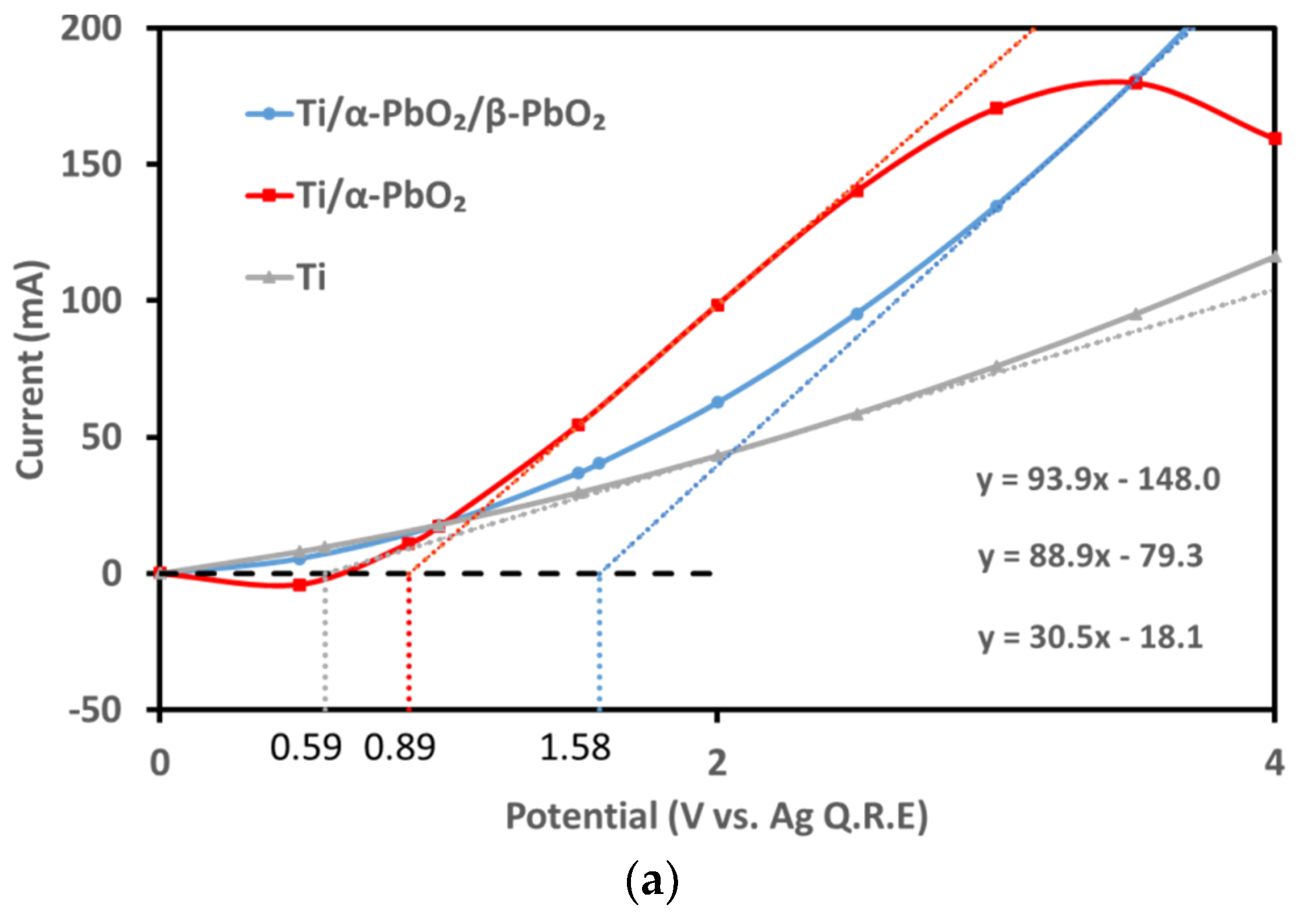
3.1.4. Electrochemical Performance
3.2. Optimization of MB Decolorization Conditions
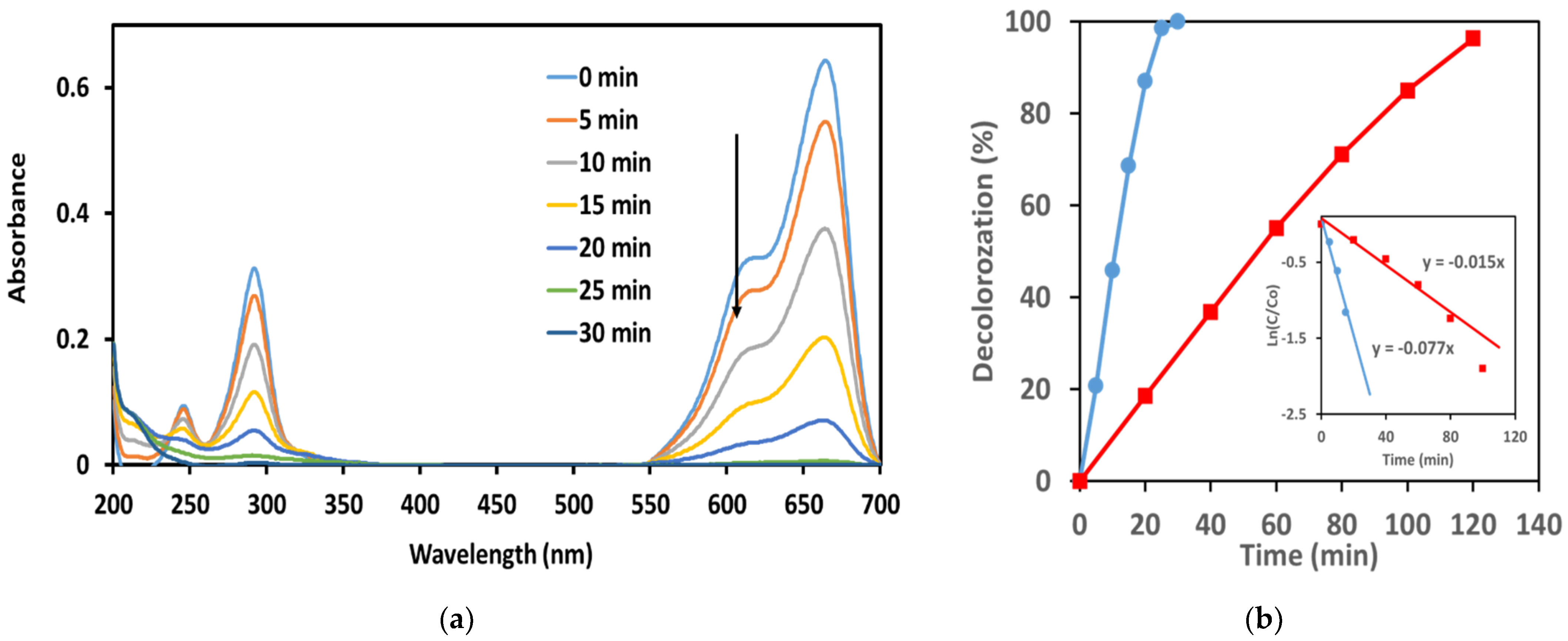
3.2.1. UV-Vis Spectra
3.2.2. Effect of Initial MB Concentration
3.2.3. Effect of Applied Cell Voltage
3.2.4. Effect of Initial pH
3.2.5. Effect of Initial Na2SO4 Concentration
3.3. Electrode Stability
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Yurtsever, A.; Sahinkaya, E.; Aktaş, Ö.; Uçar, D.; Çinar, Ö.; Wang, Z. Performances of anaerobic and aerobic membrane bioreactors for the treatment of synthetic textile wastewater. Bioresour. Technol. 2015, 192, 564–573. [Google Scholar] [CrossRef]
- Chanwala, J.; Kaushik, G.; Dar, M.A.; Upadhyay, S.; Agrawal, A. Process optimization and enhanced decolorization of textile effluent by Planococcus sp. isolated from textile sludge. Environ. Technol. Innov. 2019, 13, 122–129. [Google Scholar] [CrossRef]
- Kornmüller, A.; Cuno, M.; Wiesmann, U. Selective ozonation of polycyclic aromatic hydrocarbons in oil/water-emulsions. Water Sci. Technol. 1997, 35, 57–64. [Google Scholar] [CrossRef]
- Yıldırım, A.Ö.; Gül, Ş.; Eren, O.; Kuşvuran, E. A Comparative study of ozonation, homogeneous catalytic ozonation, and photocatalytic ozonation for C.I. Reactive Red 194 azo dye degradation. CLEAN–Soil Air Water 2011, 39, 795–805. [Google Scholar] [CrossRef]
- Nidheesh, P.; Gandhimathi, R.; Ramesh, S. Degradation of dyes from aqueous solution by Fenton processes: A review. Environ. Sci. Pollut. Res. 2013, 20, 2099–2132. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Lin, J.; Chen, Z. Remediation of water contaminated with diesel oil using a coupled process: Biological degradation followed by heterogeneous Fenton-like oxidation. Chemosphere 2017, 183, 286–293. [Google Scholar] [CrossRef]
- Singla, P.; Sharma, M.; Pandey, O.P.; Singh, K. Photocatalytic degradation of azo dyes using Zn-doped and undoped TiO2 nanoparticles. Appl. Phys. A 2014, 116, 371–378. [Google Scholar] [CrossRef]
- Neon, M.H.K.; Islam, M.S. MoO3 and Ag co-synthesized TiO2 as a novel heterogeneous photocatalyst with enhanced visible-light-driven photocatalytic activity for methyl orange dye degradation. Environ. Nanotechnol. Monit. Manag. 2019, 12, 100244. [Google Scholar]
- Teymori, M.; Khorsandi, H.; Aghapour, A.A.; Jafari, S.J.; Maleki, R. Electro-Fenton method for the removal of Malachite Green: Effect of operational parameters. Appl. Water Sci. 2020, 10, 39. [Google Scholar] [CrossRef]
- Wang, C.; Huang, Y.K.; Zhao, Q.; Ji, M. Treatment of secondary effluent using a three-dimensional electrode system: COD removal, biotoxicity assessment, and disinfection effects. Chem. Eng. J. 2014, 243, 1–6. [Google Scholar]
- Shih, Y.J.; Huang, Y.H.; Huang, C.P. Oxidation of ammonia in dilute aqueous solutions over graphite-supported α- and β-lead dioxide electrodes (PbO2@G). Electrochim. Acta 2017, 257, 444–454. [Google Scholar] [CrossRef]
- Awad, H.S.; Galwa, N.A. Electrochemical degradation of Acid Blue and Basic Brown dyes on Pb/PbO2 electrode in the presence of different conductive electrolyte and effect of various operating factors. Chemosphere 2005, 61, 1327–1335. [Google Scholar] [CrossRef]
- Wang, Y.; Shen, Z.Y.; Li, Y.; Niu, J.F. Electrochemical properties of the erbium–chitosan–fluorine–modified PbO2 electrode for the degradation of 2,4-dichlorophenol in aqueous solution. Chemosphere 2010, 79, 987–996. [Google Scholar] [CrossRef]
- Zheng, Y.H.; Su, W.Q.; Chen, S.Y.; Wu, X.Z.; Chen, X.M. Ti/SnO2–Sb2O5–RuO2/α-PbO2/β-PbO2 electrodes for pollutants degradation. Chem. Eng. J. 2011, 174, 304–309. [Google Scholar] [CrossRef]
- Vazquez-Gomez, L.; de Battisti, A.; Ferro, S.; Cerro, M.; Reyna, S.; Martínez-Huitle, C.A.; Quiroz, M.A. Anodic oxidation as green alternative for removing diethyl phthalate from wastewater using Pb/PbO2 and Ti/SnO2 anodes. CLEAN—Soil Air Water 2012, 40, 408–415. [Google Scholar] [CrossRef]
- Yahiaoui, I.; Aissani-Benissad, F.; Fourcade, F.; Amrane, A. Response surface methodology for the optimization of the electrochemical degradation of phenol on Pb/PbO2 electrode. Environ. Prog. Sustain. Energy 2012, 31, 515–523. [Google Scholar] [CrossRef]
- Li, G.; Yip, H.Y.; Wong, K.H.; Hu, C.; Qu, J.; Wong, P.K. Photoelectrochemical degradation of Methylene Blue with beta-PbO2 electrodes driven by visible light irradiation. J. Environ. Sci. China 2011, 23, 998–1003. [Google Scholar] [CrossRef]
- Sakib, A.A.M.; Masum, S.M.; Hoinkis, J.; Islam, R.; Molla, M.A.I. Synthesis of CuO/ZnO nanocomposites and their application in photodegradation of toxic textile dye. J. Compos. Sci. 2019, 3, 91. [Google Scholar] [CrossRef]
- Zhang, W.; Li, H.; Tang, J.; Lu, H.; Liu, Y. Ginger straw waste-derived porous carbons as effective adsorbents toward Methylene Blue. Molecules 2019, 24, 469. [Google Scholar] [CrossRef] [PubMed]
- Yanagi, G.; Furukawa, M.; Tateishi, I.; Katsumata, H.; Kaneco, S. Electrochemical colorization of methylene blue in solution with metal doped Ti/α, β-PbO₂ mesh electrode. Sep. Sci. Technol. 2021. [Google Scholar] [CrossRef]
- Duan, X.Y.; Ma, F.; Yuan, Z.X.; Chang, L.M.; Jin, X.T. Electrochemical degradation of phenol in aqueous solution using PbO2 anode. J. Taiwan Inst. Chem. Eng. 2013, 44, 95–102. [Google Scholar] [CrossRef]
- Devilliers, D.; Thi, M.T.D.; Mahe, E.; Le Xuan, Q. Cr(III) oxidation with lead dioxide-based anodes. Electrochim. Acta 2003, 48, 4301–4309. [Google Scholar] [CrossRef]
- Aquino, J.M.; Pereira, G.F.; Rocha-Filho, R.C.; Bocchi, N.; Biaggio, S.R. Electrochemical degradation of a real textile effluent using boron-doped diamond or β-PbO2 as anode. J. Hazard. Mater. 2011, 192, 1275–1281. [Google Scholar] [CrossRef]
- Aquino, J.M.; Rocha-Filho, R.C.; Ruotolo, L.A.M.; Bocchi, N.; Biaggio, S.R. Electrochemical degradation of a real textile wastewater using b-PbO2 and DSA® anodes. Chem. Eng. J. 2014, 251, 138–145. [Google Scholar] [CrossRef]
- Chen, Z.; Yu, Q.; Liao, D.H.; Guo, Z.C.; Wu, J. Influence of nano-CeO2 on coating structure and properties of electrodeposited Al/α-PbO2/β-PbO2. Trans. Nonferrous Met. Soc. 2013, 23, 1382–1389. [Google Scholar] [CrossRef]
- Bian, X.; Xia, Y.; Zhan, T.; Wang, L.; Zhou, W.; Dai, Q.; Chen, J. Electrochemical removal of amoxicillin using a Cu doped PbO2 electrode: Electrode characterization, operational parameters optimization and degradation mechanism. Chemosphere 2019, 233, 762–770. [Google Scholar] [CrossRef]
- Xinyu, S.; Xiaoyue, D.; Feng, X.; Limin, C. Fabrication of three-dimensional networked PbO2 anode for electrochemical oxidation of organic pollutants in aqueous solution. J. Taiwan Inst. Chem. Eng. 2019, 100, 74–84. [Google Scholar]
- Li, X.; Pletcher, D.; Walsh, F.C. Electrodeposited lead dioxide coatings. Chem. Soc. Rev. 2011, 40, 3879–3894. [Google Scholar] [CrossRef]
- Yao, Y.; Ren, B.; Yang, Y.; Huang, C.; Li, M. Preparation and electrochemical treatment application of Ce-PbO2/ZrO2 composite electrode in the degradation of acridine orange by electrochemical advanced oxidation process. J. Hazard. Mater. 2019, 361, 141–151. [Google Scholar] [CrossRef]
- Shmychkova, O.; Luk’yanenko, T.; Amadelli, R.; Velichenko, A. Physico-chemical properties of PbO2-anodes doped with Sn4+ and complex ions. J. Electroanal. Chem. 2014, 717, 196–201. [Google Scholar] [CrossRef]
- Wang, X.; Wu, Q.; Ma, H.; Ma, C.; Yu, Z.; Fu, Y.; Dong, X. Fabrication of PbO2 tipped Co3O4 nanowires for efficient photoelectrochemical decolorization of dye (reactive brilliant blue KN-R) wastewater. Sol. Energy Mater. Sol. Cells 2019, 191, 381–388. [Google Scholar] [CrossRef]
- Zhang, Y.; He, P.; Jia, L.; Li, C.; Liu, H.; Wang, S.; Zhou, S.; Dong, F. Ti/PbO2-Sm2O3 composite based electrode for highly efficient electrocatalytic degradation of alizarin yellow R. J. Colloid Interface Sci. 2019, 533, 750–761. [Google Scholar] [CrossRef]
- Li, J.; Li, M.; Li, D.; Wen, Q.; Chen, Z. Electrochemical pretreatment of coal gasification wastewater with Bi-doped PbO2 electrode: Preparation of anode, efficiency and mechanism. Chemosphere 2020, 248, 126021. [Google Scholar] [CrossRef]
- Yao, Y.; Teng, G.; Yang, Y.; Ren, B.; Cui, L. Electrochemical degradation of neutral red on PbO2/α-Al2O3 composite electrodes: Electrode characterization, byproducts and degradation mechanism. Sep. Purif. Technol. 2019, 227, 115684. [Google Scholar] [CrossRef]
- Del Nero, J.; de Araujo, R.E.; Gomes, A.S.L.; de Melo, C.P. Theoretical and experimental investigation of the second hyperpolarizabilities of methyl orange. J. Chem. Phys. 2005, 122, 104506. [Google Scholar] [CrossRef]
- Li, H.Y.; Chen, Y.; Zhang, Y.H.; Han, W.Q.; Sun, X.Y.; Li, J.S.; Wang, L.J. Preparation of Ti/PbO2-Sn anodes for electrochemical degradation of phenol. J. Electroanal. Chem. 2013, 689, 193–200. [Google Scholar] [CrossRef]
- Bhatia, D.; Datta, D.; Joshi, A.; Gupta, S.; Gote, Y. Adsorption study for the separation of isonicotinic acid from aqueous solution using activated carbon/Fe3O4 composites. J. Chem. Eng. Data 2018, 63, 436–445. [Google Scholar] [CrossRef]
- Zhang, Q.; Guo, X.; Cao, X.; Wang, D.; Wei, J. Facile preparation of a Ti/α-PbO2/β-PbO2 electrode for the electrochemical degradation of 2-chlorophenol. Chin. J. Catal. 2015, 36, 975–981. [Google Scholar] [CrossRef]
- Dai, Q.; Zhou, J.; Meng, X.; Feng, D.; Wu, C.; Chen, J. Electrochemical oxidation of cinnamic acid with Mo modified PbO2 electrode: Electrode characterization, kinetics and degradation pathway. Chem. Eng. J. 2016, 289, 239–246. [Google Scholar] [CrossRef]









| Electrode | Binding Energy (eV) | S1 Content % | S2 Content % | |
|---|---|---|---|---|
| S1 | S2 | S1/(S1 + S2) | S2/(S1 + S2) | |
| Ti/α-PbO2 | 530.9 | 528.3 | 92.6 | 7.4 |
| Ti/α-PbO2/β-PbO2 | 530.4 | 529.1 | 72.5 | 27.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Molla, M.A.I.; Yanagi, G.; Furukawa, M.; Tateishi, I.; Katsumata, H.; Kaneco, S. Optimization of Operating Conditions for Electrochemical Decolorization of Methylene Blue with Ti/α-PbO2/β-PbO2 Composite Electrode. J. Compos. Sci. 2021, 5, 117. https://doi.org/10.3390/jcs5050117
Molla MAI, Yanagi G, Furukawa M, Tateishi I, Katsumata H, Kaneco S. Optimization of Operating Conditions for Electrochemical Decolorization of Methylene Blue with Ti/α-PbO2/β-PbO2 Composite Electrode. Journal of Composites Science. 2021; 5(5):117. https://doi.org/10.3390/jcs5050117
Chicago/Turabian StyleMolla, Md. Ashraful Islam, Genta Yanagi, Mai Furukawa, Ikki Tateishi, Hideyuki Katsumata, and Satoshi Kaneco. 2021. "Optimization of Operating Conditions for Electrochemical Decolorization of Methylene Blue with Ti/α-PbO2/β-PbO2 Composite Electrode" Journal of Composites Science 5, no. 5: 117. https://doi.org/10.3390/jcs5050117
APA StyleMolla, M. A. I., Yanagi, G., Furukawa, M., Tateishi, I., Katsumata, H., & Kaneco, S. (2021). Optimization of Operating Conditions for Electrochemical Decolorization of Methylene Blue with Ti/α-PbO2/β-PbO2 Composite Electrode. Journal of Composites Science, 5(5), 117. https://doi.org/10.3390/jcs5050117






