1. Introduction
Unmanned aircraft systems were historically used exclusively by the military [
1]. Currently, the use of remotely piloted aircraft systems (RPAS) and small drones has expanded to perform civilian tasks [
1,
2] such as, supporting search and rescue operations [
2,
3,
4], monitoring weather and traffic flows [
5], delivering goods [
4,
6], and of course as a platform for aerial photography. Drones are being employed to effect change in the environment; a good example is in agriculture, where efficiencies may be made by employing drones to spray fields and track crop growth patterns [
6].
However, one of the most exciting areas of drone development is within the healthcare industries. Such applications include delivering medicines, vaccines [
7], blood [
8] and other medical supplies [
9,
10] that are urgently needed in inaccessible areas. Supply challenges are frequently caused by poor transport networks, extreme weather conditions, natural disasters or traffic congestion in urban areas [
11]. Delivery by drone could be a solution to such problems. An illustrative example is the delivery of medical supplies from the Wise County Regional airport to the clinics in rural Virginia, where the total delivery time over 39 miles was substantially reduced from 90 minutes by road to less than 30 minutes by the use of drones [
12]. United Parcel Service and Zipline have been working on a drone network to allow 50–150 daily deliveries of blood and vaccines to 20 clinics in remote areas of Rwanda, Africa [
4]. The Californian drone company, Matternet, has delivered medications to camps in Haiti following earthquakes [
6] and to remote areas in the Dominican Republic, Lesotho, Bhutan [
2], Papua New Guinea and Switzerland using automated drones with capacities of up to 2 kg, which were flown over 10 km within 18 minutes [
12]. Following the testing in Lesotho, Matternet estimated that it would cost
$900,000 to operate 50 base stations and 150 drones (24 cents/flight) throughout the capital city of Maseru, which could be more cost-effective than building roads (estimated cost to build a 2 km one-lane road =
$1 million) [
2]. In Germany, three generations of parcelcopter operated by DHL Parcel have been tested respectively to deliver blood samples across the Rhine River at Bonn (1 km), deliver medications and other urgently needed materials to Juist (a North Sea island 12 km off the coast of Germany) [
6], and deliver 130 parcels (~2 kg each) of urgently needed medicines in January–March between automated Skyports in Reit im Winkl and Winklmoosalm (two Bavarian Alpine villages), which reduced the 30-minute journey time by road in winter to eight minutes [
12].
In the healthcare context, the timely delivery of medications, vaccines and blood is crucial [
12]. Drones can potentially overcome the logistic challenges as they are not subjected to traffic delays [
13], and most importantly, they are able to reach regions that lack adequate roads faster. However, for delivery of fragile medications, blood [
14] and diagnostic clinical laboratory specimens [
15,
16,
17], drones are practical only if the quality of transported products is not adversely affected. Thus, drones as a novel method for medicine transportation, must be tested to determine their impact on medicine quality.
In the United Kingdom, drone delivery has a potential to thrive as reported in Nesta’s Flying High report July 2018 [
5]. The Flying High Challenge explored the use of drones in urban areas and the technical and economic feasibility of delivering blood samples or medical supplies between hospitals (Guy’s and St Thomas’) was evaluated [
5]. A number of challenges were identified, in particular, the legislations requiring flying drones in the line-of-sight [
18,
19]. Apart from such regulatory and safety issues, drones exhibit some technical limitations [
4] with regards to the battery life, payload capacity and maximum coverable distance [
12]. Furthermore, the misuse of drones to deliver illegal drugs to prisons has led to low public confidence and acceptance of this technology [
12,
20]. Critically however, no information on the quality of medicines delivered using drones was considered in the report, which highlights a significant knowledge gap. While a few pilot studies have been carried out on the drone delivery of medicines to remote areas [
4,
6], there has been little consideration of the impact of the flight conditions on the quality of the drone-delivered medicines. Hence, the feasibility of delivering a medicine that is highly sensitive to environmental stresses is investigated in this study.
Current unmanned aircraft systems (UAS) or drones may be generally classified into fixed-wing or rotary-wing aircraft. Fixed-wing UAS are more efficient, have longer ranges and have greater payloads; however, rotary-wing drones take off and land vertically with the ability to hover during flight. For drone delivery of medicines in urban situations, rotor-based drones are more applicable, whereas rural locations benefit from the greater range associated with the fixed-wing aircraft. Udroiu and Blaj (2016) have made a compelling case for the design of vertical take off and landing (VTOL) drones that incorporate tilting wing technology and thus combine the benefits of both rotor- and fixed-wing aircraft. For all drone configurations the same critical process parameters associated with flight will impact on the stability of medicines transported by these systems.
Insulin was chosen as the model medication in this investigation as it is one of the most important life-saving drugs in treating type I diabetes [
21]. Moreover, insulin is a sensitive peptide-based drug, which easily unfolds to cause irreversible aggregation when subjected to environmental stresses [
22], such as high temperatures and exposure to vibration/agitation. Alteration of insulin’s structure will impair its biological efficacy [
22]. This study highlights the importance of maintaining insulin quality during drone transportation. Any compromise in medicine quality is likely to be detrimental to a patient’s health and wellbeing.
The aim of our study is to examine the impact of drone transport on a form of human insulin used by diabetics. We selected the medicinal product, Actrapid and flew this product contained in its original packaging, glass vials held within a cardboard box, by a small, commercially available drone.
Under a quality by design, QbD, framework, (International Council for Harmonisation, Q8 [
23]), a set of critical quality attributes (CQA) is established to ensure quality, safety and efficacy of the medicine for the patient. Critical material attributes (CMA) represent a set of properties for an input material that should be within limits to ensure the desired quality of output material. In this study, we have selected the formation of irreversible aggregates as an indicator of insulin instability [
24]. Analytical-grade insulin was used to develop and validate the stability-indicating tests applied in this study. A set of critical process parameters (CPP), whose variability has an impact on the CQA, should be monitored or controlled to ensure the process produces the desired quality. In this study, the primary critical process parameters of interest selected were temperature and vibration, along with air pressure and relative humidity (RH%),
Table 1.
To date, there is no published work on the impact of drone delivery on insulin quality. Obtaining this information will help to determine the feasibility of drone transportation of highly sensitive medicines.
4. Discussion
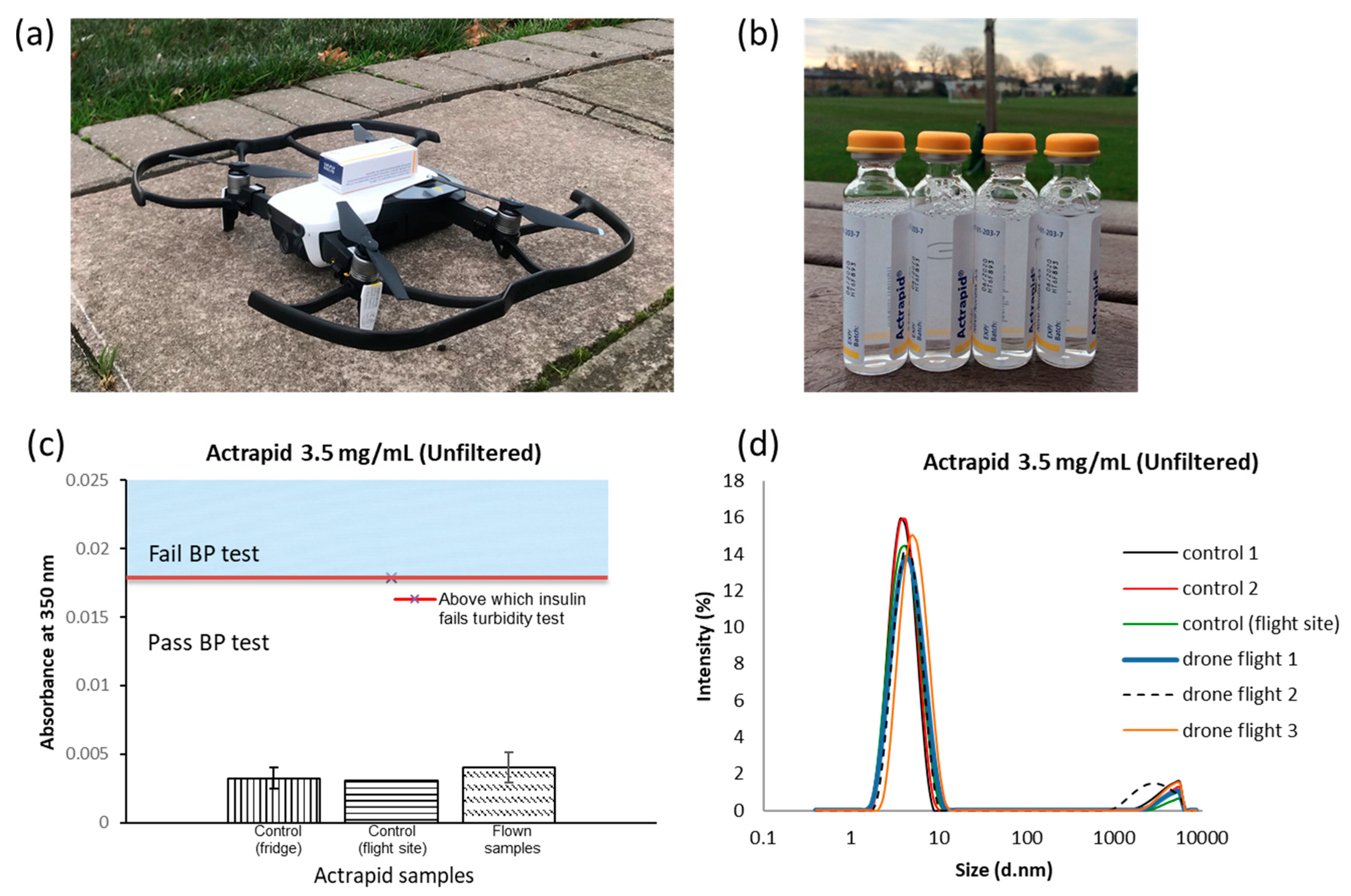
The formazin turbidity standards, the basis of the pharmacopoeia quality tests, were a dispersion of small colloidal particles. The principle of turbidity measurement is based on the attenuation of incident beams due to light scattering by particles [
26]. Thus, an increase in absorbance at 350 nm was related to the increase in the concentration of formazin particles. The robustness of formazin as an effective turbidity standard was clearly shown from the repeatability of absorbance readings (
Supplementary Materials S1). If medicines are to be regularly delivered by drones, then their quality tests upon receipt must be simple to perform, non-destructive and easy to interpret. The British Pharmacopoeia degree of opalescence test based on formazin meets these criteria, as it is based on the mixing of liquids and visual inspection of the intact glass vial [
24], which further supports the potential of insulin to be transport by drones.
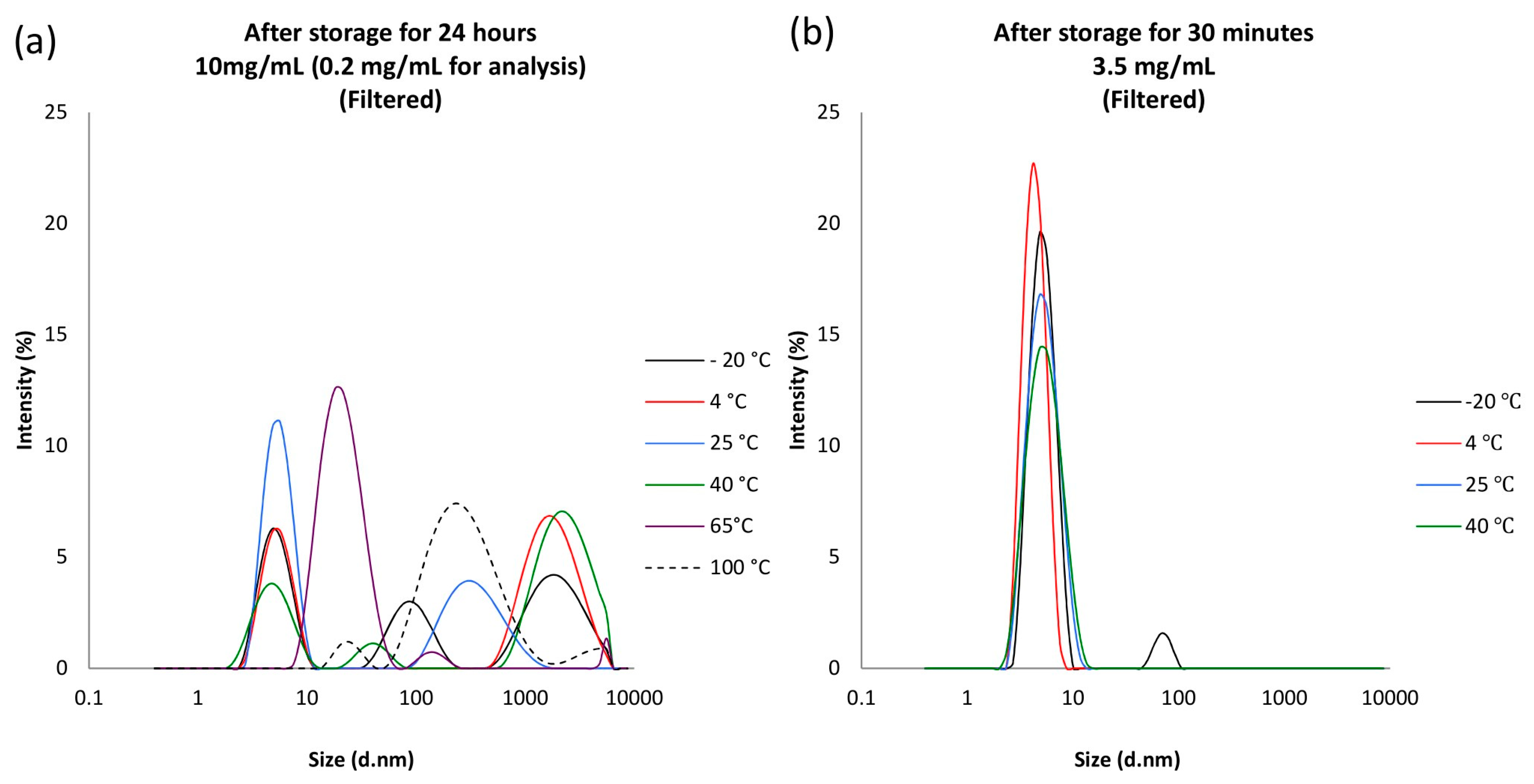
Apart from the formation of large fibrils, insulin solutions are also known to form much smaller aggregates in the nm region [
26], an order of magnitude smaller than the formazin particles [
36,
37]. DLS was used to complement the UV turbidity analysis as it is a sensitive method for measuring such small-sized aggregates, 1 to 450 nm in diameter [
38,
39]. The time-dependent fluctuations in the scattering intensity as a result of Brownian motion are measured by DLS, the frequency of these fluctuations are used to determine particle diffusion coefficients and particle size [
38]. Based on Rayleigh’s approximation, the scattering intensity of a particle is proportional to the sixth power of its diameter [
38]. DLS successfully showed the presence of the reversible smaller aggregates in all of the insulin solutions investigated, but where larger fibril aggregates appear, the BP opalescence test was definitive for identifying failing samples as a result of this physical instability.
The purpose of investigating analytical grade insulin was to develop stability testing protocols that would mimic the range of conditions possible during drone flight. Extreme temperatures (e.g., 100 °C) and storage times (24 hours) were also applied to guarantee at least some failures of insulin stability and thus validate the quality tests. It was not possible to purchase analytical grade insulin at the same concentration as that found in Actrapid, the analytical grade required dilution. Ultra-purewater (pH 7) was used to minimise heterogeneous nucleation [
27] and mimic physiological pH; however, insulin solutions for injection are typically formulated in mildly acidic conditions to maintain solubility. Although a pH of 7 is above insulin’s isoelectric point at pH 5.4, where the molecule is not charged, the authors investigated the effect of dilution by acid (0.01 M HCl) on the representative stability samples to allow comparison with the insulin solutions prepared using ultra-pure water (
Supplementary Materials S2), which demonstrated no effect on the degree of opalescence.
Physical instability, i.e., irreversible aggregation, inactivates insulin [
40]. High temperatures facilitate protein unfolding, and increase the diffusion and collision rates of the unfolded insulin molecules. Thus, at high temperatures an activation energy barrier is overcome and large aggregates form [
30,
41], as was observed for insulin solutions held at 65 and 100 °C. The mechanism of aggregation and eventual precipitation of insulin is complex, but it may be simplified by the following schematic.
High temperatures, shear stress, exposure to vibration/agitation, increase in ionic strength and contact with hydrophobic surfaces induce conformational change within the insulin molecule present in aqueous solution, leading to the unfolding of the insulin monomer [
42]. The unfolded monomers are not stable in water, and to lower the free energy of the system, they come together to form a fibril [
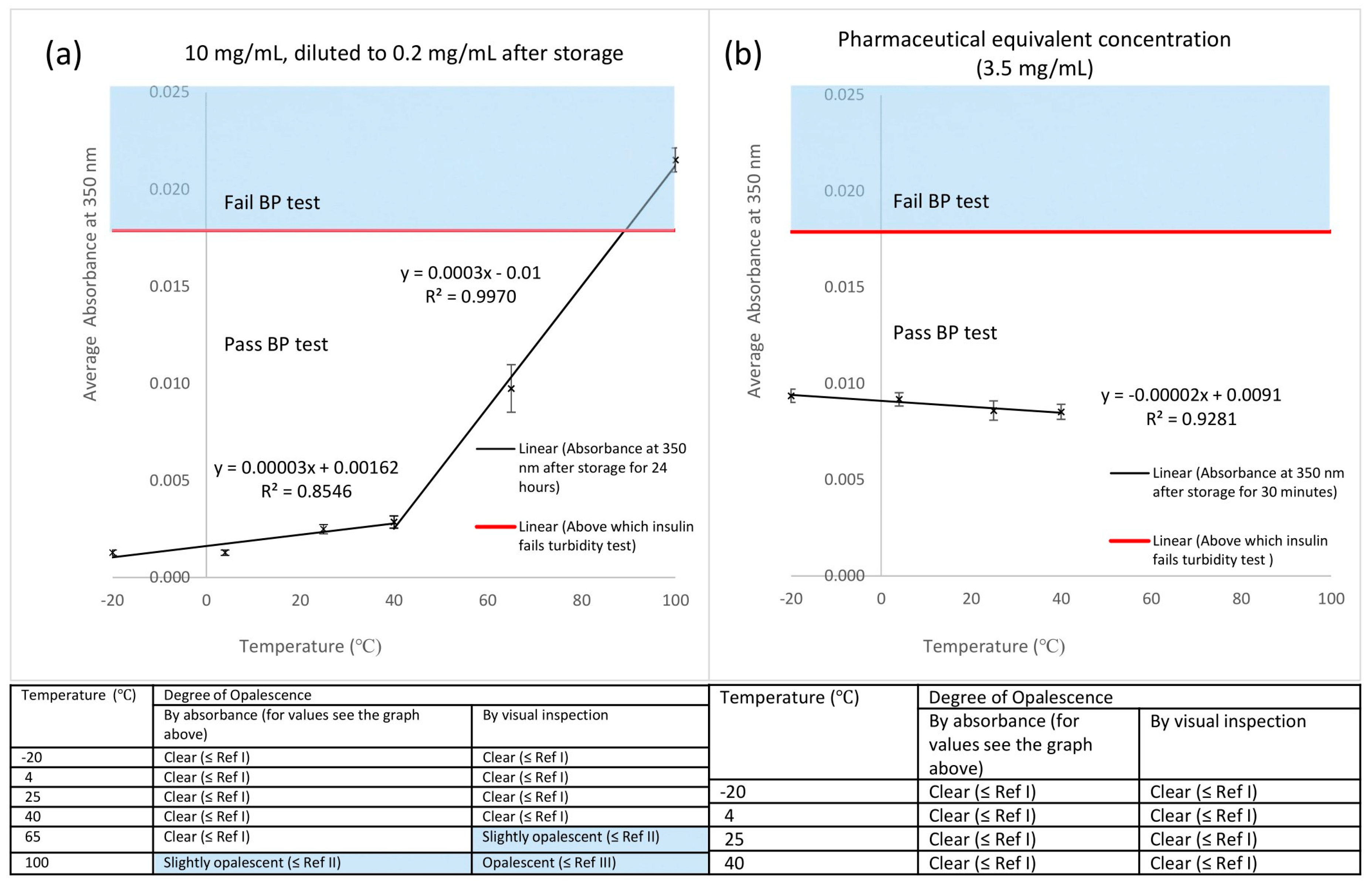
42]. Fibrils contain many hundreds of insulin molecules, where the insulin is irreversibly bound together into structures many mm long that eventually precipitates out of the solution. After storage at 65 and 100 °C for 24 hours, such fibrils are easily detected by visual inspection (
Figure 1a). To avoid the formation of fibrils, zinc ions in the form of zinc chloride are often added to pharmaceutical grade formulations of insulin to increase stability, and this is the case for Actrapid. Zinc ions stabilise the formation of the reversible smaller insulin aggregates, namely tetramers and hexamers. Insulin formulations containing these structures are regarded as stable, since they are in equilibrium with the monomeric form of insulin. Tetramers and hexamers were detected in the drone-delivered Actrapid and all solutions of insulin stored at or below 40 °C. The analytical grade of insulin used for the storage experiments did not contain zinc ions. Even after storage at 40 °C for 24 hours insulin fibrils were not detected in these samples, indicating that potentially less stable formulations of insulin could also be transported by a drone.
Settling of aggregates in solution [
43] may have contributed to the slightly lower absorbance values obtained than expected for insulin stored at 65 °C, giving results where insulin passed the turbidity test by absorbance but not by visual inspection. Whereas the large number of fibrils, i.e., precipitated insulin, after storage at 100 °C were easily detected in both the visual and UV-based turbidity assays. Although insulin stored at −20 °C passed the BP test and showed insignificant difference in absorbance readings with the control (
p = 0.71), studies report that freeze-thawing compromises insulin quality [
41,
44]. Freezing induces aggregation by local concentration of protein, causing a variation in protein distribution throughout the frozen mass [
41,
45]. This can be observed in the DLS measurements with the detection of a small population of particles close to 100 nm for the 3.5 mg/mL concentrations of insulin stored at −20 °C for 30 mins. Thus, when flying insulin samples by drone in colder environments, or at high altitude, partial freezing within the solutions needs to be considered.
The simulated laboratory tests investigating the effects of temperature and vibration explored the edge of failure and were used to validate the flight-testing experiments. The temperature range of interest for drone delivery is −20 to +40 °C [
46]. The storage temperature of −20 °C simulated drone delivery on days with sub-zero temperatures or while flying to reach, for example, communities in the Alps at high altitudes [
47]. While the 40 °C storage conditions simulated drone delivery in countries with hot climates. As insulin is highly temperature-sensitive, the development of on-board active cooling or heating devices will be important for the maintenance of insulin quality and compliance with the storage requirement made by the manufacturer of between 2 and 8 °C [
48]. However, our work indicates that higher storage temperatures for insulin over short periods may be acceptable, as supported by the samples stored at 40 °C for 30 minutes passing the BP test, which reinforces the potential for safe delivery of insulin by drones in hotter climates.
The operating temperature of the DJI Mavic air drone is between 0 to +40 °C. For specific missions, imaging Arctic environments and surveying remote infrastructures for example, several commercial drones have operated successfully at temperatures well below zero, (e.g., a DJI phantom 4 was flown at a temperature approximately −10 °C at the Tarfala Research Station Sweden [
49]. The major limitations for these applications are the reduction in working time for the lithium-based batteries. Improvements in battery technology is ongoing, thus in this study it was important to simulate in the laboratory the impact of colder temperatures (e.g., −20 °C) when considering the delivery of medicines using drones in potentially colder climates. The upper temperature limit of stability testing is set at +40 °C in pharmaceutical regulations; however, in very extreme environments, tropical climates for example, drones will experience temperatures closer to +50 °C or above. Thus, the temperature stressing experiments reported in this study covered such extreme values.
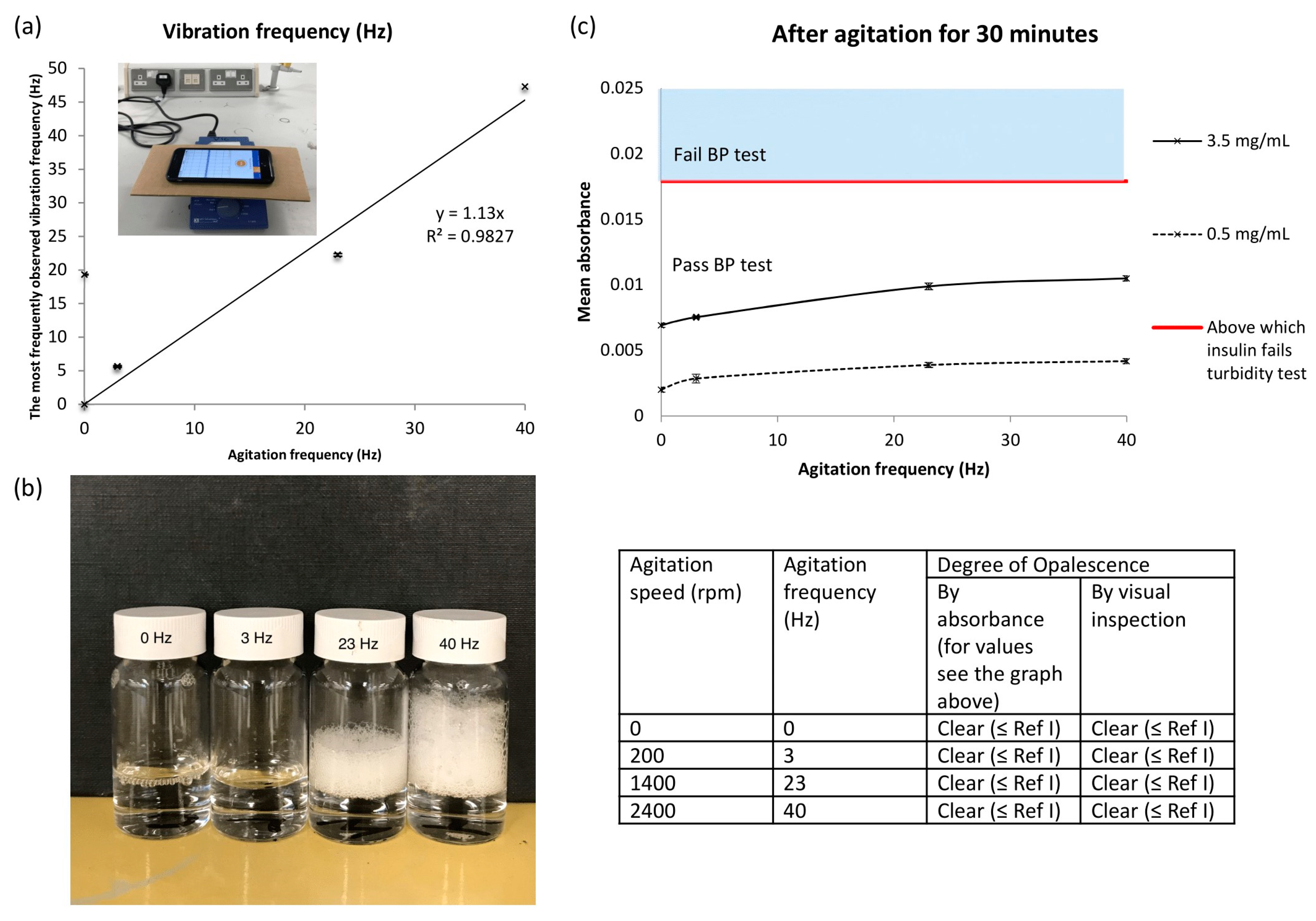
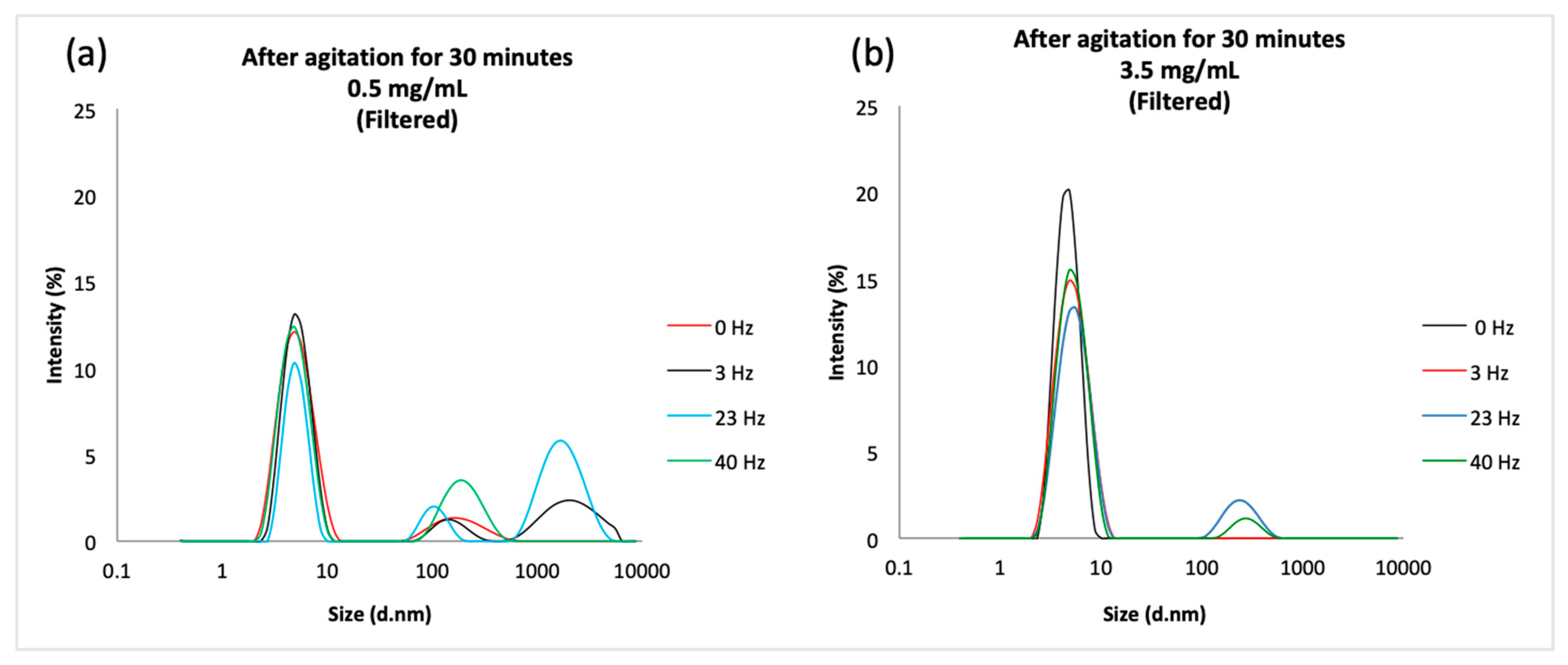
Agitation by a vortex mixer was performed to model the impact of vibrations potentially generated by drone flight. As the agitation frequency increased, the major peaks observed in the DLS remained close to 4.85 nm, which corresponded to the tetrameric form of insulin previously measured by Pease et al. [
32]. Hence, it can be deduced that insulin remained stable after agitation. Furthermore, since insulin solutions subjected to 3 Hz at 25 °C for 30 minutes passed the BP turbidity test, and 3 Hz was close to the maximum frequency observed in the drone flight tests, it would appear that vibration is not an issue for the delivery of Actrapid using the small DJI Mavic Air drone. However, in the vortexing experiments and in the drone flight tests, frothing was observed at the surface of the solutions. Intense shaking induces shear and cavitation within solutions, where rapid collapse of the bubbles formed produces shock waves and turbulent flow [
30]. This causes the protein molecules to realign at the air–water interface and expose the inner hydrophobic core of protein to the air, causing conformational changes to insulin monomers from a predominantly α-helical state to a β-sheet rich conformation [
43,
50]. Interactions between partially unfolded monomers then shift the aggregation equilibrium towards the formation of fibrils [
50]. Even though such fibril formation was not observed in the Actrapid solutions flight tested here, a small amount of froth formation was observed. Thus, measuring vibrations within liquid-based protein medications delivered by a drone still needs to be considered [
51,
52]. Possible approaches to reduce the impact of in-flight vibration include head space reduction above solutions in vials or the use of packaging materials that do not transmit vibration [
52]. During normal flight, drones are designed to have minimal vibration to increase efficiency and maintain stability [
52,
53], this may not be the case during launch or as a result of a collision or even crash landing. Innovative dampening systems are employed to protect more advanced drones from sudden vibrations or collisions, for example, the PackDrone from Dronistics which contains the drone within a cage [
51]. In case of collision, the cage works as a damper to absorb energy from the shock and protect packages [
52].
The vibrations suffered by medicines transported using rotor-based drones could be minimized by increasing the number of rotors in order to increase stability, noting that the performance of the blades must be tuned accurately. Alternatively, the gimbal technology used for camera mounts could easily be exploited for the transport of medicines. In the case of fixed-wing drones, smooth take off and landing surfaces would be optimal for reducing vibration. There is very limited data available providing the vibrations experienced by packages transported using rotor-based drones, thus a good estimate of the potential exposure is given in
Supplementary Materials Figure S4, where the most significant vibrations range between 0.1 and 10 Hz.
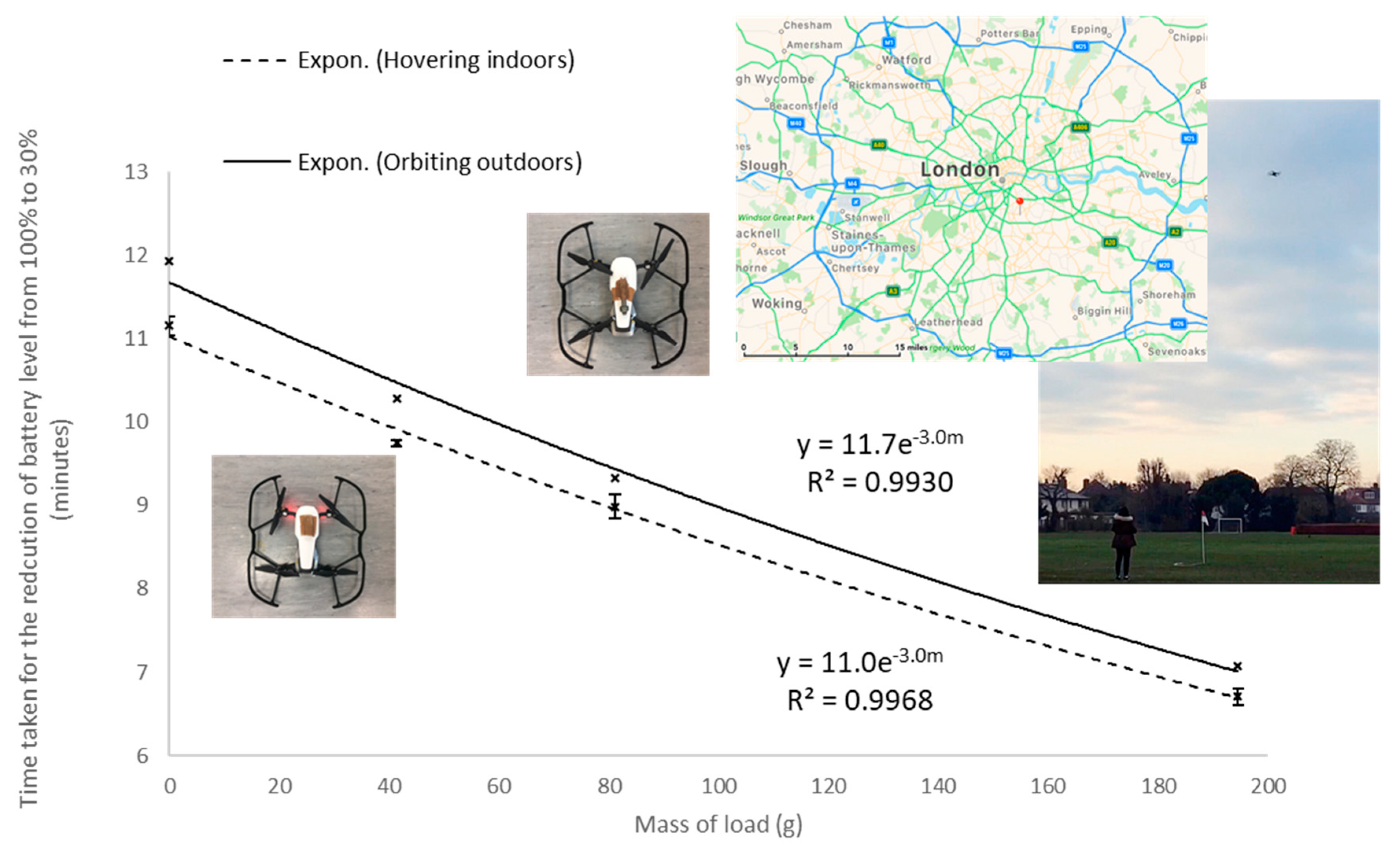
The drone used here was compact (430 g) compared to the drones currently designed for the transportation of parcels [
52,
53], but the approach towards safe flight time testing is transferable from our study to larger systems. Medicines are potentially dangerous, thus setting the battery limit of 30% is recommended to allow the safe landing for a drone transporting a medicine. Determining the relationship between the mass of the load carried and the safe flight time, or safe range, is vital as different packaged medicines have different gross weights. Modelling this relationship is essential for mapping the edge-of-failure and thus aiding in the selection of the best drone for the required delivery task. The time taken for battery level reduction from 100% to 30% decreased exponentially with the increase in mass of loads (
Section 3.4). Greater power was needed to lift heavier loads, hence the reduced flight time [
54]. Although flight times, speed and distance covered will vary depending on atmospheric conditions, for example wind speed, it was encouraging that the indoor and outdoor testing gave similar exponential decays between safe flight time and mass of load. A study on blood products transportation in Baltimore, Maryland discovered that temperature and atmospheric pressure change with altitude are small, 0.68 °C/100 m and 12 mbar/100 m, respectively [
14]. The maximum allowable altitudes for drone flights are set very conservatively; in the United Kingdom this is 400 ft or approximately 120 m. Thus, the impact of altitude on the temperature and pressures experienced by a medicine on board a drone is expected to be limited under current regulations for the United Kingdom. Applying the exponential equation for safe flight time determined from the outdoor experiments, (
Section 3.4), the maximum safe range to fly an Actrapid package (23.8 g) was just under 1 km. This was much lower than the manufacturers claim of a 4 km range unloaded, but it should be noted that DJI Mavic Air is not designed for transporting packages, and the experimental flights involved orbits around a fixed point rather than straight-line flights. Tight circular motion will reduce the efficiency of a drone due to the extra centrifugal forces. The flight speed of 1.5 m/s and wide loop of 10 m used here indicate that these additional forces are likely to be low. For the transport of Actrapid between King’s College Hospital in London and local clinics, a 1 km radius would be useful as this encompasses over 10 GP surgeries and pharmacies combined; but for the delivery of Actrapid to other locations a more powerful drone is required. Current drone designs suggest safe flight times for such applications to be close to 30 minutes [
12]. The outcome of this study indicates that insulin solutions transported for these drone flight times would arrive in a stable condition and pass the BP quality test.
Many of the barriers to the routine delivery of medicines by drones are still in place but the study reported here shows that this technology, after the required testing and validation, has the potential to deliver sensitive peptide-based drugs. The future of clinically useful drone deliveries requires the relaxation of regulations around flying beyond the line of sight [
14]. In Switzerland, drones have been approved to fly autonomously to carry blood products and cytostatic materials between hospitals in static routes since October 2017 [
46]. However, the integration of drones into air traffic control systems remains unsolved [
45], and authorisation of an approved route usually takes ~8−18 months [
46]. Battery life is yet another barrier. According to Swiss Post, the current technology allows one battery to last for 20 km (speed = 1 km/min) [
46]. Data loggers enabling live recording of the temperature of material have been developed to ensure the maintenance of the cold chain [
46]. The temperatures measured by such studies were within the range of temperatures reported in this study. As drones are extremely weight-sensitive, the maximum payload will be reduced if on board active cooling systems have to be added [
14,
46]. A number of manufacturers are positive with respect to this issue as their technologies allow a payload capacity of 2 kg, but the challenge is to design a larger carrier box to accommodate on board cooling devices [
46,
53]. Future developments could include drones with infrared/thermal imaging cameras [
54] or directional microphones, enabling quick assessment of victims in emergency situations before dispensing the required medication [
46,
52]. Securing the supply chain to avoid drone-delivered medicines being redirected for illicit purposes is an issue that must be addressed. Initiatives such as on board anti-tamper monitoring aim to ensure security and authenticity of the supply chain [
53].
Our study provides much needed stability data showing that drones have the potential to safely deliver medicines. Insulin is a very important medicine, but more studies are required that encompass a wider range of medicines that show promise for drone delivery. After discussing our work with pharmacists and clinical colleagues [
55], it was agreed that the direction of travel should be towards medicines used for emergency situations. Many medicines make good candidates for delivery by drone (
Table 2), for example; adrenaline, thrombolytics, nitroglycerin and beta blockers for cardiac arrests; vaccines for local outbreaks of disease; portable nebulizers in the case of severe asthma attacks; and antibiotics for suspected sepsis. Speed of delivery is the beneficial attribute provided by the drone. Interestingly, to avoid the creation of large stocks of opioids in care and private homes, when break through pain is registered by a patient undergoing palliative care, a quick delivery of the required medication at the required dose for the pain management specific to the patient’s needs would avoid potential redirection and abuse of surplus opioid stocks.
5. Limitations and Opportunities
The limitations of our study are concerned with the relatively small size of the drone used. Regulations prevented a large drone being flown in London, so only a small drone was used for the proof of concept. As stated, this was fit for the purpose of evaluating drone transport between hospitals and clinics within inner cities, where the flight paths are very short (1 km); but to improve, future work needs to investigate fixed-wing UAS and VTOL’s that operate over much larger distances (40 km). It is to be expected that in-flight vibration will be different for these UAS compared to the quad-rotor drone used here, which further supports measuring in-flight vibration frequencies, as described in
Section 2.4 and
Section 3.3 in this paper, when evaluating a specific VTOL for the delivery of liquid-based or fragile medicines. The study here investigated flying in orbits which is not representative of any typical UAS flight paths, especially over rural areas. However orbits are closer, in part, to the tight turns required for flight in some urban settings. Thus, the approach outlined in this study needs to be applied to medicines transported in a broader range of drones that have the capacity to fly a greater range missions that cover much longer distances, up to 40 km.
Using real-life scenario dispatching drones, that fly beyond the visual line of sight (BVLOS) would not affect the recommendations generated in this paper, see
Section 6.1, but the ranges of the critical process parameters used for stability simulation would have to be confirmed. For example, 6 and 8 rotor drones are more stable, and any medicine carried by them would be exposed to lower intensity vibrations than the quad drone used in the present paper. Real-world BVLOS missions using both rotor and fixed-wing UAS would additionally require data loggers placed in the packaging. Data loggers are commonly used in the supply chain for pharmaceuticals and record temperature, and some even vibration, over time. Such devices were precluded from this study because of the space available on the DJI Mavic Air drone, but for BVLO missions using larger drones these loggers would need to be carried onboard and within any additional packaging, to record any potential changes in temperature during the mission as a consequence of the greater flight range.
The length of the lab tests, 30 minutes, was set to mimic the typical maximum flight times of commercially available multi-rotor drones that take off and land vertically, a facility required for delivering medicines in urban environments. The drone flight tests presented here were conducted within the safe operating limits, i.e., flying from 100% battery charge level down to 30%, to allow landing with absolutely no chance of drone failure. Once loaded the safe flight times reduced to 9 minutes. The 9-minute flight vs. the 30-minute lab tests may have introduced bias, for example, a 30-minute flight may have led to the freezing of the Actrapid sample, as the temperature over the flight path was between −1 and 0 °C. Ice was not observed in the flight tests and is not expected to occur if the samples were flown for 30 minutes, as the zinc chloride present depressed the freezing point of the water in the samples. Although, the impact of freeze-thawing on insulin dispersions was fully explored in the lab and these samples still passed the BP quality tests. The vibration tests in the lab lasted for 30 minutes and were conducted at 25 °C. In the drone tests Actrapid was exposed to nine minutes of flight vibrations (0.1 to 3.4 Hz) at −1 to 0 °C. Degradation of insulin due to vibration requires insulin molecules to diffuse to the air–water interface and then unfold, both diffusion and molecular rotation are proportional with temperature, thus the chances of observing vibration initiated degradation would have been much lower in the drone flights compared to the lab tests. Thus, the lab tests were carried out for an extended time and over a wide range of vibrational frequencies in order to capture any potential vibrational degradation, none was observed.