Insights into Australian Bat Lyssavirus in Insectivorous Bats of Western Australia
Abstract
1. Introduction
2. Materials and Methods
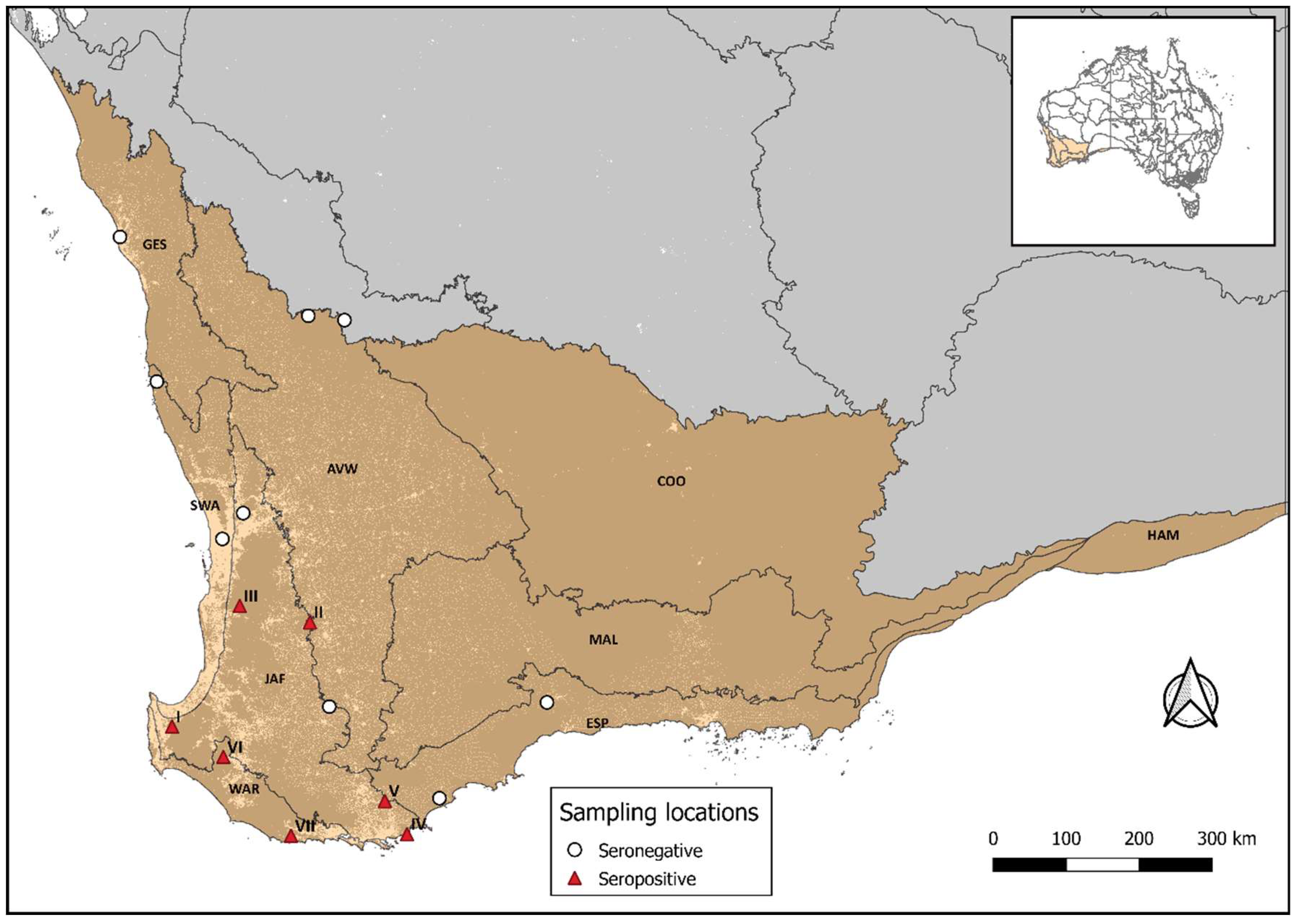
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- International Committee on Taxonomy of Viruses. Available online: https://talk.ictvonline.org/taxonomy/ (accessed on 14 February 2019).
- Fraser, G.C.; Hooper, P.T.; Lunt, R.A.; Gould, A.R.; Gleeson, L.J.; Hyatt, A.D.; Russell, G.M.; Kattenbelt, J.A. Encephalitis Caused by a Lyssavirus in Fruit Bats in Australia. Emerg. Infect. Dis. 1996, 2, 327–331. [Google Scholar] [CrossRef] [PubMed]
- Gould, A.R.; Hyatt, A.D.; Lunt, R.; Kattenbelt, J.A.; Hengstberger, S.; Blacksell, S. Characterisation of a novel lyssavirus isolated from Pteropid bats in Australia. Virus Res. 1998, 54, 165–187. [Google Scholar] [CrossRef]
- Gould, A.R.; Kattenbelt, J.A.; Gumley, S.G.; Lunt, R.A. Characterisation of an Australian bat lyssavirus variant isolated from an insectivorous bat. Virus Res. 2002, 89, 1–28. [Google Scholar] [CrossRef]
- Field, H.E. Evidence of Australian bat lyssavirus infection in diverse Australian bat taxa. Zoonoses Public Health 2018, 65, 742–748. [Google Scholar] [CrossRef] [PubMed]
- Francis, J.R.; Nourse, C.; Vaska, V.L.; Calvert, S.; Northill, J.A.; McCall, B.; Mattke, A.C. Australian Bat Lyssavirus in a child: The first reported case. Pediatrics 2014, 133, e1063–e1067. [Google Scholar] [CrossRef] [PubMed]
- Shinwari, M.W.; Annand, E.J.; Driver, L.; Warrilow, D.; Harrower, B.; Allcock, R.J.N.; Pukallus, D.; Harper, J.; Bingham, J.; Kung, N.; et al. Australian bat lyssavirus infection in two horses. Vet. Microbiol. 2014, 173, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Hanna, J.N.; Carney, I.K.; Smith, G.A.; Tannenberg, A.E.; Deverill, J.E.; Botha, J.A.; Serafin, I.L.; Harrower, B.J.; Fitzpatrick, P.F.; Searle, J.W. Australian bat lyssavirus infection: A second human case, with a long incubation period. Med. J. Aust. 2000, 172, 597–599. [Google Scholar] [PubMed]
- Australian Technical Advisory Group on Immunisation (ATAGI). Australian Immunisation Handbook. Available online: https://immunisationhandbook.health.gov.au/ (accessed on 14 February 2019).
- Field, H. The Ecology of Hendra Virus and Australian Bat Lyssavirus. Ph.D. Thesis, University of Queensland, Brisbane, Australia, 2004. [Google Scholar]
- Young, M.K.; El Saadi, D.; McCall, B.J. Preventing Australian Bat Lyssavirus: Community knowledge and risk perception of bats in southeast Queensland. Vector-Borne Zoonotic Dis. 2014, 14, 284–290. [Google Scholar] [CrossRef] [PubMed]
- Paterson, B.J.; Butler, M.T.; Eastwood, K.; Cashman, P.M.; Jones, A.; Durrheim, D.N. Cross sectional survey of human-bat interaction in Australia: Public health implications. BMC Public Health 2014, 14, 58. [Google Scholar] [CrossRef] [PubMed]
- Quinn, E.K.; Massey, P.D.; Cox-Witton, K.; Paterson, B.J.; Eastwood, K.; Durrheim, D.N. Understanding human—Bat interactions in NSW, Australia: Improving risk communication for prevention of Australian bat lyssavirus. BMC Vet. Res. 2014, 10, 144. [Google Scholar] [CrossRef] [PubMed]
- McCall, B.J.; Epstein, J.H.; Neill, A.S.; Heel, K.; Field, H.; Barrett, J.; Smith, G.A.; Selvey, L.A.; Rodwell, B.; Lunt, R. Potential exposure to Australian bat lyssavirus, Queensland, 1996-1999. Emerg. Infect. Dis. 2000, 6, 259–264. [Google Scholar] [CrossRef] [PubMed]
- Francis, J.R.; McCall, B.J.; Hutchinson, P.; Powell, J.; Vaska, V.L.; Nourse, C. Australian bat lyssavirus: Implications for public health. Med. J. Aust. 2014, 201, 647–649. [Google Scholar] [CrossRef] [PubMed]
- Churchill, S. Australian Bats, 2nd ed.; Allen and Unwin: French Forest, Australia, 1998. [Google Scholar]
- Wardell-Johnson, G.; Wardell-Johnson, A.; Bradby, K.; Robinson, T.; Bateman, P.W.; Williams, K.; Keesing, A.; Braun, K.; Beckerling, J.; Burbridge, M. Application of a Gondwanan perspective to restore ecological integrity in the south-western Australian global biodiversity hotspot. Restor. Ecol. 2016, 24, 805–815. [Google Scholar] [CrossRef]
- Smith, I.L.; Northill, J.A.; Harrower, B.J.; Smith, G.A. Detection of Australian bat lyssavirus using a fluorogenic probe. J. Clin. Virol. 2002, 25, 285–291. [Google Scholar] [CrossRef]
- Wadhwa, A.; Wilkins, K.; Gao, J.; Condori Condori, R.E.; Gigante, C.M.; Zhao, H.; Ma, X.; Ellison, J.A.; Greenberg, L.; Velasco-Villa, A.; et al. A Pan-Lyssavirus Taqman Real-Time RT-PCR assay for the detection of highly variable rabies virus and other lyssaviruses. PLoS Negl. Trop. Dis. 2017, 11, e0005258. [Google Scholar] [CrossRef] [PubMed]
- Rahmadane, I.; Certoma, A.F.; Peck, G.R.; Fitria, Y.; Payne, J.; Colling, A.; Shiell, B.J.; Beddome, G.; Wilson, S.; Yu, M.; et al. Development and validation of an immunoperoxidase antigen detection test for improved diagnosis of rabies in Indonesia. PLoS Negl. Trop. Dis. 2017, 11, e0006079. [Google Scholar] [CrossRef] [PubMed]
- Bossart, K.N.; McEachern, J.A.; Hickey, A.C.; Choudhry, V.; Dimitrov, D.S.; Eaton, B.T.; Wang, L.-F. Neutralization assays for differential henipavirus serology using Bio-Plex Protein Array Systems. J. Virol. Methods 2007, 142, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Hayman, D.T.S.; Suu-Ire, R.; Breed, A.C.; McEachern, J.A.; Wang, L.; Wood, J.L.N.; Cunningham, A.A. Evidence of Henipavirus infection in West African fruit bats. PLoS ONE 2008, 3, e2739. [Google Scholar] [CrossRef] [PubMed]
- Hayman, D.T.S.; Wang, L.-F.; Barr, J.; Baker, K.S.; Suu-Ire, R.; Broder, C.C.; Cunningham, A.A.; Wood, J.L.N. Antibodies to henipavirus or henipa-like viruses in domestic pigs in Ghana, West Africa. PLoS ONE 2011, 6, e25256. [Google Scholar] [CrossRef] [PubMed]
- Plowright, R.K.; Field, H.E.; Smith, C.; Divljan, A.; Palmer, C.; Tabor, G.; Daszak, P.; Foley, J.E. Reproduction and nutritional stress are risk factors for Hendra virus infection in little red flying foxes (Pteropus scapulatus). Proc. R. Soc. B Biol. Sci. 2008, 275, 861–869. [Google Scholar] [CrossRef] [PubMed]
- Breed, A.C.; Yu, M.; Barr, J.A.; Crameri, G.; Thalmann, C.M.; Wang, L.F. Prevalence of henipavirus and rubulavirus antibodies in pteropid bats, Papua New Guinea. Emerg. Infect. Dis. 2010, 16, 1997–1999. [Google Scholar] [CrossRef] [PubMed]
- Brown, L.D.; Cai, T.T.; Dasgupta, A. Interval estimation for a binomial proportion. Stat. Sci. 2001, 16, 101–133. [Google Scholar] [CrossRef]
- Aragon, T. epitools: Epidemiology Tools, R Package Version 0.5-10; Published 26 October 2017; Available online: https://CRAN.R-project.org/package=epitools (accessed on 21 February 2019).
- Hammarin, A.L.; Berndtsson, L.T.; Falk, K.; Nedinge, M.; Olsson, G.; Lundkvist, Å. Lyssavirus-reactive antibodies in Swedish bats. Infect. Ecol. Epidemiol. 2016, 6, 31262. [Google Scholar] [CrossRef] [PubMed]
- Wakeley, P.R.; Johnson, N.; McElhinney, L.M.; Marston, D.; Sawyer, J.; Fooks, A.R. Development of a real-time, TaqMan reverse transcription-PCR assay for detection and differentiation of lyssavirus genotypes 1, 5, and 6. J. Clin. Microbiol. 2005, 43, 2786–2792. [Google Scholar] [CrossRef] [PubMed]
- Deubelbeiss, A.; Zahno, M.-L.; Zanoni, M.; Bruegger, D.; Zanoni, R. Real-Time RT-PCR for the detection of lyssavirus species. J. Vet. Med. 2014, 2014, 476091. [Google Scholar] [CrossRef] [PubMed]
- Echevarría, J.E.; Avellón, A.; Juste, J.; Vera, M.; Ibáñez, C. Screening of active lyssavirus infection in wild bat populations by viral RNA detection on oropharyngeal swabs. J. Clin. Microbiol. 2001, 39, 3678–3683. [Google Scholar] [CrossRef] [PubMed]
- Amengual, B.; Bourhy, H.; López-Roig, M.; Serra-Cobo, J. Temporal dynamics of European bat lyssavirus type 1 and survival of Myotis myotis bats in natural colonies. PLoS ONE 2007, 2, e566. [Google Scholar] [CrossRef] [PubMed]
- Franka, R.; Johnson, N.; Muller, T.; Vos, A.; Neubert, L.; Freuling, C.; Rupprecht, C.E.; Fooks, A.R. Susceptibility of North American big brown bats (Eptesicus fuscus) to infection with European bat lyssavirus type 1. J. Gen. Virol. 2008, 89, 1998–2010. [Google Scholar] [CrossRef] [PubMed]
- Shankar, V.; Bowen, R.A.; Davis, A.D.; Rupprecht, C.E.; O’Shea, T.J. Rabies in a captive colony of big brown bats (Eptesicus fuscus). J. Wildl. Dis. 2004, 40, 403–413. [Google Scholar] [CrossRef] [PubMed]
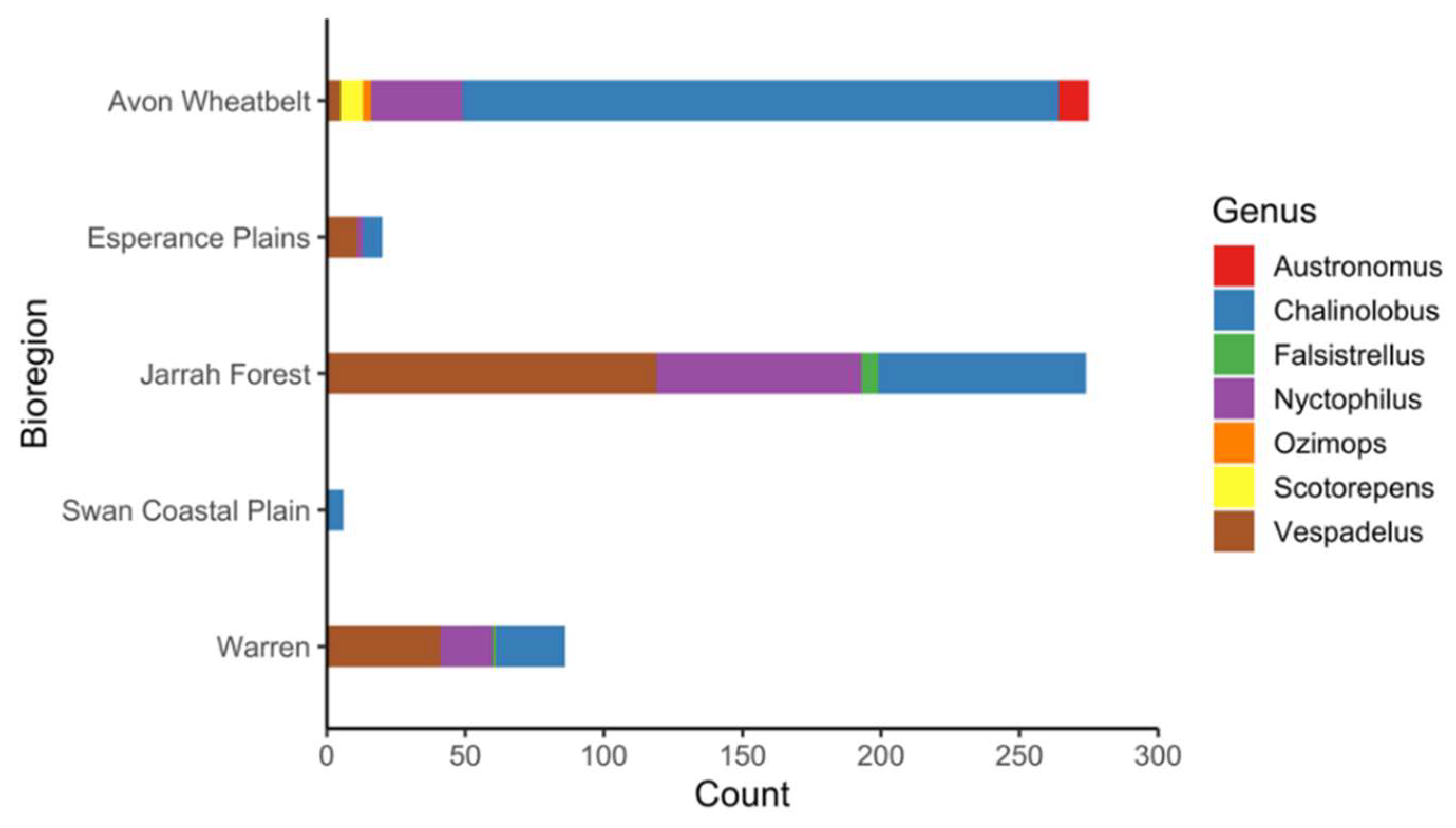
| Family | Species | Swabs | Sera | Seroprevalence 1 |
|---|---|---|---|---|
| Vespertilionidae | Chalinolobus gouldii | 287(0) | 262(2) | 0.7 (0.2–2.7) |
| Chalinolobus morio | 105(0) | 64(3) | 4.6 (1.6–12.8) | |
| Falsistrellus mackenziei | 14(0) | 7(1) | NC 2 | |
| Nyctophilus geoffroyi | 69(0) | 48(0) | ||
| Nyctophilus gouldi | 78(0) | 66(3) | 4.5 (1.5–12.5) | |
| Nyctophilus major | 12(0) | 5(1) | NC 2 | |
| Nyctophilus sp3 | 6(0) | 5(0) | ||
| Scotorepens balstoni | 13(0) | 8(0) | ||
| Vespadelus baverstocki | 6(0) | 5(0) | ||
| Vespadelus finlaysoni | 1(0) | 0 | ||
| Vespadelus regulus | 227(0) | 164(9) | 5.5 (2.9–10.1) | |
| Vespadelus sp3 | 2(0) | 1(0) | ||
| Molossidae | Austronomus australis | 13(0) | 11(0) | |
| Ozimops sp | 6(0) | 3(0) |
| Bioregion | Location | Species |
|---|---|---|
| Jarrah Forest | I | Vespadelus regulus (1) |
| II | Chalinolobus gouldii (2) | |
| III | Vespadelus regulus (2) | |
| Nyctophilus major (1) | ||
| Falsistrellus mackenziei (1) | ||
| IV | Nyctophilus gouldi (1) | |
| V | Vespadelus regulus (1) | |
| Warren | VI | Chalinolobus morio (2) |
| Nyctophilus gouldi (1) | ||
| Vespadelus regulus (1) | ||
| VII | Chalinolobus morio (1) | |
| Nyctophilus gouldi (1) | ||
| Vespadelus regulus (4) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Prada, D.; Boyd, V.; Baker, M.; Jackson, B.; O’Dea, M. Insights into Australian Bat Lyssavirus in Insectivorous Bats of Western Australia. Trop. Med. Infect. Dis. 2019, 4, 46. https://doi.org/10.3390/tropicalmed4010046
Prada D, Boyd V, Baker M, Jackson B, O’Dea M. Insights into Australian Bat Lyssavirus in Insectivorous Bats of Western Australia. Tropical Medicine and Infectious Disease. 2019; 4(1):46. https://doi.org/10.3390/tropicalmed4010046
Chicago/Turabian StylePrada, Diana, Victoria Boyd, Michelle Baker, Bethany Jackson, and Mark O’Dea. 2019. "Insights into Australian Bat Lyssavirus in Insectivorous Bats of Western Australia" Tropical Medicine and Infectious Disease 4, no. 1: 46. https://doi.org/10.3390/tropicalmed4010046
APA StylePrada, D., Boyd, V., Baker, M., Jackson, B., & O’Dea, M. (2019). Insights into Australian Bat Lyssavirus in Insectivorous Bats of Western Australia. Tropical Medicine and Infectious Disease, 4(1), 46. https://doi.org/10.3390/tropicalmed4010046





