Global Significance of Mangrove Blue Carbon in Climate Change Mitigation
Abstract
1. Introduction
2. Carbon Stocks
3. Carbon Sequestration Rates
4. Carbon Losses
5. Assessment of Global Significance
Supplementary Materials
Funding
Conflicts of Interest
References
- Nelleman, C.; Corcoran, E.; Duarte, C.M.; Valdés, L.; DeYoung, C.; Foseca, L.; Grimsditch, G. (Eds.) Blue Carbon: A Rapid Response Assessment; United Nations Environmental Programme and GRID-Arendal: Arendal, Norway, 2009. [Google Scholar]
- Laffoley, D.d.A.; Grimsditch, G. (Eds.) The Management of Natural Coastal Carbon Sinks; IUCN: Gland, Switzerland, 2009. [Google Scholar]
- Herr, D.; Pidgeon, E.; Laffoley, D. (Eds.) Blue Carbon Policy Framework: Based on the Discussion of the International Blue Carbon Policy Working Group; IUCN: Gland, Switzerland, 2012. [Google Scholar]
- Sifleet, S.; Pendleton, L.; Murray, B.C. State of the Science on Coastal Blue Carbon: A Summary for Policy Makers. In Nicholas Institute for Environmental Policy Solutions Report NIR 11-06; Nicholas Institute, Duke University: Durham, NC, USA, 2011. [Google Scholar]
- IOC. A Blueprint for Ocean and Coastal Sustainability; IOC/UNESCO: Paris, France, 2011. [Google Scholar]
- Alongi, D.M. Blue Carbon: Coastal Sequestration for Climate Change Mitigation; Springer Briefs in Climate Studies, Springer Nature: Cham, Switzerland, 2018. [Google Scholar]
- Pendleton, L.; Donato, D.C.; Murray, B.C.; Crooks, S.; Jenkins, W.A.; Sifleet, S.; Craft, C.; Fourqurean, J.W.; Kauffman, J.B.; Marbá, N.; et al. Estimating global “blue carbon: emissions from conversion and degradation of vegetated coastal ecosystems. PLoS ONE 2012, 7, e43542. [Google Scholar] [CrossRef] [PubMed]
- Huxham, M.; Whitlock, D.; Githaiga, M.; Dencer-Brown, A. Carbon in the coastal seascape: how interactions between mangrove forests, seagrass meadows and tidal marshes influence carbon storage. Curr. Forest Rept. 2018, 4, 101–110. [Google Scholar] [CrossRef]
- Taillardat, P.; Friess, D.A.; Lupascu, M. Mangrove blue carbon strategies for climate change mitigation are most effective at the national scale. Biol. Lett. 2018, 14, 20180251. [Google Scholar] [CrossRef] [PubMed]
- Alongi, D.M.; Murdiyarso, D.; Fourqurean, J.W.; Kauffman, J.B.; Hutahaean, A.; Crooks, S.; Lovelock, C.E.; Howard, J.; Herr, D.; Fortes, M.; et al. Indonesia’s blue carbon: a globally significant and vulnerable sink for seagrass and mangrove carbon. Wetl. Ecol. Manag. 2016, 24, 3–13. [Google Scholar] [CrossRef]
- Alongi, D.M. Carbon sequestration in mangrove forests. Carbon Manag. 2012, 3, 313–322. [Google Scholar] [CrossRef]
- Saderne, V.; Geraldi, N.R.; Macreadie, P.I.; Maher, D.Y.; Middelburg, J.J.; Serrano, O.; Almahasheer, H.; Arias-Ortiz, A.; Cusack, M.; Eyre, B.D.; et al. Role of carbonate burial in blue carbon budgets. Nat. Commun. 2019, 10, 1066. [Google Scholar] [CrossRef]
- Hamilton, S.E.; Casey, D. Creation of a high spatio-temporal resolution global database of continuous mangrove forest cover for the 21st century (CGMFC-21). Glob. Ecol. Biogeogr. 2016, 25, 729–738. [Google Scholar] [CrossRef]
- Jardine, S.L.; Siikamäki, J.V. A global predictive model of carbon in mangrove soils. Environ. Res. Lett. 2014, 9, 104013. [Google Scholar] [CrossRef]
- Hamilton, S.; Friess, D.A. Global carbon stocks and potential emissions due to mangrove deforestation from 2000 to 2012. Nat. Clim. Chang. 2018, 8, 240–244. [Google Scholar] [CrossRef]
- Sanders, C.J.; Maher, D.T.; Tait, D.R.; Williams, D.; Holloway, C.; Sippo, J.Z.; Santos, I.R. Are global mangrove carbon stocks driven by rainfall? J. Geophys. Res. Biogeosci. 2016, 121, 2600–2609. [Google Scholar] [CrossRef]
- Ouyang, X.; Lee, S.Y. Improved estimates on global carbon stock and carbon pools in tidal wetlands. Nat. Commun. 2020, 11, 317. [Google Scholar] [CrossRef] [PubMed]
- Giri, C.; Ochieng, E.; Tiezen, L.L.; Zhu, Z.; Singh, A.; Loveland, T.; Masek, J.; Duke, N.C. Status and distribution of mangrove forests of the world using earth observation satellite data. Glob. Ecol. Biogeogr. 2011, 20, 154–159. [Google Scholar] [CrossRef]
- Walcker, R.; Gandois, L.; Proisy, C.; Corenblit, D.; Mougin, E.; Laplanche, C.; Ray, R.; Fromard, F. Control of “blue carbon” storage by mangrove ageing: Evidence from a 66-year old chronosequence in French Guiana. Glob. Chang. Biol. 2018, 6, 2325–2338. [Google Scholar] [CrossRef] [PubMed]
- Hieu, P.V.; Dung, L.V.; Tue, N.T.; Omori, K. Will restored mangrove forests enhance sediment organic carbon and ecosystem carbon storage? Reg. Stud. Mar. Sci. 2017, 14, 43–52. [Google Scholar]
- Ha, T.H.; Marchand, C.; Aimé, J.; Dang, H.N.; Phan, N.H.; Nguyen, X.T.; Nguyen, T.K.C. Belowground carbon sequestration in mature planted mangroves (Northern Viet Nam). For. Ecol. Manag. 2018, 407, 191–199. [Google Scholar] [CrossRef]
- Arif, A.M.; Guntur, G.; Ricky, A.B.; Novianti, P.; Andik, I. Mangrove ecosystem C-stocks of Lamongan, Indonesia and its correlation with forest age. Res. J. Chem. Environ. 2017, 21, 1–8. [Google Scholar]
- Breithaupt, J.L.; Smoak, J.M.; Smith, T.J., III; Sanders, C.J.; Hoare, A. Organic carbon burial rates in mangrove sediments: strengthening the global budget. Glob. Biogeochem. Cycles 2012, 26, GB3011. [Google Scholar] [CrossRef]
- McLeod, E.; Chmura, G.L.; Bouillon, S.; Björk, M.; Duarte, C.M.; Lovelock, C.E.; Schlesinger, W.H.; Silliman, B.R. A blueprint for blue carbon: toward an improved understanding of the role of vegetated coastal habitats in sequestering CO2. Front. Ecol. Environ. 2011, 9, 552–560. [Google Scholar] [CrossRef]
- Twilley, R.R.; Chen, R.H.; Hargis, T. Carbon sinks in mangroves and their implications to carbon budget of tropical coastal ecosystems. Water Air Soil Pollut. 1992, 64, 265–288. [Google Scholar] [CrossRef]
- Jennerjahn, T.C.; Ittekot, V. Relevance of mangroves for the production and deposition of organic matter along tropical continental margins. Naturwissen. 2002, 89, 23–30. [Google Scholar] [CrossRef]
- Duarte, C.M.; Middelburg, J.J.; Caraco, N. Major role of marine vegetation on the oceanic carbon cycle. Biogeosciences 2005, 2, 1–8. [Google Scholar] [CrossRef]
- Almahasheer, H.; Serrano, O.; Duarte, C.M.; Arias-Ortiz, A.; Masque, P.; Irigoien, X. Low carbon sink capacity of Red Sea mangroves. Sci. Rep. 2017, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Pérez, A.; Libardoni, B.G.; Sanders, C.J. Factors influencing organic carbon accumulation in mangrove ecosystems. Biol. Lett. 2018, 14, 20180237. [Google Scholar] [CrossRef] [PubMed]
- Li, S.-B.; Chen, P.-H.; Huang, J.-S.; Hsueh, M.-L.; Hsieh, L.-Y.; Lee, C.-L.; Lin, H.-J. Factors regulating carbon sinks in mangrove ecosystems. Glob. Chang. Biol. 2018, 24, 4195–4210. [Google Scholar] [CrossRef]
- Cuellar-Martinez, T.; Ruiz-Fernández, A.C.; Sanchez-Cabeza, J.-A.; Pérez-Bernal, L.; Lopez-Mendoza, P.G.; Carnero-Bravo, V.; Agraz-Hernández, C.M.; van Tussenbroek, B.I.; Sandoval-Gil, J.; Cardoso-Mohedano, J.G.; et al. Temporal records of organic carbon stocks and burial rates in Mexican blue carbon coastal ecosystems throughout the Anthropocene. Glob. Planet. Chang. 2020, 192, 103215. [Google Scholar] [CrossRef]
- Lamont, K.; Saintilan, N.; Kelleway, J.J.; Mazumder, D.; Zawadzki, A. Thirty-year repeat measures of mangrove above- and below-ground biomass reveals unexpectedly high carbon sequestration. Ecosystems 2020, 23, 370–382. [Google Scholar] [CrossRef]
- Salmo, S.G., III; Malapit, V.; Garcia, M.C.A.; Pagkalinawan, H.M. Establishing rates of carbon sequestration in mangroves from an earthquake uplift event. Biol. Lett. 2019, 15, 20180799. [Google Scholar] [CrossRef]
- Bernardino, A.F.; Sanders, C.J.; Bissoli, L.B.; Gomes, L.E.D.O.; Kauffman, J.B.; Ferreira, T.O. Land use impacts on benthic bioturbation potential and carbon burial in Brazilian mangrove ecosystems. Limnol. Oceanogr. 2020. [Google Scholar] [CrossRef]
- Soper, F.M.; MacKenzie, R.A.; Sharma, S.; Cole, T.G.; Litton, C.M.; Sparks, J.P. Non-native mangroves support carbon storage, sediment carbon burial, and accretion of coastal ecosystems. Glob. Chang. Biol. 2019, 25, 4315–4326. [Google Scholar] [CrossRef]
- Pérez, A.; Machado, W.; Gutiérrez, D.; Borges, A.C.; Patchineelam, S.R.; Sanders, C.J. Carbon accumulation and storage capacity in mangrove sediments three decades after deforestation within a eutrophic bay. Mar. Pollut. Bull. 2018, 126, 275–280. [Google Scholar] [CrossRef]
- Shaltout, K.H.; Ahmed, M.T.; Alrumman, S.A.; Ahmed, D.A.; Eid, E.M. Evaluation of the carbon sequestration capacity of arid mangroves along nutrient availability and salinity gradients along the Red Sea coastline of Saudi Arabia. Oceanologia 2020, 62, 56–69. [Google Scholar] [CrossRef]
- Cusack, M.; Saderne, V.; Arias-Ortiz, A.; Masqué, P.; Krishnakumar, P.K.; Rabaoui, L.; Qurban, M.A.; Qasem, A.M.; Prihartato, P.; Loughland, R.A.; et al. Organic carbon sequestration and storage in vegetated coastal habitats along the western coast of the Arabian Gulf. Environ. Res. Lett. 2018, 13, 074007. [Google Scholar] [CrossRef]
- Sasmito, S.D.; Kuzyakov, Y.; Lubis, A.A.; Murdiyarso, D.; Hutley, L.B.; Bachri, S.; Friess, D.A.; Martius, C.; Borchard, N. Organic carbon burial and sources in soils of coastal mudflat and mangrove ecosystems. Catena 2020, 104414. [Google Scholar] [CrossRef]
- Breithaupt, J.L.; Smoak, J.M.; Sanders, C.J.; Troxler, T.G. Spatial variability of organic carbon, CaCO3 and nutrient burial rates spanning a mangrove productivity gradient in the Coastal Everglades. Ecosystems 2019, 22, 844–858. [Google Scholar] [CrossRef]
- Kusumaningtyas, M.A.; Hutahaean, A.A.; Fischer, H.W.; Pérez-Mayo, M.; Ransby, D.; Jennerjahn, T.C. Variability in the organic carbon stocks, sources, and accumulation rates of Indonesian mangrove ecosystems. Estuar. Coast Shelf Sci. 2019, 218, 310–323. [Google Scholar] [CrossRef]
- Hapsari, K.A.; Jennerjahn, T.C.; Lukas, M.C.; Karius, V.; Behling, H. Intertwined effects of climate and land use change on environmental dynamics and carbon accumulation in a mangrove-fringed coastal lagoon in Java, Indonesia. Glob. Chang. Biol. 2019, 26, 1414–1431. [Google Scholar] [CrossRef]
- Afefe, A.A.; Abbas, M.S.; Soliman, A.S.; Khedr, H.A.; Hatab, B.E. Tree biomass and soil carbon stocks of a mangrove ecosystem on the Egyptian—African Red Sea coast. Fund. Appl. Limnol. 2020, 193, 239–251. [Google Scholar] [CrossRef]
- Wilkinson, G.M.; Besterman, A.; Buelo, C.; Gephart, J.; Pace, M.L. A synthesis of modern organic carbon accumulation rates in coastal and aquatic inland ecosystems. Sci. Rep. 2018, 8, 15736. [Google Scholar] [CrossRef]
- Murdiyarso, D.; Hanggara, B.B.; Lubis, A.A. Sedimentation and soil carbon accumulation in degraded mangrove forests of North Sumatra, Indonesia. BioRxiv 2018, 32519. [Google Scholar] [CrossRef]
- Marchand, C. Soil carbon stocks and burial rates along a mangrove forest chronosequence (French Guiana). For. Ecol. Manag. 2017, 384, 92–99. [Google Scholar] [CrossRef]
- El-Hussieny, S.A.; Ismail, I.M. Role of Avicennia marina (Forssk.) Vierh. of south Sinai, Egypt in atmospheric CO2 sequestration. Int. J. Sci. Res. 2015, 6, 1935–1946. [Google Scholar]
- Eid, E.M.; Khedher, K.M.; Ayed, H.; Arshad, M.; Moatamed, A.; Mouldi, A. Evaluation of carbon stock in the sediment of two mangrove species, Avicennia marina and Rhizophora mucronata, growing in the Farasan Islands, Saudi Arabia. Oceanologia 2020, 62, 200–213. [Google Scholar] [CrossRef]
- Rosentreter, J.A.; Maher, D.T.; Erler, D.V.; Murray, R.H.; Eyre, B.D. Methane emissions partially offset “blue carbon” burial in mangroves. Sci. Adv. 2018, 4, eaao4985. [Google Scholar] [CrossRef] [PubMed]
- Maher, D.T.; Santos, I.R.; Schulz, K.G.; Call, M.; Jacobsen, G.E.; Sanders, C.J. Blue carbon oxidation revealed by radiogenic and stable isotopes in a mangrove system. Geophys. Res. Lett. 2017, 44, 4889–4896. [Google Scholar] [CrossRef]
- Kauffman, J.B.; Bernardino, A.F.; Ferreira, T.O.; Bolton, N.W.; Gomes, L.E.D.O.; Nobrega, G.N. Shrimp ponds lead to massive loss of soil carbon and greenhouse gas emissions in northeastern Brazilian mangroves. Ecol. Evol. 2018, 8, 5530–5540. [Google Scholar] [CrossRef] [PubMed]
- Kauffman, J.B.; Arifanti, V.B.; Trejo, H.H.; del Carmen Jesús García, M.; Norfolk, J.; Cifuentes, M.; Hadriyanto, D.; Murdiyarso, D. The jumbo carbon footprint of a shrimp: carbon losses from mangrove deforestation. Front. Ecol. Environ. 2017, 15, 183–188. [Google Scholar] [CrossRef]
- Lovelock, C.E.; Ruess, R.W.; Feller, I.C. CO2 efflux from cleared mangrove peat. PLoS ONE 2011, 6, e21279. [Google Scholar] [CrossRef]
- Bulmer, R.H.; Lundquist, C.J.; Schwendenmann, L. Sediment properties and CO2 efflux from intact and cleared temperate mangrove forests. Biogeosciences 2015, 12, 6169–6180. [Google Scholar] [CrossRef]
- Sharma, S.; MacKenzie, R.A.; Tieng, T.; Soben, K.; Tulyasuwan, N.; Resanond, A.; Blate, G.; Litton, C.M. The impacts of degradation, deforestation and restoration on mangrove ecosystem carbon stocks across Cambodia. Sci. Total Environ. 2020, 706, 135416. [Google Scholar] [CrossRef]
- Sidik, F.; Lovelock, C.E. CO2 efflux from shrimp ponds in Indonesia. PLoS ONE 2013, 8, e66329. [Google Scholar] [CrossRef]
- Kauffman, J.B.; Heider, C.; Norfolk, J.; Payton, F. Carbon stocks of intact mangroves and carbon emissions arising from their conversion in the Dominican Republic. Ecol. Appl. 2014, 24, 518–527. [Google Scholar] [CrossRef] [PubMed]
- Elwin, A.; Bukoski, J.J.; Jintana, V.; Robinson, E.J.Z.; Clark, J.M. Preservation and recovery of mangrove ecosystem carbon stocks in abandoned shrimp ponds. Sci. Rep. 2019, 9, 18275. [Google Scholar] [CrossRef] [PubMed]
- Arifanti, V.B.; Kauffman, J.B.; Hadriyanto, D.; Murdiyarso, D.; Diana, R. Carbon dynamics and land use carbon footprints in mangrove-converted aquaculture: The case of the Mahakam Delta, Indonesia. For. Ecol. Manag. 2019, 432, 17–29. [Google Scholar] [CrossRef]
- Lang’at, J.K.; Kairo, J.G.; Mencuccini, M.; Bouillon, S.; Skov, M.W.; Waldron, S.; Huxham, M. Rapid losses of surface elevation following tree girdling and cutting in tropical mangroves. PLoS ONE 2014, 6, e107868. [Google Scholar] [CrossRef] [PubMed]
- Cahoon, D.R.; Hensel, P.; Rybczyk, J.; McKee, K.L.; Proffitt, E.D.; Perez, B.C. Mass tree mortality leads to mangrove peat collapse at Bay Islands, Honduras after Hurricane Mitch. J. Ecol. 2003, 91, 1093–1105. [Google Scholar] [CrossRef]
- Lovelock, C.E.; Fourqurean, J.W.; Morris, J.T. Modeled CO2 emissions from coastal wetland transitions to other land uses: Tidal marshes, mangrove forests, and seagrass beds. Front. Mar. Sci. 2017, 4, 143. [Google Scholar] [CrossRef]
- Salmo, S.G., III; Gianan, E.L.D. Post-disturbance carbon stocks and rates of sequestration: Implications on “blue carbon” estimates in Philippine mangroves. Philipp. Sci. Lett. 2019, 12, 122–132. [Google Scholar]
- Peneva-Reed, E.I.; Krauss, K.W.; Bullock, E.L.; Zhu, Z.; Woltz, V.L.; Drexler, J.Z.; Conrad, J.R.; Stehman, S.V. Carbon stock losses and recovery observed for a mangrove ecosystem following a major hurricane in Southwest Florida. Estuar. Coast. Shelf Sci. 2020, 106750. [Google Scholar] [CrossRef]
- Lagomasino, D.; Fatoyinbo, T.; Lee, S.K.; Feliciano, E.; Trettin, C.; Shapiro, A.; Mangora, M.M. Measuring mangrove carbon loss and gain in deltas. Environ. Res. Lett. 2019, 14, 025002. [Google Scholar] [CrossRef]
- Duncan, C.; Primavera, J.H.; Pettorelli, N.; Thompson, J.R.; Loma, R.J.A.; Koldewey, H.J. Rehabilitating mangrove ecosystem services: A case study on the relative benefits of abandoned pond reversion from Panay Island, Philippines. Mar. Pollut. Bull. 2016, 109, 772–782. [Google Scholar] [CrossRef]
- Herrera-Silveira, J.A.; Pech-Cardenas, M.A.; Morales-Ojeda, S.M.; Cinco-Castro, S.; Camacho-Rico, A.; Sosa, J.P.C.; Mendoza-Martinez, J.E.; Pech-Poot, E.Y.; Montero, J.; Teutli-Hernandez, C. Blue carbon of Mexico, carbon stocks and fluxes: a systematic review. PeerJ 2020, 8, e8790. [Google Scholar] [CrossRef] [PubMed]
- Akhand, A.; Mukhopadhyay, A.; Chanda, A.; Mukherjee, S.; Das, A.; Das, S.; Hazra, S.; Mitra, D.; Choudhury, S.B.; Rao, K.H. Potential CO2 emission due to loss of above ground biomass from the Indian Sundarban mangroves during the last four decades. J. Indian Soc. Remote Sens. 2016, 8, 1–8. [Google Scholar] [CrossRef]
- Brinck, K.; Fischer, R.; Groeneveld, J.; Lehman, S.; De Paula, M.D.; Pütz, S.; Sexton, J.O.; Song, D.; Huth, A. High resolution analysis of tropical forest fragmentation and its impact on the global carbon cycle. Nat. Commun. 2017, 8, 14855. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-T.A. Cross-boundary exchanges of carbon and nitrogen in continental margins. In Carbon and Nutrient Fluxes in Continental Margins; Liu, K.-K., Atkinson, L., Quiñones, R., Talaue-McManus, L., Eds.; Springer: New York, NY, USA, 2010; pp. 561–574. [Google Scholar]
- Alongi, D.M.; Mukhopadhyay, S.K. Contributions of mangroves to coastal carbon cycling in low latitude seas. Agric. For. Meteorol. 2015, 213, 266–272. [Google Scholar] [CrossRef]
- Davidson, N.C.; Finlayson, C.M. Updating global coastal wetland areas presented in Davidson and Finlayson (2018). Mar. Freshw. Res. 2019, 70, 1195–1200. [Google Scholar] [CrossRef]
- Leadley, P.W.; Krug, C.B.; Alkemade, R.; Pereira, H.M.; Sumaila, U.R.; Walpole, M.; Marques, A.; Newbold, T.; Teh, L.S.L.; van Kolck, J.; et al. Progress Towards the Aichi Biodiversity Targets: An Assessment of Biodiversity Trends, Policy Scenarios and Key Actions; CBD Technical Series No. 78; Secretariat of the Convention on Biological Diversity: Montreal, QUE, Canada, 2014; Available online: http://www.cbd.int/doc/publications/1019-1020-cbd-ts-78-en.pdf (accessed on 22 June 2020).
- McKenzie, L.J.; Nordlund, L.M.; Jones, B.L.; Cullen-Unsworth, L.C.; Roelfsema, C.; Unsworth, R.K.F. The global distribution of seagrass meadows. Environ. Res. Lett. 2020. [Google Scholar] [CrossRef]
- Mora, C.; Andréfouët, S.; Costello, M.J. Coral reefs and the global network of marine protected areas. Science 2006, 312, 1750–1751. [Google Scholar] [CrossRef]
- Crossland, C.J.; Hatcher, B.G.; Smith, S.V. Role of coral reefs in global ocean production. Coral Reefs 1991, 10, 55–64. [Google Scholar] [CrossRef]
- Alongi, D.M. Coastal Ecosystem Processes; CRC Press: Boca Raton, FL, USA, 1998. [Google Scholar]
- Kinsey, D.W.; Hopley, D. The significance of coral reefs as global carbon sinks—Response to Greenhouse. Glob. Planet. Chang. 1991, 3, 363–377. [Google Scholar] [CrossRef]
- Selig, E.R.; Bruno, J.F. A global analysis of the effectiveness of marine protected areas in preventing coral loss. PLoS ONE 2010, 5, e9278. [Google Scholar] [CrossRef]
- Hedges, J.I.; Keil, R.G. Sedimentary organic matter preservation: an assessment and speculative synthesis. Mar. Chem. 1995, 49, 81–115. [Google Scholar] [CrossRef]
- Spalding, D.; Kendirli, E.; Loiver, C.D. The role of forests in global carbon budgeting. In Managing Forest Carbon in a Changing Climate; Ashton, M.S., Tyrrell, M.L., Spalding, D., Gentry, B., Eds.; Springer: Dordrecht, The Netherlands, 2012; pp. 165–179. [Google Scholar]
- Phillips, O.L.; Lewis, S.L. Evaluating the tropical forest carbon sink. Glob. Chang. Biol. 2014, 20, 2039–2041. [Google Scholar] [CrossRef] [PubMed]
- Tyrrell, M.L.; Ross, J.; Kelty, M. Carbon dynamics in the temperate forest. In Managing Forest Carbon in a Changing Climate; Ashton, M.S., Tyrrell, M.L., Spalding, D., Gentry, B., Eds.; Springer: Dordrecht, The Netherlands, 2012; pp. 77–107. [Google Scholar]
- Hansen, M.C.; Stehman, S.V.; Potapov, P.V. Quantification of global gross forest cover loss. Proc. Natl. Acad. Sci. USA 2010, 107, 8650–8655. [Google Scholar] [CrossRef] [PubMed]
- Milakovsky, B.; Frey, B.; James, T. Carbon Dynamics in the Boreal Forest. In Managing Forest Carbon in a Changing Climate; Ashton, M.S., Tyrrell, M.L., Spalding, D., Gentry, B., Eds.; Springer: Dordrecht, The Netherlands, 2012; pp. 109–135. [Google Scholar]
- Grace, J.; José, J.S.; Meir, P.; Miranda, H.S.; Montes, R.A. Productivity and carbon fluxes of tropical savannas. J. Biogeogr. 2006, 33, 387–400. [Google Scholar] [CrossRef]
- Sala, O.E. Temperate grasslands. In Global Biodiversity in a Changing Environment; Chapin, F., Sala, O.E., Huber-Sannwald, E., Eds.; Springer: New York, NY, USA, 2001; pp. 121–137. [Google Scholar]
- Dean, C.; Kirkpatrick, J.B.; Harper, R.J.; Eldridge, D.J. Optimising carbon sequestration in arid and semiarid rangelands. Ecol. Eng. 2015, 74, 148–163. [Google Scholar] [CrossRef]
- Jenkins, C.N.; Joppa, L. Expansion of the global terrestrial protected area system. Biol. Conserv. 2009, 142, 2166–2174. [Google Scholar] [CrossRef]
- Gibbon, A.; Silman, M.R.; Mahli, Y.; Fisher, J.B.; Meir, P.; Zimmermann, M.; Dargie, G.C.; Farfan, W.R.; Garcia, K.C. Ecosystem carbon storage across the grassland-forest transition in the High Andes of Manu National Park, Peru. Ecosystems 2010, 13, 1097–1111. [Google Scholar] [CrossRef]
- Mekonnen, A.; Tolera, M. Carbon stock estimation along altitudinal gradient in Sekele-Mariam dry evergreen montane forest, North-Western Ethiopia. Agric. For. Fish. 2019, 8, 48–53. [Google Scholar] [CrossRef]
- Angonese, J.G.; Grau, H.R. Assessment of swaps and persistence in land cover changes in a subtropical periurban region, NW Argentina. Landsc. Urban Plan 2014, 127, 83–93. [Google Scholar] [CrossRef]
- Martínez, M.L.; Pérez-Maqueo, O.; Vázquez, G.; Castillo-Campos, G.; García-Franco, J.; Mehltreter, K.; Equihua, M.; Landgrave, R. Effects of land use change on biodiversity and ecosystem services in tropical montane cloud forests of Mexico. For. Ecol. Manag. 2009, 258, 1856–1863. [Google Scholar] [CrossRef]
- Ray, D.K.; Nair, U.S.; Lawton, R.O.; Welch, R.M.; Pielke, R.A., Sr. Impact of land use on Costa Rican tropical montane cloud forests: Sensitivity of orographic cloud formation to deforestation in the plains. J. Geophys. Res. 2006, 111, D02108. [Google Scholar] [CrossRef]
- Kidane, Y.; Stahlmann, R.; Beierkuhnlein, C. Vegetation dynamics, and land use and land cover change in the Bale Mountains, Ethiopia. Environ. Monit. Assess. 2012, 184, 7473–7489. [Google Scholar] [CrossRef] [PubMed]
- Hailemariam, S.N.; Soromessa, T.; Teketay, D. Land use and land cover change in the Bale Mountain eco-region of Ethiopia during 1985–2015. Land 2016, 5, 41. [Google Scholar] [CrossRef]
- Kintz, D.B.; Young, K.R.; Crews-Meyer, K.A. Implications of land use/land cover change in the buffer zone of a national park in the tropical Andes. Environ. Manag. 2006, 38, 238–252. [Google Scholar] [CrossRef] [PubMed]
- Song, X.-P.; Hansen, M.C.; Stehman, S.V.; Potapov, P.V.; Tyukavina, A.; Vermote, E.F.; Townshend, J.R. Global land change 1982–2016. Nature 2018, 560, 639–643. [Google Scholar] [CrossRef]
- Evrendilek, F.; Berberoglu, S.; Taskinsu-Meydan, S.; Yilmaz, E. Quantifying carbon budgets of conifer Mediterranean forest ecosystems, Turkey. Environ. Monit. Assess. 2006, 119, 527–543. [Google Scholar] [CrossRef]
- Ruiz-Peinado, R.; Bravo-Oviedo, A.; Lopez-Senespleda, E.; Bravo, F.; del Rio, M. Forest management and carbon sequestration in the Mediterranean region: A review. For. Syst. 2017, 26, eR04S. [Google Scholar] [CrossRef]
- Bravo, F.; Bravo-Oviedo, A.; Diaz-Balteiro, L. Carbon sequestration in Spanish Mediterranean forests under two management alternatives: A modelling approach. Eur. J. For. Res. 2008, 127, 225–234. [Google Scholar] [CrossRef]
- Del Rio, M.; Barbeito, I.; Bravo-Oviedo, A.; Calama, R.; Cañellas, I.; Herrero, C.; Montero, G.; Moreno-Fernández, D.; Ruíz-Peinado, R.; Bravo, F. Mediterranean pine forests: management effects on carbon stocks. In Managing Forest Ecosystems: The Challenge of Climate Change; Bravos, F., Le May, V., Jandl, R., Eds.; Springer: Cham, Switzerland, 2017; pp. 301–327. [Google Scholar]
- Cañellas, I.; Sánchez-González, M.; Bogino, S.M.; Adame, P.; Herrero, C.; Roig, S.; Tomé, M.; Paulo., J.A.; Bravo, F. Silviculture and carbon sequestration in Mediterranean oak forests. In Managing Forest Ecosystems: The Challenge of Climate Change; Bravos, F., Le May, V., Jandl, R., Gadow, K., Eds.; Springer: Cham, Switzerland, 2008; pp. 315–336. [Google Scholar]
- Ruiz-Peinado, R.; Bravo-Oviedo, A.; Lopez-Senespleda, E.; Montero, G.; Río, M. Do thinnings influence biomass and soil carbon stocks in Meriterranean maritime pinewoods? Eur. J. For. Res. 2013, 132, 253–262. [Google Scholar] [CrossRef]
- Ylänne, H.; Olofsson, J.; Oksanen, L.; Stark, S. Consequences of grazer-induced vegetation transitions on ecosystem carbon storage in the tundra. Funct. Ecol. 2018, 32, 1091–1102. [Google Scholar] [CrossRef]
- Sørensen, M.V.; Strimbeck, R.; Nystuen, K.O.; Kapas, R.E.; Enquist, B.J.; Graae, B.J. Draining the pool? Carbon storage and fluxes in three alpine plant communities. Ecosystems 2017, 21, 316–330. [Google Scholar] [CrossRef]
- Dai, L.; Wu, G.; Zhao, J.; Kong, H.; Shao, G.; Deng, H. Carbon cycling of alpine tundra ecosystems on Changbai Mountain and its comparison with arctic tundra. Sci. China Ser. D Earth Sci. 2002, 45, 903–910. [Google Scholar] [CrossRef]
- Michaelson, G.J.; Ping, C.L.; Kimble, J.M. Carbon storage and distribution in tundra soils of Arctic Alaska, USA. Arct. Alp. Res. 1996, 28, 414–424. [Google Scholar] [CrossRef]
- Mack, M.C.; Schuur, E.A.G.; Bret-Harte, M.S.; Shaver, G.R.; Chapin, F.S., III. Ecosystem carbon storage in arctic tundra reduced by long-term nutrient fertilization. Nature 2004, 431, 440–445. [Google Scholar] [CrossRef] [PubMed]
- Campeau, A.B.; Lafleur, P.M.; Humphreys, E.R. Landscape-scale variability in soil organic carbon storage in the central Canadian Arctic. Can. J. Soil Sci. 2014, 94, 477–488. [Google Scholar] [CrossRef]
- Kaiser, C.; Meyer, H.; Biasi, C.; Rusalimova, O.; Barsukov, P.; Richter, A. Storage and mineralization of carbon and nitrogen in soils of a frost-boil tundra ecosystem in Siberia. Appl. Soil Ecol. 2005, 29, 173–183. [Google Scholar] [CrossRef]
- Sjögersten, S.; Wookey, P.A. The impact of climate change on ecosystem carbon dynamics at the Scadinavian mountain birch forest—Tundra heath ecotone. AMBIO 2009, 38, 2–10. [Google Scholar] [CrossRef]
- Kwon, H.-J.; Oechel, W.C.; Zullueta, R.C.; Hastings, S.J. Effects of climate variability on carbon sequestration among adjacent wet sedge tundra and moist tussock tundra ecosystems. J. Geophys. Res. 2006, 111, G03014. [Google Scholar] [CrossRef]
- Runkle, B.R.K.; Sachs, T.; Wille, C.; Pfeiffer, E.-M.; Kutzbach, L. Bulk partitioning the growing season net ecosystem exchange of CO2 in Siberian tundra reveals the seasonality of its carbon sequestration strength. Biogeosciences 2013, 10, 1337–1349. [Google Scholar] [CrossRef]
- Parmentier, F.J.W.; van der Molen, M.K.; van Huissteden, J.; Karsanaev, S.A.; Kononov, A.V.; Suzdalov, D.A.; Maximov, T.C.; Dolman, A.J. Longer growing seasons do not increase net carbon uptake in the northeastern Siberian tundra. J. Geophys. Res. 2011, 116, G04013. [Google Scholar] [CrossRef]
- Kutzbach, L.; Wille, C.; Pfeiffer, E.-M. The exchange of carbon dioxide between wet arctic tundra and the atmosphere at the Lena River Delta, northern Siberia. Biogeosciences 2007, 4, 1953–2005. [Google Scholar] [CrossRef]
- Leifeld, J.; Menichetti, L. The underappreciated potential of peatlands in global climate change mitigation strategies. Nat. Commun. 2018, 9, 1071. [Google Scholar] [CrossRef] [PubMed]
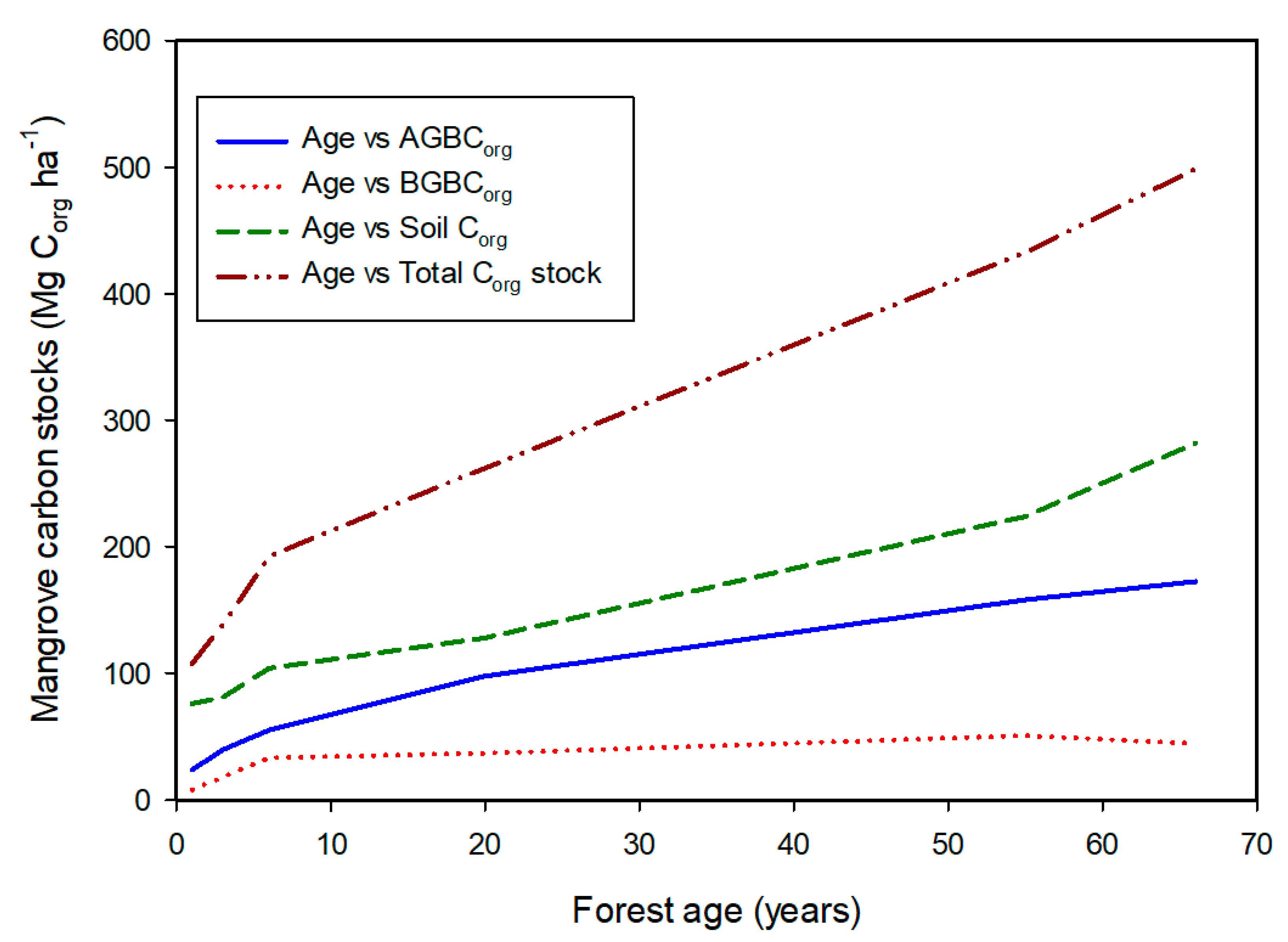
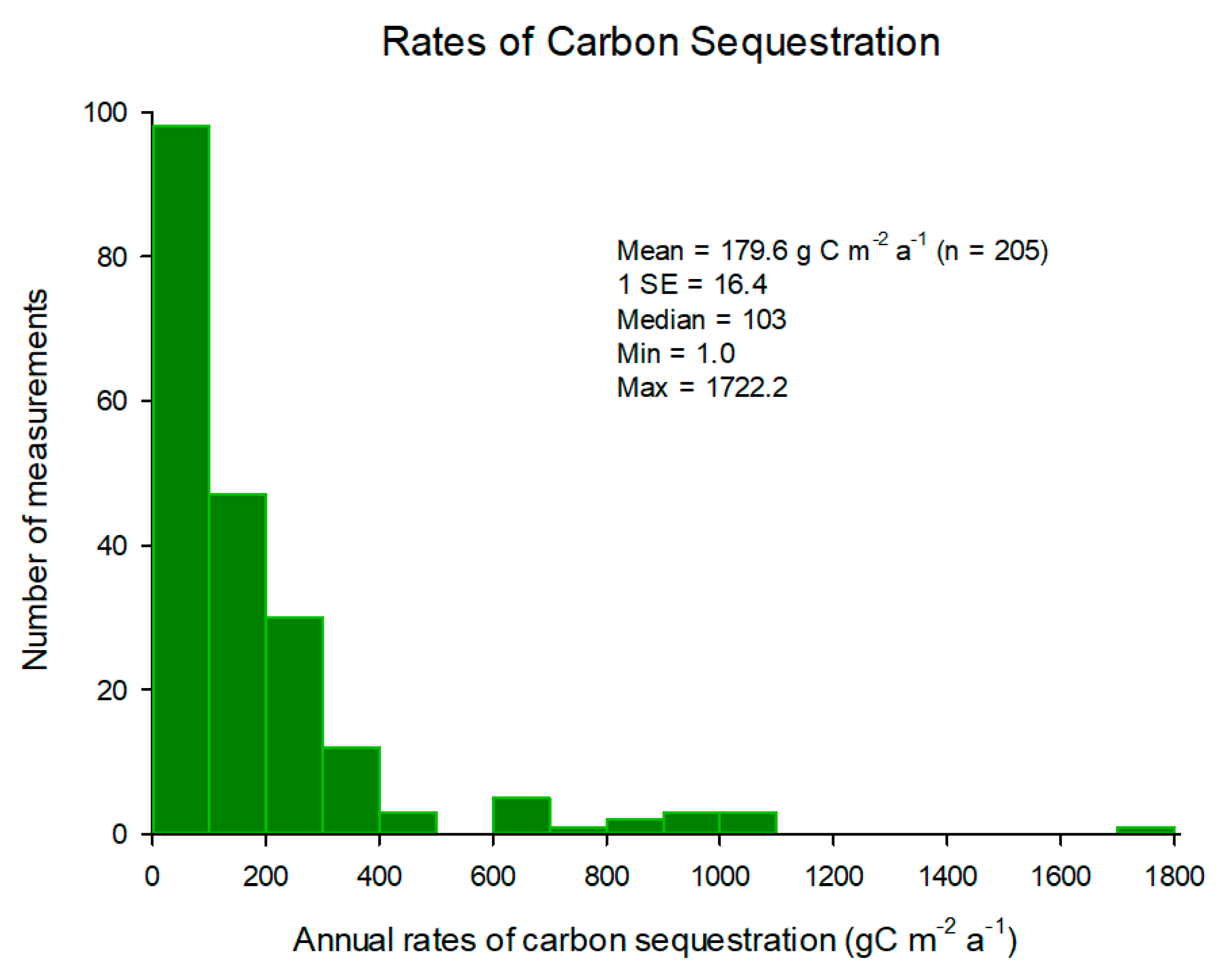
| Country | AGBCorg | BGBCorg | SCorg | Total Corg Stock |
|---|---|---|---|---|
| Africa | ||||
| Benin | 41.6 | 15.8 | ND | ND |
| Cameroon | 102.2 | 38.8 | 1961.1 | 2102.1 |
| Congo | 537.7 | 15.1 | 967.4 | 1520.2 |
| Gabon | 130.0 | 372.0 | 504.3 | 786.3 |
| Ghana | 165.1 | 37.5 | 310.9 | 466.0 |
| Guinea | 59.6 | 22.7 | ND | ND |
| Ivory Coast | 99.8 | 38.8 | ND | ND |
| Kenya | 101.1 | 68.8 | 643.6 | 806.7 |
| Liberia | 50.0 | 297.5 | 342.0 | 950.0 |
| Nigeria | 69.2 | 26.3 | ND | ND |
| Madagascar | 70.6 | 35.8 | 368.3 | 457.3 |
| Mozambique | 95.8 | 36.5 | 216.3 | 348.6 |
| Senegal | 34.0 | 401.0 | 240.0 | 675.0 |
| Sierra Leone | 62.7 | 23.8 | ND | ND |
| South Africa | 6.7 | ND | 228.1 | 234.8 |
| Tanzania | 55.7 | 50.2 | 293.4 | 397.1 |
| Togo | 42.9 | 16.3 | ND | ND |
| Southeast Asia | ||||
| Cambodia | ND | ND | ND | 657.4 |
| Indonesia | 142.0 | 335.9 | 420.1 | 794.9 |
| Malaysia | 119.7 | 5.9 | 763.0 | 894.4 |
| Myanmar | 20.7 | 18.4 | 167.0 | 206.1 |
| Philippines | 161.4 | 63.1 | 450.2 | 549.0 |
| Singapore | 105.0 | 39.9 | 307.3 | 452.3 |
| Thailand | 68.0 | 108.7 | 604.7 | 754.1 |
| Vietnam | 120.0 | 21.8 | 768.0 | 968.7 |
| South and East Asia | ||||
| Bangladesh | 81.4 | 42.3 | 438.9 | 565.6 |
| China | 89.5 | 30.3 | 380.1 | 499.9 |
| India | 88.0 | 33.6 | 81.3 | 248.5 |
| Japan | 57.9 | 27.0 | 154.2 | 239.1 |
| Pakistan | 93.3 | 39.0 | ND | ND |
| Sri Lanka | 151.7 | 30.0 | 362.1 | 543.7 |
| Central and North America and Caribbean | ||||
| Belize | 42.4 | 725.0 | 333.4 | 738.3 |
| Costa Rica | 101.4 | 484.0 | 480.5 | 845.0 |
| Dominican Republic | 50.5 | 112.3 | 690.8 | 853.5 |
| Honduras | 85.5 | 509 | 794.0 | 1222.4 |
| Mexico | 109.1 | 88.8 | 643.1 | 810.7 |
| Panama | 33.0 | 365.0 | 531.0 | 929.0 |
| USA | 62.7 | 12.6 | 201.4 | 272.5 |
| South America | ||||
| Brazil | 87.9 | 33.8 | 310.6 | 432.3 |
| Colombia | 84.2 | 382.2 | 159.0 | 648.2 |
| Ecuador | 100.7 | ND | 407.0 | 507.7 |
| French Guiana | 91.2 | 31.8 | 149.2 | 272.1 |
| Guyana | 176.5 | ND | ND | ND |
| Middle East | ||||
| Egypt | ND | ND | 389.4 | ND |
| Iran | 46.1 | 65.6 | 227.3 | 339.0 |
| Saudi Arabia | ND | ND | 92.0 | ND |
| United Arab Emirates | 25.4 | 31.7 | 123.2 | 180.4 |
| Australia and New Zealand | ||||
| Australia | 84.8 | 177.0 | 726.6 | 870.3 |
| New Zealand | 17.0 | 21.4 | 73.5 | 103.0 |
| Pacific Islands | ||||
| Hawaii | 179.3 | 78.3 | 197.1 | 464.0 |
| Kosrae | 256.4 | 237.9 | 694.1 | 1188.0 |
| Palau | 117.9 | 100.0 | 522.1 | 739.9 |
| Yap | 249.9 | 201.6 | 714.1 | 1165.7 |
| Global Means | ||||
| AGBCorg | BGBCorg | SCorg | Total Corg Stock | |
| Mean | 109.3 | 80.9 | 565.4 | 738.9 |
| ±1SE | 5.0 | 9.5 | 25.7 | 27.9 |
| n | 274 | 176 | 243 | 224 |
| Median | 94.1 | 34.1 | 500.5 | 702.5 |
| Min | 1.9 | 0.3 | 37.0 | 46.3 |
| Max | 537.7 | 866.0 | 2102.7 | 2205.0 |
| Disturbance | Location | Method for Estimating CO2 Emission | Years Since Disturbance | CO2 eq Emission | Reference |
|---|---|---|---|---|---|
| Deforestation | Belize | Flux chambers | 1 | 106 | [53] |
| 20 | 30 | ||||
| New Zealand | 0.1–8 | 21.4 | [54] | ||
| Cambodia | 10–15 | 48 | [55] | ||
| Conversion to aquaculture and/or cattle pastures | Indonesia | 25 | 16 | [56] | |
| Indonesia | 25 | 44 | |||
| Dominican Republic | Change in SCorg | 29 | 82 | [57] | |
| NE Brazil | Change in ecosystem Corg stock | 8–12 | 1392 a | [51] | |
| Mexico | 7–30 | 2610 a | [52] | ||
| Honduras | ND | 1068.4 a | |||
| Costa Rica | ND | 1811.9 a | |||
| Indonesia | ND | 2544.0 a | |||
| Dominican Republic | ND | 2781.5 a | |||
| Thailand | 10 | 179 | [58] | ||
| Mahakam delta, Borneo | 16 | 120 | [59] | ||
| Tree mortality | Kenya | Change in soil volume and gas flux | 2 | 25.3–35.6 | [60] |
| Hurricane/typhoon damage | Honduras | Difference in C inventory between disturbed and undisturbed mangroves | 2 | 18.7 | [61] |
| Global data | 30 | 33.9 | |||
| 30 | 27.2 | [62] | |||
| 30 | 20.4 | ||||
| Vietnam | Difference in C inventory between disturbed and undisturbed mangroves | 14 | 106.3 | [63] | |
| SW Florida | Loss total ecosystem Corg | 14 | 25.7–216.5 a | [64] | |
| Natural erosion, conversion to agriculture | Rufiji delta | C inventory and remote sensing | 16 | 119.7 | [65] |
| Zambezi delta | 16 | 98.9 | |||
| Ganges delta | 16 | 98.6 | |||
| Mekong delta | 16 | 88.4 | |||
| Abandoned fishponds | Philippines | ∆in C inventory abandoned and natural mangroves | 11–15 | 407.9 a | [66] |
| Various land use changes | Mexico | ∆in C inventory, loss of mangroves | 20 | 14.8 | [67] |
| Sundarbans, India | 38 | 3.7 b | [68] |
| Ecosystem | Area (106 ha) | Mean C Stock (Mg Corg ha−1) | Global Mean C Stock (Pg Corg) | Mean C Sequestration (g Corg m−2 a−1) | Global C Sequestration (Tg Corg a−1) | Current Conversion Rate (% a−1) | Carbon Emissions (Pg CO2-eq a−1) |
|---|---|---|---|---|---|---|---|
| Mangrove | 8.34 [6] | 738.9 a | 6.17 a | 179.6 a | 14.98 | 0.16 [13,15] | 0.088 b (0.036) |
| Salt Marsh | 5.50 [72] | 317.2 [6] | 1.74 | 212.0 [6] | 11.66 | 1.32 [73] | 0.084 |
| Seagrass | 16.0 [74] | 163.3 [6] | 2.61 | 220.7 [6] | 35.31 | 1.5 [7] | 0.144 |
| Coral Reef | 52.7 [75] | 0.6 [76,77] | 0.03 | 5.69 [78] | 3.0 | 0.43 [79] | 0.0005 |
| Tropical coastal ocean | 710.0 [71] | 50.7 [80] | 36.0 | 0.55 [71] | 3.9 | 0.93 c | 0.5 |
| Tropical forest | 1760 [81] | 314.2 [81] | 553.0 | 62.5 | 1100.0 [82] | 0.53 [83] | 10.8 |
| Temperate forest | 1040 [81] | 280.8 [81] | 292.1 | 28.9 | 300.0 [83] | 0.70 [84] | 7.5 |
| Boreal forest | 1370 [81] | 288.3 [85] | 395.0 | 18.0 [85] | 246.6 | 0.80 [84] | 11.6 |
| Tropical grassland/savanna | 2250 [81] | 202.4 [86] | 455.4 | 14.0 [86] | 315.0 | 0.70 [86] | 11.7 |
| Temperate grassland | 1250 [81] | 181.1 [86] | 226.4 | 16.8 | 210.0 [86] | 0.55 [87] | 4.6 |
| Desert and xericshrub land | 4550 [81] | 26.3 [88] | 119.7 | 9.5 [88] | 432.3 | 0.3 [88] | 1.3 |
| Montane grasslands/forests | 519 [89] | 173.9 [90,91] | 90.3 | ND | ND | 0.49 [92,93,94,95,96,97,98] | 1.6 |
| Mediterraneanforest | 322 [89] | 271.4 [99,100,101,102,103,104] | 87.4 | 65.8 [101,102,103] | 212.8 | ND | ND |
| Tundra | 835 [89] | 1779.6 [105,106,107,108,109,110,111] | 1486.0 | 63.2 [112,113,114,115,116] | 528.0 | ND | ND |
| Boreal peatlands | 361 [117] | 1182.8 | 427.0 [117] | 53.1 [117] | 191.7 | ND | 0.26 [117] |
| Tropical peatlands | 58.7 [117] | 2030.7 | 119.2 [117] | 54.2 [117] | 31.8 | ND | 1.48 [117] |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alongi, D.M. Global Significance of Mangrove Blue Carbon in Climate Change Mitigation. Sci 2020, 2, 67. https://doi.org/10.3390/sci2030067
Alongi DM. Global Significance of Mangrove Blue Carbon in Climate Change Mitigation. Sci. 2020; 2(3):67. https://doi.org/10.3390/sci2030067
Chicago/Turabian StyleAlongi, Daniel M. 2020. "Global Significance of Mangrove Blue Carbon in Climate Change Mitigation" Sci 2, no. 3: 67. https://doi.org/10.3390/sci2030067
APA StyleAlongi, D. M. (2020). Global Significance of Mangrove Blue Carbon in Climate Change Mitigation. Sci, 2(3), 67. https://doi.org/10.3390/sci2030067





