Organic Molecules: Is It Possible to Distinguish Aromatics from Aliphatics Collected by Space Missions in High-Speed Impacts?
Abstract
1. Introduction
Enceladus and Sampling Its Plumes
2. Materials and Methods
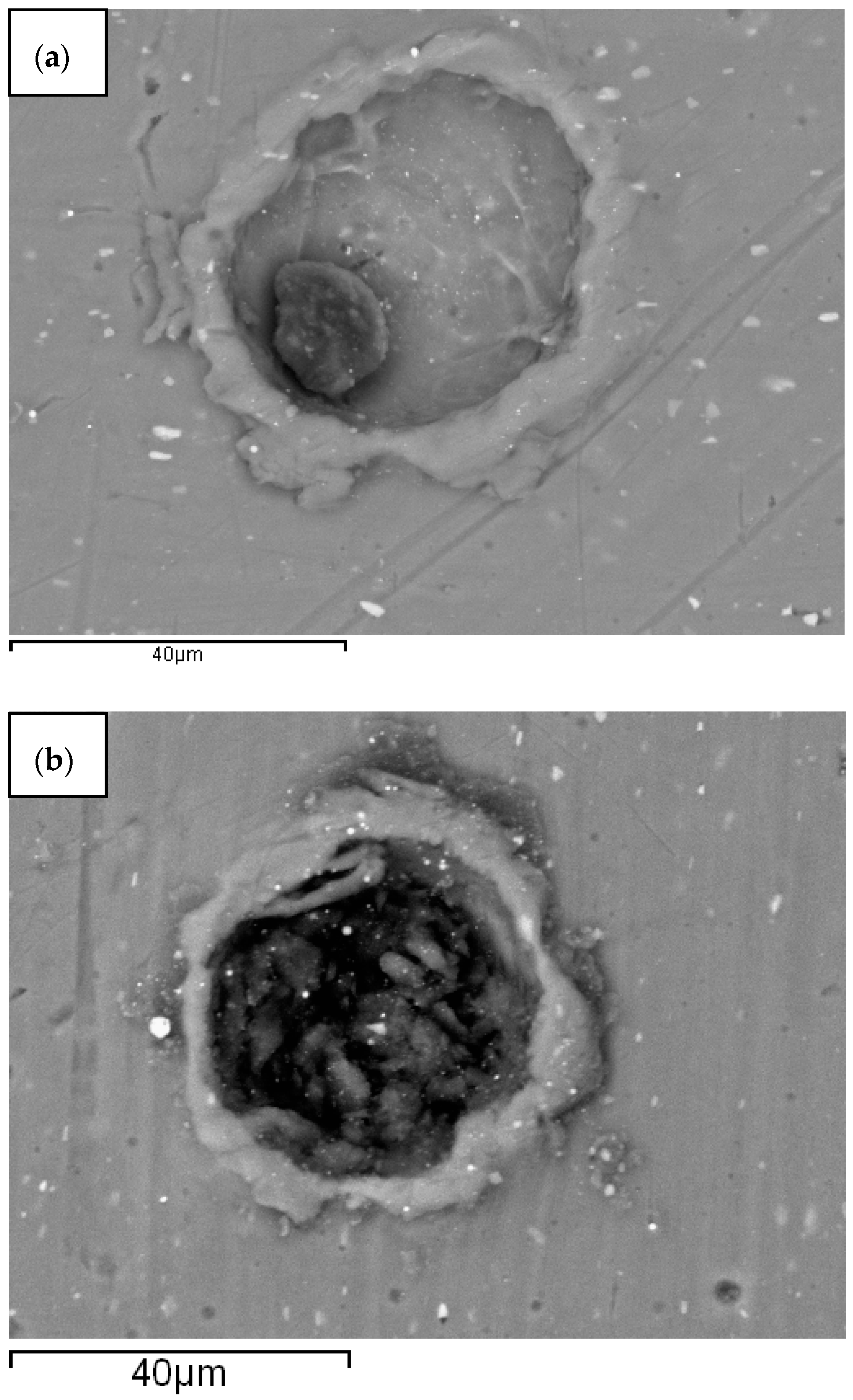
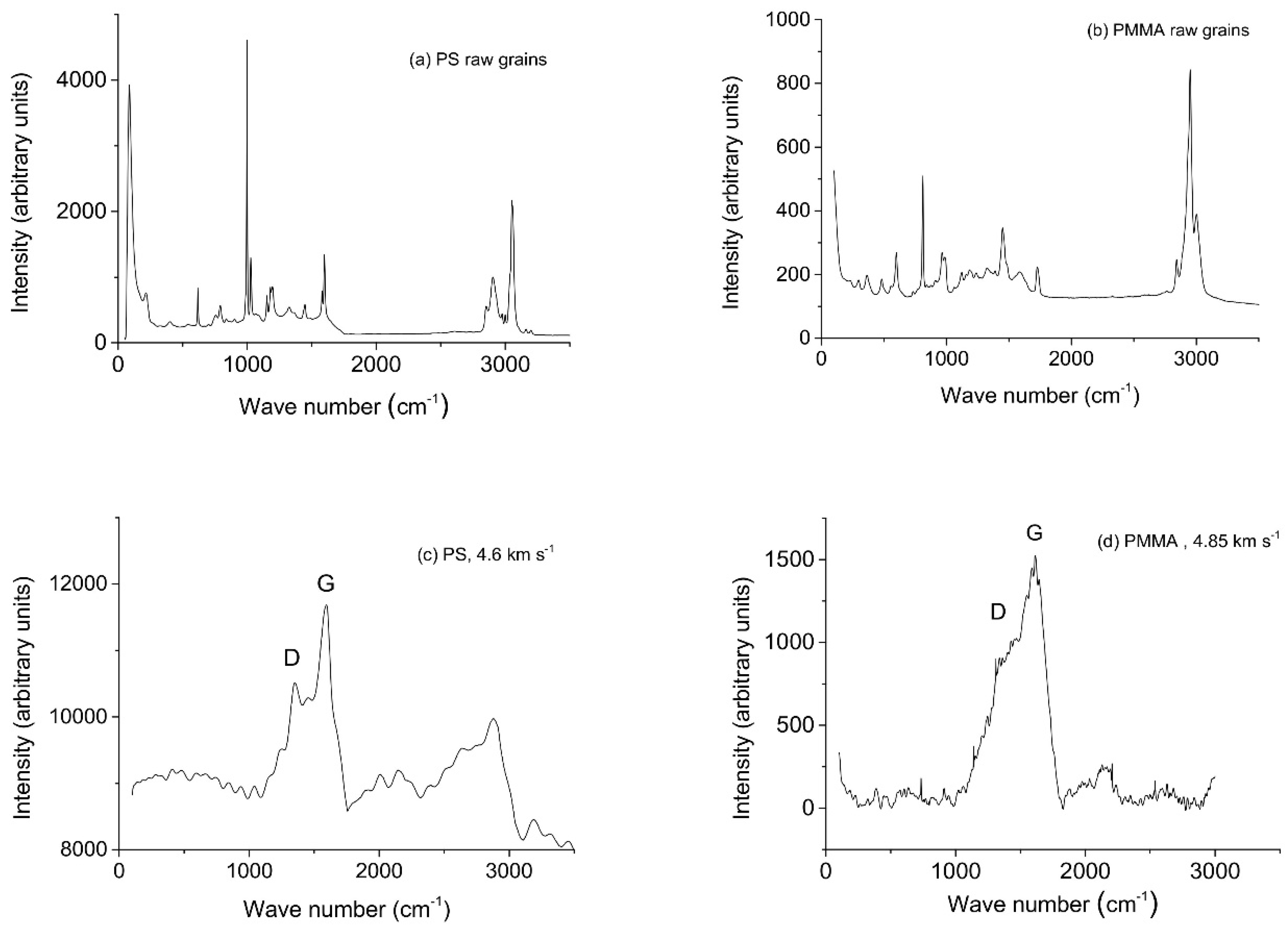
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hendrix, A.R.; Hurford, T.A.; Barge, L.M.; Bland, M.T.; Bowman, J.S.; Brinckerhoff, W.; Buratti, B.J.; Cable, M.L.; Castillo-Rogez, J.; Collins, G.C.; et al. The NASA Roadmap to Ocean Worlds. Astrobiology 2019, 19, 1–27. [Google Scholar] [CrossRef] [PubMed]
- Allamandola, L.J.; Tielens, A.G.G.M.; Barker, J.R. Polycyclic aromatic hydrocarbons and the unidentified infrared emission bands: Auto exhaust along the Milky Way. Astrophys. J. 1985, 290, L25–L28. [Google Scholar] [CrossRef]
- Tielens, A.G. Interstellar polycyclic aromatic hydrocarbon molecules. Ann. Rev. Astronomy Astrophys. 2008, 46, 289–337. [Google Scholar] [CrossRef]
- De Sanctis, M.C.; Ammannito, E.; McSween, H.Y.; Raponi, A.; Marchi, S.; Capaccioni, F.; Capria, M.T.; Carrozzo, F.G.; Ciarniello, M.; Fonte, S.; et al. Localized aliphatic organic material on the surface of Ceres. Science 2015, 355, 719–722. [Google Scholar] [CrossRef] [PubMed]
- Keller, L.P.; Bajt, S.; Baratta, G.A.; Borg, J.; Bradley, J.P.; Brownlee, D.E.; Busemann, H.; Brucato, J.R.; Burchell, M.; Colangeli, L.; et al. Infrared Spectroscopy of Comet 81P/Wild 2 Samples Returned by Stardust. Science 2006, 314, 1728–1731. [Google Scholar] [CrossRef]
- Sandford, S.A.; Aléon, J.; Alexander, C.M.; Araki, T.; Bajt, S.; Baratta, G.A.; Borg, J.; Bradley, J.P.; Brownlee, D.E.; Brucato, J.R.; et al. Organics Captured from Comet 81P/Wild 2 by the Stardust Spacecraft. Science 2006, 314, 1720–1724. [Google Scholar] [CrossRef]
- Waite, J.H.; Combi, M.R.; Ip, W.H.; Cravens, T.E.; McNutt, R.L.; Kasprzak, W.; Yelle, R.; Luhmann, J.; Niemann, H.; Gell, D.; et al. Cassini Ion and Neutral Mass Spectrometer: Enceladus Plume Composition and Structure. Science 2006, 311, 1419–1422. [Google Scholar] [CrossRef]
- Patthoff, D.A.; Kattenhorn, S.A. A fracture history on Enceladus provides evidence for a global ocean. Geophys. Res. Lett. 2011, 38, L18201. [Google Scholar] [CrossRef]
- Dougherty, M.K.; Khurana, K.K.; Neubauer, F.M.; Russell, C.T.; Saur, J.; Leisner, J.S.; Burton, M.E. Identification of a dynamic atmosphere at Enceladus with the Cassini magnetometer. Science 2006, 311, 1406–1409. [Google Scholar] [CrossRef]
- Thomas, P.C.; Tajeddine, R.; Tiscareno, M.S.; Burns, J.A.; Joseph, J.; Loredo, T.J.; Helfenstein, P.; Porco, C. Enceladus’s measured physical libration requires a global subsurface ocean. Icarus 2016, 264, 37–47. [Google Scholar] [CrossRef]
- Cadek, O.; Tobie, G.; Van Hoolst, T.; Masse, M.; Choblet, G.; Lefevre, A.; Mitri, G.; Baland, R.M.; Behounkova, M.; Bourgeois, O.; et al. Enceladus’s internal ocean and ice shell constrained from Cassini gravity, shape, and libration data. Geophys. Res. Lett. 2016, 43, 5653–5660. [Google Scholar] [CrossRef]
- Hansen, C.; Esposito, L.; Stewart, A.I.F.; Colwell, J.; Hendrix, A.; Pryor, W.; Shemansky, D.; West, R. Enceladus’ water vapor plume. Science 2006, 311, 1422–1426. [Google Scholar] [CrossRef]
- Nimmo, F.; Spencer, J.R.; Pappalardo, R.T.; Mullen, M.E. Shear heating as the origin of the plumes and heat flux on Enceladus. Nature 2007, 447, 289–291. [Google Scholar] [CrossRef]
- Porco, C.C.; Helfenstein, P.; Thomas, P.C.; Ingersoll, A.P.; Wisdom, J.; West, R.; Neukum, G.; Denk, T.; Wagner, R.; Roatsch, T.; et al. Cassini observes the active south pole of Enceladus. Science 2006, 311, 1393–1401. [Google Scholar] [CrossRef] [PubMed]
- Postberg, F.; Schmidt, J.; Hillier, J.; Kempf, S.; Srama, R. A salt-water reservoir as the source of a compositionally stratified plume on Enceladus. Nature 2011, 474, 620–622. [Google Scholar] [CrossRef] [PubMed]
- Postberg, F.; Khawaja, N.; Abel, B.; Choblet, G.; Glein, C.R.; Gudipati, M.S.; Henderson, B.L.; Hsu, H.-W.; Kempf, S.; Klenner, F.; et al. Macromolecular organic compounds from the depths of Enceladus. Nature 2018, 558, 564–568. [Google Scholar] [CrossRef] [PubMed]
- Khawaja, N.; Postberg, F.; Hillier, J.; Klenner, F.; Kemp, S.; Nölle, L.; Reviol, R.; Zou, Z.; Srama, R. Low-mass nitrogen-, oxygen-bearing, and aromatic compounds in Enceladean ice grains. Mon. Not. R. Astron. Soc. 2019, 489, 5231–5243. [Google Scholar] [CrossRef]
- McKay, C.P.; Anbar, A.D.; Porco, C.; Tsou, P. Follow the Plume: The Habitability of Enceladus. Astrobiology 2014, 14, 352–355. [Google Scholar] [CrossRef]
- National Academies of Sciences, Engineering, and Medicine. An Astrobiology Strategy for the Search for Life in the Universe; The National Academies Press: Washington, DC, USA, 2019. [Google Scholar]
- Goldsworthy, B.J.; Burchell, M.J.; Cole, M.J.; Green, S.F.; Leese, M.R.; McBride, N.; McDonnell, J.A.M.; Muller, M.; Grun, E.; Srama, R.; et al. Laboratory Calibration of the Cassini Cosmic Dust Analyser (CDA) Using New, Low Density Projectiles. Adv. Space Res. 2002, 29, 1139–1144. [Google Scholar] [CrossRef]
- Goldsworthy, B.J.; Burchell, M.J.; Cole, M.J.; Armes, S.P.; Khan, M.A.; Lascelles, S.F.; Green, S.F.; McDonnell, J.A.M.; Srama, R.; Bigger, S.W. Time of Flight Mass Spectrometry of Ions in Plasmas Produced by Hypervelocity Impacts of Organic and Mineralogical Microparticles on a Cosmic Dust Analyser. Astron. Astrophys. 2003, 409, 1151–1167. [Google Scholar] [CrossRef]
- Burchell, M.J.; Armes, S.P. Impact ionization spectra from aliphatic PMMA microparticles. Rapid Commun. Mass Spectrosc. 2011, 25, 543–550. [Google Scholar] [CrossRef] [PubMed]
- Fielding, L.A.; Hillier, J.K.; Burchell, M.J.; Armes, S.P. Space science applications for conducting polymer particles: Synthetic mimics for cosmic dust and micrometeorites. Chem. Commun. 2015, 51, 16886–16899. [Google Scholar] [CrossRef] [PubMed]
- Kearsley, A.T.; Burchell, M.J.; Hörz, F.; Cole, M.J.; Schwandt, C.S. Laboratory simulations of impacts upon aluminium foils of the Stardust spacecraft: Calibration of dust particle size from comet Wild2. Meteorit. Planet. Sci. 2006, 41, 167–180. [Google Scholar] [CrossRef]
- Kearsley, A.T.; Graham, G.A.; Burchell, M.J.; Cole, M.J.; Dai, Z.; Teslich, N.; Chater, R.J.; Wozniakiewicz, P.J.; Spratt, J.; Jones, G. Analytical scanning and transmission electron microscopy of laboratory impacts on Stardust aluminium foils: Interpreting impact crater morphology and the composition of impact residues. Meteorit. Planet. Sci. 2007, 42, 191–210. [Google Scholar] [CrossRef]
- Kearsley, A.T.; Burchell, M.J.; Price, M.C.; Graham, G.A.; Wozniakiewicz, P.J.; Cole, M.J.; Foster, N.J.; Teslich, N. Interpretation of Wild 2 dust fine structure: Comparison of Stardust aluminium foil craters to three dimensional shape of experimental impacts by artificial aggregate particles and meteorite powders. Meteorit. Planet. Sci. 2009, 44, 1489–1510. [Google Scholar] [CrossRef]
- Kearsley, A.T.; Borg, J.; Graham, G.A.; Burchell, M.J.; Cole, M.J.; Leroux, H.; Bridges, J.C.; Hörz, F.; Wozniakiewicz, P.J.; Bland, P.A.; et al. Dust from comet Wild 2: Interpreting particle size, shape, structure and composition from impact features on the Stardust aluminium foils. Meteorit. Planet. Sci. 2008, 43, 41–74. [Google Scholar] [CrossRef]
- Wozniakiewicz, P.J.; Kearsley, A.T.; Burchell, M.J.; Foster, N.J.; Cole, M.J.; Bland, P.A.; Russell, S.S. Analyses of Residues resulting from Laboratory Impacts into Aluminium 1100 Foil: Implications for Stardust Crater Analyses. Meteorit. Planet. Sci. 2009, 44, 1541–1560. [Google Scholar] [CrossRef]
- Burchell, M.J.; Kearsley, A.T. Short Period Jupiter Family Comets after Stardust. Planet. Space Sci. 2009, 57, 1146–1161. [Google Scholar] [CrossRef]
- Burchell, M.J.; Mann, J.; Creighton, J.A.; Kearsley, A.T.; Graham, G.; Franchi, I.A. Identification of Minerals and Meteoritic Materials via Raman Techniques after Capture in Hypervelocity Impacts on Aerogel. Meteorit. Planet. Sci. 2006, 41, 217–232. [Google Scholar] [CrossRef]
- Burchell, M.J.; Foster, N.J.; Kearsley, A.T.; Creighton, J.A. Identification of Mineral Impactors in Hypervelocity Impact Craters in Aluminium by Raman Spectroscopy of Residues. Meteorit. Planet. Sci. 2008, 43, 135–142. [Google Scholar] [CrossRef]
- Wozniakiewicz, P.J.; Kearsley, A.T.; Ishii, H.A.; Burchell, M.J.; Bradley, J.P.; Teslich, N.; Cole, M.J.; Price, M.C. The Origin of Crystalline Residues in Stardust Al Foils: Surviving Cometary Dust or Crystallized impact Melts? Meteorit. Planet. Sci. 2012, 47, 660–670. [Google Scholar] [CrossRef]
- Burchell, M.J.; Bowden, S.A.; Cole, M.; Price, M.C.; Parnell, J. Survival of Organic Materials in Hypervelocity Impacts of Ice on Sand, Ice, and Water in the Laboratory. Astrobiology 2014, 14, 473–485. [Google Scholar] [CrossRef]
- Burchell, M.J.; Cole, M.J.; McDonnell, J.A.M.; Zarnecki, J.C. Hypervelocity Impact Studies Using the 2 MV Van de Graaff Dust Accelerator and Two Stage Light Gas Gun of the University of Kent at Canterbury. Meas. Sci. Tech. 1999, 10, 41–50. [Google Scholar] [CrossRef]
- Schneider, N.M.; Burger, M.H.; Schaller, E.L.; Brown, M.E.; Johnson, R.E.; Kargel, J.S.; Dougherty, M.K.; Achilleos, N.A. No sodium in the vapour plumes of Enceladus. Nature 2009, 459, 1102–1104. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Itoh, N.; Shirono, K.; Fujimoto, T. Baseline Assessment for the Consistency of Raman Shifts Acquired with 26 Different Raman Systems and the Necessity of a Standardized Calibration Protocol. Anal. Sci. 2019, 35, 571–576. [Google Scholar] [CrossRef] [PubMed]
- Lascelles, S.F.; Armes, S.P.; Zhdan, P.A.; Greaves, S.J.; Brown, A.M.; Watts, J.F.; Leadley, S.R.; Luk, S.Y. Surface characterization of micrometre-sized, polypyrrole-coated polystyrene latexes: Verification of ‘core–shell’ morphology. J. Mater. Chem. 1997, 7, 1349–1355. [Google Scholar] [CrossRef]
- Thomas, K.J.; Sheeba, M.; Nampoori, V.P.N.; Vallabhan, C.P.G.; Radhakrishnan, P. Raman spectra of polymethyl methacrylate optical fibres excited by a 532 nm diode pumped solid state laser. J. Opt. Pure Appl. Opt. 2008, 10, 055303. [Google Scholar] [CrossRef]
- Pasteris, J.D.; Wopenka, B. Necessary, but not sufficient: Raman identification of disordered carbon as a signature of ancient life. Astrobiology 2003, 3, 727–738. [Google Scholar] [CrossRef]
- Melosh, H.J. Impact Cratering; Oxford University Press: Oxford, UK, 1989. [Google Scholar]
- Marsh, S.P. LASL Shock Hugoniot Data; University of California Press: Berkeley, CA, USA, 1980. [Google Scholar]
- Gault, D.E.; Heitowit, E.D. The partition of energy for hypervelocity impact craters formed in rock. Proc. Sixth Hypervelocity Impact Symp. 1963, 2, 419–456. [Google Scholar]
- Burchell, M.J.; Foster, N.J.; Ormond-Prout, J.; Dupin, D.; Armes, S.P. Extent of thermal ablation suffered by model organic microparticles during aerogel capture at hypervelocities. Meteorit. Planet. Sci. 2009, 44, 1407–1420. [Google Scholar] [CrossRef]
- New, J.S.; Mathies, R.A.; Price, M.C.; Cole, M.J.; Golozar, M.; Spathis, V.; Burchell, M.J.; Butterworth, A.L. Characterizing organic particle impacts on inert metal surfaces: Foundation for capturing organic molecules during hypervelocity transits of Enceladus plumes. Meteorit. Planet. Sci. 2020, 55, 465–479. [Google Scholar] [CrossRef] [PubMed]
- Burchell, M.J.; Graham, G.; Kearsley, A. Cosmic Dust Collection in Aerogel. Ann. Rev. Earth Planet. Sci. 2006, 34, 385–418. [Google Scholar] [CrossRef]
- Trigo-Rodríguez, J.; Domínguez, G.; Burchell, M.J.; Hörz, F.; Lorca, J. Bulbous tracks arising from hypervelocity capture in aerogel. Meteorit. Planet. Sci. 2008, 43, 75–86. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Burchell, M.; Harriss, K. Organic Molecules: Is It Possible to Distinguish Aromatics from Aliphatics Collected by Space Missions in High-Speed Impacts? Sci 2020, 2, 56. https://doi.org/10.3390/sci2030056
Burchell M, Harriss K. Organic Molecules: Is It Possible to Distinguish Aromatics from Aliphatics Collected by Space Missions in High-Speed Impacts? Sci. 2020; 2(3):56. https://doi.org/10.3390/sci2030056
Chicago/Turabian StyleBurchell, Mark, and Kathryn Harriss. 2020. "Organic Molecules: Is It Possible to Distinguish Aromatics from Aliphatics Collected by Space Missions in High-Speed Impacts?" Sci 2, no. 3: 56. https://doi.org/10.3390/sci2030056
APA StyleBurchell, M., & Harriss, K. (2020). Organic Molecules: Is It Possible to Distinguish Aromatics from Aliphatics Collected by Space Missions in High-Speed Impacts? Sci, 2(3), 56. https://doi.org/10.3390/sci2030056




