Effects of 12 Months of Vitamin D Supplementation on Physical Fitness Levels in Postmenopausal Women with Type 2 Diabetes
Abstract
1. Introduction
2. Methods
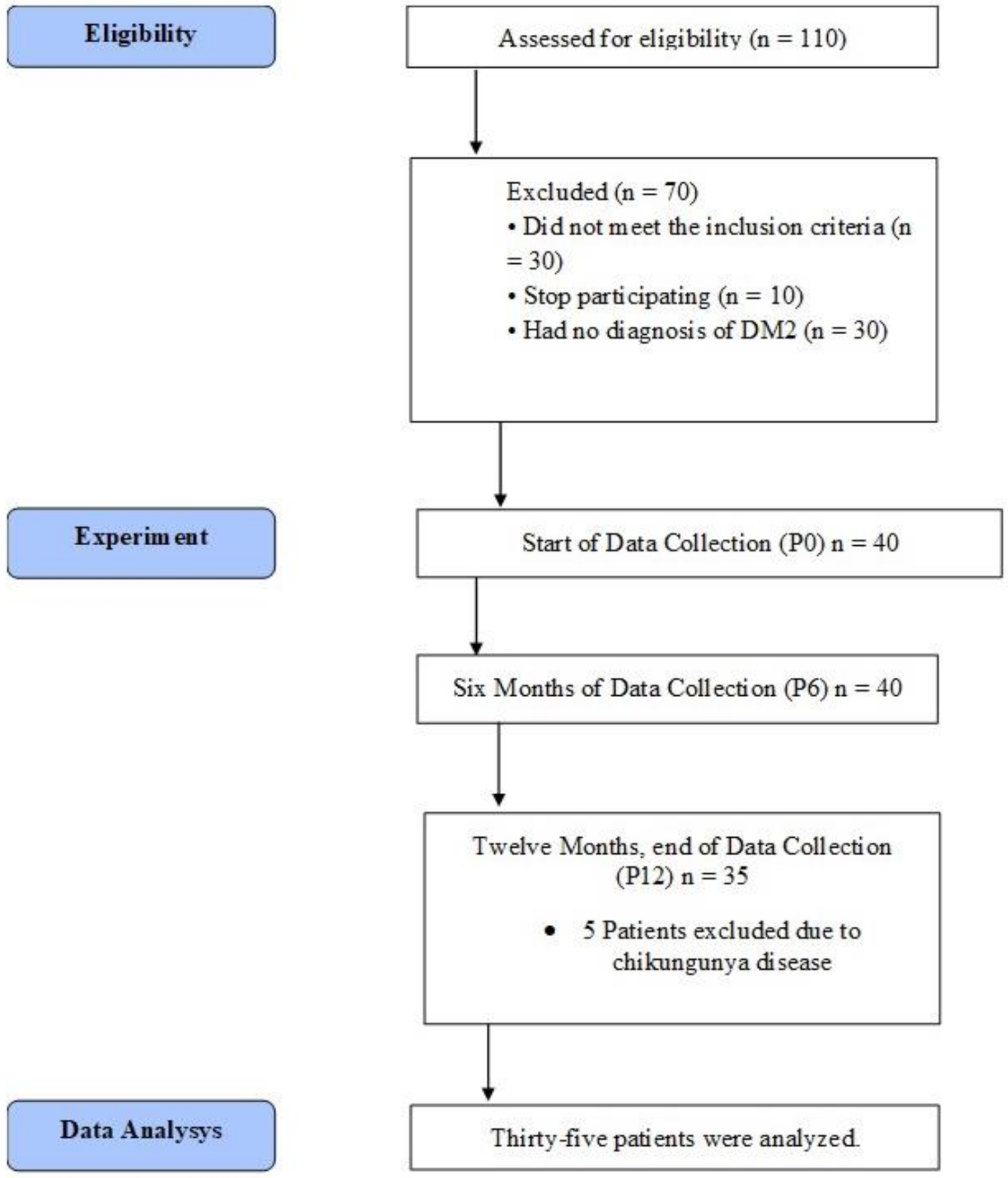
2.1. Participants and Research Design
2.2. Procedures
2.3. Physical Tests
2.4. Anthropometry and Body Composition
2.5. Blood Sample Analyses
2.6. Statistical Analyses
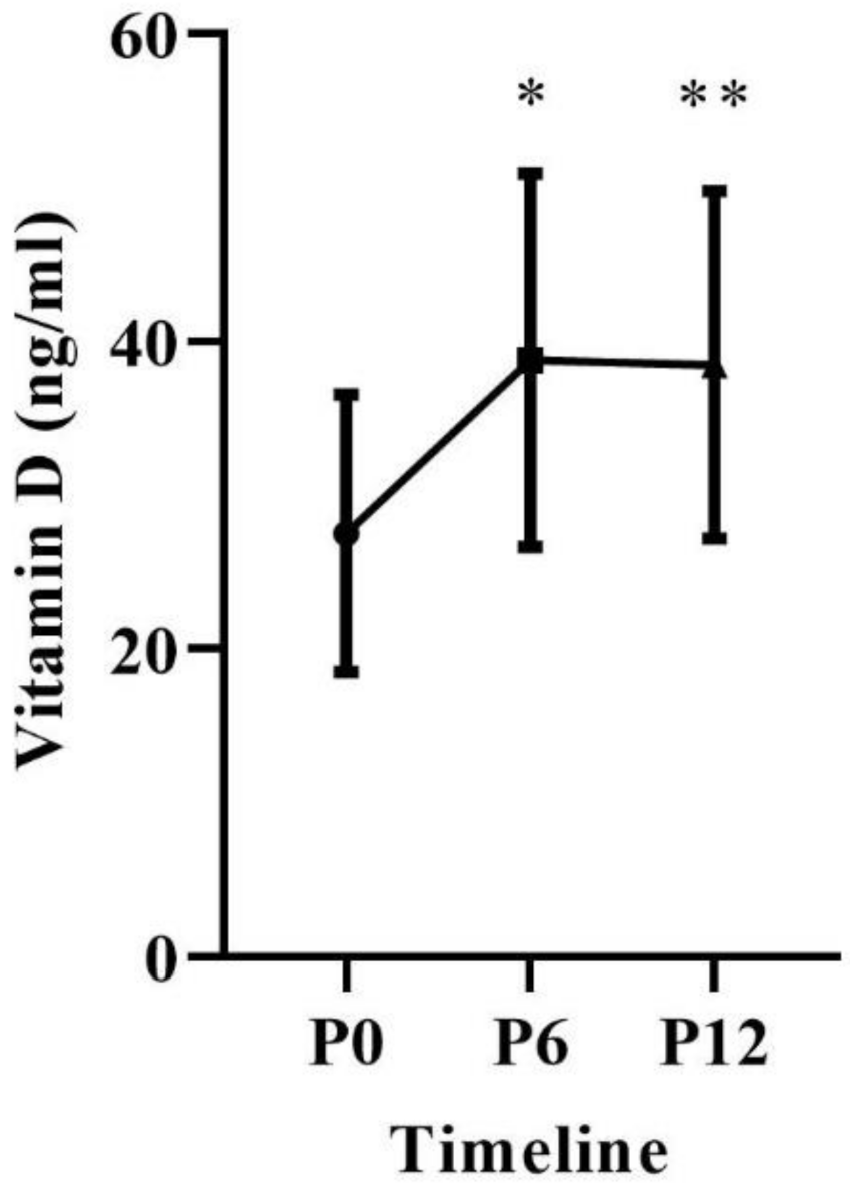
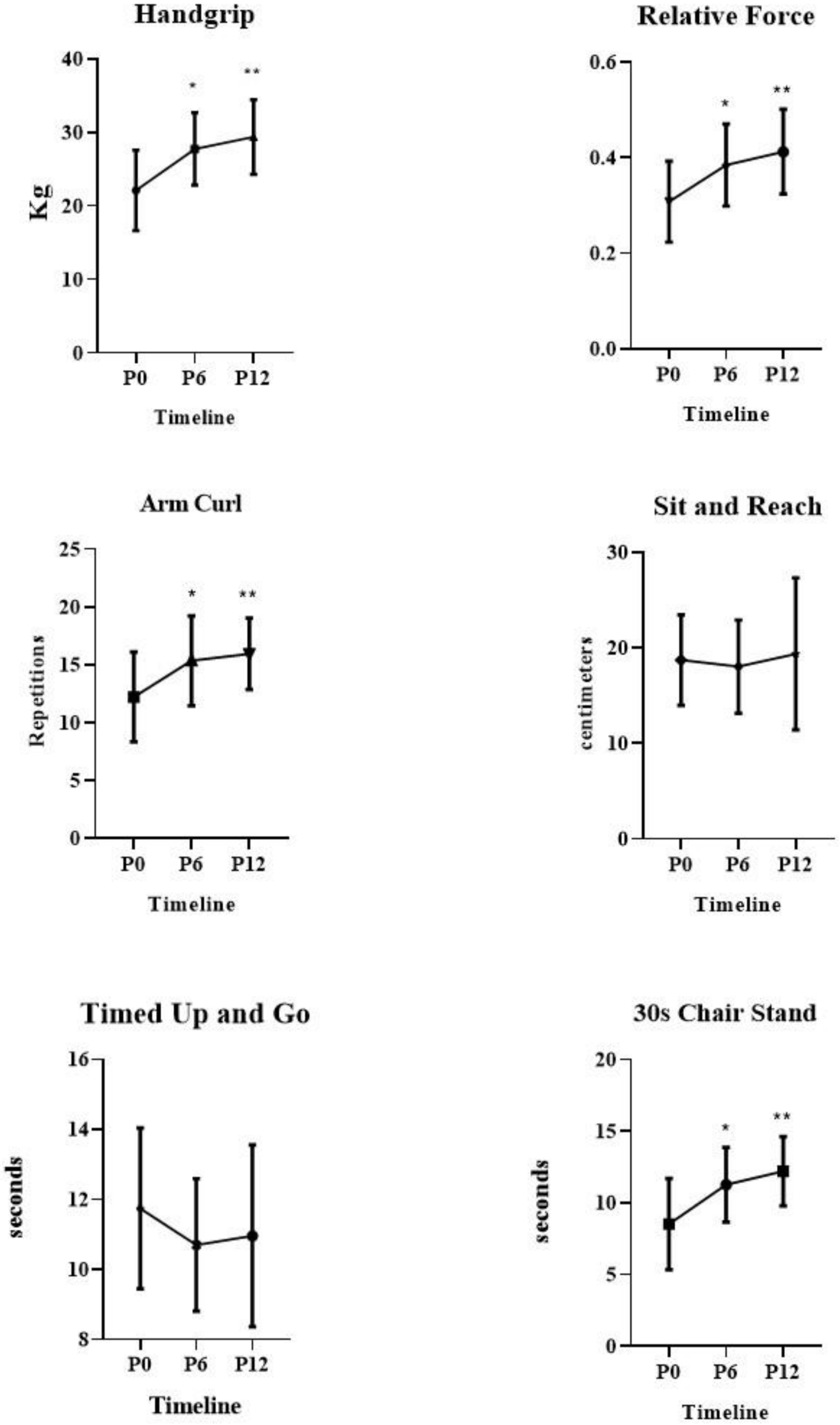
3. Results
Primary Outcomes
4. Secondary Outcomes
5. Discussion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Silverstein, N.M.; Fitzgerald, K.G. Educating a new generation of professionals in aging worldwide. Gerontol. Geriatr. Educ. 2017, 38, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Lobo, R.A.; Davis, S.R.; De Villiers, T.J.; Gompel, A.; Henderson, V.W.; Hodis, H.N.; Lumsden, M.A.; Mack, W.J.; Shapiro, S.; Baber, R. Prevention of diseases after menopause. Climacteric 2014, 17, 540–556. [Google Scholar] [CrossRef] [PubMed]
- Rose, J.; Weiser, T.G.; Hider, P.; Wilson, L.; Gruen, R.L.; Bickler, S.W. Estimated need for surgery worldwide based on prevalence of diseases: A modelling strategy for the WHO Global Health Estimate. Lancet Glob. Health 2015, 3 (Suppl. 2), S13–S20. [Google Scholar] [CrossRef]
- Zitkus, B.S. Update on the American Diabetes Association Standards of Medical Care. Nurse Pract. 2014, 39, 22–32, quiz 32–23. [Google Scholar] [CrossRef]
- Colberg, S.R.; Sigal, R.J.; Yardley, J.E.; Riddell, M.C.; Dunstan, D.W.; Dempsey, P.C.; Horton, E.S.; Castorino, K.; Tate, D.F. Physical Activity/Exercise and Diabetes: A Position Statement of the American Diabetes Association. Diabetes Care 2016, 39, 2065–2079. [Google Scholar] [CrossRef]
- Brincat, M.; Gambin, J.; Brincat, M.; Calleja-Agius, J. The role of vitamin D in osteoporosis. Maturitas 2015, 80, 329–332. [Google Scholar] [CrossRef]
- Guo, J.; Cockcroft, J.R.; Elwood, P.C.; Pickering, J.E.; Lovegrove, J.A.; Givens, D.I. Vitamin D intake and risk of CVD and all-cause mortality: Evidence from the Caerphilly Prospective Cohort Study. Public Health Nutr. 2017, 20, 2744–2753. [Google Scholar] [CrossRef]
- Lucato, P.; Solmi, M.; Maggi, S.; Bertocco, A.; Bano, G.; Trevisan, C.; Manzato, E.; Sergi, G.; Schofield, P.; Kouidrat, Y.; et al. Low vitamin D levels increase the risk of type 2 diabetes in older adults: A systematic review and meta-analysis. Maturitas 2017, 100, 8–15. [Google Scholar] [CrossRef]
- Wimalawansa, S.J. Associations of vitamin D with insulin resistance, obesity, type 2 diabetes, and metabolic syndrome. J. Steroid Biochem. Mol. Biol. 2018, 175, 177–189. [Google Scholar] [CrossRef]
- Latham, N.K.; Anderson, C.S.; Reid, I.R. Effects of vitamin D supplementation on strength, physical performance, and falls in older persons: A systematic review. J. Am. Geriatr. Soc. 2003, 51, 1219–1226. [Google Scholar] [CrossRef]
- Bentes, C.M.; Resende, M.; Miranda, H.; Netto, C.C.; Marinheiro, L.P.F. Can Vitamin D supplementation alone effective to increase a physical fitness levels in post-menopausal women with metabolic disorders? Brief Review. Diabetes Metab. Syndr. 2018, 12, 65–68. [Google Scholar] [CrossRef]
- Prestes, J.; Tibana, R.A. Muscular static strength test performance and health: Absolute or relative values. Rev. Assoc. Médica Bras. 2013, 59, 308–309. [Google Scholar] [CrossRef][Green Version]
- Podsiadlo, D.; Richardson, S. The timed “Up & Go”: A test of basic functional mobility for frail elderly persons. J. Am. Geriatr. Soc. 1991, 39, 142–148. [Google Scholar] [PubMed]
- Rikli, R.E.; Jones, C.J. Development and validation of a functional fitness test for community-residing older adults. J. Aging Phys. Act. 1999, 7, 129–161. [Google Scholar] [CrossRef]
- Finn, K.J.; Saint-Maurice, P.F.; Karsai, I.; Ihász, F.; Csányi, T. Agreement between Omron 306 and Biospace InBody 720 bioelectrical impedance analyzers (BIA) in children and adolescents. Res. Q. Exerc. Sport 2015, 86 (Suppl. 1), S58–S65. [Google Scholar] [CrossRef] [PubMed]
- Cangussu, L.M.; Nahas-Neto, J.; Orsatti, C.L.; Bueloni-Dias, F.N.; Nahas, E.A. Effect of vitamin D supplementation alone on muscle function in postmenopausal women: A randomized, double-blind, placebo-controlled clinical trial. Osteoporos. Int. 2015, 26, 2413–2421. [Google Scholar] [CrossRef]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef]
- Bischoff-Ferrari, H.A.; Giovannucci, E.; Willett, W.C.; Dietrich, T.; Dawson-Hughes, B. Estimation of optimal serum concentrations of 25-hydroxyvitamin D for multiple health outcomes. Am. J. Clin. Nutr. 2006, 84, 18–28. [Google Scholar] [CrossRef]
- Bischoff-Ferrari, H.A.; Dawson-Hughes, B.; Willett, W.C.; Staehelin, H.B.; Bazemore, M.G.; Zee, R.Y.; Wong, J.B. Effect of Vitamin D on falls: A meta-analysis. JAMA 2004, 291, 1999–2006. [Google Scholar] [CrossRef]
- Bischoff-Ferrari, H.A.; Willett, W.C.; Wong, J.B.; Stuck, A.E.; Staehelin, H.B.; Orav, E.J.; Thoma, A.; Kiel, D.; Henschkowski, J. Prevention of nonvertebral fractures with oral vitamin D and dose dependency: A meta-analysis of randomized controlled trials. Arch. Intern. Med. 2009, 169, 551–561. [Google Scholar] [CrossRef] [PubMed]
- Perez-Lopez, F.R.; Brincat, M.; Erel, C.T.; Tremollieres, F.; Gambacciani, M.; Lambrinoudaki, I.; Moen, M.H.; Schenck-Gustafsson, K.; Vujovic, S.; Rozenberg, S.; et al. EMAS position statement: Vitamin D and postmenopausal health. Maturitas 2012, 71, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Bischoff, H.A.; Borchers, M.; Gudat, F.; Duermueller, U.; Theiler, R.; Stahelin, H.B.; Dick, W. In situ detection of 1,25-dihydroxyvitamin D3 receptor in human skeletal muscle tissue. Histochem. J. 2001, 33, 19–24. [Google Scholar] [CrossRef]
- Ceglia, L.; Niramitmahapanya, S.; da Silva Morais, M.; Rivas, D.A.; Harris, S.S.; Bischoff-Ferrari, H.; Fielding, R.A.; Dawson-Hughes, B. A randomized study on the effect of vitamin D3 supplementation on skeletal muscle morphology and vitamin D receptor concentration in older women. J. Clin. Endocrinol. Metab. 2013, 98, E1927–E1935. [Google Scholar] [CrossRef]
- Annweiler, C.; Beauchet, O.; Berrut, G.; Fantino, B.; Bonnefoy, M.; Herrmann, F.; Schott, A.M. Is there an association between serum 25-hydroxyvitamin D concentration and muscle strength among older women? Results from baseline assessment of the EPIDOS study. JNHA-J. Nutr. Health Aging 2009, 13, 90–95. [Google Scholar] [CrossRef]
- Zhu, K.; Austin, N.; Devine, A.; Bruce, D.; Prince, R.L. A randomized controlled trial of the effects of vitamin D on muscle strength and mobility in older women with vitamin D insufficiency. J. Am. Geriatr. Soc. 2010, 58, 2063–2068. [Google Scholar] [CrossRef]
- Anek, A.; Bunyaratavej, N.; Jittivilai, T. Effects of Short-Term Vitamin D Supplementation on Musculoskeletal and Body Balance for Prevention of Falling in Postmenopausal Women. J. Med. Assoc. Thai 2015, 98 (Suppl. 8), S26–S31. [Google Scholar]
- Issa, C.M. Vitamin D and Type 2 Diabetes Mellitus. Adv. Exp. Med. Biol. 2017, 996, 193–205. [Google Scholar] [CrossRef] [PubMed]
- Davis, S.R.; Castelo-Branco, C.; Chedraui, P.; Lumsden, M.; Nappi, R.; Shah, D.; Villaseca, P. Understanding weight gain at menopause. Climacteric 2012, 15, 419–429. [Google Scholar] [CrossRef] [PubMed]
- Simpson, C.A.; Cusano, A.M.; Bihuniak, J.; Walker, J.; Insogna, K.L. Effect of 25(OH) vitamin D reference method procedure (RMP) alignment on clinical measurements obtained with the IDS-iSYS chemiluminescent-based automated analyzer. J. Steroid Biochem. Mol. Biol. 2015, 148, 41–46. [Google Scholar] [CrossRef]
| Mean | ± | SD | K-S | |
|---|---|---|---|---|
| Age (years) | 62.48 | ± | 7.67 | 0.067 |
| Height (cm) | 154.6 | ± | 5.11 | 0.798 |
| Body mass (kg) | 73.93 | ± | 15.43 | 0.052 |
| Waist circumference (cm) | 96.99 | ± | 14.25 | 0.636 |
| Abdominal circumference (cm) | 98.46 | ± | 11.54 | 0.666 |
| Iliac circumference (cm) | 101.42 | ± | 12.24 | 0.545 |
| Hip circumference (cm) | 104.05 | ± | 10.20 | 0.195 |
| Waist/hip ratio | 0.93 | ± | 0.08 | 0.336 |
| BMI (kg/m2) | 31.13 | ± | 5.82 | 0.404 |
| Lean body mass (kg) | 22.29 | ± | 3.23 | 0.162 |
| Fat mass (kg) | 33.17 | ± | 10.87 | 0.355 |
| Fat percentage (%) | 43.62 | ± | 6.36 | 0.269 |
| Visceral fat area (cm2) | 124.36 | ± | 31.06 | 0.248 |
| Resting Metabolic Rate (kcal/d) | 1259.54 | ± | 116.20 | 0.112 |
| Vitamin D status (ng/mL) | 27.47 | ± | 8.98 | 0.073 |
| Fasting glucose (mg/dL) | 144.65 | ± | 55.99 | 0.092 |
| Variable | Timeline | Mean | ± | Standard Deviation | Confidence Interval 95% | |
|---|---|---|---|---|---|---|
| Lower | Higher | |||||
| Age (years) | P0 | 62.64 | ± | 7.64 | 60.4 | 65.1 |
| P6 | 63.02 | ± | 7.68 | 60.6 | 65.6 | |
| P12 | 63.05 | ± | 7.30 | 60.6 | 65.6 | |
| Body Mass (Kg) | P0 | 73.80 | ± | 15.26 | 69.3 | 78.6 |
| P6 | 74.03 | ± | 14.74 | 69.5 | 78.6 | |
| P12 | 73.02 | ± | 14.17 | 68.7 | 77.8 | |
| Waist circumference (cm) | P0 | 96.93 | ± | 14.07 | 92.9 | 101.1 |
| P6 | 94.46 | ± | 13.41 | 90.2 | 98.6 | |
| P12 | 92.91 | ± | 14.02 | 88.6 | 97.6 | |
| Abdominal circumference (cm) | P0 | 98.41 | ± | 11.39 | 95.2 | 101.9 |
| P6 | 98.28 | ± | 12.15 | 94.6 | 102.1 | |
| P12 | 98.64 | ± | 11.92 | 94.8 | 102.5 | |
| Iliac circumference (cm) | P0 | 101.68 | ± | 12.19 | 98.2 | 105.6 |
| P6 | 101.82 | ± | 11.05 | 98.4 | 105.4 | |
| P12 | 103.80 | ± | 11.56 | 100.1 | 107.5 | |
| Hip circumference (cm) | P0 | 104.13 | ± | 10.07 | 101.1 | 107.3 |
| P6 | 104.60 | ± | 9.13 | 101.7 | 107.5 | |
| P12 | 104.85 | ± | 9.68 | 101.7 | 108.0 | |
| WHR | P0 | 0.93 | ± | 0.08 | 0.9 | 1.0 |
| P6 | 0.90 | ± | 0.08 | 0.9 | 0.9 | |
| P12 | 0.88 * | ± | 0.08 | 0.9 | 0.9 | |
| Lean body mass (kg) | P0 | 31.20 | ± | 5.76 | 29.4 | 33.1 |
| P6 | 31.07 | ± | 5.72 | 29.3 | 32.8 | |
| P12 | 30.72 | ± | 5.61 | 29.0 | 32.7 | |
| Bone Mass | P0 | 2.34 | ± | 0.29 | 2.3 | 2.4 |
| P6 | 2.32 | ± | 0.29 | 2.2 | 2.4 | |
| P12 | 2.31 | ± | 0.28 | 2.2 | 2.4 | |
| Lean body mass | P0 | 22.17 | ± | 3.28 | 21.2 | 23.2 |
| P6 | 22.53 | ± | 7.53 | 20.4 | 25.2 | |
| P12 | 21.71 | ± | 3.25 | 20.7 | 22.8 | |
| Fat mass | P0 | 33.24 | ± | 10.74 | 30.2 | 36.5 |
| P6 | 33.29 | ± | 10.57 | 30.0 | 36.7 | |
| P12 | 32.80 | ± | 10.25 | 29.7 | 36.3 | |
| Fat percentage (%) | P0 | 43.84 | ± | 6.42 | 41.9 | 45.9 |
| P6 | 44.35 | ± | 6.80 | 42.3 | 46.3 | |
| P12 | 44.05 | ± | 6.56 | 41.7 | 46.2 | |
| Visceral fat area | P0 | 124.09 | ± | 31.42 | 114.7 | 133.6 |
| P6 | 123.26 | ± | 29.43 | 114.5 | 132.5 | |
| P12 | 123.39 | ± | 28.95 | 114.4 | 133.5 | |
| Rest Metabolic Rate | P0 | 1255.10 | ± | 118.08 | 1219.5 | 1290.5 |
| P6 | 1249.73 | ± | 115.25 | 1215.6 | 1285.8 | |
| P12 | 1238.94 | ± | 114.32 | 1203.1 | 1277.8 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bentes, C.M.; Costa, P.B.; Resende, M.; Netto, C.; Dias, I.; da Silveira, A.L.B.; Di Masi, F.; Miranda, H.; de Carvalho, L.M.; Marinheiro, L. Effects of 12 Months of Vitamin D Supplementation on Physical Fitness Levels in Postmenopausal Women with Type 2 Diabetes. J. Funct. Morphol. Kinesiol. 2021, 6, 87. https://doi.org/10.3390/jfmk6040087
Bentes CM, Costa PB, Resende M, Netto C, Dias I, da Silveira ALB, Di Masi F, Miranda H, de Carvalho LM, Marinheiro L. Effects of 12 Months of Vitamin D Supplementation on Physical Fitness Levels in Postmenopausal Women with Type 2 Diabetes. Journal of Functional Morphology and Kinesiology. 2021; 6(4):87. https://doi.org/10.3390/jfmk6040087
Chicago/Turabian StyleBentes, Claudio Melibeu, Pablo B. Costa, Monique Resende, Claudia Netto, Ingrid Dias, Anderson Luiz Bezerra da Silveira, Fabrizio Di Masi, Humberto Miranda, Lucas Monteiro de Carvalho, and Lizanka Marinheiro. 2021. "Effects of 12 Months of Vitamin D Supplementation on Physical Fitness Levels in Postmenopausal Women with Type 2 Diabetes" Journal of Functional Morphology and Kinesiology 6, no. 4: 87. https://doi.org/10.3390/jfmk6040087
APA StyleBentes, C. M., Costa, P. B., Resende, M., Netto, C., Dias, I., da Silveira, A. L. B., Di Masi, F., Miranda, H., de Carvalho, L. M., & Marinheiro, L. (2021). Effects of 12 Months of Vitamin D Supplementation on Physical Fitness Levels in Postmenopausal Women with Type 2 Diabetes. Journal of Functional Morphology and Kinesiology, 6(4), 87. https://doi.org/10.3390/jfmk6040087








