The Effects of Alcohol Consumption on Recovery Following Resistance Exercise: A Systematic Review
Abstract
1. Introduction
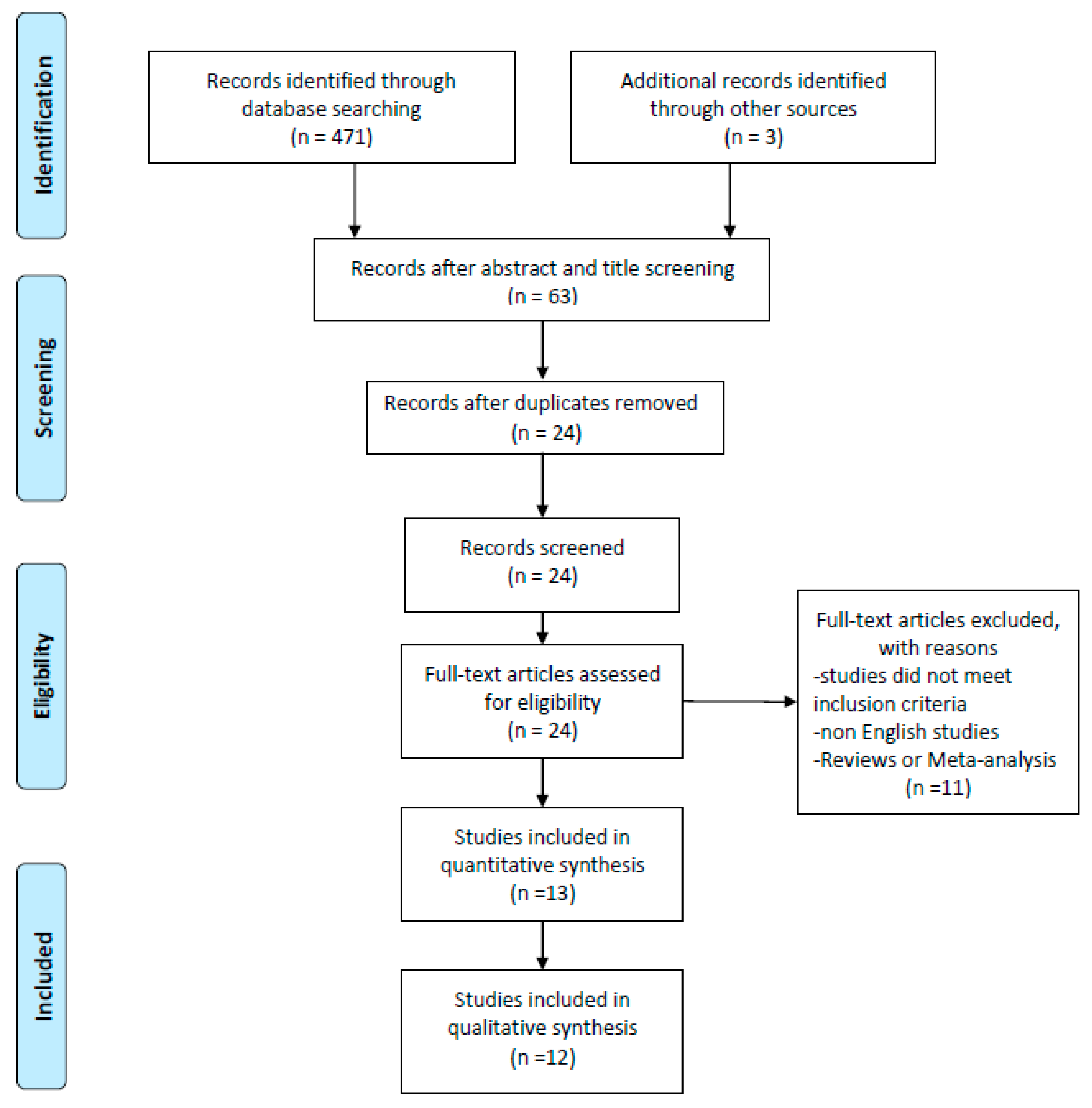
2. Materials and Methods
2.1. Inclusion and Exclusion Criteria
2.2. Eligibility Criteria
2.3. Participants
2.4. Interventions
2.5. Comparators
2.6. Outcomes
2.7. Search Strategy
2.8. Selection of Study Objects
2.9. Risk of Bias
2.10. Data Synthesis
3. Results
3.1. Biological Measures
3.1.1. Creatine Kinase
3.1.2. Heart Rate
3.1.3. Lactate
3.1.4. Blood glucose
3.1.5. Urine Measures
3.1.6. Cortisol
3.1.7. Testosterone
3.1.8. Estradiol
3.1.9. Sexual Hormone Binding Globulin
3.1.10. Leukocytes and Cytokines
3.1.11. C-reactive Protein
3.1.12. Plasma Amino Acids
3.1.13. Intracellular Signaling Proteins and Rates of Muscle Protein Synthesis
3.1.14. Calcium
3.2. Physical Measures
3.2.1. Force
3.2.2. Power
3.2.3. Muscular Endurance
3.2.4. Soreness
3.2.5. Rate of Perceived Exertion
3.3. Cognitive Function
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Schoenfeld, B. Science and Development of Muscle Hypertrophy. 2016. Available online: http://public.eblib.com/choice/publicfullrecord.aspx?p=4746185 (accessed on 13 May 2019).
- Kraemer, W.; Ratamess, N. Physiology of resistance training: Current issues. Orthop. Physic. Ther. Clin. N. Am. 2000, 9, 467–513. [Google Scholar]
- Harries, S.K.; Lubans, D.R.; Callister, R. Resistance training to improve power and sports performance in adolescent athletes: A systematic review and meta-analysis. J. Sci. Med. Sport 2012, 15, 532–540. [Google Scholar] [CrossRef] [PubMed]
- American College of Sports. American College of Sports Medicine position stand. Progression models in resistance training for healthy adults. Med. Sci. Sports Exerc. 2009, 41, 687–708. [Google Scholar] [CrossRef] [PubMed]
- Hausswirth, C. Recovery for Performance in Sport; Human Kinetics: Champaign, IL, USA, 2019. [Google Scholar]
- Barnett, A. Using recovery modalities between training sessions in elite athletes: Does it help? Sports Med. 2006, 36, 781–796. [Google Scholar] [CrossRef] [PubMed]
- Halson, S. Recovery techniques for athletes. Aspetar Sports Med. J. 2013, 26, 1–6. [Google Scholar]
- Vella, L.D.; Cameron-Smith, D. Alcohol, athletic performance and recovery. Nutrients 2010, 2, 781–789. [Google Scholar] [CrossRef]
- Parr, E.B.; Camera, D.M.; Areta, J.L.; Burke, L.M.; Phillps, S.M.; Hawley, J.A.; Coffey, V.G. Alcohol ingestion impairs maximal post-exercise rates of myofibrillar protein synthesis following a single bout of concurrent training. PLoS ONE 2014, 9, e88384. [Google Scholar] [CrossRef] [PubMed]
- Barnes, M.J. Alcohol: Impact on sports performance and recovery in male athletes. Sports Med. 2014, 44, 909–919. [Google Scholar] [CrossRef] [PubMed]
- Mager, A. ’White liquor hits black livers’: Meanings of excessive liquor consumption in South Africa in the second half of the twentieth century. Soc. Sci. Med. 2004, 59, 735–751. [Google Scholar] [CrossRef]
- Miguez, H.A. Epidemiology of alcohol consumption in Argentina. Vertex 2003, 14, 19–26. [Google Scholar]
- Pretorius, L.; Naidoo, A.; Reddy, S.P. “Kitchen cupboard drinking”: A review of South African women’s secretive alcohol addiction, treatment history, and barriers to accessing treatment. Soc. Work Public Health 2009, 24, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Rachdaoui, N.; Sarkar, D.K. Effects of alcohol on the endocrine system. Endocrinol. Metab. Clin. N. Am. 2013, 42, 593–615. [Google Scholar] [CrossRef] [PubMed]
- Lang, C.H.; Pruznak, A.M.; Nystrom, G.J.; Vary, T.C. Alcohol-induced decrease in muscle protein synthesis associated with increased binding of mTOR and raptor: Comparable effects in young and mature rats. Nutr. Metab. 2009, 6, 4. [Google Scholar] [CrossRef] [PubMed]
- Vary, T.C.; Frost, R.A.; Lang, C.H. Acute alcohol intoxication increases atrogin-1 and MuRF1 mRNA without increasing proteolysis in skeletal muscle. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2008, 294, R1777–R1789. [Google Scholar] [CrossRef] [PubMed]
- Downs, S.H.; Black, N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J. Epidemiol. Commun. Health 1998, 52, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Barnes, M.J.; Mundel, T.; Stannard, S.R. Acute alcohol consumption aggravates the decline in muscle performance following strenuous eccentric exercise. J. Sci. Med. Sport 2010, 13, 189–193. [Google Scholar] [CrossRef] [PubMed]
- Barnes, M.J.; Mundel, T.; Stannard, S.R. The effects of acute alcohol consumption on recovery from a simulated rugby match. J. Sports Sci. 2012, 30, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Clarkson, P.M.; Reichsman, F. The effect of ethanol on exercise-induced muscle damage. J. Stud. Alcohol 1990, 51, 19–23. [Google Scholar] [CrossRef]
- Haugvad, A.; Haugvad, L.; Hamarsland, H.; Paulsen, G. Ethanol Does Not Delay Muscle Recovery, but Decreases the Testosterone: Cortisol Ratio. Med. Sci. Sports Exerc. 2014, 46, 2175–2183. [Google Scholar] [CrossRef]
- Levitt, D.E.; Hiu-Ying, L.; Duplanty, A.A.; McFarlin, B.K.; Hill, D.W.; Vingreen, J.L. Effect of alcohol after muscle-damaging resistance exercise on muscular performance recovery and inflammatory capacity in women. Eur. J. Appl. Physiol. 2017, 117, 1195–1206. [Google Scholar] [CrossRef]
- McLeay, Y.; Stannard, S.R.; Mundel, L.; Foskett, A.; Barnes, M. Effect of Alcohol Consumption on Recovery From Eccentric Exercise Induced Muscle Damage in Females. Int. J. Sport. Nutr. Exerc. Metab. 2017, 27, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Murphy, A.P.; Snape, A.E.; Minett, G.M.; Skein, M.; Duffield, R. The effect of post-match alcohol ingestion on recovery from competitive rugby league matches. J. Strength Cond. Res. 2013, 27, 1304–1312. [Google Scholar] [CrossRef] [PubMed]
- Poulsen, M.B.; Jakobsen, J.; Aagaard, N.K.; Andersen, H. Motor performance during and following acute alcohol intoxication in healthy non-alcoholic subjects. Eur. J. Appl. Physiol. 2007, 101, 513–523. [Google Scholar] [CrossRef] [PubMed]
- Vingren, J.L.; Hill, D.W.; Buddhadev, H.; Duplanty, A. Postresistance exercise ethanol ingestion and acute testosterone bioavailability. Med. Sci. Sports Exerc 2013, 45, 1825–1832. [Google Scholar] [CrossRef] [PubMed]
- Barnes, M.J.; Mundel, T.; Stannard, S.R. Post-exercise alcohol ingestion exacerbates eccentric-exercise induced losses in performance. Eur. J. Appl. Physiol. 2010, 108, 1009–1014. [Google Scholar] [CrossRef] [PubMed]
- Levitt, D.E.; Idemudia, N.O.; Cregar, C.M.; Duplanty, A.A.; Hill, D.W.; Vingreen, J.L. Alcohol after Resistance Exercise Does not Affect Muscle Power Recovery. J. Strength Cond. Res. 2018. [Google Scholar] [CrossRef]
- Kalinowski, A.; Humphreys, K. Governmental standard drink definitions and low-risk alcohol consumption guidelines in 37 countries. Addiction 2016, 111, 1293–1298. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.W. Excretion of alcohol in urine and diuresis in healthy men in relation to their age, the dose administered and the time after drinking. Forensic Sci. Int. 1990, 45, 217–224. [Google Scholar] [CrossRef]
- Brunner, S.; Herber, R.; Drobesch, C.; Peters, A.; Massberg, S.; Kaab, S.; Sinner, M.F. Alcohol consumption, sinus tachycardia, and cardiac arrhythmias at the Munich Octoberfest: Results from the Munich Beer Related Electrocardiogram Workup Study (MunichBREW). Eur. Heart J. 2017, 38, 2100–2106. [Google Scholar] [CrossRef]
- Borg, G.; Domserius, M.; Kaijser, L. Effect of alcohol on perceived exertion in relation to heart rate and blood lactate. Eur. J. Appl. Physiol. Occup. Physiol. 1990, 60, 382–384. [Google Scholar] [CrossRef] [PubMed]
- Lustig, R.H. Fructose: It’s “alcohol without the buzz”. Adv. Nutr. 2013, 4, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Kelley, K.W.; Dantzer, R. Alcoholism and inflammation: Neuroimmunology of behavioral and mood disorders. Brain Behav. Immun. 2011, 25, S13–S20. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.J.; Zakhari, S.; Jung, M.K. Alcohol, inflammation, and gut-liver-brain interactions in tissue damage and disease development. World J. Gastroenterol. 2010, 16, 1304–1313. [Google Scholar] [CrossRef] [PubMed]
- Hoogeveen, A.R.; Zonderland, M.L. Relationships between testosterone, cortisol and performance in professional cyclists. Int. J. Sports Med. 1996, 17, 423–428. [Google Scholar] [CrossRef] [PubMed]
- Tataranni, P.A.; Larson, D.E.; Snitker, S.; Young, J.B.; Flatt, J.P.; Ravussin, E. Effects of glucocorticoids on energy metabolism and food intake in humans. Am. J. Physiol. 1996, 271, E317–E325. [Google Scholar] [CrossRef] [PubMed]
- Phillips, S.M.; Van Loon, L.J. Dietary protein for athletes: From requirements to optimum adaptation. J. Sports Sci. 2011, 29, S29–S38. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, B.B.; Tipton, K.D.; Miller, S.L.; Wolf, S.E.; Wolfe, R.R. An oral essential amino acid-carbohydrate supplement enhances muscle protein anabolism after resistance exercise. J. Appl. Physiol. 2000, 88, 386–392. [Google Scholar] [CrossRef]
- Kraemer, W.J.; Volek, J.S.; Bush, J.A.; Putukian, M.; Sebastianelli, W.J. Hormonal responses to consecutive days of heavy-resistance exercise with or without nutritional supplementation. J. Appl. Physiol. 1998, 85, 1544–1555. [Google Scholar] [CrossRef]
- Wilkinson, S.B.; Phillips, S.M.; Atherton, P.J.; Patel, R.; Yarasheski, K.E.; Tarnopolsky, M.A.; Rennie, M.J. Differential effects of resistance and endurance exercise in the fed state on signalling molecule phosphorylation and protein synthesis in human muscle. J. Physiol. 2008, 586, 3701–3717. [Google Scholar] [CrossRef]
- Gibbs, M.E.; Ng, K.T. Psychobiology of memory: Towards a model of memory formation. Biobehav. Rev. 1977, 1, 113–136. [Google Scholar] [CrossRef]
- Davis, H.P.; Spanis, C.W.; Squire, L.R. Inhibition of cerebral protein synthesis: Performance at different times after passive avoidance training. Pharmacol. Biochem. Behav. 1976, 4, 13–16. [Google Scholar] [CrossRef]
- Hernandez, P.J.; Abel, T. The role of protein synthesis in memory consolidation: Progress amid decades of debate. Neurobiol. Learn. Mem. 2008, 89, 293–311. [Google Scholar] [CrossRef] [PubMed]
- Faubert, J. Professional athletes have extraordinary skills for rapidly learning complex and neutral dynamic visual scenes. Sci. Rep. 2013, 3, 1154. [Google Scholar] [CrossRef] [PubMed]
| Author [Ref] (Year) | n | Aim | Alcohol (dose) | Mix | Administration Time | Measures | Resistance Training | Comparator | Outcomes (Compared to Comparator) |
|---|---|---|---|---|---|---|---|---|---|
| Barnes et al. [27] (2010) | 12 | Evaluate if ALC interacts with damaged muscles. | 1g/kg | 37.5% ALC/volume; Smirnoff Vodka in orange juice (ratio 3.2:1) | A beverage was consumed every 15 min over a total time of 90 min. | -Strength. -Peak and averaged torque. | 300 maximal eccentric contractions of the quadriceps muscles of one lower limb at an angular speed of 30°/s. | Cross-Over | No differences in acute performance measures. Decreased performance was seen after 36h following ingestion. |
| Barnes et al. [18] (2010) | 11 | To compare the effects of post-exercise ALC ingestion with that of an isocaloricnon-ALC beverage on changes in muscle performance. | 1g/kg | 37.5% ALC/volume; Smirnoff Vodka in orange juice (ratio 3.2:1). | A beverage was consumed every 15 min over a total time of 90 min. | -Soreness -Peak and averaged torque -CK | 300 maximal eccentric contractions of the quadriceps muscles of one lower limb at an angular speed of 30°/s. | Cross-Over | Peak concentric, eccentric were lower in the ALC group. No differences in CK and soreness |
| Barnes et al. [19] (2012) | 10 | To investigate the effects of post-game ALC consumption on whole-body, sport-specific performance. | 1g/kg | 37.5% ALC/volume; Smirnoff Vodka in orange juice (ratio 3.2:1). | A beverage was consumed every 15 min over a total time of 90 min. | -HR -Lactate -RPE -CMJ -HPO -CK -Cortisol and Testosterone -Leukocytes | BURST Protocol (intense 20-m shuttle run with 180° turns) | Cross-Over | HR and Lactate showed no difference. RPE varied significantly. Differences in CMJ but not in HPO were present. No differences in the leukocyte count. CK was higher in the ALC group only after 48h. Testosterone did not show any differences. Cortisol was higher in the ALC group after 36h. |
| Clarkson et al. [20] (1990) | 10 | Assess the effect of acute ALC ingestion on muscle indicators. | 0.8g/kg | Vodka 40% with orange juice (ratio 1:1) | Single dose. | -CK -Soreness -Isometric strength | 50 repetitions at a lat-pulley. | Cross-Over. | No difference in CK. No difference in soreness. No difference in strength. |
| Haugvad et al. [21] (2014) | 9 | Investigate the effects of ethanol on recovery of muscle function after RT. | -Low dose 0.6 or 0.7 g/kg -High dose 1.2 or 1.4 g/kg | 40% ethanol/volume, Absolut vodka diluted with 200-mL sugar-free lemonade(raspberry flavour) and water to a total of 1.5 L | The beverage was consumed in about 90 min. | -MVC -Power -Cortisol and Testosterone -SHBG -CK -Leukocytes. | Squats, lower limb presses, and bilateral knee extensions were performed in 4 sets with a load of 8RM with 2 min rest. | Cross-Over | MVC was decreased after the ALC trial 12h training. MVC normalized in both groups after 24h. No difference in Jump performance; Cortisol was higher at 12 at 24h in the high dose group. Neither testosterone or SHBG were influenced by ALC. Free testosterone was lower in the high dose group at 12 and 24h. No differences in the CK for any group. No differences in leukocytes. |
| Levitt et al. [22] (2017) | 13 | The effect of acute ALC consumption on muscular recovery process. | 1.09 g/kg | The ALC was diluted to 15% v/v in an artificially sweetened beverage. | The beverage volume was split into 10 equal portions; one portion was administered each minute over a 10min ingestion period. | -TNF-α -Il-1β -Il-6 -Il-8 -Il-10 -Soreness -Isometric, concentric and eccentric torque -CK | 300 maximal single-lower limb eccentric leg extensions. | Cross-Over | No difference in soreness. No difference in strength. No difference in CK. No difference in any cytokine. |
| Levitt et al. [28] (2018) | 10 | To investigate the effect of ALC consumed after heavy eccentric resistance exercise on measures of muscle power. | 1.09 g/kg | Smirnoff 40% ALC Vodka diluted to 15% v/v in an artificially sweetened beverage. | The beverage was split into 10 equal portions and one portion consumed every 3 min during the 30-min beverage ingestion period. | -Soreness -Peak power -Peak force -Jump height | 4 sets of 10 repetitions at 110% of concentric 1RM; 3 min passive rest in between sets | Cross-Over | No differences were found in peak power nor peak force or jump height. No differences were found in soreness measures. |
| McLeay et al. [23] (2017) | 8 | To investigate the effects of ALC consumption on recovery of muscle force. | 0.88 g/kg | 37.5% ALC/volume; Smirnoff Vodka in orange juice | Six drinks were consumed every 15 min over 1.5 hr. | -CK -Soreness -Isometric, concentric and eccentric torque. | 300 maximal single-lower limb eccentric leg extensions through a 60° ROM at an angular speed of 30°/s. | Cross-Over | No difference in isometric, concentric and eccentric torque. No difference in CK. No difference in muscle soreness. |
| Murphy et al. [24] (2013) | 9 | To evaluate the effects of ALC ingestion on lower-body strength and power and physiological and cognitive recovery | 1g/kg | 37.5% ALC/volume; Smirnoff Vodka in orange juice (ratio 3.2:1). | An equal volume of beverage was consumed every 20 min over a total time of 150 min | -RPE -CMJ -MVC -Urine -CK -CRP - Cortisol and Testosterone -Cognitive function | Rugby match | Cross-Over | No difference in RPE. No differences in CMJ and MVC. No difference in CK and CRP. No difference in testosterone. Large effect size for cortisol increase after 16h in the ALC group. Larger urine volume after night in the ALC group. Decreased cognitive function was observed in the ALC group. |
| Parr et al. [9] (2014) | 8 | Evaluate the effect of ALC intake on rates of myofibrillar protein synthesis following strenuous exercise | -1.5g/kg with CHO -1.5g/kg with PRO | Vodka and Orange juice (ratio 1:4) | 6 equal volumes were consumed during a 3 h period. | -Biopsy -Blood glucose -Plasma AA concentration -Intracellular signalling proteins | -8 × 5 at ,80% of 1RM -10 × 30 s high intensity intervals at 110% of PPO; 3 min rest between sets | Cross-Over | Blood ALC was higher in the CHO compared to the PRO group after 6 and 8h after consumption. Blood Glucose was higher in the ALC-CHO group after 5h.AA (EEA and BCAA) were lower in the ALC groups compared to the no ALC group.mTOR phosphorylation was higher in the no ALC group at 2 and 8h post exercise. p70S6Kphosphorylation was higher in the no ALC and the ALC-PRO group at 8h post exercise. Muscle protein synthesis was greater in the No ALC group than the ALC-PRO, which was greater than the ALC-CHO group. |
| Poulsen et al. [25] (2007) | 19 | Evaluate acute ALC intoxication on skeletal muscle function | 1.5 g/L | ALC 96% with orange juice (ratio 1:4) | 5 doses with intervals of 1h each. | -CK -Ca2+ -Strength -Endurance | MVC Isokinetic endurance and isometric knee extensors (30 extensions at a velocity of 180°/s) | Cross-Over | No differences in strength and endurance. No differences in CK. Small reduction in Ca2+ only in the ALC group. |
| Vingren et al. [26] (2013) | 8 | To examine the testosterone bioavailability and the anabolic endocrine milieu in response to acute ethanol ingestion | 1.09 g/kg | ALC was diluted to a concentration of 19% v/v absolute ethanol in an artificially sweetened and calorie-free beverage | The participants drank 1/10 of the drink each minute during a 10-min ingestion period. | -HR -RPE -Testosterone -SHBG -Lactate -Cortisol -Estradiol | 6 × 10squats starting at 80% of 1 RM and 2 min of rest between sets. | Cross-Over | No difference in HR, RPE and lactate. Serum testosterone and free testosterone was higher for ALC at 300min post exercise. FAI was higher in the ALC group. No difference in cortisol levels. No differences in estradiol. |
| Tot. 127 | Mean 1.1g/kg |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lakićević, N. The Effects of Alcohol Consumption on Recovery Following Resistance Exercise: A Systematic Review. J. Funct. Morphol. Kinesiol. 2019, 4, 41. https://doi.org/10.3390/jfmk4030041
Lakićević N. The Effects of Alcohol Consumption on Recovery Following Resistance Exercise: A Systematic Review. Journal of Functional Morphology and Kinesiology. 2019; 4(3):41. https://doi.org/10.3390/jfmk4030041
Chicago/Turabian StyleLakićević, Nemanja. 2019. "The Effects of Alcohol Consumption on Recovery Following Resistance Exercise: A Systematic Review" Journal of Functional Morphology and Kinesiology 4, no. 3: 41. https://doi.org/10.3390/jfmk4030041
APA StyleLakićević, N. (2019). The Effects of Alcohol Consumption on Recovery Following Resistance Exercise: A Systematic Review. Journal of Functional Morphology and Kinesiology, 4(3), 41. https://doi.org/10.3390/jfmk4030041




