TomoPress—In Situ Synchrotron-Based Microtomography under Axial Load
Abstract
1. Introduction
2. Method
3. Applications
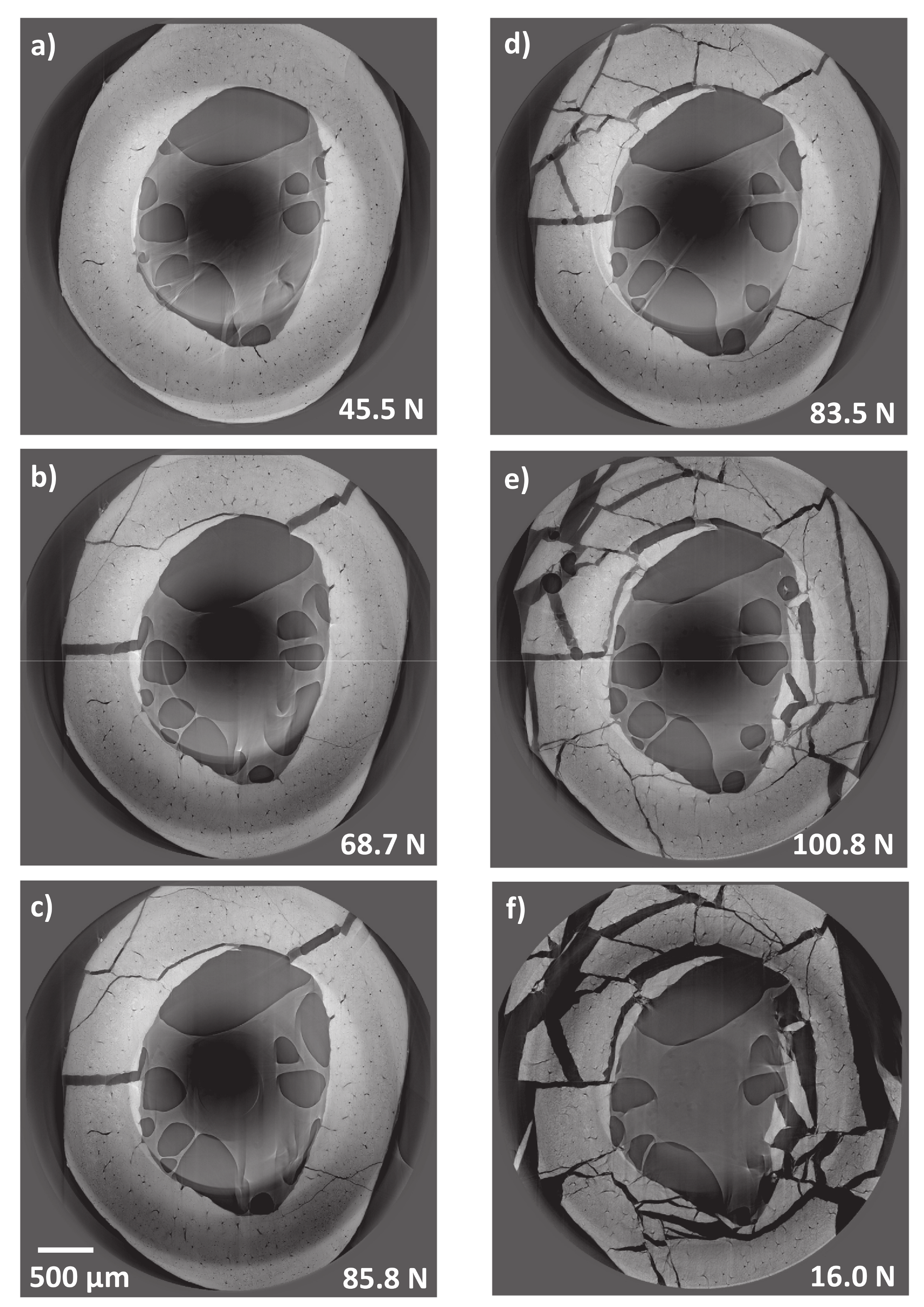
3.1. Micromechanical Response of Bio-Regenerated Rat Bone
3.2. Crack Propagation: The Case of Wood
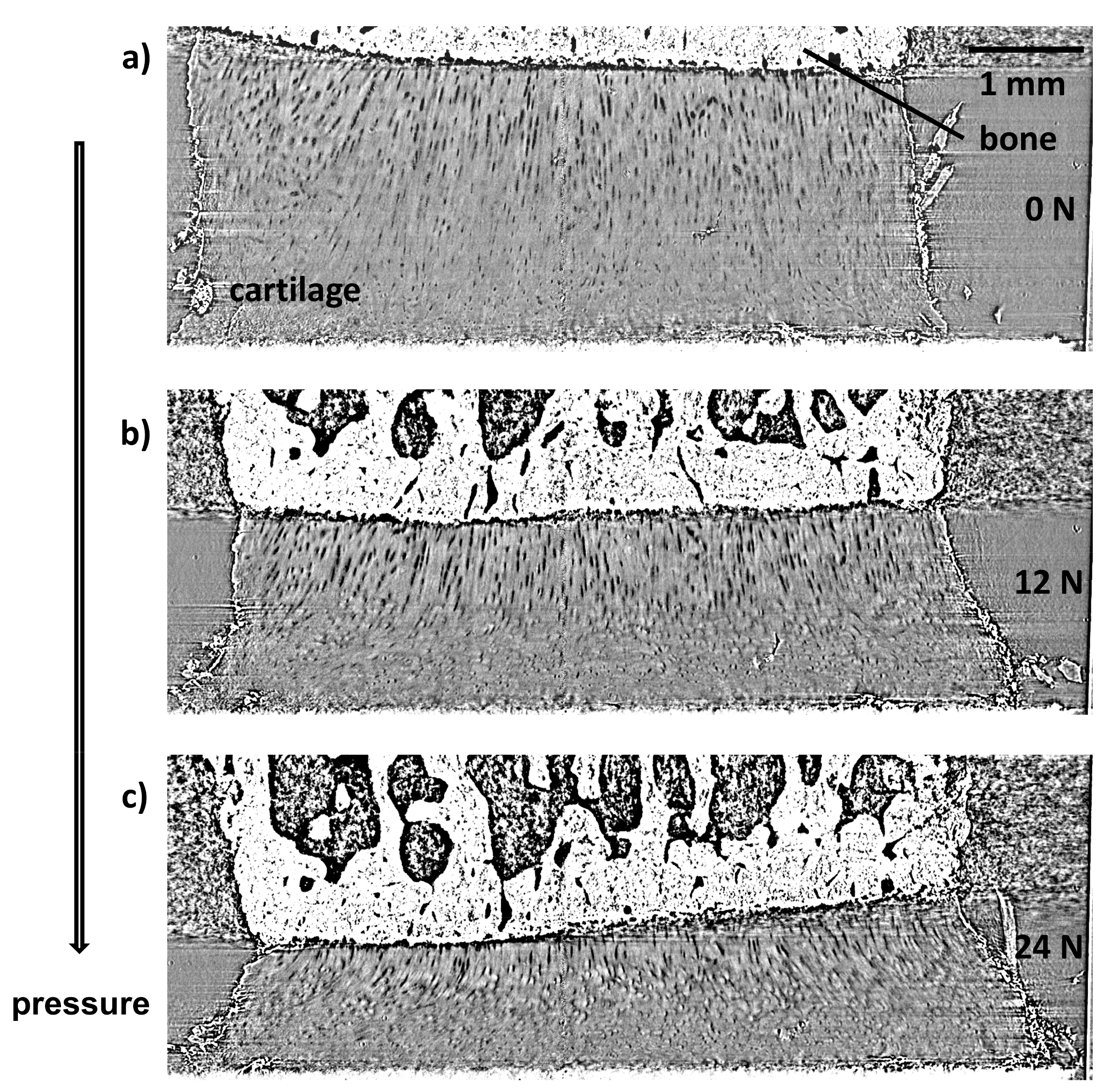
3.3. Microstructure of Cartilage under Load
4. Summary
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wiedemann, H. Synchrotron Radiation; Springer: Berlin, Germany, 2002. [Google Scholar] [CrossRef]
- Olbinado, M.P.; Just, X.; Gelet, J.L.; Lhuissier, P.; Scheel, M.; Vagovic, P.; Sato, T.; Graceffa, R.; Schulz, J.; Manusco, A.; et al. MHz frame rate hard X-ray phase-contrast imaging using synchrotron radiation. Opt. Express 2017, 25, 13857–13871. [Google Scholar] [CrossRef]
- Cloetens, P.; Barrett, R.; Baruchel, J.; Guigay, J.P.; Schlenker, M. Phase objects in synchrotron radiation hard X-ray imaging. J. Phys. D 1996, 29, 133–146. [Google Scholar] [CrossRef]
- Bonse, U.; Busch, F. X-ray computed microtomography (μCT) using synchrotron radiation (SR). Prog. Biophys. Mol. Biol. 1996, 65, 133–169. [Google Scholar] [CrossRef]
- MacDowell, A.A.; Parkinson, D.Y.; Haboub, A.; Schaible, E.; Nasiatka, J.R.; Yee, C.A.; Jameson, J.R.; Ajo-Franklin, J.B.; Brodersen, C.R.; McElrone, A.J. X-ray micro-tomography at the Advanced Light Source. In Developments in X-ray Tomography VIII; Stock, S.R., Ed.; SPIE Press: Bellingham, WA, USA, 2012; Volume 8506, p. 850618. [Google Scholar] [CrossRef]
- Wang, Y.; De Carlo, F.; Mancini, D.C.; McNulty, I.; Tieman, B.; Bresnahan, J.; Foster, I.; Insley, J.; Lange, P.; Laszewski, G.; et al. A high-throughput X-ray microtomography system at the Advanced Photon Source. Rev. Sci. Instrum. 2001, 72, 2062–2068. [Google Scholar] [CrossRef]
- Rack, A.; Weitkamp, T.; Bauer Trabelsi, S.; Modregger, P.; Cecilia, A.; dos Santos Rolo, T.; Rack, T.; Haas, D.; Simon, R.; Heldele, R.; et al. The micro-imaging station of the TopoTomo beamline at the ANKA synchrotron light source. Nucl. Instrum. Meth. B 2009, 267, 1978–1988. [Google Scholar] [CrossRef]
- Hausermann, D.; Hall, C.; Maksimenko, A.; Campbell, C. The Imaging and Medical Beam Line at the Australian Synchrotron. AIP Conf. Proc. 2010, 1266, 3–9. [Google Scholar] [CrossRef]
- Rack, A.; Zabler, S.; Müller, B.R.; Riesemeier, H.; Weidemann, G.; Lange, A.; Goebbels, J.; Hentschel, M.; Görner, W. High resolution synchrotron-based radiography and tomography using hard X-rays at the BAMline (BESSY II). Nucl. Instrum. Method A 2008, 586, 327–344. [Google Scholar] [CrossRef]
- Rau, C.; Wagner, U.; Pesic, Z.; De Fanis, A. Coherent imaging at the Diamond beamline I13. Phys. Status Solidi A 2011, 208, 2522–2525. [Google Scholar] [CrossRef]
- Drakopoulos, M.; Connolley, T.; Reinhard, C.; Atwood, R.; Magdysyuk, O.; Vo, N.; Hart, M.; Connor, L.; Humphreys, B.; Howell, G.; et al. I12: The Joint Engineering, Environment and Processing (JEEP) beamline at Diamond Light Source. J. Synchrotron Radiat. 2015, 22, 828–838. [Google Scholar] [CrossRef]
- Tromba, G.; Longo, R.; Abrami, A.; Arfelli, F.; Astolfo, A.; Bregant, P.; Brun, F.; Casarin, K.; Chenda, V.; Dreossi, D.; et al. The SYRMEP Beamline of Elettra: Clinical Mammography and Biomedical Applications. AIP Conf. Proc. 2010, 1266, 18–23. [Google Scholar] [CrossRef]
- Agrawal, A.K.; Singh, B.; Kashyap, Y.S.; Shukla, M.; Sarkar, P.S.; Sinha, A. Design, development and first experiments on the X-ray imaging beamline at Indus-2 synchrotron source RRCAT, India. J. Synchrotron Radiat. 2015, 22, 1531–1539. [Google Scholar] [CrossRef] [PubMed]
- Haibel, A.; Beckmann, F.; Dose, T.; Herzen, J.; Ogurreck, M.; Müller, M.; Schreyer, A. Latest developments in microtomography and nanotomography at PETRA III. Powder Differ. 2010, 25, 161–164. [Google Scholar] [CrossRef][Green Version]
- Stampanoni, M.; Groso, A.; Isenegger, A.; Mikuljan, G.; Chen, Q.; Meister, D.; Lange, M.; Betemps, R.; Henein, S.; Abela, R. TOMCAT: A beamline for Tomographic Microscopy and Coherent radiology experiments. AIP Conf. Proc. 2007, 879, 848–851. [Google Scholar] [CrossRef]
- Weitkamp, T.; Scheel, M.; Giorgetta, J.; Joyet, V.; Roux, V.L.; Cauchon, G.; Moreno, T.; Polack, F.; Thompson, A.; Samama, J. The tomography beamline ANATOMIX at Synchrotron SOLEIL. J. Phys. Conf. Ser. 2017, 849, 012037. [Google Scholar] [CrossRef]
- King, A.; Guignot, N.; Zerbino, P.; Boulard, E.; Desjardins, K.; Bordessoule, M.; Leclerq, N.; Le, S.; Renaud, G.; Cerato, M.; et al. Tomography and imaging at the PSICHE beam line of the SOLEIL synchrotron. Rev. Sci. Instrum. 2016, 87, 093704. [Google Scholar] [CrossRef]
- Uesugi, K.; Hoshino, M.; Takeuchi, A.; Suzuki, Y.; Yagi, N.; Nakano, T. Development of fast (sub-minute) micro-tomography. AIP Conf. Proc. 2010, 1266, 47–50. [Google Scholar] [CrossRef]
- Einfeld, D.; Hasnain, S.; Sayers, Z.; Schopper, H.; Winick, H. SESAME, a third generation synchrotron light source for the Middle East region. Radiat. Phys. Chem. 2004, 71, 693–700. [Google Scholar] [CrossRef][Green Version]
- Rack, A.; Tafforeau, P.; Riesemeier, H.; Stiller, M.; Dalügge, O.; Rack, T.; Stiller, F.; Knabe, C. 3.4 Developments in High-Resolution CT: Studying Bioregeneration by Hard X-ray Synchrotron-Based Microtomography. In Comprehensive Biomaterials II; Ducheyne, P., Ed.; Elsevier: Oxford, UK, 2017; pp. 58–77. [Google Scholar] [CrossRef]
- McCarthy, J. ESRF: A Quest for Excellence in Service to Users. Synchrotron Radiat. News 2017, 30, 31–36. [Google Scholar] [CrossRef]
- Banhart, J. (Ed.) Advanced Tomographic Methods in Materials Research and Engineering; Oxford University Press: Oxford, UK, 2008. [Google Scholar] [CrossRef]
- Stock, S.R. MicroComputed Tomography: Methodology and Applications, 2nd ed.; CRC Press: Boca Raton, FL, USA; Taylor and Francis Group: London, UK; New York, NY, USA, 2019. [Google Scholar] [CrossRef]
- Stock, S. Recent advances in X-ray microtomography applied to materials. Int. Mater. Rev. 2008, 53, 129–181. [Google Scholar] [CrossRef]
- Maire, E.; Withers, P. Quantitative X-ray tomography. Int. Mater. Rev. 2014, 59, 1–43. [Google Scholar] [CrossRef]
- Wiest, W.; Zabler, S.; Rack, A.; Fella, C.; Balles, A.; Nelson, K.; Schmelzeisen, R.; Hanke, R. In situ microradioscopy and microtomography of fatigue-loaded dental two-piece implants. J. Synchrotron Radiat. 2015, 22, 1492–1497. [Google Scholar] [CrossRef] [PubMed]
- Maire, E.; Le Bourlot, C.; Adrien, J.; Mortensen, A.; Mokso, R. 20 Hz X-ray tomography during an in situ tensile test. Int. J. Fract. 2016, 200, 3–12. [Google Scholar] [CrossRef]
- Renard, F.; Cordonnier, B.; Dysthe, D.K.; Boller, E.; Tafforeau, P.; Rack, A. A deformation rig for synchrotron microtomography studies of geomaterials under conditions down to 10 km depth in the Earth. J. Synchrotron Radiat. 2016, 23, 1030–1034. [Google Scholar] [CrossRef] [PubMed]
- Voltolini, M.; Barnard, H.; Creux, P.; Ajo-Franklin, J. A new mini-triaxial cell for combined high-pressure and high-temperature in situ synchrotron X-ray microtomography experiments up to 400 °C and 24 MPa. J. Synchrotron Radiat. 2019, 26, 238–243. [Google Scholar] [CrossRef]
- Bleuet, P.; Roux, J.P.; Dabin, Y.; Boivin, G. In situ microtomography study of human bones under strain with synchrotron radiation. In Developments in X-ray Tomography IV; Bonse, U., Ed.; SPIE Press: Bellingham, WA, USA, 2004; Volume 5535, pp. 129–136. [Google Scholar] [CrossRef]
- Weitkamp, T.; Raven, C.; Snigirev, A.A. Imaging and microtomography facility at the ESRF beamline ID 22. In Developments in X-ray Tomography II; International Society for Optics and Photonics; Bonse, U., Ed.; SPIE Press: Bellingham, WA, USA, 1999; Volume 3772, pp. 311–317. [Google Scholar] [CrossRef]
- Martínez-Criado, G.; Tucoulou, R.; Cloetens, P.; Bleuet, P.; Bohic, S.; Cauzid, J.; Kieffer, I.; Kosior, E.; Labouré, S.; Petitgirard, S.; et al. Status of the hard X-ray microprobe beamline ID22 of the European Synchrotron Radiation Facility. J. Synchrotron Radiat. 2012, 19, 10–18. [Google Scholar] [CrossRef]
- Weitkamp, T.; Tafforeau, P.; Boller, E.; Cloetens, P.; Valade, J.P.; Bernard, P.; Peyrin, F.; Ludwig, W.; Helfen, L.; Baruchel, J. Status and evolution of the ESRF beamline ID19. In AIP Conference Proceedings; Denecke, M., Walker, C.T., Eds.; American Institute of Physics: College Park, MD, USA, 2010; Volume 1221, pp. 33–38. [Google Scholar] [CrossRef]
- Cloetens, P.; Guigay, J.P.; De Martino, C.; Baruchel, J.; Schlenker, M. Fractional Talbot imaging of phase gratings with hard x rays. Opt. Lett. 1997, 22, 1059–1061. [Google Scholar] [CrossRef]
- Rack, A.; Weitkamp, T.; Assoufid, L.; Rack, T.; Zanette, I.; Morawe, C.; Kluender, R.; David, C. Protocol to study wavefront preservation capabilities of reflective X-ray optics with coherent synchrotron light. Nucl. Instrum. Method A 2013, 710, 101–105. [Google Scholar] [CrossRef]
- Douissard, P.A.; Cecilia, A.; Rochet, X.; Chapel, X.; Martin, T.; van de Kamp, T.; Helfen, L.; Baumbach, T.; Luquot, L.; Xiao, X.; et al. A versatile indirect detector design for hard X-ray microimaging. J. Instrum. 2012, 7, P09016. [Google Scholar] [CrossRef]
- Paganin, D.; Mayo, S.C.; Gureyev, T.E.; Miller, P.R.; Wilkins, S.W. Simultaneous phase and amplitude extraction from a single defocused image of a homogeneous object. J. Microsc. Oxf. 2002, 206, 33–40. [Google Scholar] [CrossRef]
- Weitkamp, T.; Haas, D.; Wegrzynek, D.; Rack, A. ANKAphase: Software for single-distance phase retrieval from inline X-ray phase-contrast radiographs. J. Synchrotron Radiat. 2011, 18, 617–629. [Google Scholar] [CrossRef]
- Erko, M.; Younes-Metzler, O.; Rack, A.; Zaslansky, P.; Young, S.L.; Milliron, G.; Chyasnavichyus, M.; Barth, F.G.; Fratzl, P.; Tsukruk, V.; et al. Micro- and nano-structural details of a spider’s filter for substrate vibrations: Relevance for low-frequency signal transmission. J. R. Soc. Interface 2015, 12, 20141111. [Google Scholar] [CrossRef] [PubMed]
- Charles-Harris, M.; del Valle, S.; Hentges, E.; Bleuet, P.; Lacroix, D.; Planell, J.A. Mechanical and structural characterisation of completely degradable polylactic acid/calcium phosphate glass scaffolds. Biomaterials 2007, 28, 4429–4438. [Google Scholar] [CrossRef] [PubMed]
- Bemmann, M.; Schulz-Kornas, E.; Hammel, J.U.; Hipp, A.; Moosmann, J.; Herrel, A.; Rack, A.; Radespiel, U.; Zimmermann, E.; Kaiser, T.M.; et al. 3D movement analysis of primate molar teeth under load using synchrotron X-ray microtomography. J. R. Soc. Interface 2020, submitted. [Google Scholar]
- Knabe, C.; Koch, C.; Rack, A.; Stiller, M. Effect of β-tricalcium phosphate particles with varying porosity on osteogenesis after sinus floor augmentation in humans. Biomaterials 2008, 29, 2249–2258. [Google Scholar] [CrossRef] [PubMed]
- Knabe, C.; Ducheyne, P. Cellular Response to Bioactive Ceramics. In Bioceramics and Their Clinical Applications; Kokubo, T., Ed.; Woodhead Publishing Inc.: Cambridge, UK, 2008; pp. 133–164. [Google Scholar] [CrossRef]
- Adel-Khattab, D.; Giacomini, F.; Peleska, B.; Gildenhaar, F.; Berger, G.; Gomes, C.; Linow, U.; Hardt, M.; Günster, J.; Stiller, M.; et al. Development of a synthetic tissue engineered 3D printed bioceramic-based bone graft with homogenously distributed osteoblasts and mineralizing bone matrix in vitro. J. Tissue Eng. Regen. Med. 2018, 12, 44–58. [Google Scholar] [CrossRef]
- Zhai, X.; Gao, J.; Nie, Y.; Guo, Z.; Kedir, N.; Claus, B.; Sun, T.; Fezzaa, K.; Xiao, X.; Chen, W.W. Real-time visualization of dynamic fractures in porcine bones and the loading-rate effect on their fracture toughness. J. Mech. Phys. Sol. 2019, 131, 358–371. [Google Scholar] [CrossRef]
- Labiche, J.C.; Mathon, O.; Pascarelli, S.; Newton, M.A.; Ferre, G.G.; Curfs, C.; Vaughan, G.; Homs, A.; Carreiras, D.F. The fast readout low noise camera as a versatile X-ray detector for time resolved dispersive extended X-ray absorption fine structure and diffraction studies of dynamic problems in materials science, chemistry, and catalysis. Rev. Sci. Instrum. 2007, 78, 0901301. [Google Scholar] [CrossRef]
- Mirone, A.; Brun, E.; Gouillart, E.; Tafforeau, P.; Kieffer, J. The PyHST2 hybrid distributed code for high speed tomographic reconstruction with iterative reconstruction and a priori knowledge capabilities. Nucl. Instrum. Method Phys. Res. B 2014, 324, 41–48. [Google Scholar] [CrossRef]
- Nieuwenhove, V.V.; Beenhouwer, J.D.; Carlo, F.D.; Mancini, L.; Marone, F.; Sijbers, J. Dynamic intensity normalization using eigen flat fields in X-ray imaging. Opt. Express 2015, 23, 27975–27989. [Google Scholar] [CrossRef]
- Smith, I.; Landis, E.; Gong, M. Fracture and Fatigue in Wood; John Wiley & Sons Ltd.: Chichester, UK; Hoboken, NJ, USA, 2003. [Google Scholar]
- Zabler, S.; Paris, O.; Burgert, I.; Fratzl, P. Moisture changes in the plant cell wall force cellulose crystallites to deform. J. Struct. Biol. 2010, 171, 133–141. [Google Scholar] [CrossRef]
- Issever, A.S.; Diederichs, G.; Majumdar, S.; Rogalla, P.; Hamm, B.K.; Lange, A.; Harwardt, M.; Hentschel, M.P.; Mueller, B.R. Analyser-based tomography images of cartilage. J. Synchrotron Radiat. 2008, 15, 525–527. [Google Scholar] [CrossRef] [PubMed]
- Stroebel, J. A Multi-Scale Imaging Approach to Understand Osteoarthritis Development. Ph.D. Thesis, Ludwig-Maximilians-Universität München, Munich, Germany, 2020. [Google Scholar]
- Mittone, A.; Manakov, I.; Broche, L.; Jarnias, C.; Coan, P.; Bravin, A. Characterization of a sCMOS-based high-resolution imaging system. J. Synchrotron Radiat. 2017, 24, 1226–1236. [Google Scholar] [CrossRef] [PubMed]



| Specification | |
|---|---|
| dimensions L × W × H [mm] | 700 × 230 × 300 |
| minimum propagation distance [mm] | 20 |
| displacement sensor accuracy [µm] | 1 |
| eccentricity tomographic axis [µm] | 1 |
| force accuracy [N] | 0.5 |
| force range [N] | 0 to 500 |
| maximum sample diameter (utilised) [mm] | 8 × 8 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rack, A.; Stroebel, J.; Rack, T.; Dabin, Y.; Knabe, C.; Stiller, M.; Coan, P.; Bleuet, P. TomoPress—In Situ Synchrotron-Based Microtomography under Axial Load. Instruments 2020, 4, 11. https://doi.org/10.3390/instruments4020011
Rack A, Stroebel J, Rack T, Dabin Y, Knabe C, Stiller M, Coan P, Bleuet P. TomoPress—In Situ Synchrotron-Based Microtomography under Axial Load. Instruments. 2020; 4(2):11. https://doi.org/10.3390/instruments4020011
Chicago/Turabian StyleRack, Alexander, Johannes Stroebel, Tatjana Rack, Yves Dabin, Christine Knabe, Michael Stiller, Paola Coan, and Pierre Bleuet. 2020. "TomoPress—In Situ Synchrotron-Based Microtomography under Axial Load" Instruments 4, no. 2: 11. https://doi.org/10.3390/instruments4020011
APA StyleRack, A., Stroebel, J., Rack, T., Dabin, Y., Knabe, C., Stiller, M., Coan, P., & Bleuet, P. (2020). TomoPress—In Situ Synchrotron-Based Microtomography under Axial Load. Instruments, 4(2), 11. https://doi.org/10.3390/instruments4020011






