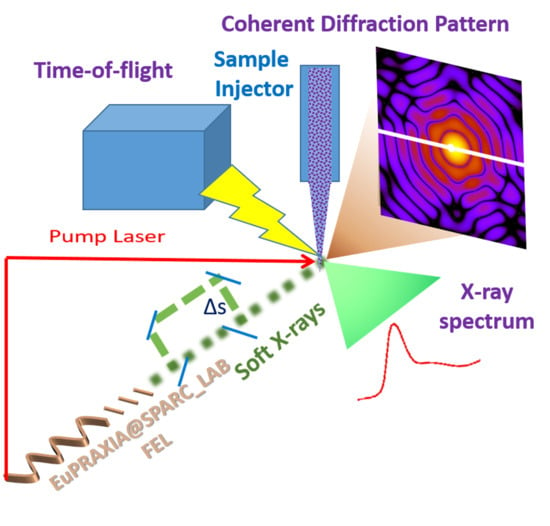
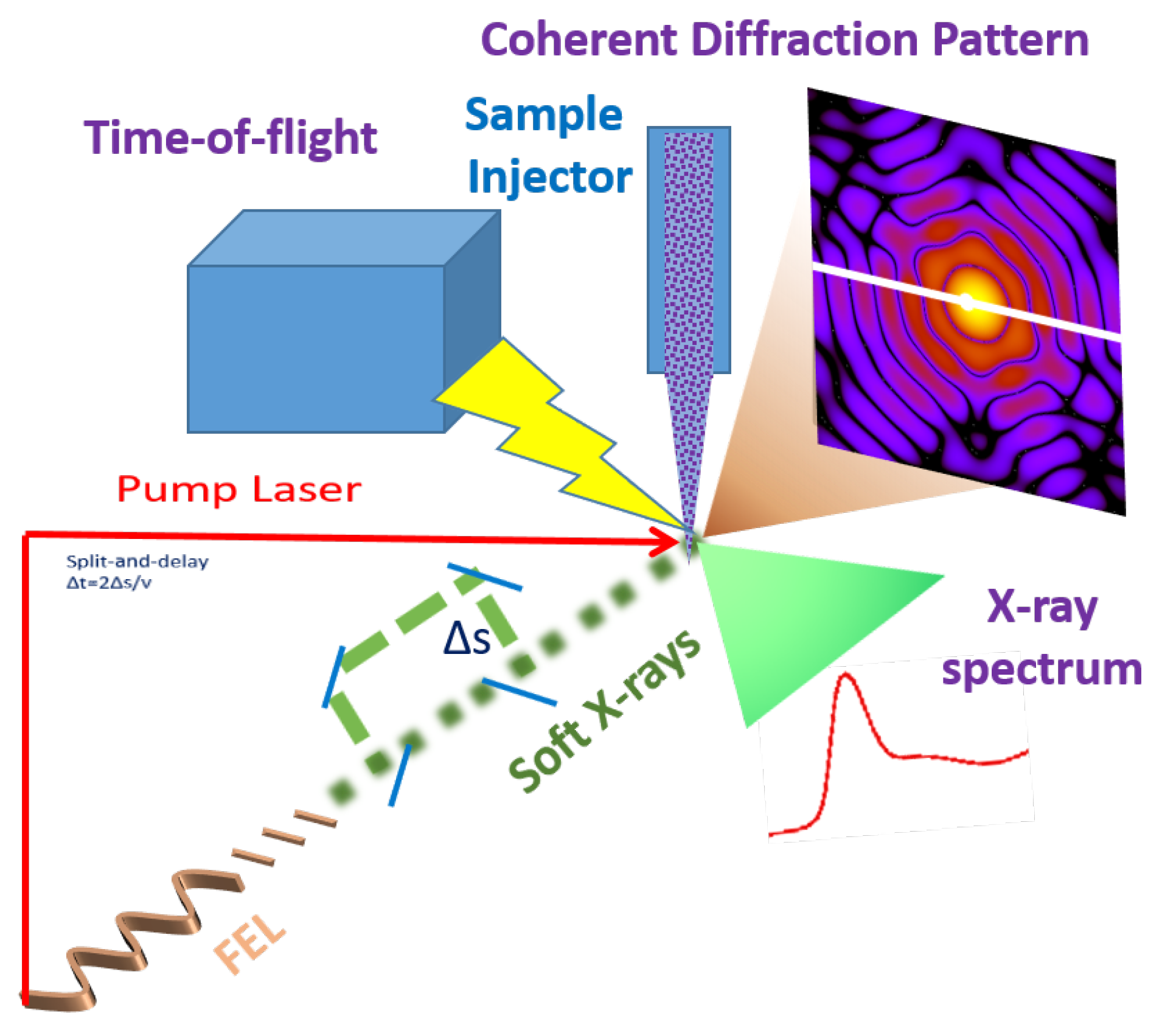
The Potential of EuPRAXIA@SPARC_LAB for Radiation Based Techniques
Abstract
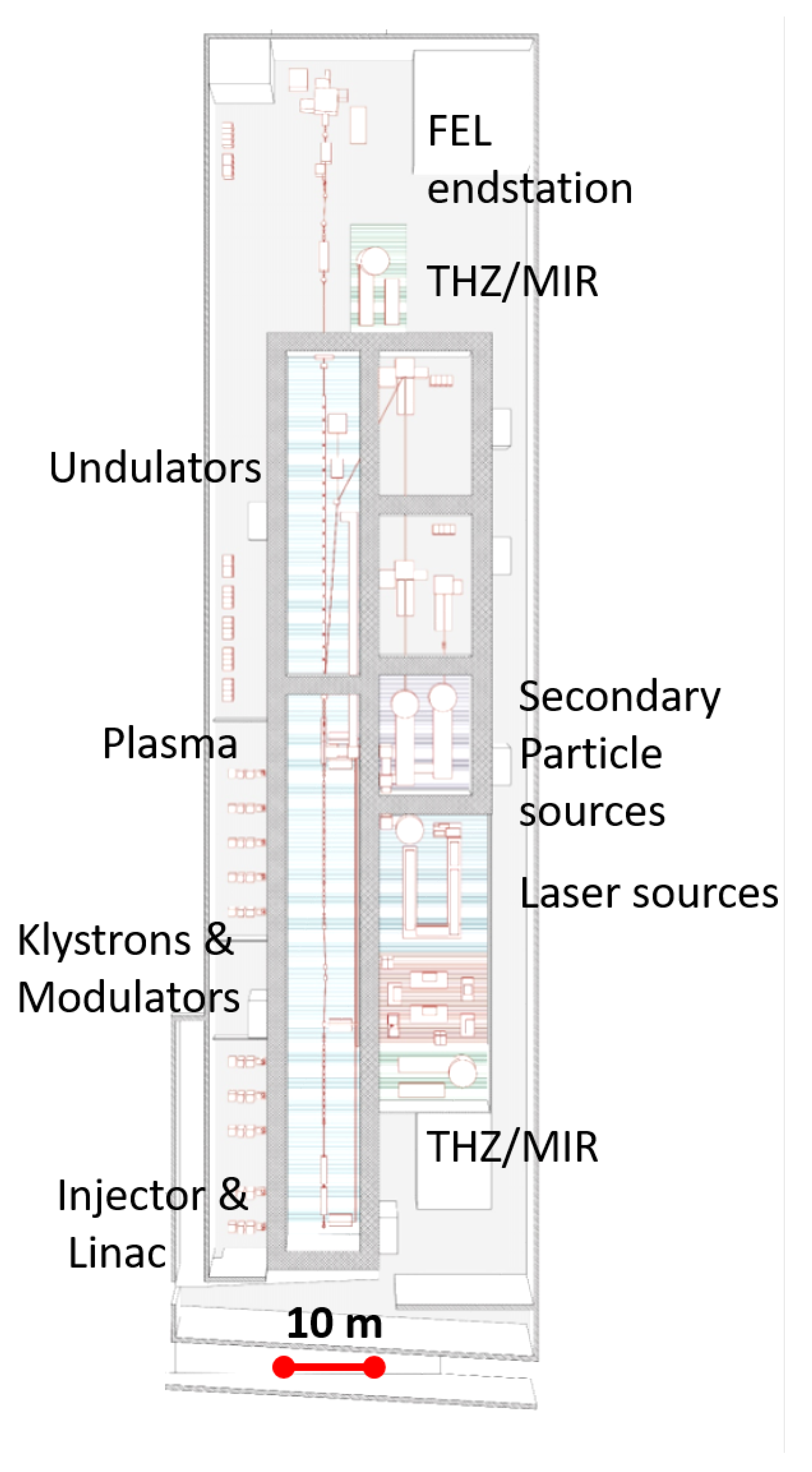
1. Introduction
2. Expected Results
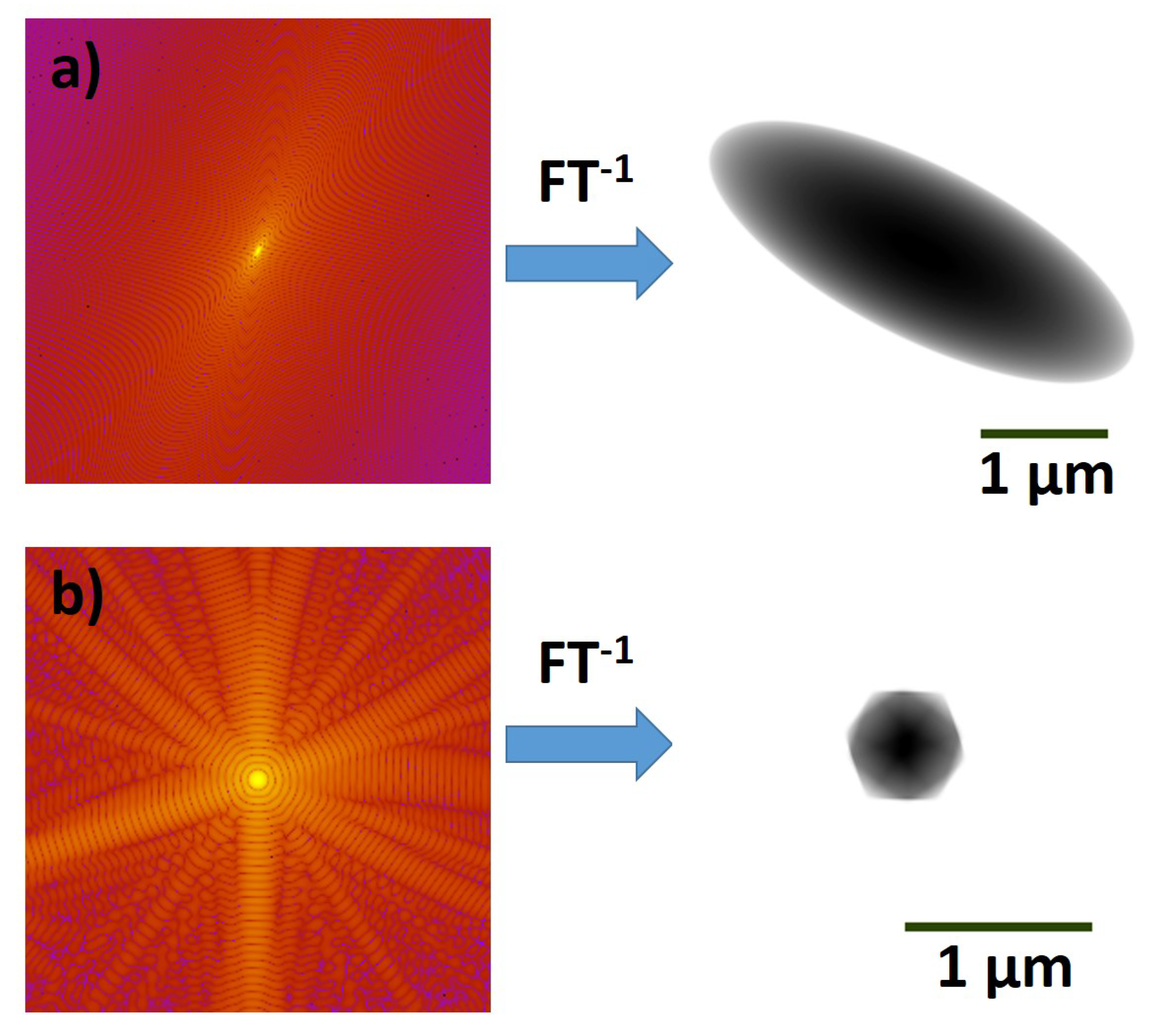
2.1. Coherent Imaging of Biological Samples
2.2. Time-Resolved X-ray Absorption Spectroscopy in the Water Window
2.3. Time-Resolved Coherent Raman Experiments with X-ray Pulses
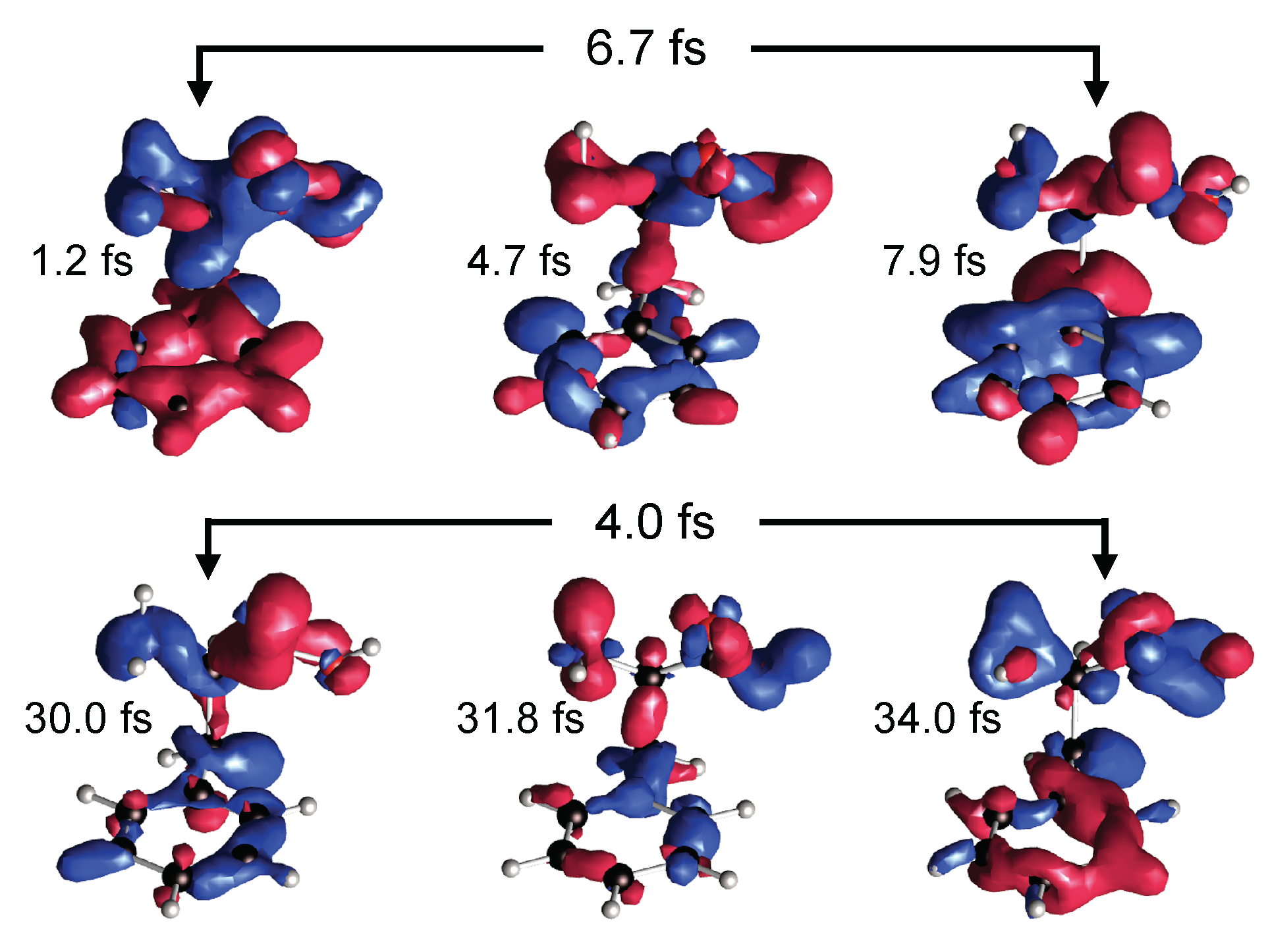
2.4. Photo-Fragmentation of Molecules
2.5. Resonant Inelastic X-ray Scattering
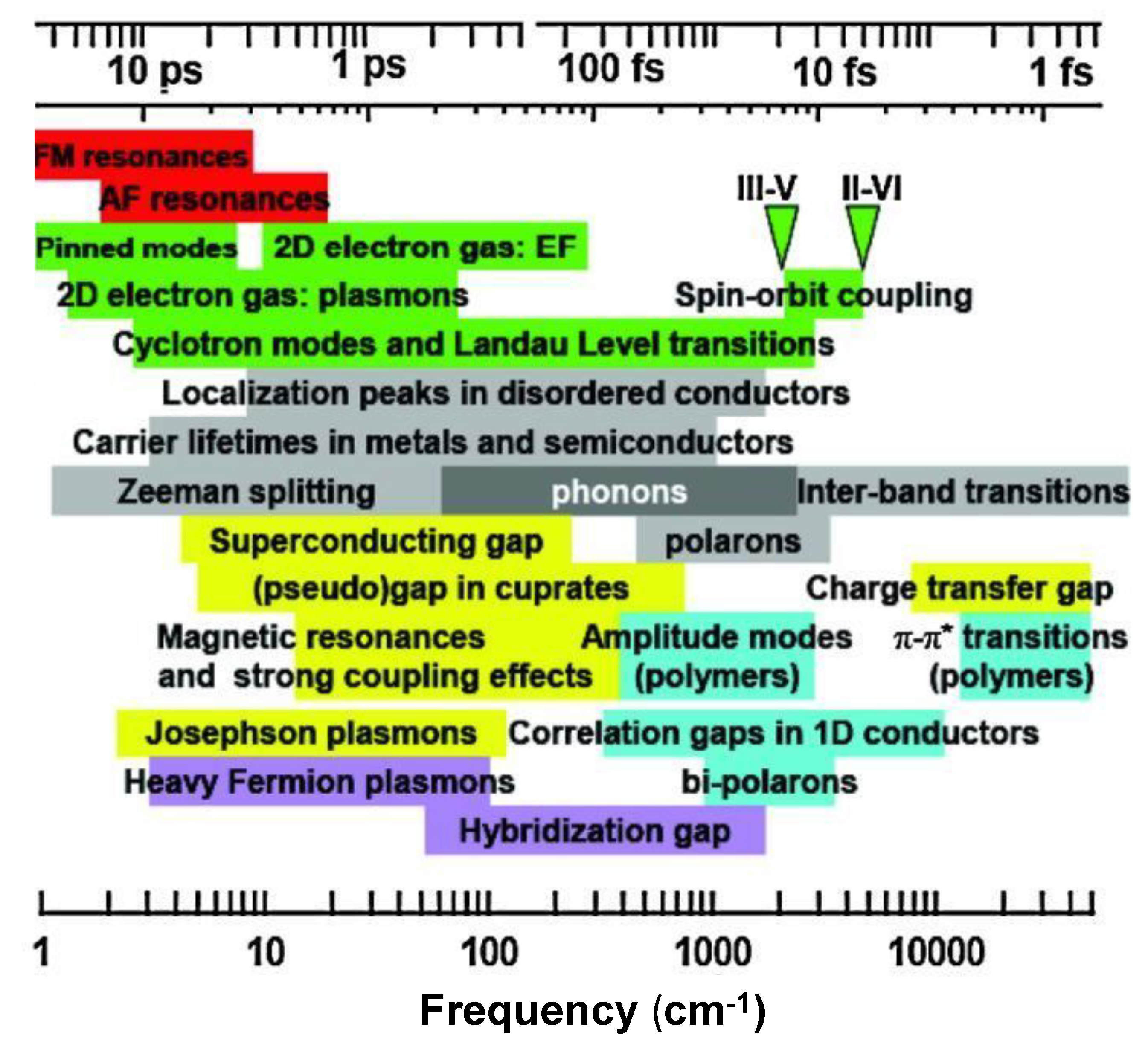
2.6. THz/MIR Sources
3. Discussion and Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AP | Actinic Pulse |
| CDI | Coherent Diffraction Imaging |
| CDR | Coherent Diffraction Radiation |
| CTR | Coherent Transition Radiation |
| CI | Conical Intersection |
| EDS | Energy Dispersive Setup |
| FEL | Free Electron Laser |
| FSRS | Femtosecond stimulated Raman scattering |
| MIR | Middle-InfraRed |
| PP | Probe Pulse |
| RIXS | Resonant Inelastic X-ray Scattering |
| RP | Raman Pulse |
| SASE | Self Amplification of SpontanEous radiation |
| SNOM | Scanning Near-field Optical Microscopy |
| THz | Tera Hertz |
| XAS | X-ray Absortpion Spectroscopy |
| X-RIVS | X-ray Impulsive Vibrational Scattering |
| XUV | eXtreme Ultra Violet |
References
- Chapman, H.N.; Fromme, P.; Barty, A.; White, T.A.; Kirian, R.A.; Aquila, A.; Hunter, M.S.; Schulz, J.; DePonte, D.P.; Weierstall, U.; et al. Femtosecond X-ray protein nanocrystallography. Nature 2011, 470, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Boutet, S.; Lomb, L.; Williams, G.J.; Barends, T.R.; Aquila, A.; Doak, R.B.; Weierstall, U.; DePonte, D.P.; Steinbrener, J.; Shoeman, R.L.; et al. High-resolution protein structure determination by serial femtosecond crystallography. Science 2012, 337, 362–364. [Google Scholar] [CrossRef] [PubMed]
- Seibert, M.M.; Ekeberg, T.; Maia, F.R.; Svenda, M.; Andreasson, J.; Jönsson, O.; Odić, D.; Iwan, B.; Rocker, A.; Westphal, D.; et al. Single mimivirus particles intercepted and imaged with an X-ray laser. Nature 2011, 470, 78–81. [Google Scholar] [CrossRef] [PubMed]
- Hantke, M.F.; Hasse, D.; Maia, F.R.; Ekeberg, T.; John, K.; Svenda, M.; Loh, N.D.; Martin, A.V.; Timneanu, N.; Larsson, D.S.; et al. High-throughput imaging of heterogeneous cell organelles with an X-ray laser. Nat. Photonics 2014, 8, 943–949. [Google Scholar] [CrossRef]
- Van Der Schot, G.; Svenda, M.; Maia, F.R.; Hantke, M.; DePonte, D.P.; Seibert, M.M.; Aquila, A.; Schulz, J.; Kirian, R.; Liang, M.; et al. Imaging single cells in a beam of live cyanobacteria with an X-ray laser. Nat. Commun. 2015, 6, 5704. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; Sun, Z.; Wang, Y.; Park, J.; Kim, S.; Gallagher-Jones, M.; Kim, Y.; Song, C.; Yao, S.; Zhang, J.; et al. Single-pulse enhanced coherent diffraction imaging of bacteria with an X-ray free-electron laser. Sci. Rep. 2016, 6, 34008. [Google Scholar] [CrossRef] [PubMed]
- Henderson, R. The potential and limitations of neutrons, electrons and X-rays for atomic resolution microscopy of unstained biological molecules. Q. Rev. Biophys. 1995, 28, 171–193. [Google Scholar] [CrossRef] [PubMed]
- Gutt, C.; Streit-Nierobisch, S.; Stadler, L.M.; Pfau, B.; Günther, C.; Könnecke, R.; Frömter, R.; Kobs, A.; Stickler, D.; Oepen, H.; et al. Single-pulse resonant magnetic scattering using a soft X-ray free-electron laser. Phys. Rev. B 2010, 81, 100401. [Google Scholar] [CrossRef]
- Ferrario, M.; Alesini, D.; Anania, M.; Artioli, M.; Bacci, A.; Bartocci, S.; Bedogni, R.; Bellaveglia, M.; Biagioni, A.; Bisesto, F.; et al. EuPRAXIA@ SPARC_LAB Design study towards a compact FEL facility at LNF. Nuclear Instrum. Methods Phys. Res. Sect. A 2018, 909, 134–138. [Google Scholar] [CrossRef]
- Villa, F.; Cianchi, A.; Coreno, M.; Dabagov, S.; Marcelli, A.; Minicozzi, V.; Morante, S.; Stellato, F. Design study of a photon beamline for a soft X-ray FEL driven by high gradient acceleration at EuPRAXIA@ SPARC_LAB. Nuclear Instrum. Methods Phys. Res. Sect. A 2018, 909, 294–297. [Google Scholar] [CrossRef]
- Oberthür, D. Biological single-particle imaging using XFELs–towards the next resolution revolution. IUCrJ 2018, 5, 663. [Google Scholar] [CrossRef] [PubMed]
- Reddy, H.K.; Yoon, C.H.; Aquila, A.; Awel, S.; Ayyer, K.; Barty, A.; Berntsen, P.; Bielecki, J.; Bobkov, S.; Bucher, M.; et al. Coherent soft X-ray diffraction imaging of coliphage PR772 at the Linac coherent light source. Sci. Data 2017, 4, 170079. [Google Scholar] [CrossRef] [PubMed]
- Rose, M.; Bobkov, S.; Ayyer, K.; Kurta, R.P.; Dzhigaev, D.; Kim, Y.Y.; Morgan, A.J.; Yoon, C.H.; Westphal, D.; Bielecki, J.; et al. Single-particle imaging without symmetry constraints at an X-ray free-electron laser. IUCrJ 2018, 5, 727–736. [Google Scholar] [CrossRef] [PubMed]
- Munke, A.; Andreasson, J.; Aquila, A.; Awel, S.; Ayyer, K.; Barty, A.; Bean, R.J.; Berntsen, P.; Bielecki, J.; Boutet, S.; et al. Coherent diffraction of single Rice Dwarf virus particles using hard X-rays at the Linac Coherent Light Source. Sci. Data 2016, 3, 160064. [Google Scholar] [CrossRef] [PubMed]
- Takayama, Y.; Inui, Y.; Sekiguchi, Y.; Kobayashi, A.; Oroguchi, T.; Yamamoto, M.; Matsunaga, S.; Nakasako, M. Coherent X-ray diffraction imaging of chloroplasts from Cyanidioschyzon merolae by using X-ray free electron laser. Plant Cell Physiol. 2015, 56, 1272–1286. [Google Scholar] [CrossRef] [PubMed]
- Van Der Schot, G.; Svenda, M.; Maia, F.R.; Hantke, M.F.; DePonte, D.P.; Seibert, M.M.; Aquila, A.; Schulz, J.; Kirian, R.A.; Liang, M.; et al. Open data set of live cyanobacterial cells imaged using an X-ray laser. Sci. Data 2016, 3, 160058. [Google Scholar] [CrossRef] [PubMed]
- Hantke, M.F.; Hasse, D.; Ekeberg, T.; John, K.; Svenda, M.; Loh, D.; Martin, A.V.; Timneanu, N.; Larsson, D.S.; Van Der Schot, G.; et al. A data set from flash X-ray imaging of carboxysomes. Sci. Data 2016, 3, 160061. [Google Scholar] [CrossRef] [PubMed]
- Ackermann, W.a.; Asova, G.; Ayvazyan, V.; Azima, A.; Baboi, N.; Bähr, J.; Balandin, V.; Beutner, B.; Brandt, A.; Bolzmann, A.; et al. Operation of a free-electron laser from the extreme ultraviolet to the water window. Nat. Photonics 2007, 1, 336. [Google Scholar] [CrossRef]
- Popp, D.; Loh, N.D.; Zorgati, H.; Ghoshdastider, U.; Liow, L.T.; Ivanova, M.I.; Larsson, M.; DePonte, D.P.; Bean, R.; Beyerlein, K.R.; et al. Flow-aligned, single-shot fiber diffraction using a femtosecond X-ray free-electron laser. Cytoskeleton 2017, 74, 472–481. [Google Scholar] [CrossRef] [PubMed]
- Stellato, F.; Fusco, Z.; Chiaraluce, R.; Consalvi, V.; Dinarelli, S.; Placidi, E.; Petrosino, M.; Rossi, G.; Minicozzi, V.; Morante, S. The effect of β-sheet breaker peptides on metal associated Amyloid-β peptide aggregation process. Biophys. Chem. 2017, 229, 110–114. [Google Scholar] [CrossRef] [PubMed]
- Carbonaro, M.; Di Venere, A.; Filabozzi, A.; Maselli, P.; Minicozzi, V.; Morante, S.; Nicolai, E.; Nucara, A.; Placidi, E.; Stellato, F. Role of dietary antioxidant (-)-epicatechin in the development of β-lactoglobulin fibrils. Biochim. Biophys. Acta 2016, 1864, 766–772. [Google Scholar] [CrossRef] [PubMed]
- Carbonaro, M.; Ripanti, F.; Filabozzi, A.; Minicozzi, V.; Stellato, F.; Placidi, E.; Morante, S.; Di Venere, A.; Nicolai, E.; Postorino, P.; et al. Human insulin fibrillogenesis in the presence of epigallocatechin gallate and melatonin: Structural insights from a biophysical approach. Int. J. Biol. Macromol. 2018, 115, 1157–1164. [Google Scholar] [CrossRef] [PubMed]
- Ekeberg, T.; Svenda, M.; Abergel, C.; Maia, F.R.; Seltzer, V.; Claverie, J.M.; Hantke, M.; Jönsson, O.; Nettelblad, C.; Van Der Schot, G.; et al. Three-dimensional reconstruction of the giant mimivirus particle with an X-ray free-electron laser. Phys. Rev. Lett. 2015, 114, 098102. [Google Scholar] [CrossRef] [PubMed]
- Hantke, M.F.; Ekeberg, T.; Maia, F.R. Condor: A simulation tool for flash X-ray imaging. J. Appl. Crystallogr. 2016, 49, 1356–1362. [Google Scholar] [CrossRef] [PubMed]
- Pertot, Y.; Schmidt, C.; Matthews, M.; Chauvet, A.; Huppert, M.; Svoboda, V.; von Conta, A.; Tehlar, A.; Baykusheva, D.; Wolf, J.P.; et al. Time-resolved X-ray absorption spectroscopy with a water window high-harmonic source. Science 2017, 355, 264–267. [Google Scholar] [CrossRef] [PubMed]
- Popmintchev, D.; Galloway, B.R.; Chen, M.C.; Dollar, F.; Mancuso, C.A.; Hankla, A.; Miaja-Avila, L.; O’Neil, G.; Shaw, J.M.; Fan, G.; et al. Near-and extended-edge X-ray-absorption fine-structure spectroscopy using ultrafast coherent high-order harmonic supercontinua. Phys. Rev. Lett. 2018, 120, 093002. [Google Scholar] [CrossRef] [PubMed]
- Chergui, M.; Zewail, A.H. Electron and X-ray Methods of Ultrafast Structural Dynamics: Advances and Applications. ChemPhysChem 2009, 10, 28–43. [Google Scholar] [CrossRef] [PubMed]
- Bressler, C.; Chergui, M. Molecular structural dynamics probed by ultrafast X-ray absorption spectroscopy. Annu. Rev. Phys. Chem. 2010, 61, 263–282. [Google Scholar] [CrossRef] [PubMed]
- Benfatto, M.; Della Longa, S.; Natoli, C. The MXAN procedure: A new method for analysing the XANES spectra of metalloproteins to obtain structural quantitative information. J. Synchrotron Radiat. 2003, 10, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Bunau, O.; Joly, Y. Self-consistent aspects of X-ray absorption calculations. J. Phys. Condens. Matter 2009, 21, 345501. [Google Scholar] [CrossRef] [PubMed]
- Rehr, J.J.; Kas, J.J.; Vila, F.D.; Prange, M.P.; Jorissen, K. Parameter-free calculations of X-ray spectra with FEFF9. Phys. Chem. Chem. Phys. 2010, 12, 5503–5513. [Google Scholar] [CrossRef] [PubMed]
- La Penna, G.; Minicozzi, V.; Morante, S.; Rossi, G.; Stellato, F. A first-principle calculation of the XANES spectrum of Cu2+ in water. J. Chem. Phys. 2015, 143, 124508. [Google Scholar] [CrossRef] [PubMed]
- Stellato, F.; Calandra, M.; D’Acapito, F.; De Santis, E.; La Penna, G.; Rossi, G.; Morante, S. Multi-scale theoretical approach to X-ray absorption spectra in disordered systems: An application to the study of Zn (II) in water. Phys. Chem. Chem. Phys. 2018, 20, 24775–24782. [Google Scholar] [CrossRef] [PubMed]
- Kubin, M.; Kern, J.; Gul, S.; Kroll, T.; Chatterjee, R.; Löchel, H.; Fuller, F.D.; Sierra, R.G.; Quevedo, W.; Weniger, C.; et al. Soft X-ray absorption spectroscopy of metalloproteins and high-valent metal-complexes at room temperature using free-electron lasers. Struct. Dyn. 2017, 4, 054307. [Google Scholar] [CrossRef] [PubMed]
- De Santis, E.; Minicozzi, V.; Proux, O.; Rossi, G.; Silva, K.I.; Lawless, M.J.; Stellato, F.; Saxena, S.; Morante, S. Cu (II)–Zn (II) Cross-Modulation in Amyloid–Beta Peptide Binding: An X-ray Absorption Spectroscopy Study. J. Phys. Chem. B 2015, 119, 15813–15820. [Google Scholar] [CrossRef] [PubMed]
- De Santis, E.; Shardlow, E.; Stellato, F.; Proux, O.; Rossi, G.; Exley, C.; Morante, S. X-ray Absorption Spectroscopy Measurements of Cu-ProIAPP Complexes at Physiological Concentrations. Condens. Matter 2019, 4, 13. [Google Scholar] [CrossRef]
- Nagler, B.; Zastrau, U.; Fäustlin, R.R.; Vinko, S.M.; Whitcher, T.; Nelson, A.; Sobierajski, R.; Krzywinski, J.; Chalupsky, J.; Abreu, E.; et al. Turning solid aluminium transparent by intense soft X-ray photoionization. Nat. Phys. 2009, 5, 693. [Google Scholar]
- Di Cicco, A.; Hatada, K.; Giangrisostomi, E.; Gunnella, R.; Bencivenga, F.; Principi, E.; Masciovecchio, C.; Filipponi, A. Interplay of electron heating and saturable absorption in ultrafast extreme ultraviolet transmission of condensed matter. Phys. Rev. B 2014, 90, 220303. [Google Scholar] [CrossRef]
- Principi, E.; Giangrisostomi, E.; Cucini, R.; Bencivenga, F.; Battistoni, A.; Gessini, A.; Mincigrucci, R.; Saito, M.; Di Fonzo, S.; D’Amico, F.; et al. Free electron laser-driven ultrafast rearrangement of the electronic structure in Ti. Struct. Dyn. 2016, 3, 023604. [Google Scholar] [CrossRef] [PubMed]
- Hatada, K.; Di Cicco, A. Modeling Non-Equilibrium Dynamics and Saturable Absorption Induced by Free Electron Laser Radiation. Appl. Sci. 2017, 7, 814. [Google Scholar] [CrossRef]
- Zewail, A.H. Femtochemistry. Past, present, and future. Pure Appl. Chem. 2000, 72, 2219–2231. [Google Scholar] [CrossRef]
- Kruglik, S.G.; Jasaitis, A.; Hola, K.; Yamashita, T.; Liebl, U.; Martin, J.L.; Vos, M.H. Subpicosecond oxygen trapping in the heme pocket of the oxygen sensor FixL observed by time-resolved resonance Raman spectroscopy. Proc. Natl. Acad. Sci. USA 2007, 104, 7408–7413. [Google Scholar] [CrossRef] [PubMed]
- Batignani, G.; Pontecorvo, E.; Ferrante, C.; Aschi, M.; Elles, C.G.; Scopigno, T. Visualizing Excited-State Dynamics of a Diaryl Thiophene: Femtosecond Stimulated Raman Scattering as a Probe of Conjugated Molecules. J. Phys. Chem. Lett. 2016, 7, 2981–2988. [Google Scholar] [CrossRef] [PubMed]
- Batignani, G.; Bossini, D.; Di Palo, N.; Ferrante, C.; Pontecorvo, E.; Cerullo, G.; Kimel, A.; Scopigno, T. Probing ultrafast photo-induced dynamics of the exchange energy in a Heisenberg antiferromagnet. Nat. Photonics 2015, 9, 506. [Google Scholar] [CrossRef]
- Dietze, D.R.; Mathies, R.A. Femtosecond stimulated Raman spectroscopy. ChemPhysChem 2016, 17, 1224–1251. [Google Scholar] [CrossRef] [PubMed]
- Ferrante, C.; Pontecorvo, E.; Cerullo, G.; Vos, M.; Scopigno, T. Direct observation of subpicosecond vibrational dynamics in photoexcited myoglobin. Nat. Chem. 2016, 8, 1137. [Google Scholar] [CrossRef] [PubMed]
- McCamant, D.W.; Kukura, P.; Yoon, S.; Mathies, R.A. Femtosecond broadband stimulated Raman spectroscopy: Apparatus and methods. Rev. Sci. Instrum. 2004, 75, 4971–4980. [Google Scholar] [CrossRef] [PubMed]
- Yoshizawa, M.; Hattori, Y.; Kobayashi, T. Femtosecond time-resolved resonance Raman gain spectroscopy in polydiacetylene. Phys. Rev. B 1994, 49, 13259. [Google Scholar] [CrossRef]
- Ferrante, C.; Batignani, G.; Fumero, G.; Pontecorvo, E.; Virga, A.; Montemiglio, L.; Cerullo, G.; Vos, M.; Scopigno, T. Resonant broadband stimulated Raman scattering in myoglobin. J. Raman Spectrosc. 2018, 49, 913–920. [Google Scholar] [CrossRef]
- Fumero, G.; Batignani, G.; Dorfman, K.E.; Mukamel, S.; Scopigno, T. On the Resolution Limit of Femtosecond Stimulated Raman Spectroscopy: Modelling Fifth-Order Signals with Overlapping Pulses. ChemPhysChem 2015, 16, 3438–3443. [Google Scholar] [CrossRef] [PubMed]
- Kukura, P.; McCamant, D.W.; Mathies, R.A. Femtosecond stimulated Raman spectroscopy. Annu. Rev. Phys. Chem. 2007, 58, 461–488. [Google Scholar] [CrossRef] [PubMed]
- Batignani, G.; Fumero, G.; Pontecorvo, E.; Ferrante, C.; Mukamel, S.; Scopigno, T. Genuine dynamics vs. cross phase modulation artefacts in Femtosecond Stimulated Raman Spectroscopy. ACS Photonics 2019, 6, 492–500. [Google Scholar] [CrossRef]
- Pontecorvo, E.; Kapetanaki, S.; Badioli, M.; Brida, D.; Marangoni, M.; Cerullo, G.; Scopigno, T. Femtosecond stimulated Raman spectrometer in the 320–520 nm range. Opt. Express 2011, 19, 1107–1112. [Google Scholar] [CrossRef] [PubMed]
- Monacelli, L.; Batignani, G.; Fumero, G.; Ferrante, C.; Mukamel, S.; Scopigno, T. Manipulating impulsive stimulated raman spectroscopy with a chirped probe pulse. J. Phys. Chem. Lett. 2017, 8, 966–974. [Google Scholar] [CrossRef] [PubMed]
- Liebel, M.; Schnedermann, C.; Wende, T.; Kukura, P. Principles and applications of broadband impulsive vibrational spectroscopy. J. Phys. Chem. A 2015, 119, 9506–9517. [Google Scholar] [CrossRef] [PubMed]
- Kuramochi, H.; Takeuchi, S.; Yonezawa, K.; Kamikubo, H.; Kataoka, M.; Tahara, T. Probing the early stages of photoreception in photoactive yellow protein with ultrafast time-domain Raman spectroscopy. Nat. Chem. 2017, 9, 660. [Google Scholar] [CrossRef] [PubMed]
- Batignani, G.; Fumero, G.; Kandada, A.R.S.; Cerullo, G.; Gandini, M.; Ferrante, C.; Petrozza, A.; Scopigno, T. Probing femtosecond lattice displacement upon photo-carrier generation in lead halide perovskite. Nat. Commun. 2018, 9, 1971. [Google Scholar] [CrossRef] [PubMed]
- Zavalin, A.; Cricenti, A.; Generosi, R.; Luce, M.; Morgan, S.; Piston, D. Nano-Raman mapping of porous glass ceramics with a scanning near-field optical microscope in collection mode. Appl. Phys. Lett. 2006, 88, 133126. [Google Scholar] [CrossRef]
- Sonntag, M.D.; Pozzi, E.A.; Jiang, N.; Hersam, M.C.; Van Duyne, R.P. Recent advances in tip-enhanced Raman spectroscopy. J. Phys. Chem. Lett. 2014, 5, 3125–3130. [Google Scholar] [CrossRef] [PubMed]
- Kowalewski, M.; Bennett, K.; Dorfman, K.E.; Mukamel, S. Catching conical intersections in the act: Monitoring transient electronic coherences by attosecond stimulated X-ray Raman signals. Phys. Rev. Lett. 2015, 115, 193003. [Google Scholar] [CrossRef] [PubMed]
- Polli, D.; Altoè, P.; Weingart, O.; Spillane, K.M.; Manzoni, C.; Brida, D.; Tomasello, G.; Orlandi, G.; Kukura, P.; Mathies, R.A.; et al. Conical intersection dynamics of the primary photoisomerization event in vision. Nature 2010, 467, 440. [Google Scholar] [CrossRef] [PubMed]
- Calegari, F.; Ayuso, D.; Trabattoni, A.; Belshaw, L.; De Camillis, S.; Anumula, S.; Frassetto, F.; Poletto, L.; Palacios, A.; Decleva, P.; et al. Ultrafast electron dynamics in phenylalanine initiated by attosecond pulses. Science 2014, 346, 336–339. [Google Scholar] [CrossRef] [PubMed]
- Lara-Astiaso, M.; Galli, M.; Trabattoni, A.; Palacios, A.; Ayuso, D.; Frassetto, F.; Poletto, L.; De Camillis, S.; Greenwood, J.; Decleva, P.; et al. Attosecond Pump–Probe Spectroscopy of Charge Dynamics in Tryptophan. J. Phys. Chem. Lett. 2018, 9, 4570–4577. [Google Scholar] [CrossRef] [PubMed]
- Beaulieu, S.; Comby, A.; Clergerie, A.; Caillat, J.; Descamps, D.; Dudovich, N.; Fabre, B.; Géneaux, R.; Légaré, F.; Petit, S.; et al. Attosecond-resolved photoionization of chiral molecules. Science 2017, 358, 1288–1294. [Google Scholar] [CrossRef] [PubMed]
- Attar, A.R.; Bhattacherjee, A.; Pemmaraju, C.; Schnorr, K.; Closser, K.D.; Prendergast, D.; Leone, S.R. Femtosecond X-ray spectroscopy of an electrocyclic ring-opening reaction. Science 2017, 356, 54–59. [Google Scholar] [CrossRef] [PubMed]
- Marciniak, A.; Despré, V.; Barillot, T.; Rouzée, A.; Galbraith, M.; Klei, J.; Yang, C.H.; Smeenk, C.; Loriot, V.; Reddy, S.N.; et al. XUV excitation followed by ultrafast non-adiabatic relaxation in PAH molecules as a femto-astrochemistry experiment. Nat. Commun. 2015, 6, 7909. [Google Scholar] [CrossRef] [PubMed]
- Nelson, T.R.; Ondarse-Alvarez, D.; Oldani, N.; Rodriguez-Hernandez, B.; Alfonso-Hernandez, L.; Galindo, J.F.; Kleiman, V.D.; Fernandez-Alberti, S.; Roitberg, A.E.; Tretiak, S. Coherent exciton-vibrational dynamics and energy transfer in conjugated organics. Nat. Commun. 2018, 9, 2316. [Google Scholar] [CrossRef] [PubMed]
- Perfetto, E.; Sangalli, D.; Marini, A.; Stefanucci, G. Ultrafast Charge Migration in XUV Photoexcited Phenylalanine: A First-Principles Study Based on Real-Time Nonequilibrium Green’s Functions. J. Phys. Chem. Lett. 2018, 9, 1353–1358. [Google Scholar] [CrossRef] [PubMed]
- Ren, X.; Wang, E.; Skitnevskaya, A.D.; Trofimov, A.B.; Gokhberg, K.; Dorn, A. Experimental evidence for ultrafast intermolecular relaxation processes in hydrated biomolecules. Nat. Phys. 2018, 14, 1062. [Google Scholar] [CrossRef]
- Pullen, M.G.; Wolter, B.; Le, A.T.; Baudisch, M.; Hemmer, M.; Senftleben, A.; Schröter, C.D.; Ullrich, J.; Moshammer, R.; Lin, C.D.; et al. Imaging an aligned polyatomic molecule with laser-induced electron diffraction. Nat. Commun. 2015, 6, 7262. [Google Scholar] [CrossRef] [PubMed]
- Erk, B.; Boll, R.; Trippel, S.; Anielski, D.; Foucar, L.; Rudek, B.; Epp, S.W.; Coffee, R.; Carron, S.; Schorb, S.; et al. Imaging charge transfer in iodomethane upon X-ray photoabsorption. Science 2014, 345, 288–291. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zhu, X.; Wolf, T.J.; Li, Z.; Nunes, J.P.F.; Coffee, R.; Cryan, J.P.; Gühr, M.; Hegazy, K.; Heinz, T.F.; et al. Imaging CF3I conical intersection and photodissociation dynamics with ultrafast electron diffraction. Science 2018, 361, 64–67. [Google Scholar] [CrossRef] [PubMed]
- Kraus, P.M.; Zürch, M.; Cushing, S.K.; Neumark, D.M.; Leone, S.R. The ultrafast X-ray spectroscopic revolution in chemical dynamics. Nature 2018, 2, 82–94. [Google Scholar] [CrossRef]
- Kuleff, A.I.; Kryzhevoi, N.V.; Pernpointner, M.; Cederbaum, L.S. Core ionization initiates subfemtosecond charge migration in the valence shell of molecules. Phys. Rev. Lett. 2016, 117, 093002. [Google Scholar] [CrossRef] [PubMed]
- Karydas, A.; Paradellis, T. Measurement of KL and LM resonant Raman scattering cross sections with a proton-induced X-ray beam. J. Phys. B 1997, 30, 1893. [Google Scholar] [CrossRef]
- Leani, J.J.; Sánchez, H.J.; Valentinuzzi, M.; Pérez, C. Determination of the oxidation state by resonant-Raman scattering spectroscopy. J. Anal. At. Spectrom. 2011, 26, 378–382. [Google Scholar] [CrossRef]
- Robledo, J.I.; Leani, J.J.; Karydas, A.G.; Migliori, A.; Pérez, C.A.; Sánchez, H.J. Energy-Dispersive Total-Reflection Resonant Inelastic X-ray Scattering as a Tool for Elemental Speciation in Contaminated Water. Anal. Chem. 2018, 90, 3886–3891. [Google Scholar] [CrossRef] [PubMed]
- Leani, J.J.; Pérez, R.D.; Robledo, J.I.; Sánchez, H. 3D-reconstruction of chemical state distributions in stratified samples by spatially resolved micro-X-ray resonant Raman spectroscopy. J. Anal. At. Spectrom. 2017, 32, 402–407. [Google Scholar] [CrossRef]
- Leani, J.J.; Robledo, J.I.; Sánchez, H.J. Quantitative speciation of manganese oxide mixtures by RIXS/RRS spectroscopy. X-ray Spectrom. 2017, 46, 507–511. [Google Scholar] [CrossRef]
- Robledo, J.I.; Saánchez, H.J.; Leani, J.J.; Pérez, C.A. Exploratory methodology for retrieving oxidation state information from X-ray resonant Raman scattering spectrometry. Anal. Chem. 2015, 87, 3639–3645. [Google Scholar] [CrossRef] [PubMed]
- Leani, J.J.; Sánchez, H.J.; Pérez, C.A. Oxide nanolayers in stratified samples studied by X-ray resonant Raman scattering at grazing incidence. J. Spectrosc. 2015, 2015, 618279. [Google Scholar] [CrossRef]
- Sanchez, H.J.; Leani, J.J.; Pérez, C.; Pèrez, R.D. Arsenic Speciation by X-ray Spectroscopy using Resonant Raman Scattering. J. Appl. Spectrosc. 2014, 80, 912–916. [Google Scholar] [CrossRef]
- Leani, J.J.; Saánchez, H.J.; Pérez, R.D.; Peérez, C. Depth profiling nano-analysis of chemical environments using resonant Raman spectroscopy at grazing incidence conditions. Anal. Chem. 2013, 85, 7069–7075. [Google Scholar] [CrossRef] [PubMed]
- Leani, J.J.; Sánchez, H.J.; Valentinuzzi, M.C.; Pérez, C.; Grenón, M. Qualitative microanalysis of calcium local structure in tooth layers by means of micro-RRS. J. Microsc. 2013, 250, 111–115. [Google Scholar] [CrossRef] [PubMed]
- Leani, J.J.; Sánchez, H.; Valentinuzzi, M.; Pérez, C. Chemical environment determination of iron oxides using RRS spectroscopy. X-ray Spectrom. 2011, 40, 254–256. [Google Scholar] [CrossRef]
- Leani, J.J.; Robledo, J.I.; Sánchez, H.J. Energy dispersive inelastic X-ray scattering (EDIXS) spectroscopy–A review. Spectrochim. Acta Part B 2019, 154, 10–24. [Google Scholar] [CrossRef]
- Chiadroni, E.; Bacci, A.; Bellaveglia, M.; Boscolo, M.; Castellano, M.; Cultrera, L.; Di Pirro, G.; Ferrario, M.; Ficcadenti, L.; Filippetto, D.; et al. The SPARC linear accelerator based terahertz source. Appl. Phys. Lett. 2013, 102, 094101. [Google Scholar] [CrossRef]
- Giorgianni, F.; Anania, M.P.; Bellaveglia, M.; Biagioni, A.; Chiadroni, E.; Cianchi, A.; Daniele, M.; Del Franco, M.; Di Giovenale, D.; Di Pirro, G.; et al. Tailoring of highly intense thz radiation through high brightness electron beams longitudinal manipulation. Appl. Sci. 2016, 6, 56. [Google Scholar] [CrossRef]
- Castellano, M.; Verzilov, V.; Catani, L.; Cianchi, A.; Orlandi, G.; Geitz, M. Measurements of coherent diffraction radiation and its application for bunch length diagnostics in particle accelerators. Phys. Rev. E 2001, 63, 056501. [Google Scholar] [CrossRef] [PubMed]
- Chiadroni, E. Bunch Length Characterization at the TTF VUV-FEL. Ph.D. Thesis, Università degli Studi di Roma “Tor Vergata”, Roma, Italy, September 2006. [Google Scholar]
- Alesini, D.; Vicario, C.; Bertolucci, S.; Biagini, M.E.; Biscari, C.; Boni, R.; Boscolo, M.; Castellano, M.; Clozza, A.; Di Pirro, G.; et al. Technical Design Report for the SPARC Advanced Photo-Injector; Palumbo, L., Rosenzweig, J.B., Eds.; INFN: Pisa, Italy, 2004. [Google Scholar]
- Van der Geer, S.; De Loos, M.; Bongerd, D. General Particle Tracer: A 3D code for accelerator and beam line design. In Proceedings of the 5th European Particle Accelerator Conference, Stockholm, Sweden, 10–14 June 1996. [Google Scholar]
- Xie, M. Design optimization for an X-ray free electron laser driven by SLAC linac. In Proceedings of the IEEE Particle Accelerator Conference, Dallas, TX, USA, 1–5 May 1995; Volume 1, pp. 183–185. [Google Scholar]
- Giorgianni, F.; Chiadroni, E.; Rovere, A.; Cestelli-Guidi, M.; Perucchi, A.; Bellaveglia, M.; Castellano, M.; Di Giovenale, D.; Di Pirro, G.; Ferrario, M.; et al. Strong nonlinear terahertz response induced by Dirac surface states in Bi2Se3 topological insulator. Nat. Commun. 2016, 7, 11421. [Google Scholar] [CrossRef] [PubMed]
- Mitrano, M.; Cantaluppi, A.; Nicoletti, D.; Kaiser, S.; Perucchi, A.; Lupi, S.; Di Pietro, P.; Pontiroli, D.; Riccò, M.; Clark, S.R.; et al. Possible light-induced superconductivity in K3C60 at high temperature. Nature 2016, 530, 461–464. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.T.; Kersting, R.; Cho, G.C. Terahertz imaging with nanometer resolution. Appl. Phys. Lett. 2003, 83, 3009–3011. [Google Scholar] [CrossRef]

| Beam Parameters | Source Parameters | ||
|---|---|---|---|
| E (GeV) | 1 | Frequency (THz) | 0.3–10 |
| Q (pC) | 200 | P (MW) | >100 |
| (m) | 30 | E (J) | ≈100 |
| Beam Parameters | Source Parameters | ||
|---|---|---|---|
| E (MeV) | 30–50 | (m) | 10–3 |
| Q (pC) | 200 | L | 3–4.4 |
| (m) | 50 | P (MW) | 140–135 |
| I (kA) | 480 | ∼10 | |
| (%) | 0.1–0.4 | E (J) | ∼60 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Balerna, A.; Bartocci, S.; Batignani, G.; Cianchi, A.; Chiadroni, E.; Coreno, M.; Cricenti, A.; Dabagov, S.; Di Cicco, A.; Faiferri, M.; et al. The Potential of EuPRAXIA@SPARC_LAB for Radiation Based Techniques. Condens. Matter 2019, 4, 30. https://doi.org/10.3390/condmat4010030
Balerna A, Bartocci S, Batignani G, Cianchi A, Chiadroni E, Coreno M, Cricenti A, Dabagov S, Di Cicco A, Faiferri M, et al. The Potential of EuPRAXIA@SPARC_LAB for Radiation Based Techniques. Condensed Matter. 2019; 4(1):30. https://doi.org/10.3390/condmat4010030
Chicago/Turabian StyleBalerna, Antonella, Samanta Bartocci, Giovanni Batignani, Alessandro Cianchi, Enrica Chiadroni, Marcello Coreno, Antonio Cricenti, Sultan Dabagov, Andrea Di Cicco, Massimo Faiferri, and et al. 2019. "The Potential of EuPRAXIA@SPARC_LAB for Radiation Based Techniques" Condensed Matter 4, no. 1: 30. https://doi.org/10.3390/condmat4010030
APA StyleBalerna, A., Bartocci, S., Batignani, G., Cianchi, A., Chiadroni, E., Coreno, M., Cricenti, A., Dabagov, S., Di Cicco, A., Faiferri, M., Ferrante, C., Ferrario, M., Fumero, G., Giannessi, L., Gunnella, R., Leani, J. J., Lupi, S., Macis, S., Manca, R., ... Villa, F. (2019). The Potential of EuPRAXIA@SPARC_LAB for Radiation Based Techniques. Condensed Matter, 4(1), 30. https://doi.org/10.3390/condmat4010030