Enhancement of Skin Mucus Immunity, Carotenoid Content, Sexual Parameters, and Growth Response in Guppy Fish (Poecilia reticulata) Fed with Green Algae (Chaetomorpha aerea) Diets
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Fish and Their Maintanance
2.2. Collection and Preparation of Seaweed
2.3. Diet Preparation and Experimental Design
2.4. Growth Performance and Fry Production
2.5. Fish Skin Mucus Collection
2.6. Skin Mucus Immunological Parameters
2.6.1. Lysozyme Activity
2.6.2. Alternative Complement Activity
2.6.3. Myeloperoxidase Activity
2.6.4. Total Immunoglobulin
2.6.5. Total Carotenoid Concentrations
2.6.6. Evaluation of Sex Steroid Hormones
2.7. Statistical Analysis
3. Results
3.1. Growth Parameters
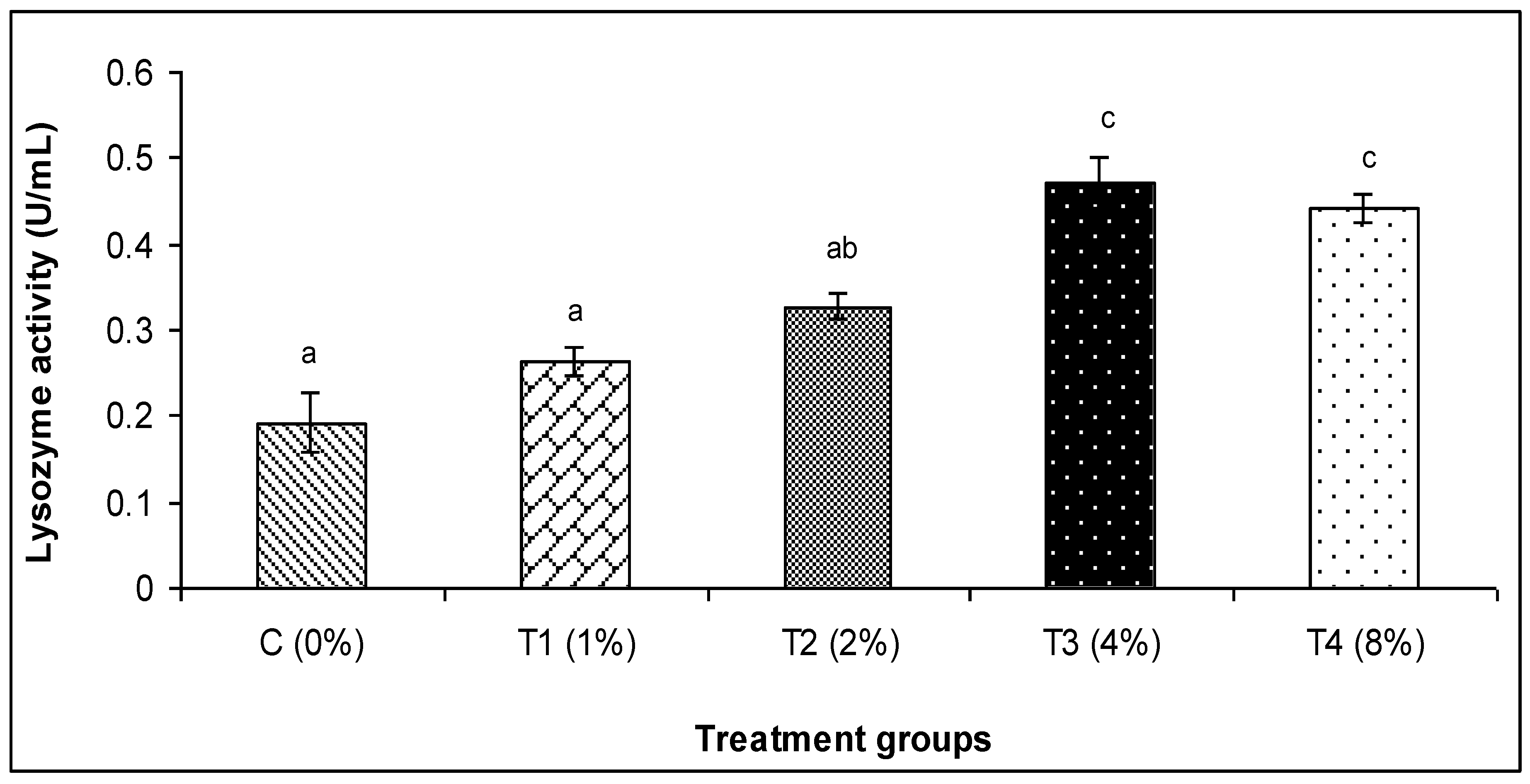
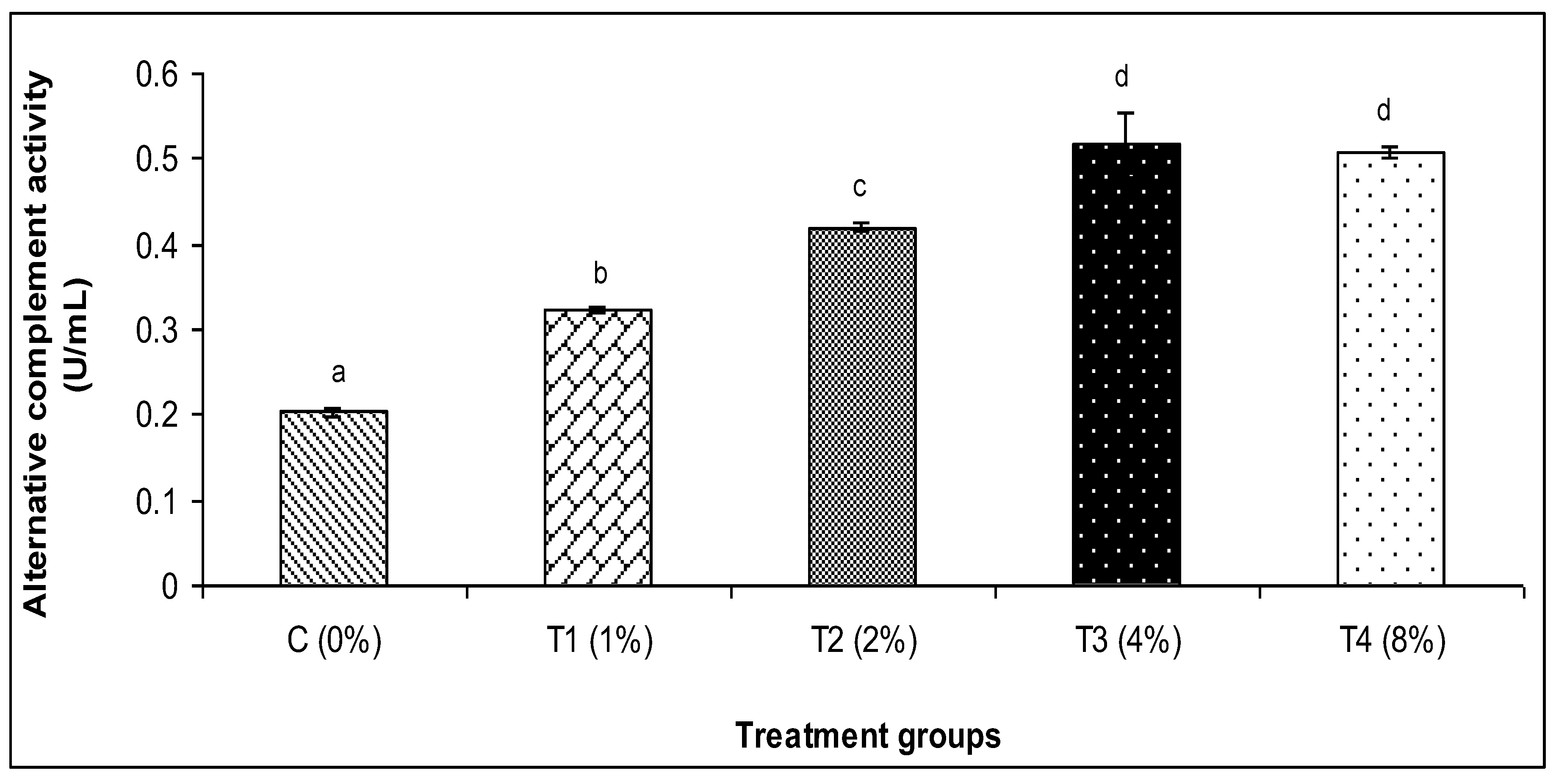
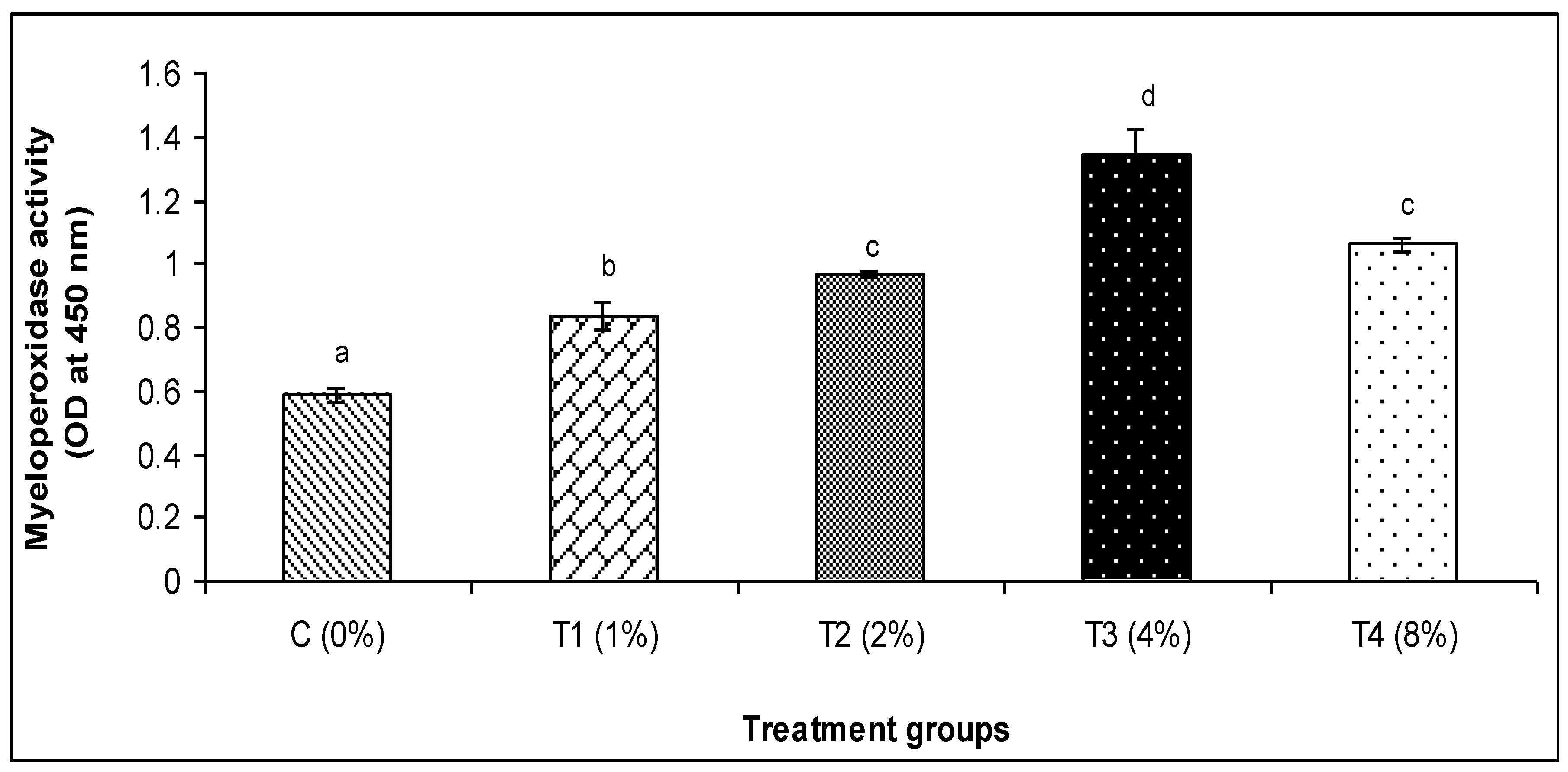
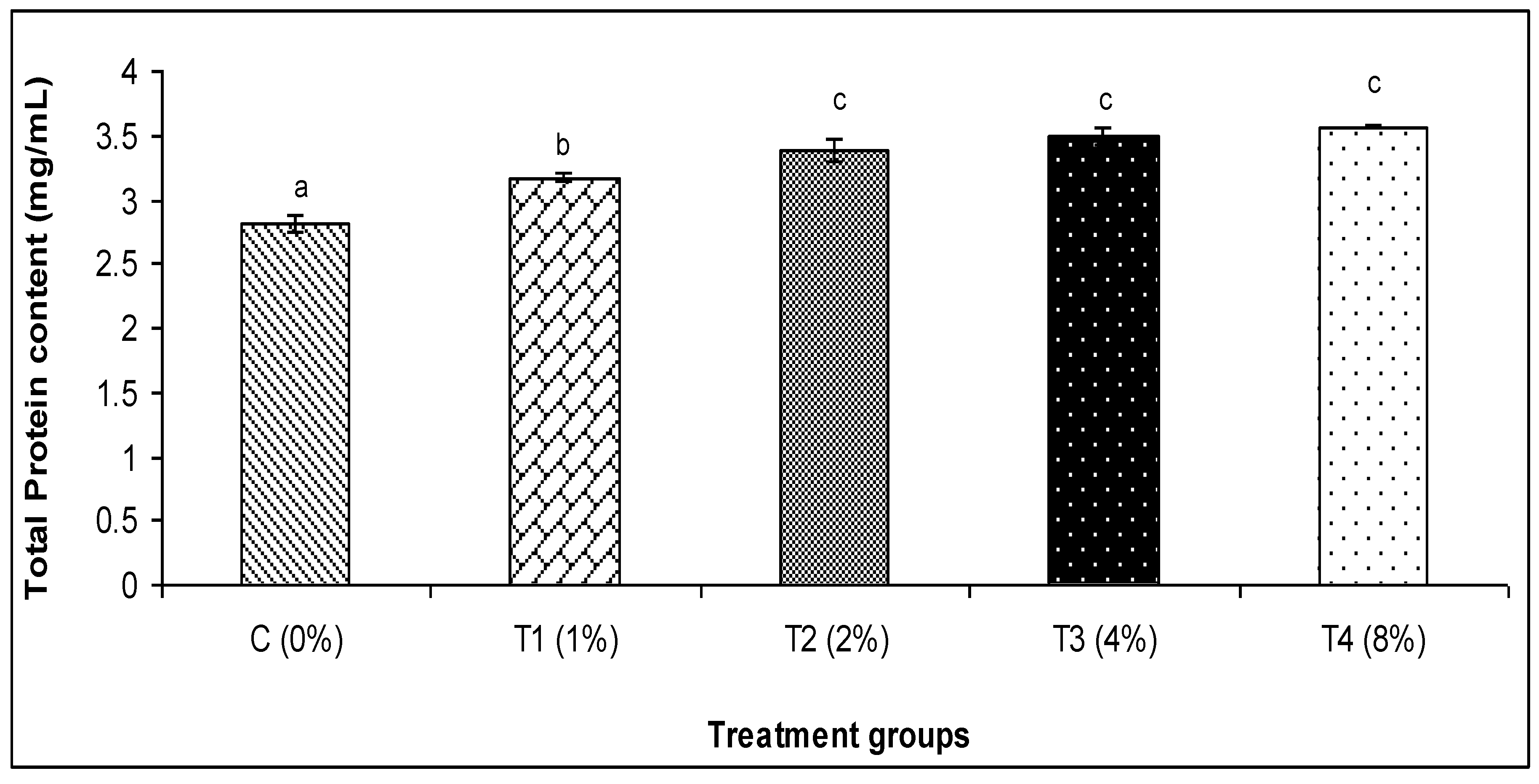
3.2. Skin Mucus Immunity
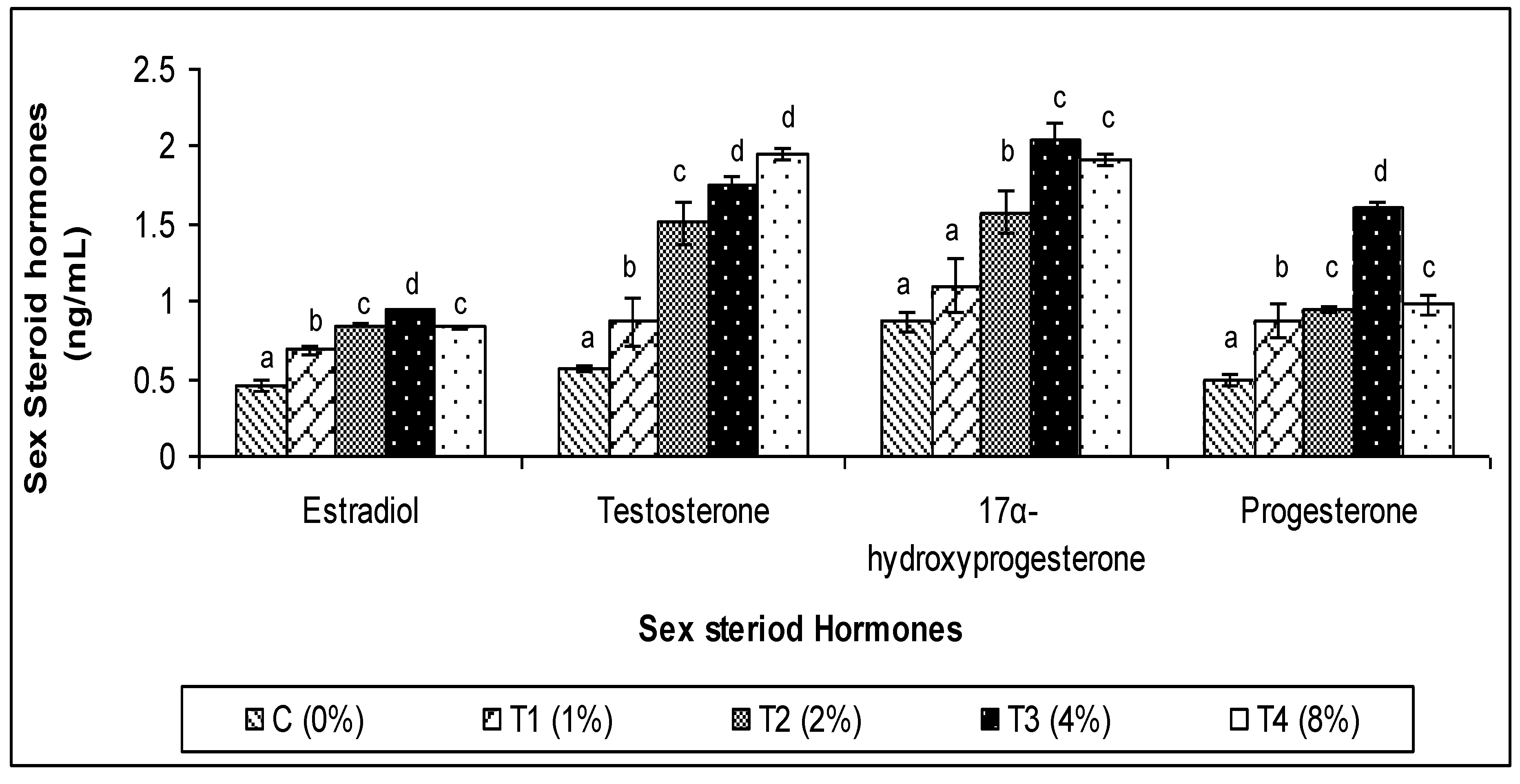
3.3. Sex Steroid Hormones
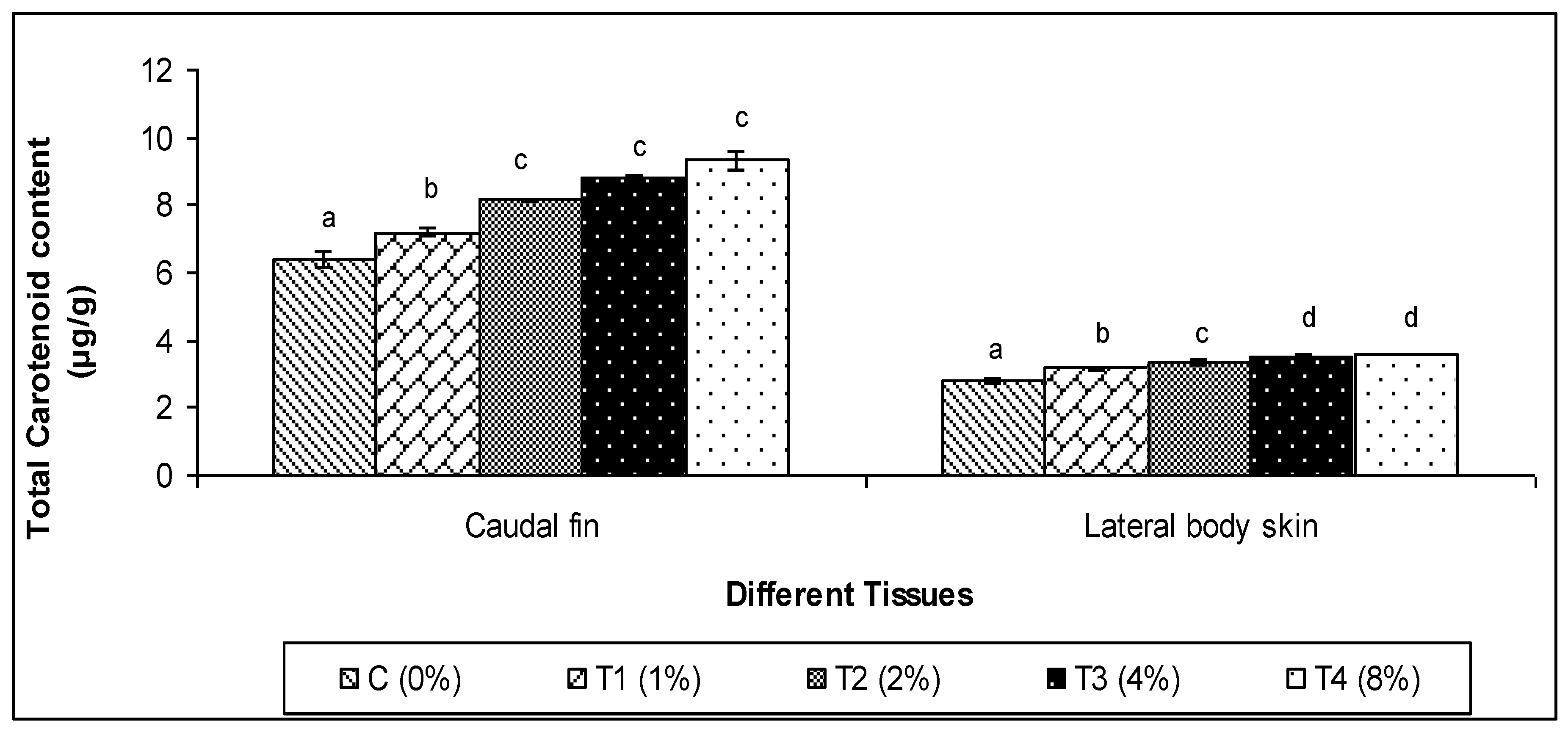
3.4. Total Carotenoid Content
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tlusty, M. The benefits and risks of aquacultural production for the aquarium trade. Aqaculture 2002, 3–4, 203–219. [Google Scholar] [CrossRef]
- Monticini, P. The Ornamental Fish Trade: Production and Commerce of Ornamental Fish: Technical-Managerial and Legislative Aspects; Food and Agriculture Organization of the United Nations: Rome, Italy, 2010; Volume 12, 134p. [Google Scholar]
- Lim, L.C.; Dhert, P.; Sorgeloos, P. Recent developments and improvements in ornamental fish packaging systems for air transport. Aquac. Res. 2003, 34, 923–935. [Google Scholar] [CrossRef]
- Hoseinifar, S.H.; Ahmadi, A.; Khalili, M.; Raeisi, M.; Van Doan, H.; Caipang, C.M. The study of antioxidant enzymes and immune-related genes expression in common carp (Cyprinuscarpio) fingerlings fed different prebiotics. Aquac. Res. 2017, 48, 5447–5454. [Google Scholar] [CrossRef]
- Awad, E.; Awaad, A. Role of medicinal plants on growth performance and immune status in fish. Fish Shellfish. Immunol. 2017, 67, 40–50. [Google Scholar] [CrossRef]
- Dawood, M.A.; Koshio, S.; Esteban, M.A. Beneficial roles of feed additives as immunostimulants in aquaculture: A review. Rev. Aquac. 2018, 10, 950–974. [Google Scholar] [CrossRef]
- Kim, S.-K.; Wijesekara, I. Development and biological activities of marine-derived bioactive peptides: A review. J. Funct. Foods 2010, 2, 1–9. [Google Scholar] [CrossRef]
- Wijesekara, I.; Kim, S.K. Angiotensin-I-converting enzyme (ACE) inhibitors from marine resources: Prospects in the pharmaceutical industry. Mar. Drugs 2010, 8, 1080–1093. [Google Scholar] [CrossRef]
- Elena, M.; Francisco, Y.; Erickson, K.L. Mailiohydrin, a Cytotoxic Chamigrene Dibromohydrin from a Phillippine Laurencia Species. J. Nat. Prod. 2001, 64, 790–791. [Google Scholar]
- Selvin, J.; Lipton, A.P. Development of a Rapid Mollusc Foot Adherence Bioassay for Detecting Potent Antifouling Bioactive Compounds. Curr. Sci. 2002, 83, 735–737. [Google Scholar]
- Daniel, N.; Sivaramakrishnan, T.; Saravanan, K.; Shalini, B.; Arunjyoti, B.; Sankar, R.; Dann Roy, S. A review on microalgae as potential fish feed ingredient. J. Andeman. Sci. Assoc. 2016, 1, 140–144. [Google Scholar]
- Younis, E.S.M.; Al-Quffail, A.S.; Al-Asgah, N.A.; Abdel-Warith, A.W.A.; Al-Hafedh, Y.S. Effect of dietary fish meal replacement by red algae, Gracilaria arcuata on growth performance and body composition of Nile tilapia, Oreochromis niloticus. Saud. J. Biol. Sci. 2018, 25, 198–203. [Google Scholar] [CrossRef] [PubMed]
- Perez-Velazquez, M.; Gatlin, I.I.I.; Gonzalez-Felix, M.L.; Garcia-Ortega, A. Partial replacement of fishmeal and fish oil by algal meals in diets of red drum Sciaenopsocellatus. Aquaculture 2018, 487, 41–50. [Google Scholar] [CrossRef]
- Leliaraert, F.; D’hondt, S.; Tyberghein, L.; Verbruggen, H.; De Clerck, O. Atypical development of Chaetomorpha antennina in culture (Cladophorales, Chlorophyta). Physiol. Res. 2011, 9, 91–97. [Google Scholar]
- Novaczek, I. A Guide to the Common Edible and Medicinal Sea Plants of the Pacific Islands; The University of the South Pacific: Suva, Fiji, 2001. [Google Scholar]
- Apaydin-Yagci, M.A.; Turna, I.I. A new record for the algal flora of Turkey: Chaetomorpha crassa (C.Ag.) Kutz. (Cladophoraceae, Chlorophyceae). Turk. J. Bot. 2002, 26, 171–174. [Google Scholar]
- Tsutsui, I.; Songphatkaew, J.; Meeanan, C. Co-culture with Chaetomorpha sp. enhanced growth performance and reduced feed conversion ratio of the giant tiger prawn, Penaeus monodon. Int. Aquat. Res. 2015, 7, 193–199. [Google Scholar] [CrossRef]
- Sattanathan, G.; Thanapal, G.; Swaminathan, S.; Kim, H.; Vijaya, R.; Kim, H.; Balasubramanian, B. Influences of dietary inclusion of algae Chaetomporpha aerea enhanced growth performance, immunity, haematological response and disease resistance of Labeo rohita challenged with Aeromonas hydrophila. Aquac. Rep. 2020, 17, 100353. [Google Scholar] [CrossRef]
- Sattanathan, G.; Tamizhazhagan, V.; Padmapriya, S.; Liu, W.C.; Balasubramanian, B. Effect of Green Algae Chaetomorpha antennina extract on growth, modulate immunity, and defenses against Edwardsiella tarda infection in Labeo rohita. Animals 2021, 10, 2033. [Google Scholar] [CrossRef]
- Song, X.; Xu, Q.; Zhou, Y.; Lin, C.; Yang, H. Growth, feed utilization and energy budgets of the sea cucumber Apostichopus japonicus with different diets containing the green tide macroalgae Chaetomorpha linum and the seagrass Zostera marina. Aquaculture 2017, 470, 157–163. [Google Scholar] [CrossRef]
- Bishat, M.; Kumar, A.; Shah, T.K. Effect of Moringa oleifera leaf powder on skin mucosal immune responses and growth performance of guppy, Poecillia reticulate (Peter, 1860). Aquac. Res. 2020, 51, 4984–4990. [Google Scholar] [CrossRef]
- Sankaran, K.; Gurnani, S. On the variation in the catalytic activity of lysozyme in fishes. Ind. J. Biochem. Biophys. 1972, 9, 162–165. [Google Scholar]
- Yano, T. Assays of hemolytic complement activity. In Techniques in Fish Immunology; Stolen, J.S., Fletcher, T.C., Anderson, D.P., Kaattari, S.L., Rowley, A.F., Eds.; SOS Publications: Fair Haven, NJ, USA, 1992; pp. 131–141. [Google Scholar]
- Quade, M.J.; Roth, J.A. A rapid direct assay to measure degranulation of bovine neutrophil primary granules. Vet. Immunol. Immunopathol. 1997, 58, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Siwicki, A.K.; Anderson, D.P.; Rumsey, G.L. Dietary intake of immunostimulants by rainbow trout affects non-specific immunity and protection against furunculosis. Vet. Immunol. Immunopathol. 1994, 41, 125–139. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.R.; Pham, M.A.; Lee, S.M. Effects of dietary paprika and lipid levels on growth and skin pigmentation of Pale Chub (Zacco platypus). Asian-Aust. J. Anim. Sci. 2012, 23, 724–732. [Google Scholar] [CrossRef]
- Feist, G.; Schreck, C.B.; Fitzpatrick, M.S.; Redding, J.M. Sex steroid profiles of coho salmon (Oncorhynchus kisutch) during early development and sexual differentiation. Gen. Comp. Endocrinol. 2009, 80, 299–313. [Google Scholar] [CrossRef]
- Kagawa, H.; Young, G.; Nagahama, Y. In vitro estradiol-17 β and testosterone production by ovarian follicles of the goldfish, Carassius auratus. Gen. Comp. Endocrinol. 1984, 54, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Varijakzhan, D.; Chong, C.M.; Abushelaibi, A.; Lai, K.S.; Lim, S.H.E. Middle Eastern plant extracts: An alternative to modern medicine problems. Molecules 2020, 25, 1126. [Google Scholar] [CrossRef]
- Sharma, A.; Deo, A.D.; Riteshkumar, S.T.; Chanu, T.I.; Das, A. Effect of Withania somnifera (L. Dunal) root as a feed additive on immunological parameters and disease resistance to Aeromonas hydrophila in Labeo rohita (Hamilton) fingerlings. Fish. Shellfish. Immunol. 2010, 29, 508–512. [Google Scholar] [CrossRef]
- Srinivasan, G.; Babu, P.; Murugeswari, V. Effect of neem products and insecticides on the egg parasitoids, Trichogramma spp. (Trichogrammatidae: Hymenoptera). Pestic. Res. J. 2001, 13, 250–253. [Google Scholar]
- Motlagh, A.H.; Paolucci, M.; Lashkarizadeh Bami, M.; Safari, O. Sexual parameters, digestive enzyme activities, and growth performance of guppy (Poecilia reticulata) fed garlic (Allium sativum) extract supplemented diets. J. World Aquac. Soc. 2020, 51, 1087–1097. [Google Scholar] [CrossRef]
- Hindu, S.V.; Chandrasekaran, N.; Mukherjee, A.; Thomas, J. A review on the impact of seaweed polysaccharide on the growth of probiotic bacteria and its application in aquaculture. Aquac. Int. 2018, 27, 227–238. [Google Scholar] [CrossRef]
- Sattanathan, G.; Tamizhazhagan, V.; Raza, N.; Shah, S.Q.A.; Hussain, M.Z.; Kim, K.H. Effects of Green Alga, Chaetomorpha aerea Extract on Non-Specific Immune Responses and Disease Resistance against Edwardsiella tarda Infection in Labeo rohita. Appl. Sci. 2021, 11, 4325. [Google Scholar] [CrossRef]
- Elizondo-Gonzalez, R.; Quiroz-Guzman, E.; Escobedo-Fregoso, C.; Magallon-Servin, P.; Pena-Rodríguez, A. Use of seaweed Ulva lactuca for water bioremediation and as feed additive for white shrimp Litopenaeus vannamei. PeerJ 2018, 5, 44–59. [Google Scholar] [CrossRef]
- Moghanlou, K.S.; Isfahani, E.N.; Dorafshan, S.; Tukmechi, A.; Aramli, M.S. Effects of dietary supplementation with Stachys lavandulifolia Vahl extract on growth performance, hemato-biochemical and innate immunity parameters of rainbow trout (Oncorhynchus mykiss). Anim. Feed Sci. Technol. 2017, 237, 98–105. [Google Scholar] [CrossRef]
- Gabriel, N.N.; Qiang, J.; He, J.; Ma, X.Y.; Kpundeh, M.D. Dietary Aloe vera supplementation on growth performance, some haemato-biochemical parameters and disease resistance against Streptococcus iniae in tilapia. Fish. Shellfish. Immunol. 2015, 44, 504–514. [Google Scholar] [CrossRef] [PubMed]
- Parrillo, L.; Coccia, E.; Volpe, M.G.; Siano, F.; Pagliarulo, C.; Scioscia, E.; Paolucci, M. Olive mill wastewater enriched diet positively affects growth, oxidative and immune status and intestinal microbiota in the crayfish, Astacus leptodactylus. Aquaculture 2017, 473, 161–168. [Google Scholar] [CrossRef]
- Farsani, M.N.; Hoseinifar, S.H.; Rashidian, G.; Farsani, H.G.; Ashouri, G.; Van Doan, H. Dietary effects of Coriandrum sativum extract on growth performance, physiological and innate immune responses and resistance of rainbow trout (Oncorhynchus mykiss) against Yersinia ruckeri. Fish. Shellfish. Immunol. 2019, 91, 233–240. [Google Scholar] [CrossRef]
- Chakraborty, S.B.; Horn, P.; Hancz, C. Application of phytochemicals as growth-promoters and endocrine modulators in fish culture. Rev. Aquac. 2014, 6, 1–19. [Google Scholar] [CrossRef]
- Mukherjee, P.K.; Biswas, R.; Sharma, A.; Banerjee, S.; Biswas, S.; Katiyar, C.K. Validation of medicinal herbs for anti-tyrosinase potential. J. Herb. Med. 2018, 14, 1–16. [Google Scholar] [CrossRef]
- Beck, V.; Unterrieder, E.; Krenn, L.; Kubelka, W.; Jungbauer, A. Comparison of hormonal activity (estrogen, androgen and progestin) of standardized plant extracts for large scale use in hormone replacement therapy. J. Steroid Biochem. Mol. Biol. 2003, 84, 259–268. [Google Scholar] [CrossRef]
- Yeganeh, S.; Sotoudeh, A.; Movaffagh, A.N. Effects of Tribulus terrestris extract on growth and reproductive performance of male convict cichlid (Cichlasoma nigrofasciatum). Turk. J. Fish. Aquat. Sci. 2017, 17, 1003–1007. [Google Scholar] [CrossRef]
- Davis, K.B.; Goudie, C.A.; Simco, B.A. Influence of dihydrotestosterone on sex determination in channel catfish and blue catfish: Period of developmental sensitivity. Gen. Comp. Endocrinol. 1992, 86, 147–151. [Google Scholar] [CrossRef] [PubMed]
- Patiňo, R.; Davis, K.B.; Shoore, J.E. Sex differentiation of channel catfish gonads: Normal development and effects of temperature. J. Exp. Zool. 1996, 276, 209–218. [Google Scholar] [CrossRef]
- Tzchori, I.; Degani, G.; Elisha, R. The influence of phytoestrogens and oestradiol-17β on growth and sex determination in the European eel (Anguilla anguilla). Aquacult. Res. 2004, 35, 1213–1219. [Google Scholar] [CrossRef]
- Green, C.C.; Kelly, A.M. Effects of the estrogen mimic genistein as a dietary component on sex differentiation and ethoxyresorufin-O-deethylase (EROD) activity in channel catfish (Ictalurus punctatus). Fish. Physiol. Biochem. 2009, 35, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Forsatkar, M.N.; HedayatiRad, M.; Luchiari, A.C. “Not tonight zebrafish”: The effects of Ruta graveolens on reproduction. Pharm. Biol. 2018, 56, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Reyes, L.I.; Constantinides, S.V.; Bhandari, S.; Frongillo, E.A.; Schreinemachers, P.; Wertheim-Heck, S. Actions in global nutrition initiatives to promote sustainable healthy diets. Glob. Food Sec. 2021, 31, 100585. [Google Scholar] [CrossRef]
- Idenyi, J.N.; Eya, J.C.; Ogbonna, J.C. Characterization of strains of Chlorella from Abakaliki, Nigeria, for the production of high-value products under variable temperatures. J. Appl. Phycol. 2021, 33, 275–285. [Google Scholar] [CrossRef]
- Batista, A.P.; Niccolai, A.; Fradinho, P.; Fragoso, S.; Bursic, I.; Rodolfi, L.; Biondi, L.; Tredici, R.; Sousa, I.; Raymundo, A. Microalgae biomass as an alternative ingredient in cookies: Sensory, physical and chemical properties, antioxidant activity and in vitro digestibility. Algal. Res. 2017, 26, 161–171. [Google Scholar] [CrossRef]
- Moheimani, N.R.; Vadiveloo, A.; Ayre, J.M.; Pluske, J.R. Nutritional profile and in vitro digestibility of microalgae grown in anaerobically digested piggery effluent. Algal. Res. 2018, 35, 362–369. [Google Scholar] [CrossRef]
- Shah, M.R.; Lutzu, G.A.; Alam, M.D.; Pallab Sarker, M.A.; Chowdhury, K.; Parsaeimehr, A.; Liang, Y.; Daroch, M. Microalgae in aquafeeds for a sustainable aquaculture industry. J. Appl. Phycol. 2018, 30, 197–213. [Google Scholar] [CrossRef]
- Nethravathy, M.U.; Jitendra, G.; Sandeep, N.; Mudliar, A.; Shekh, Y. Recent advances in microalgal bioactives for food, feed, and healthcare products: Commercial potential, market space, and sustainability. Compr. Rev. Food Sci. Food Saf. 2019, 18, 1882–1897. [Google Scholar]
- Mabeau, S.; Fleurence, J. Seaweed in food products: Biochemical and nutritional aspects. Trends Food Sci. Technol. 1993, 4, 103–107. [Google Scholar] [CrossRef]
- Fleurence, J. Seaweed proteins: Biochemical, nutritional aspects and potential uses. Trends Food Sci. Technol. 1999, 10, 25–28. [Google Scholar] [CrossRef]
- Galland-Irmouli, A.-V.; Fleurence, J.; Lamghari, R.; Luçon, M.; Rouxel, C.; Barbaroux, O.; Bronowicki, J.-P.; Villaume, C.; Guéant, J.-L. Nutritional value of proteins from edible seaweed palmaria palmata (dulse). J. Nutr. Biochem. 1999, 10, 353–359. [Google Scholar] [CrossRef] [PubMed]
- Pihlanto-Leppälä, A. Bioactive peptides derived from bovine whey proteins: Opioid and aceinhibitory peptides. Trends Food Sci. Technol. 2000, 11, 347–356. [Google Scholar] [CrossRef]
- Mayer, A.M.; Gustafson, K.R. Marine pharmacology in 2000: Antitumor and cytotoxic compounds. Int. J. Cancer 2003, 105, 291–299. [Google Scholar] [CrossRef] [PubMed]
- Elias, R.J.; Kellerby, S.S.; Decker, E.A. Antioxidant activity of proteins and peptides. Crit. Rev. Food Sci. Nutr. 2008, 48, 430–441. [Google Scholar] [CrossRef]
- Hajiuon, B. Effects of garlic (Allium sativum L.) hydroalcoholic extract on estrogen, progesterone and testosterone levels in rats exposed to cell phone radiation. Zahedan J. Res. Med. Sci. 2014, 16, 20–25. [Google Scholar]
- Yanar, M.; Buyukcapar, H.M.; Yanar, Y. Effects of hot and sweet red peppers (Capsicum annum) as feed supplements on pigmentation, sensory properties and weight gain of rainbow trout (Onchorhynchus mykiss). Ann. Anim. Sci. 2016, 16, 825–834. [Google Scholar] [CrossRef]
- Yigit, N.O.; Bahadır Koca, S.; Özmen, Ö.; Didinen, B.I.; Metin, S. The effects of dietary administration with high level red pepper (Capsicum annuum) on growth performance, coloration, histology and protection against Aeromonas sobria in yellow tail cichlid, Pseudotropheusacei. Acta Aquat. Turc. 2019, 15, 340–346. [Google Scholar] [CrossRef]
- Yimaz, S.; Ergun, S. Effect of red pepper (Capsicum annum) on pigmentation blue streak hap (Labidpchromis caeruleus). Isr. J. Aquac.-Bamidgeh 2011, 63, 633–638. [Google Scholar]
- Yilmaz, S.; Ergun, S.; Soytas, N. Enhancement of growth performance, and pigmentation in red Oreochromis mossambicus associated with dietary intake Astaxanthin, Paprikla, or Capsicum. Isr. J. Aquac.-Bamidgeh 2013, 65, 1–7. [Google Scholar]
- Silmestad, R.; Fossen, T.; Vagen, I.M. Onions: A Source of unique dietary flavonoids. J. Agric. Food Chem. 2007, 55, 10067–10080. [Google Scholar] [CrossRef]
- Wang, X. Allicin attentunates lipopolysaccharides induced acute lung injury in neonatal rates via the PI3K/Akt pathway. Mol. Med. Rep. 2018, 17, 6777–6783. [Google Scholar] [PubMed]
- Giuliano, G.; Aquilani, R.; Dharmapuri, S. Metabolic engineering of plant carotenoids. Trends Plant Sci. 2000, 5, 406–409. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.K.; Jha, A.K.; Pal, A.K.; Venktaeshwarlu, G. Use of natural carotenoids for pigmentation in fishes. Nat. Prod. Radiance 2007, 6, 46–49. [Google Scholar]
- Raymundo, A.; Gouveida, L.; Batista, A.P.; Empis, J.; Sousa, I. Fat mimetic capacity of Chlorella vulgaris biomas in oil-in-water food emulsions stabilized by pea protein. Food Res. Int. 2005, 38, 961–965. [Google Scholar] [CrossRef]
- Sales, K.; Janseens, G.P. Nutrient requirements of ornamental fish. Aquat. Living Resour. 2003, 16, 533–540. [Google Scholar] [CrossRef]
- Guroy, B.; Şahin, I.; Mantoğlu, S.; Kayalı, S. Spirulina as a natural carotenoid source on growth, pigmentation and reproductive performance of yellow tail cichlid Pseudotropheusacei. Aquac. Int. 2012, 20, 869–878. [Google Scholar] [CrossRef]
- Güroy, D.; Güroy, B.; Merrifield, D.L.; Ergün, S.; Tekinay, A.A.; Yiğit, M. Effect of dietary Ulva and Spirulina on weight loss and body composition of rainbow trout, Oncorhynchus mykiss (Walbaum), during a starvation period. J Anim. Physiol. Anim. Nutr. (Berl.) 2011, 95, 320–327. [Google Scholar] [CrossRef] [PubMed]
- Spolaore, P.; Joannis-Cassan, C.; Duran, E.; Isambert, A. Commercial applications of microalgae. J. Biosci. Bioeng. 2006, 101, 87–96. [Google Scholar] [CrossRef]
- Nakagawa, H.; Montgomery, W.L. Algae. In Dietary Supplements for the Health and Quality of Cultured Fish; Nakagawa, H., Sato, M., Gatlin, D.M., Eds.; Cabi International: Cambridge, UK, 2007; pp. 133–167. [Google Scholar]
- Sattanathan, G.; Liu, W.L.; Padmapriya, S.S.; Pushparaj, K.; Sureshkumar, S.; Lee, J.W.; Balasubramanian, B.; Kim, I.H. Effects of Dietary Blend of Algae Extract Supplementation on Growth, Biochemical, Haemato-Immunological Response, and Immune Gene Expression in Labeo rohita with Aeromonas hydrophila Post-Challenges. Fishes 2023, 8, 7. [Google Scholar] [CrossRef]
- James, R.; Sampath, K.; Thangarathinam, R.; Vasudevan, I. Effect of dietary Spirulina level on growth, fertility, coloration and leucocyte count in red swordtail, Xiphophorus helleri. Isr. J. Aquac.-Bamidgeh 2006, 58, 97–104. [Google Scholar] [CrossRef]
- James, R.; Vasudhevan, I.; Sampath, K. Interaction of Spirulina with different levels of vitamin E on growth, reproduction, and coloration in goldfish (Carassius auratus). Isr. J. Aquac.-Bamidgeh 2009, 61, 330–338. [Google Scholar] [CrossRef]
- Vasudhevan, I.; James, R. Effect of optimum Spirulina along with different levels of vitamin C incorporated diets on growth, reproduction and coloration in goldfish Carassius auratus (Linnaeus, 1758). Indian J. Fish. 2011, 58, 101–106. [Google Scholar]
- Khanzadeh, M.; Fereidouni, A.E.; Berenjestanaki, S.S. Effects of partial replacement of fish meal with Spirulina platensis meal in practical diets on growth, survival, body composition, and reproductive performance of three-spot gourami (Trichopodustrichopterus) (Pallas, 1770). Aquac. Int. 2016, 24, 69–84. [Google Scholar] [CrossRef]

| Ingredients | Quantity (in g/kg Diet) |
|---|---|
| Rice bran | 170 |
| Soya bean meal | 550 |
| Dry fish meal | 50 |
| Wheat flour | 20 |
| Corn flour | 150 |
| Vegetable oil (v/w) | 25 |
| Iodine salt | 10 |
| Vitamin mineral mixture | 25 |
| Parameters | Control | T1 | T2 | T3 | T4 |
|---|---|---|---|---|---|
| Initial weight (g) | 0.019 ± 0.0006 a | 0.019 ± 0.0007 a | 0.019 ± 0.0005 a | 0.019 ± 0.0006 a | 0.019 ± 0.0003 a |
| Final weight (g) | 0.028 ± 0.0011 a | 0.031 ± 0.0006 b | 0.034 ± 0.0004 c | 0.040 ± 0.0008 d | 0.041 ± 0.0007 d |
| Weight gain (g) | 0.010 ± 0.0011 a | 0.012 ± 0.0005 a | 0.015 ± 0.0005 b | 0.021 ± 0.0008 c | 0.022 ± 0.0005 c |
| Specific growth rate (%) | 0.41 ± 0.008 a | 0.46 ± 0.003 b | 0.58 ± 0.006 c | 0.74 ± 0.006 d | 0.77 ± 0.005 e |
| Feed conversion ratio (g) | 0.077 ± 0.0009 d | 0.062 ± 0.0007 c | 0.050 ± 0.0003 b | 0.035 ± 0.0006 a | 0.034 ± 0.001 a |
| Fry production | 0.735 ± 0.004 a | 1.00 ± 0.114 b | 1.55 ± 0.04 c | 1.83 ± 0.060 d | 2.06 ± 0.04 e |
| Survival rate (%) | 100 ± 0.00 a | 100 ± 0.00 a | 100 ± 0.00 a | 100 ± 0.00 a | 100 ± 0.00 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Govindharajan, S.; Balasubramanian, B.; Thapo, V.; Venkatalakshmi, S.; Liu, W.-C. Enhancement of Skin Mucus Immunity, Carotenoid Content, Sexual Parameters, and Growth Response in Guppy Fish (Poecilia reticulata) Fed with Green Algae (Chaetomorpha aerea) Diets. Fishes 2024, 9, 101. https://doi.org/10.3390/fishes9030101
Govindharajan S, Balasubramanian B, Thapo V, Venkatalakshmi S, Liu W-C. Enhancement of Skin Mucus Immunity, Carotenoid Content, Sexual Parameters, and Growth Response in Guppy Fish (Poecilia reticulata) Fed with Green Algae (Chaetomorpha aerea) Diets. Fishes. 2024; 9(3):101. https://doi.org/10.3390/fishes9030101
Chicago/Turabian StyleGovindharajan, Sattanathan, Balamuralikrishnan Balasubramanian, Vivi Thapo, Sournamanikam Venkatalakshmi, and Wen-Chao Liu. 2024. "Enhancement of Skin Mucus Immunity, Carotenoid Content, Sexual Parameters, and Growth Response in Guppy Fish (Poecilia reticulata) Fed with Green Algae (Chaetomorpha aerea) Diets" Fishes 9, no. 3: 101. https://doi.org/10.3390/fishes9030101
APA StyleGovindharajan, S., Balasubramanian, B., Thapo, V., Venkatalakshmi, S., & Liu, W.-C. (2024). Enhancement of Skin Mucus Immunity, Carotenoid Content, Sexual Parameters, and Growth Response in Guppy Fish (Poecilia reticulata) Fed with Green Algae (Chaetomorpha aerea) Diets. Fishes, 9(3), 101. https://doi.org/10.3390/fishes9030101










