First Evidence of Secondary Sexual Dimorphism in the Freshwater Fish Family Botiidae: A Newly Recognised Synapomorphy of Loaches (Cypriniformes: Cobitoidea)
Abstract
1. Introduction
2. Materials and Methods
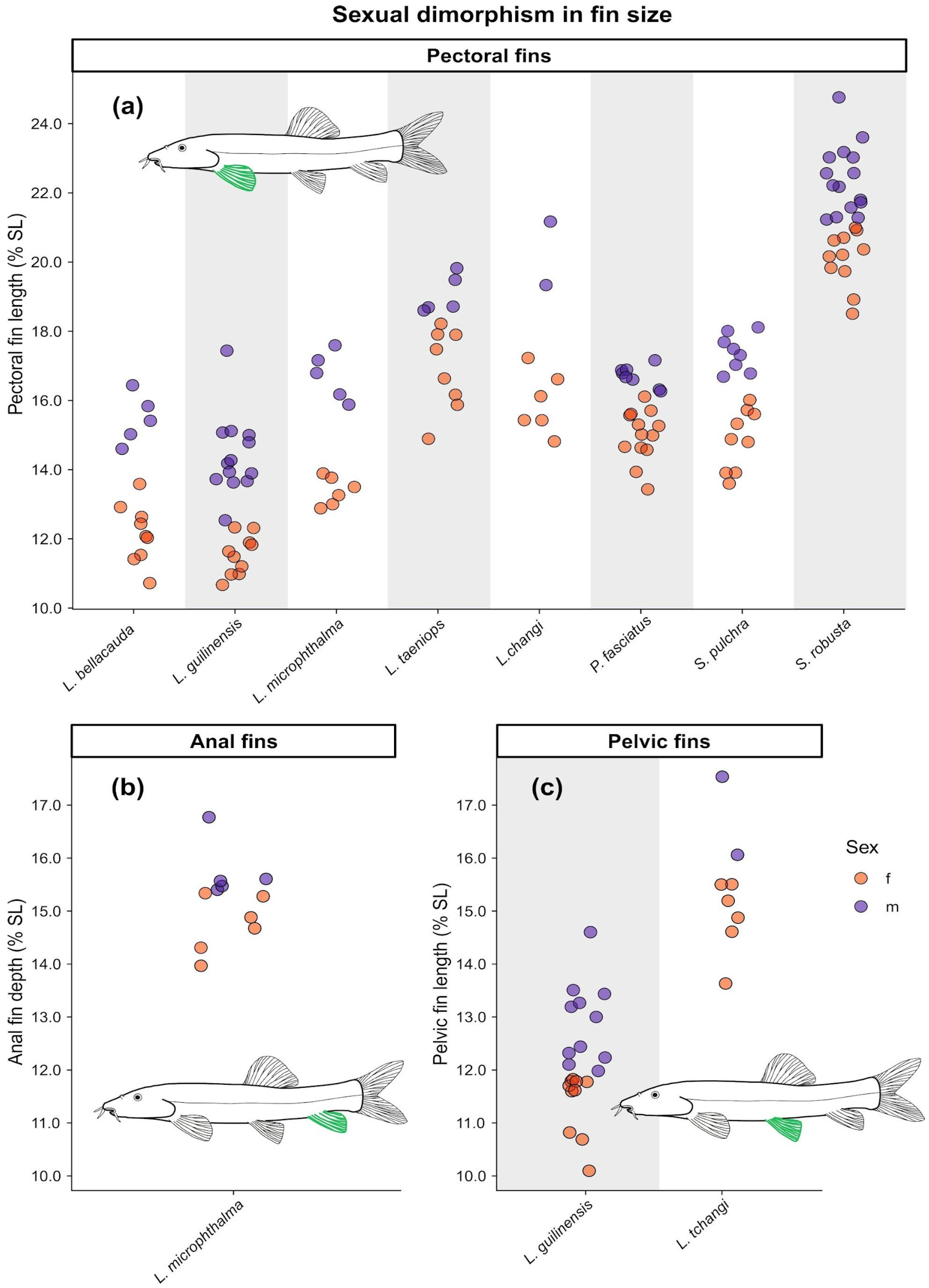
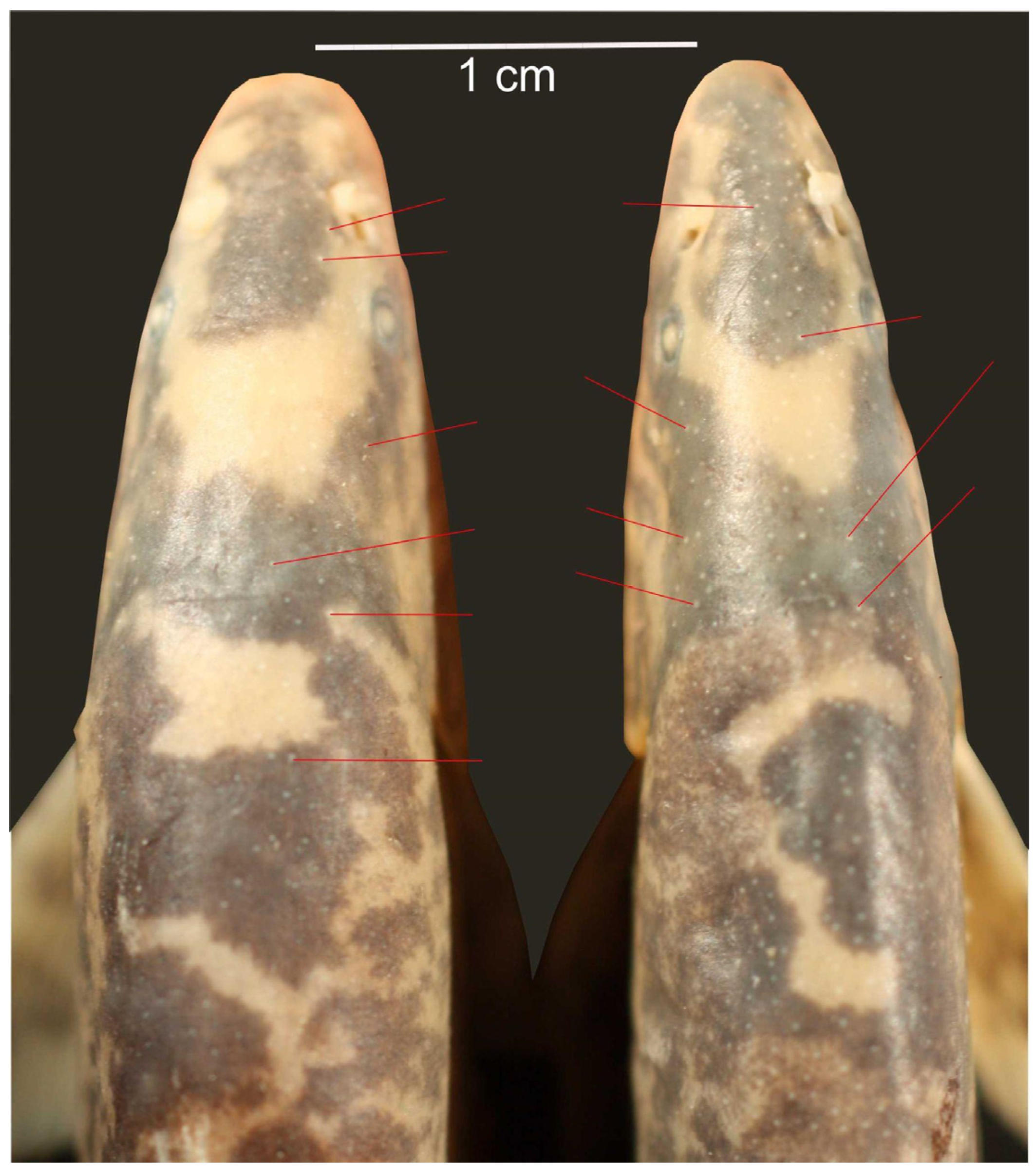
3. Results
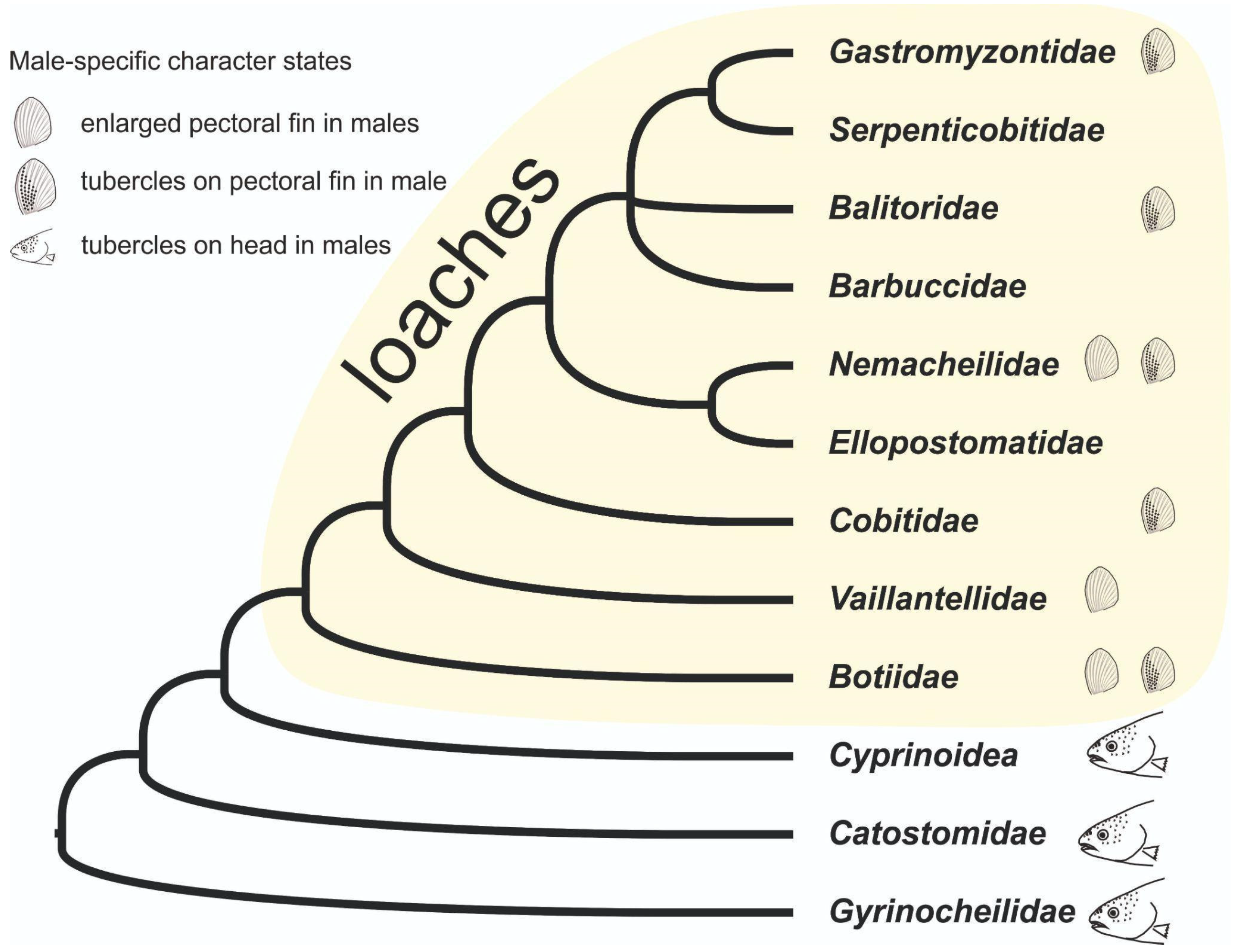
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nelson, J.S. Fishes of the World, 4th ed.; Wiley: Hoboken, NJ, USA, 2006; 752p. [Google Scholar]
- Šlechtová, V.; Bohlen, J.; Tan, H.H. Families of Cobitoidea (Teleostei; Cypriniformes) as revealed from nuclear genetic data and the position of the mysterious genera Barbucca, Psilorhynchus, Serpenticobitis and Vaillantella. Mol. Phylogen. Evol. 2007, 44, 1358–1365. [Google Scholar] [CrossRef]
- Kottelat, M. Conspectus cobitidum: An inventory of the loaches of the world (Teleostei: Cypriniformes: Cobitoidei). Raffles Bull. Zool. 2012, 26, 1–199. [Google Scholar]
- Stout, C.C.; Tan, M.; Lemmon, A.R.; Lemmon, E.M.; Armbruster, J.W. Resolving Cypriniformes relationships using an anchored enrichment approach. BMC Evol. Biol. 2016, 16, 244. [Google Scholar] [CrossRef] [PubMed]
- Hirt, M.V.; Arratia, G.; Chen, W.J.; Mayden, R.L. Effects of gene choice, base composition and rate heterogeneity on inference and estimates of divergence times in cypriniform fishes. Biol. J. Linn. Soc. 2017, 121, 319–339. [Google Scholar] [CrossRef]
- Tan, M.; Armbruster, J.W. Phylogenetic classification of extant genera of fishes of the order Cypriniformes (Teleostei: Ostariophysi). Zootaxa 2018, 4476, 6–39. [Google Scholar] [CrossRef] [PubMed]
- Sawada, Y. Phylogeny and zoogeography of the superfamily Cobitoidea (Cyprinoidei: Cypriniformes). Mem. Fac. Fish. Hokkaido Univ. 1982, 28, 65–223. [Google Scholar]
- Conway, K.W. Osteology of the South Asian Genus Psilorhynchus McClelland, 1839 (Teleostei: Ostariophysi: Psilorhynchidae), with investigation of its phylogenetic relationships within the order Cypriniformes. Zool. J. Linn. Soc. 2011, 163, 50–154. [Google Scholar] [CrossRef]
- Bohlen, J.; Šlechtová, V. Phylogenetic position of the fish genus Ellopostoma (Teleostei: Cypriniformes) using molecular genetic data. Ichthyol. Explor. Freshw. 2009, 20, 157–162. [Google Scholar]
- Saitoh, K.; Sado, T.; Mayden, R.L.; Hanzawa, N.; Nakamura, K.; Nishida, M.; Miya, M. Mitogenomic Evolution and Interrelationships of the Cypriniformes (Actinopterygii: Ostariophysi): The first evidence toward resolution of higher-level relationships of the world’s largest freshwater fish clade based on 59 whole mitogenome sequences. J. Mol. Evol. 2006, 63, 826–841. [Google Scholar] [CrossRef]
- Nelson, J.S.; Grande, T.C.; Wilson, M.V.H. Fishes of the World, 5th ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2016; 707p. [Google Scholar]
- Rainboth, W.J. Fishes of the Cambodian Mekong. In FAO Species Identification Field Guide for Fishery Purposes; FAO: Rome, Italy, 1996; 265p. [Google Scholar]
- Tan, H.H.; Lim, K.K.P. A new species of Ellopostoma (Teleostei: Cypriniformes: Balitoridae) from Peninsular Thailand. Raffles Bull. Zool. 2002, 50, 453–457. [Google Scholar]
- Bohlen, J.; Li, F.; Šlechtová, V. Phylogenetic position of the genus Bibarba as revealed from molecular genetic data (Teleostei: Cobitidae). Ichthyol. Explor. Freshw. 2019, 29, 1–8. [Google Scholar] [CrossRef]
- Basumatary, B.; Singh, Y.L. Redescription of Neoeucirrhichthys maydelli (Teleostei: Cobitidae) from Brahmaputra drainage, Assam, Northeastern India. Rec. Zool. Surv. India 2023, 123, 151–158. [Google Scholar] [CrossRef]
- Roberts, T.R. Quintabarbates bicolor, a new genus and species of cobitid Fish from the Middle Chindwin Basin in Myanmar. Int. J. Ichthyol. 2020, 26, 17–20. [Google Scholar]
- Bănărescu, P. Zoogeography of Fresh Waters, Vol. 2: Distribution and Dispersal of Freshwater Animals in North America and Eurasia; AULA: Wiesbaden, Germany, 1992; 511p. [Google Scholar]
- Wootton, R.J.; Smith, C. Reproductive Biology of Teleost Fishes; Wiley-Blackwell: Oxford, UK, 2015; 496p. [Google Scholar]
- Bohlen, J. Similarities and differences in the reproductive biology of loaches (Cobitis and Sabanejewia) under laboratory conditions. Folia Zool. 2000, 49, 179–186. [Google Scholar]
- Bohlen, J. First report on the spawning behaviour of a golden spined loach, Sabanejewia vallachica (Teleostei: Cobitidae). Folia Zool. 2008, 57, 139–146. [Google Scholar]
- Bohlen, J. Spawning marks in spined loaches (Cobitis taenia, Cobitidae, Teleostei). Folia Zool. 2008, 57, 168–172. [Google Scholar]
- Morii, K.; Takakura, K.I. Reproductive behavior of endangered spined loach Cobitis magnostriata in the field. J. Ethol. 2022, 40, 167–174. [Google Scholar] [CrossRef]
- Buj, I.; Šanda, R.; Marčić, Z.; Ćaleta, M.; Mrakovčić, M. Sexual dimorphism of five Cobitis species (Cypriniformes, Actinopterygii) in the Adriatic watershed. Folia Zool. 2015, 64, 97–103. [Google Scholar] [CrossRef]
- Šedivá, A. On the sexual dimorphism of stone loach Barbatula barbatula (Balitoridae). Folia Zool. 2001, 50, 201–208. [Google Scholar]
- Kottelat, M.; Tan, H.H. Kottelatlimia hipporhynchos, a new species of loach from southern Borneo (Teleostei: Cobitidae). Zootaxa 2008, 1967, 63–72. [Google Scholar] [CrossRef]
- Kottelat, M. Mustura celata, a new genus and species of loaches from northern Myanmar, and an overview of Physoschistura and related taxa (Teleostei: Nemacheilidae). Ichthyol. Explor. Freshw. 2018, 28, 289–314. [Google Scholar] [CrossRef]
- Nalbant, T. Studies on loaches (Pisces: Ostariophysi; Cobitoidea). I. An evaluation of the valid genera of Cobitinae. Trav. Mus. Hist. Nat. “Grigore Antipa” 1994, 34, 375–380. [Google Scholar]
- Kottelat, M. Barbucca heokhuii, a new species of loach from central Borneo (Teleostei: Barbuccidae). Raffles Bull. Zool. 2025, 73, 54–66. [Google Scholar] [CrossRef]
- Šlechtová, V.; Bohlen, J.; Perdices, A. Molecular phylogeny of the freshwater fish family Cobitidae (Cypriniformes: Teleostei): Delimitation of genera, mitochondrial introgression and evolution of sexual dimorphism. Mol. Phylogen. Evol. 2008, 47, 812–831. [Google Scholar] [CrossRef] [PubMed]
- Wiley, M.L.; Collette, B.B. 1970. Breeding tubercles and contact organs in fishes: Their occurrence, structure and significance. Bull. Am. Mus. Nat. Hist. 1970, 143, 143–216. [Google Scholar]
- Freyhof, J.; Serov, D.V. Review of the genus Sewellia with description of two new species from Vietnam (Cypriniformes: Balitoridae). Ichthyol. Explor. Freshw. 2000, 11, 217–240. [Google Scholar]
- Kottelat, M. Indochinese nemacheilines. In A Revision of Nemacheiline Loaches (Pisces: Cypriniformes) of Thailand, Burma, Laos, Cambodia and Southern Viet Nam; Pfeil: München, Germany, 1990; 544p. [Google Scholar]
- Conway, K.W.; Kottelat, M. Physoschistura mango, a new miniature species of loach from Myanmar (Teleostei: Nemacheilidae). Raf. Bull. Zool. 2023, 71, 681–701. [Google Scholar]
- Gong, X.; Zhang, E. Bashimyzon cheni, a new genus and species of sucker loach (Teleostei, Gastromyzontidae) from South China. Zoosyst. Evol. 2024, 100, 309–324. [Google Scholar] [CrossRef]
- Nalbant, T.T. Sixty million years of evolution. Part one: Family Botiidae (Pisces: Ostariophysi:Cobitoidea). Trav. Mus. Hist. Nat. “Grigore Antipa” 2002, 44, 309–333. [Google Scholar]
- Chen, W.-J.; Lheknim, V.; Mayden, R.L. Molecular phylogeny of the Cobitoidea (Teleostei: Cypriniformes) revisited: Position of enigmatic loach Ellopostoma resolved with six nuclear genes. J. Fish Biol. 2009, 75, 2197–2208. [Google Scholar] [CrossRef]
- Wickham, H. Ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2023; Available online: https://www.R-project.org/ (accessed on 2 October 2025).
- Bohlen, J.; Šlechtová, V. A new genus and two new species of loaches (Teleostei: Nemacheilidae) from Myanmar. Ichthyol. Explor. Freshw. 2011, 22, 1–10. [Google Scholar]
- Branson, B.A. Observations on the Distribution of Nuptial Tubercles in Some Catostomid Fishes. Trans. Kans. Acad. Sci. 1961, 64, 360–372. [Google Scholar] [CrossRef]
- Akin, D.R.; Jenkins, R.E.; Armbruster, J.W. Description of the Apalachicola Redhorse (Catostomidae: Moxostoma). Zootaxa 2025, 5711, 181–202. [Google Scholar] [CrossRef]
- Jenkins, R.E.; Favrot, S.D.; Freeman, B.J.; Albanese, B.; Armbruster, J.W. Description of the Sicklefin Redhorse (Catostomidae: Moxostoma). Ichthyol. Herpetol. 2025, 113, 27–43. [Google Scholar] [CrossRef]
- Rupprecht, R.J.; Jahn, L.A. Biological notes on blue suckers in the Mississippi River. Trans. Am. Fish. Soc. 1980, 109, 323–326. [Google Scholar] [CrossRef]
- Stanley, D.R. Sexual dimorphism of pelvic fin shape in four species of Catostomidae. Trans. Am. Fish. Soc. 1988, 117, 600–602. [Google Scholar] [CrossRef]
- Laskar, B.A.; Sarma, D.; Das, D.N. Biometrics and Sexual dimorphism of Neolissochilus hexagonolepis (McClelland). Int. J. Fish. Aquat. Stud. 2013, 1, 61–65. [Google Scholar]
- Johnston, C.E.; Knight, C.L. Life-history traits of the bluenose shiner, Pteronotropis welaka (Cypriniformes: Cyprinidae). Copeia 1999, 1999, 200–205. [Google Scholar] [CrossRef]
- Smith, C.; Reichard, M.; Jurajda, P.; Przybylski, M. The reproductive ecology of the Europeanan bitterling (Rhodeus sericeus). J. Zool. 2004, 262, 107–124. [Google Scholar] [CrossRef]
- Li, F.; Liao, T.Y.; Arai, R. Two new species of Rhodeus (Teleostei: Cyprinidae: Acheilognathinae) from the river Yangtze, China. J. Vertebr. Biol. 2020, 69, 1–17. [Google Scholar] [CrossRef]
- Ryu, T.; Okamoto, K.; Ansai, S.; Nakao, M.; Kumar, A.; Iguchi, T.; Ogino, Y. Gene duplication of androgen receptor as an evolutionary driving force underlying the diversity of sexual characteristics in teleost fishes. Zool. Sci. 2024, 41, 68–76. [Google Scholar] [CrossRef]
- Katemo Manda, B.; Snoeks, J.; Decru, E.; Bills, R.; Vreven, E. Enteromius thespesios (Teleostei: Cyprinidae): A new minnow species with remarkable sexual dimorphism from the south-eastern part of the upper Congo River. J. Fish Biol. 2020, 96, 1160–1175. [Google Scholar] [CrossRef]
- Gunawickrama, K.B. Intraspecific variation in morphology and sexual dimorphism in Puntius singhala (Teleostei: Cyprinidae). Ceylon J. Sci. Biol. Sci. 2009, 37, 167–175. [Google Scholar] [CrossRef][Green Version]
- Ahnelt, H.; Keckeis, H. Breeding tubercles and spawning behavior in Chondrostoma nasus (Telesotei: Cyprinidae): A correlation? Ichthyol. Explor. Freshw. 1994, 5, 321–330. [Google Scholar]
- Chen, X.Y.; Arratia, G. Breeding tubercles of Phoxinus (Teleostei: Cyprinidae): Morphology, distribution, and phylogenetic implications. J. Morphol. 1996, 228, 127–144. [Google Scholar] [CrossRef]
- Chacko, J.J.; Sekharan, N.M. Sexual dimorphism in structures, size and shape of the cyprinid Nilgiri melon barb, Haludaria fasciata. Fish. Aquat. Life 2022, 30, 138–148. [Google Scholar] [CrossRef]



| Species | Voucher Deposit | Geographic Origin | Number and Size Males (mm SL) | Number and Size Females (mm SL) |
|---|---|---|---|---|
| Leptobotia bellacauda | SNHM 20160220-20160225, ZRC 54802, IAPG A8651-8655 | PR China, Anhui prov., Qiupu River | 4 (46.8–71.7) | 9 (47.6–87.6) |
| IAPG A10062 | PR China, Anhui prov., Ningguo River | 1 (64.7) | 0 | |
| Leptobotia guilinensis | IAPG A8861-8883 | PR China, Guangxi prov., Guilin market | 13 (58.6–74.4) | 10 (64.6–80.4) |
| Leptobotia microphthalma | IAPG A8536-8543 | PR China, Sichuan prov., Lueshan market | 4 (69.0–73.7) | 5 (66.2–80.0) |
| IAPG A5284 | PR China, Guangxi prov., Li River | 0 | 1 (70.7) | |
| IAPG A9124 | PR China, Sichuan prov., Jiu Ling River | 1 (76.3) | 0 | |
| Leptobotia tchangi | IAPG A9167-9172 | PR China, Zhejiang prov., Xinchang county | 2 (62.6–65.1) | 5 (65.1–111.8) |
| IAPG A10066-10068 | PR China, Anhui prov., Jixi county | 0 | 1 (48.3) | |
| Leptobotia taeniops | IAPG A8544-8548 | PR China, Sichuan prov., Luzhou market | 0 | 6 (84.5–91.0) |
| IAPG A8549 | PR China, Sichuan prov., Yibingmarket | 1 (73.5) | 0 | |
| IAPG A8562-8566, A8797-8798 | PR China, Jiangxi prov., Nanchang market | 4 (68.3–100.3) | 3 (75.1–81.6) | |
| Parabotia fasciatus | IAPG A8471-8500, A8780-8793 | PR China, Guangxi prov., Nanning market | 8 (95.7–117.5) | 13 (89.2–151.6) |
| Sinibotia pulchra | IAPG A8889-8891, A8900 | PR China, Guangxi prov., Li River | 1 (71.6) | 3 (65.5–73.8) |
| IAPG A8615-8631 | PR China, Fujian prov., Min River | 8 (54.7–78.4) | 4 (56.2–71.7) | |
| IAPG A9102-9106 | PR China, Guangxi prov., Nanning market | 3 (65.5–75.1) | 2 (62.6–70.0) | |
| IAPG A121-123 | Ornamental fish trade | 1 (78.0) | 2 (71.8–76.5) | |
| Sinibotia robusta | IAPG A8837-8860 | PR China, Guangxi prov., Guilin market | 15 (51.4–61.7) | 11 (54.4–66.2) |
| Species | Sex (n) | Pectoral Fin Length (In %SL) | Ventral Fin Length (In %SL) | Anal Fin Depth (In %SL) | Tubercles on Pectoral Fin |
|---|---|---|---|---|---|
| Leptobotia bellacauda | male (5) | 15.0–15.9 | – | – | yes |
| female (9) | 10.7–12.9 | – | – | no | |
| Leptobotia guilinensis | male (13) | 12.5–17.4 | 11.0–14.6 | – | – |
| female (10) | 10.7–12.3 | 10.1–11.8 | – | – | |
| Leptobotia microphthalma | male (5) | 15.9–17.6 | – | 15.4–16.8 | yes |
| female (6) | 12.9–13.9 | – | 14.0–15.3 | no | |
| Leptobotia taeniops | male (5) | 18.7–19.8 | – | – | – |
| female (8) | 14.9–18.2 | – | – | – | |
| Leptobotia tchangi | male * (2) | 19.3–21.2 | 16.1–17.5 | – | yes |
| female (6) | 14.8–17.2 | 13.6–15.5 | – | no | |
| Parabotia fasciatus | male (8) | 16.3–17.2 | – | – | – |
| female (13) | 13.4–16.1 | – | – | – | |
| Sinibotia pulchra | male (8) | 16.7–18.1 | – | – | – |
| female (9) | 13.6–16.2 | – | – | – | |
| Sinibotia robusta | male (15) | 21.2–24.8 | – | – | – |
| female (11) | 18.5–21.6 | – | – | – |
| Character | SS Between | SS Within | df Between | df Within | F | p-Value | |
|---|---|---|---|---|---|---|---|
| Leptobotia bellacauda |  | 35.145 | 7.992 | 1 | 12 | 52.770 | 9.95 × 10-6 |
| Leptobotia guilinensis |  | 46.556 | 19.041 | 1 | 21 | 51.346 | 4.59 × 10-7 |
| Leptobotia guilinensis |  | 12.408 | 9.650 | 1 | 19 | 24.430 | 9.04 × 10-5 |
| Leptobotia microphthalma |  | 30.424 | 2.792 | 1 | 9 | 98.072 | 3.88 × 10-6 |
| Leptobotia microphthalma |  | 2.893 | 2.723 | 1 | 9 | 9.563 | 0.0129 |
| Leptobotia taeniops |  | 14.522 | 10.840 | 1 | 11 | 14.736 | 2.75 × 10-3 |
| Leptobotia tchangi |  | 28.167 | 5.733 | 1 | 6 | 29.477 | 1.62 × 10-3 |
| Leptobotia tchangi |  | 5.510 | 3.568 | 1 | 6 | 9.266 | 0.0227 |
| Parabotia fasciatus |  | 14.785 | 7.445 | 1 | 19 | 37.730 | 6.64 × 10-6 |
| Sinibotia pulchra |  | 27.151 | 8,150 | 1 | 15 | 49.965 | 3.82 × 10-6 |
| Sinibotia robusta |  | 34.300 | 20.485 | 1 | 24 | 40.184 | 1.49 × 10-6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bohlen, J.; Dvořák, T.; Šlechtová, V. First Evidence of Secondary Sexual Dimorphism in the Freshwater Fish Family Botiidae: A Newly Recognised Synapomorphy of Loaches (Cypriniformes: Cobitoidea). Fishes 2025, 10, 648. https://doi.org/10.3390/fishes10120648
Bohlen J, Dvořák T, Šlechtová V. First Evidence of Secondary Sexual Dimorphism in the Freshwater Fish Family Botiidae: A Newly Recognised Synapomorphy of Loaches (Cypriniformes: Cobitoidea). Fishes. 2025; 10(12):648. https://doi.org/10.3390/fishes10120648
Chicago/Turabian StyleBohlen, Jörg, Tomáš Dvořák, and Vendula Šlechtová. 2025. "First Evidence of Secondary Sexual Dimorphism in the Freshwater Fish Family Botiidae: A Newly Recognised Synapomorphy of Loaches (Cypriniformes: Cobitoidea)" Fishes 10, no. 12: 648. https://doi.org/10.3390/fishes10120648
APA StyleBohlen, J., Dvořák, T., & Šlechtová, V. (2025). First Evidence of Secondary Sexual Dimorphism in the Freshwater Fish Family Botiidae: A Newly Recognised Synapomorphy of Loaches (Cypriniformes: Cobitoidea). Fishes, 10(12), 648. https://doi.org/10.3390/fishes10120648






