Self-Actuated Paper and Wood Models: Low-Cost Handcrafted Biomimetic Compliant Systems for Research and Teaching
Abstract
:1. Introduction
1.1. Motivation
1.2. Plant Concept Generators
1.2.1. Flower of the Bird of Paradise (Strelitzia reginae)
1.2.2. False Rose of Jericho (Selaginella lepidophylla)
1.2.3. Snap-Trap of the Carnivorous Waterwheel Plant (Aldrovanda vesiculosa)
1.2.4. Blooming of the Lily Flower (Lilium spec.)
1.2.5. Opening of the Legume Seed Pod
1.2.6. Snap-Trap of the Carnivorous Venus Flytrap (Dionaea muscipula)
2. Materials and Methods
2.1. Paper-Based Models
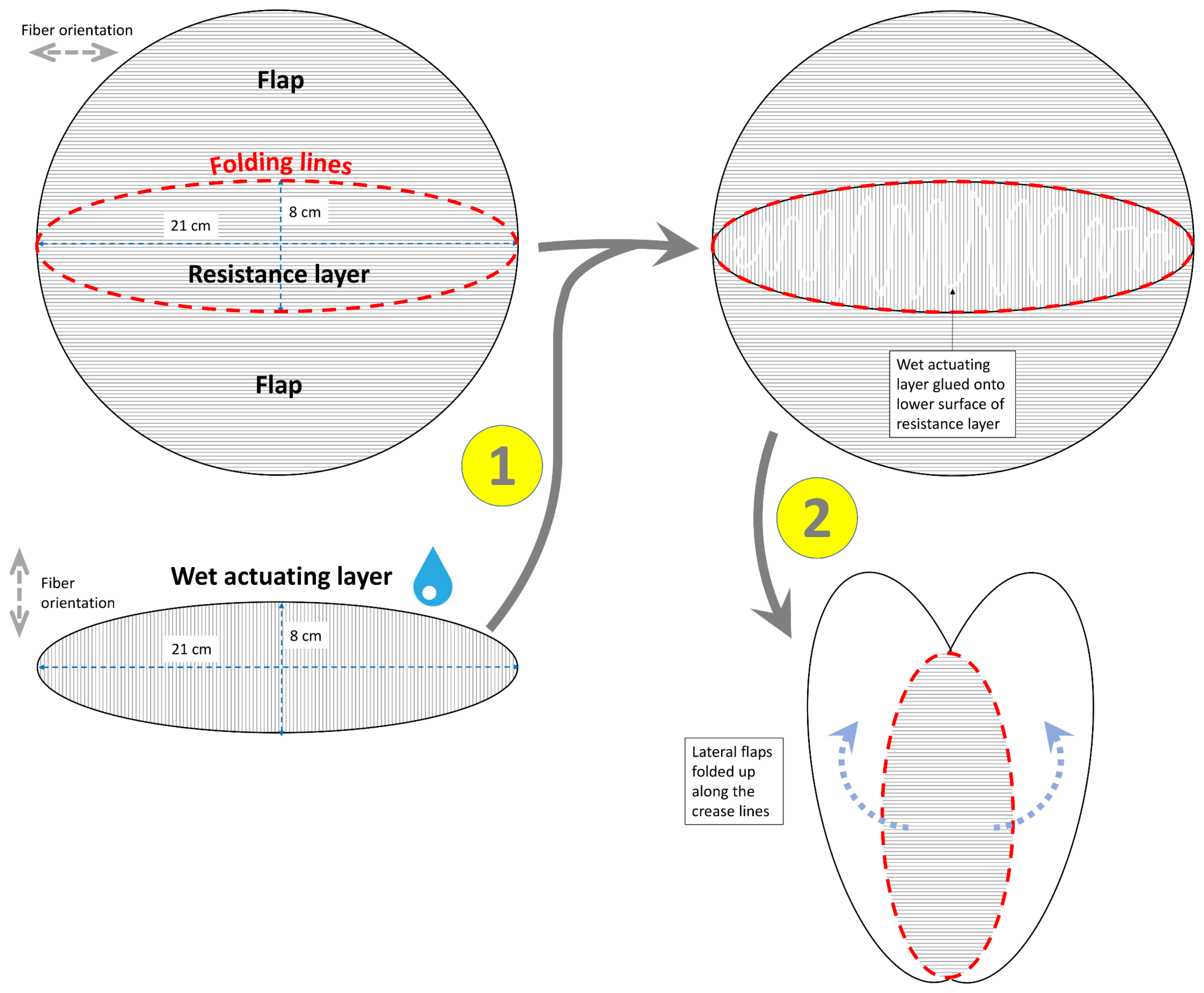
2.1.1. Flower of the Bird of Paradise (S. reginae)
2.1.2. False Rose of Jericho (S. lepidophylla)
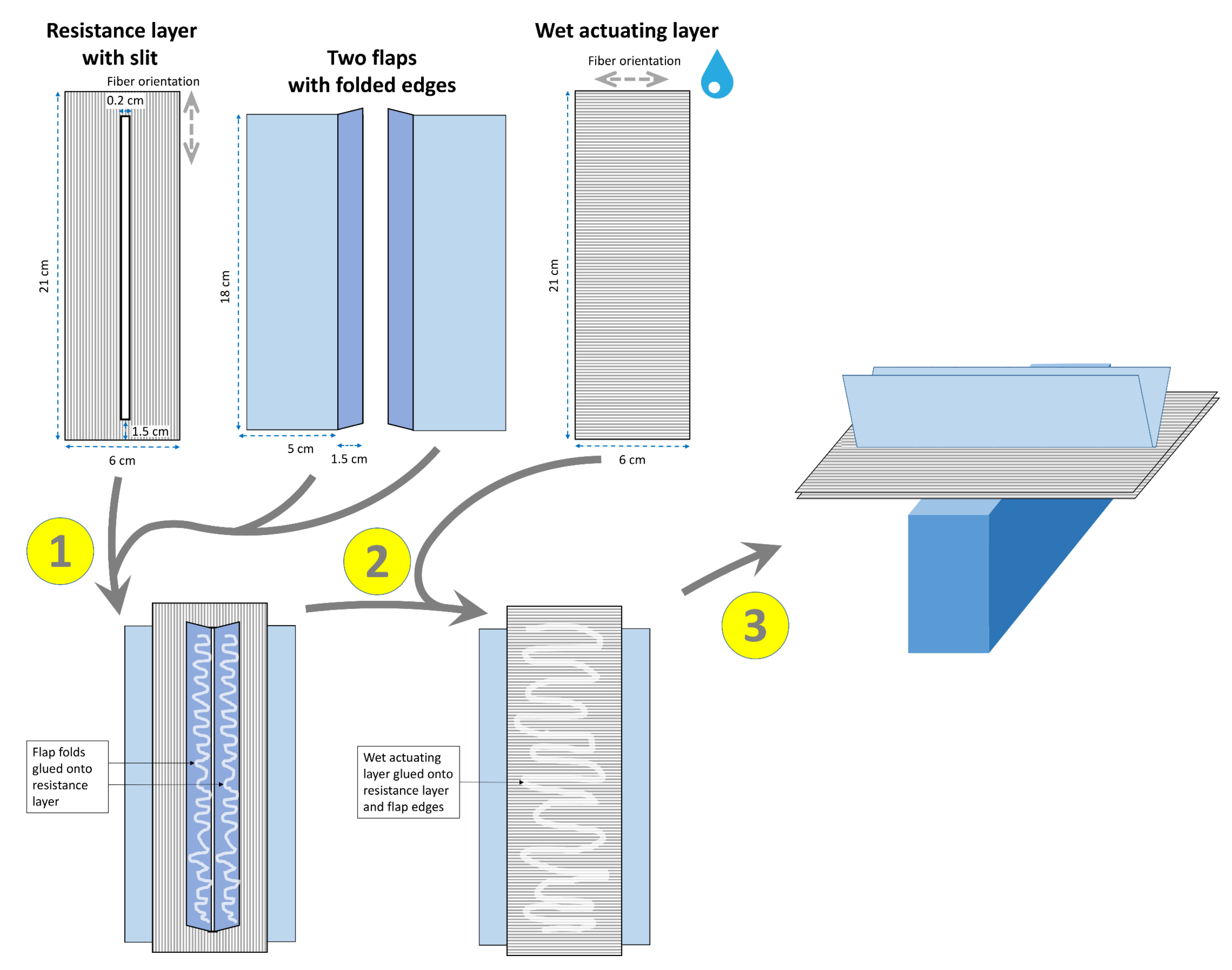
2.1.3. Snap-Trap of the Carnivorous Waterwheel Plant (A. vesiculosa)
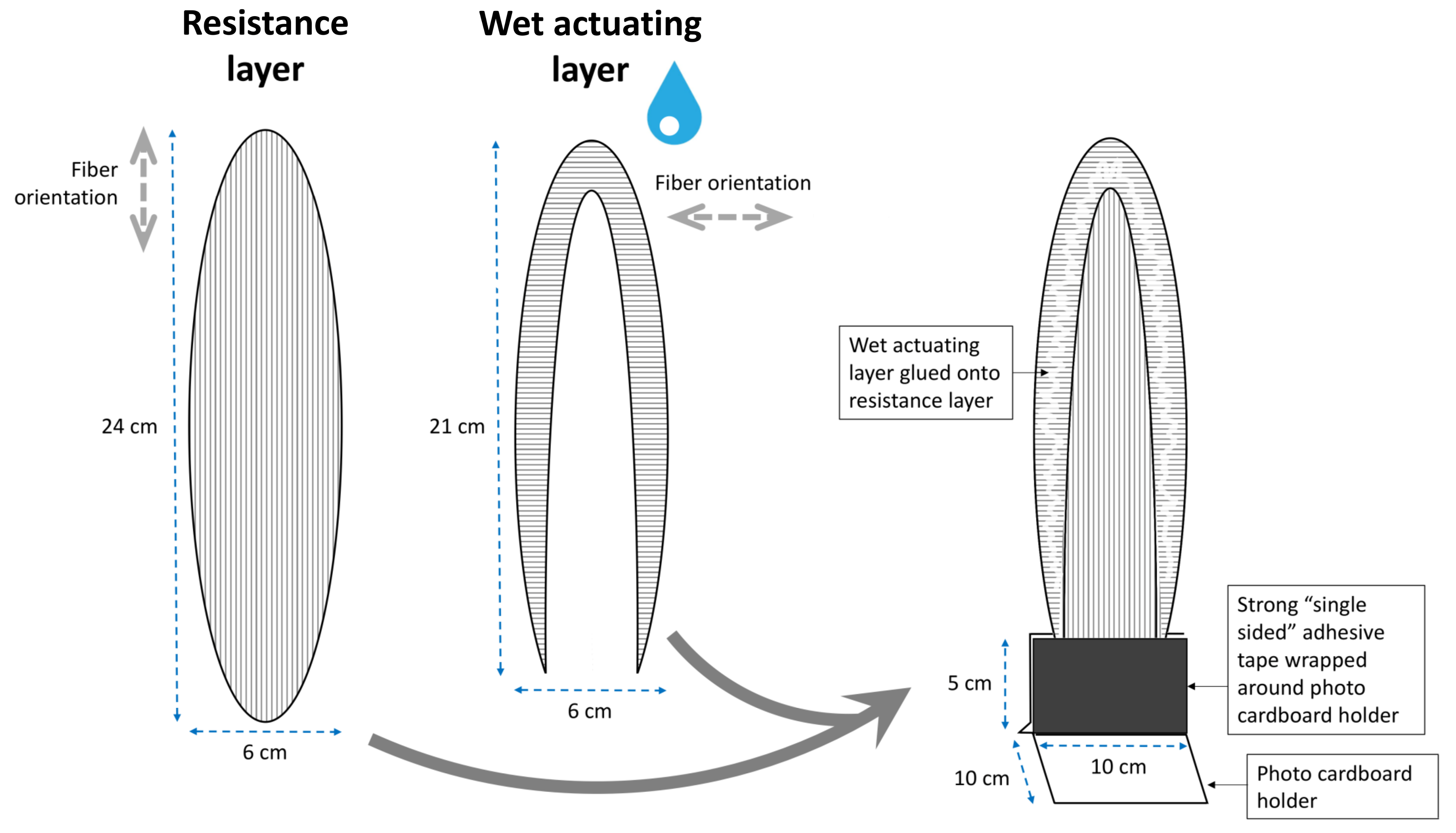
2.1.4. Blooming of the Lily Flower (L. Spec.)
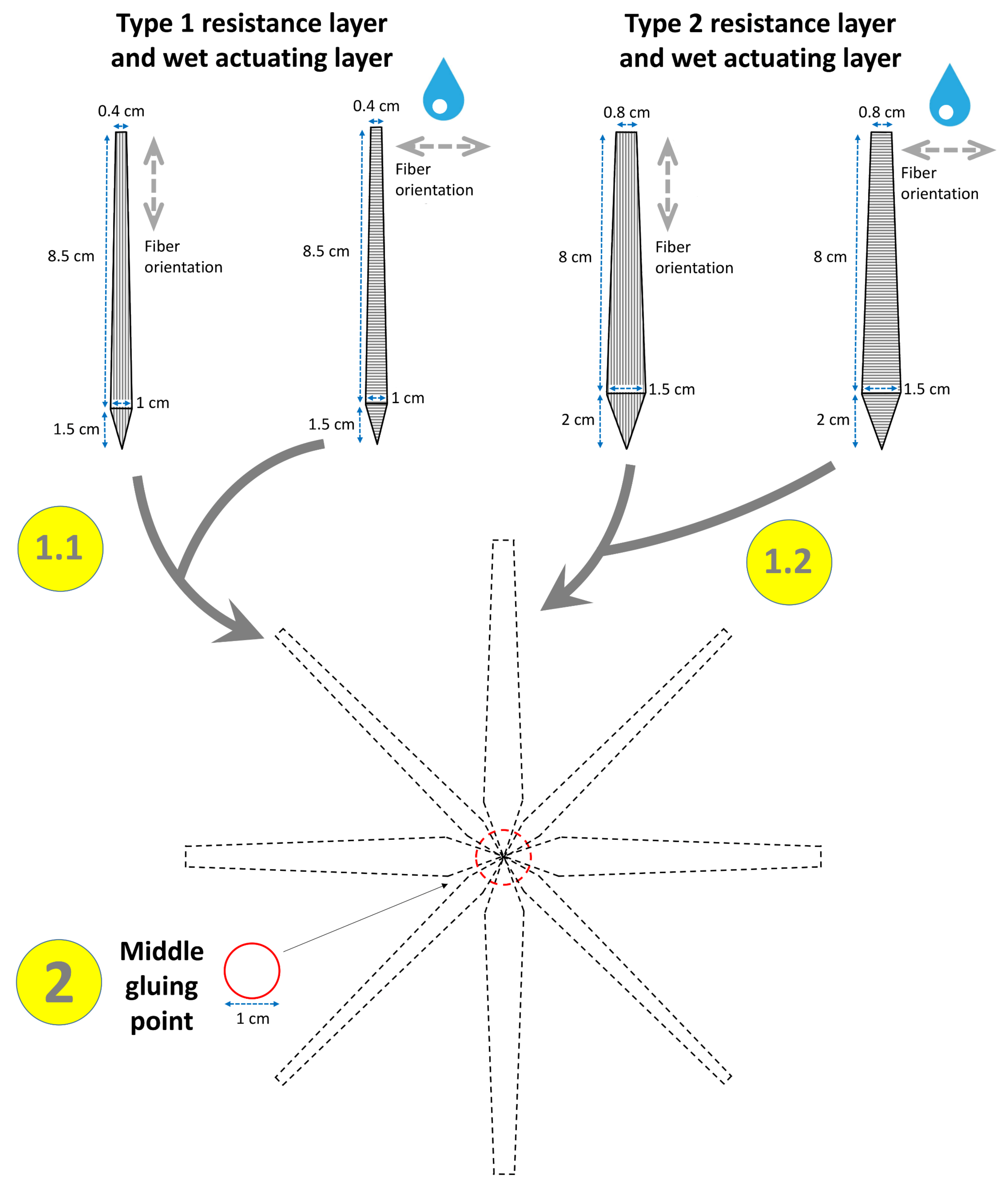
2.1.5. Opening of the Legume
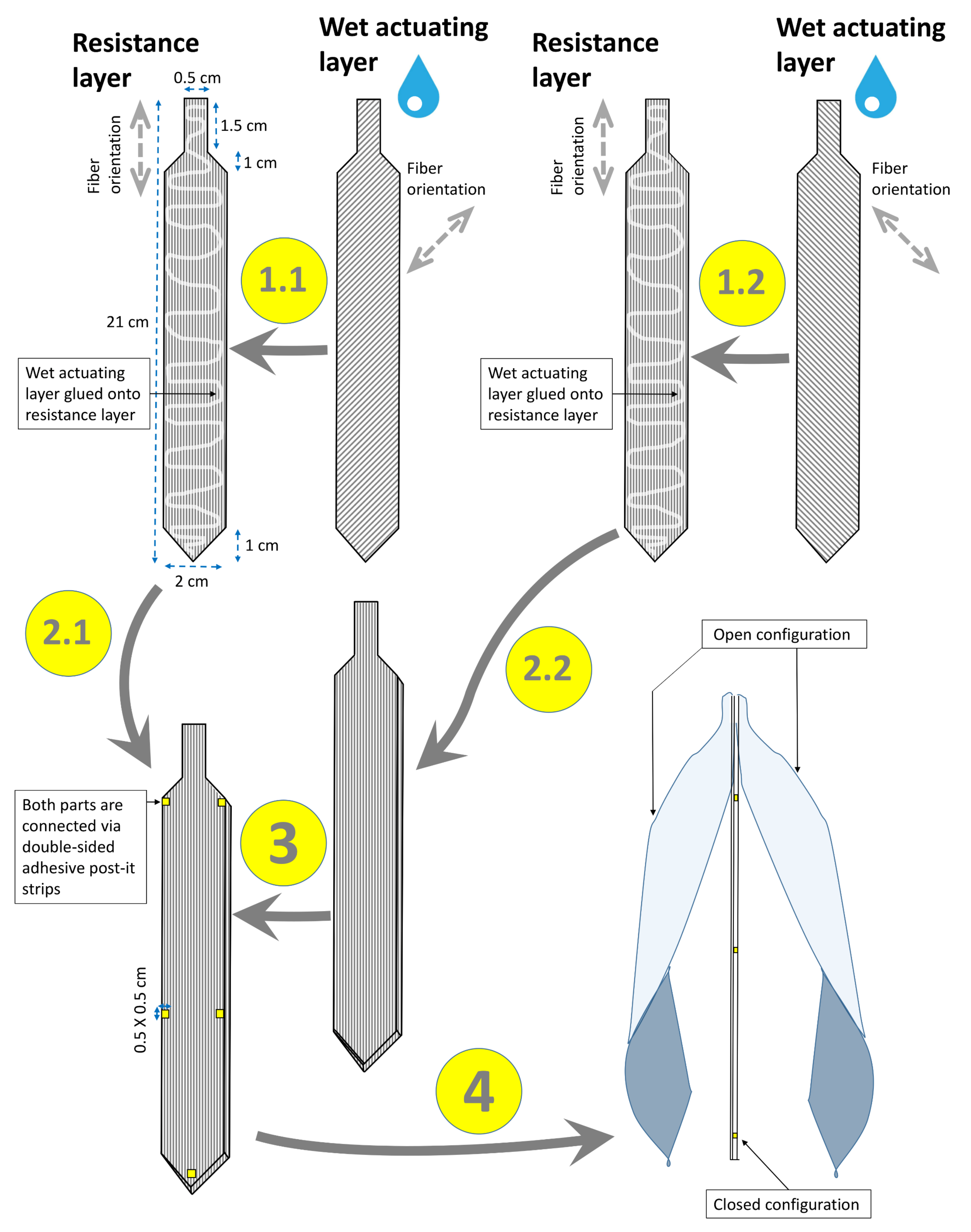
2.2. Paper-Polymer Model
Snap-Trap of the Carnivorous Venus Flytrap (D. muscipula)
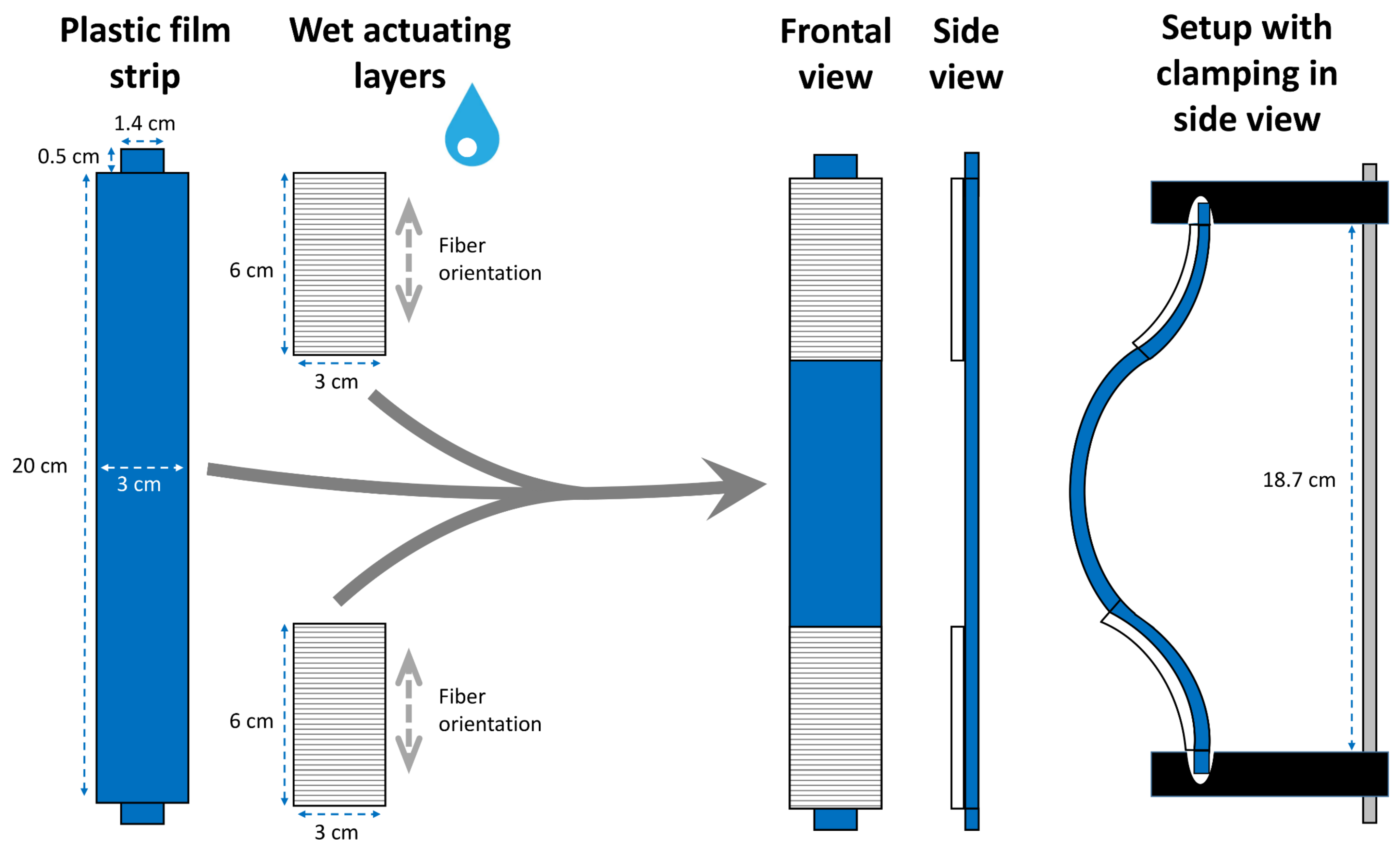
2.3. Wood-Polymer Models
Snap-Trap of the Carnivorous Waterwheel Plant (A. vesiculosa)
3. Results
3.1. Paper-Based Models
3.1.1. Flower of the Bird of Paradise (S. reginae)
3.1.2. False Rose of Jericho (S. lepidophylla)
3.1.3. Snap-Trap of the Carnivorous Waterwheel Plant (A. vesiculosa)
3.1.4. Blooming of the Lily Flower (L. spec.)
3.1.5. Opening of the Legume
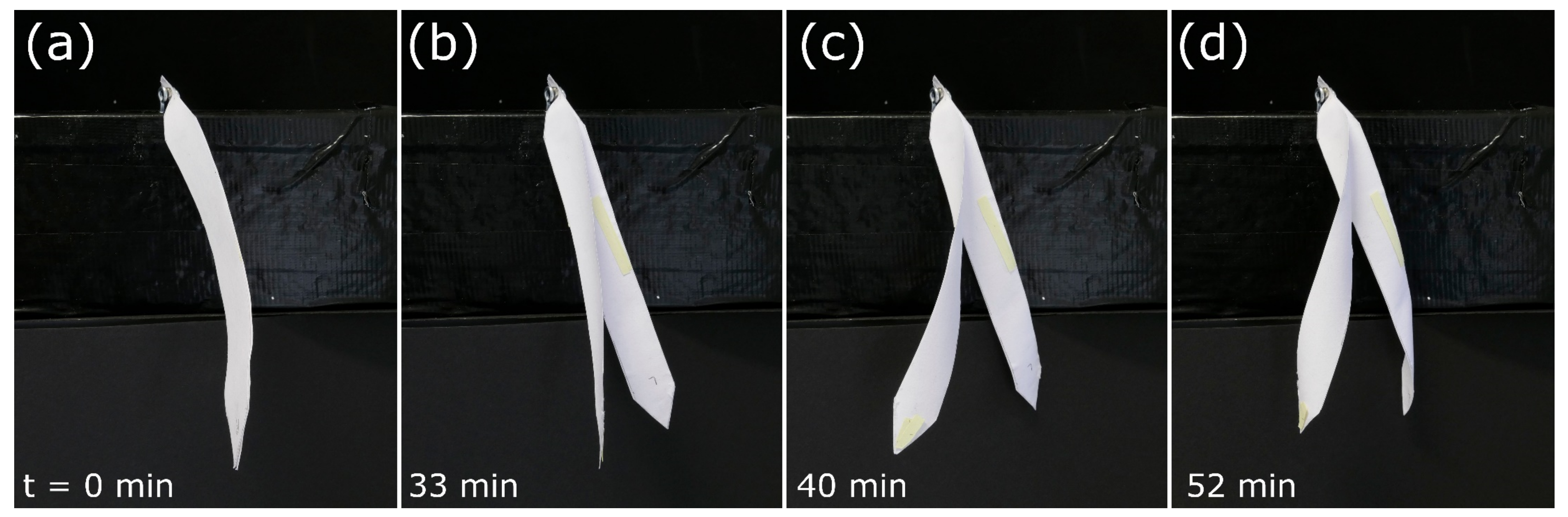
3.2. Paper-Polymer Model
Snap-Trap of the Carnivorous Venus Flytrap (D. muscipula)
3.3. Wood-Polymer Models
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Skotheim, J.; Mahadevan, L. Physical limits and design principles for plant and fungal movements. Science 2005, 308, 1308–1310. [Google Scholar] [CrossRef] [Green Version]
- Dumais, J.; Forterre, Y. “Vegetable dynamicks”: The role of water in plant movements. Annu. Rev. Fluid Mech. 2012, 44, 453–478. [Google Scholar] [CrossRef] [Green Version]
- Forterre, Y. Slow, fast and furious: Understanding the physics of plant movements. J. Exp. Bot. 2013, 64, 4745–4760. [Google Scholar] [CrossRef] [Green Version]
- Poppinga, S.; Masselter, T.; Speck, T. Faster than their prey: New insights into the rapid movements of active carnivorous plants traps. BioEssays 2013, 35, 649–657. [Google Scholar] [CrossRef]
- Weintraub, M. Leaf movements in Mimosa pudica L. N. Phytol. 1952, 50, 357–382. [Google Scholar] [CrossRef]
- Dawson, C.; Vincent, J.F.; Rocca, A.M. How pine cones open. Nature 1997, 390, 668. [Google Scholar] [CrossRef]
- Forterre, Y.; Skotheim, J.M.; Dumais, J.; Mahadevan, L. How the Venus flytrap snaps. Nature 2005, 433, 421–425. [Google Scholar] [CrossRef]
- Sachse, R.; Westermeier, A.; Mylo, M.; Nadasdi, J.; Bischoff, M.; Speck, T.; Poppinga, S. Snapping mechanics of the Venus flytrap (Dionaea muscipula). Proc. Natl. Acad. Sci. USA 2020, 117, 16035–16042. [Google Scholar] [CrossRef] [PubMed]
- Howell, L.L. Compliant Mechanisms; Wiley: New York, NY, USA, 2001. [Google Scholar]
- Poppinga, S.; Körner, A.; Sachse, R.; Born, L.; Westermeier, A.S.; Hesse, L.; Knippers, J.; Bischoff, M.; Gresser, G.T.; Speck, T. Compliant mechanisms in plants and architecture. In Biomimetic Research for Architecture and Building Construction: Biological Design and Integrative Structures; Knippers, J., Speck, T., Nickel, K., Eds.; Springer: Berlin/Heidelberg, Germany, 2016; pp. 169–193. [Google Scholar]
- Burgert, I.; Fratzl, P. Actuation systems in plants as prototypes for bioinspired devices. Philos. Trans. R. Soc. A 2009, 28, 1541–1557. [Google Scholar] [CrossRef] [PubMed]
- Schleicher, S.; Lienhard, J.; Poppinga, S.; Speck, T.; Knippers, J. A methodology for transferring principles of plant movements to elastic systems in architecture. Comput. Aided Des. 2015, 60, 105–117. [Google Scholar] [CrossRef]
- Li, S.; Wang, K.W. Plant-inspired adaptive structures and materials for morphing and actuation: A review. Bioinsp. Biomim. 2017, 12, 011001. [Google Scholar] [CrossRef] [PubMed]
- Mathijsen, D. The rise of compliant mechanisms: composites’ role in enabling an engineering revolution. Reinf. Plast. 2020, 64, 254–260. [Google Scholar] [CrossRef]
- Guo, Q.; Dai, E.; Han, X.; Xie, S.; Chao, E.; Chen, Z. Fast nastic motion of plants and bioinspired structures. J. R. Soc. Interface 2015, 12, 20150598. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Quian, X.; Zhao, Y.; Alsaid, Y.; Wang, X.; Hua, M.; Galy, T.; Gopalakrishna, H.; Yang, Y.; Cui, J.; Liu, N.; et al. Artificial phototropism for omnidirectional tracking and harvesting of light. Nat. Nanotechnol. 2019, 14, 1048–1055. [Google Scholar] [CrossRef]
- Harlow, W.M.; Côté, W.A.; Day, A.C. The opening mechanism of pine cone scales. J. For. 1964, 62, 538–540. [Google Scholar] [CrossRef]
- Reyssat, E.; Mahadevan, L. Hygromorphs: From pine cones to biomimetic bilayers. J. R. Soc. Interface 2009, 6, 951–957. [Google Scholar] [CrossRef] [Green Version]
- Ionov, L. Biomimetic hydrogel-based actuating systems. Adv. Funct. Mater. 2013, 23, 4555–4570. [Google Scholar] [CrossRef]
- Stoychev, G.; Guiducci, L.; Turcaud, S.; Dunlop, J.W.C.; Ioniv, L. Hole-programmed superfast multistep folding of hydrogel bilayers. Adv. Funct. Mater. 2016, 26, 7733–7739. [Google Scholar] [CrossRef]
- Le Duigou, A.; Requile, S.; Beaugrand, J.; Scarpa, F.; Castro, M. Natural fibres actuators for smart bio-inspired hygromorph biocomposites. Smart Mater. Struct. 2017, 26, 125009. [Google Scholar] [CrossRef] [Green Version]
- Erb, R.M.; Sander, J.S.; Grisch, R.; Studart, A.R. Self-shaping composites with programmable bioinspired microstructures. Nat. Commun. 2013, 4, 1712. [Google Scholar] [CrossRef] [Green Version]
- Bargardi, F.L.; Le Ferrand, H.; Libanori, R.; Studart, A.R. Bio-inspired self-shaping ceramics. Nat. Commun. 2016, 7, 13912. [Google Scholar] [CrossRef]
- Gladman, A.S.; Matsumoto, E.A.; Nuzzo, R.G.; Mahadevan, L. Biomimetic 4D printing. Nat. Mater. 2016, 15, 413–418. [Google Scholar] [CrossRef]
- Correa, D.; Menges, A. Fused filament fabrication for multi-kinematic-state climate-responsive aperture. In Fabricate 2017; Menges, A., Sheil, B., Glynn, R., Eds.; UCL Press: London, UK, 2017; pp. 190–195. [Google Scholar]
- Joshi, S.; Rawat, K.; Karunakaran, C.; Rajamohan, V.; Mathew, A.T.; Koziol, K.; Thakur, V.K.; Balan, A.S.S. 4D printing of materials for the future: Opportunities and challenges. Appl. Mater. Today 2020, 18, 100490. [Google Scholar] [CrossRef]
- Correa, D.; Poppinga, S.; Mylo, M.D.; Westermeier, A.S.; Bruchmann, B.; Menges, A.; Speck, T. 4D pine scale: Biomimetic 4D printed autonomous scale and flap structures capable of multi-phase movement. Philos. Trans. R. Soc. A 2020, 378, 20190445. [Google Scholar] [CrossRef]
- Poppinga, S.; Correa, D.; Bruchmann, B.; Menges, A.; Speck, T. Plant movements as concept generators for the development of biomimetic compliant mechanisms. Integr. Comp. Biol. 2020, 60, 886–895. [Google Scholar] [CrossRef] [PubMed]
- Poppinga, S.; Lienhard, J.; Schleicher, S.; Speck, O.; Knippers, J.; Speck, T.; Masselter, T. Paradiesvogelblume trifft Architektur—Bionische Innovation für gelenkfreie technische Anwendungen. Prax. Nat. Biol. Sch. 2012, 5, 31–35. [Google Scholar]
- Poppinga, S.; Metzger, A.; Speck, O.; Masselter, T.; Speck, T. Schnappen, schleudern, saugen: Fallenbewegungen fleischfressender Pflanzen. Biol. Unserer Zeit 2013, 43, 352–361. [Google Scholar] [CrossRef]
- Salmén, L.; Boman, R.; Fellers, C.; Htun, M. The implications of fiber and sheet structure for the hygroexpansivity of paper. Nord. Pulp. Paper. Res. J. 1987, 4, 127–131. [Google Scholar] [CrossRef]
- Skaar, C. Wood-Water Relation; Springer: Berlin/Heidelberg, Germany, 1988. [Google Scholar]
- Brauner, L.; Rau, W. Versuche zur Bewegungsphysiologie der Pflanzen; Pflanzenphysiologische Praktika Band 3; Springer: Berlin/Heidelberg, Germany, 1966. [Google Scholar]
- Rowan, M.K. Bird pollination of Strelitzia. Ostrich 1974, 45, 40. [Google Scholar]
- Kronestedt, E.; Walles, B. Anatomy of the Strelitzia reginae flower (Strelitziaceae). Nord. J. Bot. 1986, 6, 307–320. [Google Scholar] [CrossRef]
- Lienhard, J.; Schleicher, S.; Poppinga, S.; Masselter, T.; Milwich, M.; Speck, T.; Knippers, J. Flectofin: A hingeless flapping mechanism inspired by nature. Bioinspir. Biomim. 2011, 6, 045001. [Google Scholar] [CrossRef]
- Poppinga, S.; Smaij, J.; Westermeier, A.S.; Horstmann, M.; Kruppert, S.; Tollrian, R.; Speck, T. Prey capture analyses in the carnivorous aquatic waterwheel plant (Aldrovanda vesiculosa L., Droseraceae). Sci. Rep. 2019, 9, 18590. [Google Scholar] [CrossRef] [Green Version]
- Lebkuecher, J.G.; Eickmeier, W.G. Physiological benefits of stem curling for resurrection plants in the field. Ecology 1993, 74, 1073–1080. [Google Scholar] [CrossRef]
- Rafsanjani, A.; Brulé, V.; Western, T.L.; Pasini, D. Hydro-responsive curling of the resurrection plant Selaginella lepidophylla. Sci. Rep. 2015, 5, 8064. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ashida, J. Studies on the leaf movement of Aldrovanda vesiculosa L. I. Process and mechanism of the movement. Mem. Coll. Sci. Kyoto Imp. Univ. Ser. B 1934, 9, 141–244. [Google Scholar]
- Westermeier, A.; Sachse, R.; Poppinga, S.; Vögele, P.; Adamec, L.; Speck, T.; Bischoff, M. How the carnivorous waterwheel plant (Aldrovanda vesiculosa) snaps. Proc. Roy. Soc. B 2018, 285, 20180012. [Google Scholar] [CrossRef] [Green Version]
- Poppinga, S.; Joyeux, M. Different mechanics of snap-trapping in the two closely related carnivorous plants Dionaea muscipula and Aldrovanda vesiculosa. Phys. Rev. E 2011, 84, 041928. [Google Scholar] [CrossRef] [Green Version]
- Körner, A.; Born, L.; Mader, A.; Sachse, R.; Saffarian, S.; Westermeier, A.S.; Poppinga, S.; Bischoff, M.; Gresser, G.T.; Milwich, M.; et al. Flectofold—A biomimetic compliant shading device for complex free form facades. Smart Mater. Struct. 2018, 27, 017001. [Google Scholar] [CrossRef]
- Liang, H.; Mahadevan, L. Growth, geometry, and mechanics of a blooming lily. Proc. Natl. Acad. Sci. USA 2011, 108, 5516–5521. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ridley, H. The Dispersal of Fruits throughout the World; L. Reeve & Co., LTD: Ashford, UK, 1930. [Google Scholar]
- Armon, S.; Efrati, E.; Kupferman, R.; Sharon, E. Geometry and mechanics in the opening of chiral seed pods. Science 2011, 333, 1726–1730. [Google Scholar] [CrossRef] [Green Version]
- Harrington, M.J.; Razghandi, K.; Ditsch, F.; Guiducci, L.; Rüggeberg, M.; Dunlop, J.W.C.; Fratzl, P.; Neinhuis, C.; Burgert, I. Origami-like unfolding of hydro-actuated ice plant seed capsules. Nat. Commun. 2011, 2, 337. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Melzer, B.; Steinbrecher, T.; Seidel, R.; Kraft, O.; Schwaiger, R.; Speck, T. The attachment strategy of English ivy: A complex mechanism acting on several hierarchical levels. J. R. Soc. Interface 2010, 7, 1383–1389. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abraham, Y.; Tamburu, C.; Klein, E.; Dunlop, J.W.C.; Fratzl, P.; Raviv, U.; Elbaum, R. Tilted cellulose arrangement as a novel mechanism for hygroscopic coiling in the stork’s bill awn. J. R. Soc. Interface 2012, 9, 640–647. [Google Scholar] [CrossRef] [Green Version]










Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poppinga, S.; Schenck, P.; Speck, O.; Speck, T.; Bruchmann, B.; Masselter, T. Self-Actuated Paper and Wood Models: Low-Cost Handcrafted Biomimetic Compliant Systems for Research and Teaching. Biomimetics 2021, 6, 42. https://doi.org/10.3390/biomimetics6030042
Poppinga S, Schenck P, Speck O, Speck T, Bruchmann B, Masselter T. Self-Actuated Paper and Wood Models: Low-Cost Handcrafted Biomimetic Compliant Systems for Research and Teaching. Biomimetics. 2021; 6(3):42. https://doi.org/10.3390/biomimetics6030042
Chicago/Turabian StylePoppinga, Simon, Pablo Schenck, Olga Speck, Thomas Speck, Bernd Bruchmann, and Tom Masselter. 2021. "Self-Actuated Paper and Wood Models: Low-Cost Handcrafted Biomimetic Compliant Systems for Research and Teaching" Biomimetics 6, no. 3: 42. https://doi.org/10.3390/biomimetics6030042
APA StylePoppinga, S., Schenck, P., Speck, O., Speck, T., Bruchmann, B., & Masselter, T. (2021). Self-Actuated Paper and Wood Models: Low-Cost Handcrafted Biomimetic Compliant Systems for Research and Teaching. Biomimetics, 6(3), 42. https://doi.org/10.3390/biomimetics6030042







