Design of a Fixed-Bed Column with Vegetal Biomass and Its Recycling for Cr (VI) Treatment
Abstract
1. Introduction
2. Materials and Methods
2.1. E. Crassipes Acquisition and Conditioning
2.2. Preparation of Cr (VI) Solutions
2.3. Obtaining Iron-Modified Biomass
2.4. Development of Fixed-Bed Column
2.5. Process Experiments
2.6. Cr (VI) Measurement
2.7. Elution Assays with EDTA
2.8. Biomass Characterization
2.9. Model Evaluation
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Taka, A.L.; Klink, M.J.; Mbianda, X.Y.; Naidoo, E.B. Chitosan nanocomposites for water treatment by fixed-bed continuous flow column adsorption: A review. Carbohydr. Polym. 2020, 255, 117398. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Liu, Z.; Jiang, Y.; Li, F.; Xue, B.; Dong, Z.; Ding, M.; Chen, R.; Yang, Q.; An, T.; et al. Micro-structure, surface properties and adsorption capacity of ball-milled cellulosic biomass derived biochar based mineral composites synthesized via carbon-bed pyrolysis. Appl. Clay Sci. 2020, 199, 105877. [Google Scholar] [CrossRef]
- Yoshinaga, M.; Ninomiya, H.; Al Hossain, M.A.; Sudo, M.; Akhand, A.A.; Ahsan, N.; Alim, A.; Khalequzzaman, M.; Iida, M.; Yajima, I.; et al. A comprehensive study including monitoring, assessment of health effects and development of a remediation method for chromium pollution. Chemosphere 2018, 201, 667–675. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, L.M.; Cunha-Oliveira, T.; Sobral, M.C.; Abreu, P.L.; Alpoim, M.C.; Urbano, A.M. Impact of carcinogenic chromium on the cellular response to proteotoxic stress. Int. J. Mol. Sci. 2019, 20, 4901. [Google Scholar] [CrossRef] [PubMed]
- Sayago, U.F.C. Design of a sustainable development process between phytoremediation and production of bioethanol with Eichhornia crassipes. Environ. Monit. Assess. 2019, 191, 221. [Google Scholar] [CrossRef] [PubMed]
- Bhatti, H.N.; Mahmood, Z.; Kausar, A.; Yakout, S.M.; Shair, O.H.; Iqbal, M. Biocomposites of polypyrrole, polyaniline and sodium alginate with cellulosic biomass: Adsorption-desorption, kinetics and thermodynamic studies for the removal of 2, 4-dichlorophenol. Int. J. Biol. Macromol. 2020, 153, 146–157. [Google Scholar] [CrossRef] [PubMed]
- Carreño-Sayago, U.F.; Rodríguez-Parra, C. Eichhornia crassipes (Mart.) Solms: An integrated phytoremediation and bioenergy system. Rev. Chapingo Ser. Cienc. For. 2019, 25, 399–411. [Google Scholar] [CrossRef]
- Sayago, U.F.C. Design and development of a biotreatment of E. crassipes for the decontamination of water with Chromium (VI). Sci. Rep. 2021, 11, 9326. [Google Scholar] [CrossRef] [PubMed]
- Sayago, U.F.C.; Castro, Y.P.; Rivera, L.R.C.; Mariaca, A.G. Estimation of equilibrium times and maximum capacity of adsorption of heavy metals by E. crassipes. Environ. Monit. Assess. 2019, 192, 141. [Google Scholar] [CrossRef] [PubMed]
- Yi, Z.J.; Yao, J.; Chen, H.L.; Wang, F.; Yuan, Z.M.; Liu, X. Uranium biosorption from aqueous solution onto Eichhornia crassipes. J. Environ. Radioact. 2016, 154, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Al-Ghouti, M.A.; Al-Absi, R.S. Mechanistic understanding of the adsorption and thermodynamic aspects of cationic methylene blue dye onto cellulosic olive stones biomass from wastewater. Sci. Rep. 2020, 10, 15928. [Google Scholar] [CrossRef]
- Abdulrazak, S.; Hussaini, K.; Sani, H.M. Evaluation of removal efficiency of heavy metals by low-cost activated carbon prepared from African palm fruit. Appl. Water Sci. 2017, 7, 3151–3155. [Google Scholar] [CrossRef]
- Deng, L.; Geng, M.; Zhu, D.; Zhou, W.; Langdon, A.; Wu, H.; Yu, Y.; Zhu, Z.; Wang, Y. Effect of chemical and biological degumming on the adsorption of heavy metal by cellulose xanthogenate prepared from Eichhornia crassipes. Bioresour. Technol. 2012, 107, 41–45. [Google Scholar] [CrossRef]
- Zhou, Y.; Fu, S.; Zhang, L.; Zhan, H.; Levit, M.V. Use of carboxylated cellulose nanofibrils-filled magnetic chitosan hydrogel beads as adsorbents for Pb (II). Carbohydr. Polym. 2014, 101, 75–82. [Google Scholar] [CrossRef]
- Kumar, V.; Singh, J.; Kumar, P. Regression models for removal of heavy metals by water hyacinth (Eichhornia crassipes) from wastewater of pulp and paper processing industry. Environ. Sustain. 2020, 3, 35–44. [Google Scholar] [CrossRef]
- Purnomo, R.O. Delignifikasi Adsorben Dari Batang Eceng Gondok (Eichhornia crassipes) Untuk Adsorpsi Nikel (II) Menggunakan Metode Batch. Diploma Thesis, Universitas Negeri Malang, Malang, Indonesia, 2018. [Google Scholar]
- Gaballah, M.S.; Ismail, K.; Aboagye, D.; Ismail, M.M.; Sobhi, M.; Stefanakis, A.I. Effect of design and operational parameters on nutrients and heavy metal removal in pilot floating treatment wetlands with Eichhornia Crassipes treating polluted lake water. Environ. Sci. Pollut. Res. 2021, 28, 25664–25678. [Google Scholar] [CrossRef]
- Mitra, T.; Singha, B.; Bar, N.; Das, S.K. Removal of Pb (II) ions from aqueous solution using water hyacinth root by fixed-bed column and ANN modeling. J. Hazard. Mater. 2014, 273, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Nag, S.; Mondal, A.; Mishra, U.; Bar, N.; Das, S.K. Removal of chromium (VI) from aqueous solutions using rubber leaf powder: Batch and column studies. Desalination Water Treat. 2016, 57, 16927–16942. [Google Scholar] [CrossRef]
- Aydına, S.; Nura, H.M.; Traorea, A.M.; Yıldırımb, E.; Emikb, S. Fixed bed column adsorption of vanadium from water using amino-functional polymeric adsorbent. J. Desalination Water Treat. 2021, 144, 155–159. [Google Scholar] [CrossRef]
- Worch, E. Adsorption Technology in Water Treatment: Fundamentals, Processes, and Modeling; Walter de Gruyter: Berlin, Germany, 2012. [Google Scholar]
- Benítez, L.T.; Tovar, C.T.; Bolívar, W.M.; Ortiz, Á.V. Estudio de modificación química y física de biomasa (Citrus sinensis y Musa paradisiaca) para la adsorción de metales pesados en solución. Rev. Luna Azul 2014, 39, 124–142. [Google Scholar] [CrossRef]
- Cañizares-Villanueva, R.O. Biosorción de metales pesados mediante el uso de biomasa microbiana. Rev. Latinoam. Microbiol. -Mex. 2000, 42, 131–143. [Google Scholar]
- Shim, Y.S.; Kim, Y.K.; Kong, S.H.; Rhee, S.W.; Lee, W.K. The adsorption characteristics of heavy metals by various particle sizes of MSWI bottom ash. Waste Manag. 2003, 23, 851–857. [Google Scholar] [CrossRef]
- Júnior, W.N.; Silva, M.G.C.; Vieira, M.G.A. Competitive fixed-bed biosorption of Ag (I) and Cu (II) ions on Sargassum filipendula seaweed waste. J. Water Process Eng. 2020, 36, 101294. [Google Scholar] [CrossRef]
- Lin, S.; Yang, H.; Na, Z.; Lin, K. A novel biodegradable arsenic adsorbent by immobilization of iron oxyhydroxide (FeOOH) on the root powder of long-root Eichhornia crassipes. Chemosphere 2018, 192, 258–266. [Google Scholar] [CrossRef]
- Wei, Y.; Fang, Z.; Zheng, L.; Tsang, E.P. Biosynthesized iron nanoparticles in aqueous extracts of Eichhornia crassipes and its mechanism in the hexavalent chromium removal. Appl. Surf. Sci. 2017, 399, 322–329. [Google Scholar] [CrossRef]
- Hokkanen, S.; Repo, E.; Lou, S.; Sillanpää, M. Removal of arsenic (V) by magnetic nanoparticle activated microfibrillated cellulose. Chem. Eng. J. 2014, 260, 886–894. [Google Scholar] [CrossRef]
- Dhankhar, R.; Hooda, A. Fungal biosorption–an alternative to meet the challenges of heavy metal pollution in aqueous solutions. Environ. Technol. 2011, 32, 467–491. [Google Scholar] [CrossRef]
- Park, D.; Yun, Y.S.; Park, J.M. Mechanisms of the removal of hexavalent chromium by biomaterials or biomaterial-based activated carbons. J. Hazard. Mater. 2006, 137, 1254–1257. [Google Scholar] [CrossRef]
- Park, D.; Yun, Y.S.; Jo, J.H.; Park, J.M. Mechanism of hexavalent chromium removal by dead fungal biomass of Aspergillus niger. Water Res. 2005, 39, 533–540. [Google Scholar] [CrossRef]
- Chatterjee, A.; Abraham, J. Desorption of heavy metals from metal loaded sorbents and e-wastes: A review. Biotechnol. Lett. 2019, 41, 319–333. [Google Scholar] [CrossRef]
- Chen, S.; Yue, Q.; Gao, B.; Li, Q.; Xu, X.; Fu, K. Adsorption of hexavalent chromium from aqueous solution by modified corn stalk: A fixed-bed column study. Bioresour. Technol. 2012, 113, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Yang, L.; Yang, Z.; Yang, H.; Li, A.; Cheng, R. Preparation of chitosan/poly (acrylic acid) magnetic composite microspheres and applications in the removal of copper (II) ions from aqueous solutions. J. Hazard. Mater. 2012, 229, 371–380. [Google Scholar] [CrossRef] [PubMed]
- Anirudhan, T.S.; Shainy, F. Adsorption behaviour of 2-mercaptobenzamide modified itaconic acid-grafted-magnetite nanocellulose composite for cadmium (II) from aqueous solutions. J. Ind. Eng. Chem. 2015, 32, 157–166. [Google Scholar] [CrossRef]
- Golie, W.M.; Upadhyayula, S. Continuous fixed-bed column study for the removal of nitrate from water using chitosan/alumina composite. J. Water Process Eng. 2016, 12, 58–65. [Google Scholar] [CrossRef]
- Tejada-Tovar, C.N.; Villabona-Ortíz, A.; Alvarez-Bajaire, G.; Granados-Conde, C. Influencia de la altura del lecho sobre el comportamiento dinámico de columna de lecho fijo en la biosorción de mercurio. TecnoLógicas 2019, 20, 71–81. [Google Scholar] [CrossRef][Green Version]
- Singh, J.; Kumar, V.; Kumar, P. Kinetics and prediction modeling of heavy metal phytoremediation from glass industry effluent by water hyacinth (Eichhornia crassipes). Int. J. Environ. Sci. Technol. 2021, 19, 5481–5492. [Google Scholar] [CrossRef]
- Saralegui, A.B.; Willson, V.; Caracciolo, N.; Piol, M.N.; Boeykens, S.P. Macrophyte biomass productivity for heavy metal adsorption. J. Environ. Manag. 2021, 289, 112398. [Google Scholar] [CrossRef]
- Ajmani, A.; Shahnaz, T.; Subbiah, S.; Narayanasamy, S. Hexavalent chromium adsorption on virgin, biochar, and chemically modified carbons prepared from Phanera vahlii fruit biomass: Equilibrium, kinetics, and thermodynamics approach. Environ. Sci. Pollut. Res. 2019, 26, 32137–32150. [Google Scholar] [CrossRef]
- Gupta, S.; Jain, A.K. Biosorption of Ni (II) from aqueous solutions and real industrial wastewater using modified A. barbadensis Miller leaves residue powder in a lab scale continuous fixed bed column. Clean. Eng. Technol. 2021, 5, 100349. [Google Scholar] [CrossRef]
- De Sá Costa, H.P.; da Silva, M.G.C.; Vieira, M.G.A. Fixed bed biosorption and ionic exchange of aluminum by brown algae residual biomass. J. Water Process Eng. 2021, 42, 102117. [Google Scholar] [CrossRef]
- Han, X.; Zhang, Y.; Zheng, C.; Yu, X.; Li, S.; Wei, W. Enhanced Cr (VI) removal from water using a green synthesized nanocrystalline chlorapatite: Physicochemical interpretations and fixed-bed column mathematical model study. Chemosphere 2021, 264, 128421. [Google Scholar] [CrossRef] [PubMed]
- Ghasemabadi, S.M.; Baghdadi, M.; Safari, E.; Ghazban, F. Investigation of continuous adsorption of Pb (II), As (III), Cd (II), and Cr (VI) using a mixture of magnetic graphite oxide and sand as a medium in a fixed-bed column. J. Environ. Chem. Eng. 2018, 6, 4840–4849. [Google Scholar] [CrossRef]
- Jiang, X.; An, Q.D.; Xiao, Z.Y.; Zhai, S.R.; Shi, Z. Versatile core/shell-like alginate@ polyethylenimine composites for efficient removal of multiple heavy metal ions (Pb2+, Cu2+, CrO42−): Batch and fixed-bed studies. Mater. Res. Bull. 2019, 118, 110526. [Google Scholar] [CrossRef]





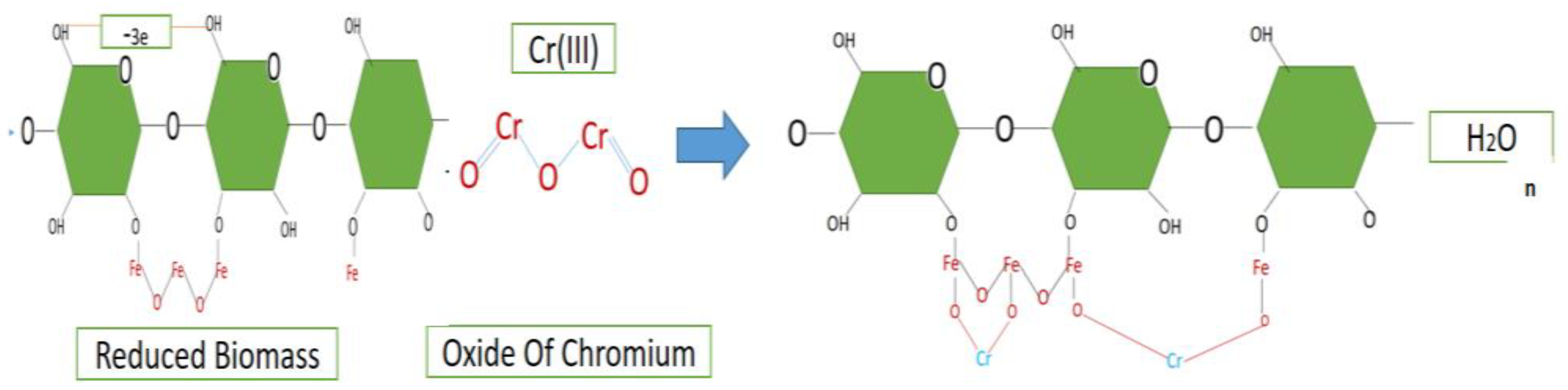



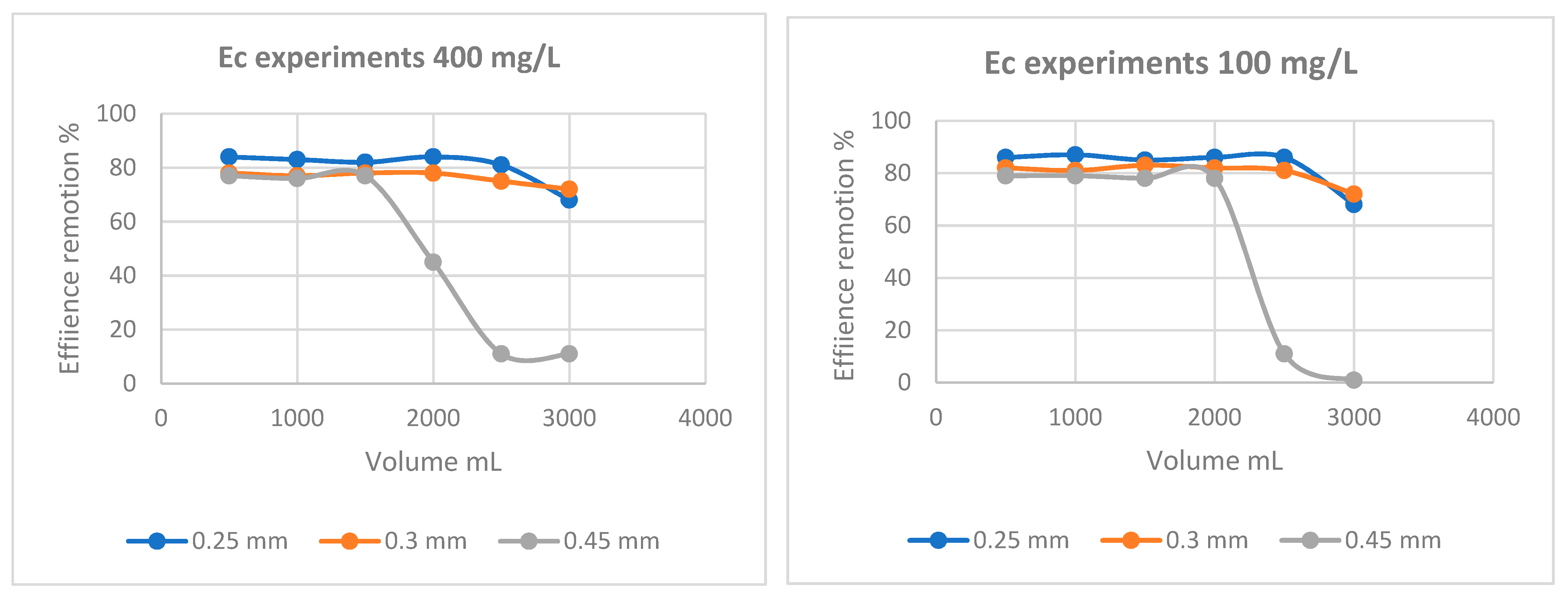

| Thomas model | |
| Yoon model | |
| Bohart model | Tb = |
| Element | Weight | Percentage % |
|---|---|---|
| Carbon | 42.64 | 52.67 |
| Oxygen | 44.15 | 40.94 |
| Sodium | 2.13 | 1.37 |
| Manganese | 0.31 | 0.19 |
| Potassium | 1.45 | 0.55 |
| Calcium | 2.16 | 0.80 |
| Element | Weight | Percentage % |
|---|---|---|
| Carbon | 31.64 | 46.67 |
| Oxygen | 39.15 | 42.34 |
| Iron | 26.13 | 8.37 |
| Element | Weight | Percentages % |
|---|---|---|
| Carbon | 37.6 | 50.67 |
| Oxygen | 30.3 | 38.94 |
| Chromium | 17.13 | 7.4 |
| Iron | 12.2.3 | 7.9 |
| Element | Weight | Percentages % |
|---|---|---|
| Carbon | 53.86 | 60.96 |
| Oxygen | 45.62 | 38.76 |
| Chromium | 0.25 | 0.14 |
| Diameter | Bed Volume (mL) | Bed Mass (g) | Bed Density (g/mL) | Microparticle Volume (mm) | Mass of Particles (mg) | Particle Density (mg/mm) | |
|---|---|---|---|---|---|---|---|
| 0.25 | 80 | 50 | 0.67 | 0.008 | 0.00418 | 1.91 | 0.7 |
| 0.35 | 82 | 50 | 0.69 | 0.014 | 0.018 | 1.26 | 0.5 |
| 0.425 | 84 | 50 | 0.7 | 0.0335 | 0.028 | 0.83 | 0.3 |
| EC | Kth | q (Thomas) | q Design | |
|---|---|---|---|---|
| 400 mg/L | Iron | 0.0537 | 17.9 | 17.6 |
| 400 mg/L | 0.10 | 8.33 | 8 |
| EC | Bohart | Yoon | Thomas | |
|---|---|---|---|---|
| 400 mg/L | K | 0.048 | 0.039 | 0.048 |
| 400 mg/L | R2 | 0.9542 | 0.9444 | 0.954 |
| 100 mg/L | K | 0.047 | 0.039 | 0.047 |
| 100 mg/L | R2 | 0.89 | 0.89 | 0.96 |
| ECFe | Bohart | Yoon | Thomas | |
|---|---|---|---|---|
| 400 mg/L | K | 0.061 | 0.05 | 0.061 |
| 400 mg/L | R2 | 0.90 | 0.9444 | 0.966 |
| 100 mg/L | K | 0.060 | 0.039 | 0.060 |
| 100 mg/L | R2 | 0.89 | 0.89 | 0.94 |
| Reference | Biomass | Contaminate Treated | Recycling | Capacity (mg/g) | Capacity (mg/g) with Equation (11) |
|---|---|---|---|---|---|
| Present article | ECFe | Cr (VI) | EDTA | 16 | 46 |
| Present article | EC | Cr (VI) | EDTA | 11 | 29 |
| [37] | Phanera vahlii | Cr (VI) | NaOH | 30 | 62 |
| [38] | A. barbadensis Miller | Ni (II) | HCl | 14 | 20 |
| [39] | Brown algae | Al (III) | CaCl2 EDTA HNO3 HCl | 12 | 20 30 35 35 |
| [40] | Green synthesized nanocrystalline chlorapatite | Cr (VI) | NaOH | 20 | 35 |
| [41] | Graphite | Cr (VI) | HNO₃ | 20 | 52 |
| [42] | Biochar alginate | Cr (VI) | HCl | 30 | 62 |
| [43] | Pine cone shell | Pb (II) | HCl | 22 | 30 |
| [44] | Brown algae | Cu (II) | CaCl2 | 14 | 54 |
| [45] | Cassava | Cr (VI) | EDTA | 14 | 40 |
| Cycle | |||||
|---|---|---|---|---|---|
| Model | Parameter | 1 | 2 | 3 | 4 |
| Thomas | R2 | 0.99 | 0.99 | 0.98 | 0.97 |
| kt | 0.048 | 0.044 | 0.036 | 0.033 | |
| qt | 14.2 | 9.2 | 3.0 | 2.8 | |
| Bohart | R2 | 0.88 | 0.98 | 0.97 | 0.9 |
| kb | 0.962 | 0.854 | 0.804 | 0.77 | |
| Yoon | R2 | 0.9 | 0.8 | 0.94 | 0.93 |
| ky | 0.944 | 0.88 | 0.80 | 0.75 | |
| Cycle | |||||
|---|---|---|---|---|---|
| Model | Parameter | 1 | 2 | 3 | 4 |
| Thomas | R2 | 0.91 | 0.90 | 0.85 | 0.85 |
| kt | 0.038 | 0.034 | 0.025 | 0.022 | |
| qt | 6.5 | 5.6 | 5.6 | 4.2 | |
| Bohart | R2 | 0.99 | 0.99 | 0.98 | 0.97 |
| kb | 0.862 | 0.854 | 0.77 | 0.66 | |
| Yoon | R2 | 0.88 | 0.9 | 0.91 | 0.92 |
| ky | 0.844 | 0.80 | 0.77 | 0.63 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carreño Sayago, U.F.; Piñeros Castro, Y.; Conde Rivera, L.R. Design of a Fixed-Bed Column with Vegetal Biomass and Its Recycling for Cr (VI) Treatment. Recycling 2022, 7, 71. https://doi.org/10.3390/recycling7050071
Carreño Sayago UF, Piñeros Castro Y, Conde Rivera LR. Design of a Fixed-Bed Column with Vegetal Biomass and Its Recycling for Cr (VI) Treatment. Recycling. 2022; 7(5):71. https://doi.org/10.3390/recycling7050071
Chicago/Turabian StyleCarreño Sayago, Uriel Fernando, Yineth Piñeros Castro, and Laura Rosa Conde Rivera. 2022. "Design of a Fixed-Bed Column with Vegetal Biomass and Its Recycling for Cr (VI) Treatment" Recycling 7, no. 5: 71. https://doi.org/10.3390/recycling7050071
APA StyleCarreño Sayago, U. F., Piñeros Castro, Y., & Conde Rivera, L. R. (2022). Design of a Fixed-Bed Column with Vegetal Biomass and Its Recycling for Cr (VI) Treatment. Recycling, 7(5), 71. https://doi.org/10.3390/recycling7050071






