Release of Trace Elements from Bottom Ash from Hazardous Waste Incinerators
Abstract
:1. Introduction
2. Materials and Methods
2.1. Material Characterization
2.2. Extractions/Leaching Test
2.3. Analysis and Quality Control
3. Results
3.1. Single Extractions
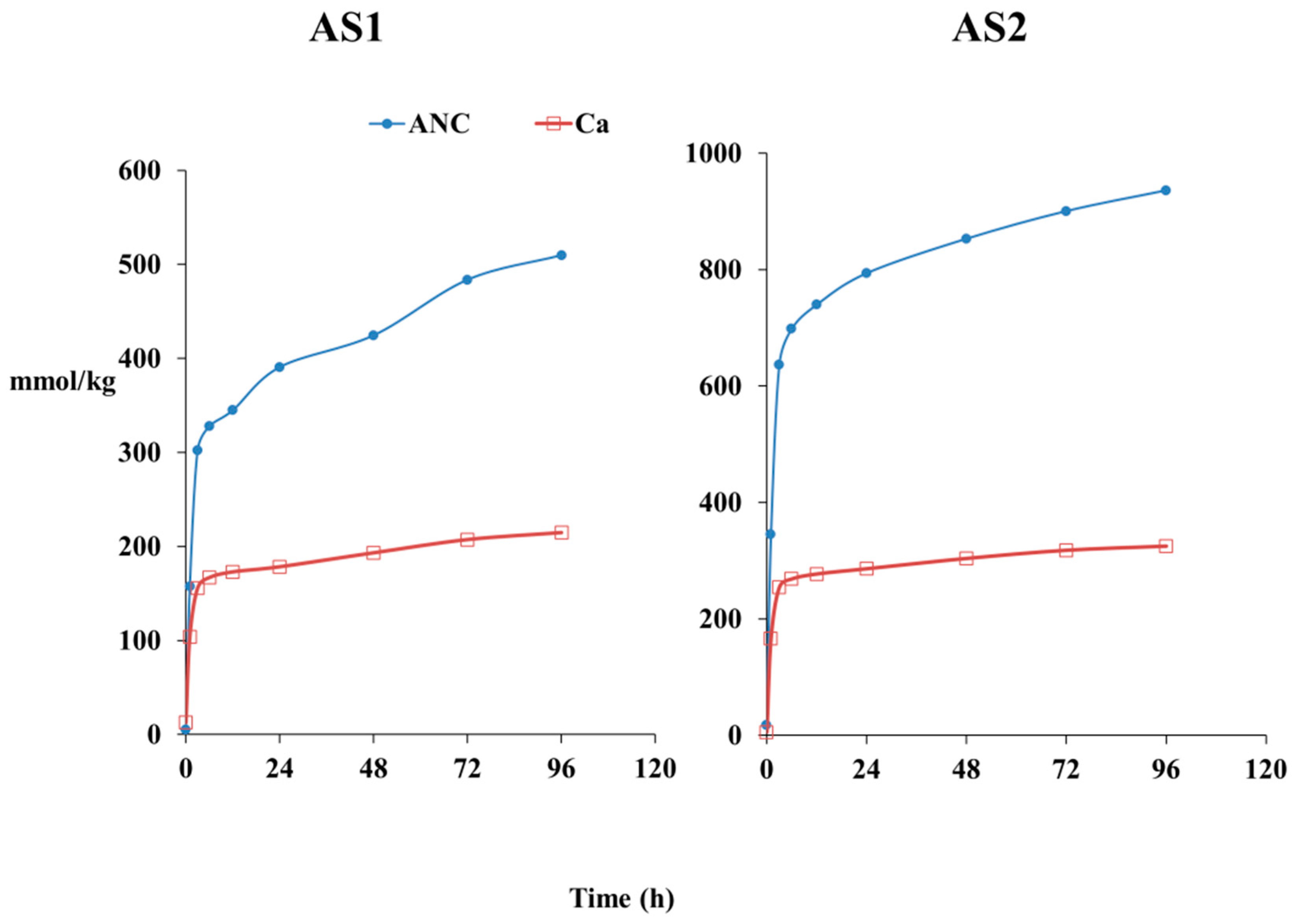
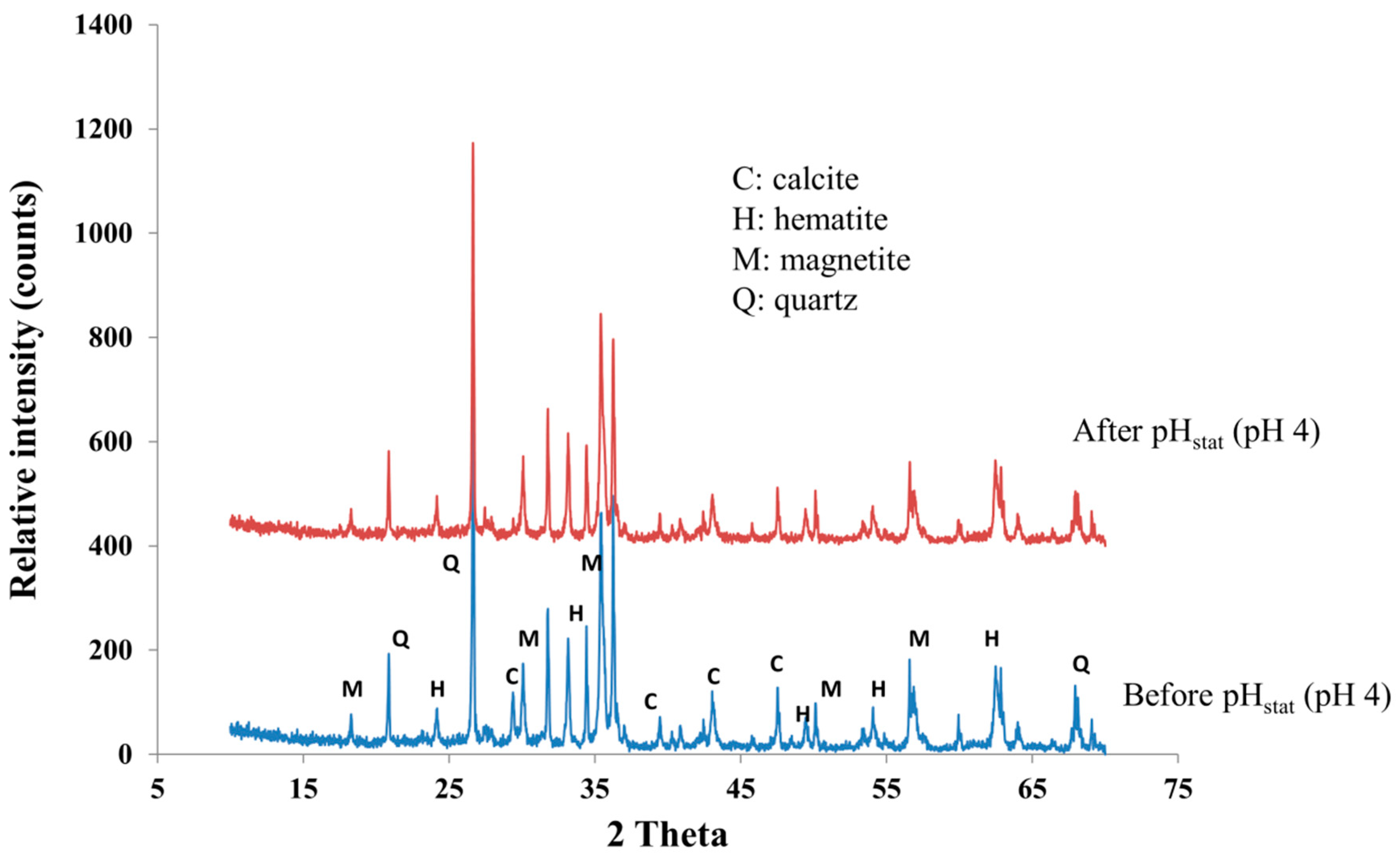
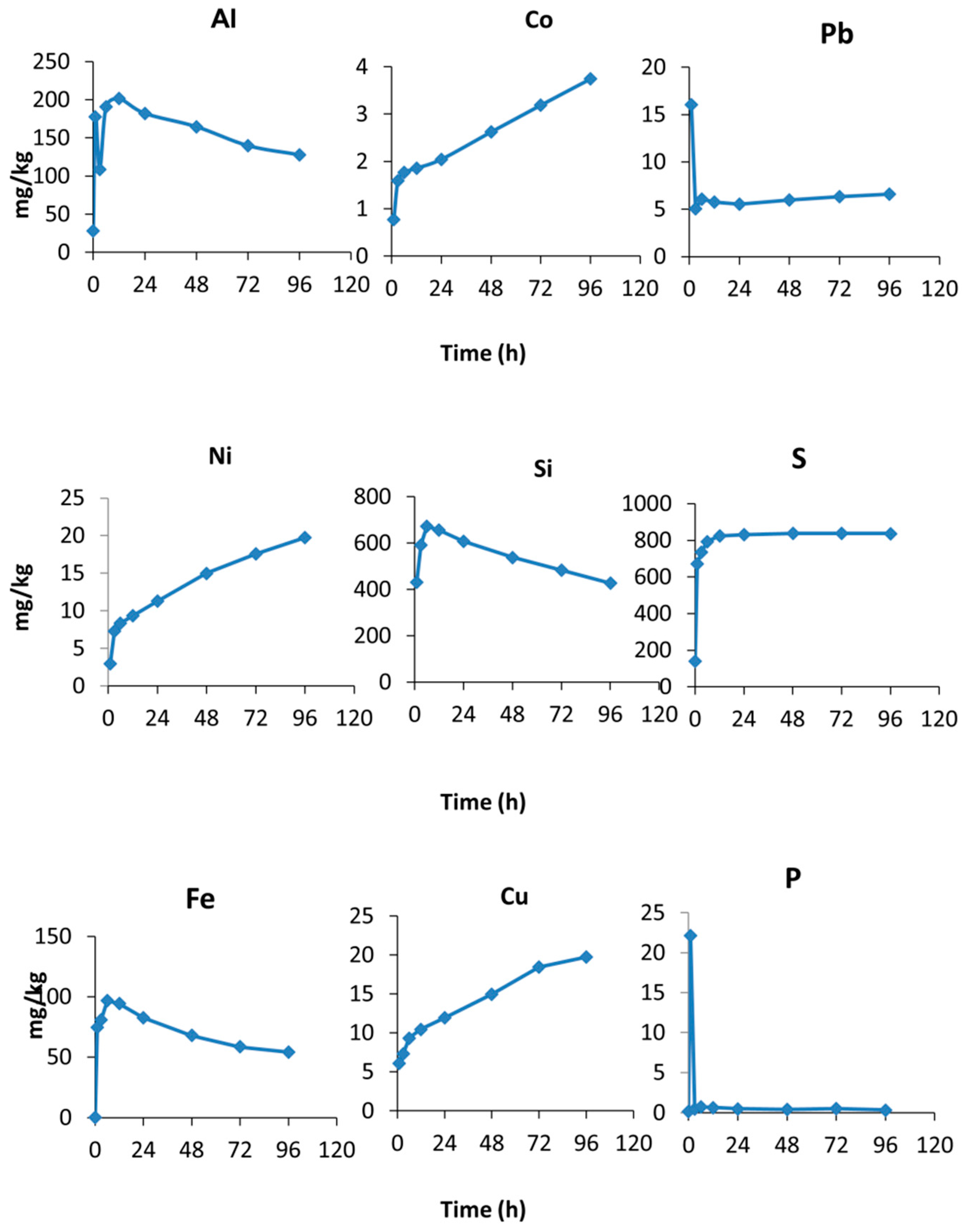
3.2. Acid Neutralization Capacity (ANC) and Trace Element Release at pH 4
4. Discussion
4.1. Potential Release of Trace Elements Based on Different Extractions/Leaching Test
4.2. Kinetics of Trace Element Release during Leaching Test at pH 4
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mineral Commodity Summaries 2013. Available online: https://minerals.usgs.gov/minerals/pubs/mcs/2013/mcs2013.pdf (accessed on 10 July 2018).
- Hunt, A.J.; Farmer, T.J.; Clark, J.H. Elemental sustainability & the importance of scarce element recovery. In Element Recovery and Sustainability; Hunt, A.J., Ed.; RSC Publishing: Cambridge, UK, 2013; Volume 22, pp. 1–28. [Google Scholar]
- Chlopecka, A.; Bacon, J.R.; Wilson, M.J.; Kay, J. Forms of cadmium, lead and zinc in contaminated soils from southwest Poland. J. Environ. Qual. 1996, 25, 69–79. [Google Scholar] [CrossRef]
- Kabata-Pendias, A.; Mukherjee, A.B. Trace Elements from Soil to Human; Springer: Berlin/Heidelberg, Germany; New York, NY, USA, 2007; pp. 1–549. [Google Scholar]
- Veli, S.; Kirli, L.; Alyuz, B.; Durmusoglu, E. Characterization of bottom ash, fly ash, and filter cake produced from hazardous waste incineration. Polish J. Environ. Stud. 2008, 17, 139–145. [Google Scholar]
- Cho, J.H.; Eom, Y.; Park, J.M.; Lee, S.B.; Hong, J.H.; Lee, T.G. Mercury leaching characteristics of waste treatment residues generated from various sources in Korea. Waste Manag. 2013, 33, 1675–1681. [Google Scholar] [CrossRef] [PubMed]
- CEWEP. Bottom Ash Fact Sheet 2016. Available online: http://www.cewep.eu/wp-content/uploads/2017/09/FINAL-Bottom-Ash-factsheet.pdf (accessed on 4 June 2017).
- Nowak, B.; Rocha, S.F.; Aschenbrenner, P.; Rechberger, H.; Winter, F. Heavy metal removal from MSW fly ash by means of chlorination and thermal treatment: Influence of the chloride type. Chem. Eng. J. 2012, 179, 178–185. [Google Scholar] [CrossRef]
- Pani, G.K.; Rath, P.; Maharana, L.; Barik, R.; Senapati, P.K. Assessment of heavy metals and rheological characteristics of coal ash samples in presence of some selective additives. Int. J. Environ. Sci. Technol. 2016, 13, 725–731. [Google Scholar] [CrossRef]
- Kosson, D.S.; Van der Sloot, H.A.; Sanchez, F.; Garrabrants, A.C. An integrated framework for evaluating leaching in waste management and utilization of secondary materials. Environ. Eng. Sci. 2002, 19, 159–204. [Google Scholar] [CrossRef]
- Zou, Z.; Qiu, R.; Zhang, W.; Dong, H.; Zhao, Z.; Zhang, T.; Wei, X.; Cai, X. The study of operating variables in soil washing with EDTA. Environ. Pollut. 2009, 157, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Sahuquillo, A.; Rigol, A.; Rauret, G. Overview of the use of leaching/extraction tests for risk assessment of trace metals in contaminated soils and sediments. Trends Anal. Chem. 2003, 22, 152–159. [Google Scholar] [CrossRef]
- Ure, A.M. 1996 Single extraction schemes for soil analysis and related applications. Sci. Total Environ. 1996, 178, 3–10. [Google Scholar] [CrossRef]
- CEN/TS 14997. Characterization of Waste—Leaching Behavior Tests—Influence of pH on Leaching with Continuous pH-Control; CEN (Comité Européen de Normalisation): Brussels, Belgium, 2006. [Google Scholar]
- Polettini, A.; Pomi, R. The leaching behavior of incinerator bottom ash as affected by accelerated ageing. J. Hazard. Mater. 2004, 113, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Cornelis, G.; Van Gerven, T.; Vandecasteele, C. Antimony leaching from MSWI bottom ash: Modelling of the effect of pH and carbonation. Waste Manag. 2012, 32, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Van Zomeren, A.; van der Sloot, H. Systematic Leaching Behaviour of Worldwide MSWI Bottom Ashes in Spite of Their Variability in Content Sustainable Landfilling; ECN Report; CISA Publisher: Padova, Italy, 2013. [Google Scholar]
- Adama, M.; Esena, R. Heavy metal contamination of soils around a hospital waste incinerator bottom ash dumps site. J. Environ. Pub. Health 2016, 2016, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Dung, T.T.T.; Vassilieva, E.; Golreihan, A.; Phung, N.K.; Swennen, R.; Cappuyns, V. Potentially toxic elements in bottom ash from hazardous waste incinerators: an integrated approach to assess the potential release in relation to solid-phase characteristics. J. Mater. Cycles Waste Manag. 2017, 19, 1194–1203. [Google Scholar] [CrossRef]
- Nurmesniemi, H.; Poykio, R.; Kuokkanen, T.; Ramo, J. Chemical sequential extraction of heavy metals and sulphur in bottom ash and in fly ash from a pulp and paper mill complex. Waste Manag. Res. 2008, 26, 389–399. [Google Scholar] [CrossRef] [PubMed]
- Bayuseno, A.P.; Schmahl, W.W. Understanding the chemical and mineralogical properties of the inorganic portion of MSWI bottom ash. Waste Manag. 2010, 30, 1509–1520. [Google Scholar] [CrossRef] [PubMed]
- Manskinen, K.; Pöykiö, R. Comparison of the total and fractionated heavy metal and sulphur concentrations in bottom ash and fly ash from a large-sized (120 MW ) power plant of a fluting board mill. Chemija 2011, 22, 46–55. [Google Scholar]
- Cappuyns, V.; Swennen, R. The application of pH(stat) leaching tests to assess the pH-dependent release of trace metals from soils, sediments and waste materials. J. Hazard. Mater. 2008, 158, 185–195. [Google Scholar] [CrossRef] [PubMed]
- Van Gerven, T.; Cooreman, H.; Imbrecht, K.; Hindrix, K.; Vandecasteele, C. Extraction of heavy metals from municipal solid waste incinerator (MSWI) bottom ash with organic solutions. J. Hazard. Mater. 2007, 140, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Solid Waste—National Technical Regulation on Hazardous Waste Thresholds. Available online: http://vea.gov.vn/vn/vanbanphapquy/layykiengopy/Documents/QCVNnguongCTNH.doc (accessed on 10 July 2018).
- Quevauviller, P. Operationally defined extraction procedures for soil and sediment analysis I. Standardization. Trends Anal. Chem. 1998, 17, 289–298. [Google Scholar] [CrossRef]
- Quevauviller, P.; Rauret, G.; Rubio, R.; Lopez-Sanchez, J.F.; Ure, A.; Bacon, J.; Muntau, H. Certified reference materials for the quality control of EDTA-and acetic acid-extractable contents of trace elements in sewage sludge amended soils (CRMs 483 and 484). Fresenius J. Anal. Chem. 1997, 357, 611–618. [Google Scholar] [CrossRef]
- Li, L.Y.; Ohtsubo, M.; Higashi, T.; Yamaoka, S.; Morishita, T. Leachability of municipal solid waste ashes in simulated landfill conditions. Waste Manag. 2007, 27, 932–945. [Google Scholar] [CrossRef] [PubMed]
- Jamali, M.K.; Kazi, T.G.; Arain, M.B.; Afridi, H.I.; Jalbani, N.; Kandhro, G.A.; Shah, A.Q.; Baig, J.A. Heavy metal accumulation in different varieties of wheat (Triticum aestivum L.) grown in soil amended with domestic sewage sludge. J. Hazard. Mater. 2009, 164, 1386–1391. [Google Scholar] [CrossRef] [PubMed]
- Soriano-Disla, J.M.; Gómez, I.; Navarro-Pedreño, J.; Lag-Brotons, A. Evaluation of single chemical extractants for the prediction of heavy metal uptake by barley in soils amended with polluted sewage sludge. Plant Soil 2010, 327, 303–314. [Google Scholar] [CrossRef]
- Nugteren, H. Limitations of combustion ashes: ‘From threat to profit’. In Combustion Residues: Current, Novel and Renewable Applications; Cox, M., Nugteren, H., Janssen-Jurkovičvá, M., Eds.; John Wiley & Sons: West Sussex, UK, 2008; pp. 137–198. [Google Scholar]
- Martell, A.E.; Smith, R.M. Critical Stability Constants. Other Organic Ligands, 3rd ed.; Plenum Press: New York, NY, USA, 1977. [Google Scholar]
- Martell, A.E.; Smith, R.M. Critical Stability Constants. First Supplement, 5; Plenum Press: New York, NY, USA, 1982. [Google Scholar]
- Kim, C.; Lee, Y.; Ong, S.K. Factors affecting EDTA extraction of lead from lead-contaminated soils. Chemosphere 2003, 51, 845–853. [Google Scholar] [CrossRef]
- Papassiopi, N.; Tambouris, S.; Kontopoulos, A. Removal of heavy metals from calcareous contaminated soils by EDTA leaching. Water Air Soil Pollut. 1999, 109, 1–15. [Google Scholar] [CrossRef]
- Ganne, P.; Cappuyns, V.; Vervoort, A.; Buvé, L.; Swennen, R. Leachability of heavy metals and arsenic from slags of metal extraction industry at Angleur (eastern Belgium). Sci. Tot. Environ. 2006, 356, 69–85. [Google Scholar] [CrossRef] [PubMed]
- Meima, J.A.; van der Weijden, R.D.; Eighmy, T.T.; Comans, R.N.J. Carbonation processes in municipal solid waste incinerator bottom ash and their effect on the leaching of copper and molybdenum. Appl. Geochem. 2002, 17, 1503–1513. [Google Scholar] [CrossRef]
- Kaibouchi, S.; Germain, P. Comparative study of physico-chemical and environmental characteristics of (MSWI) bottom ash resulting from classical and selective collection for a valorization in road construction. In Progress on the Road to Sustainability, Fifth International Conference on the Environmental and Technical Implications of Construction with Alternative Materials, San Sebastian, Spain, 4–6 June 2003; Ortiz de Urbina, G., Goumans, H., Eds.; 2003; pp. 645–653. [Google Scholar]
- Van Gerven, T.; Van Keer, E.; Arickx, S.; Jaspers, M.; Wauters, G.; Vandecasteele, C. Carbonation of MSWI-bottom ash to decrease heavy metal leaching, in view of recycling. Waste Manag. 2005, 25, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Liu, J.; Yu, Q.; Jin, Y.; Nie, Y. Leaching characteristics of heavy metals in municipal solid waste incinerator fly ash. J. Environ. Sci. Health A 2005, 40, 1975–1985. [Google Scholar] [CrossRef] [PubMed]
- Fedje, K.K.; Ekberg, C.; Skarnemark, G.; Steenari, B.M. Removal of hazardous metals from MSW fly ash—An evaluation of ash leaching methods. J. Hazard. Mater. 2010, 173, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Olsson, S.; van Schaik, J.W.J.; Gustafsson, J.P.; Kleja, D.B.; van Hees, P.A.W. Copper (II) binding to dissolved organic matter fractions in municipal solid waste incinerator bottom ash leachate. Environ. Sci. Technol. 2007, 41, 4286–4291. [Google Scholar] [CrossRef] [PubMed]
- Arickx, S.; Van Gerven, T.; Boydens, E.; L’Hoëst, P.; Blanpain, B.; Vandecasteele, C. Speciation of Cu in MSWI bottom ash and its relation to Cu leaching. Appl. Geochem. 2008, 23, 3642–3650. [Google Scholar] [CrossRef]
- Meima, J.A.; Comans, R.N.J. Reducing Sb-leaching from municipal solid waste incinerator bottom ash by addition of sorbent minerals. J. Geochem. Explor. 1998, 62, 299–304. [Google Scholar] [CrossRef]
- Kirby, C.S.; Rimstidt, J.D. Interaction of municipal solid waste ash with water. Environ. Sci. Technol. 1994, 28, 443–451. [Google Scholar] [CrossRef] [PubMed]
- Pickering, W.F. Metal ion speciation- soils and sediments (a review). Ore Geol. Rev. 1986, 1, 83–146. [Google Scholar] [CrossRef]
- Cornelis, G.; Johnson, C.A.; Van Gerven, T.; Vandecasteele, C. Leaching mechanisms of oxyanionic metalloid and metal species in alkaline solid wastes: A review. Appl. Geochem. 2008, 23, 955–976. [Google Scholar] [CrossRef]



| Type of Bottom Ash | Unit | This Study [19] | [20] | [21] | [22] | [18] | |
|---|---|---|---|---|---|---|---|
| Hazardous Waste Incinerators | Industrial Wastes | Municipal Solid Waste Incinerators (MSWI) | Coal, Peat and Forest Residues | Hospital Medical Waste Incinerators | |||
| Element | AS1 * | AS2 ** | |||||
| Al | % | 6.06 ± 0.33 | 2.90 ± 0.12 | - | 6.4 | 1.22 | |
| As | mg·kg−1 | 7 ± 1 | 77 ± 75 | 21.4 | 21 | 3.9 | |
| Ca | % | 1.91 ± 0.02 | 3.85 ± 0.18 | 6.26 | 9.7 | - | |
| Cd | mg·kg−1 | 1 ± 0.03 | 2 ± 0.1 | <0.3 | 14 | <3.0 | 7.54 |
| Co | mg·kg−1 | 928 ± 23 | 63 ± 11 | 7.8 | 67 | 2.9 | |
| Cr | mg·kg−1 | 573 ± 45 | 804 ± 185 | 50.6 | 1158 | 10.9 | 99.30 |
| Cu | mg·kg−1 | 1126 ± 196 | 818 ± 188 | 33.5 | 7743 | 16.9 | |
| Fe | % | 3.92 ± 0.15 | 23.82 ± 3.53 | - | 8.9 | 0.84 | |
| K | mg·kg−1 | 3800 ± 200 | 2400 ± 2 | 2910 | 9000 | - | |
| Mg | mg·kg−1 | 1900 ± 6 | 7900 ± 617 | 5850 | 15,000 | - | |
| Mn | mg·kg−1 | 377 ± 3 | 3597 ± 259 | 1450 | 1000 | 425.0 | |
| Mo | mg·kg−1 | 278 ± 7 | 42 ± 1 | 1.1 | 99 | <1.0 | |
| Ni | mg·kg−1 | 1373 ± 77 | 233 ± 22 | 24.3 | 356 | 6.3 | |
| P | mg·kg−1 | 2000 ± 47 | 1000 ± 41 | 3430 | 4000 | - | |
| Pb | mg·kg−1 | 63 ± 4 | 817 ± 85 | 5.1 | 1022 | <3.0 | 143.80 |
| S | mg·kg−1 | 1130 ± 56 | 3820 ± 348 | 1580 | 4950 | 59.4 | |
| Zn | mg·kg−1 | 930 ± 2 | 1461 ± 291 | 340 | 7732 | 256.0 | 16,417.69 |
| OC | % | 3.24 ± 0.01 | 0.95 ± 0.04 | - | <0.5 | ||
| pH | 8.64 ± 0.05 | 9.40 ± 0.70 | - | ||||
| Calcite | % | 1.0 | 3.6 | - | 0.5 | ||
| Corundum | % | 6.9 | - | - | 4.5 | ||
| Hematite | % | - | 7.2 | - | 1.9 | ||
| Magnetite | % | 3.1 | 13.1 | - | 4.2 | ||
| Mullite | % | 1.9 | - | - | |||
| Quartz | % | 10.3 | 7.8 | 31.4 | |||
| Rutile | % | - | 2.6 | 0.8 | |||
| Element | Ammonium-EDTA 0.05 mol·L−1 (This Work) | Ammonium-EDTA 0.05 mol·L−1 (Certified Values) | Acetic Acid 0.43 mol·L−1 (This work) | Acetic Acid 0.43 mol·L−1 (Certified Values) |
|---|---|---|---|---|
| Cd | 20.7 ± 0.7 | 20.4 ± 1.3 | 19.6 ± 0.5 | 18.3 ± 0.6 |
| Cr | 37.6 ± 9.8 | 28.6 ± 2.6 | 26.3 ± 1.6 | 18.7 ± 1.0 |
| Cu | 192 ± 7 | 215 ± 11 | 38.2 ± 1.2 | 33.5 ± 1.6 |
| Ni | 24.2 ± 1.4 | 28.7 ± 1.7 | 25.5 ± 1.5 | 25.8 ± 1.0 |
| Pb | 189 ± 17 | 229 ± 8 | 1.9 ± 0.2 | 3.1 ± 0.25 |
| Zn | 529 ± 16 | 612 ± 19 | 659 ± 34 | 620 ± 24 |
| Element | AS1 (%) | AS2 (%) | ||
|---|---|---|---|---|
| Major elements | ||||
| EDTA | CH3COOH | EDTA | CH3COOH | |
| Al | 0.2 | 4 | 1 | 20 |
| Ca | 87 | 100 | 45 | 87 |
| Fe | 1 | 7 | 0.1 | 2 |
| K | 5 | 10 | 8 | 19 |
| Mg | 25 | 47 | 7 | 50 |
| Mn | 9 | 23 | 1 | 11 |
| P | 13 | 13 | 5 | 12 |
| S | 98 | 100 | 36 | 37 |
| Trace elements | ||||
| As | 14 | < 6 | 2 | < 0.5 |
| Cd | 27 | 65 | < 3 | < 3 |
| Co | 3 | 17 | 3 | 25 |
| Cr | 0.6 | 3 | 0.2 | 4 |
| Cu | 22 | 33 | 11 | 29 |
| Mo | 10 | 6 | 10 | < 0.9 |
| Ni | 2 | 15 | 3 | 27 |
| Pb | 9 | < 1 | 31 | 42 |
| Zn | 20 | 43 | 6 | 29 |
| Heading | AS1 (%) | AS2 (%) |
|---|---|---|
| Major Elements | ||
| Al | 0.1 | 0.4 |
| Ca | 45 | 34 |
| Fe | 0.3 | 0.02 |
| K | 5 | 7 |
| Mg | 14 | 14 |
| Mn | 5 | 2 |
| P | 0.03 | 0.04 |
| S | 54 | 22 |
| Trace Elements | ||
| As | <1 | <1 |
| Cd | <6 | <3 |
| Co | 3 | 6 |
| Cr | 0.04 | 0.02 |
| Cu | 3 | 2 |
| Mo | 1 | 0.3 |
| Ni | 3 | 6 |
| Pb | <0.2 | 1 |
| Zn | 9 | 3 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dung, T.T.T.; Vassilieva, E.; Swennen, R.; Cappuyns, V. Release of Trace Elements from Bottom Ash from Hazardous Waste Incinerators. Recycling 2018, 3, 36. https://doi.org/10.3390/recycling3030036
Dung TTT, Vassilieva E, Swennen R, Cappuyns V. Release of Trace Elements from Bottom Ash from Hazardous Waste Incinerators. Recycling. 2018; 3(3):36. https://doi.org/10.3390/recycling3030036
Chicago/Turabian StyleDung, Tran Thi Thu, Elvira Vassilieva, Rudy Swennen, and Valérie Cappuyns. 2018. "Release of Trace Elements from Bottom Ash from Hazardous Waste Incinerators" Recycling 3, no. 3: 36. https://doi.org/10.3390/recycling3030036





