Anode-Free Rechargeable Sodium-Metal Batteries
Abstract
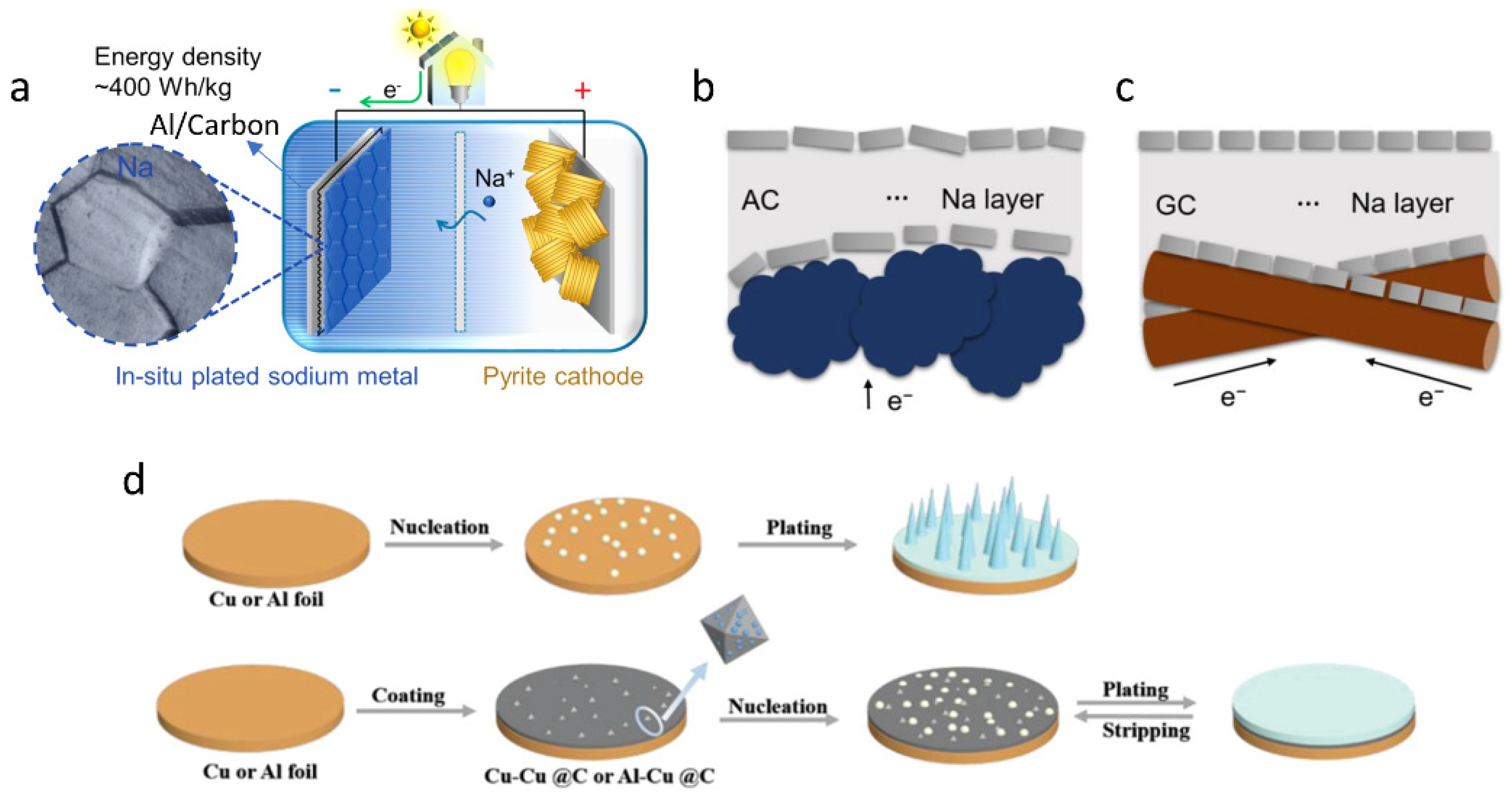
1. Introduction
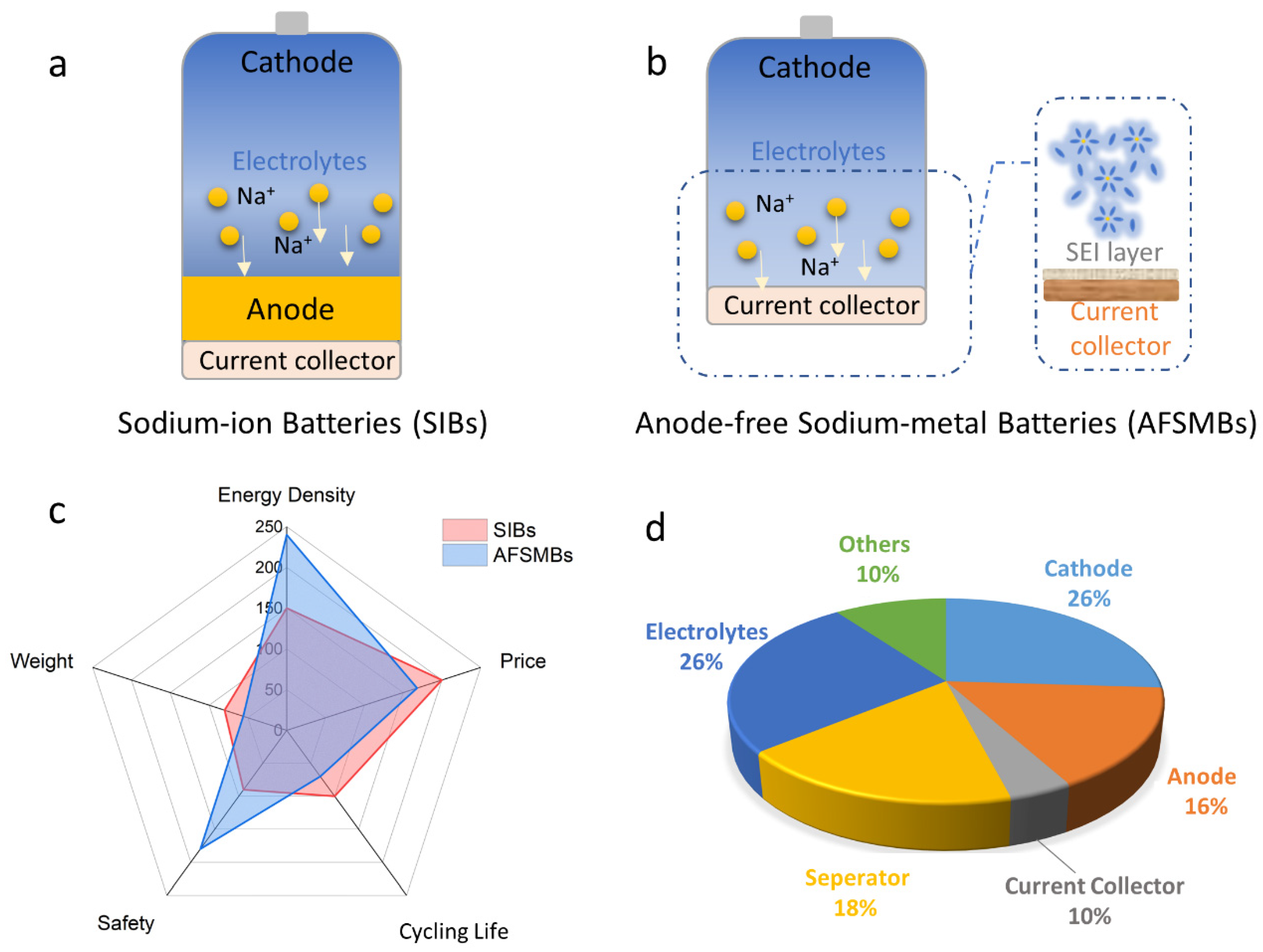
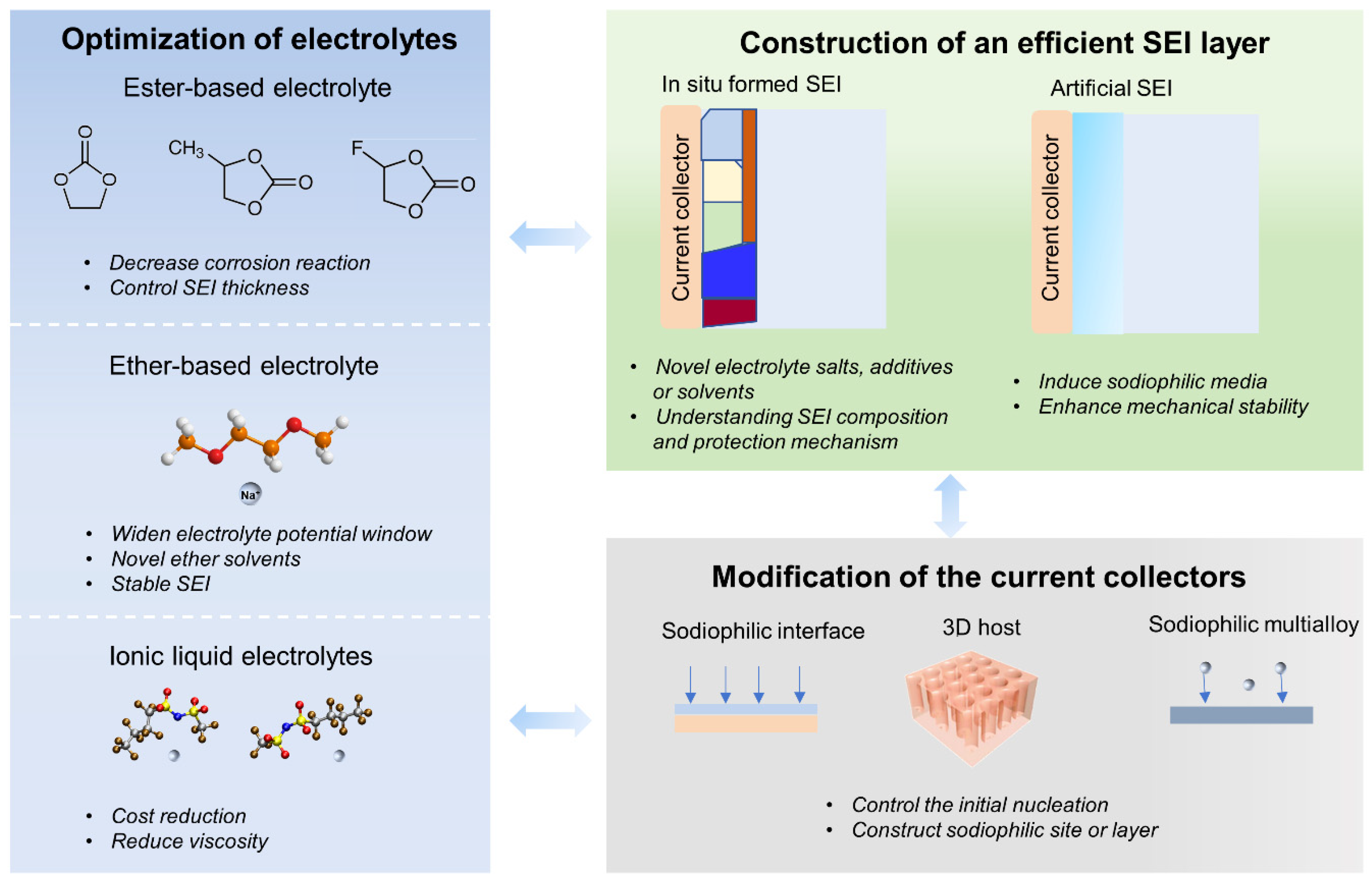
2. State-of-the-Art Anode-Free Rechargeable Sodium Metal Batteries
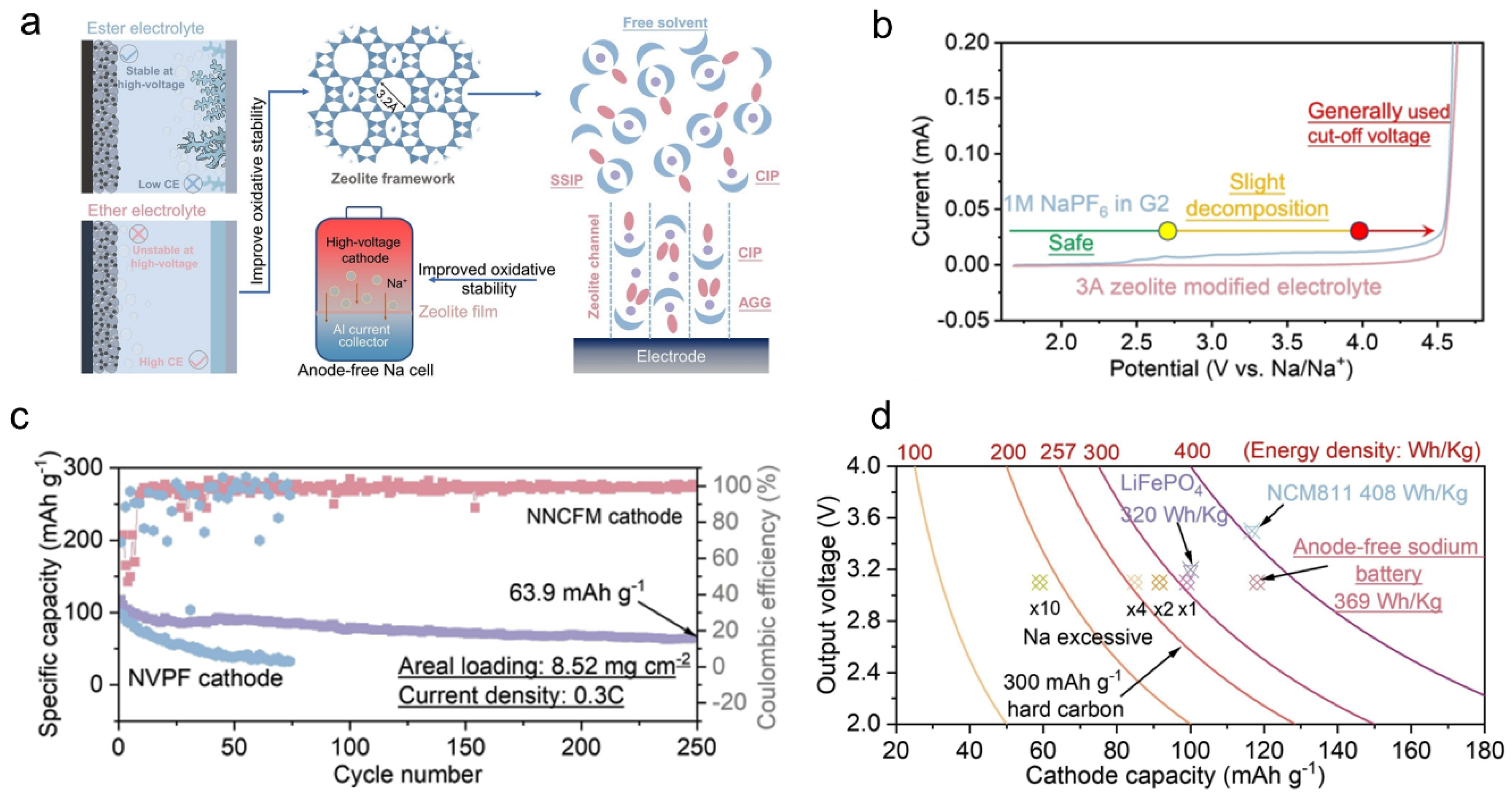
2.1. The Optimization of Electrolytes
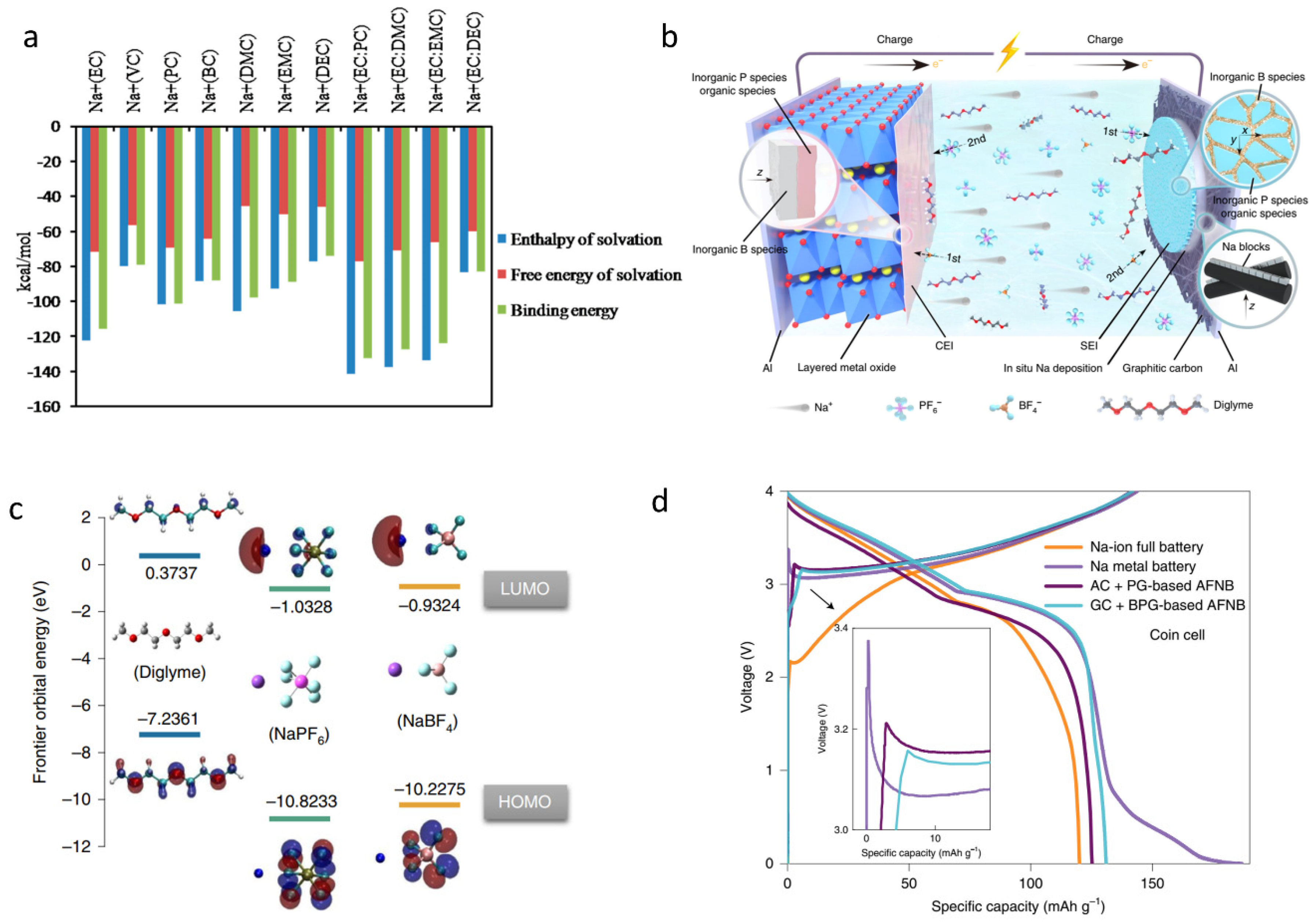
2.2. The Construction of an Efficient SEI Layer
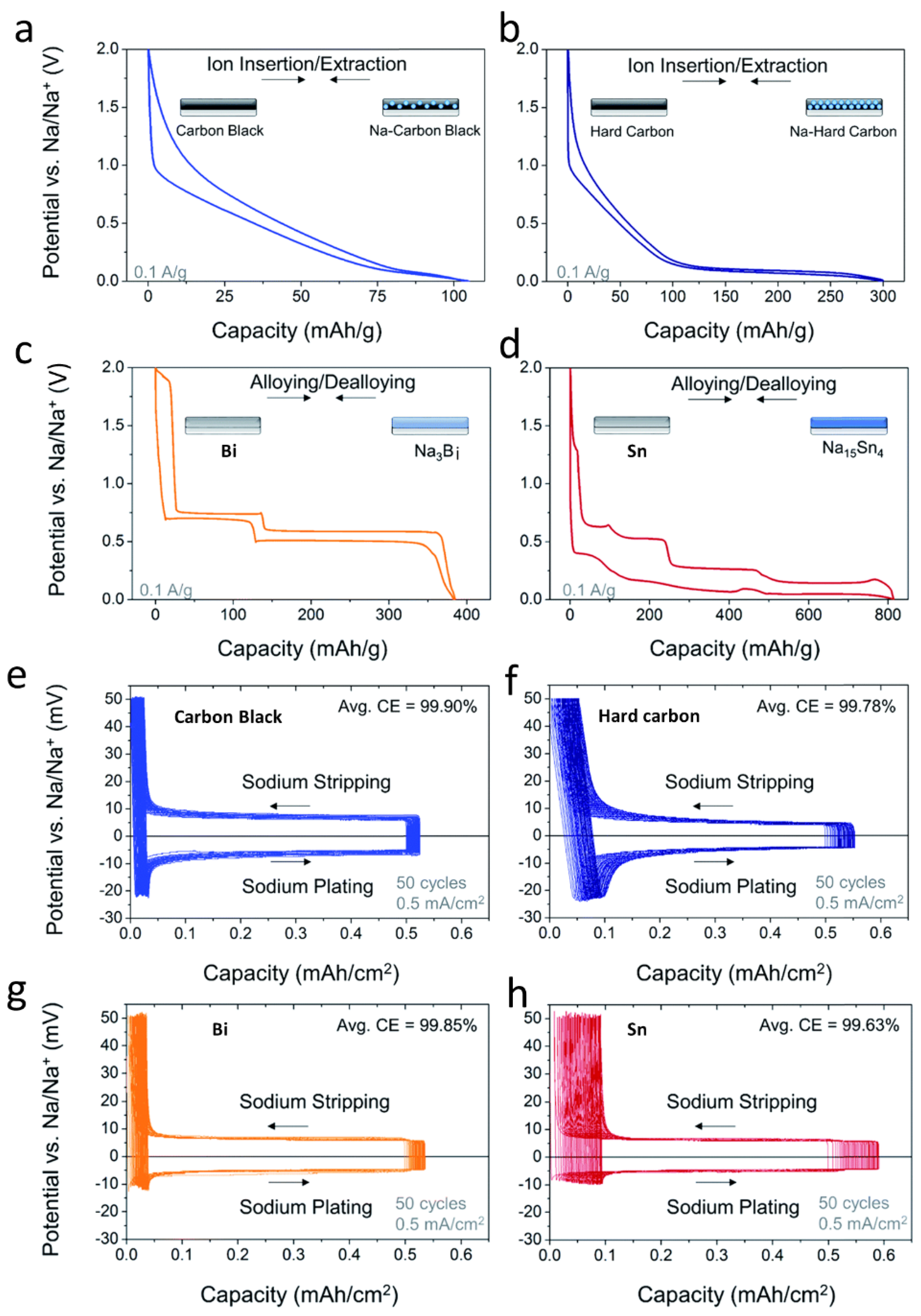
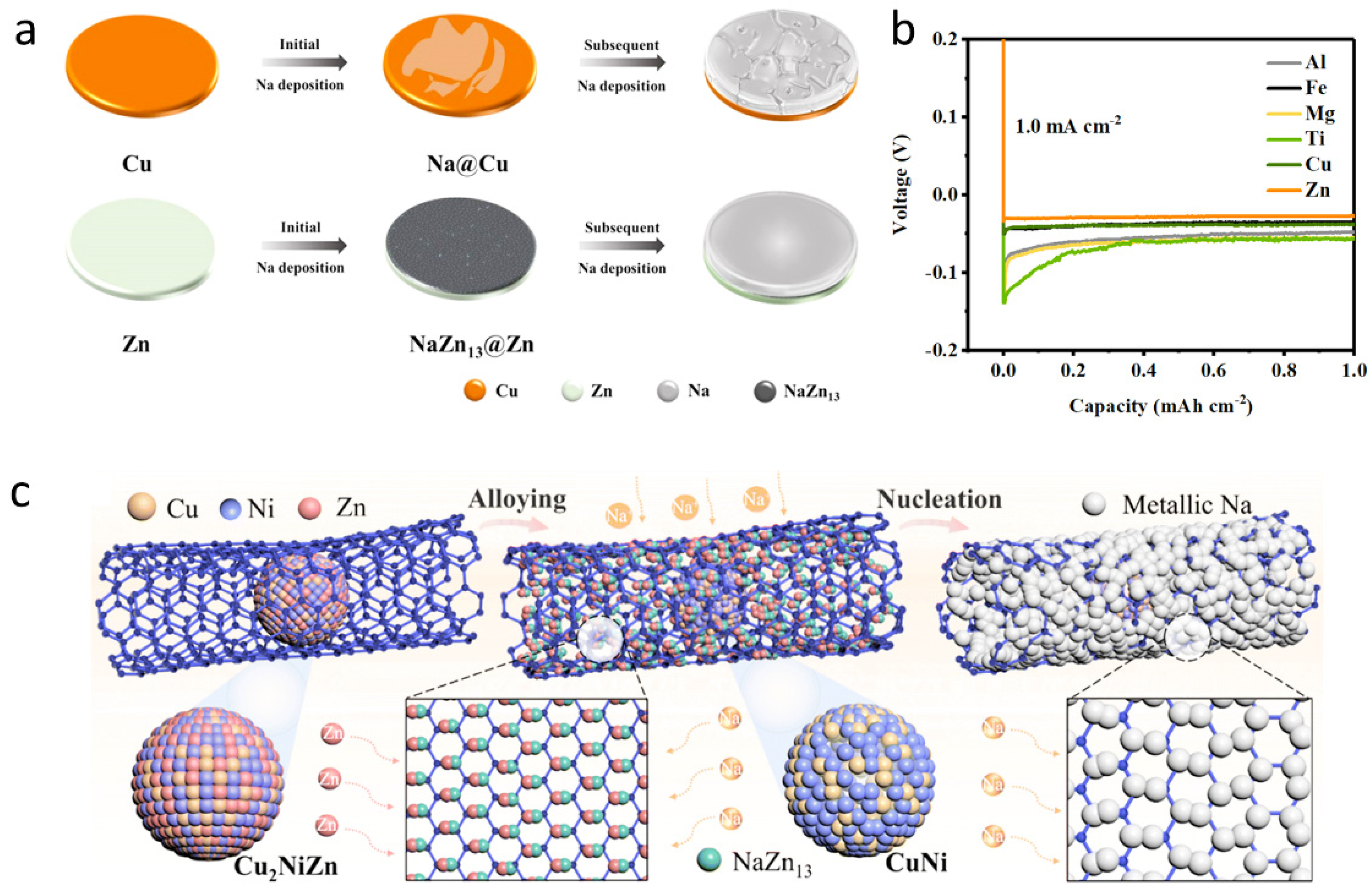
2.3. Modification of the Current Collectors to Regulate Sodium Deposition
3. Summary and Perspective
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Yabuuchi, N.; Kubota, K.; Dahbi, M.; Komaba, S. Research development on sodium-ion batteries. Chem. Rev. 2014, 114, 11636–11682. [Google Scholar] [CrossRef] [PubMed]
- Pan, H. Room-temperature stationary sodium-ion batteries for large-scale electric energy storage. Energy Environ. Sci. 2013, 6, 2338–2360. [Google Scholar] [CrossRef]
- Grey, C.P.; Hall, D.S. Prospects for lithium-ion batteries and beyond—A 2030 vision. Nat. Commun. 2020, 11, 6279. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Gao, T.; Fan, X.; Han, F.; Wang, C. Electrochemical Techniques for Intercalation Electrode Materials in Rechargeable Batteries. Acc. Chem. Res. 2017, 50, 1022–1031. [Google Scholar] [CrossRef]
- Winter, M.; Barnett, B.; Xu, K. Before Li Ion Batteries. Chem. Rev. 2018, 118, 11433–11456. [Google Scholar] [CrossRef]
- Hwang, J.Y.; Myung, S.T.; Sun, Y.K. Sodium-ion batteries: Present and future. Chem. Soc. Rev. 2017, 46, 3529–3614. [Google Scholar] [CrossRef]
- Yang, M.; Luo, J.; Guo, X.; Chen, J.; Cao, Y.; Chen, W. Aqueous Rechargeable Sodium-Ion Batteries: From Liquid to Hydrogel. Batteries 2022, 8, 180. [Google Scholar] [CrossRef]
- Peters, J.; Peña Cruz, A.; Weil, M. Exploring the Economic Potential of Sodium-Ion Batteries. Batteries 2019, 5, 10. [Google Scholar] [CrossRef]
- Choi, J.W.; Aurbach, D. Promise and reality of post-lithium-ion batteries with high energy densities. Nat. Rev. Mater. 2016, 1, 16013. [Google Scholar] [CrossRef]
- Zhu, M.; Wang, G.; Liu, X.; Guo, B.; Xu, G.; Huang, Z.; Wu, M.; Liu, H.K.; Dou, S.X.; Wu, C. Dendrite-Free Sodium Metal Anodes Enabled by a Sodium Benzenedithiolate-Rich Protection Layer. Angew. Chem. Inter. Ed. 2020, 132, 6658–6662. [Google Scholar] [CrossRef]
- Gao, L.; Chen, J.; Chen, Q.; Kong, X. The chemical evolution of solid electrolyte interface in sodium metal batteries. Sci. Adv. 2022, 8, eabm4606. [Google Scholar] [CrossRef]
- Shakourian-Fard, M.; Kamath, G.; Smith, K.; Xiong, H.; Sankaranarayanan, S.K.R.S. Trends in Na-Ion Solvation with Alkyl-Carbonate Electrolytes for Sodium-Ion Batteries: Insights from First-Principles Calculations. J. Phys. Chem. C 2015, 119, 22747–22759. [Google Scholar] [CrossRef]
- Zhang, J.-G.; Xu, W.; Xiao, J.; Cao, X.; Liu, J. Lithium Metal Anodes with Nonaqueous Electrolytes. Chem. Rev. 2020, 120, 13312–13348. [Google Scholar] [CrossRef]
- Ponrouch, A.; Monti, D.; Boschin, A.; Steen, B.; Johansson, P.; Palacín, M.R. Non-aqueous electrolytes for sodium-ion batteries. J. Mater. Chem. A 2015, 3, 22–42. [Google Scholar] [CrossRef]
- Cresce, A.V.; Russell, S.M.; Borodin, O.; Allen, J.A.; Schroeder, M.A.; Dai, M.; Peng, J.; Gobet, M.P.; Greenbaum, S.G.; Rogers, R.E.; et al. Solvation behavior of carbonate-based electrolytes in sodium ion batteries. Phys. Chem. Chem. Phys. 2017, 19, 574–586. [Google Scholar] [CrossRef]
- Kumar, H.; Detsi, E.; Abraham, D.P.; Shenoy, V.B. Fundamental Mechanisms of Solvent Decomposition Involved in Solid-Electrolyte Interphase Formation in Sodium Ion Batteries. Chem. Mater. 2016, 28, 8930–8941. [Google Scholar] [CrossRef]
- Slater, M.D.; Kim, D.; Lee, E.; Johnson, C.S. Sodium-ion batteries. Adv. Funct. Mater. 2013, 23, 947–958. [Google Scholar] [CrossRef]
- Zhang, L.; Tsolakidou, C.; Mariyappan, S.; Tarascon, J.-M.; Trabesinger, S. Unraveling gas evolution in sodium batteries by online electrochemical mass spectrometry. Energy Storage Mater. 2021, 42, 12–21. [Google Scholar] [CrossRef]
- Chen, X.; Shen, X.; Li, B.; Peng, H.J.; Cheng, X.B.; Li, B.Q.; Zhang, X.Q.; Huang, J.Q.; Zhang, Q. Ion-Solvent Complexes Promote Gas Evolution from Electrolytes on a Sodium Metal Anode. Angew. Chem. Int. Ed. Engl. 2018, 57, 734–737. [Google Scholar] [CrossRef]
- Li, K.; Zhang, J.; Lin, D.; Wang, D.-W.; Li, B.; Lv, W.; Sun, S.; He, Y.-B.; Kang, F.; Yang, Q.-H.; et al. Evolution of the electrochemical interface in sodium ion batteries with ether electrolytes. Nat. Commun. 2019, 10, 725. [Google Scholar] [CrossRef]
- Carbone, L.; Munoz, S.; Gobet, M.; Devany, M.; Greenbaum, S.; Hassoun, J. Characteristics of glyme electrolytes for sodium battery: Nuclear magnetic resonance and electrochemical study. Electrochim. Acta 2017, 231, 223–229. [Google Scholar] [CrossRef]
- Louli, A.J.; Eldesoky, A.; Weber, R.; Genovese, M.; Coon, M.; deGooyer, J.; Deng, Z.; White, R.T.; Lee, J.; Rodgers, T.; et al. Diagnosing and correcting anode-free cell failure via electrolyte and morphological analysis. Nat. Energy 2020, 5, 693–702. [Google Scholar] [CrossRef]
- Li, Y.; Zhou, Q.; Weng, S.; Ding, F.; Qi, X.; Lu, J.; Li, Y.; Zhang, X.; Rong, X.; Lu, Y.; et al. Interfacial engineering to achieve an energy density of over 200 Wh kg−1 in sodium batteries. Nat. Energy 2022, 7, 511–519. [Google Scholar] [CrossRef]
- Wu, D.; Zhu, C.; Wu, M.; Wang, H.; Huang, J.; Tang, D.; Ma, J. Highly Oxidation-Resistant Electrolyte for 4.7 V Sodium Metal Batteries Enabled by Anion/Cation Solvation Engineering. Angew. Chem. Inter. Ed. 2022, e202214198. [Google Scholar] [CrossRef]
- Zheng, J.; Chen, S.; Zhao, W.; Song, J.; Engelhard, M.H.; Zhang, J.-G. Extremely Stable Sodium Metal Batteries Enabled by Localized High-Concentration Electrolytes. ACS Energy Lett. 2018, 3, 315–321. [Google Scholar] [CrossRef]
- Lu, Z.; Yang, H.; Yang, Q.H.; He, P.; Zhou, H. Building a Beyond Concentrated Electrolyte for High-Voltage Anode-Free Rechargeable Sodium Batteries. Angew. Chem. Int. Ed. Engl. 2022, 61, e202200410. [Google Scholar] [CrossRef]
- Wang, J.; Yamada, Y.; Sodeyama, K.; Chiang, C.H.; Tateyama, Y.; Yamada, A. Superconcentrated electrolytes for a high-voltage lithium-ion battery. Nat. Commun. 2016, 7, 12032. [Google Scholar] [CrossRef]
- Hou, J.; Lu, L.; Wang, L.; Ohma, A.; Ren, D.; Feng, X.; Li, Y.; Li, Y.; Ootani, I.; Han, X.; et al. Thermal runaway of Lithium-ion batteries employing LiN(SO2F)2-based concentrated electrolytes. Nat. Commun. 2020, 11, 5100. [Google Scholar] [CrossRef]
- Chen, S.; Zheng, J.; Mei, D.; Han, K.S.; Engelhard, M.H.; Zhao, W.; Xu, W.; Liu, J.; Zhang, J.G. High-Voltage Lithium-Metal Batteries Enabled by Localized High-Concentration Electrolytes. Adv. Mater 2018, 30, 1706102. [Google Scholar] [CrossRef]
- Zhu, N.; Zhang, K.; Wu, F.; Bai, Y.; Wu, C. Ionic Liquid-Based Electrolytes for Aluminum/Magnesium/Sodium-Ion Batteries. Energy Mater. Adv. 2021, 2021, 1–29. [Google Scholar] [CrossRef]
- Yang, Q.; Zhang, Z.; Sun, X.-G.; Hu, Y.-S.; Xing, H.; Dai, S. Ionic liquids and derived materials for lithium and sodium batteries. Chem. Soc. Rev. 2018, 47, 2020–2064. [Google Scholar] [CrossRef]
- Wibowo, R.; Aldous, L.; Rogers, E.I.; Ward Jones, S.E.; Compton, R.G. A study of the Na/Na+ redox couple in some room temperature ionic liquids. J. Phys. Chem. C 2010, 114, 3618–3626. [Google Scholar] [CrossRef]
- Sun, H.; Zhu, G.; Xu, X.; Liao, M.; Li, Y.Y.; Angell, M.; Gu, M.; Zhu, Y.; Hung, W.H.; Li, J.; et al. A safe and non-flammable sodium metal battery based on an ionic liquid electrolyte. Nat. Commun. 2019, 10, 3302. [Google Scholar] [CrossRef]
- Zhou, W.; Zhang, M.; Kong, X.; Huang, W.; Zhang, Q. Recent Advance in Ionic-Liquid-Based Electrolytes for Rechargeable Metal-Ion Batteries. Adv. Sci. 2021, 8, 2004490. [Google Scholar] [CrossRef]
- Meng, T.; Young, K.-H.; Wong, D.; Nei, J. Ionic Liquid-Based Non-Aqueous Electrolytes for Nickel/Metal Hydride Batteries. Batteries 2017, 3, 4. [Google Scholar] [CrossRef]
- Shadike, Z.; Lee, H.; Borodin, O.; Cao, X.; Fan, X.; Wang, X.; Lin, R.; Bak, S.M.; Ghose, S.; Xu, K.; et al. Identification of LiH and nanocrystalline LiF in the solid-electrolyte interphase of lithium metal anodes. Nat. Nanotechnol. 2021, 16, 549–554. [Google Scholar] [CrossRef]
- Peled, E.; Menkin, S. Review—SEI: Past, Present and Future. J. Electrochem. Soc. 2017, 164, A1703–A1719. [Google Scholar] [CrossRef]
- Parejiya, A.; Amin, R.; Dixit, M.B.; Essehli, R.; Jafta, C.J.; Iii, D.; Belharouak, I. Improving Contact Impedance via Electrochemical Pulses Applied to Lithium–Solid Electrolyte Interface in Solid-State Batteries. ACS Energy Lett. 2021, 6, 3669–3675. [Google Scholar] [CrossRef]
- Czz, A.; Bcz, A.; Chong, Y.B.; Xqz, A.; Jqh, B.; Ym, C.; Xx, D.; Hong, L.E.; Qiang, Z.A. Liquid phase therapy to solid electrolyte–electrode interface in solid-state Li metal batteries: A review. Energy Storage Mater. 2020, 24, 75–84. [Google Scholar]
- Liang, G.; Mo, F.; Ji, X.; Zhi, C. Non-metallic charge carriers for aqueous batteries. Nat. Rev. Mater. 2021, 6, 109–123. [Google Scholar] [CrossRef]
- Sui, Y.; Ji, X. Anticatalytic Strategies to Suppress Water Electrolysis in Aqueous Batteries. Chem. Rev. 2021, 121, 6654–6695. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Weng, S.; Luo, W.; Chen, B.; Zhang, X.; Gu, Z.; Wang, H.; Ye, X.; Liu, X.; Huang, L.; et al. Deciphering the Role of Fluoroethylene Carbonate towards Highly Reversible Sodium Metal Anodes. Research 2022, 2022, 9754612. [Google Scholar] [CrossRef]
- Jin, Y.; Kneusels, N.-J.H.; Marbella, L.E.; Castillo-Martínez, E.; Magusin, P.C.M.M.; Weatherup, R.S.; Jónsson, E.; Liu, T.; Paul, S.; Grey, C.P. Understanding Fluoroethylene Carbonate and Vinylene Carbonate Based Electrolytes for Si Anodes in Lithium Ion Batteries with NMR Spectroscopy. J. Am. Chem. Soc. 2018, 140, 9854–9867. [Google Scholar] [CrossRef] [PubMed]
- Han, B.; Zou, Y.; Zhang, Z.; Yang, X.; Shi, X.; Meng, H.; Wang, H.; Xu, K.; Deng, Y.; Gu, M. Probing the Na metal solid electrolyte interphase via cryo-transmission electron microscopy. Nat. Commun. 2021, 12, 3066. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, Y.; Pei, A.; Yan, K.; Sun, Y.; Wu, C.-L.; Joubert, L.-M.; Chin, R.; Koh, A.L.; Yu, Y. Atomic structure of sensitive battery materials and interfaces revealed by cryo–electron microscopy. Science 2017, 358, 506–510. [Google Scholar] [CrossRef] [PubMed]
- Zachman, M.J.; Tu, Z.; Choudhury, S.; Archer, L.A.; Kourkoutis, L.F. Cryo-STEM mapping of solid–liquid interfaces and dendrites in lithium-metal batteries. Nature 2018, 560, 345–349. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, S.; Wang, W.; Li, B. Stabilizing sodium metal anode through facile construction of organic-metal interface. J. Energy Chem. 2022, 66, 133–139. [Google Scholar] [CrossRef]
- Yu, Z.; Cui, Y.; Bao, Z. Design Principles of Artificial Solid Electrolyte Interphases for Lithium-Metal Anodes. Cell Rep. Phys. Sci. 2020, 1, 100119. [Google Scholar] [CrossRef]
- Li, N.-W.; Yin, Y.-X.; Yang, C.-P.; Guo, Y.-G. An Artificial Solid Electrolyte Interphase Layer for Stable Lithium Metal Anodes. Adv. Mater. 2016, 28, 1853–1858. [Google Scholar] [CrossRef]
- Kim, M.S.; Ryu, J.-H.; Deepika; Lim, Y.R.; Nah, I.W.; Lee, K.-R.; Archer, L.A.; Il Cho, W. Langmuir–Blodgett artificial solid-electrolyte interphases for practical lithium metal batteries. Nat. Energy 2018, 3, 889–898. [Google Scholar] [CrossRef]
- Wang, H.; Wang, C.; Matios, E.; Li, W. Critical Role of Ultrathin Graphene Films with Tunable Thickness in Enabling Highly Stable Sodium Metal Anodes. Nano Lett. 2017, 17, 6808–6815. [Google Scholar] [CrossRef]
- Hou, Z.; Wang, W.; Chen, Q.; Yu, Y.; Zhao, X.; Tang, M.; Zheng, Y.; Quan, Z. Hybrid protective layer for stable sodium metal anodes at high utilization. ACS Appl. Mater. Interfaces 2019, 11, 37693–37700. [Google Scholar] [CrossRef]
- Qian, J.; Li, Y.; Zhang, M.; Luo, R.; Wang, F.; Ye, Y.; Xing, Y.; Li, W.; Qu, W.; Wang, L. Protecting lithium/sodium metal anode with metal-organic framework based compact and robust shield. Nano Energy 2019, 60, 866–874. [Google Scholar] [CrossRef]
- Sun, B.; Li, P.; Zhang, J.; Wang, D.; Munroe, P.; Wang, C.; Notten, P.H.; Wang, G. Dendrite-free sodium-metal anodes for high-energy sodium-metal batteries. Adv Mater 2018, 30, 1801334. [Google Scholar] [CrossRef]
- Xu, F.; Qu, C.; Lu, Q.; Meng, J.; Zhang, X.; Xu, X.; Qiu, Y.; Ding, B.; Yang, J.; Cao, F.; et al. Atomic Sn–enabled high- utilization, large-capacity, and long-life Na anode. Sci. Adv. 2022, 8, eabm7489. [Google Scholar] [CrossRef]
- Luo, W.; Lin, C.F.; Zhao, O.; Noked, M.; Zhang, Y.; Rubloff, G.W.; Hu, L. Ultrathin surface coating enables the stable sodium metal anode. Adv. Energy Mater. 2017, 7, 1601526. [Google Scholar] [CrossRef]
- Tomich, A.; Park, J.; Son, S.-B.; Kamphaus, E.; Lyu, X.; Dogan, F.; Carta, V.; Gim, J.; Li, T.; Cheng, L. A Carboranyl Electrolyte Enabling Highly Reversible Sodium Metal Anodes via a “Fluorine-Free” SEI. Angew. Chem. Inter. Ed. 2022, e202208158. [Google Scholar] [CrossRef]
- Kim, J.; Kim, J.; Jeong, J.; Park, J.; Park, C.-Y.; Park, S.; Lim, S.G.; Lee, K.T.; Choi, N.-S.; Byon, H.R. Designing fluorine-free electrolytes for stable sodium metal anodes and high-power seawater batteries via SEI reconstruction. Energy Environ. Sci. 2022, 15, 4109–4118. [Google Scholar] [CrossRef]
- Liang, P.; Sun, H.; Huang, C.L.; Zhu, G.; Tai, H.C.; Li, J.; Wang, F.; Wang, Y.; Huang, C.J.; Jiang, S.K. A Nonflammable High-Voltage 4.7 V Anode-Free Lithium Battery. Adv. Mater. 2022, 2207361. [Google Scholar] [CrossRef]
- Yun, Q.; He, Y.B.; Lv, W.; Zhao, Y.; Li, B.; Kang, F.; Yang, Q.H. Chemical dealloying derived 3D porous current collector for Li metal anodes. Adv. Mater. 2016, 28, 6932–6939. [Google Scholar] [CrossRef]
- Lu, L.-L.; Ge, J.; Yang, J.-N.; Chen, S.-M.; Yao, H.-B.; Zhou, F.; Yu, S.-H. Free-standing copper nanowire network current collector for improving lithium anode performance. Nano Lett. 2016, 16, 4431–4437. [Google Scholar] [CrossRef] [PubMed]
- Yue, Y.; Liang, H. 3D current collectors for lithium-ion batteries: A topical review. Small Methods 2018, 2, 1800056. [Google Scholar] [CrossRef]
- Cohn, A.P.; Metke, T.; Donohue, J.; Muralidharan, N.; Share, K.; Pint, C.L. Rethinking sodium-ion anodes as nucleation layers for anode-free batteries. J. Mater. Chem. A 2018, 6, 23875–23884. [Google Scholar] [CrossRef]
- Cohn, A.P.; Muralidharan, N.; Carter, R.; Share, K.; Pint, C.L. Anode-Free Sodium Battery through in Situ Plating of Sodium Metal. Nano Lett. 2017, 17, 1296–1301. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhang, H.; Wu, F.; Zarrabeitia, M.; Geiger, D.; Kaiser, U.; Varzi, A.; Passerini, S. Sodiophilic Current Collectors Based on MOF-Derived Nanocomposites for Anode-Less Na-Metal Batteries. Adv. Energy Mater. 2022, 12, 2202293. [Google Scholar] [CrossRef]
- Li, S.; Jiang, M.; Xie, Y.; Xu, H.; Jia, J.; Li, J. Developing High-Performance Lithium Metal Anode in Liquid Electrolytes: Challenges and Progress. Adv. Mater. 2018, 30, e1706375. [Google Scholar] [CrossRef]
- Jin, S.; Jiang, Y.; Ji, H.; Yu, Y. Advanced 3D Current Collectors for Lithium-Based Batteries. Adv. Mater. 2018, 30, 1802014. [Google Scholar] [CrossRef]
- Zheng, J.; Archer, L.A. Controlling electrochemical growth of metallic zinc electrodes: Toward affordable rechargeable energy storage systems. Sci. Adv. 2021, 7, eabe0219. [Google Scholar] [CrossRef]
- Pu, J.; Li, J.; Zhang, K.; Zhang, T.; Li, C.; Ma, H.; Zhu, J.; Braun, P.V.; Lu, J.; Zhang, H. Conductivity and lithiophilicity gradients guide lithium deposition to mitigate short circuits. Nat. Commun. 2019, 10, 1896. [Google Scholar] [CrossRef]
- Chen, Y.; Yue, M.; Liu, C.; Zhang, H.; Yu, Y.; Li, X.; Zhang, H. Long Cycle Life Lithium Metal Batteries Enabled with Upright Lithium Anode. Adv. Funct. Mater. 2019, 29, 1806752. [Google Scholar] [CrossRef]
- Zhang, W.; Zhuang, H.L.; Fan, L.; Gao, L.; Lu, Y. A “cation-anion regulation” synergistic anode host for dendrite-free lithium metal batteries. Sci. Adv. 2018, 4, eaar4410. [Google Scholar] [CrossRef]
- Chi, S.-S.; Liu, Y.; Song, W.-L.; Fan, L.-Z.; Zhang, Q. Prestoring Lithium into Stable 3D Nickel Foam Host as Dendrite-Free Lithium Metal Anode. Adv. Funct. Mater. 2017, 27, 1700348. [Google Scholar] [CrossRef]
- Liang, Z.; Yan, K.; Zhou, G.; Pei, A.; Zhao, J.; Sun, Y.; Xie, J.; Li, Y.; Shi, F.; Liu, Y.; et al. Composite lithium electrode with mesoscale skeleton via simple mechanical deformation. Sci. Adv. 2019, 5, eaau5655. [Google Scholar] [CrossRef]
- Wang, P.; Zhang, G.; Wei, X.Y.; Liu, R.; Gu, J.J.; Cao, F.F. Bioselective Synthesis of a Porous Carbon Collector for High-Performance Sodium-Metal Anodes. J. Am. Chem. Soc. 2021, 143, 3280–3283. [Google Scholar] [CrossRef]
- Payne, C.M.; Knott, B.C.; Mayes, H.B.; Hansson, H.; Himmel, M.E.; Sandgren, M.; Ståhlberg, J.; Beckham, G.T. Fungal Cellulases. Chem. Rev. 2015, 115, 1308–1448. [Google Scholar] [CrossRef]
- Lee, K.; Lee, Y.J.; Lee, M.J.; Han, J.; Lim, J.; Ryu, K.; Yoon, H.; Kim, B.H.; Kim, B.J.; Lee, S.W. A 3D Hierarchical Host with Enhanced Sodiophilicity Enabling Anode-Free Sodium-Metal Batteries. Adv. Mater. 2022, 34, e2109767. [Google Scholar] [CrossRef]
- Tang, S.; Zhang, Y.Y.; Zhang, X.G.; Li, J.T.; Wang, X.Y.; Yan, J.W.; Wu, D.Y.; Zheng, M.S.; Dong, Q.F.; Mao, B.W. Stable Na Plating and Stripping Electrochemistry Promoted by In Situ Construction of an Alloy-Based Sodiophilic Interphase. Adv Mater 2019, 31, e1807495. [Google Scholar] [CrossRef]
- Wang, G.; Zhang, Y.; Guo, B.; Tang, L.; Xu, G.; Zhang, Y.; Wu, M.; Liu, H.-K.; Dou, S.-X.; Wu, C. Core–Shell C@Sb Nanoparticles as a Nucleation Layer for High-Performance Sodium Metal Anodes. Nano Lett. 2020, 20, 4464–4471. [Google Scholar] [CrossRef]
- Wang, H.; Matios, E.; Wang, C.; Luo, J.; Lu, X.; Hu, X.; Zhang, Y.; Li, W. Tin nanoparticles embedded in a carbon buffer layer as preferential nucleation sites for stable sodium metal anodes. J. Mater. Chem. A 2019, 7, 23747–23755. [Google Scholar] [CrossRef]
- Sun, Z.; Jin, H.; Ye, Y.; Xie, H.; Jia, W.; Jin, S.; Ji, H. Guiding Sodium Deposition through a Sodiophobic–Sodiophilic Gradient Interfacial Layer for Highly Stable Sodium Metal Anodes. ACS Appl. Energy Mater. 2021, 4, 2724–2731. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, C.; Matios, E.; Luo, J.; Hu, X.; Yue, Q.; Kang, Y.; Li, W. Sodium Deposition with a Controlled Location and Orientation for Dendrite-Free Sodium Metal Batteries. Adv. Energy Mater. 2020, 10, 2002308. [Google Scholar] [CrossRef]
- Liu, C.; Xie, Y.; Li, H.; Xu, J.; Zhang, Z. In Situ Construction of Sodiophilic Alloy Interface Enabled Homogenous Na Nucleation and Deposition for Sodium Metal Anode. J. Electrochem. Soc. 2022, 169, 080521. [Google Scholar] [CrossRef]
- Bai, M.; Tang, X.; Liu, S.; Wang, H.; Liu, Y.; Shao, A.; Zhang, M.; Wang, Z.; Ma, Y. An anodeless, mechanically flexible and energy/power dense sodium battery prototype. Energy Environ. Sci. 2022, 15, 4686–4699. [Google Scholar] [CrossRef]
- Chen, Q.; He, H.; Hou, Z.; Zhuang, W.; Zhang, T.; Sun, Z.; Huang, L. Building an artificial solid electrolyte interphase with high-uniformity and fast ion diffusion for ultralong-life sodium metal anodes. J. Mater. Chem. A 2020, 8, 16232–16237. [Google Scholar] [CrossRef]
- Han, J.; He, G. Capacity-Limited Na–M foil Anode: Toward Practical Applications of Na Metal Anode. Small 2021, 17, 2102126. [Google Scholar] [CrossRef]
- Choi, J.-H.; Ha, C.-W.; Choi, H.-Y.; Lee, S.-M. Carbon embedded SnSb composite tailored by carbothermal reduction process as high performance anode for sodium-ion batteries. J. Ind. Eng. Chem. 2018, 60, 451–457. [Google Scholar] [CrossRef]
- Rehnlund, D.; Lindgren, F.; Böhme, S.; Nordh, T.; Zou, Y.; Pettersson, J.; Bexell, U.; Boman, M.; Edström, K.; Nyholm, L. Lithium trapping in alloy forming electrodes and current collectors for lithium based batteries. Energy Environ. Sci. 2017, 10, 1350–1357. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ni, Q.; Yang, Y.; Du, H.; Deng, H.; Lin, J.; Lin, L.; Yuan, M.; Sun, Z.; Sun, G. Anode-Free Rechargeable Sodium-Metal Batteries. Batteries 2022, 8, 272. https://doi.org/10.3390/batteries8120272
Ni Q, Yang Y, Du H, Deng H, Lin J, Lin L, Yuan M, Sun Z, Sun G. Anode-Free Rechargeable Sodium-Metal Batteries. Batteries. 2022; 8(12):272. https://doi.org/10.3390/batteries8120272
Chicago/Turabian StyleNi, Qiao, Yuejiao Yang, Haoshen Du, Hao Deng, Jianbo Lin, Liu Lin, Mengwei Yuan, Zemin Sun, and Genban Sun. 2022. "Anode-Free Rechargeable Sodium-Metal Batteries" Batteries 8, no. 12: 272. https://doi.org/10.3390/batteries8120272
APA StyleNi, Q., Yang, Y., Du, H., Deng, H., Lin, J., Lin, L., Yuan, M., Sun, Z., & Sun, G. (2022). Anode-Free Rechargeable Sodium-Metal Batteries. Batteries, 8(12), 272. https://doi.org/10.3390/batteries8120272








