Carbon/High-Entropy Alloy Nanocomposites: Synergistic Innovations and Breakthrough Challenges for Electrochemical Energy Storage
Abstract
1. Introduction
- Structural Stability: The high-entropy effect lowers the Gibbs free energy, enabling the alloy to maintain a single-phase structure under high-temperature, corrosive, or irradiation environments [32,33]. For instance, refractory HEA like Nb25Mo25Ta25W25 and V20Nb20Mo20Ta20W20 remain stable and disordered, retaining a single-phase body-centered cubic structure without superlattice reflections even after exposure to 1400 °C [34].
- Enhanced Catalytic Activity: Multi-element synergy tunes the d-band center position, optimizing the adsorption free energy of reaction intermediates. For example, the design of PtFeCoNiCu high-entropy alloys adheres to three core principles: atomic size compatibility (radius 1.24–1.39 Å), high mixing entropy (1.576 R), and electronic synergistic effects [36,37]. The comparable atomic sizes facilitate the formation of face-centered cubic (fcc) solid solutions, while the high mixing entropy suppresses elemental segregation through positive dissolution energies confirmed based on DFT calculations, thereby enhancing structural stability. The electronic effects manifest as follows: electronegativity differences induce electron transfer, resulting in a negative shift of 0.3 eV in Pt 4f binding energy and a downward shift of 0.36 eV in the d-band center, which optimizes the -OOH adsorption energy (ΔG = 0.369–0.428 eV). Specifically, Fe/Co/Ni synergistically activates the O-O bond with low activation barriers (0.291–0.326 eV), while Cu modulates electron density to prevent overoxidation. This multi-active-site synergistic mechanism endows the material with exceptional ORR performance, achieving a mass activity of 1.738 A/mg Pt (surpassing commercial Pt/C by 15.8 times) and unprecedented stability with only 3 mV decay after 10,000 cycles.
- Improved Ion Storage Capacity: Some HEAs possess open crystal structures, exhibiting lithium-ion diffusion coefficients significantly superior to graphite [38].
- Conductive Network Construction: Three-dimensional conductive frameworks formed by carbon nanotubes (CNTs) or reduced graphene oxide can reduce overall electrode resistance below 10 Ω/sq [43].
- Mechanical Stress Buffering: Carbon coatings (e.g., amorphous carbon) absorb volume changes of active materials through elastic deformation, enabling silicon anodes to achieve double the capacity after 800 cycles [44].
- Multi-scale Mass Transport Optimization: Hierarchically porous carbon (micro-meso-macro pores) provides rapid ion transport channels, allowing SCs to achieve power densities exceeding 512 kW/kg [45].
- Interfacial Chemistry Regulation: Heteroatom doping (N, S, P) alters the electron distribution of the carbon skeleton, enhancing the chemical anchoring of polysulfides (capacity decay rate < 0.032% per cycle over 500 cycles in Li-S batteries) [46].
- In Li-S batteries, carbon materials (especially porous carbon) provide physical confinement and some chemical adsorption to capture polysulfides, while HEAs exert strong chemical adsorption and efficient catalytic action to accelerate polysulfide conversion. This synergy forms a “physical confinement + chemical adsorption + efficient catalysis” triple mechanism, effectively suppressing the shuttle effect [56].
- In SIBs, the composite leverages the sodium-storage capacity and buffering effect of carbon (e.g., hard carbon) combined with the potentially suitable open structure of HEAs for Na+ storage, jointly improving rate performance and cycle stability [57].
2. Synergistic Mechanisms of Carbon/HEA Nanocomposites
2.1. Interfacial Electronic Reconstruction and Band Structure Modulation Mechanism
2.2. Nano-Confinement and Steric Hindrance Stabilization Mechanism
2.3. Multiscale Structure Synergy and Strain-Adaptive Mechanism
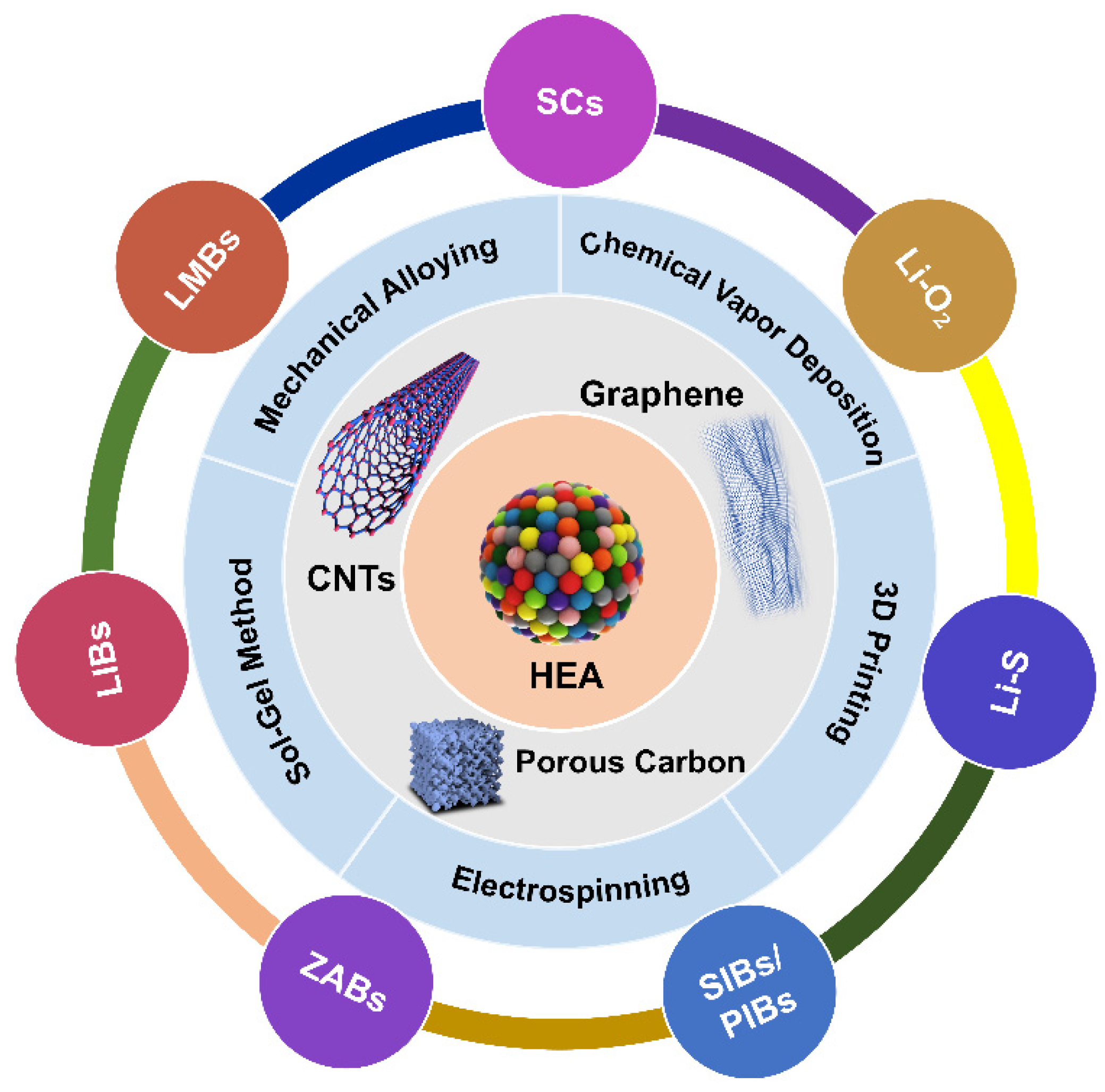
3. Preparation Methods
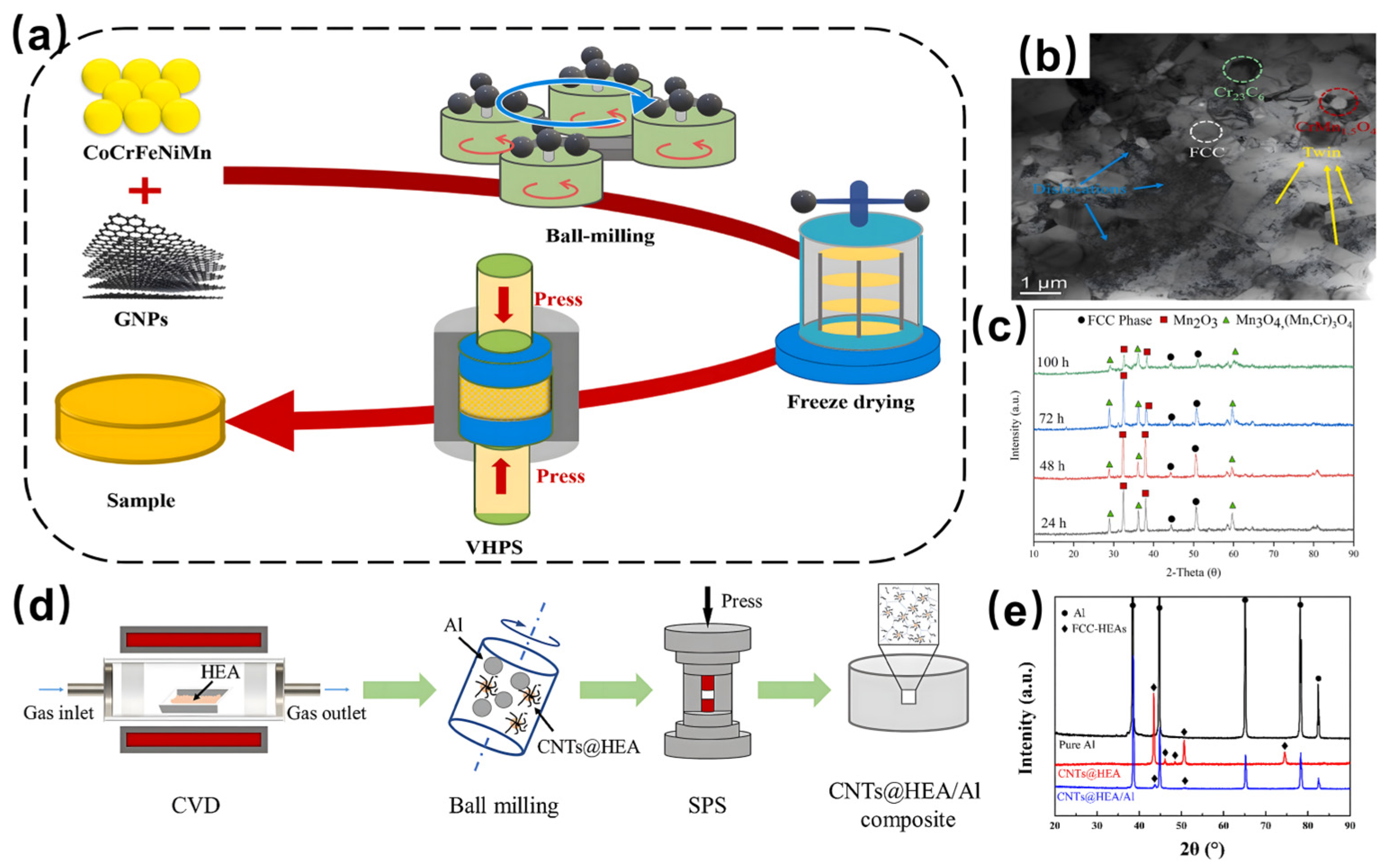
3.1. Mechanical Alloying
3.2. Chemical Vapor Deposition (CVD)
3.3. Sol-Gel Method
3.4. Electrospinning
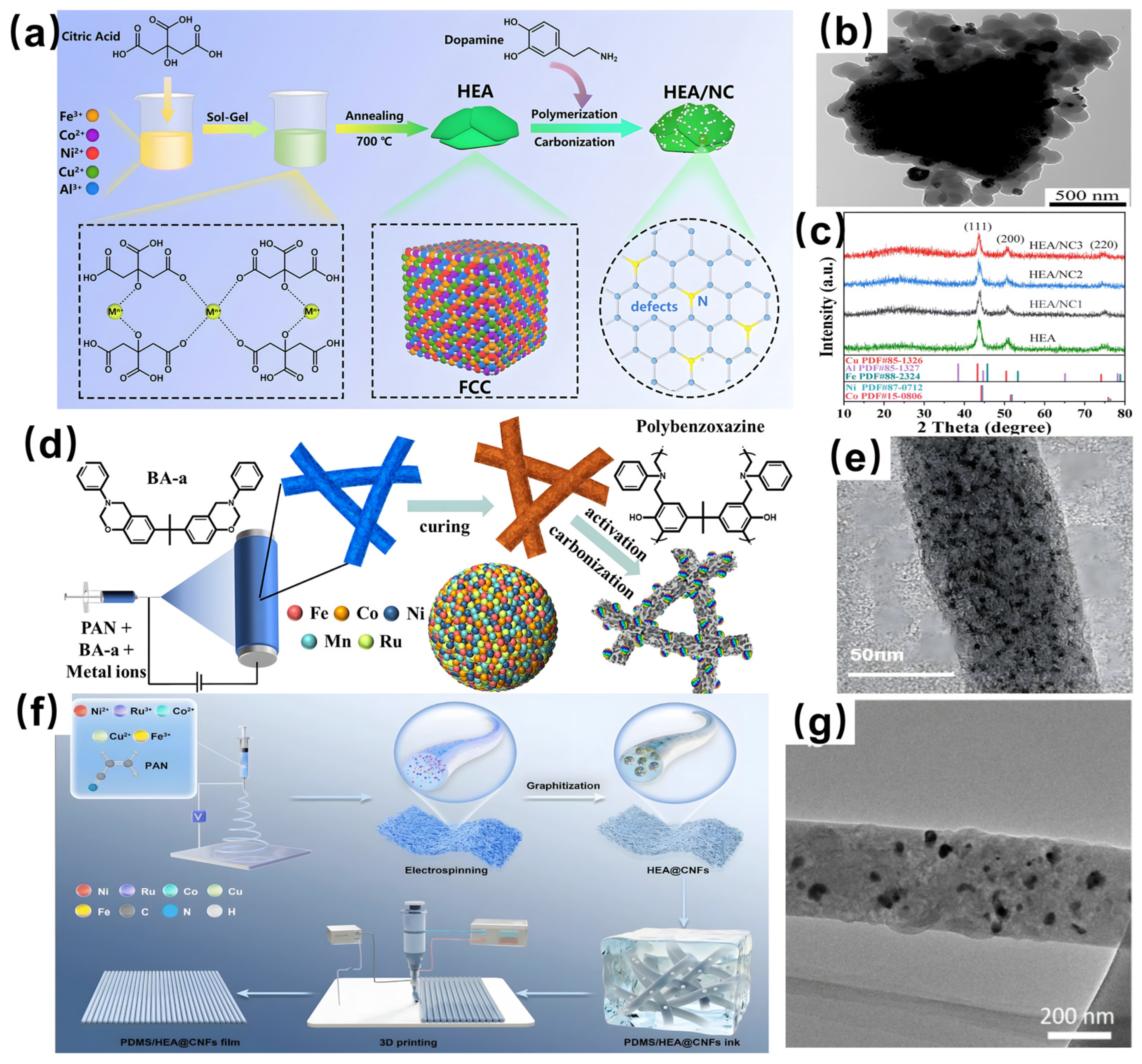
3.5. 3D Printing
4. EES Applications
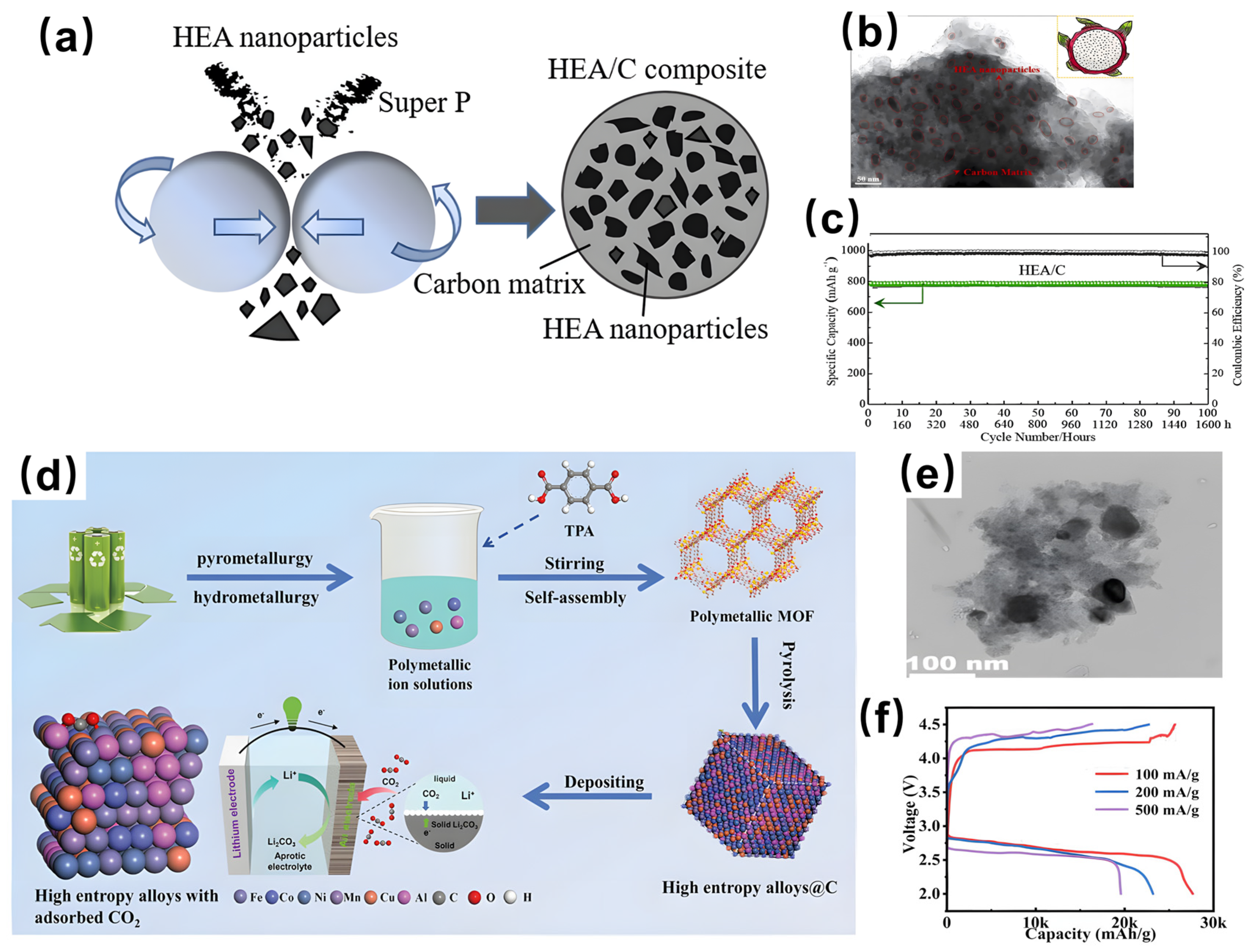
4.1. Lithium-Ion Batteries (LIBs)
4.2. Lithium Metal Batteries (LMBs)
4.3. Lithium-Oxygen Batteries (Li-O2)
4.4. Sodium/Potassium-Ion Batteries (SIBs/PIBs)
4.5. Lithium-Sulfur Batteries (Li-S)
4.6. Zinc–Air Batteries (ZABs)
4.7. Supercapacitors (SCs)
5. Summary and Outlook
- (1)
- Complex and Costly Synthesis: The multi-principal element nature of HEAs makes the precise control over synthesis difficult, requiring meticulous regulation of elemental ratios and reaction conditions. Traditional HEA preparation often relies on high temperatures, high pressures, and inert atmospheres, demanding stringent equipment specifications, significantly increasing energy consumption and production costs, and severely hindering large-scale production. Elemental segregation during multi-component mixing affects material homogeneity and reproducibility, leading to inconsistent product quality.
- (2)
- Ambiguous Interfacial Interaction Mechanisms: The interfacial bonding strength and interaction mechanisms between carbon materials and HEAs remain unclear. During electrochemical cycling, differences in thermal expansion coefficients and chemical activity can cause stress concentration and structural mismatch at the interface, reducing interfacial stability. This can lead to composite structural failure, shortening the cycle life of energy storage devices and impacting long-term operational stability.
- (3)
- Lagging Theoretical Research: While experiments demonstrate the excellent performance of carbon/HEA composites in EES, theoretical research providing a deep understanding of their intrinsic mechanisms at the atomic and electronic levels lags behind. The microscopic action mechanisms of high-entropy effects in complex composite systems are debated. The lack of comprehensive theoretical models hinders material design, performance optimization, and the full exploitation of material potential.
- (4)
- Performance-Cost Trade-off: Pursuing high performance often necessitates the use of rare or expensive elements in HEAs, increasing material costs. However, energy-storage devices are highly cost-sensitive in practical applications. Achieving a balance between high performance and low cost is a critical challenge for the large-scale adoption of these materials.
- (5)
- Slow Industrialization Process: Transitioning from lab-scale R&D to industrial production presents numerous obstacles for carbon/HEA composites. Existing preparation processes struggle to meet large-scale demands, lacking standardized production workflows and quality control systems. Furthermore, the incomplete industry chain and insufficient upstream–downstream collaboration further delay industrialization.
- (1)
- Precise Multi-scale Structural Design and Control:
- Atomic Scale: Utilize advanced computational simulations (e.g., Density Functional Theory—DFT) to precisely control the HEA elemental composition and ratio, optimize the electronic structure, and precisely regulate active sites.
- Nanoscale: Finely tune the HEA nanoparticle size, shape, and distribution, combined with the nanostructural features of carbon materials (e.g., graphene layer number, CNT diameter/length), to achieve efficient synergy.
- Macroscale: Construct composites with three-dimensionally ordered porous structures, optimizing the pore architecture and connectivity to promote ion transport and electrolyte wetting, enhancing the overall energy-storage performance. Examples include designing gradient-structured composites for efficient ion transport/storage across scales and boosting the energy density, power density, and cycling stability.
- (2)
- Cross-disciplinary Integration for New Material Systems: Integrate knowledge from materials science, chemistry, physics, and computational science to develop novel HEA systems and carbon/HEA composites.
- Materials Science: Focus on synthesis, structural characterization, and performance testing.
- Chemistry: Deepen understanding of reaction mechanisms and surface chemistry.
- Physics: Provide theoretical support from perspectives of electronic structure and energy conversion.
- Computational Science: Employ machine learning, molecular dynamics simulations, etc., to accelerate material screening and design. Use machine learning algorithms to screen HEA components with specific properties, combined with experimental validation, to develop high-performance composites for novel energy-storage/conversion devices (e.g., all-solid-state batteries, fuel cells), expanding application fields.
- (3)
- Innovation and Development of Sustainable Synthesis Technologies: Develop green, low-energy-consumption synthesis technologies for sustainable material production.
- Explore bio-templating methods, utilizing specific structures of biomacromolecules or microorganisms to synthesize complex-structured composites, reducing chemical usage and energy consumption.
- Optimize existing processes (e.g., improved Joule heating for precise control and higher energy efficiency, lowering costs).
- Enhance research on recycling and reuse technologies to achieve green practices throughout the material lifecycle, laying the foundation for large-scale application.
- (4)
- Deep Application of Intelligent In situ Characterization Techniques: Employ intelligent in situ characterization techniques:
- In situ XRD: Real-time monitoring of crystal structure evolution during cycling.
- In situ TEM: Observation of nanostructural and interfacial changes.
- In situ XPS: Analysis of surface element chemical states and electronic structure evolution.
- (5)
- Material Surface Engineering Optimization: Utilize surface modification techniques (e.g., ALD, CVD) to introduce functional coatings or active sites onto composite surfaces.
- Coatings can improve hydrophilicity/hydrophobicity, optimize electrolyte wettability, and promote fast ion transport.
- Introducing active sites can enhance electrocatalytic activity and reduce reaction overpotentials (e.g., depositing ultra-thin metal oxide coatings to boost ORR catalytic performance in ZABs, improving efficiency and stability).
- Surface engineering can also enhance corrosion resistance, preventing HEA oxidation/dissolution in electrolytes and extending device lifespan.
- (6)
- Research on Novel Electrolyte Compatibility: Develop novel electrolytes highly compatible with carbon/HEA composites for different systems (e.g., LIBs, Li-S).
- Create electrolytes with high ionic conductivity, wide electrochemical windows, and good thermal stability, suppressing polysulfide shuttling in Li-Ss.
- Improve electrode–electrolyte interfacial compatibility using special additives or ionic liquid solvents, enhancing the cycle performance and Coulombic efficiency.
- Deeply study interaction mechanisms between electrolytes and composite surfaces to optimize formulations, reduce side reactions, and improve device safety and reliability.
- (7)
- Research on Dynamic Response Mechanisms of Composite Structures: Utilize advanced characterization and numerical simulations to deeply investigate the dynamic response mechanisms during cycling:
- Structural evolution, stress distribution changes, and electrochemical reaction kinetics.
- Molecular dynamic simulations of HEA nanoparticle diffusion within the carbon matrix and interaction changes during cycling.Understanding these mechanisms aids in optimizing material design, improving stability and reliability under complex operating conditions, and providing a theoretical basis for high-performance devices.
- (8)
- Device Integration and System Optimization: Extend research from electrode materials to the level of complete energy-storage device systems for integration and optimization.
- Study the compatibility and synergy between internal components (e.g., electrode-separator–electrolyte interfaces).
- Optimize device structure design to increase energy/power density.
- Tailor thermal management, safety management, and energy-management strategies to application scenarios (e.g., develop efficient heat dissipation structures, design safety mechanisms against overcharge/overdischarge/thermal runaway).
Author Contributions
Funding
Conflicts of Interest
References
- Nandy, S.; Fortunato, E.; Martins, R. Green economy and waste management: An inevitable plan for materials science. Prog. Nat. Sci. 2022, 32, 1–9. [Google Scholar] [CrossRef]
- Larcher, D.; Tarascon, J.M. Towards greener and more sustainable batteries for electrical energy storage. Nat. Chem. 2014, 7, 19–29. [Google Scholar] [CrossRef] [PubMed]
- de la Peña, O.S.; Víctor, A.; Costa, R.D. Recent advances towards sustainable materials and processes for energy conversion and storage. Adv. Energy Mater. 2021, 11, 2102874. [Google Scholar] [CrossRef]
- Zhang, W.; Lu, J.; Guo, Z. Challenges and future perspectives on sodium and potassium ion batteries for grid-scale energy storage. Mater. Today 2021, 50, 400–417. [Google Scholar] [CrossRef]
- Hirsh, H.S.; Li, Y.; Tan, D.H.S.; Zhang, M.; Zhao, E.; Meng, Y.S. Sodium-ion batteries paving the way for grid energy storage. Adv. Energy Mater. 2020, 10, 2001274. [Google Scholar] [CrossRef]
- Fagiolari, L.; Sampò, M.; Lamberti, A.; Amici, J.; Francia, C.; Bodoardo, S.; Bella, F. Integrated energy conversion and storage devices: Interfacing solar cells, batteries and supercapacitors. Energy Storage Mater. 2022, 51, 400–434. [Google Scholar] [CrossRef]
- Baptista, A.C.; Martins, J.I.; Fortunato, E.; Martins, R.; Borges, J.P.; Ferreira, I. Thin and flexible bio-batteries made of electrospun cellulose-based membranes. Biosens. Bioelectron. 2011, 26, 2742–2745. [Google Scholar] [CrossRef]
- Ferreira, I.; Bras, B.; Correia, N.; Barquinha, P.; Fortunato, E.; Martins, R. Performance and applications. J. Disp. Technol. 2010, 6, 332–335. [Google Scholar] [CrossRef]
- Carvalho, J.T.; Correia, A.; Cordeiro, N.J.A.; Coelho, J.; Lourenço, S.A.; Fortunato, E.; Martins, R.; Pereira, L. MoS2 decorated carbon fiber yarn hybrids for the development of freestanding flexible supercapacitors. NPJ 2D Mater. Appl. 2024, 8, 20. [Google Scholar] [CrossRef]
- Goodenough, J.B.; Park, K.-S. The Li-ion rechargeable battery: A perspective. J. Am. Chem. Soc. 2013, 135, 1167–1176. [Google Scholar] [CrossRef]
- Sada, K.; Darga, J.; Manthiram, A. Challenges and prospects of sodium-ion and potassium-ion batteries for mass production. Adv. Energy Mater. 2023, 13, 2302321. [Google Scholar] [CrossRef]
- Wang, J.; Li, L.; Hu, H.; Hu, H.; Guan, Q.; Huang, M.; Jia, L.; Adenusi, H.; Tian, K.V.; Zhang, J.; et al. Toward dendrite-free metallic lithium anodes: From structural design to optimal electrochemical diffusion kinetics. ACS Nano 2022, 16, 17729–17760. [Google Scholar] [CrossRef]
- Wu, F.; Maier, J.; Yu, Y. Guidelines and trends for next-generation rechargeable lithium and lithium-ion batteries. Chem. Soc. Rev. 2020, 49, 1569–1614. [Google Scholar] [CrossRef]
- Fu, X.; Beatty, D.N.; Gaustad, G.G.; Ceder, G.; Roth, R.; Kirchain, R.E.; Bustamante, M.; Babbitt, C.; Olivetti, E.A. Perspectives on cobalt supply through 2030 in the face of changing demand. Environ. Sci. Technol. 2020, 54, 2985–2993. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Bi, Z.; Zhang, A.; Das, P.; Lin, H.; Wu, Z.-S. High-voltage and fast-charging lithium cobalt oxide cathodes: From key challenges and strategies to future perspectives. Engineering 2024, 37, 105–127. [Google Scholar] [CrossRef]
- Xiao, Y.; Xiao, J.; Zhao, H.; Li, J.; Zhang, G.; Zhang, D.; Guo, X.; Gao, H.; Wang, Y.; Chen, J.; et al. Prussian blue analogues for sodium-ion battery cathodes: A review of mechanistic insights, current challenges, and future pathways. Small 2024, 20, 2401957. [Google Scholar] [CrossRef] [PubMed]
- Olabi, A.G.; Abbas, Q.; Abdelkareem, M.A.; Alami, A.H.; Mirzaeian, M.; Sayed, E.T. Carbon-based materials for supercapacitors: Recent progress, challenges and barriers. Batteries 2022, 9, 19. [Google Scholar] [CrossRef]
- Wang, W.; Wang, Y.; Yuan, L.; You, C.; Wu, J.; Liu, L.; Ye, J.; Wu, Y.; Fu, L. Recent advances in modification strategies of silicon-based lithium-ion batteries. Nano Res. 2022, 16, 3781–3803. [Google Scholar] [CrossRef]
- Li, H.; Li, Y.; Zhang, L. Designing principles of advanced sulfur cathodes toward practical lithium-sulfur batterierus. SusMat 2022, 2, 34–64. [Google Scholar] [CrossRef]
- Shen, Z.; Huang, J.; Xie, Y.; Wei, D.; Chen, J.; Shi, Z. Solid electrolyte interphase on lithium metal anodes. ChemSusChem 2024, 17, e202301777. [Google Scholar] [CrossRef]
- Bruce, P.G.; Freunberger, S.A.; Hardwick, L.J.; Tarascon, J.-M. Li-O2 and Li-S batteries with high energy storage. Nat. Mater. 2011, 11, 19–29. [Google Scholar] [CrossRef]
- Versaci, D.; Canale, I.; Goswami, S.; Amici, J.; Francia, C.; Fortunato, E.; Martins, R.; Pereira, L.; Bodoardo, S. Molybdenum disulfide/polyaniline interlayer for lithium polysulphide trapping in lithium-sulphur batteries. J. Power Source. 2022, 521, 230945. [Google Scholar] [CrossRef]
- Yeh, J.W.; Chen, S.K.; Lin, S.J.; Gan, J.Y.; Chin, T.S.; Shun, T.T.; Tsau, C.H.; Chang, S.Y. Nanostructured high-entropy alloys with multiple principal elements: Novel alloy design concepts and outcomes. Adv. Eng. Mater. 2004, 6, 299–303. [Google Scholar] [CrossRef]
- Ma, Y.; Ma, Y.; Wang, Q.; Schweidler, S.; Botros, M.; Fu, T.; Hahn, H.; Brezesinski, T.; Breitung, B. High-entropy energy materials: Challenges and new opportunities. Energ. Environ. Sci. 2021, 14, 2883–2905. [Google Scholar] [CrossRef]
- Zhou, Z.; Ma, Y.; Brezesinski, T.; Breitung, B.; Wu, Y.; Ma, Y. Improving upon rechargeable battery technologies: On the role of high-entropy effects. Energ. Environ. Sci. 2025, 18, 19–52. [Google Scholar] [CrossRef]
- Ma, Y.; Du, H.; Zheng, S.; Zhou, Z.; Zhang, H.; Ma, Y.; Passerini, S.; Wu, Y. High-entropy approach vs. traditional doping strategy for layered oxide cathodes in alkali-metal-ion batteries: A comparative study. Energy Storage Mater. 2025, 79, 104295. [Google Scholar] [CrossRef]
- Ma, Y.; Zhou, Z.; Brezesinski, T.; Ma, Y.; Wu, Y. Stabilizing Layered Cathodes by High-Entropy Doping. Research 2024, 7, 0503. [Google Scholar] [CrossRef]
- Dong, Y.; Zhou, Z.; Ma, Y.; Zhang, H.; Meng, F.; Wu, Y.; Ma, Y. Layered-structured sodium-ion cathode materials: Advancements through high-entropy approaches. ACS Energy Lett. 2024, 11, 5096–5119. [Google Scholar] [CrossRef]
- Wang, T.; Tan, X.; Ma, Y.; Ma, Y.; Wu, Y. High-entropy materials regulating lithium polysulfides for advanced lithium-sulfur batteries. Chem. Eng. J. 2025, 519, 165580. [Google Scholar] [CrossRef]
- Oketola, A.M.; Adegbola, T.A.; Jamiru, T.; Ogunbiyi, O.; Salifu, S. Advances in high-entropy alloy research: Unraveling fabrication a techniques, microstructural transformations, and mechanical properties. J. Bio-Tribo-Corrosio. 2025, 11, 79. [Google Scholar] [CrossRef]
- Barman, S.; Gupta, K.K.; Dey, S. Molecular dynamics-based explanation of the reinforcement geometry effects on CNT/graphene-reinforced Al0.3 CoCrFeNi high-entropy alloys. Sci. Rep. 2025, 15, 24984. [Google Scholar] [CrossRef]
- Kumar, D. Recent advances in tribology of high entropy alloys: A critical review. Prog. Mater. Sci. 2023, 136, 101106. [Google Scholar] [CrossRef]
- Zhang, Y.; Zuo, T.T.; Tang, Z.; Gao, M.C.; Dahmen, K.A.; Liaw, P.K.; Lu, Z.P. Microstructures and properties of high-entropy alloys. Prog. Mater. Sci. 2014, 61, 1–93. [Google Scholar] [CrossRef]
- Senkov, O.N.; Wilks, G.B.; Scott, J.M.; Miracle, D.B. Mechanical properties of Nb25Mo25Ta25W25 and V20Nb20Mo20Ta20W20 refractory high entropy alloys. Intermetallics 2011, 19, 698–706. [Google Scholar] [CrossRef]
- Gludovatz, B.; Hohenwarter, A.; Catoor, D.; Chang, E.H.; George, E.P.; Ritchie, R.O. A fracture-resistant high-entropy alloy for cryogenic applications. Science 2014, 345, 1153–1158. [Google Scholar] [CrossRef]
- Ren, J.-T.; Chen, L.; Wang, H.-Y.; Yuan, Z.-Y. High-entropy alloys in electrocatalysis: From fundamentals to applications. Chem. Soc. Rev. 2023, 52, 8319–8373. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Ning, F.; Qi, J.; Feng, G.; Wang, Y.; Song, J.; Yang, T.; Liu, X.; Chen, L.; Xia, D. PtFeCoNiCu high-entropy solid solution alloy as highly efficient electrocatalyst for the oxygen reduction reaction. iScience 2023, 26, 105890. [Google Scholar] [CrossRef]
- Wei, Y.; Liu, X.; Yao, R.; Qian, J.; Yin, Y.; Li, D.; Chen, Y. Embedding the high entropy alloy nanoparticles into carbon matrix toward high performance Li-ion batteries. J. Alloys Compd. 2023, 938, 168610. [Google Scholar] [CrossRef]
- Zhao, J.; Wei, Z.; Chen, N.; Meng, F.; Tian, R.; Zeng, Y.; Du, F. High-entropy alloy anodes for low-strain and high-volumetric lithium-ion storage at ambient and subzero temperatures. Energy Storage Mater. 2024, 65, 103127. [Google Scholar] [CrossRef]
- Wu, D.-H.; Haq, M.U.; Zhang, L.; Feng, J.-J.; Yang, F.; Wang, A.-J. Noble metal-free FeCoNiMnV high entropy alloy anchored on N-doped carbon nanotubes with prominent activity and durability for oxygen reduction and zinc–air batteries. J. Colloid Interf. Sci. 2024, 662, 149–159. [Google Scholar] [CrossRef]
- Wang, X.; Yang, C.; Li, J.; Chen, X.A.; Yang, K.; Yu, X.; Lin, D.; Zhang, Q.; Wang, S.; Wang, J.; et al. Insights of heteroatoms doping-enhanced bifunctionalities on carbon based energy storage and conversion. Adv. Funct. Mater. 2020, 31, 2009109. [Google Scholar] [CrossRef]
- Sun, Y.; Sun, J.; Sanchez, J.S.; Xia, Z.; Xiao, L.; Chen, R.; Palermo, V. Surface chemistry and structure manipulation of graphene-related materials to address the challenges of electrochemical energy storage. Chem. Commun. 2023, 59, 2571–2583. [Google Scholar] [CrossRef]
- Li, X.; Zhang, G.; Bai, X.; Sun, X.; Wang, X.; Wang, E.; Dai, H. Highly conducting graphene sheets and Langmuir-Blodgett films. Nat. Nanotechnol. 2008, 3, 538–542. [Google Scholar] [CrossRef] [PubMed]
- Qi, C.; Li, S.; Yang, Z.; Xiao, Z.; Zhao, L.; Yang, F.; Ning, G.; Ma, X.; Wang, C.; Xu, J.; et al. Suitable thickness of carbon coating layers for silicon anode. Carbon 2022, 86, 530–538. [Google Scholar] [CrossRef]
- Xu, J.; Tan, Z.; Zeng, W.; Chen, G.; Wu, S.; Zhao, Y.; Ni, K.; Tao, Z.; Ikram, M.; Ji, H.; et al. A hierarchical carbon derived from sponge-templated activation of graphene oxide for high-performance supercapacitor electrodes. Adv. Mater. 2016, 28, 5222–5228. [Google Scholar] [CrossRef]
- Song, Z.; Lu, X.; Hu, Q.; Ren, J.; Zhang, W.; Zheng, Q.; Lin, D. Synergistic confining polysulfides by rational design a N/P co-doped carbon as sulfur host and functional interlayer for high-performance lithium-sulfur batteries. J. Power Source 2019, 421, 23–31. [Google Scholar] [CrossRef]
- Başgöz, Ö.; Güngör, A.; Güler, Ö.; Erdem, E. High-entropy alloys and oxides as supercapacitor electrodes: A structural and electrochemical perspective for energy storage. Adv. Sustain. Syst. 2025, 9, 2500201. [Google Scholar] [CrossRef]
- Yang, Z.; Tian, J.; Yin, Z.; Cui, C.; Qian, W.; Wei, F. Carbon nanotube- and graphene-based nanomaterials and applications in high-voltage supercapacitor: A review. Carbon 2019, 141, 467–480. [Google Scholar] [CrossRef]
- Zhang, J.; Jiang, J.; Li, H.; Zhao, X.S. A high-performance asymmetric supercapacitor fabricated with graphene-based electrodes. Energ. Environ. Sci. 2011, 4, 4009–4015. [Google Scholar] [CrossRef]
- Hussain, I.; Lamiel, C.; Ahmad, M.; Chen, Y.; Shuang, S.; Javed, M.S.; Yang, Y.; Zhang, K. High entropy alloys as electrode material for supercapacitors: A review. J. Energy Storage 2021, 44, 103405. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, Y. High-entropy alloys in catalyses and supercapacitors: Progress, prospects. Nano Energy 2022, 104, 107958. [Google Scholar] [CrossRef]
- Feng, D.; Dong, Y.; Nie, P.; Zhang, L.; Qiao, Z.-A. CoNiCuMgZn high entropy alloy nanoparticles embedded onto graphene sheets via anchoring and alloying strategy as efficient electrocatalysts for hydrogen evolution reaction. Chem. Eng. J. 2022, 430, 132883. [Google Scholar] [CrossRef]
- Ashwini, R.; Kumar, M.K.P.; Rekha, M.Y.; Santosh, M.S.; Srivastava, C. Optimization of NiFeCrCoCu high entropy alloy nanoparticle-graphene (HEA-G) composite for the enhanced electrochemical sensitivity towards urea oxidation. J. Alloys Compd. 2022, 903, 163846. [Google Scholar] [CrossRef]
- Xu, X.; Du, Y.; Wang, C.; Guo, Y.; Zou, J.; Zhou, K.; Zeng, Z.; Liu, Y.; Li, L. High-entropy alloy nanoparticles on aligned electronspun carbon nanofibers for supercapacitors. J. Alloys Compd. 2020, 822, 153642. [Google Scholar] [CrossRef]
- Wang, J.; Liu, W.; Wang, Y.; Guo, Y.; Liu, M.; Ye, C.; Wang, S.; Zou, Q. AgCuInCdZn high-entropy alloy nanoparticles-embedded in porous carbon fibers for long-cycling lithium metal anodes. Chem. Eng. J. 2023, 477, 146884. [Google Scholar] [CrossRef]
- Yao, Y.; Zhao, Z.; Niu, R.; Chen, J.; Wang, X. Rationally designed FeCoNiCrMn high-entropy alloys nanoparticles hybridized rGO modified separator for highly efficient lithium-sulfur batteries. Chem. Phys. Lett. 2024, 839, 141124. [Google Scholar] [CrossRef]
- Liu, B.; Li, M.; Wu, Y.; Ren, R.; Ge, Q.; Fan, L.; Qin, P.; Qian, J.; Ding, X. High-entropy BiSnSbCuAl nanoalloys conformed in carbon fibers as fast-charging and high-capacity anode material for sodium-ion batteries. J. Power Sources 2025, 652, 237600. [Google Scholar] [CrossRef]
- Ujah, C.O.; Kallon, D.V.V.; Aigbodion, V.S. Study on properties of high entropy alloys reinforced with carbon nanotubes/graphene-A review. J. Alloys Metall. Syst. 2024, 8, 100117. [Google Scholar] [CrossRef]
- Gao, X.; Chen, R.; Liu, T.; Fang, H.; Qin, G.; Su, Y.; Guo, J. High-entropy alloys: A review of mechanical properties and deformation mechanisms at cryogenic temperatures. J. Mater. Sci. 2022, 57, 6573–6606. [Google Scholar] [CrossRef]
- Bolar, S.; Ito, Y.; Fujita, T. Future prospects of high-entropy alloys as next-generation industrial electrode materials. Chem. Sci. 2024, 15, 8664–8722. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, Y.; Xing, P.; Li, X.; Du, Q.; Fan, X.; Cai, Z.; Yin, R.; Yao, Y.; Gan, W. Self-encapsulation of high-entropy alloy nanoparticles inside carbonized wood for highly durable electrocatalysis. Adv. Mater. 2024, 36, 2402391. [Google Scholar] [CrossRef]
- Zhao, M.; Wu, T.; Liu, D.; Huang, Y.; Zhao, L.; Tang, Y.; Shen, M.; Hu, Y.; Zhang, J.; Li, J.; et al. Effect of carbon fiber on microstructure evolution and surface properties of FeCoCrNiCu high-entropy alloy coatings. Mater. Corros. 2019, 71, 430–439. [Google Scholar] [CrossRef]
- Liu, Y.; Yuan, J.; Zhou, J.; Pan, K.; Zhang, R.; Zhao, R.; Li, L.; Huang, Y.; Liu, Z. Laser solid-phase synthesis of graphene shell-encapsulated high-entropy alloy nanoparticles. Light Sci. App. 2024, 13, 270. [Google Scholar] [CrossRef]
- Wang, Z.; Ge, H.; Liu, S.; Li, G.; Gao, X. High-entropy alloys to activate the sulfur cathode for lithium-sulfur batteries. Energy Environ. Mater. 2022, 6, e12358. [Google Scholar] [CrossRef]
- Pourghaz, A.; Rajabi, M.; Torabi, M.; Amirnejad, M. The impressive improvement of CoCrFeMnNi high entropy alloy mechanical properties and pitting corrosion resistance by incorporation of carbon nanotube reinforcement. Intermetallics 2023, 63, 108073. [Google Scholar] [CrossRef]
- Khan, I.; Khan, S.; Wu, S.-Y.; Liu, L.; Alodhayb, A.N.; Mead, J.L.; Ali, S.; Hassan, S.U.; Chen, H.-T.; Ju, S.-P.; et al. Advanced fabrication of graphene-integrated high-entropy alloy@carbon nanocomposites as superior multifunctional electrocatalysts. ACS Appl. Mater. Interfaces 2025, 17, 21033–21052. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Yang, P.; Wang, Y.; Wang, S. High entropy alloy nanoparticles decorated carbon-based electrode as interfacial Li-ion localized accelerators for anode-free lithium metal batteries. Carbon 2024, 228, 119432. [Google Scholar] [CrossRef]
- Xiao, W.; Xu, H.; Pei, Y.; Hu, L.; Yang, Z. High entropy alloy/graphite composite as a new strategy to enhance the performance of lithium-ion batteries. Inorg. Chem. Commun. 2025, 178, 114594. [Google Scholar] [CrossRef]
- Vaidya, M.; Muralikrishna, G.M.; Murty, B.S. High-entropy alloys by mechanical alloying: A review. J. Mater. Res. 2019, 34, 664–686. [Google Scholar] [CrossRef]
- Kumar, A.; Singh, A.; Suhane, A. Mechanically alloyed high entropy alloys: Existing challenges and opportunities. J. Mater. Res. Technol. 2022, 17, 2431–2456. [Google Scholar] [CrossRef]
- Martin, P.; Aguilar, C.; Cabrera, J.M. A review on mechanical alloying and spark plasma sintering of refractory high-entropy alloys: Challenges, microstructures, and mechanical behavior. J. Mater. Res. Technol. 2024, 30, 1900–1928. [Google Scholar] [CrossRef]
- Liu, C.; Jiang, X.; Sun, H.; Liu, T.; Wu, Z.; Yang, L. Steam oxidation properties of graphene reinforced bioinspired laminated CoCrFeNiMn high-entropy alloy matrix composites at 1000 °C. Mater. Today Commun. 2024, 38, 107962. [Google Scholar]
- Singh, S.; Shaikh, S.M.; Kumar MK, P.K.; Murty, B.S.; Srivastava, C. Microstructural homogenization and substantial improvement in corrosion resistance of mechanically alloyed FeCoCrNiCu high entropy alloys by incorporation of carbon nanotubes. Materialia 2020, 14, 100917. [Google Scholar] [CrossRef]
- Peng, Y.Z.; You, X.; Li, C.J.; Yang, C.M.Y.; Xu, Z.Y.; Lu, Q.; Xu, G.Y.; Wang, Y.R.; Feng, Z.X.; Bao, R.; et al. Strength-ductility synergy in a novel carbon nanotube-high entropy alloy co-reinforced aluminum matrix composite. Compos. Part A Appl. Sci. Manuf. 2024, 181, 108116. [Google Scholar] [CrossRef]
- Han, C.X.; Zhi, J.Q.; Zeng, Z.; Wang, Y.S.; Zhou, B.; Gao, J.; Wu, Y.X.; He, Z.Y.; Wang, X.M.; Yu, S.W. Synthesis and characterization of nano-polycrystal diamonds on refractory high entropy alloys by chemical vapour deposition. Appl. Surf. Sci. 2023, 623, 157108. [Google Scholar] [CrossRef]
- Lu, D.; Fu, X.; Guo, D.; Ma, W.; Sun, S.; Shao, G.; Zhou, Z. Challenges and opportunities in 2D high-entropy alloy electrocatalysts for sustainable energy conversion. SusMat 2023, 3, 730–748. [Google Scholar] [CrossRef]
- Hassan, S.U.; Hou, L.; Yang, Y.; Qamar, T.H.; Wang, S. Optimizing the electromagnetic wave attenuation properties of carbon encapsulated FeCoNiCuMnx high entropy alloy nanoparticles. Carbon 2024, 229, 119502. [Google Scholar] [CrossRef]
- Ciriminna, R.; Pagliaro, M. Open challenges in sol-gel science and technology. J. Sol-Gel Sci. Technol. 2022, 101, 29–36. [Google Scholar] [CrossRef]
- Wang, Q.; Wang, J.; Jiang, S.; Li, P. Recent progress in sol-gel method for designing and preparing metallic and alloy nanocrystals. Acta. Phys. Chim. Sin. 2019, 35, 1186–1206. [Google Scholar] [CrossRef]
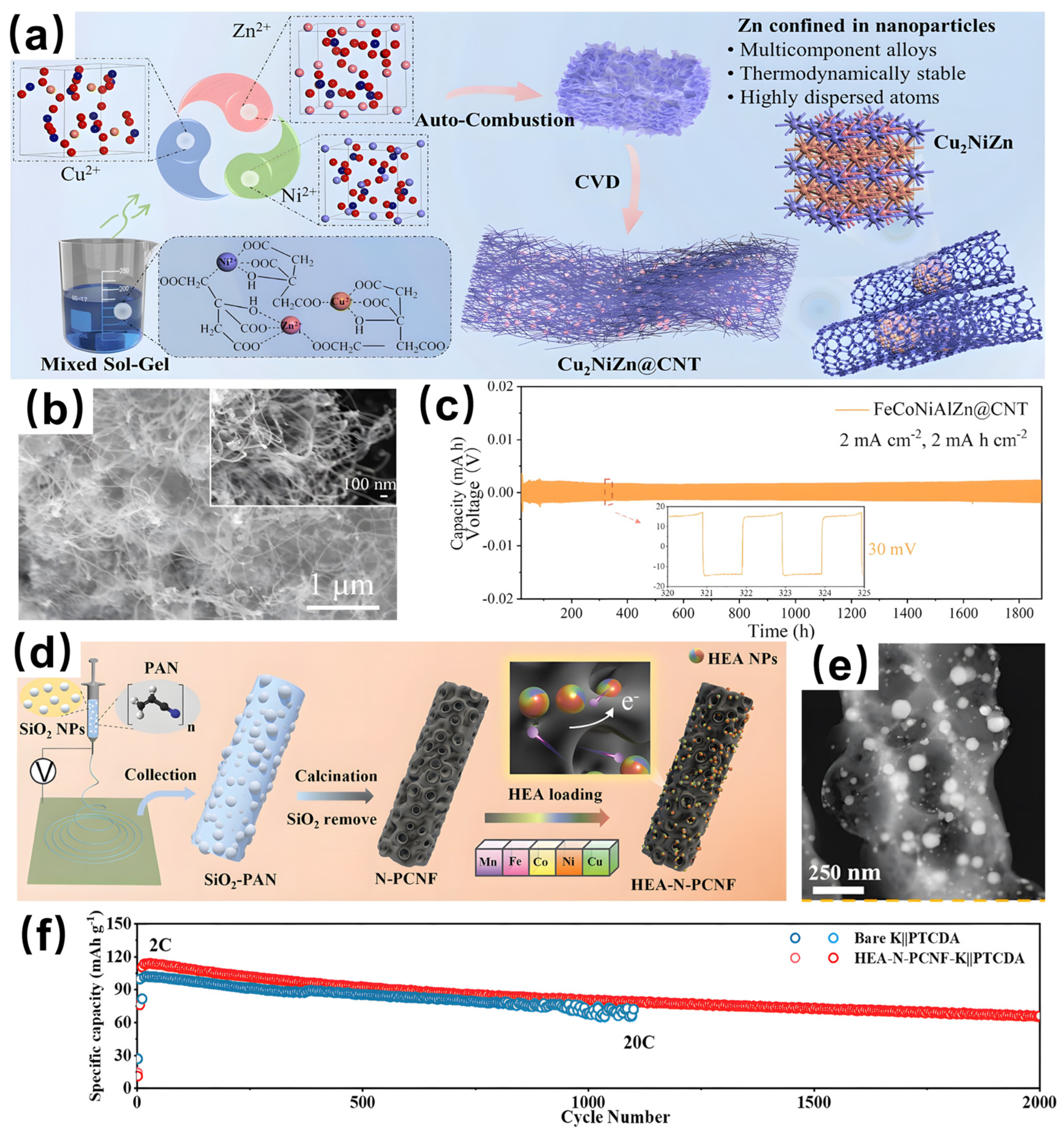
- Niu, B.; Zhang, F.; Ping, H.; Li, N.; Zhou, J.; Lei, L.; Xie, J.; Zhang, J.; Wang, W.; Fu, Z. Sol-gel autocombustion synthesis of nanocrystalline high-entropy alloys. Sci. Rep. 2017, 7, 3421. [Google Scholar] [CrossRef]
- Zhang, Q.; Ye, Y.; Sun, L.; Sun, P.; Wei, J.; Gan, Q. A heterojunction of high-entropy alloy and nitrogen-doped carbon nanospheres for efficient electromagnetic wave absorption. J. Mater. Chem. C 2025, 13, 7205–7218. [Google Scholar] [CrossRef]
- Yu, Y.; Cui, W.; Xu, Z.; Wang, S.; Jiang, W.; Sun, R.; Qi, L.; Pan, K. High-entropy Pt18Ni26Fe15Co14Cu27 nanocrystalline crystals in situ grown on reduced graphene oxide with excellent electromagnetic absorption properties. J. Colloid Interf. Sci. 2023, 639, 193–202. [Google Scholar] [CrossRef]
- Ji, D.; Lin, Y.; Guo, X.; Ramasubramanian, B.; Wang, R.; Radacsi, N.; Jose, R.; Qin, X.; Ramakrishna, S. Electrospinning of nanofibres. Nat. Rev. Methods Primers 2024, 4, 1. [Google Scholar] [CrossRef]
- Keirouz, A.; Wang, Z.; Reddy, V.S.; Nagy, Z.K.; Vass, P.; Buzgo, M.; Ramakrishna, S.; Radacsi, N. The history of electrospinning: Past, present, and future developments. Adv. Mater. Technol. 2023, 8, 2201723. [Google Scholar] [CrossRef]
- Li, H.; Zhu, H.; Sun, S.; Hao, J.; Zhu, Z.; Xu, F.; Lu, S.; Duan, F.; Du, M. Thermodynamically driven metal diffusion strategy for controlled synthesis of high-entropy alloy electrocatalysts. Chem. Commun. 2021, 57, 10027–10030. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Zhang, B.; Wen, X.; Wang, L.; Jin, M.; Wu, H.; Qin, S.; Yang, X.; Liu, Y.; Yu, G. Bottom-up synthesis of high-entropy alloy/carbon nanofiber with magnetic-dielectric synergy for low-filler and broadband electromagnetic wave absorption. ACS Appl. Nano Mater. 2024, 7, 26706–26716. [Google Scholar] [CrossRef]
- Wang, L.; Wang, C.; Mu, Y.; Fan, J.; Yang, X.; Yu, C.; Guo, B.; Zeng, G. Mesoporous high-entropy-alloy electrocatalysts via electrospinning for enhanced alkaline water electrolysis. Fuel 2025, 391, 134800. [Google Scholar] [CrossRef]
- Wu, H.; Qi, Y.; Yin, R.; Long, Y.; Song, J.; Xie, P.; Zhong, J.; Wang, C.; Hou, Q.; Fan, R.; et al. Biocompatible radio frequency epsilon-near-zero materials for wearable electronics. Adv. Compos. Hybrid Mater. 2024, 8, 32. [Google Scholar] [CrossRef]
- Haile, B.S.; Pal, V.; Pal, T.; Slathia, S.; Jigi, G.M.; Negedu, S.D.; Tiwari, N.; Singh, H.; Joseph, A.; Olu, F.E.; et al. Direct ink writing (3D printing) of robust, highly efficient, double-half-heusler thermoelectric high-entropy alloy. Adv. Eng. Mater. 2025, 27, 2402283. [Google Scholar] [CrossRef]
- Ai, J.; Liu, S.; Zhang, Y.; Han, Y.; Liu, B.; Yin, Y.; Ma, H.; Feng, J. 3D-printed high-entropy alloy nanoarchitectures. Small 2025, 21, 2409900. [Google Scholar] [CrossRef]
- Bajaj, D.; Feng, A.; Qu, S.; Chen, Z.; Li, D.; Chen, D.L. Deformation behavior of 3D-printed high-entropy alloys: A critical review. Adv. Eng. Mater. 2023, 26, 2300615. [Google Scholar] [CrossRef]
- Yan, T.; Ye, X.; He, E.; Gao, Q.; Wang, Y.; Ye, Y.; Wu, H. 3D-Printed (CoCrFeMnNi)3O4@C-GR dual core-shell composites: Multilevel control and mechanisms of microwave absorption performance. Chem. Eng. J. 2024, 497, 154777. [Google Scholar] [CrossRef]
- Yi, J.; Deng, Q.; Cheng, H.; Zhu, D.; Zhang, K.; Yang, Y. Unique hierarchically structured high-entropy alloys with multiple adsorption sites for rechargeable Li-CO2 batteries with high capacity. Small 2024, 20, 2401146. [Google Scholar] [CrossRef]
- Wen, Z.; Mu, X.; Sun, X.; Xu, Z.; Zheng, M.; Zhou, H.; He, P. Boosting Li-CO2 battery performance via high-entropy alloy catalysts: Insights into configurational entropy effect. Angew. Chem. Int. Edit. 2025, 64, e202424121. [Google Scholar] [CrossRef]
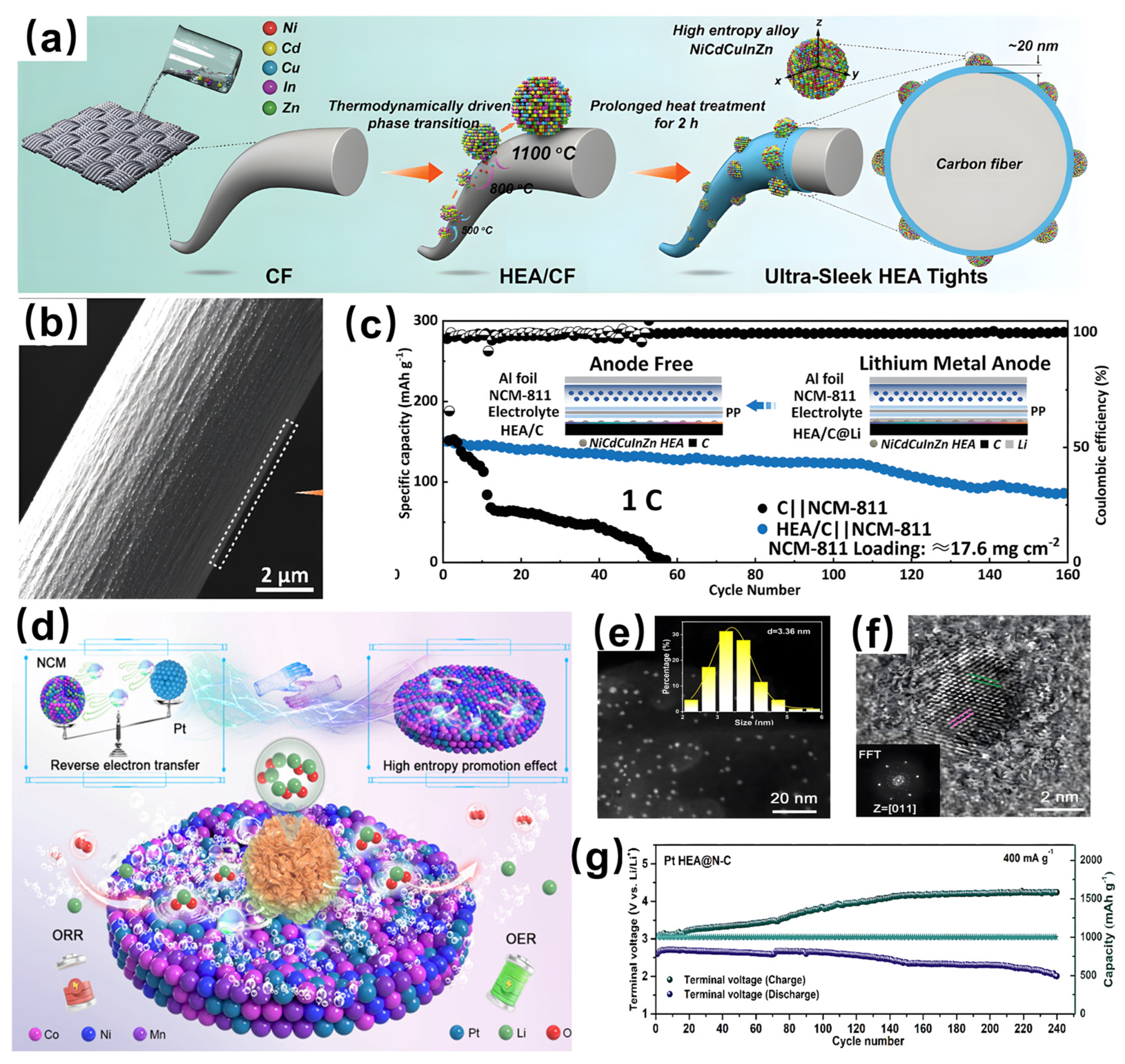
- Wang, J.; Wang, Y.; Lu, X.; Qian, J.; Yang, C.; Manke, I.; Song, H.; Liao, J.; Wang, S.; Chen, R. Ultra-sleek high entropy alloy tights: Realizing superior cyclability for anode-free battery. Adv. Mater. 2023, 36, 2308257. [Google Scholar] [CrossRef]
- Wang, P.; Guo, S.; Xu, Y.; Yuan, X.; Tian, Y.; Xu, B.; Zhao, Z.; Wang, Y.; Li, J.; Wang, X.; et al. Upcycling spent cathodes from Li-ion batteries into a high-entropy alloy catalyst with reverse electron transfer for Li-O2 batteries. ACS Nano 2025, 19, 17589–17605. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.; Zhang, Q.; Yang, Q.; Hu, Z.; Zhao, Y. High-entropy alloy nanoparticles functionalized with reduced graphene oxide as a high-performance cathode for lithium–oxygen batteries. Nanoscale 2025, 17, 10581–10588. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.; Rao, Y.; Shi, W.; Yang, J.; Ning, W.; Li, H.; Yao, Y.; Zhou, H.; Guo, S. Sabatier relations in electrocatalysts based on high-entropy alloys with wide-distributed d-band centers for Li-O2 batteries. Angew. Chem. Int. Ed. 2023, 62, e202310894. [Google Scholar] [CrossRef] [PubMed]
- Bai, M.; Tang, X.; Liu, S.; Wang, H.; Liu, Y.; Shao, A.; Zhang, M.; Wang, Z.; Ma, Y. An anodeless, mechanically flexible and energy/power dense sodium battery prototype. Energy Environ. Sci. 2022, 15, 4686–4699. [Google Scholar] [CrossRef]
- Zhang, R.; Xue, H.; Du, D.; Shen, Y.; Zheng, J.; Li, C.; Feng, Z.; Sun, T. Regulation of configurational entropy to realize long cycle lifespan of high entropy alloy anodes for potassium batteries. Adv. Funct. Mater. 2025, 35, 2422218. [Google Scholar] [CrossRef]
- Chang, C.B.; Tseng, Y.Y.; Lu, Y.R.; Yang, Y.C.; Tuan, H.Y. High entropy induced local charge enhancement promotes Frank-Van der merwe growth for dendrite-free potassium metal batteries. Adv. Funct. Mater. 2024, 34, 2411193. [Google Scholar] [CrossRef]
- Zheng, D.; Zha, J.; Wang, Y.; Wei, Z.; Qi, J.; Wei, F.; Meng, Q.; Xue, X.; Zhao, D.; Li, Y.; et al. Co0.2Sb0.2Fe0.2Mn0.2Ni0.2 high-entropy alloy carbon nanofiber as anode for lithium/potassium ion batteries. J. Mater. Sci. 2025, 60, 3926–3939. [Google Scholar] [CrossRef]
- Han, F.; Zhang, L.; Jin, Q.; Ma, X.; Zhang, Z.; Sun, Z.; Zhang, X.; Wu, L. High rate and long-cycle life of lithium-sulfur battery enabled by high d-band center of high-entropy alloys. ACS Nano 2025, 19, 9182–9195. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Ren, Y.; Sun, D.; Wang, B.; Wu, H.; Bian, H.; Cao, J.; Cao, X.; Ding, F.; Lu, J.; et al. High entropy alloy nanoparticles dual-decorated with nitrogen-doped carbon and carbon nanotubes as promising electrocatalysts for lithium-sulfur batteries. J. Mater. Sci. Technol. 2024, 188, 98–104. [Google Scholar] [CrossRef]
- Cao, X.; Gao, Y.; Wang, Z.; Zeng, H.; Song, Y.; Tang, S.; Luo, L.; Gong, S. FeNiCrCoMn high-entropy alloy nanoparticles loaded on carbon nanotubes as bifunctional oxygen catalysts for rechargeable zinc-air batteries. ACS Appl. Mater. Interfaces 2023, 15, 32365–32375. [Google Scholar] [CrossRef]
- Han, J.; Zhang, W.; Liu, K.; Zheng, H.; Li, Y.; Luo, L.; Gong, S.; Jia, Y.; Liang, X. PtFeCoNiMoY high-entropy alloy nanoparticles as bifunctional oxygen catalysts for zinc air batteries. Appl. Surf. Sci. 2025, 687, 162238. [Google Scholar] [CrossRef]
- Yao, Y.; Li, Z.; Dou, Y.; Jiang, T.; Zou, J.; Lim, S.Y.; Norby, P.; Stamate, E.; Jensen, J.O.; Zhang, W. High entropy alloy nanoparticles encapsulated in graphitised hollow carbon tubes for oxygen reduction electrocatalysis. Dalton Trans. 2023, 52, 4142–4151. [Google Scholar] [CrossRef]
- Shen, E.; Song, X.; Chen, Q.; Zheng, M.; Bian, J.; Liu, H. Spontaneously forming oxide layer of high entropy alloy nanoparticles deposited on porous carbons for supercapacitors. ChemElectroChem 2021, 8, 260–269. [Google Scholar] [CrossRef]
- Mohanty, G.C.; Verma, A. Supercapacitive study of a high entropy alloy and green carbon composite electrode with a wide potential window (1.3 V) and its application towards an aqueous symmetric device. New J. Chem. 2025, 49, 521–529. [Google Scholar] [CrossRef]

| Technology | Advantages | Disadvantages | Application Scenarios |
|---|---|---|---|
| Mechanical Alloying | Low cost, easy to scale up | Easy to introduce impurities, uneven particle size | Bulk materials, energy storage electrodes |
| CVD | Strong interface bonding, controllable structure | Expensive equipment, high energy consumption at a high temperature | Thin films, catalytic materials |
| Sol-Gel | Porous structure, high specific surface area | Calcination shrinkage, low mechanical strength | Porous electrodes, catalyst carriers |
| Electrospinning | Flexible fibers, high specific surface area | Low fiber strength, difficult mass production | Flexible devices, sensors |
| 3D Printing | Customized complex structures, high precision | Limited material selection, complex post-processing | Bionic structures, functional devices |
| Type | Material | Capacity | Stability | Ref. |
|---|---|---|---|---|
| LIBs | HEAs/C | 1462 mAh/g | 1600 h | [38] |
| HEA/C | 1881 mAh/g | 63.6% after 1000 cycles | [68] | |
| FeCoNiMnCuAl@C | 27,664 mAh/g | 134 cycles | [93] | |
| FeCoNiCuRu/CNFs | 6160 mAh/g | 550 cycles | [94] | |
| LMBs | HEA/PCF | 177.9 mAh/g | 99.5% after 200 cycles | [55] |
| HEA/CF | 197.9 mAh/g | 99.2% after 160 cycles | [67] | |
| HEA/C | 166.3 mAh/g | 99.5% after 160 cycles | [95] | |
| Li-O2 | Pt HEAs@N-C | 1000 mAh/g | 240 cycles | [96] |
| PtFeCoNiCu@rGO | 13,949 mAh/g | 148 cycles | [97] | |
| HEAs/C | 4000 mAh/g | 2000 h | [98] | |
| SIBs/PIBs | FeCoNiAlZn@CNT | 10 mAh/cm2 | 93.7% after 200 cycles | [99] |
| HEAs-NPs@NC | 513 mAh/g | 96.6% after 3000 cycles | [100] | |
| HEA-N-PCNF | 120 mAh/g | 2350 h | [101] | |
| HEA-CNFs | 1164 mAh/g | 1400 mAh/g after 1000 cycles | [102] | |
| Li-S | PCFCN-HEA/HCS/HCNB | 1221.6 mAh/g | 82.9% after 200 cycles | [103] |
| CNT/HEA-NC | 622.5 mA h/g | only 0.03% per cycle after 300 cycles | [104] | |
| FeCoNiCuMn HEA-NC | 1079.5 mAh/g | 0.3% per cycle for 160 cycles | [64] | |
| ZABs | PtFeCoNiMoY/CNT | 797 mA·h/g | 80 h | [106] |
| FeCoNiMnCu HEA@N-GHCT | 630.29 mA·h/g | >200 h | [107] | |
| Fe12Ni23Cr10Co30Mn25/CNT | 760 mA·h/g | 256 h | [105] | |
| SCs | HEA-NP@MOL/HCPC | 495.4 F/g | 94.7% after 15,000 cycles | [108] |
| FCNCM@Green carbon | 450 F/g | 94.7% after 15,000 cycles | [109] | |
| FeNiCoMnMg HEA-NPs/ACNFs | 203 F/g | 89.2% after 2000 cycles | [54] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, L.; Li, H.; Dong, Y.; Rong, W.; Zhou, N.; Dang, R.; Xu, J.; Cao, Q.; Pan, C. Carbon/High-Entropy Alloy Nanocomposites: Synergistic Innovations and Breakthrough Challenges for Electrochemical Energy Storage. Batteries 2025, 11, 317. https://doi.org/10.3390/batteries11090317
Sun L, Li H, Dong Y, Rong W, Zhou N, Dang R, Xu J, Cao Q, Pan C. Carbon/High-Entropy Alloy Nanocomposites: Synergistic Innovations and Breakthrough Challenges for Electrochemical Energy Storage. Batteries. 2025; 11(9):317. https://doi.org/10.3390/batteries11090317
Chicago/Turabian StyleSun, Li, Hangyu Li, Yu Dong, Wan Rong, Na Zhou, Rui Dang, Jianle Xu, Qigao Cao, and Chunxu Pan. 2025. "Carbon/High-Entropy Alloy Nanocomposites: Synergistic Innovations and Breakthrough Challenges for Electrochemical Energy Storage" Batteries 11, no. 9: 317. https://doi.org/10.3390/batteries11090317
APA StyleSun, L., Li, H., Dong, Y., Rong, W., Zhou, N., Dang, R., Xu, J., Cao, Q., & Pan, C. (2025). Carbon/High-Entropy Alloy Nanocomposites: Synergistic Innovations and Breakthrough Challenges for Electrochemical Energy Storage. Batteries, 11(9), 317. https://doi.org/10.3390/batteries11090317