Research on Modification of Fe3O4 Magnetic Nanoparticles with Two Silane Coupling Agents
Abstract
1. Introduction
2. Materials and Experiments
2.1. Materials
2.2. Experiments
2.3. Characterization
3. Results and Discussion
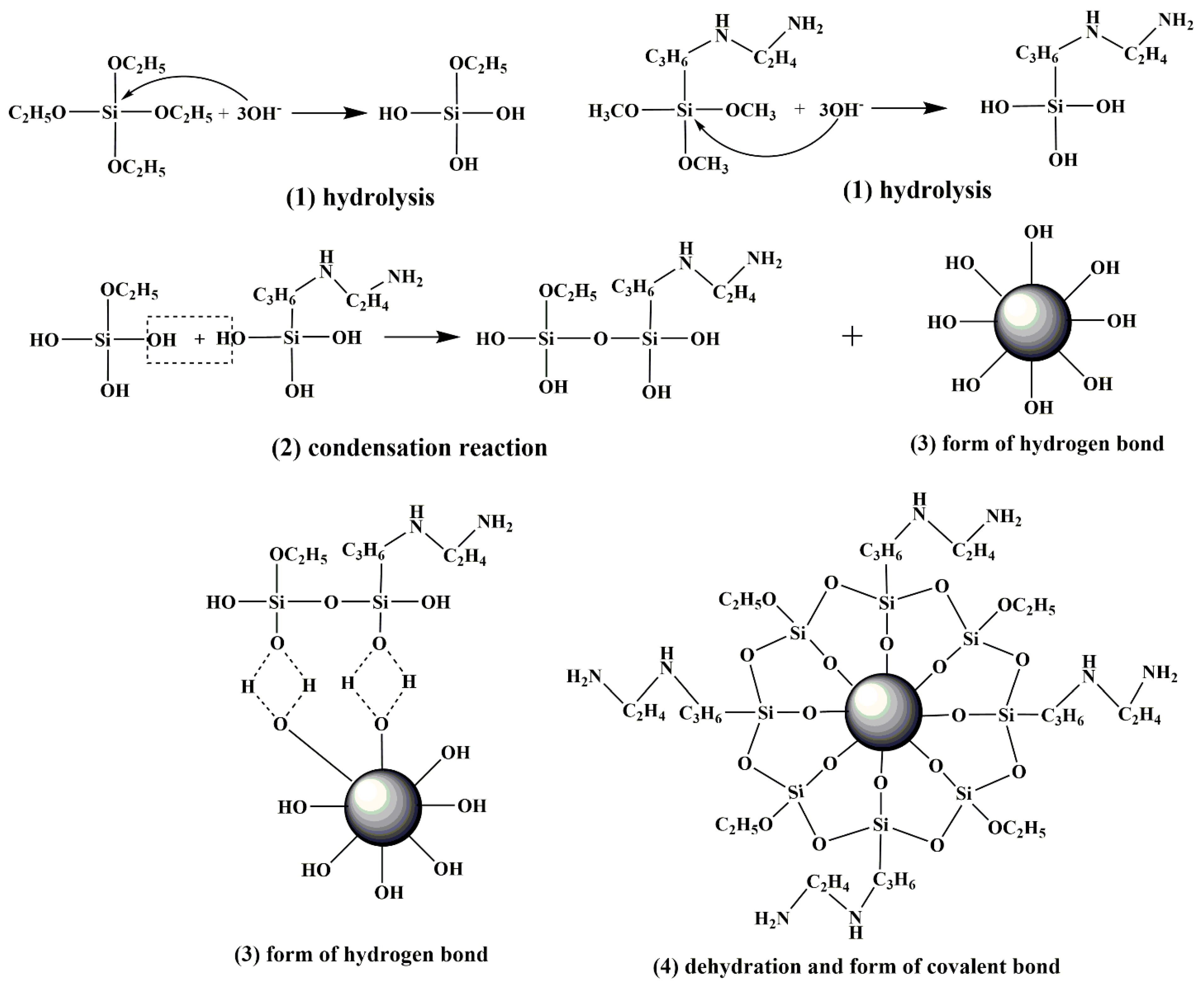
3.1. Research on Modification Mechanism of Two Silane Coupling Agents
3.2. Thermal Stability Analysis
3.3. Component Analysis
- (1)
- FT-IR spectroscopy analysis
- (2)
- X-ray photoelectron spectroscopy analysis
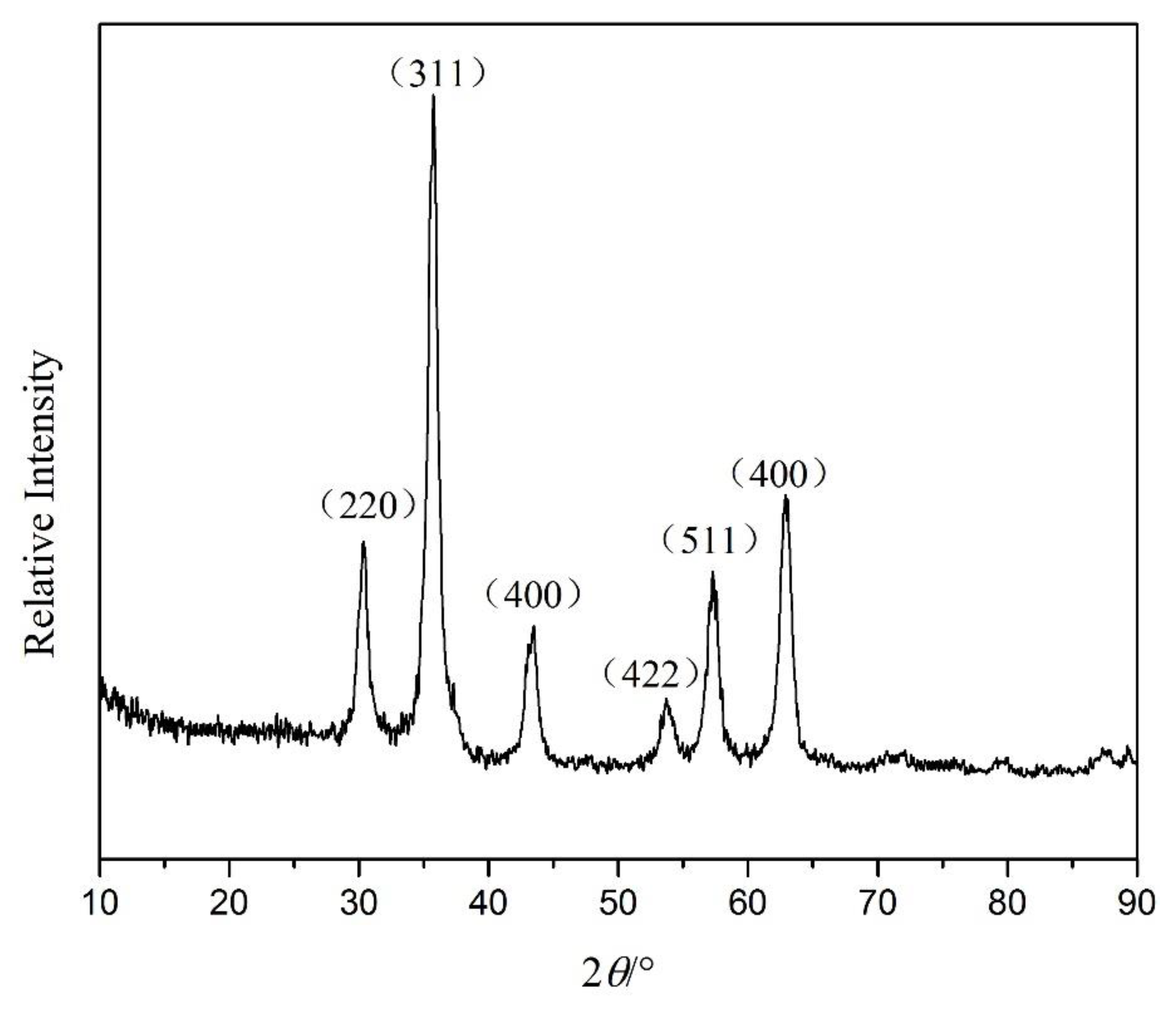
3.4. Crystal Structure
3.5. Micromorphology Analysis
3.6. Particle Size and Size Distribution
3.7. Low Surface Tension
3.8. Contact Angle
3.9. Magnetic Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yachi, T.; Matsubara, M.; Shen, C.; Asami, S.; Milbrandt, N.B.; Ju, M.; Wickramasinghe, S.; Samia, A.C.S.; Muramatsu, A.; Kanie, K. Water-Dispersible Fe3O4 Nanoparticles Modified with Controlled Numbers of Carboxyl Moieties for Magnetic Induction Heating. ACS Appl. Nano Mater. 2021, 4, 7395–7403. [Google Scholar] [CrossRef]
- Mehrez, Z.; El Cafsi, A. Heat exchange enhancement of ferrofluid flow into rectangular channel in the presence of a magnetic field. Appl. Math. Comput. 2021, 391, 14. [Google Scholar] [CrossRef]
- Frey, N.A.; Peng, S.; Cheng, K.; Sun, S. Magnetic Nanoparticles:Synthesis, Functionalization, and Applications in Bioimaging and Magnetic Energy Storage. Chem. Soc. Rev. 2009, 38, 2532–2542. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.Z.; Yan, X.Y. Nanozymes: Biomedical Applications of Enzymatic Fe3O4 Nanoparticles from In Vitro to In Vivo. In Biological and Bio-Inspired Nanomaterials: Properties and Assembly Mechanisms; Perrett, S., Buell, A.K., Knowles, T.P.J., Eds.; Springer: Berlin/Heidelberg, Germany, 2019; Volume 1174, pp. 291–312. [Google Scholar]
- Lee, J.S.; Song, Y.J.; Hsu, H.S.; Lin, C.R.; Huang, J.Y.; Chen, J. Magnetic enhancement of carbon-encapsulated magnetite nanoparticles. J. Alloy. Compd. 2019, 790, 716–722. [Google Scholar] [CrossRef]
- Guo, Z.W.; Adolfsson, E.; Tam, P.L. Nanostructured micro particles as a low-cost and sustainable catalyst in the recycling of PET fiber waste by the glycolysis method. Waste Manag. 2021, 126, 559–566. [Google Scholar] [CrossRef]
- Guo, Y.; Zhang, D.Y.; Yang, Y.; Wang, Y.B.; Bai, Z.X.; Chu, P.K.; Luo, Y.S. MXene-encapsulated hollow Fe3O4 nanochains embedded in N-doped carbon nanofibers with dual electronic pathways as flexible anodes for high-performance Li-ion batteries. Nanoscale 2021, 13, 4624–4633. [Google Scholar] [CrossRef]
- Han, J.; Wartenberg, M.G.; Chan, H.L.; Derby, B.K.; Li, N.; Scully, J.R. Electrochemical stability, physical, and electronic properties of thermally pre-formed oxide compared to artificially sputtered oxide on Fe thin films in aqueous chloride. Corros. Sci. 2021, 186, 109456. [Google Scholar] [CrossRef]
- Zeng, Y.; Zhou, N.; Xiong, C.H.; Huang, Z.Y.; Du, G.P.; Fan, Z.Y.; Chen, N. Highly stretchable silicone rubber nanocomposites incorporated with oleic acid-modified Fe3O4 nanoparticles. J. Appl. Polym. Sci. 2022, 139, 51476. [Google Scholar] [CrossRef]
- Nguyen, H.; Wang, Y.Z.; Moglia, D.; Fu, J.Y.; Zheng, W.Q.; Orazov, M.; Vlachos, D.G. Production of renewable oleo-furan surfactants by cross-ketonization of biomass-derived furoic acid and fatty acids. Catal. Sci. Technol. 2021, 11, 2762–2769. [Google Scholar] [CrossRef]
- Choudhary, S.; Sachdeva, A.; Kumar, P. Time-based analysis of stability and thermal efficiency of flat plate solar collector using iron oxide nanofluid. Appl. Therm. Eng. 2021, 183, 13. [Google Scholar] [CrossRef]
- Seal, P.; Alam, A.; Borgohain, C.; Paul, N.; Babu, P.D.; Borah, J.P. Optimization of self heating properties of Fe3O4 using PEG and amine functionalized MWCNT. J. Alloy. Compd. 2021, 882, 9. [Google Scholar] [CrossRef]
- Chen, H.; Zheng, J.; Qiao, L.; Ying, Y.; Jiang, L.; Che, S. Surface modification of NdFe12Nx magnetic powder using silane coupling agent KH550. Adv. Powder Technol. 2015, 26, 618–621. [Google Scholar] [CrossRef]
- Zhu, K.; Duan, Y.; Wang, F.; Gao, P.; Jia, H.; Ma, C.; Wang, C. Silane-modified halloysite/Fe3O4 nanocomposites: Simultaneous removal of Cr(VI) and Sb(V) and positive effects of Cr(VI) on Sb(V) adsorption. Chem. Eng. J. 2017, 311, 236–246. [Google Scholar] [CrossRef]
- Yang, W.; Shi, Z.; Li, M.; Zhang, Y.; Wang, Z. Surface Organic Modification of Nano-Sb2O3 Particles with Silane Coupling Agent. Integr. Ferroelectr. 2020, 208, 83–90. [Google Scholar] [CrossRef]
- Mohammad-Beigi, H.; Yaghmaei, S.; Roostaazad, R.; Bardania, H.; Arpanaei, A. Effect of pH, citrate treatment and silane-coupling agent concentration on the magnetic, structural and surface properties of functionalized silica-coated iron oxide nanocomposite particles. Phys. E 2011, 44, 618–627. [Google Scholar] [CrossRef]
- Abedi, M.; Abolmaali, S.S.; Abedanzadeh, M.; Borandeh, S.; Samani, S.M.; Tamaddon, A.M. Citric acid functionalized silane coupling versus post-grafting strategy for dual pH and saline responsive delivery of cisplatin by Fe3O4/carboxyl functionalized mesoporous SiO2 hybrid nanoparticles: A-synthesis, physicochemical and biological characterization. Mater. Sci. Eng. C 2019, 104, 109922. [Google Scholar] [CrossRef]
- Slimani, S.; Meneghini, C.; Abdolrahimi, M.; Talone, A.; Murillo, J.P.M.; Barucca, G.; Yaacoub, N.; Imperatori, P.; Illes, E.; Smari, M.; et al. Spinel Iron Oxide by the Co-Precipitation Method: Effect of the Reaction Atmosphere. Appl. Sci. 2021, 11, 5433. [Google Scholar] [CrossRef]
- Eskandari, M.J.; Hasanzadeh, I. Size-controlled synthesis of Fe3O4 magnetic nanoparticles via an alternating magnetic field and ultrasonic-assisted chemical co-precipitation. Mater. Sci. Eng. B 2021, 266, 115050. [Google Scholar] [CrossRef]
- Joseph, Y.; Ranke, W.; Weiss, W. Water on FeO(111) and Fe3O4(111): Adsorption Behavior on Different Surface Terminations. J. Phys. Chem. B 2000, 104, 3224–3236. [Google Scholar] [CrossRef]
- Breitbach, M.; Bathen, D.; Schmidt-Traub, H. Effect of Ultrasound on Adsorption and Desorption Processes. Ind. Eng. Chem. Res. 2003, 42, 5635–5646. [Google Scholar] [CrossRef]
- Cornell, R.M.; Schwertmann, U. The Iron Oxides: Structure, Properties, Reactions, Occurrences and Uses; John Wiley & Sons: New York, NY, USA, 1996. [Google Scholar]
- Cui, H.C.; Li, D.C.; Zhang, Z.L. Preparation and characterization of Fe3O4 magnetic nanoparticles modified by perfluoropolyether carboxylic acid surfactant. Mater. Lett. 2015, 143, 38–40. [Google Scholar] [CrossRef]
- Lin, C.L.; Lee, C.F.; Chiu, W.Y. Preparation and Properties of Poly(acrylic Acid) Oligomer Stabilized Superparamagnetic Ferrofluid. J. Colloid Interface Sci. 2005, 291, 411–420. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.c.; Hou, Y.l.; Sun, S.h. Magnetic Core/Shell Fe3O4/Au and Fe3O4/Au/Ag Nanoparticles with Tunable Plasmonic Properties. JACS 2007, 129, 8698–8699. [Google Scholar] [CrossRef]
- Maity, D.; Agrawal, D.C. Synthesis of Iron Oxide Nanoparticles Under Oxidizing Environment and Their Stabilization in Aqueous and Non-aqueous Media. J. Magn. Magn. Mater. 2007, 308, 46–55. [Google Scholar] [CrossRef]
- Bhattacharya, S.; Gao, Y.; Korampally, V.; Othman, M.T.; Grant, S.A.; Gangopadhyay, K.; Gangopadhyay, S. Mechanics of plasma exposed spin-on-glass (SOG) and polydimethyl siloxane (PDMS) surfaces and their impact on bond strength. Appl. Surf. Sci. 2007, 253, 4220–4225. [Google Scholar] [CrossRef]
- Liao, M.H.; Chen, D.H. Preparation and Characterization of a Novel Magnetic Nano-adsorbent. J. Mater. Chem. 2002, 12, 3654–3659. [Google Scholar] [CrossRef]
- Yamaura, M.; Camilo, R.L.; Sampaio, L.C.; Macedo, M.A.; Nakamura, M.; Toma, H.E. Preparation and Characterization of (3-aminopropyl) Triethoxysilane-coated Magnetite Nanoparticles. J. Magn. Magn. Mater. 2004, 279, 210–217. [Google Scholar] [CrossRef]
- Peddis, D.; Cannas, C.; Musinu, A.; Piccaluga, G. Magnetism in Nanoparticles: Beyond the Effect of Particle Size. Chem.-A Eur. J. 2009, 15, 7822–7829. [Google Scholar] [CrossRef]









| Element | Fe2p | O1s | Si2p | C1s | N1s |
|---|---|---|---|---|---|
| Content | 17.34 | 51.49 | 9.14 | 17.61 | 4.42 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cui, H.; Zhang, J.; Lu, J.; Li, Z.; Li, D. Research on Modification of Fe3O4 Magnetic Nanoparticles with Two Silane Coupling Agents. Magnetochemistry 2023, 9, 1. https://doi.org/10.3390/magnetochemistry9010001
Cui H, Zhang J, Lu J, Li Z, Li D. Research on Modification of Fe3O4 Magnetic Nanoparticles with Two Silane Coupling Agents. Magnetochemistry. 2023; 9(1):1. https://doi.org/10.3390/magnetochemistry9010001
Chicago/Turabian StyleCui, Hongchao, Jiajia Zhang, Jingjing Lu, Zhenkun Li, and Decai Li. 2023. "Research on Modification of Fe3O4 Magnetic Nanoparticles with Two Silane Coupling Agents" Magnetochemistry 9, no. 1: 1. https://doi.org/10.3390/magnetochemistry9010001
APA StyleCui, H., Zhang, J., Lu, J., Li, Z., & Li, D. (2023). Research on Modification of Fe3O4 Magnetic Nanoparticles with Two Silane Coupling Agents. Magnetochemistry, 9(1), 1. https://doi.org/10.3390/magnetochemistry9010001







