Chiral Dy(III) Fluorescent Single-Molecule Magnet Based on an Achiral Flexible Ligand
Abstract
1. Introduction
2. Results
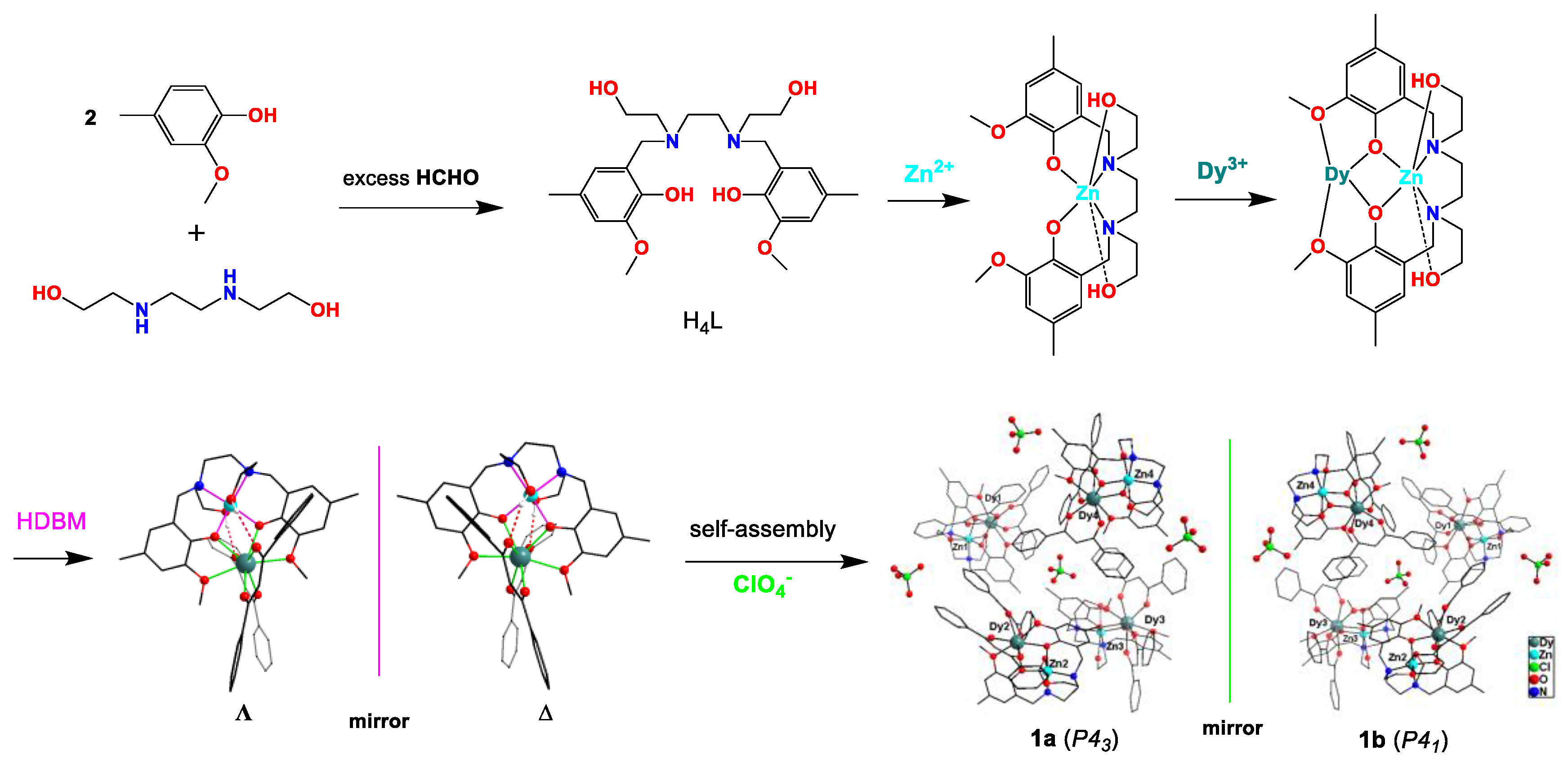
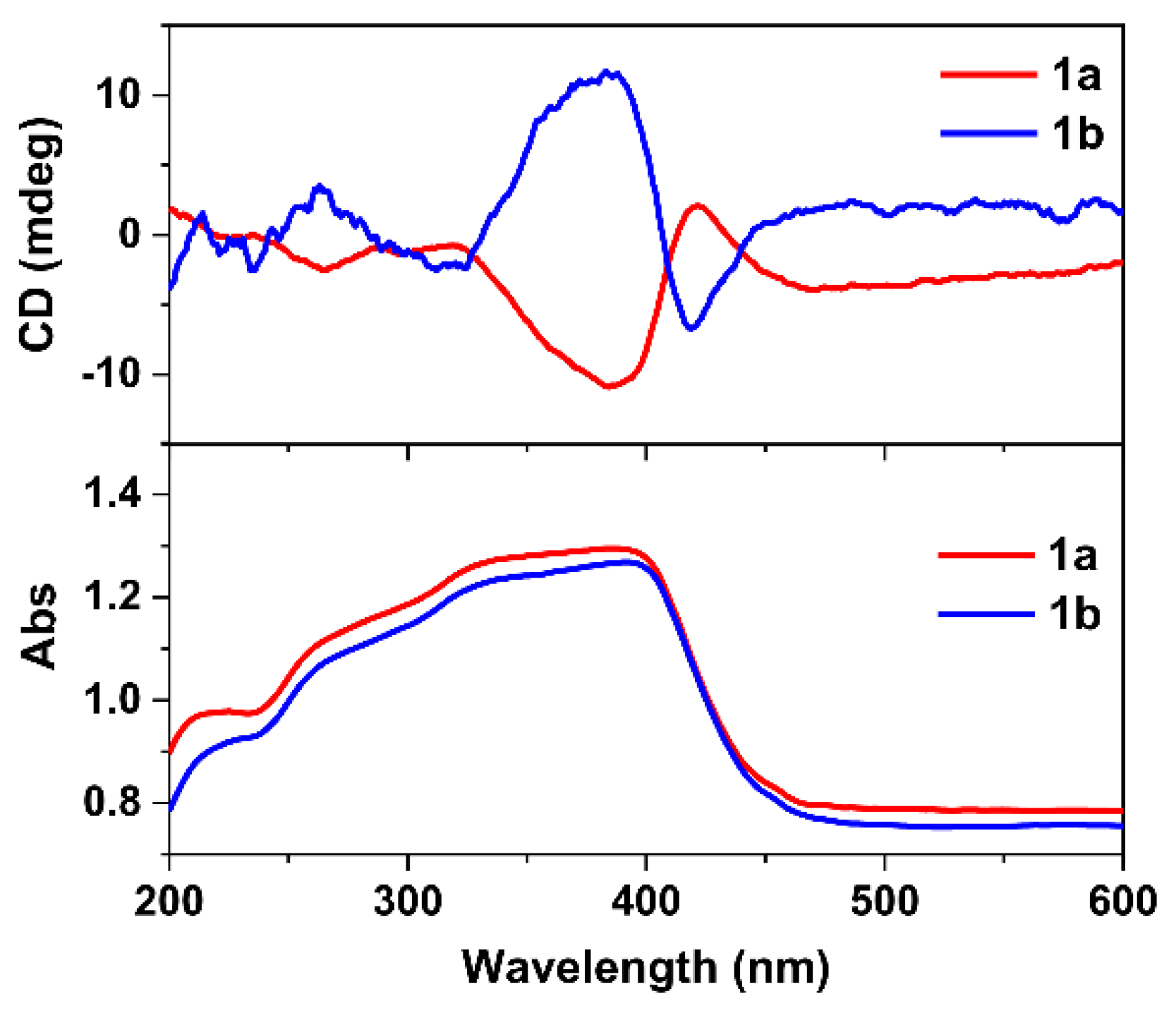
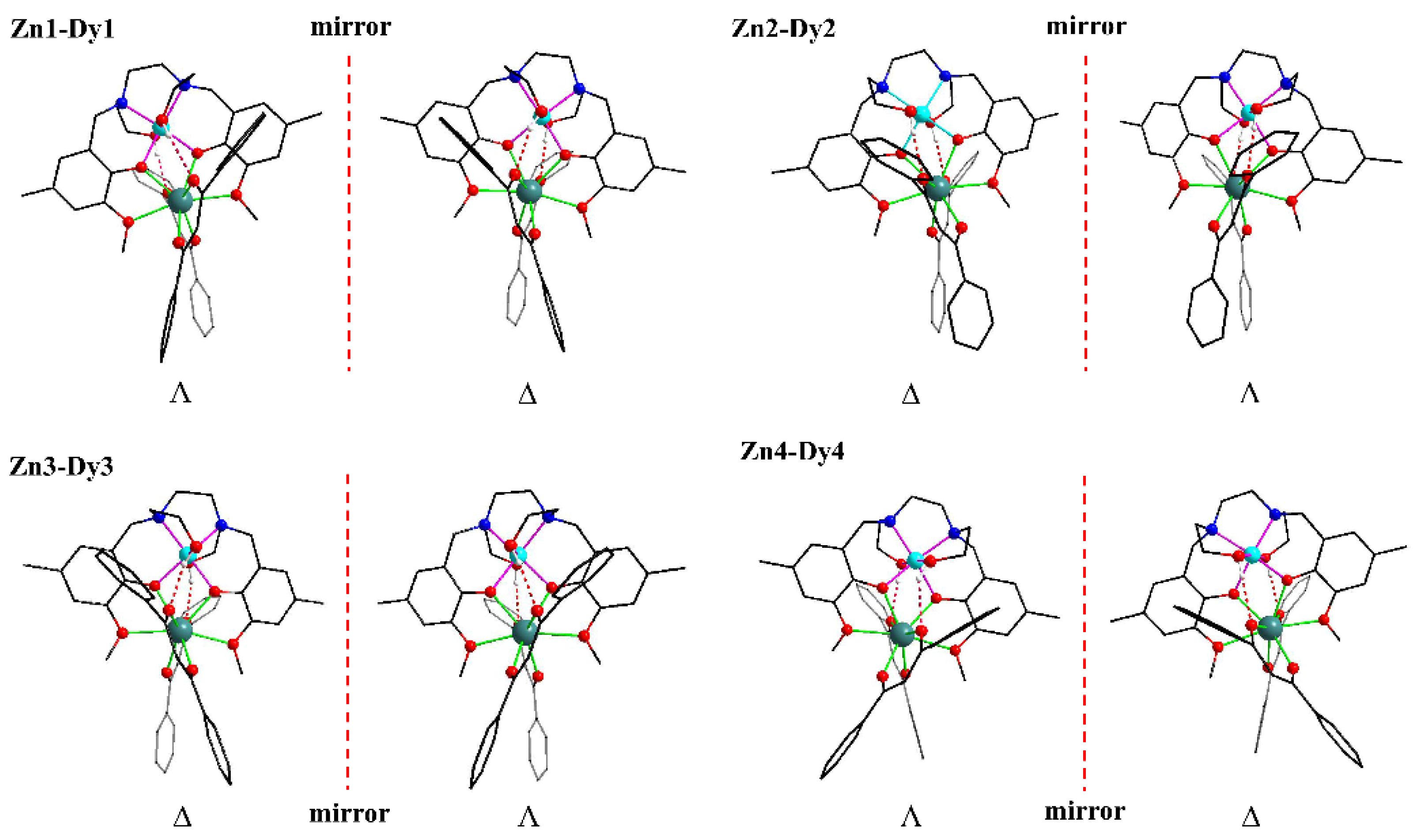
2.1. Crystal Structures
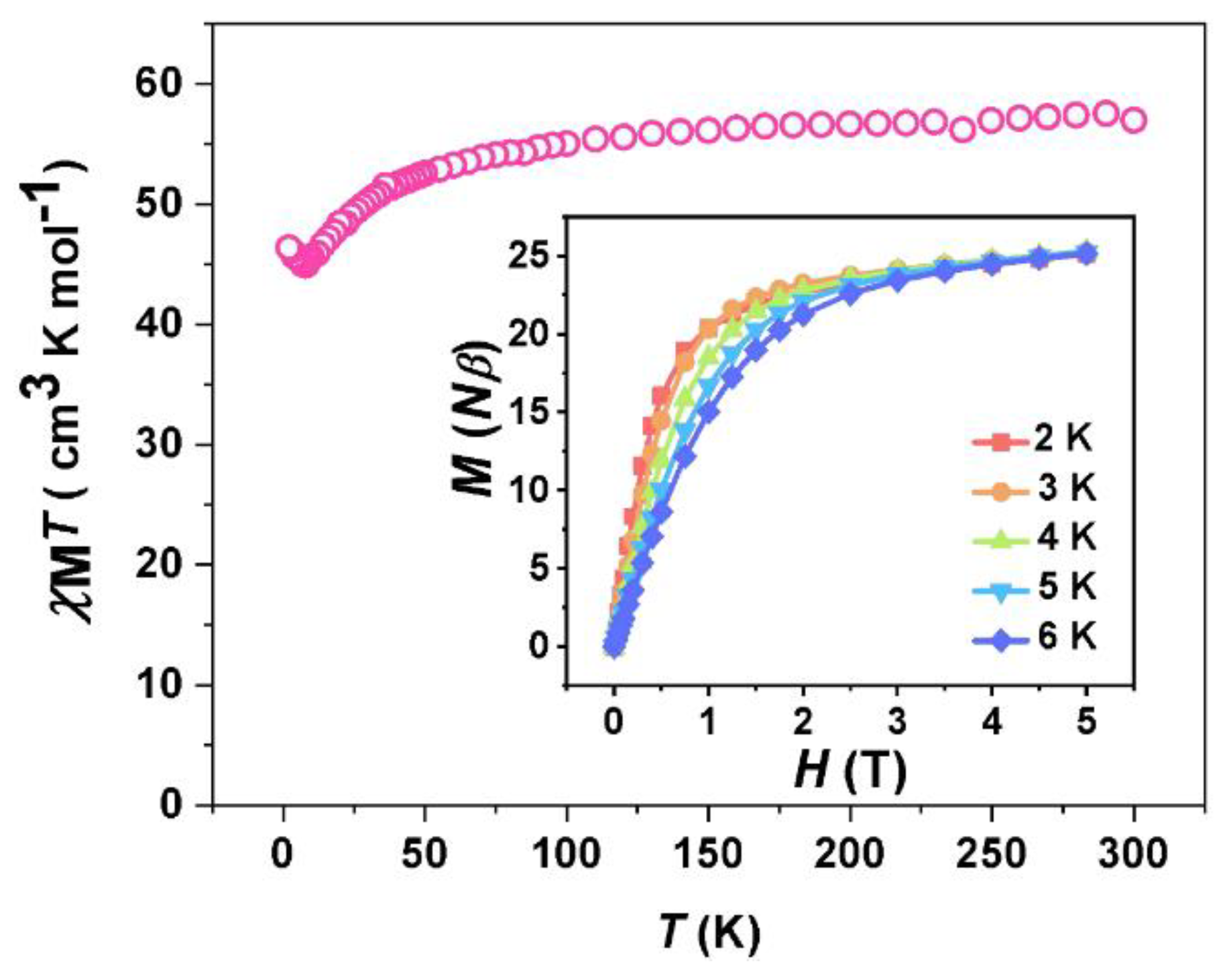
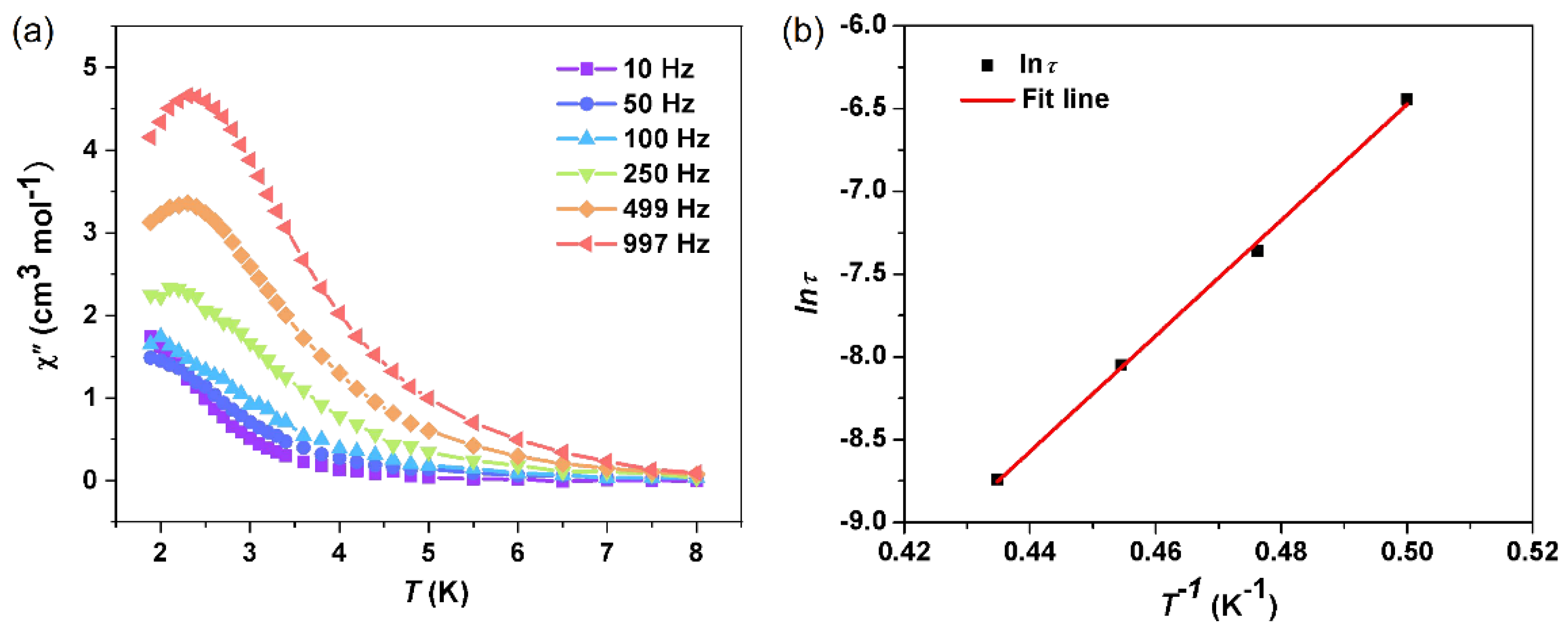
2.2. Magnetic Properties
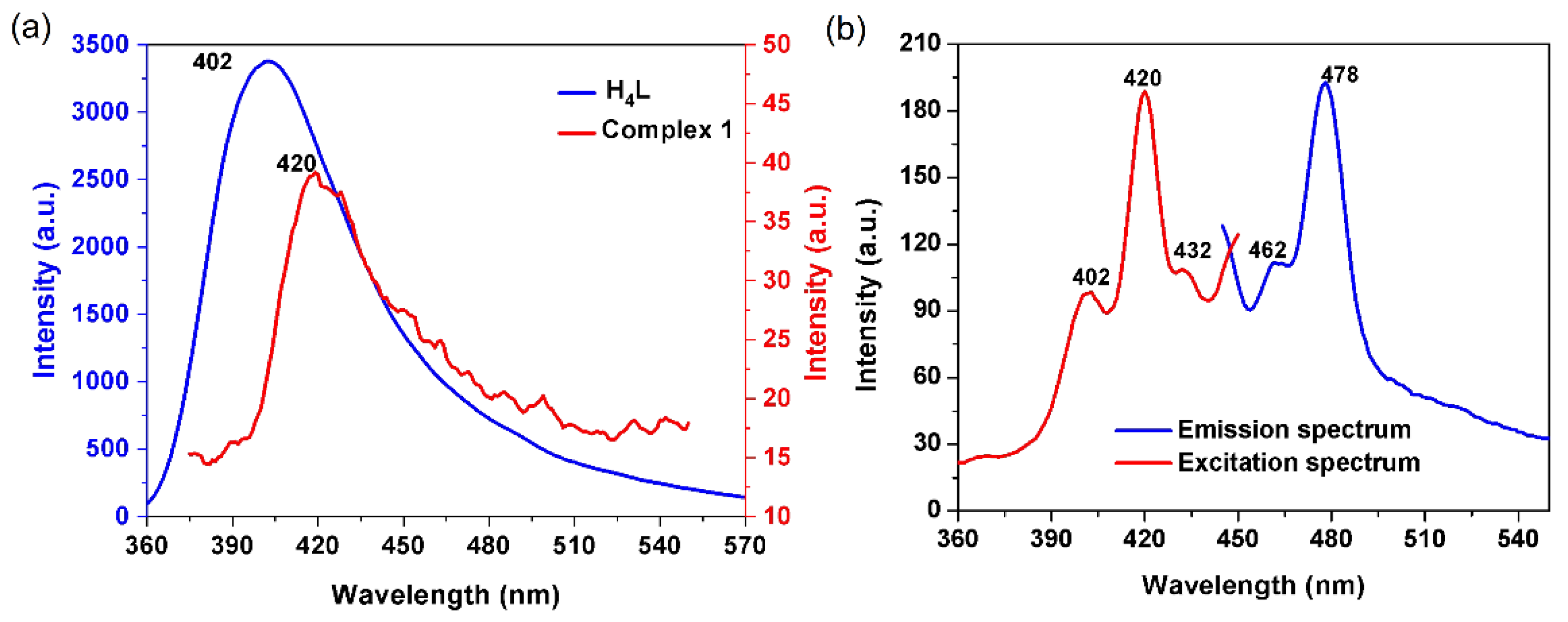
2.3. Luminescent Properties
3. Materials and Methods
3.1. Materials
3.2. The Preparation of Complex 1
3.3. Physical Measurements
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Conflicts of Interest
References
- White, K.A.; Chengelis, D.A.; Gogick, K.A.; Stehman, J.; Rosi, N.L.; Petoud, S. Near-Infrared Luminescent Lanthanide MOF Barcodes. J. Am. Chem. Soc. 2009, 131, 18069–18071. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, L.; Deng, R.; Tian, J.; Zong, Y.; Jin, D.; Liu, X. Multicolor barcoding in a single upconversion crystal. J. Am. Chem. Soc. 2014, 136, 4893–4896. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Yan, B. Luminescent lanthanide barcodes based on postsynthetic modified nanoscale metal–organic frameworks. J. Mater. Chem. C 2014, 2, 7411–7416. [Google Scholar] [CrossRef]
- Zhao, Y.; Shum, H.C.; Chen, H.; Adams, L.L.; Gu, Z.; Weitz, D.A. Microfluidic generation of multifunctional quantum dot barcode particles. J. Am. Chem. Soc. 2011, 133, 8790–8793. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Haushalter, R.C.; Haushalter, R.W.; Shi, Y.; Zhang, Y.; Ding, K.; Zhao, D.; Stucky, G.D. Rare-earth upconverting nanobarcodes for multiplexed biological detection. Small 2011, 7, 1972–1976. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.; Zhu, Y.X.; Wu, K.; Chen, L.; Hou, Y.J.; Yin, S.Y.; Wang, H.P.; Fan, Y.N.; Su, C.Y. Epitaxial Growth of Hetero-Ln-MOF Hierarchical Single Crystals for Domain- and Orientation-Controlled Multicolor Luminescence 3D Coding Capability. Angew. Chem. Int. Ed. Engl. 2017, 56, 14582–14586. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Zhang, P.; Gao, G.; Gao, D.; Yang, Y.; Liu, H.; Wang, Y.; Gong, P.; Cai, L. Near-infrared-emitting two-dimensional codes based on lattice-strained core/(doped) shell quantum dots with long fluorescence lifetime. Adv. Mater. 2014, 26, 6313–6317. [Google Scholar] [CrossRef]
- Shikha, S.; Salafi, T.; Cheng, J.; Zhang, Y. Versatile design and synthesis of nano-barcodes. Chem. Soc. Rev. 2017, 46, 7054–7093. [Google Scholar] [CrossRef]
- Ishii, A.; Miyasaka, T. Direct detection of circular polarized light in helical 1D perovskite-based photodiode. Sci. Adv. 2020, 6, eabd3274. [Google Scholar] [CrossRef]
- Zeng, M.; Ren, A.; Wu, W.; Zhao, Y.; Zhan, C.; Yao, J. Lanthanide MOFs for inducing molecular chirality of achiral stilbazolium with strong circularly polarized luminescence and efficient energy transfer for color tuning. Chem. Sci. 2020, 11, 9154–9161. [Google Scholar] [CrossRef]
- Lu, J.; Guo, M.; Tang, J. Recent Developments in Lanthanide Single-Molecule Magnets. Chem.-Asian J. 2017, 12, 2772–2779. [Google Scholar] [CrossRef] [PubMed]
- Marin, R.; Brunet, G.; Murugesu, M. Shining New Light on Multifunctional Lanthanide Single-Molecule Magnets. Angew. Chem.-Int. Ed. 2020, 60, 1728–1746. [Google Scholar] [CrossRef]
- Meng, Y.-S.; Jiang, S.-D.; Wang, B.-W.; Gao, S. Understanding the Magnetic Anisotropy toward Single-Ion Magnets. Acc. Chem. Res. 2016, 49, 2381–2389. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.; Zhao, C.; Feng, T.; Liu, X.; Ying, X.; Li, X.-L.; Zhang, Y.-Q.; Tang, J. Air-Stable Chiral Single-Molecule Magnets with Record Anisotropy Barrier Exceeding 1800 K. J. Am. Chem. Soc. 2021, 143, 10077–10082. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-C.; Liu, J.-L.; Ungur, L.; Liu, J.; Li, Q.-W.; Wang, L.-F.; Ni, Z.-P.; Chibotaru, L.F.; Chen, X.-M.; Tong, M.-L. Symmetry-Supported Magnetic Blocking at 20 K in Pentagonal Bipyramidal Dy(III) Single-Ion Magnets. J. Am. Chem. Soc. 2016, 138, 2829–2837. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.-H.; Zhai, Y.-Q.; Chen, W.-P.; Ding, Y.-S.; Zheng, Y.-Z. Air-Stable Hexagonal Bipyramidal Dysprosium(III) Single-Ion Magnets with Nearly Perfect D-6h Local Symmetry. Chem.-A Eur. J. 2019, 25, 16219–16224. [Google Scholar] [CrossRef]
- Yang, Q.Y.; Pan, M.; Wei, S.C.; Li, K.; Du, B.B.; Su, C.Y. Linear Dependence of Photoluminescence in Mixed Ln-MOFs for Color Tunability and Barcode Application. Inorg. Chem. 2015, 54, 5707–5716. [Google Scholar] [CrossRef]
- Zhou, Y.; Yan, B. Ratiometric multiplexed barcodes based on luminescent metal–organic framework films. J. Mater. Chem. C 2015, 3, 8413–8418. [Google Scholar] [CrossRef]
- Gao, M.L.; Wang, W.J.; Liu, L.; Han, Z.B.; Wei, N.; Cao, X.M.; Yuan, D.Q. Microporous Hexanuclear Ln(III) Cluster-Based Metal-Organic Frameworks: Color Tunability for Barcode Application and Selective Removal of Methylene Blue. Inorg. Chem. 2017, 56, 511–517. [Google Scholar] [CrossRef]
- Yao, Y.; Gao, Z.; Lv, Y.; Lin, X.; Liu, Y.; Du, Y.; Hu, F.; Zhao, Y.S. Heteroepitaxial Growth of Multiblock Ln-MOF Microrods for Photonic Barcodes. Angew. Chem.-Int. Ed. 2019, 58, 13803–13807. [Google Scholar] [CrossRef]
- Du, B.B.; Zhu, Y.X.; Pan, M.; Yue, M.Q.; Hou, Y.J.; Wu, K.; Zhang, L.Y.; Chen, L.; Yin, S.Y.; Fan, Y.N.; et al. Direct white-light and a dual-channel barcode module from Pr(III)-MOF crystals. Chem. Commun. (Camb.) 2015, 51, 12533–12536. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.Q.; Baxter, B.C.; Brower, K.; Diaz-Botia, C.A.; DeRisi, J.L.; Fordyce, P.M.; Thorn, K.S. Programmable Microfluidic Synthesis of Over One Thousand Uniquely Identifiable Spectral Codes. Adv. Opt. Mater. 2017, 5, 1600548. [Google Scholar] [CrossRef]
- Liu, M.-J.; Yuan, J.; Wang, B.-L.; Wu, S.-T.; Zhang, Y.-Q.; Liu, C.-M.; Kou, H.-Z. Spontaneous Resolution of Chiral Co(III)Dy(III) Single-Molecule Magnet Based on an Achiral Flexible Ligand. Cryst. Growth Des. 2018, 18, 7611–7617. [Google Scholar] [CrossRef]
- Sunatsuki, Y.; Fujita, K.; Maruyama, H.; Suzuki, T.; Ishida, H.; Kojima, M.; Glaser, R. Chiral Crystal Structure of a P212121 Kryptoracemate Iron(II) Complex with an Unsymmetric Azine Ligand and the Observation of Chiral Single Crystal Circular Dichroism. Cryst. Growth Des. 2014, 14, 3692–3695. [Google Scholar] [CrossRef]
- Tshuva, E.Y.; Gendeziuk, N.; Kol, M. Single-step synthesis of salans and substituted salans by Mannich condensation. Tetrahedron Lett. 2001, 42, 6405–6407. [Google Scholar] [CrossRef]
- Zeng, M.; Zhou, Z.-Y.; Wu, X.-R.; Liu, C.-M.; Kou, H.-Z. Assembly of a Heterotrimetallic Zn2Dy2Ir Pentanuclear Complex toward Multifunctional Molecular Materials. Inorg. Chem. 2022, 61, 14275–14281. [Google Scholar] [CrossRef]
- Zeng, M.; Ji, S.-Y.; Wu, X.-R.; Zhang, Y.-Q.; Liu, C.-M.; Kou, H.-Z. Magnetooptical Properties of Lanthanide(III) Metal-Organic Frameworks Based on an Iridium(III) Metalloligand. Inorg. Chem. 2022, 61, 3097–3102. [Google Scholar] [CrossRef]
- Liu, C.-M.; Sun, R.; Hao, X.; Wang, B. Chiral Co-Crystals of (S)- or (R)-1,1′-Binaphthalene-2,2′-diol and Zn2Dy2 Tetranuclear Complexes Behaving as Single-Molecule Magnets. Cryst. Growth Des. 2021, 21, 4346–4353. [Google Scholar] [CrossRef]
- Guo, Y.-N.; Xu, G.-F.; Gamez, P.; Zhao, L.; Lin, S.-Y.; Deng, R.; Tang, J.; Zhang, H.-J. Two-Step Relaxation in a Linear Tetranuclear Dysprosium(III) Aggregate Showing Single-Molecule Magnet Behavior. J. Am. Chem. Soc. 2010, 132, 8538. [Google Scholar] [CrossRef]
- Li, L.; Qin, F.; Zhou, Y.; Zheng, Y.; Miao, J.; Zhang, Z. Photoluminescence and time -resolved -luminescence of CaWO 4: Dy 3+phosphors. J. Lumin. 2020, 224, 117308. [Google Scholar] [CrossRef]
- Zeng, M.; Zhan, C.; Yao, J. Novel bimetallic lanthanide metal-organic frameworks (Ln-MOFs) for colour-tuning through energy-transfer between visible and near-infrared emitting Ln3+ ions. J. Mater. Chem. C 2019, 7, 2751–2757. [Google Scholar] [CrossRef]
- Cui, Y.; Yue, Y.; Qian, G.; Chen, B. Luminescent Functional Metal-Organic Frameworks. Chem. Rev. 2012, 112, 1126–1162. [Google Scholar] [CrossRef] [PubMed]

| Complexes | 1a | 1b |
|---|---|---|
| formula | C225H262Cl4Dy4N8O66Zn4 | C224H257Cl4Dy4N8O64.5Zn4 |
| formula weight | 5187.95 | 5146.90 |
| T/K | 173(2) | 173(2) |
| crystal system | tetragonal | tetragonal |
| space group | P43 | P41 |
| a, b/Å | 29.7479(2) | 29.7491(2) |
| c/Å | 25.3023(3) | 25.3017(2) |
| α, β, γ/° | 90 | 90 |
| V/Å3 | 22,391.0(4) | 22,392.2(3) |
| Z | 4 | 4 |
| ρcalc/g⋅cm−3 | 1.539 | 1.527 |
| μ/mm−1 | 8.583 | 8.562 |
| F(000) | 10,192 | 10,500 |
| data/restraints/parameters | 29,484/102/2724 | 34,446/103/2666 |
| GOF on F2 | 1.019 | 1.013 |
| Rint | 0.0253 | 0.0253 |
| R1, wR2 [I ≥ 2σ (I)] | 0.0338, 0.0860 | 0.0420, 0.1092 |
| R1, wR2 (all data) | 0.0357, 0.0872 | 0.0442, 0.1111 |
| Flack parameter | −0.0025(14) | 0.0012(16) |
| CCDC | 2,209,058 | 2,209,059 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zeng, M.; Miao, L.; Wu, X.-R.; Liu, C.-M.; Kou, H.-Z. Chiral Dy(III) Fluorescent Single-Molecule Magnet Based on an Achiral Flexible Ligand. Magnetochemistry 2022, 8, 166. https://doi.org/10.3390/magnetochemistry8120166
Zeng M, Miao L, Wu X-R, Liu C-M, Kou H-Z. Chiral Dy(III) Fluorescent Single-Molecule Magnet Based on an Achiral Flexible Ligand. Magnetochemistry. 2022; 8(12):166. https://doi.org/10.3390/magnetochemistry8120166
Chicago/Turabian StyleZeng, Min, Lin Miao, Xue-Ru Wu, Cai-Ming Liu, and Hui-Zhong Kou. 2022. "Chiral Dy(III) Fluorescent Single-Molecule Magnet Based on an Achiral Flexible Ligand" Magnetochemistry 8, no. 12: 166. https://doi.org/10.3390/magnetochemistry8120166
APA StyleZeng, M., Miao, L., Wu, X.-R., Liu, C.-M., & Kou, H.-Z. (2022). Chiral Dy(III) Fluorescent Single-Molecule Magnet Based on an Achiral Flexible Ligand. Magnetochemistry, 8(12), 166. https://doi.org/10.3390/magnetochemistry8120166










