A Dy2 Complex Constructed by TCNQ·− Radical Anions with Slow Magnetic Relaxation Behavior †
Abstract
1. Introduction
2. Results
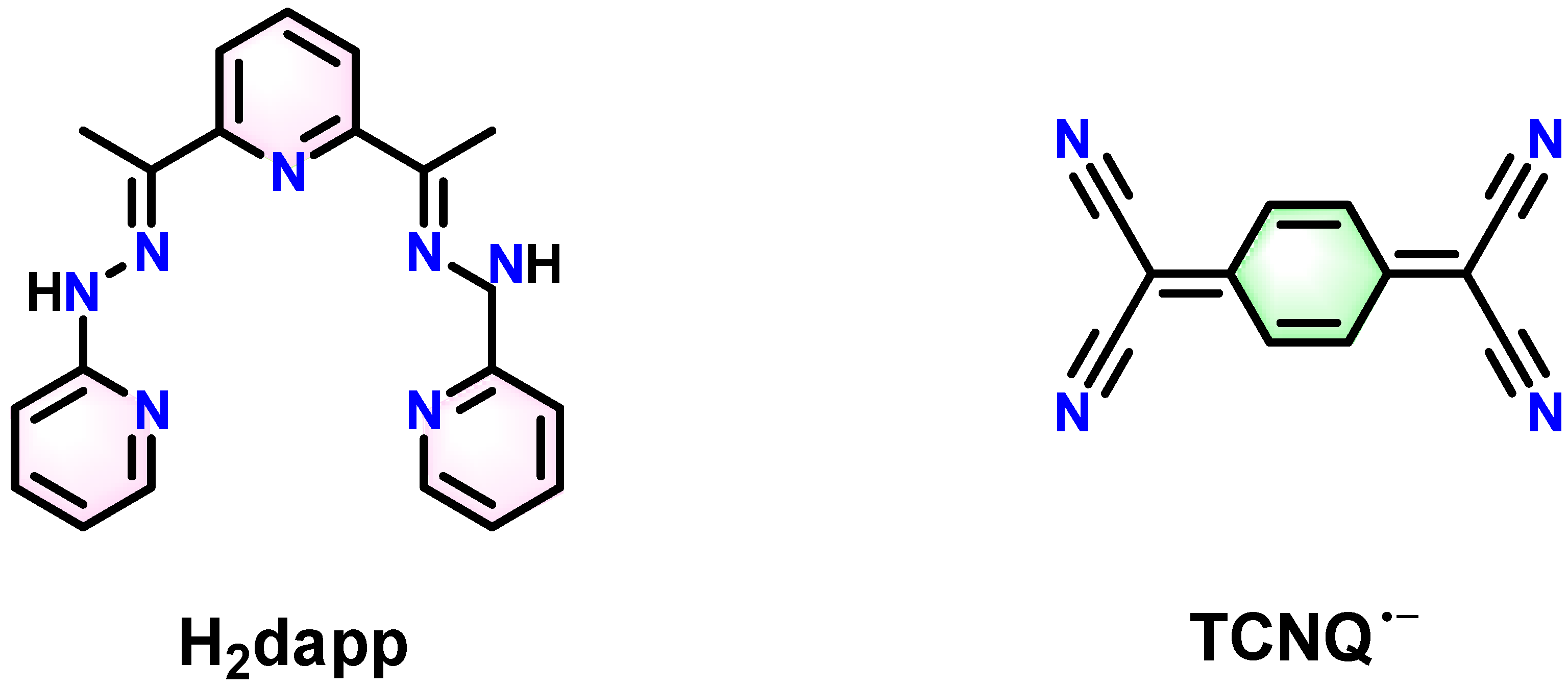
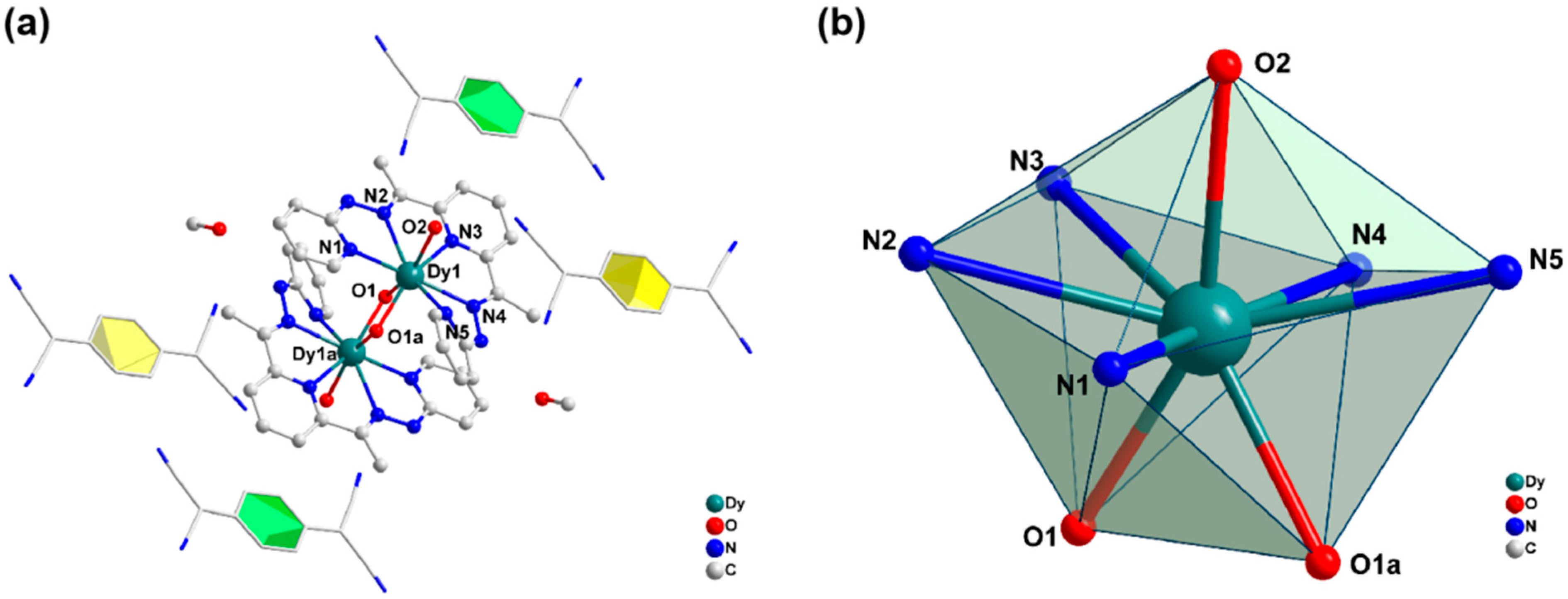
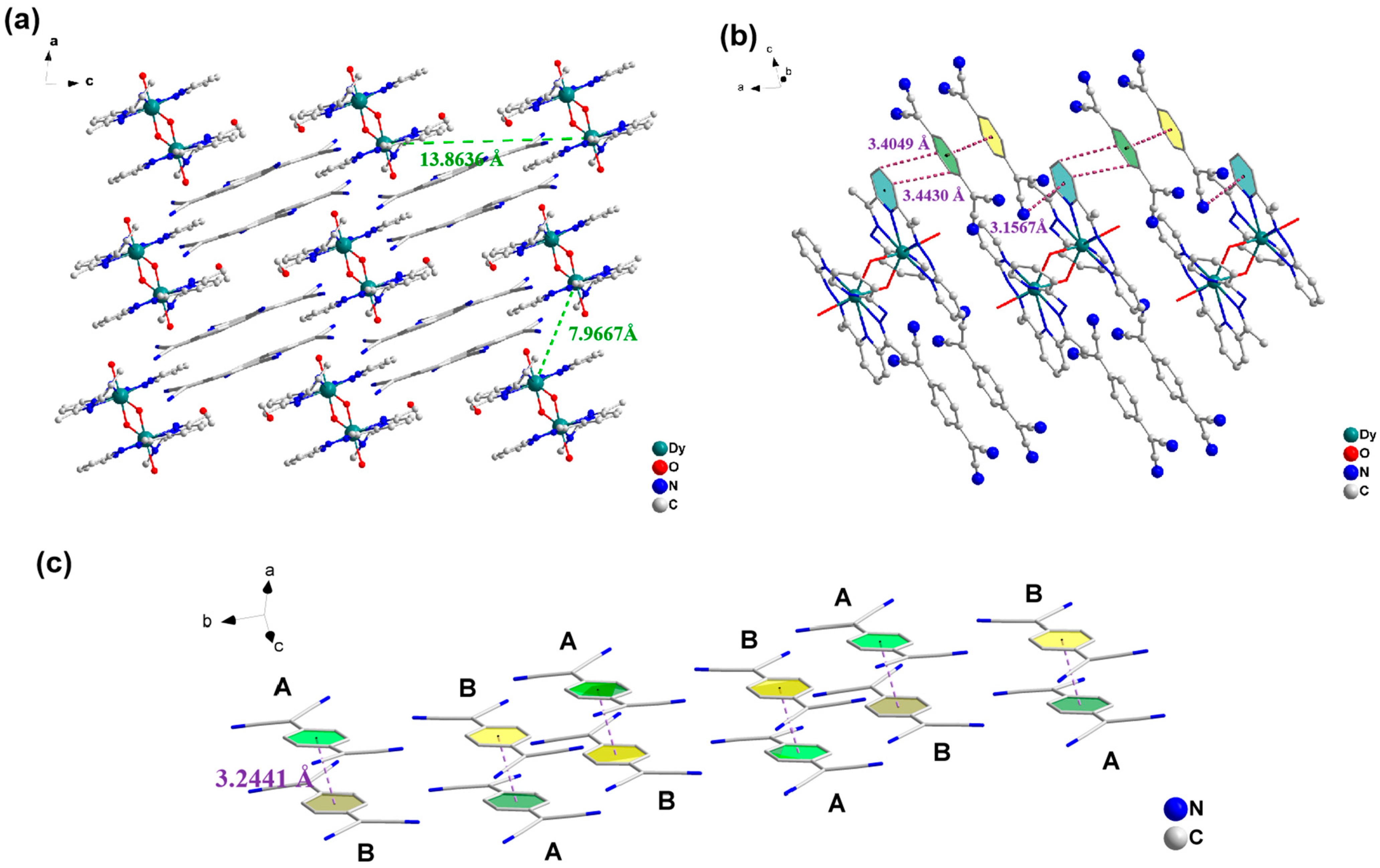
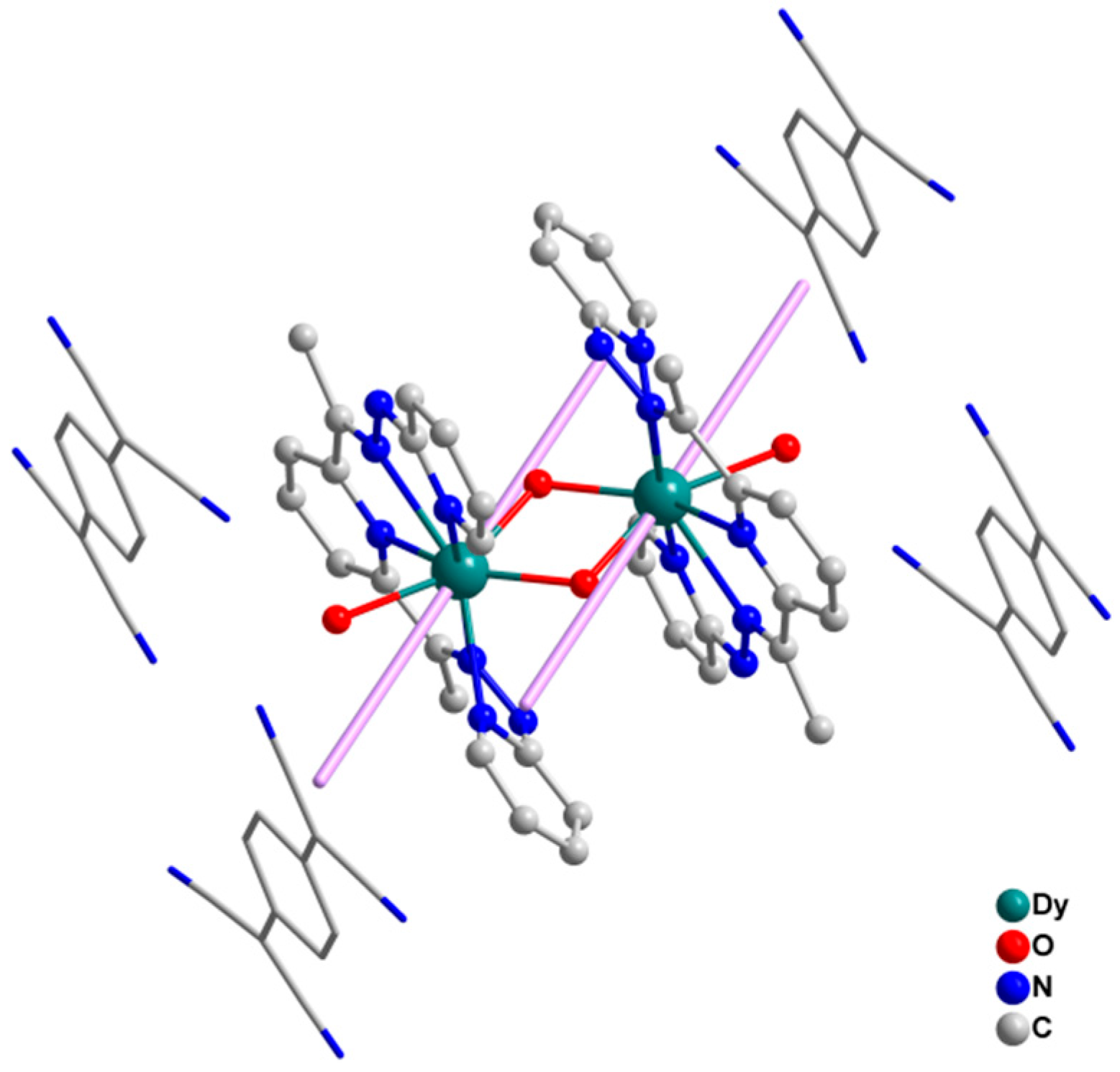
2.1. Crystal Structure Description
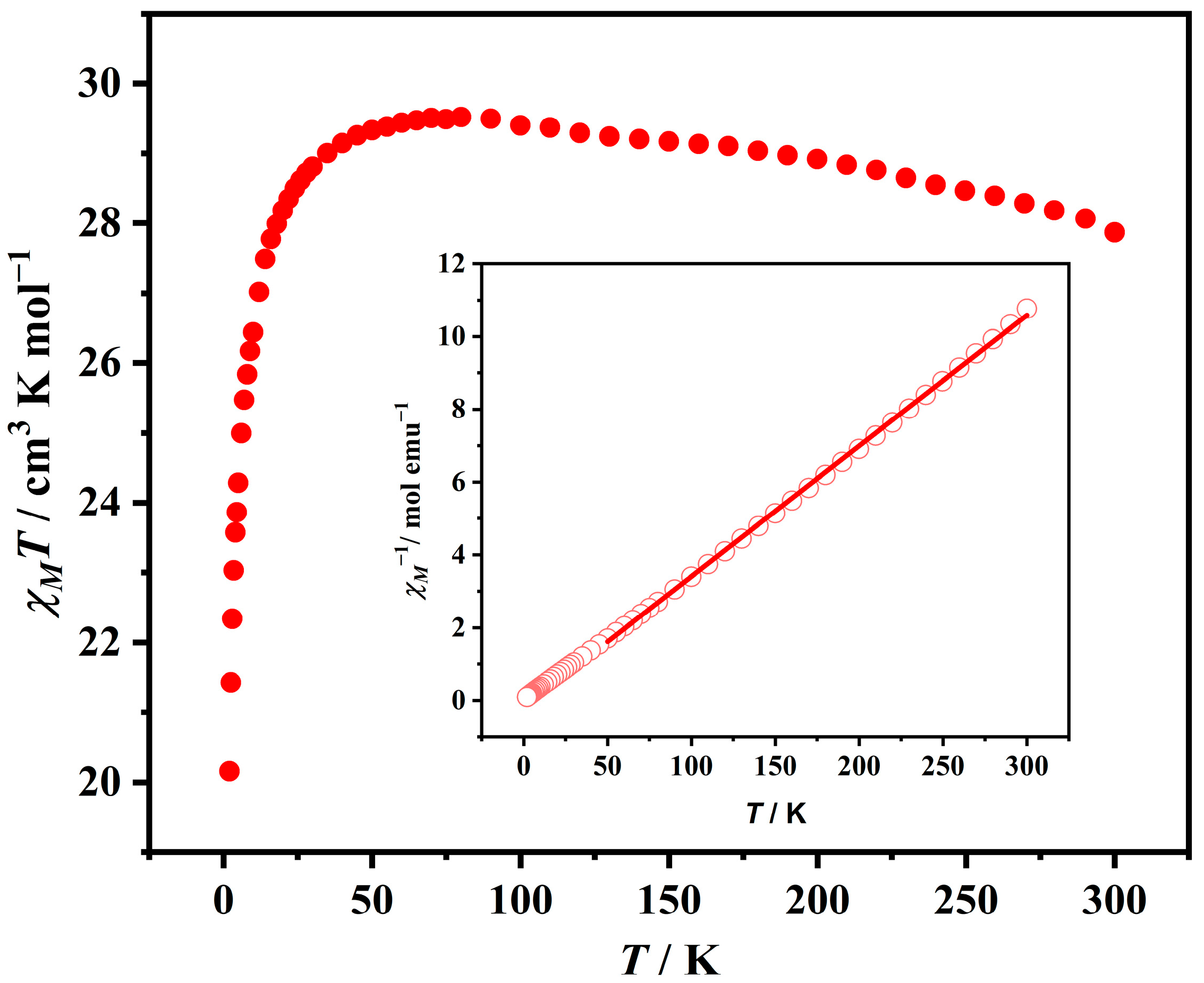
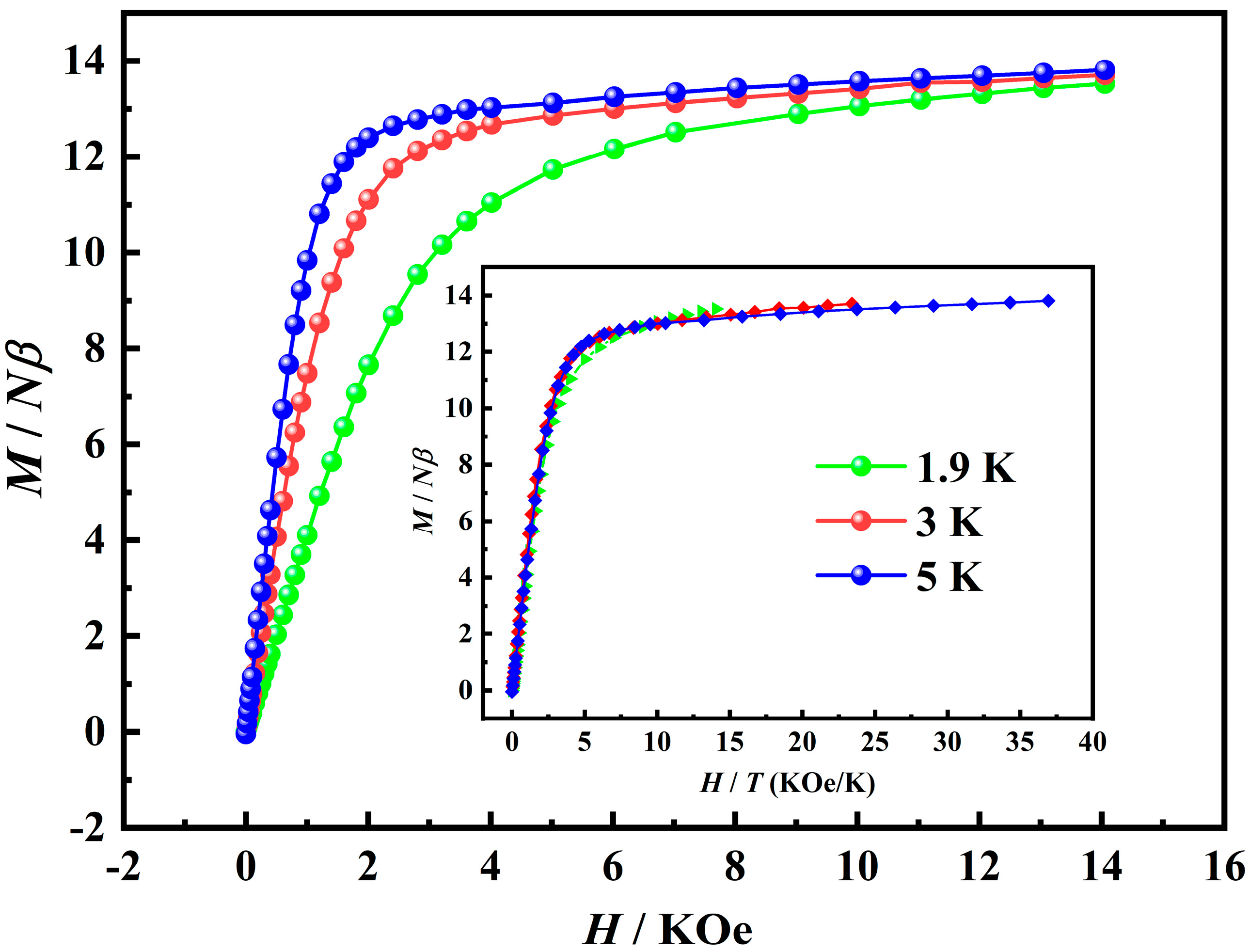
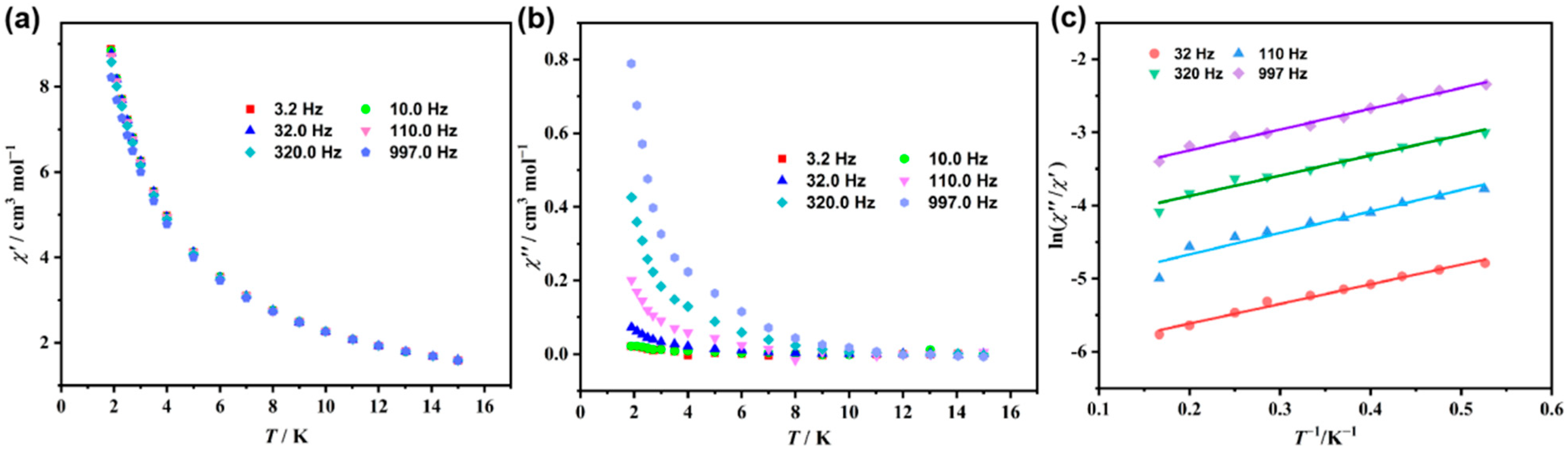
2.2. Magnetic Properties
3. Materials and Methods
3.1. Materials and Characterizations
3.2. Synthesis of [Dy2(H2dapp)2(μ-OH)2(H2O)2] 4TCNQ 2CH3OH
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhu, Z.; Tang, J. Lanthanide single-molecule magnets with high anisotropy barrier: Where to from here? Natl. Sci. Rev. 2022, 9, nwac194. [Google Scholar] [CrossRef]
- Mannini, M.; Pineider, F.; Sainctavit, P.; Danieli, C.; Otero, E.; Sciancalepore, C.; Talarico, A.M.; Arrio, M.-A.; Cornia, A.; Gatteschi, D.; et al. Magnetic memory of a single-molecule quantum magnet wired to a gold surface. Nat. Mater. 2009, 8, 194–197. [Google Scholar] [CrossRef]
- Troiani, F.; Affronte, M. Molecular spins for quantum information technologies. Chem. Soc. Rev. 2011, 40, 3119–3129. [Google Scholar] [CrossRef]
- Aromí, G.; Aguilà, D.; Gamez, P.; Luis, F.; Roubeau, O. Design of magnetic coordination complexes for quantum computing. Chem. Soc. Rev. 2012, 41, 537–546. [Google Scholar] [CrossRef]
- Emerson-King, J.; Gransbury, G.K.; Whitehead, G.F.S.; Vitorica-Yrezabal, I.J.; Rouzières, M.; Clérac, R.; Chilton, N.F.; Mills, D.P. Isolation of a Bent Dysprosium Bis(amide) Single-Molecule Magnet. J. Am. Chem. Soc. 2024, 146, 3331–3342. [Google Scholar] [CrossRef]
- Liu, M.; Chen, Y.-C.; Wang, H.; Shang, T.; Tong, M.-L.; Layfield, R.A.; Mansikkamäki, A.; Guo, F.-S. A Linear Dysprosium(II) Metallocene with a High Effective Energy Barrier and Magnetic Hysteresis up to 70 Kelvin. J. Am. Chem. Soc. 2025, 147, 18307–18316. [Google Scholar] [CrossRef]
- Borah, A.; Murugavel, R. Magnetic relaxation in single-ion magnets formed by less-studied lanthanide ions Ce(III), Nd(III), Gd(III), Ho(III), Tm(II/III) and Yb(III). Coord. Chem. Rev. 2022, 453, 214288. [Google Scholar] [CrossRef]
- Goodwin, C.A.P.; Ortu, F.; Reta, D.; Chilton, N.F.; Mills, D.P. Molecular magnetic hysteresis at 60 kelvin in dysprosocenium. Nature 2017, 548, 439–442. [Google Scholar] [CrossRef]
- Guo, F.-S.; Day, B.M.; Chen, Y.-C.; Tong, M.-L.; Mansikkamäki, A.; Layfield, R.A. Magnetic hysteresis up to 80 kelvin in a dysprosium metallocene single-molecule magnet. Science 2018, 362, 1400–1403. [Google Scholar] [CrossRef]
- Emerson-King, J.; Gransbury, G.K.; Atkinson, B.E.; Blackmore, W.J.A.; Whitehead, G.F.S.; Chilton, N.F.; Mills, D.P. Soft magnetic hysteresis in a dysprosium amide–alkene complex up to 100 kelvin. Nature 2025, 643, 125–129. [Google Scholar] [CrossRef]
- Li, H.-D.; Wu, S.-G.; Tong, M.-L. Lanthanide–radical single-molecule magnets: Current status and future challenges. Chem. Commun. 2023, 59, 6159–6170. [Google Scholar] [CrossRef]
- Zhu, Z.; Paul, S.; Zhao, C.; Wu, J.; Ying, X.; Ungur, L.; Wernsdorfer, W.; Meyer, F.; Tang, J. Record Quantum Tunneling Time in an Air-Stable Exchange-Bias Dysprosium Macrocycle. J. Am. Chem. Soc. 2024, 146, 18899–18904. [Google Scholar] [CrossRef]
- Canaj, A.B.; Dey, S.; Martí, E.R.; Wilson, C.; Rajaraman, G.; Murrie, M. Insight into D6h Symmetry: Targeting Strong Axiality in Stable Dysprosium(III) Hexagonal Bipyramidal Single-Ion Magnets. Angew. Chem. Int. Ed. 2019, 58, 14146–14151. [Google Scholar] [CrossRef]
- Liu, J.-L.; Chen, Y.-C.; Tong, M.-L. Symmetry strategies for high performance lanthanide-based single-molecule magnets. Chem. Soc. Rev. 2018, 47, 2431–2453. [Google Scholar] [CrossRef]
- Peng, Y.; Powell, A.K. What do 3d-4f butterflies tell us? Coord. Chem. Rev. 2021, 426, 213490. [Google Scholar] [CrossRef]
- Liu, K.; Shi, W.; Cheng, P. Toward heterometallic single-molecule magnets: Synthetic strategy, structures and properties of 3d–4f discrete complexes. Coord. Chem. Rev. 2015, 289–290, 74–122. [Google Scholar] [CrossRef]
- Vaz, M.G.F.; Andruh, M. Molecule-based magnetic materials constructed from paramagnetic organic ligands and two different metal ions. Coord. Chem. Rev. 2021, 427, 213611. [Google Scholar] [CrossRef]
- Demir, S.; Jeon, I.-R.; Long, J.R.; Harris, T.D. Radical ligand-containing single-molecule magnets. Coord. Chem. Rev. 2015, 289–290, 149–176. [Google Scholar] [CrossRef]
- Liu, F.; Spree, L.; Krylov, D.S.; Velkos, G.; Avdoshenko, S.M.; Popov, A.A. Single-Electron Lanthanide-Lanthanide Bonds Inside Fullerenes toward Robust Redox-Active Molecular Magnets. Acc. Chem. Res. 2019, 52, 2981–2993. [Google Scholar] [CrossRef]
- Jin, C.-Y.; Mei, X.-L.; Zhou, Y.; Li, L.; Sutter, J.-P. Single-Molecule Magnet Behavior in a Tb-Nitronyl Nitroxide Radical Network with [Tb3(NIT)2] Nodes. Inorg. Chem. 2024, 63, 23829–23836. [Google Scholar] [CrossRef]
- Li, H.; Sun, Z.; Sun, J.; Xi, L.; Guo, J.; Sun, G.; Xie, J.; Ma, Y.; Li, L. Single-molecule magnet behavior in a CuII-decorated {DyIII2} complex with nitronyl nitroxide biradicals. J. Mater. Chem. C 2018, 6, 2060–2068. [Google Scholar] [CrossRef]
- Ballesteros-Rivas, M.; Ota, A.; Reinheimer, E.; Prosvirin, A.; Valdés-Martinez, J.; Dunbar, K.R. Highly Conducting Coordination Polymers Based on Infinite M(4,4′-bpy) Chains Flanked by Regular Stacks of Non-Integer TCNQ Radicals. Angew. Chem. Int. Ed. 2011, 50, 9703–9707. [Google Scholar] [CrossRef]
- Wang, Z.-X.; Zhang, X.; Zhang, Y.-Z.; Li, M.-X.; Zhao, H.; Andruh, M.; Dunbar, K.R. Single-Chain Magnetic Behavior in a Hetero-Tri-Spin Complex Mediated by Supramolecular Interactions with TCNQF.− Radicals. Angew. Chem. Int. Ed. 2014, 53, 11567–11570. [Google Scholar] [CrossRef]
- Benner, F.; La Droitte, L.; Cador, O.; Le Guennic, B.; Demir, S. Magnetic hysteresis and large coercivity in bisbenzimidazole radical-bridged dilanthanide complexes. Chem. Sci. 2023, 14, 5577–5592. [Google Scholar] [CrossRef]
- Kaim, W.; Moscherosch, M. The coordination chemistry of TCNE, TCNQ and related polynitrileπ acceptors. Coord. Chem. Rev. 1994, 129, 157–193. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, Z.-X.; Xie, H.; Li, M.-X.; Woods, T.J.; Dunbar, K.R. A cobalt(II) spin-crossover compound with partially charged TCNQ radicals and an anomalous conducting behavior. Chem. Sci. 2016, 7, 1569–1574. [Google Scholar] [CrossRef]
- Üngör, Ö.; Shatruk, M. Transition metal complexes with fractionally charged TCNQ radical anions as structural templates for multifunctional molecular conductors. Polyhedron 2020, 177, 114254. [Google Scholar] [CrossRef]
- Taponen, A.I.; Ayadi, A.; Lahtinen, M.K.; Oyarzabal, I.; Bonhommeau, S.; Rouzières, M.; Mathonière, C.; Tuononen, H.M.; Clérac, R.; Mailman, A. Room-Temperature Magnetic Bistability in a Salt of Organic Radical Ions. J. Am. Chem. Soc. 2021, 143, 15912–15917. [Google Scholar] [CrossRef]
- Zhang, X.; Xie, H.; Ballesteros-Rivas, M.; Woods, T.J.; Dunbar, K.R. Conducting Molecular Nanomagnet of DyIII with Partially Charged TCNQ Radicals. Chem.—A Eur. J. 2017, 23, 7448–7452. [Google Scholar] [CrossRef]
- Zhou, Y.-Q.; Deng, W.; Du, S.-N.; Chen, Y.-C.; Ruan, Z.-Y.; Wu, S.-G.; Liu, J.-L.; Tong, M.-L. An alkoxyborate-bridging Dy2 single-molecule magnet with ferromagnetic coupling. Inorg. Chem. Front. 2024, 11, 1061–1069. [Google Scholar] [CrossRef]
- Zhou, Y.-Q.; Yao, C.-Y.; Ruan, Z.-Y.; Lyu, B.-H.; Du, S.-N.; Wu, S.-G.; Chen, Y.-C.; Deng, W.; Liu, J.-L.; Tong, M.-L. Proton control of Raman relaxation in bis-hydrazone single-molecule magnets. Chin. Chem. Lett. 2025, 36, 111179. [Google Scholar] [CrossRef]
- Llunell, M.; Casanova, D.; Cirera, J.; Alemany, P.; Alvarez, S. SHAPE 2.1; University of Barcelona: Barcelona, Spain, 2013. [Google Scholar]
- Tiunova, A.V.; Korchagin, D.V.; Kazakova, A.V.; Shilov, G.V.; Buravov, L.I.; Dmitriev, A.I.; Zhidkov, M.V.; Zverev, V.N.; Yagubskii, E.B.; Aldoshin, S.M. Solvent and Dihalide Substituent Effects on Crystal Structure, Spin-Crossover Behavior and Conductivity of the Cationic Mn(III) Complexes with Electroactive TCNQ Counteranions. Eur. J. Inorg. Chem. 2024, 27, e202300749. [Google Scholar] [CrossRef]
- Zhang, J.; Yan, P.; Li, G.; Hou, G.; Suda, M.; Einaga, Y. Systematic investigation of an array of TCNQ lanthanide complexes: Synthesis, structure and magnetic properties. Dalton Trans. 2009, 10466–10473. [Google Scholar] [CrossRef]
- Wang, J.; Li, Q.-W.; Wu, S.-G.; Chen, Y.-C.; Wan, R.-C.; Huang, G.-Z.; Liu, Y.; Liu, J.-L.; Reta, D.; Giansiracusa, M.J.; et al. Opening Magnetic Hysteresis by Axial Ferromagnetic Coupling: From Mono-Decker to Double-Decker Metallacrown. Angew. Chem. Int. Ed. 2021, 60, 5299–5306. [Google Scholar] [CrossRef]
- Lu, Y.-B.; Jian, F.-M.; Jin, S.; Zhao, J.-W.; Xie, Y.-R.; Luo, G.-T. Three-Dimensional Extended Frameworks Constructed from Dinuclear Lanthanide(III) 1,4-Naphthalenedicarboxylate Units with Bis(2,2′-biimidazole) Templates: Syntheses, Structures, and Magnetic and Luminescent Properties. Cryst. Growth Des. 2014, 14, 1684–1694. [Google Scholar] [CrossRef]
- Kahn, M.L.; Sutter, J.-P.; Golhen, S.; Guionneau, P.; Ouahab, L.; Kahn, O.; Chasseau, D. Systematic Investigation of the Nature of The Coupling between a Ln(III) Ion (Ln = Ce(III) to Dy(III)) and Its Aminoxyl Radical Ligands. Structural and Magnetic Characteristics of a Series of {Ln(organic radical)2} Compounds and the Related {Ln(Nitrone)2} Derivatives. J. Am. Chem. Soc. 2000, 122, 3413–3421. [Google Scholar] [CrossRef]
- Xi, L.; Li, H.; Sun, J.; Ma, Y.; Tang, J.; Li, L. Designing Multicoordinating Nitronyl Nitroxide Radical Toward Multinuclear Lanthanide Aggregates. Inorg. Chem. 2020, 59, 443–451. [Google Scholar] [CrossRef]
- Lv, X.-H.; Yang, S.-L.; Li, Y.-X.; Zhang, C.-X.; Wang, Q.-L. A family of lanthanide compounds based on nitronyl nitroxide radicals: Synthesis, structure, magnetic and fluorescence properties. RSC Adv. 2017, 7, 38179–38186. [Google Scholar] [CrossRef]
- Ruan, L.; Tong, J.; Luo, F.; Qin, G.; Tian, F.; Jiao, X.; Zhang, X. Magnetic Improvement and Relaxation Mechanism of the Tb-Phthalocyanine Single-Molecule Magnet by Absorbing CH2Cl2 Molecules. J. Phys. Chem. C 2021, 125, 10165–10172. [Google Scholar] [CrossRef]
- Tian, H.; Liu, R.; Wang, X.; Yang, P.; Li, Z.; Li, L.; Liao, D. Magnetic Slow Relaxation in Cyclic TbIII-Nitronyl Nitroxide Radical Complexes. Eur. J. Inorg. Chem. 2009, 2009, 4498–4502. [Google Scholar] [CrossRef]
- Zhao, J.; Luo, J.; Chen, L.; Yuan, J.; Li, H.; Ma, P.; Wang, J.; Niu, J. Novel 1-D double-chain organic–inorganic hybrid polyoxotungstates constructed from dimeric copper–lanthanide heterometallic silicotungstate units. CrystEngComm 2012, 14, 7981–7993. [Google Scholar] [CrossRef]
- Luis, F.; Bartolomé, J.; Fernández, J.F.; Tejada, J.; Hernández, J.M.; Zhang, X.X.; Ziolo, R. Thermally activated and field-tuned tunneling in Mn12Ac studied by ac magnetic susceptibility. Phys. Rev. B 1997, 55, 11448–11456. [Google Scholar] [CrossRef]
- Bartolomé, J.; Filoti, G.; Kuncser, V.; Schinteie, G.; Mereacre, V.; Anson, C.E.; Powell, A.K.; Prodius, D.; Turta, C. Magnetostructural correlations in the tetranuclear series of {Fe3LnO2} butterfly core clusters: Magnetic and Mössbauer spectroscopic study. Phys. Rev. B 2009, 80, 014430. [Google Scholar] [CrossRef]
- Gatteschi, D.; Sessoli, R. Quantum Tunneling of Magnetization and Related Phenomena in Molecular Materials. Angew. Chem. Int. Ed. 2003, 42, 268–297. [Google Scholar] [CrossRef]
- Xi, L.; Han, J.; Huang, X.; Li, L. Nitronyl Nitroxide Biradical-Based Binuclear Lanthanide Complexes: Structure and Magnetic Properties. Magnetochemistry 2020, 6, 48. [Google Scholar] [CrossRef]
- Jin, C.-Y.; Zhang, Q.; Tang, J.; Cheng, P.; Li, L. Unusual zig-zag-shaped Ln-radical coordination networks derived from a nitronyl nitroxide with two imidazole groups: Single-crystal-to-single-crystal transformations and magnetic properties. Dalton Trans. 2025, 54, 7718–7725. [Google Scholar] [CrossRef]
- Wang, X.; Han, J.; Huang, X.; Li, L. LnIII-NiII heterometallic compounds linked by nitronyl nitroxides: Structure and magnetism. Inorg. Chem. Commun. 2021, 134, 108983. [Google Scholar] [CrossRef]
- Chilton, N.F.; Collison, D.; McInnes, E.J.L.; Winpenny, R.E.P.; Soncini, A. An electrostatic model for the determination of magnetic anisotropy in dysprosium complexes. Nat. Commun. 2013, 4, 2551. [Google Scholar] [CrossRef]
- Wang, Y.; Zeng, Z.; Luo, S.; Guo, Y.; Liu, X. Enhancing the single-molecule magnetic performance of β-diketonate Dy(III) complexes by modulating the coordination microenvironment and magnetic interaction: From a mononuclear to a dinuclear structure. Dalton Trans. 2024, 53, 17272–17280. [Google Scholar] [CrossRef]
- Zhang, W.; Xu, S.-M.; Zhu, Z.-X.; Ru, J.; Zhang, Y.-Q.; Yao, M.-X. Strong intramolecular DyIII–DyIII magnetic couplings up to 15.00 cm−1 in phenoxyl-bridged dinuclear 4f complexes. New J. Chem. 2020, 44, 2083–2090. [Google Scholar] [CrossRef]
- Zhang, W.-Y.; Tian, Y.-M.; Li, H.-F.; Chen, P.; Sun, W.-B.; Zhang, Y.-Q.; Yan, P.-F. A series of dinuclear Dy(III) complexes bridged by 2-methyl-8-hydroxylquinoline: Replacement on the periphery coordinated β-diketonate terminal leads to different single-molecule magnetic properties. Dalton Trans. 2016, 45, 3863–3873. [Google Scholar] [CrossRef]
- Shen, F.-X.; Pramanik, K.; Brandão, P.; Zhang, Y.-Q.; Jana, N.C.; Wang, X.-Y.; Panja, A. Macrocycle supported dimetallic lanthanide complexes with slow magnetic relaxation in Dy2 analogues. Dalton Trans. 2020, 49, 14169–14179. [Google Scholar] [CrossRef]
- Wang, W.-M.; Zhang, H.-X.; Wang, S.-Y.; Shen, H.-Y.; Gao, H.-L.; Cui, J.-Z.; Zhao, B. Ligand Field Affected Single-Molecule Magnet Behavior of Lanthanide(III) Dinuclear Complexes with an 8-Hydroxyquinoline Schiff Base Derivative as Bridging Ligand. Inorg. Chem. 2015, 54, 10610–10622. [Google Scholar] [CrossRef]
- Guo, M.; Zhang, Y.-Q.; Zhu, Z.; Tang, J. Dysprosium Compounds with Hula-Hoop-like Geometries: The Influence of Magnetic Anisotropy and Magnetic Interactions on Magnetic Relaxation. Inorg. Chem. 2018, 57, 12213–12221. [Google Scholar] [CrossRef]
- Leng, J.-D.; Liu, J.-L.; Lin, W.-Q.; Gómez-Coca, S.; Aravena, D.; Ruiz, E.; Tong, M.-L. Unprecedented ferromagnetic dipolar interaction in a dinuclear holmium(III) complex: A combined experimental and theoretical study. Chem. Commun. 2013, 49, 9341–9343. [Google Scholar] [CrossRef]
- Curry, J.D.; Robinson, M.A.; Busch, D.H. Metal complexes derived from substituted hydrazones of 2,6-diacetylpyridine. Inorg. Chem. 1967, 6, 1570–1574. [Google Scholar] [CrossRef]
- Melby, L.R.; Harder, R.J.; Hertler, W.R.; Mahler, W.; Benson, R.E.; Mochel, W.E. Substituted Quinodimethans. II. Anion-radical Derivatives and Complexes of 7,7,8,8-Tetracyanoquinodimethan. J. Am. Chem. Soc. 1962, 84, 3374–3387. [Google Scholar] [CrossRef]
- Zhong, L.; Chen, W.-B.; OuYang, Z.-J.; Yang, M.; Zhang, Y.-Q.; Gao, S.; Schulze, M.; Wernsdorfer, W.; Dong, W. Unprecedented one-dimensional chain and two-dimensional network dysprosium(III) single-molecule toroics with white-light emission. Chem. Commun. 2020, 56, 2590–2593. [Google Scholar] [CrossRef]
- Wang, H.-S.; Chen, Y.; Hu, Z.-B.; Yin, C.-L.; Zhang, Z.; Pan, Z.-Q. Modulation of the directions of the anisotropic axes of DyIII ions through utilizing two kinds of organic ligands or replacing DyIII ions by FeIII ions. CrystEngComm 2019, 21, 5429–5439. [Google Scholar] [CrossRef]
- Wang, H.-S.; Zhang, K.; Song, Y.; Pan, Z.-Q. Recent advances in 3d-4f magnetic complexes with several types of non-carboxylate organic ligands. Inorganica Chim. Acta 2021, 521, 120318. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXS-2014, Program for Solution of Crystal Structures; University of Göttingen: Göttingen, Germany, 2014. [Google Scholar]
- Sheldrick, G.M. SHELXL-2014, Program for Refinement of Crystal Structures; University of Göttingen: Göttingen, Germany, 2014. [Google Scholar]
| Dy2 Complex | Point Group | Shortest Intermolecular Dy···Dy Distance | Magnetic Coupling | The Slow Relaxation Process | ||
|---|---|---|---|---|---|---|
| Field (Oe) | Ueff (K) | τ0 (s) | ||||
| [Dy2(dmpd)6(bmp)] | D4d | 10.487 Å | AF (J = −0.524 cm−1) | 0 | 87.29 | 5.48 × 10−7 |
| [Dy2(dbm)2(LH3)2]·H2O | D4d | 9.423 Å | F (J = 15.00 cm−1) | 0 | 76.18 | 2.54 × 10−8 |
| [Dy(hfac)3]2(μ-HMq)2 | D4d | 10.028 Å | F (J = 1.00 cm−1) | 0 | 9.2 | 1.7 × 10−5 |
| [Dy2(L5)(tfac)4] | D4d | 9.214 Å | F (J = 2.72 cm−1) | 0 | 79.0 | 3.3 × 10−8 |
| [Dy2(hfac)4(L3)2] | D2d | 10.172 Å | F (/) | 0 | 6.8 | 9.12 × 10−6 |
| [Dy2(bfac)4(L4)2]·C7H16 | D2d | 9.502 Å | AF (/) | 0 | 25.7 | 1.64 × 10−6 |
| [Dy2(H2dapp)2(μ-OH)2(H2O)2]·4TCNQ·2CH3OH (this work) | D4d | 7.967 Å | F (/) | 0 | 2.82 | 6.88 × 10−6 |
| Complex 1 | |
|---|---|
| Empirical formula | C88H68Dy2N30O6 |
| Formula weight | 1966.70 |
| Temperature/K | 120.01(10) |
| Crystal system | triclinic |
| Space group | Pī |
| a [Å] | 10.41860(10) |
| b [Å] | 13.5613(2) |
| c [Å] | 15.3557(2) |
| a [deg] | 75.1470(10) |
| b [deg] | 81.0730(10) |
| g [deg] | 85.4510(10) |
| Volume [Å3] | 2069.93(5) |
| Z | 1 |
| ρcalc [g/cm−3] | 1.578 |
| μ [mm−1] | 10.163 |
| F(000) | 986 |
| Reflections collected | 25526 |
| Unique/parameters | 8352/577 |
| R(int) | 0.0584 |
| Goodness-of-fit on F2 | 1.161 |
| R1, wR2 [I > 2σ(I)] | 0.0419, 0.1132 |
| R1, wR2 (all data) | 0.0431, 0.1140 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.; Qin, S.; Li, X.; Zuo, W.; Wang, Q.; Li, L.; Ma, Y.; Tang, J.; Zhao, B. A Dy2 Complex Constructed by TCNQ·− Radical Anions with Slow Magnetic Relaxation Behavior. Magnetochemistry 2025, 11, 75. https://doi.org/10.3390/magnetochemistry11090075
Wang X, Qin S, Li X, Zuo W, Wang Q, Li L, Ma Y, Tang J, Zhao B. A Dy2 Complex Constructed by TCNQ·− Radical Anions with Slow Magnetic Relaxation Behavior. Magnetochemistry. 2025; 11(9):75. https://doi.org/10.3390/magnetochemistry11090075
Chicago/Turabian StyleWang, Xirong, Shijia Qin, Xiulan Li, Wenjing Zuo, Qinglun Wang, Licun Li, Yue Ma, Jinkui Tang, and Bin Zhao. 2025. "A Dy2 Complex Constructed by TCNQ·− Radical Anions with Slow Magnetic Relaxation Behavior" Magnetochemistry 11, no. 9: 75. https://doi.org/10.3390/magnetochemistry11090075
APA StyleWang, X., Qin, S., Li, X., Zuo, W., Wang, Q., Li, L., Ma, Y., Tang, J., & Zhao, B. (2025). A Dy2 Complex Constructed by TCNQ·− Radical Anions with Slow Magnetic Relaxation Behavior. Magnetochemistry, 11(9), 75. https://doi.org/10.3390/magnetochemistry11090075







