A Status Review on the Importance of Mulberry (Morus spp.) and Prospects towards Its Cultivation in a Controlled Environment
Abstract
1. Introduction
2. Significance of Mulberry (Morus spp.)
2.1. Medicinal Value of Mulberry
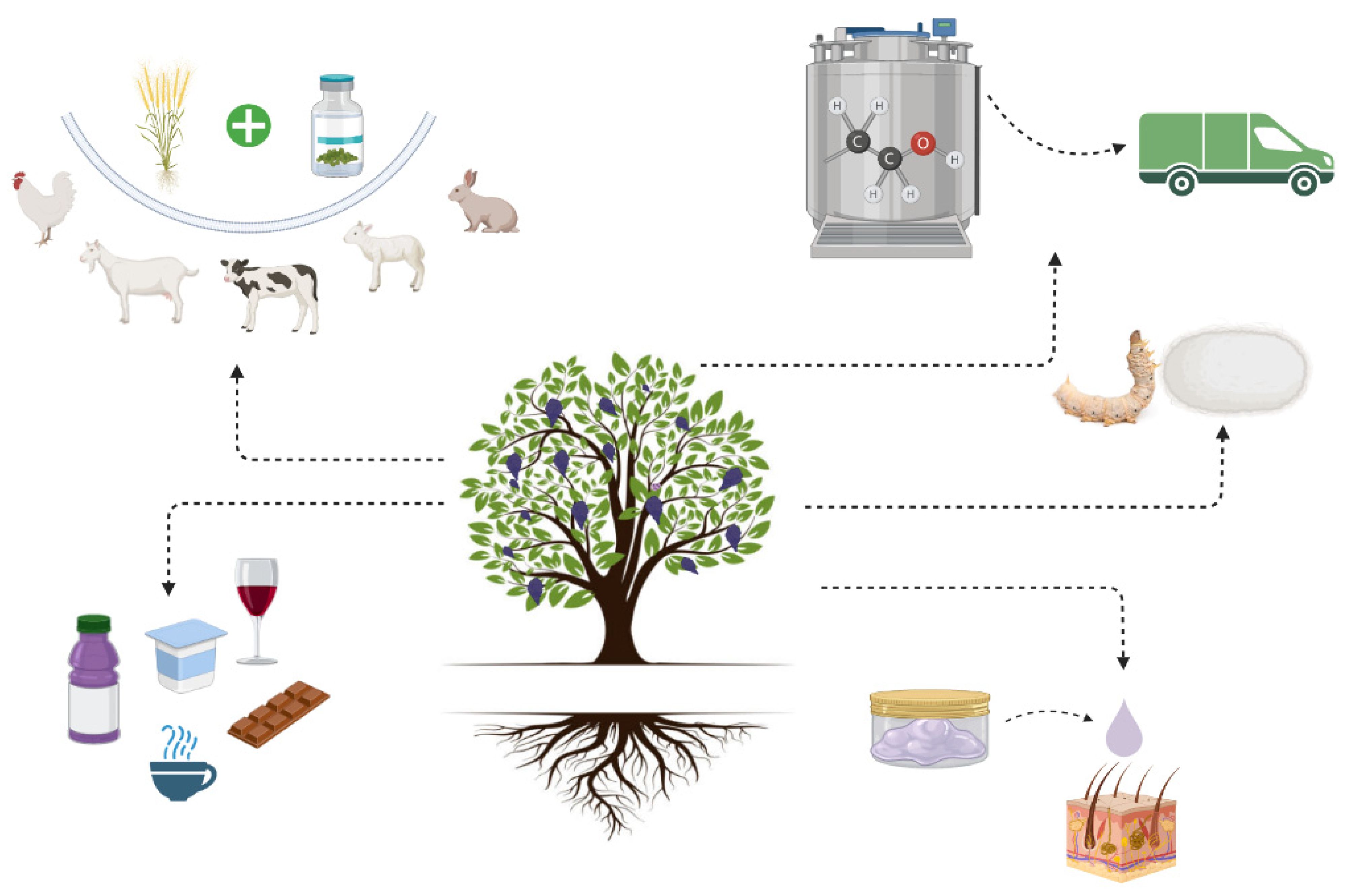
2.2. Economic Impact of Mulberry
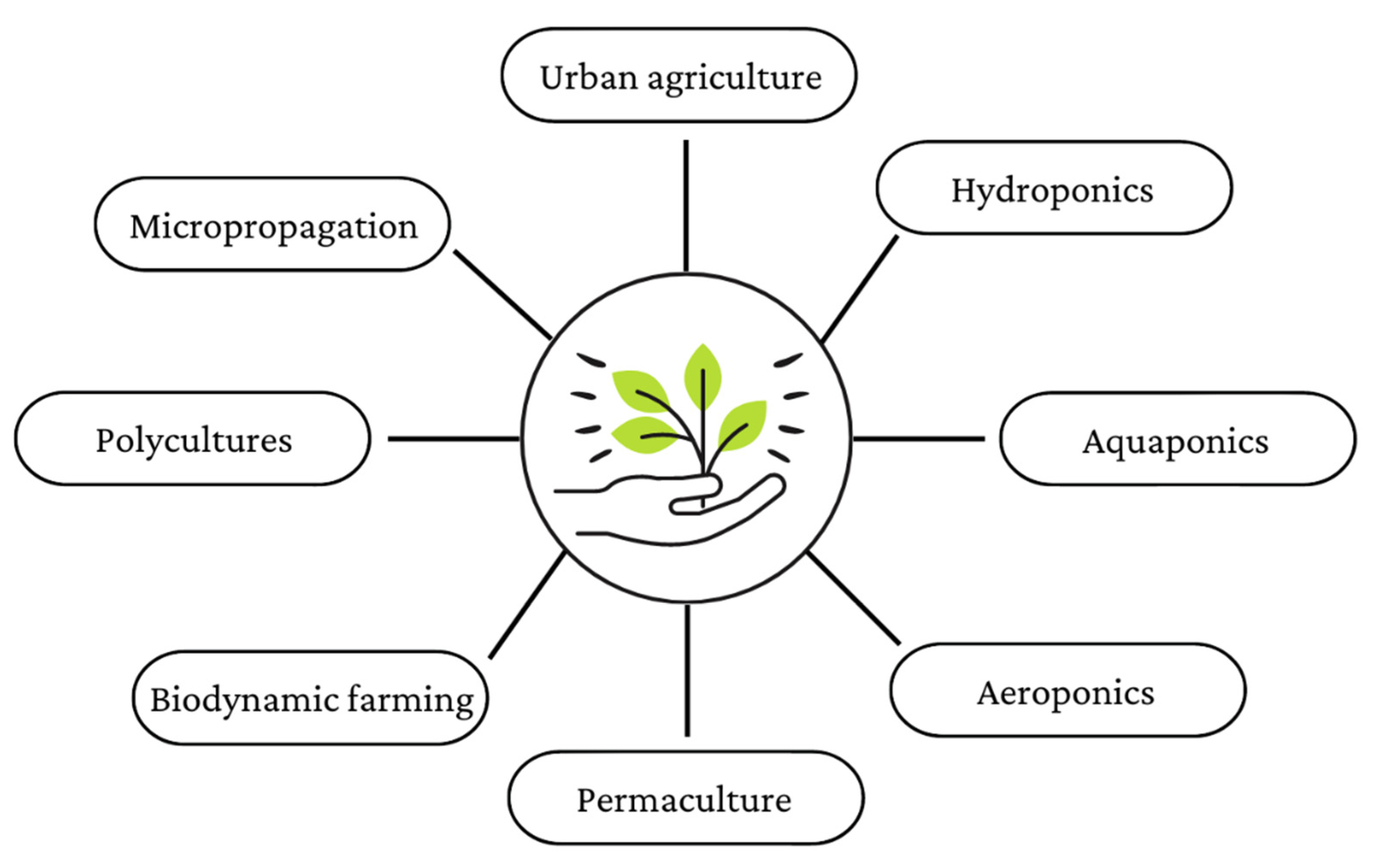
3. Alternative Farming Methods
3.1. Hydroponic System
3.2. Aquaponic System
3.3. Aeroponic System
3.4. Plants Adapted to Hydroponic, Aquaponic and Aeroponic Conditions
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Memete, A.R.; Timar, A.V.; Vuscan, A.N.; Miere, F.; Venter, A.C.; Vicas, S.I. Phytochemical Composition of Different Botanical Parts of Morus Species, Health Benefits and Application in Food Industry. Plants 2022, 11, 152. [Google Scholar] [CrossRef] [PubMed]
- Jan, B.; Parveen, R.; Zahiruddin, S.; Khan, M.U.; Mohapatra, S.; Ahmad, S. Nutritional constituents of mulberry and their potential applications in food and pharmaceuticals: A review. Saudi J. Biol. Sci. 2021, 28, 3909–3921. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Sharma, A.; Thakur, J.; Murali, S.; Bali, K. Mulberry as a Life Savior—A Review. J. Pharmacogn. Phytochem. 2020, 9, 2445–2451. [Google Scholar]
- Paşca, I.; Morar, R.; Dezmirean, D.; Matei, A.; Marghitaş, L. Sericicultură Teoretică şi Practică; Risoprint: Cluj-Napoca, Romania, 2008. [Google Scholar]
- Bhattacharjya, D.; Sadat, A.; Dam, P.; Buccini, D.F.; Mondal, R.; Biswas, T.; Biswas, K.; Sarkar, H.; Bhuimali, A.; Kati, A.; et al. Current concepts and prospects of mulberry fruits for nutraceutical and medicinal benefits. Curr. Opin. Food Sci. 2021, 40, 121–135. [Google Scholar] [CrossRef]
- Chen, X.; Sohouli, M.H.; Nateghi, M.; Melekoglu, E.; Fatahi, S. Impact of mulberry consumption on cardiometabolic risk factors: A systematic review and meta-analysis of randomized-controlled trials. J. Clin. Pharm. Ther. 2022, 47, 1982–1993. [Google Scholar] [CrossRef]
- Ma, Y.; Lv, W.; Gu, Y.; Yu, S. 1-Deoxynojirimycin in mulberry (Morus indica L.) leaves ameliorates stable angina pectoris in patients with coronary heart disease by improving antioxidant and anti-inflammatory capacities. Front. Pharmacol. 2019, 10, 569. [Google Scholar] [CrossRef]
- Wang, Y.; Yu, Z.; Jiang, J.; Li, Y.; Yu, S. Mulberry leaf attenuates atherosclerotic lesions in patients with coronary heart disease possibly via 1-Deoxynojirimycin: A placebo-controlled, double-blind clinical trial. J. Food Biochem. 2021, 45, e13573. [Google Scholar] [CrossRef]
- Zhang, L.; Su, S.; Zhu, Y.; Guo, J.; Guo, S.; Qian, D.; Ouyang, Z.; Duan, J.-A. Mulberry leaf active components alleviate type 2 diabetes and its liver and kidney injury in db/db mice through insulin receptor and TGF-β/Smads signaling pathway. Biomed. Pharmacother. 2019, 112, 108675. [Google Scholar] [CrossRef]
- Duan, Y.; Dai, H.; An, Y.; Cheng, L.; Shi, L.; Lv, Y.; Li, H.; Wang, C.; He, C.; Zhang, H.; et al. Mulberry Leaf Flavonoids Inhibit Liver Inflammation in Type 2 Diabetes Rats by Regulating TLR4/MyD88/NF-B Signaling Pathway. Evid. Based Complement. Altern. Med. 2022, 2022, 3354062. [Google Scholar] [CrossRef]
- Liu, Z.Z.; Liu, Q.H.; Liu, Z.; Tang, J.W.; Chua, E.G.; Li, F.; Xiong, X.S.; Wang, M.M.; Wen, P.B.; Shi, X.Y.; et al. Ethanol extract of mulberry leaves partially restores the composition of intestinal microbiota and strengthens liver glycogen fragility in type 2 diabetic rats. BMC Complement. Med. Ther. 2021, 21, 172. [Google Scholar] [CrossRef]
- Thaipitakwong, T.; Supasyndh, O.; Rasmi, Y.; Aramwit, P. A randomized controlled study of dose-finding, efficacy, and safety of mulberry leaves on glycemic profiles in obese persons with borderline diabetes. Complement. Ther. Med. 2020, 49, 102292. [Google Scholar] [CrossRef]
- Sericulture Technology: Mulberry Cultivation. Available online: https://agritech.tnau.ac.in/sericulture/seri_mulberry%20cultivation.html (accessed on 10 February 2023).
- Kumar Yadav, V.; Padhan, D.; Sobhana, V.; Sen, S.; Josepha, M.; Santha, P.C.; Chandrashekar, M.N.; Praveen Kumar, K.; Tewary, P. Chemical Science Review and Letters Effect of Organic vis-à-vis Conventional Cultivation Practices on Growth and Yield of Mulberry (Morus alba L.). Chem. Sci. Rev. Lett. 2020, 9, 571–577. [Google Scholar]
- Xin, Y.; Meng, S.; Ma, B.; He, W.; He, N. Mulberry genes MnANR and MnLAR confer transgenic plants with resistance to Botrytis cinerea. Plant Sci. 2020, 296, 110473. [Google Scholar] [CrossRef] [PubMed]
- Gnanesh, B.N.; Arunakumar, G.S.; Tejaswi, A.; Supriya, M.; Manojkumar, H.B.; Devi, S.S. Characterization and Pathogenicity of Lasiodiplodia theobromae Causing Black Root Rot and Identification of Novel Sources of Resistance in Mulberry Collections. Plant Pathol. J. 2022, 38, 272–286. [Google Scholar] [CrossRef] [PubMed]
- Paul, M.; Chowdhury, T.; Saha, S. Antimicrobial peptide: A competent tool for plant disease control in mulberry—A review. Vegetos 2022. [Google Scholar] [CrossRef]
- Xu, Y.Q.; Wang, H.; Qin, R.L.; Fang, L.J.; Liu, Z.; Yuan, S.S.; Gai, Y.P.; Ji, X.L. Characterization of NPR1 and NPR4 genes from mulberry (Morus multicaulis) and their roles in development and stress resistance. Physiol. Plant. 2019, 167, 302–316. [Google Scholar] [CrossRef]
- Sun, Z.; Kumar, R.M.S.; Li, J.; Yang, G.; Xie, Y. In Silico search and biological validation of MicroR171 family related to abiotic stress response in mulberry (Morus alba). Hortic. Plant J. 2022, 8, 184–194. [Google Scholar] [CrossRef]
- Litwińczuk, W.; Jacek, B. Micropropagation of mountain mulberry (Morus bombycis Koidz.) ‘kenmochi’ on cytokinin-free medium. Plants 2020, 9, 1533. [Google Scholar] [CrossRef]
- Sarkar, T.; Ravindra, K.N.; Doss, S.G.; Kumar, P.M.P.; Tewary, P. In vitro regeneration of mulberry plants from seedling explants of Morus indica cv. G4 through direct organogenesis. Trees 2022, 36, 113–125. [Google Scholar] [CrossRef]
- Vijayan, K.; Tikader, A.; Teixeira da Silva, J.A. Application of Tissue Culture Techniques for Propagation and Crop Improvement in Mulberry (Morus spp.). Tree For. Sci. Biotechnol. 2011, 5, 1–13. [Google Scholar]
- Dubey, V.; Khan, S.; Shah, K.W.; Raghuwanshi, R.K. Standardization of Protocol for In Vitro Micropropagation of Morus alba L., an Important Economical and Medicinal Plant. Pharm. Biosci. J. 2020, 8, 46–51. [Google Scholar] [CrossRef]
- Van Delden, S.H.; SharathKumar, M.; Butturini, M.; Graamans, L.J.A.; Heuvelink, E.; Kacira, M.; Kaiser, E.; Klamer, R.S.; Klerkx, L.; Kootstra, G.; et al. Current status and future challenges in implementing and upscaling vertical farming systems. Nat. Food 2021, 2, 944–956. [Google Scholar] [CrossRef]
- Khan, H. Medicinal Plants in Light of History: Recognized Therapeutic Modality. J. Evid. Based Complement. Altern. Med. 2014, 19, 216–219. [Google Scholar] [CrossRef] [PubMed]
- Patwardhan, B.; Warude, D.; Pushpangadan, P.; Bhatt, N. Ayurveda and Traditional Chinese Medicine: A Comparative Overview. Adv. Access Publ. 2005, 2, 465–473. [Google Scholar] [CrossRef]
- Jamshidi-Kia, F.; Lorigooini, Z.; Amini-khoei, H. Medicinal plants: Past history and future perspective. J. Herbmed. Pharmacol. 2018, 7, 1–7. [Google Scholar] [CrossRef]
- Dasari, S.; Njiki, S.; Mbemi, A.; Yedjou, C.G. Pharmacological Effects of Cisplatin Combination with Natural Products in Cancer Chemotherapy. Int. J. Mol. Sci. 2022, 23, 1532. [Google Scholar] [CrossRef]
- Huang, M.; Jian, J.; Jian, L. Natural Products in Cancer Therapy: Past, Present and Future. Nat. Prod. Bioprospect. 2021, 11, 5–13. [Google Scholar] [CrossRef]
- Elrayess, R.A.; El-Hak, H.N.G. Anticancer Natural Products: A Review. Cancer Stud. Mol. Med. 2019, 5, 11–22. [Google Scholar] [CrossRef]
- Deepa, M.; Sureshkumar, T.; Satheeshkumar, P.K.; Priya, S. Purified mulberry leaf lectin (MLL) induces apoptosis and cell cycle arrest in human breast cancer and colon cancer cells. Chem. Biol. Interact. 2012, 200, 38–44. [Google Scholar] [CrossRef]
- Sik, J.; Lee, D.; Rak, S.; Wook, J.; Choi, C.; Su, T.; Sung, K.; Hyun, K. Chemical characterization of cytotoxic indole acetic acid derivative from mulberry fruit (Morus alba L.) against human cervical cancer. Bioorg. Chem. 2018, 76, 28–36. [Google Scholar]
- Erden, Y. Sour black mulberry (Morus nigra L.) causes cell death by decreasing mutant p53 expression in HT-29 human colon cancer cells. Food Biosci. 2021, 42, 101113. [Google Scholar] [CrossRef]
- Cheng, K.; Wang, C.; Chang, Y. Mulberry fruits extracts induce apoptosis and autophagy of liver cancer cell and prevent hepatocarcinogenesis in vivo. J. Food Drug Anal. 2019, 28, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Liao, S.; Liu, J.; Xu, M.; Zheng, J. Evaluation of the Liver Cancer Prevention of Anthocyanin Extracts from Mulberry (Morus alba L.) Variety PR-01. Adv. Biosci. Biotechnol. 2018, 9, 423–442. [Google Scholar] [CrossRef]
- Ding, B.; Lv, Y.; Zhang, Y. Anti-tumor effect of morusin from the branch bark of cultivated mulberry in Bel-7402 cells via the MAPK pathway. R. Soc. Chem. 2016, 6, 17396–17404. [Google Scholar] [CrossRef]
- Dalkiliç, S.; Dalkiliç, L.K.; İnci, Ş.; Korkmaz, İ.; Kırbağ, S. Investigation of cytotoxic effect of black mulberry (Morus nigra L.) fruit. Indian J. Tradit. Knowl. 2021, 20, 54–58. [Google Scholar]
- Fallah, S.; Karimi, A.; Panahi, G.; Nejad, S.G.; Fadaei, R.; Seifi, M. Human colon cancer HT-29 cell death responses to doxorubicin and Morus Alba leaves flavonoid extract. Cell Mol. Biol. 2016, 62, 72–77. [Google Scholar]
- Pranata, R.; Henrina, J.; Matthew, W.; Lawrensia, S.; Huang, I. Diabetes and COVID-19: The past, the present, and the future. Metabolism 2021, 121, 154814. [Google Scholar] [CrossRef]
- Marlen, S.; Mata, R.; Andrade-Cetto, A. Molecules Isolated from Mexican Hypoglycemic Plants: A Review. Molecules 2020, 25, 4145. [Google Scholar]
- Yang, C.; Deng, X.; Tang, B.; Wang, X. Study on Hypoglycemic Effect of Mulberry Leaf Extract Based on Big Data analysis. J. Phys. Conf. Ser. 2021, 1774, 022105. [Google Scholar] [CrossRef]
- Mohammadi, A.; Naik, P.R. Evaluation of hypoglycemic effect of Morus alba in an animal model. Indian J. Pharmacol. 2008, 40, 15–18. [Google Scholar]
- Eo, H.J.; Park, J.H.; Park, G.H.; Lee, M.H.; Lee, J.R.; Koo, J.S.; Jeong, J.B. Anti-inflammatory and anti-cancer activity of mulberry (Morus alba L.) root bark. BMC Complement. Altern. Med. 2014, 14, 200. [Google Scholar] [CrossRef] [PubMed]
- Yin, X.L.; Liu, H.Y.; Zhang, Y.Q. Mulberry branch bark powder significantly improves hyperglycemia and regulates insulin secretion in type II diabetic mice. Food Nutr. Res. 2017, 61, 1368847. [Google Scholar] [CrossRef]
- Qiu, F.; Zhang, Y.Q. Metabolic effects of mulberry branch bark powder on diabetic mice based on GC-MS metabolomics approach. Nutr. Metab. 2019, 16, 10. [Google Scholar] [CrossRef] [PubMed]
- Mahesh, D.S.; Vidhathri, B.S.; Vidyashree, D.N.; Narayanaswamy, T.K.; Subbarayappa, C.T.; Muthuraju, R. Biochemical Composition and Pharmacological Properties of Mulberry (Morus spp.)—A Review. Int. J. Curr. Microbiol. Appl. Sci. 2017, 6, 2207–2217. [Google Scholar] [CrossRef]
- Dhiman, S.; Kumar, V.; Mehta, C.M.; Gat, Y.; Kaur, S. Bioactive compounds, health benefits and utilisation of Morus spp.—A comprehensive review. J. Hortic. Sci. Biotechnol. 2020, 95, 8–18. [Google Scholar] [CrossRef]
- Hu, L.; Wang, C.; Guo, X.; Chen, D.; Zhou, W.; Chen, X.; Zhang, Q. Flavonoid Levels and Antioxidant Capacity of Mulberry Leaves: Effects of Growth Period and Drying Methods. Front. Plant Sci. 2021, 12, 684974. [Google Scholar] [CrossRef]
- Szwajgier, D.; Borowiec, K.; Pustelniak, K. The neuroprotective effects of phenolic acids: Molecular mechanism of action. Nutrients 2017, 9, 477. [Google Scholar] [CrossRef]
- Flaczyk, E.; Kobus-Cisowska, J.; Przeor, M.; Korczak, J.; Remiszewski, M.; Korbas, E.; Buchowski, M. Chemical characterization and antioxidative properties of Polish variety of Morus alba L. leaf aqueous extracts from the laboratory and pilot-scale processes. Agric. Sci. 2013, 4, 141–147. [Google Scholar] [CrossRef]
- Abotaleb, M.; Liskova, A.; Kubatka, P.; Büsselberg, D. Therapeutic potential of plant phenolic acids in the treatment of cancer. Biomolecules 2020, 10, 221. [Google Scholar] [CrossRef]
- Yan, X.; Qi, M.; Li, P.; Zhan, Y.; Shao, H. Apigenin in cancer therapy: Anti-cancer effects and mechanisms of action. Cell Biosci. 2017, 7, 50. [Google Scholar] [CrossRef]
- Islam, A.; Islam, M.S.; Rahman, M.K.; Uddin, M.N.; Akanda, M.R. The pharmacological and biological roles of eriodictyol. Arch. Pharm. Res. 2020, 43, 582–592. [Google Scholar] [CrossRef] [PubMed]
- Pari, L.; Mohamed Jalaludeen, A. Protective role of sinapic acid against arsenic—Induced toxicity in rats. Chem. Biol. Interact. 2011, 194, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Shang, P.; Li, D. Luteolin: A Flavonoid that has multiple cardio-protective effects and its molecular mechanisms. Front. Pharmacol. 2017, 8, 692. [Google Scholar] [CrossRef] [PubMed]
- Gundogdu, M.; Muradoglu, F.; Sensoy, R.I.G.; Yilmaz, H. Determination of fruit chemical properties of Morus nigra L., Morus alba L. and Morus rubra L. by HPLC. Sci. Hortic. 2011, 132, 37–41. [Google Scholar] [CrossRef]
- Du, Q.; Zheng, J.; Xu, Y. Composition of anthocyanins in mulberry and their antioxidant activity. J. Food Compos. Anal. 2008, 21, 390–395. [Google Scholar] [CrossRef]
- Thomas Pannakal, S.; Eilstein, J.; Prasad, A.; Ekhar, P.; Shetty, S.; Peng, Z.; Bordier, E.; Boudah, S.; Paillat, L.; Marrot, L.; et al. Comprehensive characterization of naturally occurring antioxidants from the twigs of mulberry (Morus alba) using on-line high-performance liquid chromatography coupled with chemical detection and high-resolution mass spectrometry. Phytochem. Anal. 2022, 33, 105–114. [Google Scholar] [CrossRef]
- Yulistiani, D.; Jelan, Z.A.; Liang, J.B.; Yaakub, H.; Abdullah, N. Effects of supplementation of mulberry (Morus alba) foliage and urea-rice bran as fermentable energy and protein sources in sheep fed urea-treated rice straw based diet. Asian-Australas J. Anim. Sci. 2015, 28, 494–501. [Google Scholar] [CrossRef]
- Salinas-Chavira, J.; Castillo-Martínez, O.; Ramirez-Bribiesca, J.E.; Mellado, M. Effect of increasing levels of white mulberry leaves (Morus alba) on ruminal dry matter degradability in lambs. Trop. Anim. Health Prod. 2011, 43, 995–999. [Google Scholar] [CrossRef]
- Islam, M.R.; Siddiqui, M.N.; Khatun, A.; Siddiky, M.N.A.; Rahman, M.Z.; Bostami, A.B.M.R.; Selim, A.S.M. Dietary effect of mulberry leaf (Morus alba) meal on growth performance and serum cholesterol level of broiler chickens. SAARC J. Agric. 2014, 12, 79–89. [Google Scholar] [CrossRef]
- Attila, C.R.; Cristian, R.; Alina, N.; Mihaela, R.M.; Veronica, L.; Alexandru, A.E. Molecular and bioinformatics analysis of the relative expression profiles of dorsal, Toll-1, Relish and Duox genes in young versus old diutinus workers of Apis mellifera. Rom. Biotechnol. Lett. 2016, 21, 11513–11526. [Google Scholar]
- Rahmathulla, V.K.; Tilak, R.; Rajan, R.K. Influence of Moisture Content of Mulberry Leaf on Growth and Silk Production in Bombyx mori L. Caspian J. Env. Sci. 2006, 4, 25–30. [Google Scholar]
- Susanti, S. and Tanjung, M. Effect Of The Quality Of Mulberry Leaves Morus alba Against The Silkworm Nutrition Index Bombyx mori L. (Lepidoptera:Bombicidae). Int. J. Ecophysiol. 2021, 3, 1. [Google Scholar]
- Gug, K. Physiological and Whitening Effects of Morus alba Extracts. J. Chosun Nat. Sci. 2012, 5, 46–52. [Google Scholar] [CrossRef][Green Version]
- Cai, M.; Mu, L.; Wang, Z.L.; Liu, J.Y.; Liu, T.L.; Wanapat, M.; Huang, B.Z. Assessment of mulberry leaf as a potential feed supplement for animal feeding in P.R. China. Asian-Australas. J. Anim. Sci. 2019, 32, 1145–1152. [Google Scholar] [CrossRef]
- On-Nom, N.; Suttisansanee, U.; Tongmai, J.; Khemthong, C.; Chamchan, R.; Prangthip, P.; Hanboonkunupakarn, B.; Chupeerach, C. Consumption of Anthocyanin-Rich Mulberry Fruit Jelly with a High-Fat Meal Decreases Postprandial Serum Cardiometabolic Risk Factors in Dyslipidemia Subjects. J. Nutr. Metab. 2020, 2020, 1370951. [Google Scholar] [CrossRef]
- Rohela, G.K.; Shukla, P.; Kumar, R.M.; Chowdhury, S.R. Mulberry (Morus spp.): An ideal plant for sustainable development. Trees For. People 2020, 2, 100011. [Google Scholar] [CrossRef]
- Jiang, Y.; Huang, R.; Yan, X.; Jia, C.; Jiang, S.; Long, T. Mulberry for environmental protection. Pak. J. Bot. 2017, 49, 781–788. [Google Scholar]
- Das, S.; Chatterjee, A.; Pal, T.K. Organic farming in India: A vision towards a healthy nation. Food Qual. Saf. 2021, 4, 69–76. [Google Scholar] [CrossRef]
- Durán-Lara, E.F.; Valderrama, A.; Marican, A. Natural organic compounds for application in organic farming. Agriculture 2010, 10, 41. [Google Scholar] [CrossRef]
- Łuczka, W.; Kalinowski, S. Barriers to the development of organic farming: A polish case study. Agriculture 2020, 10, 536. [Google Scholar] [CrossRef]
- Sakthivel, N.; Ravikumar, J.; Chikkanna; Mukund, V.K.; Bindroo, B.B.; Sivaprasad, V. Organic Farming in Mulberry: Recent Breakthrough; Regional Sericultural Research Station: Salem, India, 2014. [Google Scholar]
- Chakraborty, B.; Kundu, M.; Chattopadhyay, R.N. Organic Farming with Bio-mulching—A New Paradigm for Sustainable Leaf Yield & Quality of Mulberry (Morus alba L.) under Rainfed Lateritic Soil Condition. Agric. Agric. Sci. Procedia 2016, 11, 31–37. [Google Scholar]
- Population. Available online: https://www.un.org/en/global-issues/population (accessed on 4 June 2022).
- Zhang, Z.; Rod, M.; Hosseinian, F. A Comprehensive Review on Sustainable Industrial Vertical Farming Using Film Farming Technology. Sustain. Agric. Res. 2020, 10, 46. [Google Scholar] [CrossRef]
- Saxena, N.N. The Review on Techniques of Vertical Farming. Int. J. Mod. Agric. 2021, 10, 732–738. [Google Scholar]
- Gumisiriza, M.S.; Ndakidemi, P.; Nalunga, A.; Mbega, E.R. Building sustainable societies through vertical soilless farming: A cost-effectiveness analysis on a small-scale non-greenhouse hydroponic system. Sustain. Cities Soc. 2022, 83, 103923. [Google Scholar] [CrossRef]
- Lei, C.; Engeseth, N.J. Comparison of growth characteristics, functional qualities, and texture of hydroponically grown and soil-grown lettuce. LWT 2021, 150, 111931. [Google Scholar] [CrossRef]
- Sronsri, C.; Sittipol, W.; U-yen, K. Quantity and quality of lettuce (Lactuca sativa L.) grown by a circulating hydroponic method with a Halbach array magnetizer. J. Food Compos. Anal. 2022, 108, 104460. [Google Scholar] [CrossRef]
- Rattan, S.; Partap, M.; Kanika; Kumar, S.; Warghat, A.R. Nutrient feeding approach enhances the vegetative growth biomass, volatile oil composition, and myristicin content in hydroponically cultivated Petroselinum crispum (Mill.) Nyman. J. Appl. Res. Med. Aromat. Plants 2022, 26, 100359. [Google Scholar] [CrossRef]
- Musa, A.; Hassan, M.; Hamada, M.; Aliyu, F.; Aliyu, F.M. Low-Power Deep Learning Model for Plant Disease Detection for Smart-Hydroponics Using Knowledge Distillation Techniques. J. Low Power Electron. Appl. 2022, 12, 24. [Google Scholar] [CrossRef]
- Shrivastava, A.; Nayak, C.K.; Dilip, R.; Samal, S.R.; Rout, S.; Ashfaque, S.M. Automatic robotic system design and development for vertical hydroponic farming using IoT and big data analysis. Mater. Today Proc. 2021. [Google Scholar] [CrossRef]
- Sharma, N.; Acharya, S.; Kumar, K.; Singh, N.; Chaurasia, O.P. Hydroponics as an advanced technique for vegetable production: An overview. J. Soil Water Conserv. 2018, 17, 364. [Google Scholar] [CrossRef]
- Asao, T. Hydroponics—A Standard Methodology for Plant Biological Researches; InTech: Rijeka, Croatia, 2012. [Google Scholar]
- Domingues, D.S.; Takahashi, H.W.; Camara, C.A.P.; Nixdorf, S.L. Automated system developed to control pH and concentration of nutrient solution evaluated in hydroponic lettuce production. Comput. Electron. Agric. 2012, 84, 53–61. [Google Scholar] [CrossRef]
- Sakurai, M.; Sato, S.; Fukushima, T.; Konishi, T. Characteristics of Morus alba L. Cultured by In-Room Hydroponics. Am. J. Plant Sci. 2022, 13, 91–108. [Google Scholar] [CrossRef]
- Goddek, S.; Joyce, A.; Kotzen, B.; Burnell Editors, G.M. Aquaponics Food Production Systems Combined Aquaculture and Hydroponic Production Technologies for the Future; Springer: Berlin/Heidelberg, Germany, 2019. [Google Scholar] [CrossRef]
- Diver, S. Aquaponics-Integration of Hydroponics with Aquaculture; ATTRA Publication: Davis, CA, USA, 2009. [Google Scholar]
- Kledal, P.R.; Thorarinsdottir, R. Aquaponics: A Commercial Niche for Sustainable Modern Aquaculture. In Sustainable Aquaculture; Springer International Publishing: Cham, Switzerland, 2018; pp. 173–190. [Google Scholar]
- Danner, R.I.; Mankasingh, U.; Anamthawat-Jonsson, K.; Thorarinsdottir, R.I. Designing aquaponic production systems towards integration into greenhouse farming. Water 2019, 11, 2123. [Google Scholar] [CrossRef]
- Lobillo-Eguíbar, J.; Fernández-Cabanás, V.M.; Bermejo, L.A.; Pérez-Urrestarazu, L. Economic sustainability of small-scale aquaponic systems for food self-production. Agronomy 2020, 10, 1468. [Google Scholar] [CrossRef]
- Wei, Y.; Li, W.; An, D.; Li, D.; Jiao, Y.; Wei, Q. Equipment and Intelligent Control System in Aquaponics: A Review. IEEE Access 2019, 7, 169306–169326. [Google Scholar] [CrossRef]
- Baganz, G.F.M.; Junge, R.; Portella, M.C.; Goddek, S.; Keesman, K.J.; Baganz, D.; Staaks, G.; Shaw, C.; Lohrberg, F.; Kloas, W. The aquaponic principle—It is all about coupling. Rev. Aquac. 2022, 14, 252–264. [Google Scholar] [CrossRef]
- Ghamkhar, R.; Hartleb, C.; Wu, F.; Hicks, A. Life cycle assessment of a cold weather aquaponic food production system. J. Clean. Prod. 2020, 244, 118767. [Google Scholar] [CrossRef]
- Love, D.C.; Fry, J.P.; Li, X.; Hill, E.S.; Genello, L.; Semmens, K.; Thompson, R.E. Commercial aquaponics production and profitability: Findings from an international survey. Aquaculture 2015, 435, 67–74. [Google Scholar] [CrossRef]
- Yang, T.; Kim, H.J. Nutrient management regime affects water quality, crop growth, and nitrogen use efficiency of aquaponic systems. Sci. Hortic. 2019, 256, 108619. [Google Scholar] [CrossRef]
- Yang, T.; Kim, H.J. Characterizing nutrient composition and concentration in tomato-, basil-, and lettuce-based aquaponic and hydroponic systems. Water 2020, 12, 1259. [Google Scholar] [CrossRef]
- Ulum, M.H.; Ibadillah, A.F.; Alfita, R.; Aji, K.; Rizkyandi, R. Smart Aquaponic System Based Internet of Things (IoT); Institute of Physics Publishing: Bristol, UK, 2019. [Google Scholar]
- Dhal, S.B.; Jungbluth, K.; Lin, R.; Sabahi, S.P.; Bagavathiannan, M.; Braga-Neto, U.; Kalafatis, S. A Machine-Learning-Based IoT System for Optimizing Nutrient Supply in Commercial Aquaponic Operations. Sensors 2022, 22, 3510. [Google Scholar] [CrossRef] [PubMed]
- Lauguico, S.C.; Concepcion, R.S.; Alejandrino, J.D.; Tobias, R.R.; Macasaet, D.D.; Dadios, E.P. A comparative analysis of machine learning algorithms modeled from machine vision-based lettuce growth stage classification in smart aquaponics. Int. J. Environ. Sci. Dev. 2020, 11, 442–449. [Google Scholar] [CrossRef]
- Reyes-Yanes, A.; Martinez, P.; Ahmad, R. Towards automated aquaponics: A review on monitoring, IoT, and smart systems. J. Clean. Prod. 2020, 263, 121571. [Google Scholar] [CrossRef]
- Asciuto, A.; Schimmenti, E.; Cottone, C.; Borsellino, V. A financial feasibility study of an aquaponic system in a Mediterranean urban context. Urban For. Urban Green. 2019, 38, 397–402. [Google Scholar] [CrossRef]
- Greenfeld, A.; Becker, N.; Bornman, J.F.; Spatari, S.; Angel, D.L. Monetizing environmental impact of integrated aquaponic farming compared to separate systems. Sci. Total Environ. 2021, 792, 148459. [Google Scholar] [CrossRef]
- Suárez-Cáceres, G.P.; Lobillo-Eguíbar, J.; Fernández-Cabanás, V.M.; Quevedo-Ruiz, F.J.; Pérez-Urrestarazu, L. Polyculture production of vegetables and red hybrid tilapia for self-consumption by means of micro-scale aquaponic systems. Aquac. Eng. 2021, 95, 102181. [Google Scholar] [CrossRef]
- Weathers, P.J.; Zobel¢, R.W. Aeroponics for the culture of organisms, tissues and cells. Biotechnol. Adv. 1992, 10, 93–115. [Google Scholar] [CrossRef]
- Carter, W. A method of growing plants in water vapor to facilitate examination of roots. Phytopathology 1942, 32, 623–625. [Google Scholar]
- Buckseth, T.; Sharma, A.K.; Pandey, K.K.; Singh, B.P.; Muthuraj, R. Methods of pre-basic seed potato production with special reference to aeroponics—A review. Sci. Hortic. 2016, 204, 79–87. [Google Scholar] [CrossRef]
- Kratsch, H.A.; Graves, W.R.; Gladon, R.J. Aeroponic system for control of root-zone atmosphere. Environ. Exp. Bot. 2006, 55, 70–76. [Google Scholar] [CrossRef]
- Gwynn-Jones, D.; Dunne, H.; Donnison, I.; Robson, P.; Sanfratello, G.M.; Schlarb-Ridley, B.; Hughes, K.; Convey, P. Can the optimisation of pop-up agriculture in remote communities help feed the world? Glob. Food Secur. 2018, 18, 35–43. [Google Scholar] [CrossRef]
- Johnson, M.; Villani, T.S.; Azmat, A.; Simon, J.E.; Both, A.J. Evaluation of algal biomass production on vertical aeroponic substrates. Algal Res. 2015, 10, 240–248. [Google Scholar] [CrossRef]
- Wang, M.; Dong, C.; Gao, W. Evaluation of the growth, photosynthetic characteristics, antioxidant capacity, biomass yield and quality of tomato using aeroponics, hydroponics and porous tube-vermiculite systems in bio-regenerative life support systems. Life Sci. Space Res. 2019, 22, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Patil, N.L.; Kulkarni, A.A.; Amalnerkar, D.; Kamble, S.C. Exploration of wheatgrass as functional food by using urban agriculture models for regulating growth & nutrients. S. Afr. J. Bot. 2022, 151, 284–289. [Google Scholar]
- Pasch, J.; Appelbaum, S.; Palm, H.W.; Knaus, U. Growth of Basil (Ocimum basilicum) in Aeroponics, DRF, and Raft Systems with Effluents of African Catfish (Clarias gariepinus) in Decoupled Aquaponics (s.s.). AgriEngineering 2021, 3, 559–574. [Google Scholar] [CrossRef]
- Tunio, M.H.; Hussain Tunio, M.; Gao, J. Technological Modernization and Its Influence on Agriculture Sustainability, Aeroponics Systems in Belt and Road Countries. In Proceedings of the 10th Sino-Foreign Postgraduate Academic Forum Jiangsu University, Zhenjiang, China, 22 November 2020. [Google Scholar]
- De Miranda, F.R.; da Silva, V.B.; dos Santos, F.S.R.; Rossetti, A.G.; da Silva, C.d.F.B. Production of strawberry cultivars in closed hydroponic systems and coconut fibre substrate. Rev. Ciência Agronômica 2014, 45, 833–841. [Google Scholar] [CrossRef]
- Tzortzakis, N.; Chrysargyris, A.; Asao, T.; Asaduzzaman, M.; Raihan Talukder, M.; Tanaka, H.; Ueno, M.; Kawaguchi, M.; Yano, S.; Ban, T. Production of Low-Potassium Content Melon Through Hydroponic Nutrient Management Using Perlite Substrate. Front. Plant Sci. 2018, 9, 1382. [Google Scholar]
- Bakiu, R.; Tafaj, C.; Taci, J. First Study about Aquaponic Systems in Albania. J. Mar. Biol. Aquac. Res. 2018, 1, 1–7. [Google Scholar] [CrossRef]
- Fernández-Cabanás, V.M.; Delgado, A.; Lobillo-Eguíbar, J.R.; Pérez-Urrestarazu, L. Early production of strawberry in aquaponic systems using commercial hydroponic bands. Aquac. Eng. 2022, 97, 102242. [Google Scholar] [CrossRef]
- Rodríguez-Delfín, A.; Molina, L. Advances of Hydroponics in Latin America. Acta Hortic. 2012, 947, 23–32. [Google Scholar] [CrossRef]
- Krastanova, M.; Sirakov, I.; Ivanova-Kirilova, S.; Yarkov, D.; Orozova, P. Aquaponic systems: Biological and technological parameters. Biotechnol. Biotechnol. Equip. 2022, 36, 305–316. [Google Scholar] [CrossRef]
- Hayden, A.L. Aeroponic and Hydroponic Systems for Medicinal Herb, Rhizome, and Root Crops. HortScience 2006, 41, 536–538. [Google Scholar] [CrossRef]
- Kumar Naik, P. Review-Production and utilisation of hydroponics fodder. Indian J. Anim. Nutr. 2015, 32, 1–9. [Google Scholar]
- Amankwaa-Yeboah, P.; Bessah, E. Assessment of Aquaponics-based Food Production System Effluents on the Performance of Maize. Int. J. Agric. Res. Rev. 2017, 5, 615–627. [Google Scholar]
- Germer, J.; Sauerborn, J.; Asch, F.; de Boer, J.; Schreiber, J.; Weber, G.; Müller, J. Skyfarming an ecological innovation to enhance global food security. J. Verbrauch. Lebensm. 2011, 6, 237–251. [Google Scholar] [CrossRef]
- Wimmerova, L.; Keken, Z.; Solcova, O.; Bartos, L.; Spacilova, M. A Comparative LCA of Aeroponic, Hydroponic, and Soil Cultivations of Bioactive Substance Producing Plants. Sustainability 2022, 14, 2421. [Google Scholar] [CrossRef]
- Hilmi, E.; Sari, L.K.; Cahyo, T.N.; Mahdiana, A.; Soedibya, P.H.T.; Sudiana, E. Survival and growth rates of mangroves planted in vertical and horizontal aquaponic systems in North Jakarta, Indonesia. Biodiversitas 2022, 23, 687–694. [Google Scholar] [CrossRef]
- Mangaiyarkarasi, R. Aeroponics System for Production of Horticultural Crops. Madras Agric. J. 2020, 1, 32–40. [Google Scholar]
- Mărghitaș, L.; Dezmirean, D.; Pașca, I. Sericicultura; Mediamira: Cluj-Napoca, Romania, 2003. [Google Scholar]
- Jürkenbeck, K.; Heumann, A.; Spiller, A. Sustainability matters: Consumer acceptance of different vertical farming systems. Sustainability 2019, 11, 4052. [Google Scholar] [CrossRef]
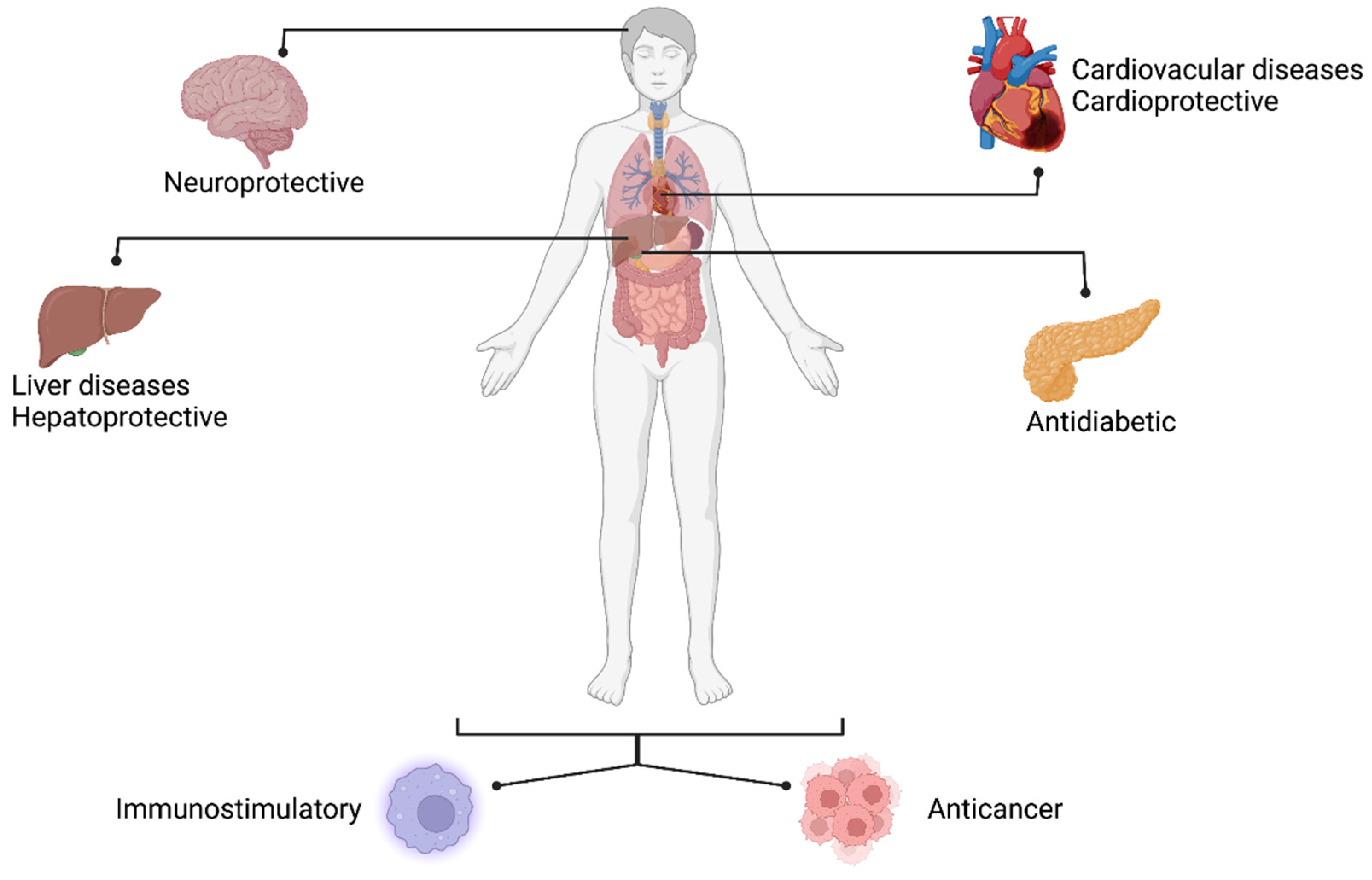


| Plant Part | Health Benefits | Biological Compounds | References |
|---|---|---|---|
| Leaves | Anti-diabetic activity | 1-Deoxynojirimycin, Moranolin, 2-aryl-benzofuran | [46,47] |
| Anticancer activity | Morin, Kuwanon S, 8-granilapigenin, ciclomulberrin, morusin, ciclomorusin, atalantoflavones, Kaempherol-3-O-glucoside (Astragalin) and derivates, Quercetin and derivates, Epicatechin, moracins, phenolic acids (gallic, protocatechuic, vanilinc, p-coumaric, ferulic), apigenin and derivates | [46,47,48,49,50,51,52] | |
| Antioxidant activity | Quercitrin, rutin, Eriodictyol, gallic acid, chlorogenic acid, sinapic acid | [47,48,49,50,53,54] | |
| Hypolipidemic effect | 1-Deoxynojirimycin, Caffeic acid, Cyanidin-3-O-rutinoside | [46,47,49] | |
| Neuroprotective activity | γ-aminobutyric acid, gallic acid, chlorogenic acid | [46,48,49] | |
| Anti-inflammatory effects | Chlorogenic acid, Caffeic acid | [47,49,50] | |
| Anti-viral activity | Cyanidin-3-O-rutinoside, 1-Deoxynojirimycin | [46,47] | |
| Cardio-protective | Mulberrofurans, Luteolins | [48,55] | |
| Fruits | Anti-diabetic activity | Polysaccharides (FMAP, MFP, MFP-1, MFP-2, MP, PMF1, PMF2, PMF3, MFP3P, JS-MP-1) | [47] |
| Anticancer activity | Vanillic acid, caffeic acid, ferulic acid protocatechuic acid, gallic acid | [49,51,56] | |
| Antioxidant activity | Isoquercitrin, rutin, phenolic acids (gallic, chlorogenic, phloridzin, citric, malic, tartaric, lactic, succinic, fumaric, acetic), anthocians (Cyanidin-3-O-β-D-glucopiranoside, Cyanidin 3-β-D-glucopyranoside, Cyanidin-7-O- β-D-glucopyranoside, Cyanidin-3-O-(6′′-O-α-rhamnopyranosyl-β-D-glucopyranoside), Cyanidin-3-O-(6′’-O-α-rhamnopyranosyl-β-D-galactopyranoside), catechins | [47,48,49,50,51,56,57,58] | |
| Neuroprotective | Quercetin and derivates, gallic acid, chlorogenic acid, Cyanidin-3-O-β-D-glucopiranoside | [46,49,56,57] | |
| Anti-inflammatory effects | Chlorogenic acid, p-coumaric acid, Caffeic acid, Protocatechuic acid, o-coumaric acid | [47,49,56,57] | |
| Root bark | Anti-diabetic activity | 1-Deoxynojirimycin | [56] |
| Anticancer activity | Morusin, oxyhidromorusin, moracins, Glycoside,5,2′Dihidroxyflavone-7, 4′-di-O-D-glucoside | [46,47,56] | |
| Antioxidant activity | Mulberroside A, Mulberoside C | [47,59] | |
| Neuroprotective and hepatoprotective activities | Sanggenones (B, D, E, G, M, O, T) | [56] | |
| Anti-inflammatory effects | Mulberrofurans, Morusin, oxyhidromorusin, sanggenones | [47,56] | |
| Anti-viral activity | Leachianone G, 1-Deoxynojirimycin, Eudraflavone B hydroperoxide, α-acetyl-amyrin | [46,47,60] | |
| Anti-bacterial effect | Kuwanones (G, H, M, L, O) | [47] | |
| Anti-hypertensive effect | Moracenin A, Moracenin C, Moracenin D | [56] |
| Type of the Crop | Examples | Hydroponic Culture | Aquaponic Culture | Aeroponic Culture | References |
|---|---|---|---|---|---|
| Fruits | strawberries, melons | ✓ | ✓ | ✓ | [116,117,118,119,120] |
| Vegetables | tomatoes, spinach, lettuce, green beans, bell peppers, cucumbers, eggplants, celery, potatoes | ✓ | ✓ | ✓ | [118,120,121] |
| Spices | basil, parsley, mint, coriander, ginger, echinacea, licorice | ✓ | ✓ | ✓ | [84,121,122] |
| Cereals | rice, maize, sorghum, oats, wheat | ✓ | ✓ | ✓ | [84,123,124,125] |
| Woody plants | Coffee tree, bamboo, mangrove tree | ✓ | ✓ | ✓ | [126] |
| Mulberry | ✓ | [87] | |||
| Othe plants | Indian aloe, roses, philodendron, Cineraria | ✓ | ✓ | ✓ | [84,123,126,127,128] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baciu, E.-D.; Baci, G.-M.; Moise, A.R.; Dezmirean, D.S. A Status Review on the Importance of Mulberry (Morus spp.) and Prospects towards Its Cultivation in a Controlled Environment. Horticulturae 2023, 9, 444. https://doi.org/10.3390/horticulturae9040444
Baciu E-D, Baci G-M, Moise AR, Dezmirean DS. A Status Review on the Importance of Mulberry (Morus spp.) and Prospects towards Its Cultivation in a Controlled Environment. Horticulturae. 2023; 9(4):444. https://doi.org/10.3390/horticulturae9040444
Chicago/Turabian StyleBaciu, Ecaterina-Daniela, Gabriela-Maria Baci, Adela Ramona Moise, and Daniel Severus Dezmirean. 2023. "A Status Review on the Importance of Mulberry (Morus spp.) and Prospects towards Its Cultivation in a Controlled Environment" Horticulturae 9, no. 4: 444. https://doi.org/10.3390/horticulturae9040444
APA StyleBaciu, E.-D., Baci, G.-M., Moise, A. R., & Dezmirean, D. S. (2023). A Status Review on the Importance of Mulberry (Morus spp.) and Prospects towards Its Cultivation in a Controlled Environment. Horticulturae, 9(4), 444. https://doi.org/10.3390/horticulturae9040444









