Effects of Rootstock on Water Stress, Physiological Parameters, and Growth of the Pistachio Tree
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Stem Water Potential
2.3. Photosynthesis Measurements
2.4. Determination of Chlorophyll and Other Pigments
2.5. Biomass Weight and Plant Growth
2.6. Statistical Analysis
3. Results
3.1. Stem Water Potential (MPa)
3.2. Gas Exchange and Photosynthesis
3.3. Efficiency of Carboxylation and Water Use
3.4. Chlorophyll and Other Pigments
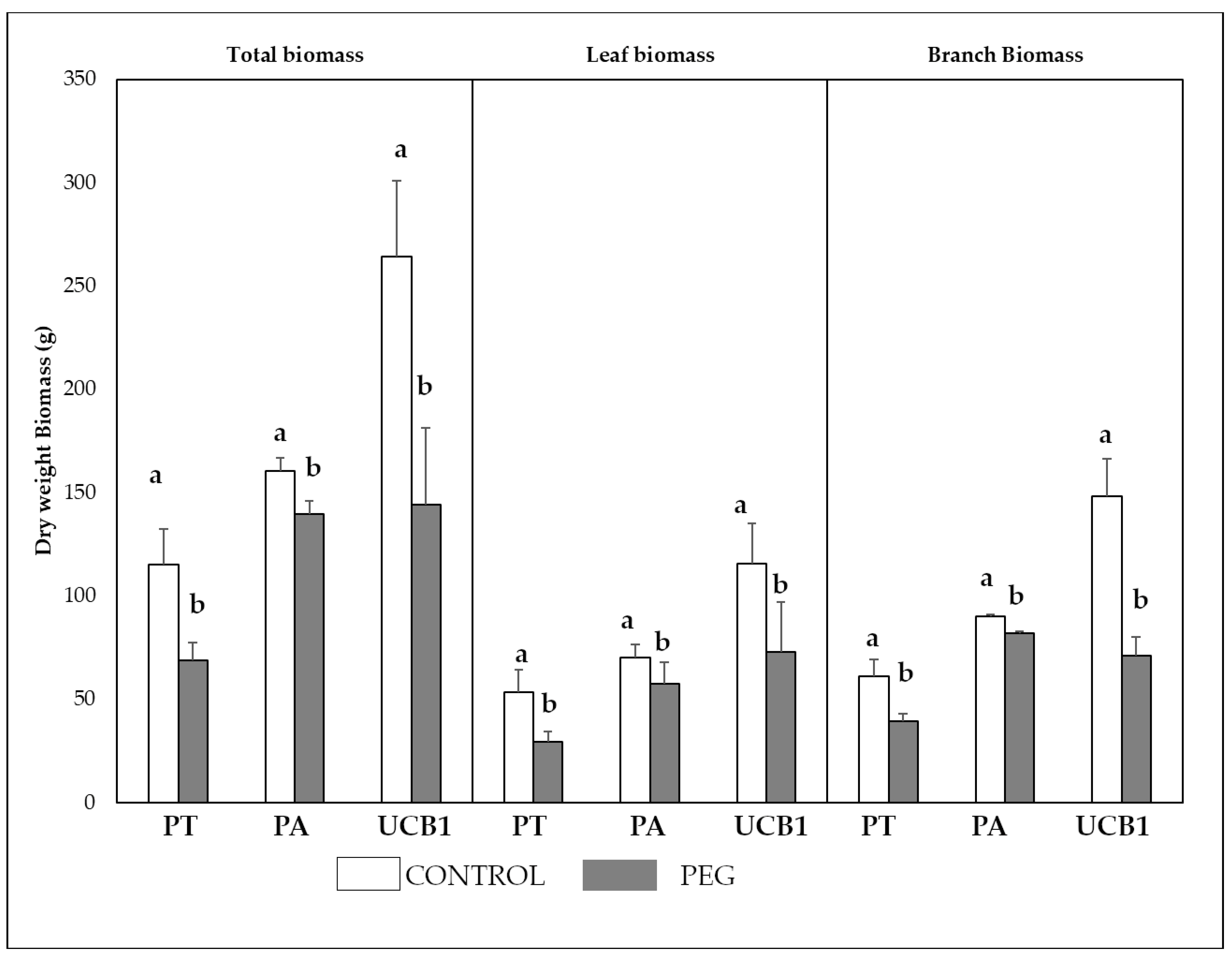
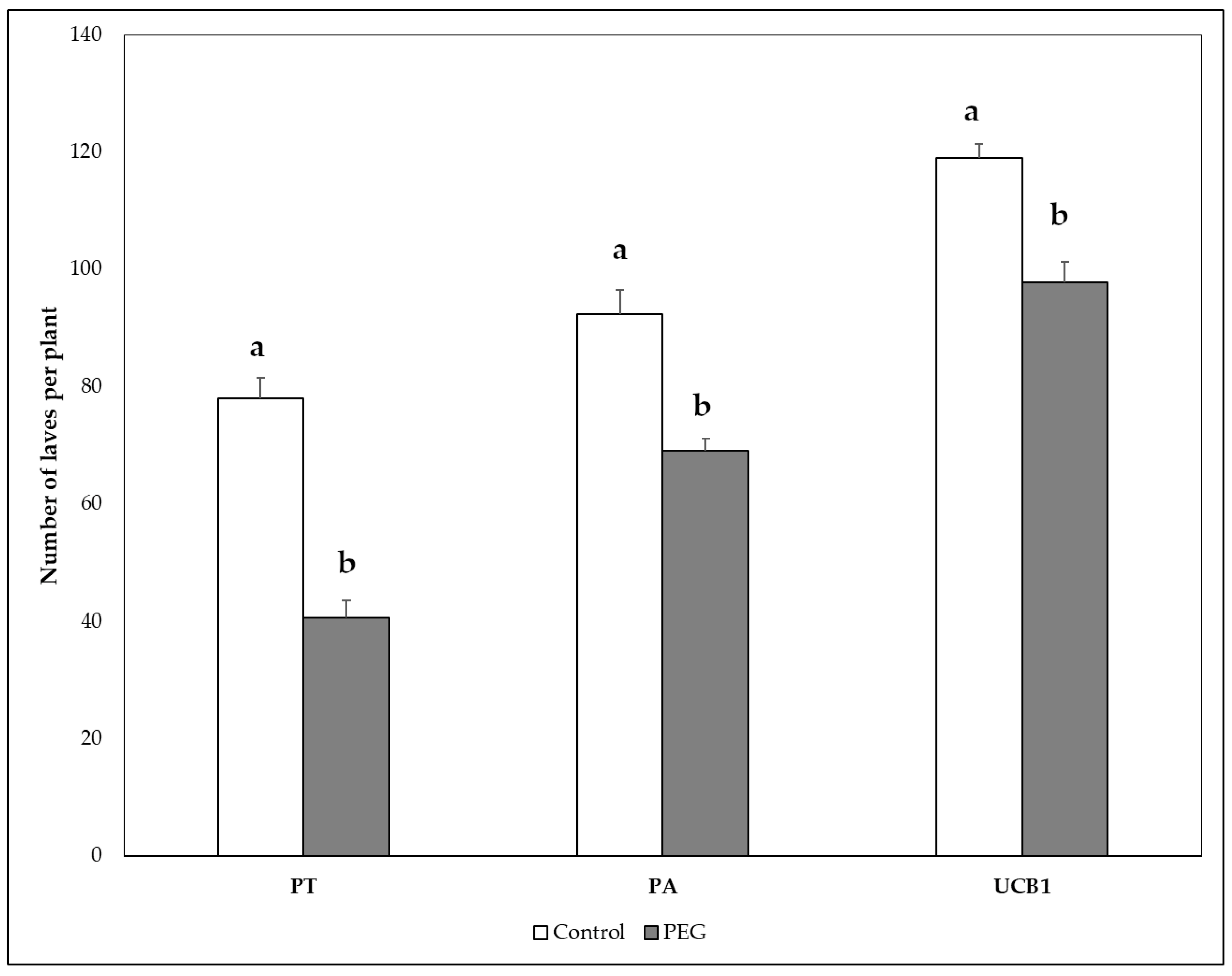
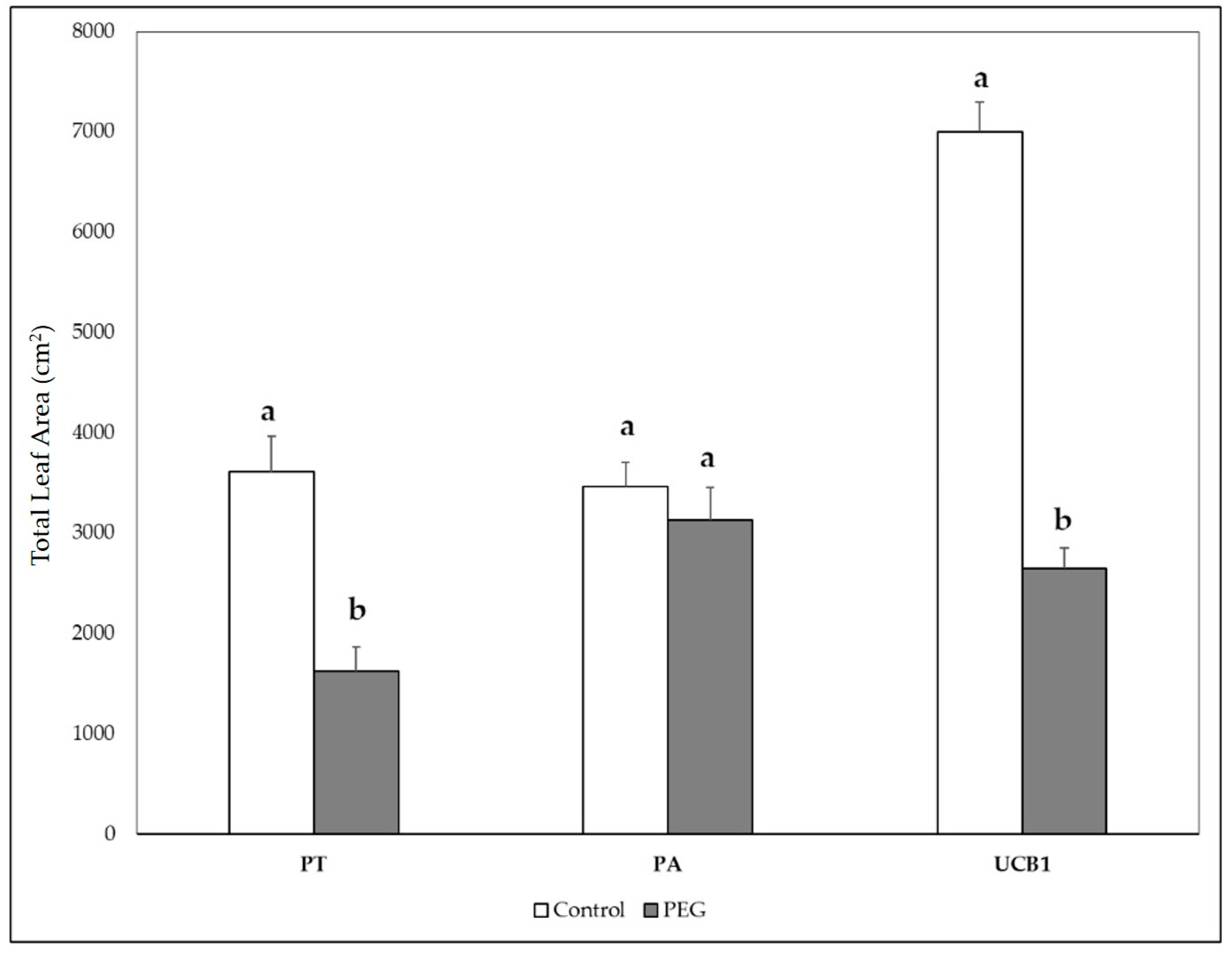
3.5. Biomass Weight and Plant Growth
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Behboudian, M.H.; Walker, R.R.; Törökfalvy, E. Effects of water stress and salinity on photosynthesis on pistachio. Sci. Hortic. 1986, 29, 251–261. [Google Scholar] [CrossRef]
- Galindo, A.; Collado-González, J.; Griñán, I.; Corell, M.; Centeno, A.; Martín-Palomo, M.J.; Girón, P.; Rodríguez, Z.N.; Cruz, H.; Memmi, A.A.; et al. Deficit irrigation and emerging fruit crops as a strategy to save water in Mediterranean semiarid agrosystems. Agric. Water Manag. 2018, 202, 311–324. [Google Scholar] [CrossRef]
- Spiegel-Roy, P.; Mazigh, D.; Evenari, M. Response of Pistachio to Low Soil Moisture Conditions. J. Am. Soc. Hortic. Sci. 1977, 102, 470–473. [Google Scholar] [CrossRef]
- Lin, T.S.; Crane, J.C.; Ryugo, K.; Polito, V.S.; DeJong, T.M. Comparative study of leaf morphology, photosynthesis, and leaf conductance in selected Pistacia species. J. Am. Soc. Hortic. Sci. 1984, 109, 325–330. [Google Scholar] [CrossRef]
- Germana, C. The response of pistachio trees to water stress as affected by two different rootstocks. In Proceedings of the II International Symposium on Irrigation of Horticultural Crops, Chania (Crete), Greece, 9–13 September 1996. [Google Scholar]
- Kanber, R.; Yazar, A.; Önder, S.; Köksal, H. Irrigation response of pistachio (Pistacia vera L.). Irrig. Sci. 1993, 14, 7–14. [Google Scholar] [CrossRef]
- Goldhamer, D.A. Tree water requirements and regulated deficit irrigation. In Pistachio Production Manual, 4th ed.; Ferguson, L., Ed.; University of California: Davis, CA, USA, 2005; pp. 103–116. [Google Scholar]
- Iniesta, F.; Testi, L.; Goldhamer, D.A.; Fereres, E. Quantifying reductions in consumptive water use under regulated deficit irrigation in pistachio (Pistacia vera L.). Agric. Water Manag. 2008, 95, 877–886. [Google Scholar] [CrossRef]
- Marino, G.; Di Martino, S.; Roxas, A.A.; Caruso, T.; Ferguson, L.; Barone, E.; Marra, F.P. Sustainability of pistachio production (Pistacia vera L.) under supplemental irrigation in a Mediterranean climate. Sci. Hortic. 2018, 241, 260–266. [Google Scholar] [CrossRef]
- Goldhamer, D.A.; Beede, R.; Moore, J.M.; Weinberger, G.; Menezes, J., Jr. Water use requirements and physiological response to water stress in pistachio. In Annual Report of the California Pistachio Commission, Crop Year 1982–1983; University of California: Davis, CA, USA, 1983; pp. 53–57. [Google Scholar]
- Goldhamer, D.A.; Kjelgren, R.; Beede, R.; Williams, L.; Moore, M.J.; Lane, J.; Weinberger, G.; Menezes, J. Water use requirements of pistachio trees and response to water stress. In Annual Report of the California Pistachio Commission, Crop Year 1984–1985; University of California: Davis, CA, USA, 1985; pp. 85–92. [Google Scholar]
- Goldhamer, D.A. Irrigation management. In Pistachio Production; Ferguson, L., Ed.; Center for Fruit and Nut Research and Information: Davis, CA, USA, 1995; pp. 71–81. [Google Scholar]
- Memmi, H.; Gijón, M.C.; Couceiro, J.F.; Pérez-López, D. Water stress thresholds for regulated deficit irrigation in pistachio trees: Rootstock influence and effects on yield quality. Agric. Water Manag. 2016, 164, 58–72. [Google Scholar] [CrossRef]
- Kermani, M.M.; Salehi, F. Determination of pistachio crop evapotranspiratation (ETc). In Proceedings of the IV International Symposium on Pistachios and Almonds, Teheran, Iran, 22 May 2005. [Google Scholar]
- Testi, L.; Goldhamer, D.A.; Iniesta, F.; Salinas, M. Crop water stress index is a sensitive water stress indicator in pistachio trees. Irrig. Sci. 2008, 26, 395–405. [Google Scholar] [CrossRef]
- Anuario estadistica. Ministerio de Agricultura, Pesca y Alimentación. MAPA. 2022. Available online: https://www.mapa.gob.es/es/estadistica/temas/publicaciones/anuario-de-estadistica/default.aspx (accessed on 23 October 2023).
- Encuesta sobre Superficie y Rendimientos Cultivos. Ministerio de Agricultura, Pesca y Alimentación. 2022. Available online: https://www.mapa.gob.es/es/estadistica/temas/estadisticas-agrarias/agricultura/esyrce/ (accessed on 23 October 2023).
- Moriana, A.; Memmi, H.; Centeno, A.; Martín-Palomo, M.J.; Corell, M.; Torrecillas, A.; Pérez-López, D. Influence of rootstock on pistachio (Pistacia vera L. cv Kerman) water relations. Agric. Water Manag. 2018, 202, 263–270. [Google Scholar] [CrossRef]
- Ranjbarfordoei, A.; Samson, R.; Van Damme, P.; Lemeur, R. Effects of drought stress induced by polyethylene glycol on pigment content and photosynthetic gas exchange of Pistacia khinjuk and P. mutica. Photosynthetica 2000, 38, 443–447. [Google Scholar] [CrossRef]
- Arzani, K.; Ghasemi, M.; Yadollahi, A.; Hokmabadi, H. Study of foliar epidermal anatomy of four pistachio rootstocks under water stress. Idesia 2013, 31, 101–107. [Google Scholar] [CrossRef]
- Esmaeilpour, A.; Van Labeke, M.C.; Samson, R.; Van Damme, P. Osmotic stress affects physiological responses and growth characteristics of three pistachio cultivars. Acta Physiol. Plant. 2015, 37, 123. [Google Scholar] [CrossRef]
- Esmaeilpour, A.; Van Labeke, M.C.; Samson, R.; Boeckx, P.; Van Damme, P. Variation in biochemical characteristics, water status, stomata features, leaf carbon isotope composition and its relationship to water use efficiency in pistachio (Pistacia vera L.) cultivars under drought stress condition. Sci. Hortic. 2016, 211, 158–166. [Google Scholar] [CrossRef]
- Khoyerdi, F.F.; Shamshiri, M.H.; Estaji, A. Changes in some physiological and osmotic parameters of several pistachio genotypes under drought stress. Sci. Hortic. 2016, 198, 44–51. [Google Scholar] [CrossRef]
- Rahneshan, Z.; Nasibi, F.; Moghadam, A.A. Effects of salinity stress on some growth, physiological, biochemical parameters and nutrients in two pistachio (Pistacia vera L.) rootstocks. J. Plant Interact. 2018, 13, 73–82. [Google Scholar] [CrossRef]
- De Palma, L.; Novello, V. Effect of drip irrigation on leaf gas exchanges and stem water potential in pistachio cv. Larnaka. In Proceedings of the II International Symposium on Pistachios and Almonds, Davis, CA, USA, 1 August 1998. [Google Scholar]
- Sedaghati, N.; Hokmabadi, H. Optimizing pistachio irrigation management using the relationship between echo-physiological characteristics and water stress. J. Agric. Sci. Technol. 2015, 17, 189–200. [Google Scholar]
- Jones, H.G. How do rootstocks control shoot water relations? New Phytol. 2012, 194, 301–303. [Google Scholar] [CrossRef]
- Ezzahouani, A.; Williams, L.E. The influence of rootstock on leaf water potential, yield, and berry composition of Ruby Seedless grapevines. Am. J. Enol. Vitic. 1995, 46, 559–563. [Google Scholar] [CrossRef]
- Atkinson, C.J.; Else, M.A.; Taylor, L.; Dover, C.J. Root and stem hydraulic conductivity as determinants of growth potential in grafted trees of apple (Malus pumila Mill.). J. Exp. Bot. 2003, 54, 1221–1229. [Google Scholar] [CrossRef]
- Motisi, A.; Caruso, T.; Grutta, I.; Marra, F.P.; Pernice, F. Canopy architecture appraisal by fractal dimension of ‘Flordastar’ peach trees grafted onto different rootstocks. In Proceedings of the VIII International Symposium on Canopy, Rootstocks and Environmental Physiology in Orchard Systems, Budapest, Hungary, 13–18 June 2004. [Google Scholar]
- Kumar, P.; Rouphael, Y.; Cardarelli, M.; Colla, G. Vegetable grafting as a tool to improve drought resistance and water use efficiency. Front. Plant Sci. 2017, 8, 1130. [Google Scholar] [CrossRef] [PubMed]
- Ghrab, M.; Ben Mimoun, M.; Triki, H.; Gouta, H. Evaluation of the Performance of Seventeen Male Pistachio-Three Specimens. In Proceedings of the III International Symposium on Pistachios and Almonds, Zaragoza, Spain, 20–24 May 2001. [Google Scholar]
- Gijón, M.C.; Gimenez, C.; Perez-López, D.; Guerrero, J.; Couceiro, J.F.; Moriana, A. Rootstock influences the response of pistachio (Pistacia vera L. cv. Kerman) to water stress and rehydration. Sci. Hortic. 2010, 125, 666–671. [Google Scholar] [CrossRef]
- Memmi, H.; Centeno, A.; Moriana, A.; Corell, M.; Torrecillas, A.; Pérez, D. Influence of rootstocks on pistachio (Pistacia vera L.) water relations. In Proceedings of the XIII Simposio Hispano-Portugués de Relaciones Hídricas en las Plantas, Pamplona, Spain, 18–20 October 2016. [Google Scholar]
- Memmi, H.; Couceiro, J.F.; Gijón, M.C.; Pérez-López, D. Impact of wáter stress, environment and rootstock on the diurnal behavior of stem wáter potential and leaf conductance in pistachio (Pistacia vera L.). Span. J. Agric. Res. 2016, 14, 0804. [Google Scholar] [CrossRef]
- Preston, A.P. Apples for amateurs. GC HTJ 1980, 187, 15–17. [Google Scholar]
- Osmolovskaya, N.; Shumilina, J.; Kim, A.; Didio, A.; Grishina, T.; Bilova, T.; Keltsieva, O.A.; Zhukov, V.; Tikhonovich, I.; Tarakhovskaya, E.; et al. Methodology of drought stress research: Experimental setup and physiological characterization. Int. J. Mol. Sci. 2018, 19, 4089. [Google Scholar] [CrossRef] [PubMed]
- Bressan, R.A.; Handa, A.K.; Handa, S.; Hasegawa, P.M. Growth and water relations of cultured tomato cells after adjustment to low external water potentials. Plant Physiol. 1982, 70, 1303–1309. [Google Scholar] [CrossRef] [PubMed]
- Hohl, M.; Schöpfer, P. Growth at reduced turgor: Irreversible and reversible cell-wall extension of maize coleoptiles and its implications for the theory of cell growth. Planta 1982, 187, 209–217. [Google Scholar] [CrossRef]
- Lu, Z.; Neumann, P.M. Water-stressed maize, barley and rice seedlings show species diversity in mechanisms of leaf growth inhibition. J. Exp. Bot. 1998, 49, 1945–1952. [Google Scholar] [CrossRef]
- Hoagland, D.R.; Arnon, D.I. The water-culture method for growing plants without soil. Circ. Calif. Agric. Exp. Stn. 1950, 347, 32. [Google Scholar]
- Michel, B.E.; Kaufmann, M.R. The osmotic potential of polyethylene glycol 6000. Plant Physiol. 1973, 51, 914–916. [Google Scholar] [CrossRef]
- Kramer, P.J.; Boyer, J.S. Water Relations of Plants and Soils, 1st ed.; Academic press: San Diego, CA, USA, 1995; pp. 167–199. [Google Scholar]
- Begg, J.E.; Turner, N.C. Water potential gradients in field tobacco. Plant Physiol. 1970, 46, 343–346. [Google Scholar] [CrossRef] [PubMed]
- Tuberosa, R. Phenotyping for drought tolerance of crops in the genomics era. Front. Physiol. 2012, 3, 347. [Google Scholar] [CrossRef] [PubMed]
- Boopathi, N.M.; Swapnashri, G.; Kavitha, P.; Sathish, S.; Nithya, R.; Ratnam, W.; Kumar, A. Introgression, evaluation and bulked segregant analysis of major yield QTL under water stress into indigenous elite line for low water. Rice Sci. 2012, 19, 1–6. [Google Scholar]
- Guerrero, J.; Couceiro, J.F.; Moriana, A. Selection of terebinth (Pistacia terebinthus L.) trees as seed producers for pistachio (Pistacia vera L.) rootstocks in the Castilla La Mancha (Spain). FAO-Nucis Newsl. 2002, 11, 25–29. [Google Scholar]
- Arpaci, S.; Acar, S.; Karadag, H.S.; Atli, H.S.; Ak, B.E.; Sarpkaya, K. Comparison of growing of pistachio rooststocks on different planting intervals. In ISHS Acta Horticulturae 1028; VI International Symposium on Almonds and Pistachios; International Society For Horticultural Science: Korbeek-Lo, Belgium, 2013. [Google Scholar]
- Taiz, L.; Zeiger, E. Photosynthesis. In Plant Physiology, 5th ed.; Sinauer Associates Inc.: Sunderland, MA, USA, 2010. [Google Scholar]
- Iacono, F.; Bucella, A.; Peterlunger, E. Water stress and rootstock influence on leaf gas exchange of grafted and ungrafted grapevines. Sci. Hortic. 1998, 75, 27–39. [Google Scholar] [CrossRef]
- Nezami, S.R.; Yadollahi, A.; Hokmabadi, H.; Tajabadipour, A. Changes in Drought Tolerance Mechanism at Different Times of Stress and Re-hydration in Hybrid Pistachio Rootstock. J. Nuts 2021, 12, 333–360. [Google Scholar]
- Blum, A. Effective use of water (EUW) and not water-use efficiency (WUE) is the target of crop yield improvement under drought stress. Field Crops Res. 2009, 112, 119–123. [Google Scholar] [CrossRef]
- Parida, A.K.; Das, A.B.; Sanada, Y.; Mohanty, P. Effects of salinity on biochemical components of the mangrove, Aegiceras corniculatum. Aquat. Bot. 2004, 80, 77–87. [Google Scholar] [CrossRef]
- Grace, S.C.; Logan, B.A. Energy dissipation and radical scavenging by the plant phenylpropanoid pathway. Trans. R. Soc. Lond. Ser. B Biol. Sci. 2000, 355, 1499–1510. [Google Scholar] [CrossRef]
- Sánchez-Rodríguez, E.; Moreno, D.A.; Ferreres, F.; del Mar Rubio-Wilhelmi, M.; Ruiz, J.M. Differential responses of five cherry tomato varieties to water stress: Changes on phenolic metabolites and related enzymes. Phytochemistry 2011, 72, 723–729. [Google Scholar] [CrossRef]
- Bürling, K.; Cerovic, Z.G.; Cornic, G.; Ducruet, J.M.; Noga, G.; Hunsche, M. Fluorescence-based sensing of drought-induced stress in the vegetative phase of four contrasting wheat genotypes. Environ. Exp. Bot. 2013, 89, 51–59. [Google Scholar] [CrossRef]
- Chalker-Scott, L. Environmental significance of anthocyanins in plant stress responses. Photochem. Photobiol. 1999, 70, 1–9. [Google Scholar] [CrossRef]
- Álvarez, S.; Rodríguez, P.; Broetto, F.; Sánchez-Blanco, M.J. Long term responses and adaptive strategies of Pistacia lentiscus under moderate and severe deficit irrigation and salinity: Osmotic and elastic adjustment, growth, ion uptake and photosynthetic activity. Agric. Water Manag. 2018, 202, 253–262. [Google Scholar] [CrossRef]
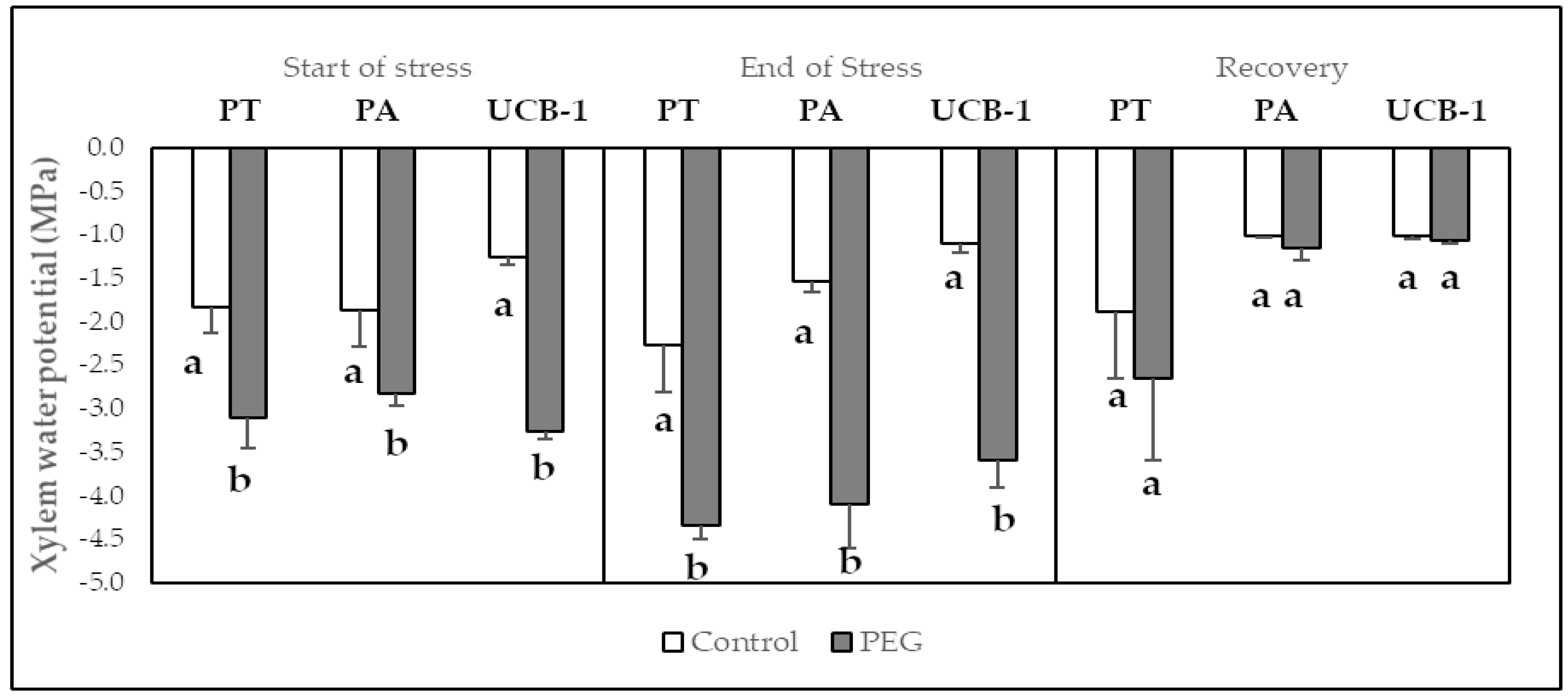
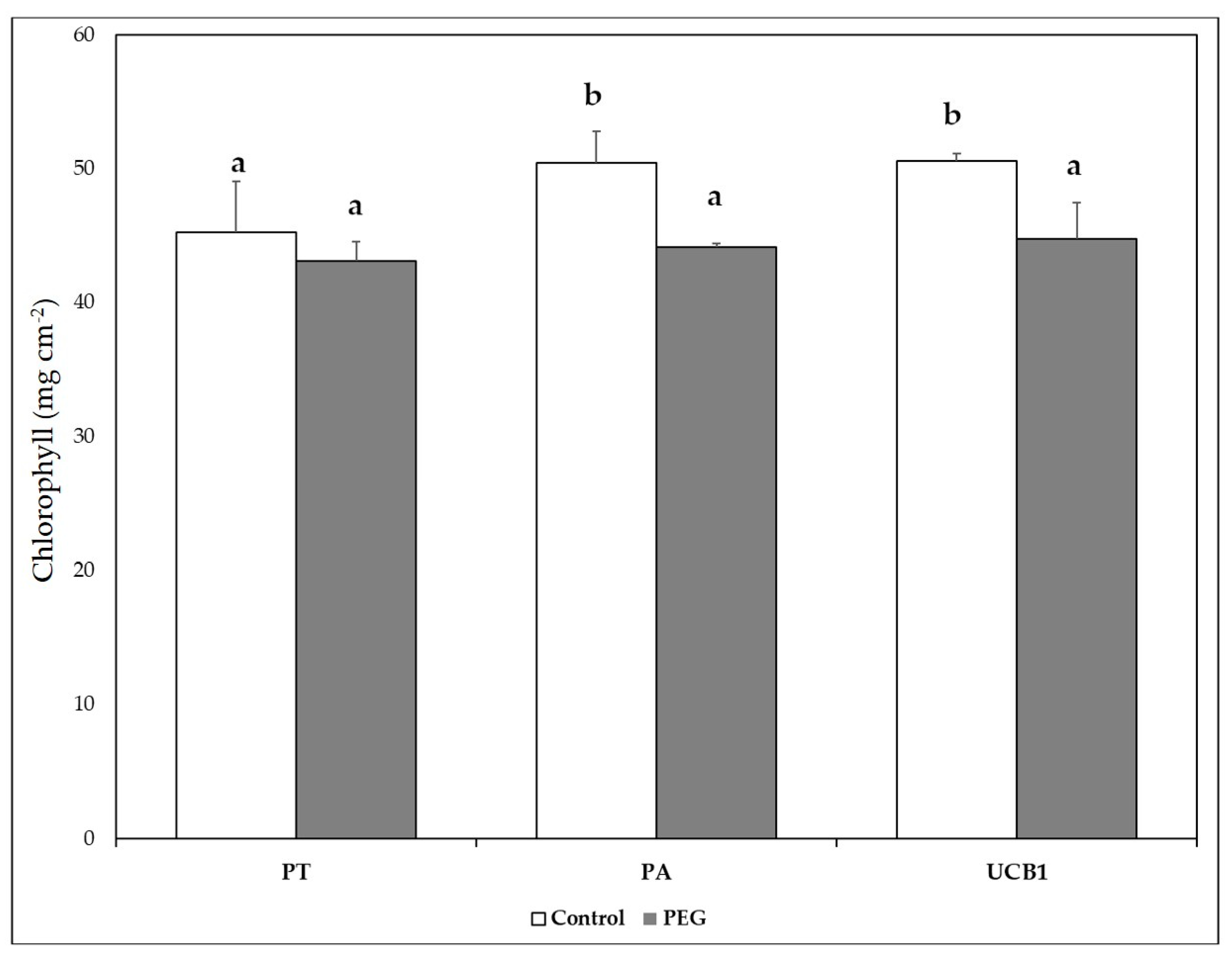
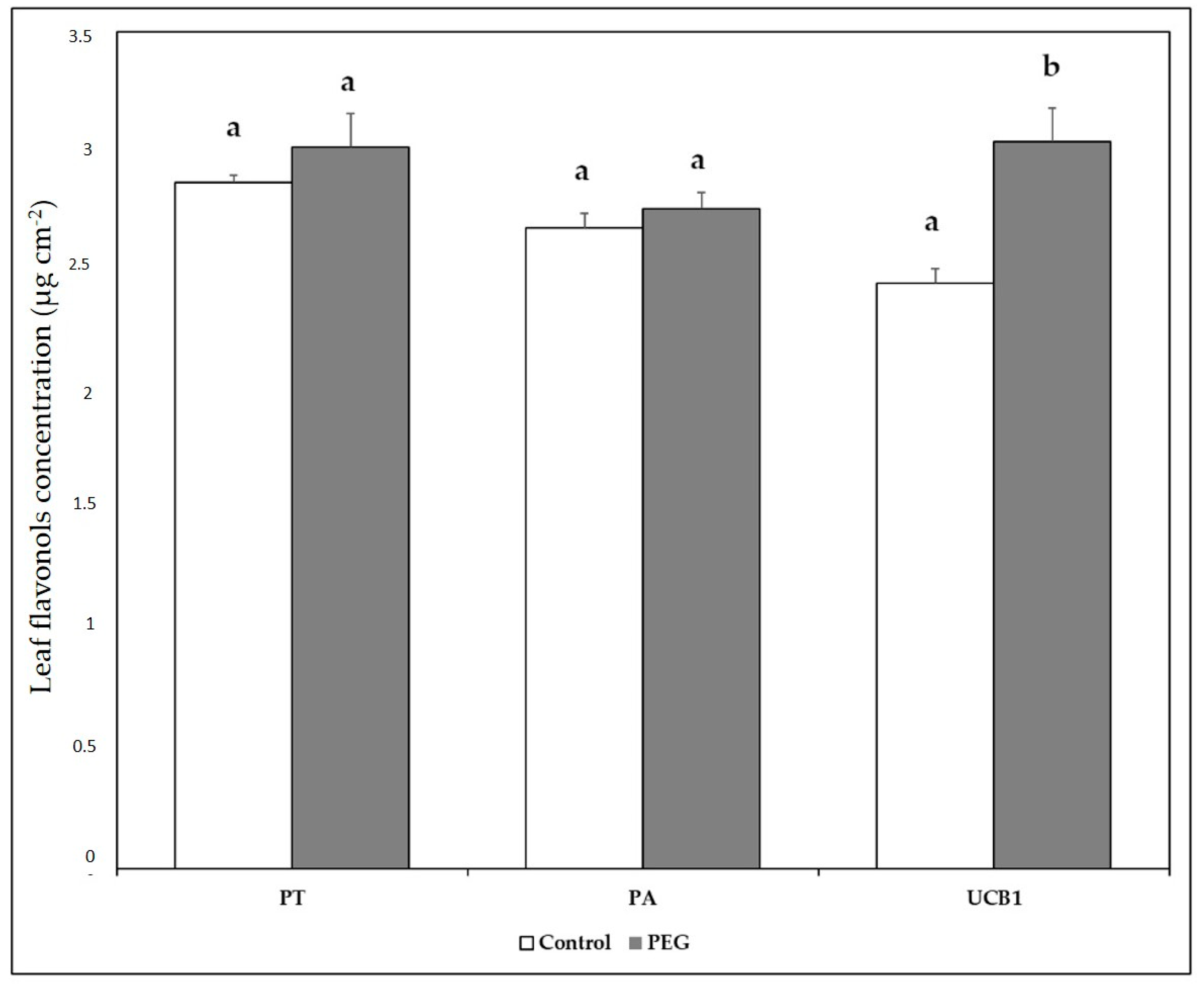
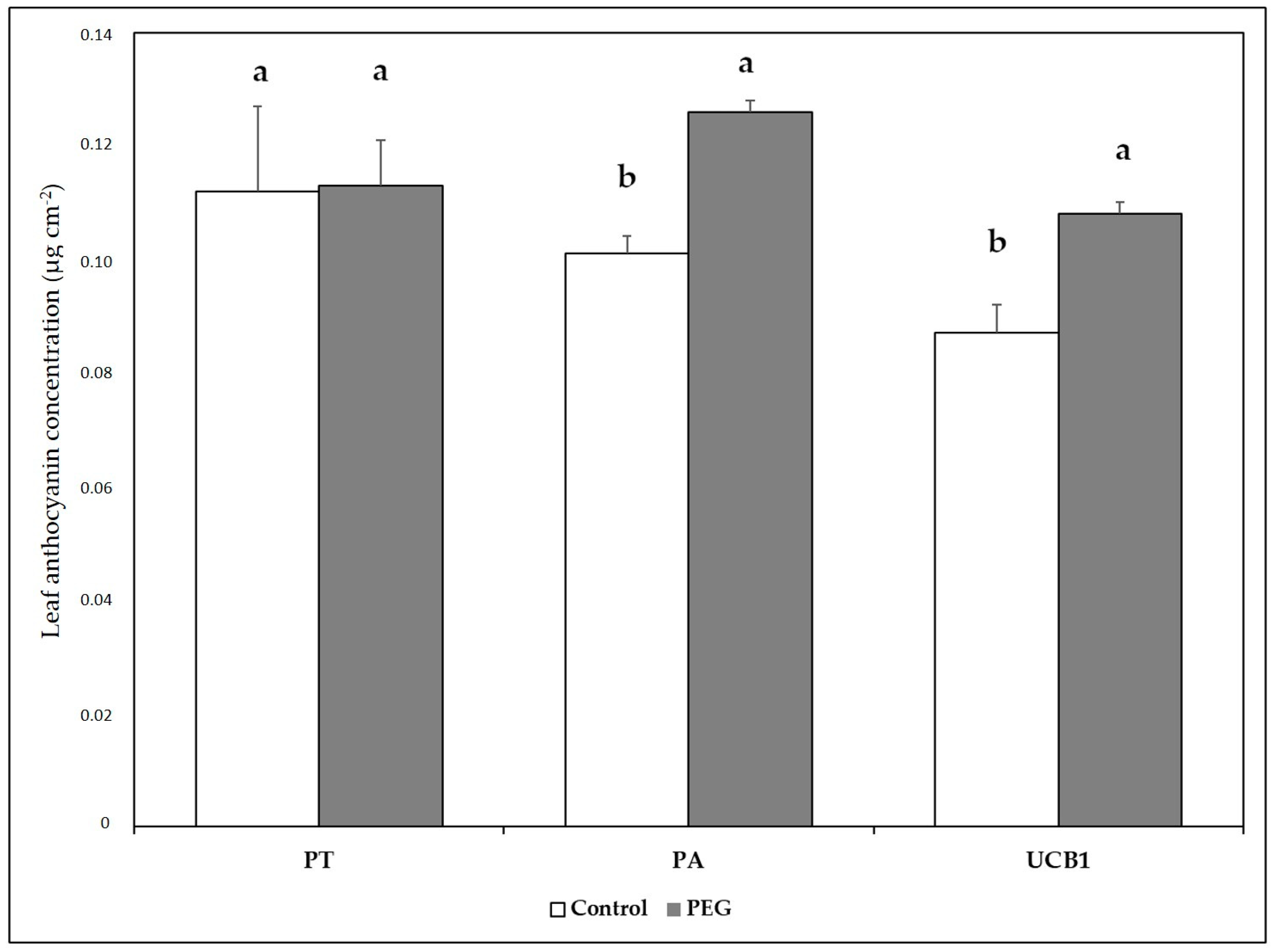
| Start of Stress | End of Stress | Recovery | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Rootstock | Trait | Ci | E | gs | A | Ci | E | Gs | A | Ci | E | gs | A |
| P. terebinthus | Control | 240 ± 5 | 5.4 ± 0.8 | 0.18 ± 0.04 | 12.9 ± 2.0 | 231 ± 18 | 3.6 ± 0.5 | 0.2 ± 0.04 | 14.2 ± 1.4 | 266 ± 24 | 3.0 ± 0.1 | 0.19 ± 0.01 | 14.6 ± 1.5 |
| PEG | 184 ± 15 | 4.5 ± 0.5 | 0.12 ± 0.02 | 13.7 ± 1.0 | 223 ± 17 | 1.7 ± 0.0 | 0.06 ± 0.01 | 5.9 ± 0.8 | 201 ± 28 | 2.4 ± 0.7 | 0.12 ± 0.05 | 11.5 ± 2.8 | |
| P. atlantica | Control | 219 ± 4 | 6.7 ± 0.4 | 0.18 ± 0.02 | 14.9 ± 1.5 | 228 ± 8 | 4.2 ± 0.1 | 0.21 ± 0.01 | 15.7 ± 0.9 | 256 ± 7 | 3.6 ± 0.2 | 0.17 ± 0.02 | 12.8 ± 0.7 |
| PEG | 200 ± 11 | 5.3 ± 0.7 | 0.12 ± 0.02 | 11.7 ± 1.1 | 164 ± 2 | 1.8 ± 0.0 | 0.06 ± 0.01 | 7.3 ± 0.3 | 248 ± 3 | 2.6 ± 0.5 | 0.10 ± 0.05 | 8.5 ± 2.1 | |
| UCB-1 | Control | 219 ± 7 | 7.9 ± 0.1 | 0.22 ± 0.01 | 17.4 ± 0.9 | 217 ± 7 | 5.1 ± 0.1 | 0.23 ± 0.01 | 17.6 ± 0.8 | 231 ± 12 | 4.3 ± 0.2 | 0.20 ± 0.01 | 16.0 ± 1.1 |
| PEG | 233 ± 24 | 7.1 ± 0.2 | 0.18 ± 0.01 | 13.5 ± 1.8 | 198 ± 7 | 2.6 ± 0.2 | 0.08 ± 0.01 | 8.4 ± 0.5 | 207 ± 6 | 4.2 ± 0.2 | 0.14 ± 0.02 | 15.2 ± 0.1 | |
| Rootstock effect | P. terebinthus | 226 ± 14 | 5.2 ± 0.6 | 0.17 ± 0.03 | 13.1 ± 1.4 | 227 ± 11 | 2.6 ± 0.5 | 0.13 ± 0.04 | 10.1 ± 2.0 | 233 ± 22 | 2.7 ± 0.4 | 0.16 ± 0.03 | 13.0 ± 1.4 |
| P. atlantica | 209 ± 7 | 6.0 ± 0.5 | 0.15 ± 0.02 | 13.3 ± 1.1 | 196 ± 15 | 3.0 ± 0.5 | 0.13 ± 0.04 | 11.5 ± 1.9 | 252 ± 4 | 3.1 ± 0.3 | 0.14 ± 0.03 | 10.7 ± 1.4 | |
| UCB-1 | 226 ± 12 | 7.5 ± 0.2 | 0.20 ± 0.01 | 15.4 ± 1.2 | 207 ± 6 | 3.8 ± 0.6 | 0.15 ± 0.04 | 13.0 ± 2.1 | 219 ± 8 | 4.2 ± 0.1 | 0.17 ± 0.02 | 15.6 ± 0.5 | |
| Trait effect | Control | 226 ± 4 | 6.7 ± 0.4 | 0.19 ± 0.02 | 15.1 ± 1.0 | 225 ± 6 | 4.3 ± 0.3 | 0.21 ± 0.01 | 15.8 ± 0.7 | 252 ± 10 | 3.6 ± 0.2 | 0.19 ± 0.01 | 14.4 ± 0.7 |
| PEG | 212 ± 13 | 5.9 ± 0.5 | 0.15 ± 0.01 | 12.8 ± 0.9 | 195 ± 10 | 2.0 ± 0.2 | 0.06 ± 0.01 | 7.2 ± 0.5 | 218 ± 11 | 3.1 ± 0.4 | 0.12 ± 0.02 | 11.7 ± 1.4 | |
| ANOVA | Rootstock | Ns 1 | 0.007 | 0.04 | ns | 0.05 | 0.001 | Ns | 0.02 | ns | 0.007 | ns | 0.035 |
| Treatment | 0.05 | 0.02 | 0.03 | ns | 0.007 | 0.001 | 0.001 | 0.001 | 0.03 | ns | 0.02 | ns | |
| P × T | 0.03 | ns | ns | ns | ns | Ns | Ns | ns | ns | ns | ns | ns | |
| Start of Stress | End of Stress | Recovery | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Rootstock | Trait | A/Ci | A/gs | WUE | Fv/Fm | A/Ci | A/gs | WUE | Fv/Fm | A/Ci | A/gs | WUE | Fv/Fm |
| P. terebinthus | Control | 0.05 ± 0.008 | 73.1 ± 6.9 | 2.4 ± 0.1 | 0.81 ± 0.01 | 0.06 ± 0.006 | 76.9 ± 14.3 | 4.0 ± 0.4 | 0.83 ± 0.001 | 0.06 ± 0.011 | 75.8 ± 10.0 | 4.8 ± 0.6 | 0.79 ± 0.02 |
| PEG | 0.07 ± 0.001 | 113.7 ± 9.1 | 3.0 ± 0.2 | 0.79 ± 0.01 | 0.03 ± 0.006 | 106.7 ± 8.3 | 3.5 ± 0.4 | 0.82 ± 0.002 | 0.06 ± 0.007 | 109.0 ± 28.7 | 5.0 ± 0.4 | 0.80 ± 0.01 | |
| P. atlantica | Control | 0.07 ± 0.008 | 84.8 ± 0.4 | 2.2 ± 0.1 | 0.80 ± 0.02 | 0.07 ± 0.007 | 74.2 ± 4.1 | 3.8 ± 0.2 | 0.83 ± 0.004 | 0.05 ± 0.003 | 74.4 ± 2.8 | 3.6 ± 0.0 | 0.81 ± 0.01 |
| PEG | 0.06 ± 0.003 | 101.0 ± 9.8 | 2.2 ± 0.1 | 0.80 ± 0.02 | 0.04 ± 0.002 | 132.4 ± 1.2 | 4.1 ± 0.1 | 0.81 ± 0.004 | 0.03 ± 0.009 | 89.3 ± 5.2 | 3.3 ± 0.1 | 0.78 ± 0.02 | |
| UCB-1 | Control | 0.08 ± 0.007 | 77.5 ± 2.4 | 2.2 ± 0.1 | 0.82 ± 0.01 | 0.08 ± 0.006 | 77.2 ± 1.6 | 3.5 ± 0.1 | 0.84 ± 0.002 | 0.07 ± 0.009 | 80.6 ± 1.8 | 3.7 ± 0.1 | 0.79 ± 0.02 |
| PEG | 0.06 ± 0.013 | 75.9 ± 11.0 | 1.9 ± 0.3 | 0.81 ± 0.01 | 0.04 ± 0.001 | 112.9 ± 6.1 | 3.2 ± 0.1 | 0.82 ± 0.004 | 0.07 ± 0.002 | 93.0 ± 4.6 | 3.7 ± 0.1 | 0.78 ± 0.01 | |
| Rootstock effect | P. terebinthus | 0.06 ± 0.007 | 83.3 ± 11.3 | 2.6 ± 0.2 | 0.81 ± 0.01 | 0.04 ± 0.009 | 91.8 ± 9.9 | 3.8 ± 0.3 | 0.82 ± 0.002 | 0.06 ± 0.006 | 92.4 ± 15.5 | 4.9 ± 0.3 | 0.80 ± 0.01 |
| P. atlantica | 0.06 ± 0.004 | 92.9 ± 5.7 | 2.2 ± 0.1 | 0.80 ± 0.01 | 0.06 ± 0.006 | 103.3 ± 13.1 | 4.0 ± 0.1 | 0.82 ± 0.005 | 0.04 ± 0.005 | 81.9 ± 4.2 | 3.4 ± 0.1 | 0.80 ± 0.01 | |
| UCB-1 | 0.07 ± 0.008 | 76.7 ± 5.0 | 2.1 ± 0.2 | 0.82 ± 0.01 | 0.06 ± 0.009 | 95.0 ± 8.5 | 3.4 ± 0.1 | 0.83 ± 0.004 | 0.07 ± 0.004 | 86.8 ± 3.5 | 3.7 ± 0.1 | 0.79 ± 0.01 | |
| Trait effect | Control | 0.07 ± 0.005 | 78.4 ± 2.7 | 2.3 ± 0.1 | 0.81 ± 0.01 | 0.07 ± 0.004 | 76.1 ± 4.3 | 3.8 ± 0.2 | 0.83 ± 0.002 | 0.06 ± 0.005 | 77.0 ± 3.2 | 4.0 ± 0.3 | 0.80 ± 0.01 |
| PEG | 0.06 ± 0.006 | 92.1 ± 8.2 | 2.2 ± 0.2 | 0.80 ± 0.01 | 0.04 ± 0.003 | 117.3 ± 4.9 | 3.6 ± 0.2 | 0.82 ± 0.002 | 0.05 ± 0.006 | 97.1 ± 9.1 | 4.0 ± 0.3 | 0.79 ± 0.01 | |
| ANOVA | Rootstock | Ns 1 | 0.05 | 0.02 | ns | 0.02 | ns | Ns | ns | 0.007 | ns | 0.001 | ns |
| Treatment | ns | 0.01 | Ns | ns | 0.001 | 0.001 | Ns | 0.001 | ns | ns | ns | ns | |
| P × T | ns | 0.03 | 0.02 | ns | ns | ns | Ns | ns | ns | ns | ns | ns | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernández-Suela, E.; Garcia-Estringana, P.; de Andrés, E.F.; Ramírez-Martín, N.; Alegre, J. Effects of Rootstock on Water Stress, Physiological Parameters, and Growth of the Pistachio Tree. Horticulturae 2023, 9, 1305. https://doi.org/10.3390/horticulturae9121305
Fernández-Suela E, Garcia-Estringana P, de Andrés EF, Ramírez-Martín N, Alegre J. Effects of Rootstock on Water Stress, Physiological Parameters, and Growth of the Pistachio Tree. Horticulturae. 2023; 9(12):1305. https://doi.org/10.3390/horticulturae9121305
Chicago/Turabian StyleFernández-Suela, Eduardo, Pablo Garcia-Estringana, E. Francisco de Andrés, Noelia Ramírez-Martín, and Jesus Alegre. 2023. "Effects of Rootstock on Water Stress, Physiological Parameters, and Growth of the Pistachio Tree" Horticulturae 9, no. 12: 1305. https://doi.org/10.3390/horticulturae9121305
APA StyleFernández-Suela, E., Garcia-Estringana, P., de Andrés, E. F., Ramírez-Martín, N., & Alegre, J. (2023). Effects of Rootstock on Water Stress, Physiological Parameters, and Growth of the Pistachio Tree. Horticulturae, 9(12), 1305. https://doi.org/10.3390/horticulturae9121305





