Effect of Chitosan-Poly(Acrylic Acid) Complexes and Two Nutrient Solutions on the Growth and Yield of Two Habanero Pepper Cultivars
Abstract
1. Introduction
2. Materials and Methods
2.1. Establishment of the Experiment
2.2. Synthesis of the Non-Stoichiometric Interpolyelectrolyte CS–PAA Complexes and Foliar Applications
2.3. Nutrient Solutions
2.4. Growth, Biomass and Yield Parameters
2.5. Parameters Measured in Green and Ripe Fruits
2.6. Statistical Analysis
3. Results
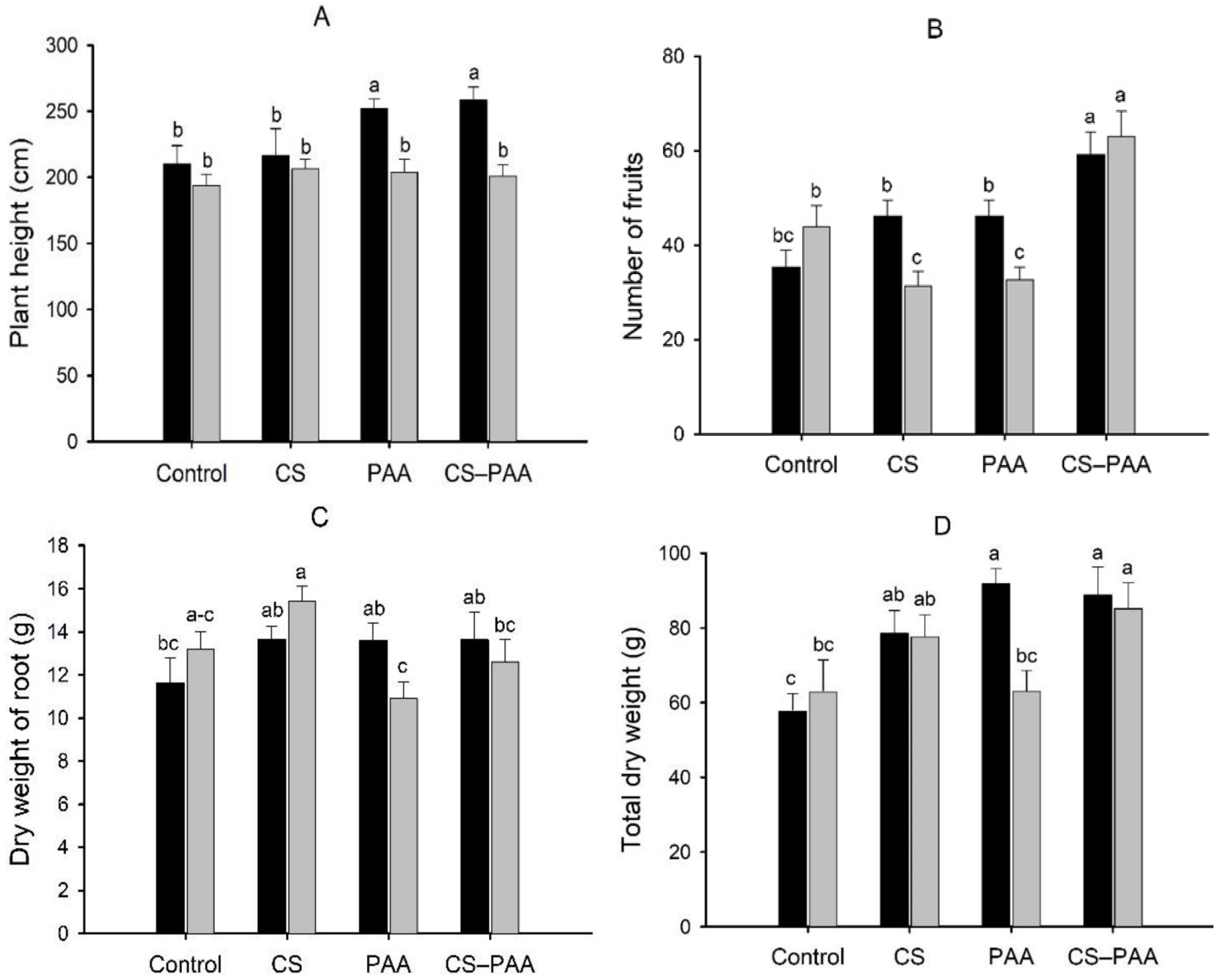
3.1. Effect of Foliar Applications
3.2. Effect of Nutrient Solutions
3.3. Effect of Cultivars
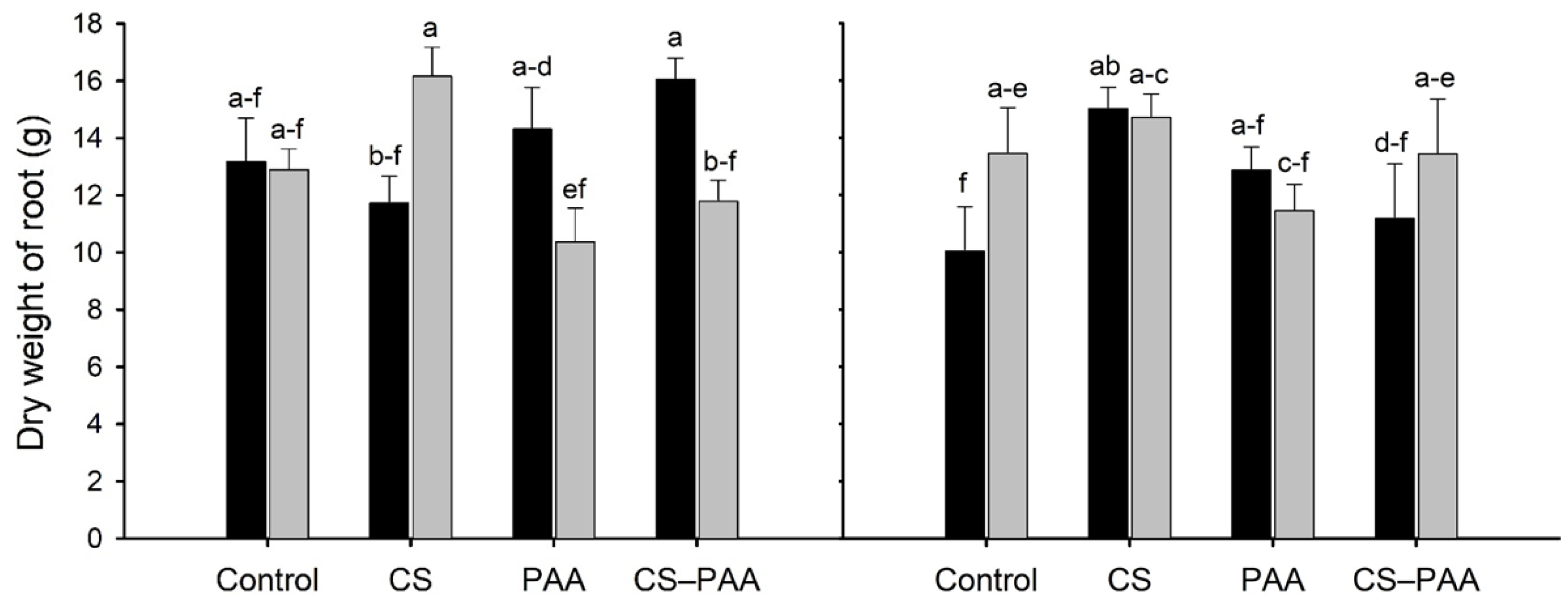
3.4. Effect of the Interactions on the Growth, Biomass, and Yield of the Habanero Pepper
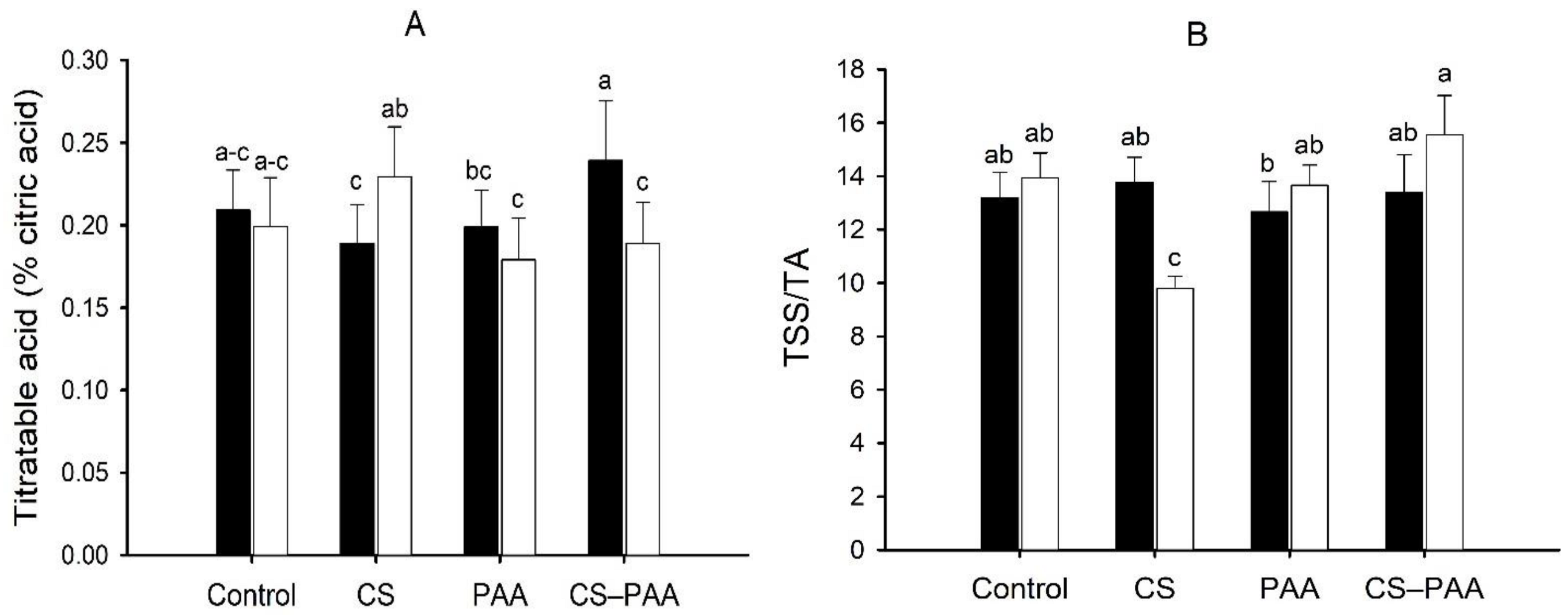
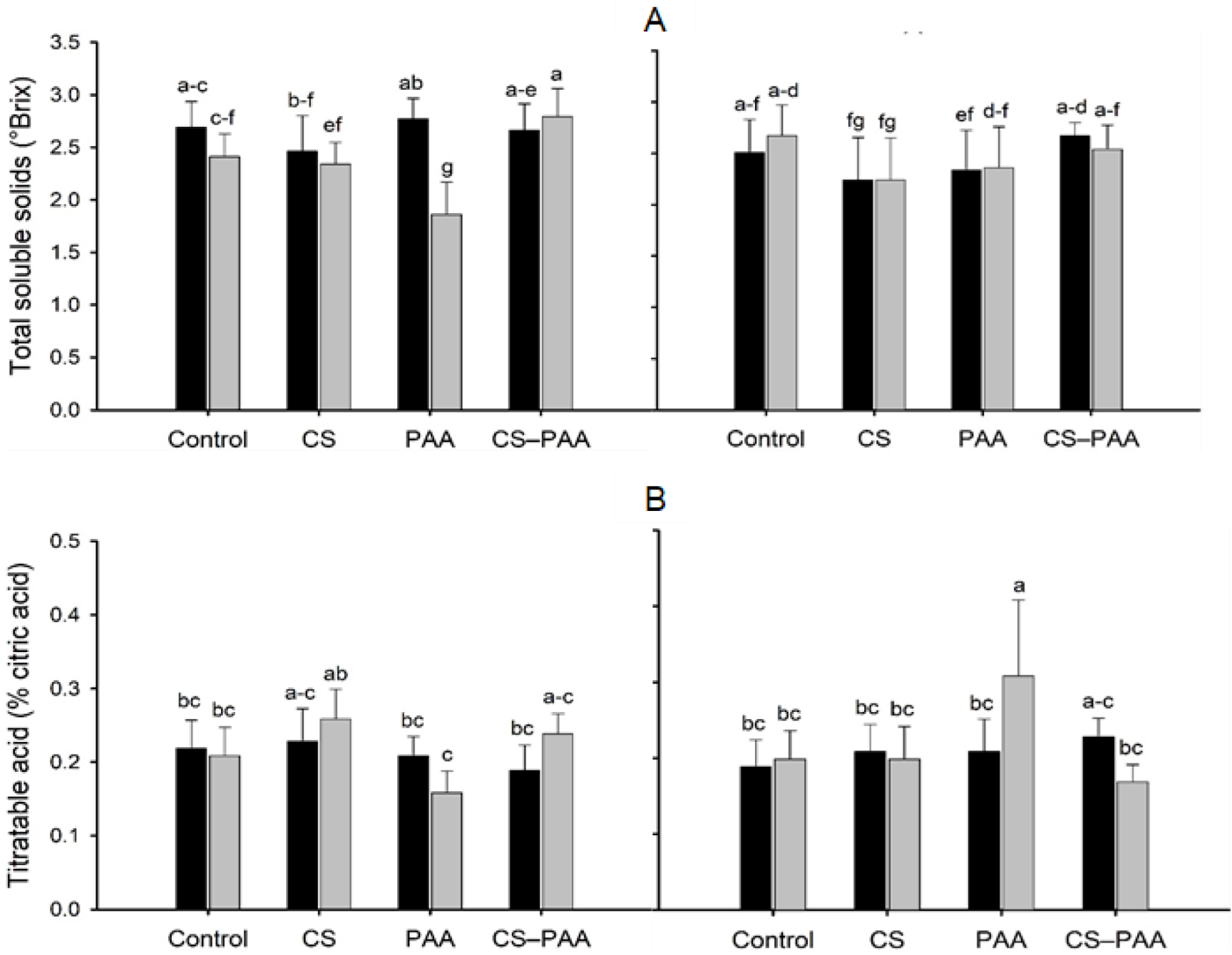
3.5. Effect of the Interactions on the Green and Ripe Fruits of the Habanero Peppers
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kaya, M.; Baran, T.; Erdoğan, S.; Menteş, A.; Özüsağlam, M.A.; Çakmak, Y.S. Physicochemical Comparison of Chitin and Chitosan Obtained from Larvae and Adult Colorado Potato Beetle (Leptinotarsa decemlineata). Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 45, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Kaya, M.; Cakmak, Y.S.; Baran, T.; Asan-Ozusaglam, M.; Mentes, A.; Tozak, K.O. New Chitin, Chitosan, and O-Carboxymethyl Chitosan Sources from Resting Eggs of Daphnia Longispina (Crustacea); with Physicochemical Characterization, and Antimicrobial and Antioxidant Activities. Biotechnol. Bioprocess Eng. 2014, 19, 58–69. [Google Scholar] [CrossRef]
- Malerba, M.; Cerana, R. Chitosan Effects on Plant Systems. Int. J. Mol. Sci. 2016, 17, 996. [Google Scholar] [CrossRef] [PubMed]
- Kashyap, P.L.; Xiang, X.; Heiden, P. Chitosan Nanoparticle Based Delivery Systems for Sustainable Agriculture. Int. J. Biol. Macromol. 2015, 77, 36–51. [Google Scholar] [CrossRef] [PubMed]
- Mukhtar Ahmed, K.B.; Khan, M.M.A.; Siddiqui, H.; Jahan, A. Chitosan and Its Oligosaccharides, a Promising Option for Sustainable Crop Production—A Review. Carbohydr. Polym. 2020, 227, 115331. [Google Scholar] [CrossRef] [PubMed]
- Mujtaba, M.; Khawar, K.M.; Camara, M.C.; Carvalho, L.B.; Fraceto, L.F.; Morsi, R.E.; Elsabee, M.Z.; Kaya, M.; Labidi, J.; Ullah, H.; et al. Chitosan-Based Delivery Systems for Plants: A Brief Overview of Recent Advances and Future Directions. Int. J. Biol. Macromol. 2020, 154, 683–697. [Google Scholar] [CrossRef]
- Lopez-Moya, F.; Suarez-Fernandez, M.; Lopez-Llorca, L. Molecular Mechanisms of Chitosan Interactions with Fungi and Plants. Int. J. Mol. Sci. 2019, 20, 332. [Google Scholar] [CrossRef]
- Hidangmayum, A.; Dwivedi, P.; Katiyar, D.; Hemantaranjan, A. Application of Chitosan on Plant Responses with Special Reference to Abiotic Stress. Physiol. Mol. Biol. Plants 2019, 25, 313–326. [Google Scholar] [CrossRef]
- Sharif, R.; Mujtaba, M.; Ur Rahman, M.; Shalmani, A.; Ahmad, H.; Anwar, T.; Tianchan, D.; Wang, X. The Multifunctional Role of Chitosan in Horticultural Crops; A Review. Molecules 2018, 23, 872. [Google Scholar] [CrossRef]
- Malerba, M.; Cerana, R. Recent Advances of Chitosan Applications in Plants. Polymers 2018, 10, 118. [Google Scholar] [CrossRef]
- Ortega-Ortíz, H.; Benavides-Mendoza, A.; Flores-Olivas, A.; Ledezma-Pérez, A. Use of the Interpolyelectrolyte Complexes of Poly(Acrylic Acid)-Chitosan as Inductors of Tolerance Against Pathogenic Fungi in Tomato (Lycopersicon esculentum Mill. Var. Floradade). Macromol. Biosci. 2003, 3, 566–570. [Google Scholar] [CrossRef]
- Bai, M.; Wilske, B.; Buegger, F.; Esperschütz, J.; Bach, M.; Frede, H.-G.; Breuer, L. Relevance of Nonfunctional Linear Polyacrylic Acid for the Biodegradation of Superabsorbent Polymer in Soils. Environ. Sci. Pollut. Res. 2015, 22, 5444–5452. [Google Scholar] [CrossRef] [PubMed]
- Zhao, T.; Zhang, K.; Chen, J.; Shi, X.; Li, X.; Ma, Y.; Fang, G.; Xu, S. Changes in Heavy Metal Mobility and Availability in Contaminated Wet-Land Soil Remediated Using Lignin-Based Poly(Acrylic Acid). J. Hazard. Mater. 2019, 368, 459–467. [Google Scholar] [CrossRef] [PubMed]
- Cabrera-De la Fuente, M.; Ortega-Ortiz, H.; Juárez-Maldonado, A.; Sandoval-Rangel, A.; González-Morales, S.; Cadenas-Pliego, G.; Benavides-Mendoza, A. Use of Chitosan-Polyacrylic Acid (CS–PAA) Complex, Chitosan-Polyvinyl Alcohol (CS-PVA) and Chitosan Hydrogels in Greenhouses as a Carrier for Beneficial Elements, Nanoparticles, and Microorganisms. Acta Hortic. 2020, 1296, 1153–1160. [Google Scholar] [CrossRef]
- Benavides-Mendoza, A.; Burgos-Limón, D.; Ortega-Ortiz, H.; Ramírez, H. Benzoic Acid and Poly(Acrylic Acid)-Chitosan in Tomato Quality and Yield in Calcareous Soil. Terra Latinoam. 2007, 25, 261–268. [Google Scholar]
- Abd El-Aziz, M.E.; Morsi, S.M.M.; Salama, D.M.; Abdel-Aziz, M.S.; Abd Elwahed, M.S.; Shaaban, E.A.; Youssef, A.M. Preparation and Characterization of Chitosan/Polyacrylic Acid/Copper Nanocomposites and Their Impact on Onion Production. Int. J. Biol. Macromol. 2019, 123, 856–865. [Google Scholar] [CrossRef]
- Leija-Martínez, P.; Benavides-Mendoza, A.; Cabrera-De La Fuente, M.; Robledo-Olivo, A.; Ortega-Ortíz, H.; Sandoval-Rangel, A.; González-Morales, S. Lettuce Biofortification with Selenium in Chitosan-Polyacrylic Acid Complexes. Agronomy 2018, 8, 275. [Google Scholar] [CrossRef]
- Calvo, P.; Nelson, L.; Kloepper, J.W. Agricultural Uses of Plant Biostimulants. Plant Soil 2014, 383, 3–41. [Google Scholar] [CrossRef]
- Food and Agriculture Information Service (SIAP). Statistical Yearbook of Agricultural Production. 2020. Available online: https://nube.siap.gob.mx/cierreagricola/ (accessed on 21 February 2021).
- López-Gómez, J.D.; Sotelo-Nava, H.; Villegas-Torres, O.G.; Andrade-Rodríguez, M. Yield and quality of habanero chili in response to driving pruning and nutritional regime. Rev. Mex. Cienc. Agríc. 2020, 11, 315–325. [Google Scholar] [CrossRef][Green Version]
- Tucuch-Haas, C.J.; Alcántar-González, G.; Santizo-Rincón, J.A.; Ordaz-Chaparro, V.M.; Larqué-Saavedra, A. Production and quality of habanero pepper (Capsicum Chinense Jacq.) with different NH4+/NO3− ratios and size of substrate particles. Terra Latinoam. 2012, 30, 9–15. [Google Scholar]
- Tapia-Vargas, M.; Larios-Guzmán, A.; Díaz-Sánchez, D.D.; Ramírez-Ojeda, G.; Hernández-Pérez, A.; Vidales-Fernández, I.; Guillén-Andrade, H. Black habanero pepper (Capsicum chinense Jacq.) hydroponics production. Rev. Fitotec. Mex. 2016, 39, 241–245. [Google Scholar] [CrossRef]
- Meneses-Lazo, R.E.; Garruña, R. The Habanero Pepper (Capsicum Chinense Jacq.) as a Study Plant Mothe in Mexico. Trop. Subtrop. Agroecosyst. 2020, 23, 1–17. [Google Scholar]
- Steiner, A.A. A Universal Method for Preparing Nutrient Solutions of a Certain Desired Composition. Plant Soil 1961, 15, 134–154. [Google Scholar] [CrossRef]
- He, Y.; Bose, S.K.; Wang, W.; Jia, X.; Lu, H.; Yin, H. Pre-Harvest Treatment of Chitosan Oligosaccharides Improved Strawberry Fruit Quality. Int. J. Mol. Sci. 2018, 19, 2194. [Google Scholar] [CrossRef]
- Hernández-Pérez, O.I.; Valdez-Aguilar, L.A.; Alia-Tejacal, I.; Cartmill, A.D.; Cartmill, D.L. Tomato Fruit Yield, Quality, and Nutrient Status in Response to Potassium: Calcium Balance and Electrical Conductivity in the Nutrient Solution. J. Soil Sci. Plant Nutr. 2020, 20, 484–492. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis of the AOAC, Methods 932.06, 925.09, 985.29, 923.03; Association of Official Analytical Chemists: Arlington, VA, USA, 1990. [Google Scholar]
- Pichyangkura, R.; Chadchawan, S. Biostimulant Activity of Chitosan in Horticulture. Sci. Hortic. 2015, 196, 49–65. [Google Scholar] [CrossRef]
- Chookhongkha, N.; Miyagawa, S.; Jirakiattikul, Y.; Photchanachai, S. Chili Growth and Seed Productivity as Affected by Chitosan. In Proceedings of the International Conference on Agriculture Technology and Food Sciences (ICATFS’2012), Manila, Philippines, 17–18 November 2012; pp. 146–149. [Google Scholar]
- Esyanti, R.R.; Dwivany, F.M.; Mahani, S.; Nugrahapraja, H.; Meitha, K. Foliar Application of Chitosan Enhances Growth and Modulates Expression of Defense Genes in Chilli Pepper (Capsicum annuum L.). Aust. J. Crop Sci. 2019, 13, 55–60. [Google Scholar] [CrossRef]
- Dzung, P.D.; Phu, D.V.; Du, B.D.; Ngoc, L.S.; Duy, N.N.; Hiet, H.D.; Nghia, D.H.; Thang, N.T.; Le, B.V.; Hien, N.Q. Effect of Foliar Application of Oligochitosan with Different Molecular Weight on Growth Promotion and Fruit Yield Enhancement of Chili Plant. Plant Prod. Sci. 2017, 20, 389–395. [Google Scholar] [CrossRef]
- Mahmood, N.; Abbasi, N.A.; Hafiz, I.A.; Ali, I.; Zakia, S. Effect of Biostimulants on Growth, Yield and Quality of Bell Pepper Cv. Yolo Wonder. Pak. J. Agric. Sci. 2017, 54, 311–317. [Google Scholar] [CrossRef]
- Li, K.; Xing, R.; Liu, S.; Li, P. Chitin and Chitosan Fragments Responsible for Plant Elicitor and Growth Stimulator. J. Agric. Food Chem. 2020, 68, 12203–12211. [Google Scholar] [CrossRef]
- Zhang, X.; Li, K.; Xing, R.; Liu, S.; Li, P. Metabolite Profiling of Wheat Seedlings Induced by Chitosan: Revelation of the Enhanced Carbon and Nitrogen Metabolism. Front. Plant Sci. 2017, 8, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, M.; Hasanuzzaman, M.; Rahman, M.; Khan, M.A.R.; Bhowmik, P.; Mahmud, N.U.; Tanveer, M.; Islam, T. Mechanism of Plant Growth Promotion and Disease Suppression by Chitosan Biopolymer. Agriculture 2020, 10, 624. [Google Scholar] [CrossRef]
- Zhang, X.; Li, K.; Xing, R.; Liu, S.; Chen, X.; Yang, H.; Li, P. MiRNA and MRNA Expression Profiles Reveal Insight into Chitosan-Mediated Regulation of Plant Growth. J. Agric. Food Chem. 2018, 66, 3810–3822. [Google Scholar] [CrossRef] [PubMed]
- El-Tanahy, A.M.M.; Mahmoud, A.R.; Abde-Mouty, M.M.; Ali, A.H. Effect of Chitosan Doses and Nitrogen Sources on the Growth, Yield and Seed Quality of Cowpea. Aust. J. Basic. Appl. Sci. 2012, 6, 115–121. [Google Scholar]
- Dwivany Fenny, M.; Karlia, M.; Kuswati, K.; Esyanti Rizkita, R.; Husna, N. Chitosan Improving Growth in Chili (Capsicum annuum L.) Plants and Acting through Distinct Gene Regulation between Cultivars. Res. J. Biotechnol. 2021, 16, 87–92. [Google Scholar]
- Ravi Kumar, M.N.V. A Review of Chitin and Chitosan Applications. React. Funct. Polym. 2000, 46, 1–27. [Google Scholar] [CrossRef]
- Jakobsen, S.T. Interaction between Plant Nutrients: III. Antagonism between Potassium, Magnesium and Calcium. Acta Agric. Scand. B Soil Plant Sci. 1993, 43, 1–5. [Google Scholar] [CrossRef]
- Labroo, M.R.; Studer, A.J.; Rutkoski, J.E. Heterosis and Hybrid Crop Breeding: A Multidisciplinary Review. Front. Genet. 2021, 12, 234. [Google Scholar] [CrossRef]
| Substance | Concentration (M) | Dilution (mL L−1) |
|---|---|---|
| CS | 0.04 | 50 |
| PAA | 0.04 | 50 |
| CS–PAA | 0.04 | 100 |
| Nutrient Solution | K+ | Ca2+ | Mg2+ | NO3− | H2PO4− | SO42− |
|---|---|---|---|---|---|---|
| NSA | 7.5 | 9 | 4 | 12 | 1.5 | 7 |
| NSB | 9 | 7.5 | 4 | 12 | 1.5 | 7 |
| Nutrient Solution | Ca(NO3)2·4H2O | MgSO4·7H2O | KH2PO4 | Mg(NO3)2·6H2O | K2SO4 | KNO3 |
|---|---|---|---|---|---|---|
| NSA | 1.063 | 0.495 | 0.205 | - | 0.257 | 0.308 |
| NSB | 0.886 | 0.284 | 0.205 | 0.218 | 0.409 | 0.287 |
| Plant Height (cm) | Stem Diameter (mm) | Number of Fruits | Leaf–Stem Dry Weight (g) | Root Dry Weight (g) | Total Dry Weight (g) | Yield per Plant (g) | |
|---|---|---|---|---|---|---|---|
| Foliar Applications (FA) | |||||||
| Control | 202.87 b | 9.10 b | 39.89 b | 48.13 b | 12.44 b | 60.57 b | 663.26 b |
| CS | 212.27 ab | 9.24 b | 39.03 b | 63.71 a | 14.45 a | 78.29 a | 719.69 b |
| PAA | 228.74 a | 10.23 a | 39.67 b | 65.41 a | 12.30 b | 77.71 a | 720.30 b |
| CS–PAA | 230.32 a | 10.36 a | 61.26 a | 74.01 a | 13.17 ab | 87.18 a | 956.68 a |
| ANOVA | * | ** | *** | *** | * | *** | *** |
| Nutrient Solution (NS) | |||||||
| NSA | 221.07 | 10.02 a | 47.22 | 66.64 | 13.36 | 80.07 | 834.52 a |
| NSB | 216.03 | 9.44 b | 42.70 | 58.98 | 12.82 | 71.80 | 695.45 b |
| ANOVA | ns | * | ns | ns | ns | ns | ** |
| Cultivars (C) | |||||||
| Chichen Itza | 235.06 a | 9.65 | 46.94 | 66.31 | 13.10 | 79.47 | 827.04 a |
| Jaguar | 202.04 b | 9.82 | 42.98 | 59.32 | 13.08 | 72.40 | 702.92 b |
| ANOVA | *** | ns | ns | ns | ns | ns | ** |
| Interactions | |||||||
| FA*NS | ns | ns | ns | ns | ns | ns | ns |
| FA*C | * | ns | ** | ns | * | * | ns |
| NS*C | ns | ns | ns | ns | ns | ns | * |
| FA*NS*C | ns | ns | ns | ns | * | ns | ns |
| Total Soluble Solids (°Brix) | Titratable Acid (% Citric Acid) | TSS/TA | pH | |||||
|---|---|---|---|---|---|---|---|---|
| Green | Ripe | Green | Ripe | Green | Ripe | Green | Ripe | |
| Foliar Applications (FA) | ||||||||
| Control | 2.58 a | 3.30 b | 0.21 | 0.20 | 13.61 a | 16.38 | 5.54 | 5.38 |
| CS | 2.33 b | 3.38 b | 0.21 | 0.23 | 11.84 b | 15.96 | 5.57 | 5.38 |
| PAA | 2.37 b | 3.28 b | 0.19 | 0.22 | 13.21 ab | 16.91 | 5.56 | 5.25 |
| CS–PAA | 2.68 a | 3.69 a | 0.22 | 0.21 | 14.52 a | 19.02 | 5.64 | 5.41 |
| ANOVA | *** | ** | ns | ns | * | ns | ns | ns |
| Nutrient Solution (NS) | ||||||||
| NSA | 2.52 | 3.33 | 0.21 | 0.21 | 13.30 | 16.49 | 5.57 | 5.33 |
| NSB | 2.46 | 3.44 | 0.20 | 0.21 | 13.29 | 17.64 | 5.59 | 5.38 |
| ANOVA | ns | ns | ns | ns | ns | ns | ns | ns |
| Cultivars (C) | ||||||||
| Chichen Itza | 2.55 a | 3.40 | 0.21 | 0.21 | 13.44 | 16.68 | 5.54 b | 5.25 b |
| Jaguar | 2.42 b | 3.42 | 0.20 | 0.22 | 13.15 | 17.45 | 5.62 a | 5.46 a |
| ANOVA | * | ns | ns | ns | ns | ns | * | ** |
| Interactions | ||||||||
| FA*NS | ns | ns | * | ns | ** | ns | ns | ns |
| FA*C | ns | ns | ns | ns | ns | ns | ns | ns |
| NS*C | * | ns | ns | ns | ns | ns | ns | ns |
| FA*NS*C | ** | ns | ns | * | ns | ns | ns | ns |
| Chichen Itza | Jaguar | |
|---|---|---|
| NSA | 944.96 a | 724.09 b |
| NSB | 709.13 b | 681.76 b |
| Total Soluble Solids (°Brix) of Green Fruits | pH of Ripe Fruits | |||
|---|---|---|---|---|
| Chichen Itza | Jaguar | Chichen Itza | Jaguar | |
| NSA | 2.65 a | 2.38 b | 5.16 b | 5.50 a |
| NSB | 2.45 b | 2.46 b | 5.34 a | 5.42 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palacios-Torres, R.E.; Santos-Chavez, A.; Ortega-Ortiz, H.; Ramírez-Seañez, A.R.; Yam-Tzec, J.A.; Amador-Mendoza, A.; Juárez-Maldonado, A.; Reyes-Osornio, M.; Hernández-Hernández, H. Effect of Chitosan-Poly(Acrylic Acid) Complexes and Two Nutrient Solutions on the Growth and Yield of Two Habanero Pepper Cultivars. Horticulturae 2022, 8, 201. https://doi.org/10.3390/horticulturae8030201
Palacios-Torres RE, Santos-Chavez A, Ortega-Ortiz H, Ramírez-Seañez AR, Yam-Tzec JA, Amador-Mendoza A, Juárez-Maldonado A, Reyes-Osornio M, Hernández-Hernández H. Effect of Chitosan-Poly(Acrylic Acid) Complexes and Two Nutrient Solutions on the Growth and Yield of Two Habanero Pepper Cultivars. Horticulturae. 2022; 8(3):201. https://doi.org/10.3390/horticulturae8030201
Chicago/Turabian StylePalacios-Torres, Rogelio Enrique, Amadeo Santos-Chavez, Hortensia Ortega-Ortiz, Ana Rosa Ramírez-Seañez, José Antonio Yam-Tzec, Adolfo Amador-Mendoza, Antonio Juárez-Maldonado, Maribel Reyes-Osornio, and Hipólito Hernández-Hernández. 2022. "Effect of Chitosan-Poly(Acrylic Acid) Complexes and Two Nutrient Solutions on the Growth and Yield of Two Habanero Pepper Cultivars" Horticulturae 8, no. 3: 201. https://doi.org/10.3390/horticulturae8030201
APA StylePalacios-Torres, R. E., Santos-Chavez, A., Ortega-Ortiz, H., Ramírez-Seañez, A. R., Yam-Tzec, J. A., Amador-Mendoza, A., Juárez-Maldonado, A., Reyes-Osornio, M., & Hernández-Hernández, H. (2022). Effect of Chitosan-Poly(Acrylic Acid) Complexes and Two Nutrient Solutions on the Growth and Yield of Two Habanero Pepper Cultivars. Horticulturae, 8(3), 201. https://doi.org/10.3390/horticulturae8030201







