Phenotyping Olive Cultivars for Drought Tolerance Using Leaf Macro-Characteristics
Abstract
1. Introduction
2. Material and Methods
2.1. Overall Approach
2.2. First Experiment
2.3. Second Experiment
2.4. Statistical Analysis
3. Results and Discussion
3.1. Traits Variability within the Collection
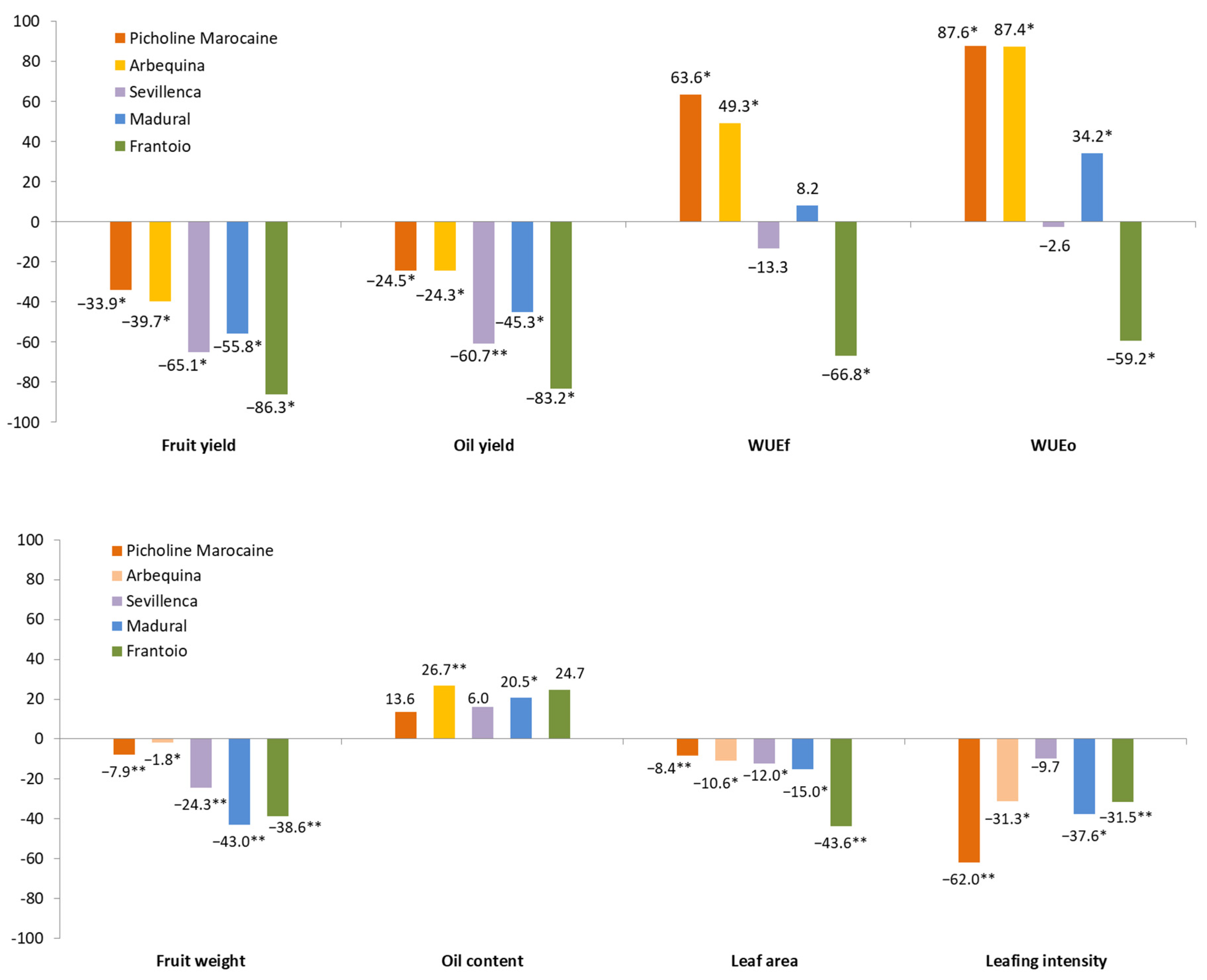
3.2. Olive Varieties Response to Drought
3.3. Correlations
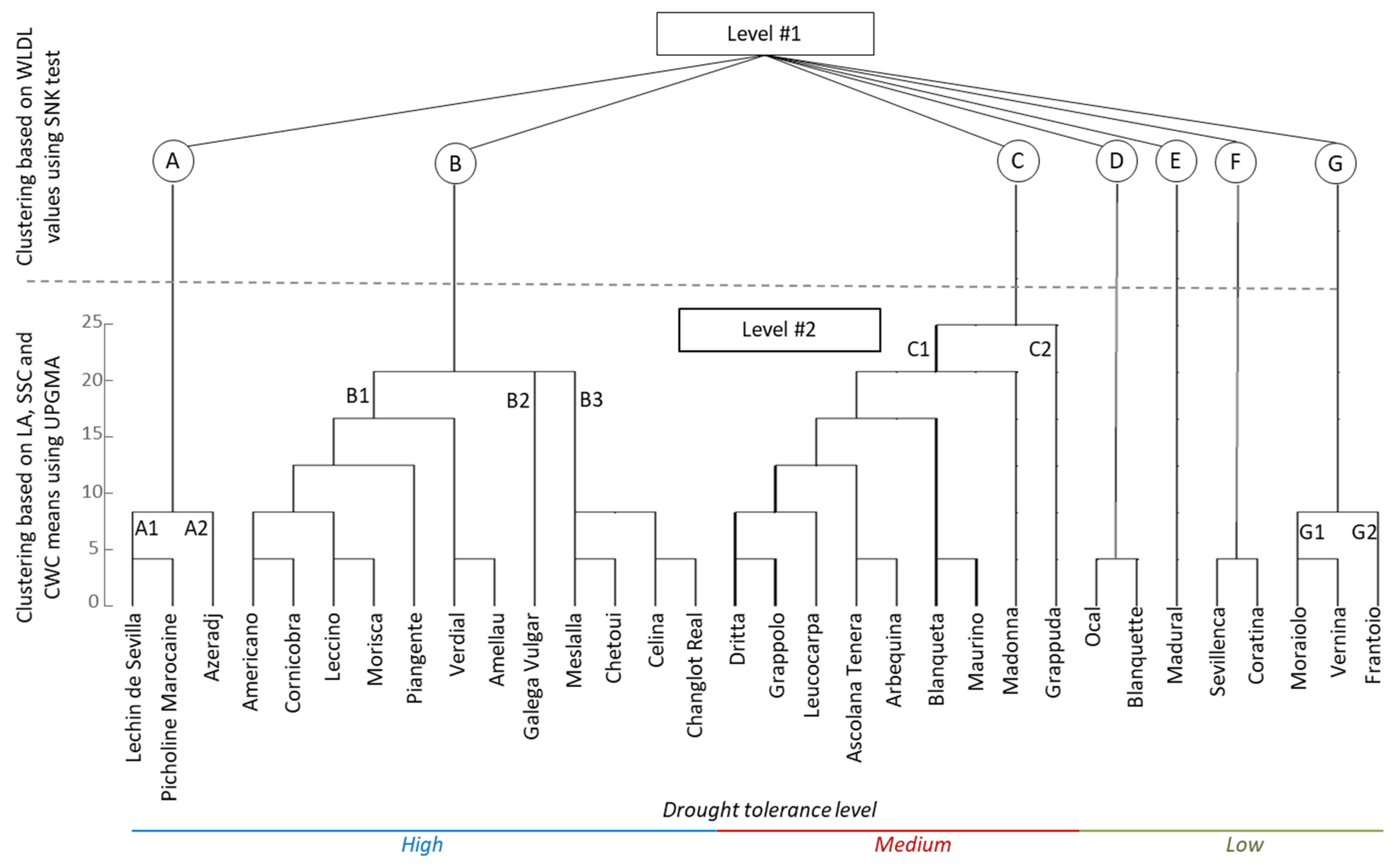
3.4. Hierarchical Clustering
4. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cramer, W.; Guiot, J.; Fader, M.; Garrabou, J.; Gattuso, J.P.; Iglesias, A.; Lange, M.A.; Lionello, P.; Llasat, M.C.; Paz, S.; et al. Climate change and interconnected risks to sustainable development in the Mediterranean. Nat. Clim. Change 2018, 8, 972–980. [Google Scholar] [CrossRef]
- Fraga, H.; Moriondo, M.; Leolini, L.; Santos, J.A. Mediterranean Olive Orchards under Climate Change: A Review of Future Impacts and Adaptation Strategies. Agronomy 2021, 11, 56. [Google Scholar] [CrossRef]
- Bari, A. Assessment of plant genetic resources for Water-Use Efficiency (WUE): Managing water scarcity. In Proceedings of the Bioversity International Workshop for North Africa and West Asia, Marrakech, Morocco, 10–12 October 2005; Bioversity International: Roma, Italy, 2007. [Google Scholar]
- Fernández, J.E.; Perez-Martin, A.; Torres-Ruiz, J.M.; Cuevas, M.; Rodríguez, D.C.M.; Farag, S.; Morales-Sillero, A.; García, J.; Hernández-Santana, V.; Diaz-Espejo, A. A regulated deficit irrigation strategy for hedgerow olive orchards with high plant density. Plant Soil 2013, 372, 279–295. [Google Scholar] [CrossRef]
- Loumou, A.; Giourga, C. Olive groves: The life and identity of the Mediterranean. Agric. Hum. Values 2003, 20, 87–95. [Google Scholar] [CrossRef]
- Tous, J.; Romero, A.; Hermoso, J.F. New trends in olive orchard design for continuous mechanical harvesting. Adv. Hortic. Sci. 2010, 24, 43–52. [Google Scholar]
- Tognetti, R.; D’Andria, R.; Morelli, G.; Calandrelli, D.; Fragnito, F. Irrigation effects on daily and seasonal variations of trunk sap flow and leaf water relations in olive trees. Plant Soil 2004, 263, 249–264. [Google Scholar] [CrossRef]
- Bacelar, E.A.; Santos, D.L.; Moutinho-Pereira, J.M.; Lopes, J.I.; Goncalves, B.C.; Ferreira, T.C.; Correia, C.M. Physiological behavior, oxidative damage and antioxidative protection of olive trees grown under different irrigation regimes. Plant Soil 2007, 292, 1. [Google Scholar] [CrossRef]
- Diaz-Rueda, P.; Franco-Navarro, J.D.; Messora, R.; Espartero, J.; Rivero-Núñez, C.M.; Aleza, P.; Capote, N.; Cantos, M.; García-Fernández, J.L.; De Cires, A. SILVOLIVE, a germplasm collection of wild subspecies with high genetic variability as a source of rootstocks and resistance genes for olive breeding. Front. Plant Sci. 2020, 11, 629. [Google Scholar] [CrossRef]
- Cruz de Carvalho, M.H. Drought stress and reactive oxygen species: Production, scavenging and signaling. Plant Signal. Behav. 2008, 3, 156–165. [Google Scholar] [CrossRef]
- Sofo, A.; Dichio, B.; Xiloyannis, C.; Masia, A. Antioxidant defences in olive trees during drought stress: Changes in activity of some antioxidant enzymes. Funct. Plant Biol. 2005, 32, 45–53. [Google Scholar] [CrossRef]
- Proietti, P.; Nasini, L.; Reale, L.; Caruso, T.; Ferranti, F. Productive and vegetative behavior of olive cultivars in super high-density olive grove. Sci. Agric. 2015, 72, 20–27. [Google Scholar] [CrossRef]
- Ennajeh, M.; Tounekti, T.; Vadel, A.M.; Khemira, H.; Cochard, H. Water relations and drought-induced embolism in olive (Olea europaea) varieties ‘Meski’ and ‘Chemlali’ during severe drought. Tree Physiol. 2008, 28, 971–976. [Google Scholar] [CrossRef] [PubMed]
- Dichio, B.; Xiloyannis, C.; Angelopoulos, K.; Nuzzo, V.; Bufo, S.A.; Celano, G. Drought-induced variations of water relations parameters in Olea europaea. Plant Soil 2003, 257, 381–389. [Google Scholar] [CrossRef]
- Karimi, S.; Rahemi, M.; Rostami, A.A.; Sedaghat, S. Drought effects on growth, water content and osmoprotectants in four olive cultivars with different drought tolerance. Int. J. Fruit Sci. 2018, 18, 254–267. [Google Scholar] [CrossRef]
- Sofo, A.; Manfreda, S.; Fiorentino, M.; Dichio, B.; Xiloyannis, C. The olive tree: A paradigm for drought tolerance in Mediterranean climates. Hydrol. Earth Syst. Sci. 2008, 12, 293–301. [Google Scholar] [CrossRef]
- Naoum, S.; Albalawneh, A.; Ayoub, S.; Diab, M.; Amayreh, I.; Ammoush, M.; Kawaleet, B.; Daoud, L. Productivity of water, growth and yield of olive trees under deficit irrigation. Acta Hortic. 2018, 1199, 261–266. [Google Scholar] [CrossRef]
- Guerfel, M.; Baccouri, O.; Boujnah, D.; Chaïbi, W.; Zarrouk, M. Impacts of water stress on gas exchange, water relations, chlorophyll content and leaf structure in the two main Tunisian olive (Olea europaea L.) cultivars. Sci. Hortic. 2009, 119, 257–263. [Google Scholar] [CrossRef]
- Torres-Ruiz, J.M.; Diaz-Espejo, A.; Morales-Sillero, A.; Martín-Palomo, M.J.; Mayr, S.; Beikircher, B.; Fernández, J.E. Shoot hydraulic characteristics, plant water status and stomatal response in olive trees under different soil water conditions. Plant Soil 2013, 373, 77–87. [Google Scholar] [CrossRef]
- Ahmadipour, S.; Arji, I.; Ebadi, A.; Abdossi, V. Physiological and biochemical responses of some olive cultivars (Olea europaea L.) to water stress. Cell. Mol. Biol. 2018, 64, 20–29. [Google Scholar] [CrossRef]
- Anjum, S.A.; Xie, X.Y.; Wang, L.C.; Saleem, M.F.; Man, C.; Lei, W. Morphological, physiological and biochemical responses of plants to drought stress. Afr. J. Agric. Res. 2011, 6, 2026–2032. [Google Scholar]
- Fernández, J.E.; Diaz-Espejo, A.; Infante, J.M.; Durán, P.; Palomo, M.J.; Chamorro, V.; Villagarcía, L. Water relations and gas exchange in olive trees under regulated deficit irrigation and partial rootzone drying. Plant Soil 2006, 284, 273–291. [Google Scholar] [CrossRef]
- Fereres, E.; Pruitt, W.O.; Beutel, J.A.; Henderson, D.W.; Holzapfel, E.; Shulbach, H.; Uriu, K. Evapotranspiration and drip irrigation scheduling. In: Drip irrigation management. (Fereres, E. Technical Editor). Univ. Calif. Div. Agric. Sci. 1981, 21259, 8–13. [Google Scholar]
- Schwabe, W.W.; Lionakis, S.M. Leaf attitude in olive in relation to drought resistance. J. Hortic. Sci. 1996, 71, 157–166. [Google Scholar] [CrossRef]
- Gitz, D.C.; Baker, J.T. Methods for creating stomatal impressions directly onto achievable slides. Agron. J. 2009, 101, 232–236. [Google Scholar] [CrossRef]
- Bewick, T.A.; Shilling, D.G.; Querns, R. Evaluation of epicuticular wax removal from whole leaves with chloroform. Weed Technol. 1993, 7, 706–716. [Google Scholar] [CrossRef]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.T.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Gholami, R.; Zahedi, S.M. Identifying superior drought-tolerant olive genotypes and their biochemical and some physiological responses to various irrigation levels. J. Plant Nutr. 2019, 42, 2057–2069. [Google Scholar] [CrossRef]
- Ennajeh, M.; Vadel, A.M.; Cochard, H.; Khemira, H. Comparative impacts of water stress on the leaf anatomy of a drought-resistant and a drought-sensitive olive cultivar. J. Hortic. Sci. Biotechnol. 2010, 85, 289–294. [Google Scholar] [CrossRef]
- Guerfel, M.; Boujnah, D.; Baccouri, B.; Zarrouk, M. Evaluation of morphological and physiological traits for drought tolerance in 12 Tunisian olive varieties (Olea europaea L.). J. Agron. 2007, 6, 356–361. [Google Scholar] [CrossRef]
- Kardiman, R.; Raebild, A. Relationship between stomatal density, size and speed of opening in Sumatran rainforest species. Tree Physiol. 2018, 38, 696–705. [Google Scholar] [CrossRef]
- Yadollahi, A.; Arzani, K.; Ebadi, A.; Wirthensohn, M.; Karimi, S. The response of different almond genotypes to moderate and severe water stress in order to screen for drought tolerance. Sci. Hortic. 2011, 129, 403–413. [Google Scholar]
- Chaari-Rkhis, A.; Maalej, M.; Chelli-Chaabouni, A.; Fki, L.; Drira, N. Photosynthesis parameters during acclimatization of in vitro-grown olive plantlets. Photosynthetica 2015, 53, 613–616. [Google Scholar] [CrossRef]
- Galdon-Armero, J.; Fullana-Pericas, M.; Mulet, P.A.; Conesa, M.A.; Martin, C.; Galmes, J. The ratio of trichomes to stomata is associated with water use efficiency in Solanum lycopersicum (tomato). Plant J. 2018, 96, 607–619. [Google Scholar]
- Ludlow, M.M.; Muchow, R.C. A critical evaluation of traits for improving crop yields in water-limited environments. Adv. Agron. 1990, 43, 107–153. [Google Scholar]
- Pierantozzi, P.; Torres, M.; Bodoira, R.; Maestri, D. Water relations, biochemical–physiological and yield responses of olive trees (Olea europaea L. cvs. Arbequina and Manzanilla) under drought stress during the pre-flowering and flowering period. Agric. Water Manag. 2013, 125, 13–25. [Google Scholar] [CrossRef]
- Gomez-del-Campo, M.; Pérez-Expósito, M.Á.; Hammami, S.B.; Centeno, A.; Rapoport, H.F. Effect of varied summer deficit irrigation on components of olive fruit growth and development. Agric. Water Manag. 2014, 137, 84–91. [Google Scholar] [CrossRef]
- Schuster, A.C.; Burghardt, M.; Riederer, M. The ecophysiology of leaf cuticular transpiration: Are cuticular water permeabilities adapted to ecological conditions? J. Exp. Bot. 2017, 68, 5271–5279. [Google Scholar] [CrossRef] [PubMed]
- Mingjie, C.; Yi, Z.; Xiangrui, K.; Zhenghua, D.; Huiwen, Z.; Zhaoxi, Y.; Jianheng, Q.; Changsong, C. Leaf cuticular transpiration barrier organization in tea tree under normal growth conditions. Front. Plant Sci. 2021, 12, 1316–1332. [Google Scholar]
- Zhang, Q.; Chen, G.; Huang, J.; Peng, C. Comparison of the ability to control water loss in the detached leaves of Wedelia trilobata, Wedelia chinensis, and their hybrid. Plants 2020, 9, 1227. [Google Scholar] [CrossRef]
- Medina-Alonso, M.G.; Navas, J.F.; Cabezas, J.M.; Weiland, C.M.; Ríos-Mesa, D.; Lorite, I.J.; de la Rosa, R. Differences on flowering phenology under Mediterranean and Subtropical environments for two representative olive cultivars. Environ. Exp. Bot. 2020, 180, 104239. [Google Scholar] [CrossRef]
- Kour, D.; Bakshi, P.; Wali, V.K.; Sharma, N.; Sharma, A.; Iqbal, M. Alternate bearing in olive: A review. Int. J. Curr. Microbiol. Appl. Sci. 2018, 7, 2281–2297. [Google Scholar]
- Hadiddou, A.; Oukabli, A.; Moudaffar, C.; Mamouni, A.; Gaboun, F.; Mekaoui, A.; Hssaini, L.; El Fechtali, M. Evaluation des performances de production de 14 variétés d’olivier (Olea europaea L.) nationales et méditerranéennes dans deux systèmes contrastés de culture (pluvial et irrigué) au Maroc. Al Awamia 2013, 127, 22–43. [Google Scholar]
- Mezghani, M.A.; Charfi, C.M.; Gouiaa, M.; Labidi, F. Vegetative and reproductive behaviour of some olive tree varieties (Olea europaea L.) under deficit irrigation regimes in semi-arid conditions of Central Tunisia. Sci. Hortic. 2012, 146, 143–152. [Google Scholar]
- Tognetti, R.; d’Andria, R.; Lavini, A.; Morelli, G. The effect of deficit irrigation on crop yield and vegetative development of Olea europaea L. (cvs. Frantoio and Leccino). Eur. J. Agron. 2006, 25, 356–364. [Google Scholar] [CrossRef]
- Iniesta, F.; Testi, L.; Orgaz, F.; Villalobos, F.J. The effects of regulated and continuous deficit irrigation on the water use, growth and yield of olive trees. Eur. J. Agron. 2009, 30, 258–265. [Google Scholar]
- Fernandes-Silva, A.A.; Ferreira, T.C.; Correia, C.M.; Malheiro, A.C.; Villalobos, F.J. Influence of different irrigation regimes on crop yield and water use efficiency of olive. Plant Soil 2010, 333, 35–47. [Google Scholar]
- Turktas, M.; Inal, B.; Okay, S.; Erkilic, E.G.; Dundar, E.; Hernandez, P.; Unver, T. Nutrition metabolism plays an important role in the alternate bearing of the olive tree (Olea europaea L.). PLoS ONE 2013, 8, e59876. [Google Scholar] [CrossRef]
- Mailer, R.; Ayton, J. Effect of irrigation and water stress on olive oil quality and yield based on a four year study. Acta Hortic. 2011, 888, 63–72. [Google Scholar] [CrossRef]
- Dag, A.; Naor, A.; Ben-Gal, A.; Harlev, G.; Zipori, I.; Schneider, D.; Kerem, Z. The effect of water stress on super-high-density ‘Koroneiki’ olive oil quality. J. Sci. Food Agric. 2015, 95, 2016–2020. [Google Scholar]
- Brito, C.; Dinis, L.T.; Moutinho-Pereira, J.; Correia, C.M. Drought stress effects and olive tree acclimation under a changing climate. Plants 2019, 8, 232. [Google Scholar] [CrossRef]
- Deng, S.; Yin, Q.; Zhang, S.; Shi, K.; Jia, Z.; Ma, L. Drip irrigation affects the morphology and distribution of olive roots. Hortscience 2017, 52, 1298–1306. [Google Scholar] [CrossRef]
- Ristic, Z.; Jenks, M.A. Leaf cuticle and water loss in maize lines differing in dehydration avoidance. J. Plant Physiol. 2002, 159, 645–651. [Google Scholar] [CrossRef]
- Hopper, D.W.; Ghan, R.; Cramer, G.R. A rapid dehydration leaf assay reveals stomatal response differences in grapevine genotypes. Hortic. Res. 2014, 1, 2. [Google Scholar] [PubMed]
- Drake, P.L.; Froend, R.H.; Franks, P.J. Smaller, faster stomata: Scaling of stomatal size, rate of response, and stomatal conductance. J. Exp. Bot. 2013, 64, 495–505. [Google Scholar]
- Bacelar, E.A.; Correia, C.M.; Moutinho-Pereira, J.M.; Gonçalves, B.C.; Lopes, J.I.; Torres-Pereira, J.M. Sclerophylly and leaf anatomical traits of five field-grown olive cultivars growing under drought conditions. Tree Physiol. 2004, 24, 233–239. [Google Scholar] [CrossRef]
- Bosabalidis, A.M.; Kofidis, G. Comparative effects of drought stress on leaf anatomy of two olive cultivars. Plant Sci. 2002, 163, 375–379. [Google Scholar]
- Farooq, M.; Hussain, M.; Wahid, A.; Siddique, K.H.M. Drought stress in plants: An overview. In Plant Responses to Drought Stress: From Morphological to Molecular Features; Aroca, R., Ed.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 1–33. ISBN 9783642326530. [Google Scholar]
- Wang, C.; He, J.; Zhao, T.H.; Cao, Y.; Wang, G.; Sun, B.; Li, M.H. The smaller the leaf is, the faster the leaf water loses in a temperate forest. Front. Plant Sci. 2019, 10, 58. [Google Scholar]
- Chehab, H.; Mechri, B.; Mariem, F.B.; Hammami, M.; Elhadj, S.B.; Braham, M. Effect of different irrigation regimes on carbohydrate partitioning in leaves and wood of two table olive cultivars (Olea europaea L. cv. Meski and Picholine). Agric. Water Manag. 2009, 96, 293–298. [Google Scholar] [CrossRef]
- De Pascali, M.; Vergine, M.; Negro, C.; Greco, D.; Vita, F.; Sabella, E.; Luvisi, A. Xylella fastidiosa and drought stress in olive trees: A complex relationship mediated by soluble sugars. Biology 2022, 11, 112. [Google Scholar] [CrossRef]
- Bacelar, E.A.; Moutinho-Pereira, J.M.; Gonçalves, B.C.; Lopes, J.I.; Correia, C.M. Physiological responses of different olive genotypes to drought conditions. Acta Physiol. Plant. 2009, 31, 611–621. [Google Scholar] [CrossRef]
- Marin, L.; Benlloch, M.; Fernandez-Escobar, R. Screening of olive cultivars for salt tolerance. Sci. Hortic. 1995, 64, 113–116. [Google Scholar] [CrossRef]
| Variety | Origin | Variety | Origin |
|---|---|---|---|
| Amellau | France | Grappolo | Italy |
| Americano | Italy | Leucocarpa | Italy |
| Arbequina * | Spain | Leccino | Italy |
| Ascolana Tenra | Italy | Lechin de Sevilla | Spain |
| Azeradj | Algeria | Madonna | Italy |
| Blanqueta | Spain | Madural * | Portugal |
| Blanquette | France | Maurino | Italy |
| Cellina | Italy | Meslalla | Morocco |
| Changlot Real | Spain | Moraiolo | Italy |
| Chetoui | Tunisia | Morisca | Spain |
| Cornicabra | Spain | Ocal | Spain |
| Coratina | Italy | Piangente | Italy |
| Grappuda | Italy | Picholine Marocaine * | Morocco |
| Dritta | Italy | Sevillenca * | Spain |
| Frantoio * | Italy | Verdial | Portugal |
| Galega Vulgar | Portugal | Vernina | Italy |
| Min | Max | Mean | Std. Deviation | Mean Square | ANOVA p-Value | |
| Leaf area (cm2) | 2.9 | 9.5 | 5.00 | 1.5 | 18.7 | <0.001 |
| Petiole elasticity (°) | 8.0 | 36.0 | 20.6 | 8.1 | 240.2 | <0.001 |
| Water loss in detached leaves (%) | 3.6 | 11.6 | 6.6 | 3.2 | 2.8 | <0.001 |
| Stomatal density (no mm−2) | 222.6 | 470.1 | 337.9 | 58.3 | 6796.6 | 0.002 |
| Stomatal length (µm) | 11.4 | 18.7 | 16.3 | 1.7 | 5.8 | 0.001 |
| Trichomes density (no mm−2) | 120.5 | 204.4 | 158.5 | 24.1 | 207.1 | <0.001 |
| Trichome width (µm) | 103.6 | 183.4 | 138.8 | 25.5 | 1582.3 | <0.001 |
| Trichomes per stoma | 0.3 | 0.7 | 0.5 | 0.1 | 0.03 | <0.001 |
| TAI (mm2 mm−2) | 1.1 | 4.8 | 2.4 | 1.0 | 9.9 | <0.001 |
| SSC (mg gdw−1) | 15.8 | 536.9 | 167.3 | 138.7 | 0.05 | <0.001 |
| CWC (µg cm−2) | 44.7 | 606.2 | 240.4 | 177.0 | 1817.3 | <0.001 |
| Varieties | LA | SD | SL | TD | TW | TD/SD | TAI | PE | WLDL | SSC | CWC |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Amellau | 5.7 bcde | 338.1 abc | 16.1 ab | 174.2 fghi | 129.1 cdef | 0.5 bc | 2.3 cd | 13.1 abc | 5.4 ab | 292.3 bcd | 141.4 abc |
| Americano | 4.4 abc | 354.5 abc | 15.7 ab | 131.8 abc | 176.9 g | 0.4 ab | 3.2 fgh | 8.0 a | 4.9 ab | 49.3 ab | 105.9 a |
| Arbequina | 3.9 abc | 404.4 abc | 14.5 ab | 170.3 fghi | 116.1 abcd | 0.4 abc | 1.8 abcd | 18.0 efg | 6.9 abc | 117.7 ab | 163.5 abc |
| Ascolana Tenra | 7.1 def | 296.9 abc | 17.9 c | 153.2 defg | 119.9 abcd | 0.5 bc | 1.7 abcd | 28.0 ghi | 6.9 abc | 176.6 abc | 184.3 bcd |
| Azeradj | 7.4 ef | 370.8 abc | 15.0 ab | 124.3 ab | 130.3 cdef | 0.3 ab | 1.7 abcd | 22.1 defg | 4.2 a | 164.4 abc | 44.7 a |
| Blanqueta | 5.7 bcde | 429.3 bc | 14.4 ab | 194.7 ij | 110.5 abc | 0.4 bc | 1.9 bcd | 36.0 j | 6.2 abc | 62.9 ab | 58.4 a |
| Blanquette | 3.1 a | 354.5 abc | 15.3 ab | 162.4 efgh | 182.7 g | 0.5 bc | 4.3 jk | 17.0 bcde | 7.3 bc | 142.5 ab | 481.5 cd |
| Cellina | 4.2 abc | 313.3 abc | 17.2 c | 194.4 ij | 133.1 cdef | 0.6 bc | 2.7 efg | 23.1 efg | 5.3 ab | 268.0 abcd | 510.5 def |
| Changlot Real | 4.1 abc | 255.5 ab | 18.6 c | 167.2 fghi | 158.3 defg | 0.6 c | 3.3 fgh | 31.0 hij | 5.4 ab | 254.2 abcd | 451.7 de |
| Chetoui | 4.9 abc | 429.3 bc | 14.4 c | 150.4 defg | 103.6 a | 0.3 ab | 1.3 ab | 18.1 cdef | 5.0 ab | 72.5 ab | 483.2 ef |
| Cornicabra | 4.1 abc | 304.9 abc | 17.2 c | 138.0 abcd | 153.5 defg | 0.4 bc | 2.5 def | 13.1 abc | 5.4 ab | 42.0 ab | 104.2 a |
| Coratina | 6.3 cde | 304.8 abc | 17.3 c | 127.6 abc | 171.5 fg | 0.4 abc | 2.9 fgh | 18.9 cdef | 9.7 de | 230.2 abcd | 352.0 ef |
| Grappuda | 4.1 abc | 387.3 abc | 14.9 ab | 188.7 hij | 180.9 g | 0.5 bc | 4.8 l | 9.1 a | 7.1 abc | 487.2 e | 250.4 bcd |
| Dritta | 3.9 abc | 222.6 a | 18.7 c | 127.0 abc | 131.1 cdef | 0.6 bc | 1.7 abcd | 24.0 efg | 6.7 abc | 43.2 ab | 192.1 bcd |
| Frantoio | 5.2 abcd | 330.6 abc | 16.4 ab | 180.9 ghij | 126.7 bcde | 0.5 bc | 2.3 cd | 11.0 ab | 11.5 e | 134.4 ab | 339.1 de |
| Galega Vulgar | 4.5 abc | 313.6 abc | 17.2 c | 204.4 j | 104.0 a | 0.6 c | 1.7 abcd | 28.0 ghi | 5.3 ab | 536.9 f | 298.7 bcd |
| Grappolo | 9.5 g | 445.5 bc | 13.7 ab | 127.0 abc | 124.8 bcde | 0.3 a | 1.5 abcd | 22.0 defg | 7.1 abc | 17.7 a | 196.0 de |
| Leucocarpa | 4.7 abc | 371.5 abc | 14.9 ab | 176.8 ghij | 115.2 abcd | 0.5 bc | 1.8 abcd | 18.0 cdef | 6.7 abc | 57.6 ab | 281.5 bcd |
| Leccino | 5.4 abcd | 271.9 abc | 18.4 c | 127.0 abc | 107.6 ab | 0.5 bc | 1.1 a | 22.1 defg | 4.4 ab | 31.1 ab | 66.9 a |
| Lechin de Sevilla | 3.7 ab | 247.7 ab | 18.6 c | 155.7 defg | 164.6 efg | 0.6 c | 3.3 ghi | 36.0 j | 3.6 a | 386.1 cde | 60.7 a |
| Madonna | 8.5 fg | 330.0 abc | 17.1 c | 136.9 abcd | 153.3 defg | 0.4 abc | 2.5 cde | 11.0 abc | 6.7 abc | 280.1 abcd | 164.1 cd |
| Madural | 3.7 ab | 346.5 abc | 15.8 ab | 145.0 bcde | 142.5 defg | 0.4 abc | 2.3 cd | 25.1 fgh | 8.6 cde | 198.1 abcd | 110.5 ab |
| Maurino | 5.1 abcd | 470.1 c | 11.4 a | 178.1 ghij | 121.2 abcd | 0.4 ab | 2.0 bcd | 16.1 bcde | 7.0 abc | 167.7 abc | 59.3 a |
| Meslalla | 5.4 abcd | 297.0 abc | 17.6 c | 150.7 defg | 140.9 defg | 0.5 bc | 2.3 cd | 8.0 a | 4.6 ab | 90.9 ab | 518.8 ef |
| Moraiolo | 4.6 abc | 345.5 abc | 16.0 ab | 120.5 a | 149.6 defg | 0.3 ab | 2.1 cd | 31.1 ij | 10.2 e | 95.6 ab | 517.8 ef |
| Morisca | 5.2 abcd | 396.3 abc | 14.8 ab | 167.0 fghi | 115.8 abcd | 0.4 abc | 1.8 abcd | 21.1 defg | 5.4 ab | 15.8 a | 77.8 a |
| Ocal | 4.9 abc | 280.4 abc | 18.4 c | 197.0 ij | 108.9 abc | 0.7 c | 1.8 abcd | 15.0 abcd | 7.3 bc | 47.5 ab | 488.0 ef |
| Piangente | 3.9 abc | 337.8 abc | 16.1 ab | 157.9 defg | 166.9 efg | 0.5 bc | 3.4 hij | 34.1 j | 5.1 ab | 88.1 ab | 112.9 ab |
| Picholine Mar. | 3.4 a | 288.6 abc | 18.3 c | 170.7 fghi | 166.1 efg | 0.6 bc | 3.7 ijk | 25.0 fgh | 3.8 a | 414.1 de | 107.3 a |
| Sevillenca | 2.9 a | 305.0 abc | 17.2 c | 150.1 defg | 142.2 defg | 0.5 bc | 2.4 cde | 23.0 efg | 9.6 de | 93.0 ab | 79.8 a |
| Verdial | 4.0 abc | 321.5 abc | 17.1 c | 175.6 fghi | 183.4 g | 0.5 bc | 4.6 kl | 23.0 efg | 5.2 ab | 235.5 abcd | 83.8 a |
| Vernina | 5.1 abcd | 346.5 abc | 16.0 ab | 146.8 cdef | 109.5 abc | 0.4 abc | 1.4 abc | 9.1 a | 11.5 e | 62.5 ab | 606.2 g |
| Year | Variety | Fruit Yield | Oil Yield | Fruit Weight | Oil Content | Leaf Area | Leaves dm−1 | WUEf | WUEo | |
|---|---|---|---|---|---|---|---|---|---|---|
| 2019 | Picholine | WT | 28.6 d | 4.0 c | 5.1 c | 13.9 ab | 5.7 de | 14.3 c | 3.1 bc | 0.4 b |
| ST | 19.5 c | 3.4 c | 4.7 c | 17.6 bc | 5.3 c | 5.7 a | 5.3 d | 0.9 d | ||
| Arbequina | WT | 21.4 c | 3.2 c | 1.7 a | 14.8 ab | 6.6 g | 11.0 b | 2.3 b | 0.3 b | |
| ST | 13.3 b | 2.5 bc | 1.7 a | 19.1 c | 6.0 ef | 8.0 ab | 3.6 c | 0.7 c | ||
| Sevillenca | WT | 24.1 cd | 3.5 c | 2.2 a | 14.4 ab | 4.8 b | 10.0 ab | 2.6 bc | 0.4 b | |
| ST | 9.4 b | 1.4 ab | 1.7 a | 15.4 abc | 4.3 a | 9.7 ab | 2.6 bc | 0.4 b | ||
| Madural | WT | 3.9 a | 0.4 a | 3.9 b | 11.8 a | 6.3 fg | 7.3 ab | 0.4 a | 0.1 a | |
| ST | 1.2 a | 0.2 a | 2.2 a | 14.5 ab | 5.4 cd | 5.0 a | 0.3 a | 0.1 a | ||
| Frantoio | WT | 11.9 b | 1.8 b | 2.1 a | 15.2 abc | 8.9 h | 9.3 ab | 1.3 a | 0.2 ab | |
| ST | 0.6 a | 0.1 a | 1.3 a | 19.4 c | 5.1 bc | 6.7 ab | 0.2 a | 0.03 a | ||
| 2020 | Picholine | WT | 25.0 e | 3.6 d | 5.1 c | 14.5 abc | 5.6 d | 14.7 f | 2.8 cd | 0.4 c |
| ST | 16.0 cd | 2.3 bcd | 4.7 c | 14.6 abc | 5.1 bc | 5.3 ab | 4.3 e | 0.6 d | ||
| Arbequina | WT | 20.0 de | 3.1 cd | 1.7 a | 15.1 abc | 6.5 e | 11.3 e | 2.3 bcd | 0.3 bc | |
| ST | 11.7 bc | 2.2 abcd | 1.7 a | 18.8 c | 5.8 d | 7.3 bcd | 3.2 d | 0.6 d | ||
| Sevillenca | WT | 21.8 de | 3.4 cd | 2.2 a | 14.78 abc | 4.8 b | 10.3 e | 2.5 bcd | 0.4 bc | |
| ST | 6.7 ab | 1.2 ab | 1.7 a | 18.6 bc | 4.2 a | 8.7 cde | 1.8 abc | 0.3 bc | ||
| Madural | WT | 6.4 ab | 0.7 ab | 4.0 b | 11.7 a | 6.2 e | 7.7 bcd | 0.7 a | 0.1 a | |
| ST | 3.6 a | 0.5 a | 2.3 a | 13.8 ab | 5.2 c | 4.3 a | 1.0 a | 0.1 ab | ||
| Frantoio | WT | 13.3 c | 1.8 abc | 2.1 a | 13.9 ab | 8.8 f | 9.7 de | 1.5 ab | 0.2 abc | |
| ST | 3.0 a | 0.5 a | 1.3 a | 16.9 bc | 4.9 bc | 6.3 abc | 0.8 a | 0.1 ab | ||
| Water treatment (T) | *** | *** | *** | *** | *** | *** | *** | *** | ||
| Genotype (G) | *** | *** | *** | * | * | ns | *** | *** | ||
| Year (Y) | * | * | ns | * | ns | ns | * | * | ||
| T × G | *** | * | *** | ns | *** | *** | *** | *** | ||
| T × Y | ns | Ns | ns | ns | ns | ns | ns | ns | ||
| G × Y | ns | Ns | ns | ns | ns | ns | ns | ns | ||
| T × G × Y | ns | Ns | ns | ns | ns | ns | ns | ns | ||
| Leaf Traits | TSI | |||||||
|---|---|---|---|---|---|---|---|---|
| Fruit Yield | Oil Yield | Fruit Weight | Oil Content | Leaf Area | Leafing Intensity | WUEf | WUEo | |
| Leaf area | −0.647 | −0.570 | −0.375 | 0.669 | −0.881 * | −0.132 | −0.647 | −0.570 |
| Stomatal density | 0.068 | 0.127 | 0.203 | 0.462 | 0.025 | 0.213 | 0.068 | 0.127 |
| Stomatal length | 0.113 | 0.059 | −0.010 | −0.376 | 0.087 | −0.340 | 0.113 | 0.059 |
| Trichomes density | −0.049 | 0.001 | 0.325 | 0.651 | −0.512 | −0.404 | −0.049 | 0.001 |
| Trichome width | 0.340 | 0.311 | 0.030 | −0.247 | 0.388 | −0.538 | 0.340 | 0.311 |
| Petiole elasticity | 0.593 | 0.558 | 0.167 | −0.400 | 0.845 | −0.202 | 0.593 | 0.558 |
| Trichomes per stoma | −0.030 | −0.044 | 0.080 | 0.054 | −0.297 | −0.486 | −0.030 | −0.044 |
| Trichomes area index | 0.383 | 0.373 | 0.201 | 0.004 | 0.254 | −0.719 | 0.383 | 0.373 |
| WDLD | −0.973 ** | −0.939 * | −0.857 | 0.153 | −0.918 * | 0.238 | −0.973 ** | −0.939 * |
| SSC | 0.503 | 0.534 | 0.282 | 0.324 | 0.329 | −0.916 * | 0.503 | 0.534 |
| CWC | −0.674 | −0.619 | −0.328 | 0.528 | −0.930 * | −0.021 | −0.674 | −0.619 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Razouk, R.; Hssaini, L.; Alghoum, M.; Adiba, A.; Hamdani, A. Phenotyping Olive Cultivars for Drought Tolerance Using Leaf Macro-Characteristics. Horticulturae 2022, 8, 939. https://doi.org/10.3390/horticulturae8100939
Razouk R, Hssaini L, Alghoum M, Adiba A, Hamdani A. Phenotyping Olive Cultivars for Drought Tolerance Using Leaf Macro-Characteristics. Horticulturae. 2022; 8(10):939. https://doi.org/10.3390/horticulturae8100939
Chicago/Turabian StyleRazouk, Rachid, Lahcen Hssaini, Mohamed Alghoum, Atman Adiba, and Anas Hamdani. 2022. "Phenotyping Olive Cultivars for Drought Tolerance Using Leaf Macro-Characteristics" Horticulturae 8, no. 10: 939. https://doi.org/10.3390/horticulturae8100939
APA StyleRazouk, R., Hssaini, L., Alghoum, M., Adiba, A., & Hamdani, A. (2022). Phenotyping Olive Cultivars for Drought Tolerance Using Leaf Macro-Characteristics. Horticulturae, 8(10), 939. https://doi.org/10.3390/horticulturae8100939





