Rehydration and Textural Properties of Dried Konjac Noodles: Effect of Alkaline and Some Gelling Agents
Abstract
:1. Introduction
2. Experimental Section
2.1. Material
2.2. Production of Konjac Noodles by Using Alkaline Solution
2.3. Production of Konjac Noodles by Using Sodium Alginate and Calcium Chloride
2.4. Drying Experiments
2.5. Rehydration Properties
2.6. Texture Profile Analysis
2.7. Microstructure of Konjac Noodles
2.8. Statistical Analysis
3. Results and Discussion
3.1. Production of Konjac Noodles
3.2. Drying Experiments
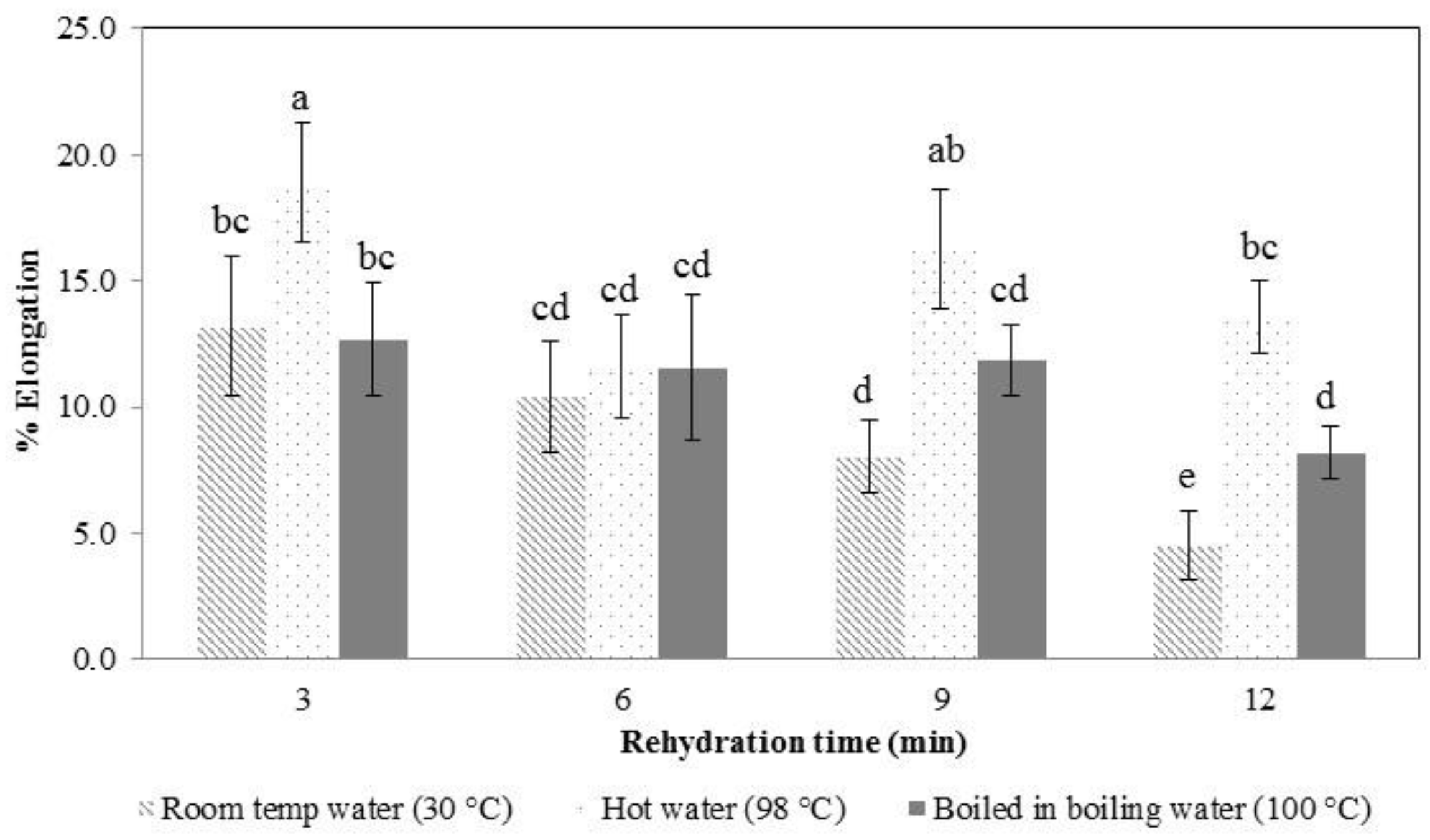
3.3. Rehydration Properties, Texture Profile Analysis, and Microstructure Observation
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Impaprasert, R.; Borompichaichartkul, C.; Srzednicki, G. A new drying approach to enhance quality of konjac glucomannan extracted from Amorph. Muelleri. Dry. Technol. Int. J. 2014, 32, 851–860. [Google Scholar] [CrossRef]
- Konjac Flour. Available online: http://www.fao.org/gsfaonline/additives/details.html?id=10 (accessed on 15 August 2015).
- Zhang, Y.Q.; Xie, B.J.; Gan, X. Advance in the applications of konjac glucomannan and its derivatives. Carbohyd. Polym. 2005, 60, 27–31. [Google Scholar] [CrossRef]
- Tye, R.J. Konjac flour: Properties and applications. Food Technol. 1991, 45, 86–92. [Google Scholar]
- Chua, M.; Baldwin, T.C.; Hocking, T.J.; Chan, K. Traditional uses and potential health benefits of Amorphophallus konjac K Koch ex N.E.Br. J. Ethnopharmacol. 2010, 128, 268–278. [Google Scholar] [CrossRef] [PubMed]
- Shah, B.R.; Li, B.; Wang, L.; Liu, S.; Li, Y.; Wei, X.; Weiping, J.; Zhenshun, L. Health benefits of konjac glucomannan with special focus on diabetes. Bioact. Carbohydr. Diet. Fiber 2015, 5, 179–187. [Google Scholar] [CrossRef]
- Dave, V.; McCarthy, S.P. Review of Konjac Glucomannan. J. Environ. Polym. Degr. 1997, 5, 237–243. [Google Scholar]
- Nishinari, K.; Zhang, H. Recent advances in the understanding of heat set gelling polysaccharides. Trends Food Sci. Technol. 2004, 15, 305–312. [Google Scholar] [CrossRef]
- Jimenez-Colmenero, F.; Cofrades, S.; Herrero, A.M.; Solas, M.T.; Ruiz-Capillas, C. Konjac gel for use as potential fat analogue for healthier meat product development: Effect of chilled and frozen storage. Food Hydrocoll. 2013, 30, 351–357. [Google Scholar] [CrossRef]
- Harding, S.E.; Smith, I.H.; Lawson, C.J.; Gahlerd, R.J.; Wood, S. Studies on macromolecular interactions in ternary mixtures of konjac glucomannan, xanthan gum and sodium alginate. Carbohydr. Polym. 2011, 83, 329–338. [Google Scholar] [CrossRef]
- Wang, J.; Liu, C.; Shuai, Y.; Cui, X.; Nie, L. Controlled release of anticancer drug using graphene oxide as a drug-binding effector in konjac glucomannan/sodium alginate hydrogels. Colloids Surf. B Biointerfaces 2014, 113, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Association of Official Analytical Chemists. Official Methods of Analysis, 18th ed.; Association of Official Analytical Chemists International: Washington, DC, USA, 2006. [Google Scholar]
- Chen, J.Y.; Zhang, H.; Miao, Y. The effect of quantity of salt on the drying characteristics of fresh noodles. Agric. Agric. Sci. Procedia. 2014, 2, 207–211. [Google Scholar] [CrossRef]
- Zhou, Y.; Cao, H.; Hou, M.; Nirasawa, S.; Tatsumi, E.; Foster, T.J.; Cheng, Y. Effect of konjac glucomannan on physical and sensory properties of noodles made from low-protein wheat flour. Food Res. Int. 2013, 51, 879–885. [Google Scholar] [CrossRef]
- Kaur, A.; Singh, N.; Kaur, S.; Katyal, M.; Virdi, A.S.; Kaur, D.; Ahlawat, A.K.; Singh, A.H. Relationship of various flour properties with noodle making characteristics among durum wheat varieties. Food Chem. 2015, 188, 517–526. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Takahashi, R.; Kobayashi, S.; Kawase, T.; Nishinari, K. Gelation behavior of native and acetylated konjac glucomannan. Biomacromolecules 2002, 3, 1296–1303. [Google Scholar] [CrossRef] [PubMed]
- Maekaji, K. Mechanism of gelation of konjac mannan. Agric. Biol. Chem. 1974, 38, 315–321. [Google Scholar] [CrossRef]
- Williams, M.A.; Foster, T.J.; Martin, D.R.; Norton, I.T.; Yoshimura, M.; Nishinari, K. A molecular description of the gelation mechanism of konjac mannan. Biomacromolecules 2000, 1, 440–450. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, M.; Nishinari, K. Dynamic viscoelastic study on the gelation of konjac glucomannan with different molecular weights. Food Hydrocoll. 1999, 13, 227–233. [Google Scholar] [CrossRef]
- Zhang, H.; Yoshimura, M.; Nishinari, K.; Williams, M.A.K.; Foster, T.J.; Norton, I.T. Gelation behaviour of konjac glucomannan with different molecular weights. Biopolymers 2001, 59, 38–50. [Google Scholar] [CrossRef]
- Solo-de-Zaldivar, B.; Herranz, B.; Borderias, J. First steps in using glucomannan to make thermostable gels for potential use in mince fish restructuration. Int. J. Food Eng. 2012, 8, 1–10. [Google Scholar] [CrossRef]
- Lapasin, R.; Pricl, S. Rheology of Industrial Polysaccharides: Theory and Applications; Aspen Publishers: Gaithersburg, MD, USA, 1999. [Google Scholar]
- Herranz, B.; Tovar, C.A.; Solo-de-Zaldívar, B.; Borderias, A.J. Effect of alkalis on konjac glucomannan gels for use as potential gelling agents in restructured seafood products. Food Hydrocoll. 2012, 27, 145–153. [Google Scholar] [CrossRef]
- Grant, G.T.; Morris, E.R.; Rees, D.A.; Smith, P.J.C.; Thom, D. Biological interactions between polysaccharides and divalent cations: The Egg-Box model. FEBS Lett. 1973, 32, 195–198. [Google Scholar] [CrossRef]
- Chuajiw, W.; Nakano, M.; Takatori, K.; Kojima, T.; Wakimoto, Y.; Fukushima, Y. Effects of amine, amine salt and amide on the behaviour of carbon dioxide absorption into calcium hydroxide suspension to precipitate calcium carbonate. J. Environ. Sci. 2013, 25, 2507–2515. [Google Scholar] [CrossRef]
- Luo, X.G.; He, P.; Lin, X.Y. The mechanism of sodium hydroxide solution promoting the gelation of Konjac glucomannan (KGM). Food Hydrocoll. 2013, 30, 92–99. [Google Scholar] [CrossRef]
- Jang, A.; Kim, H.; Shim, J.; Lee, S.K.; Lee, S. Correlation of thermal conductivity of instant noodles with their textural property for rehydration study. J. Texture Stud. 2016, 47, 87–91. [Google Scholar] [CrossRef]
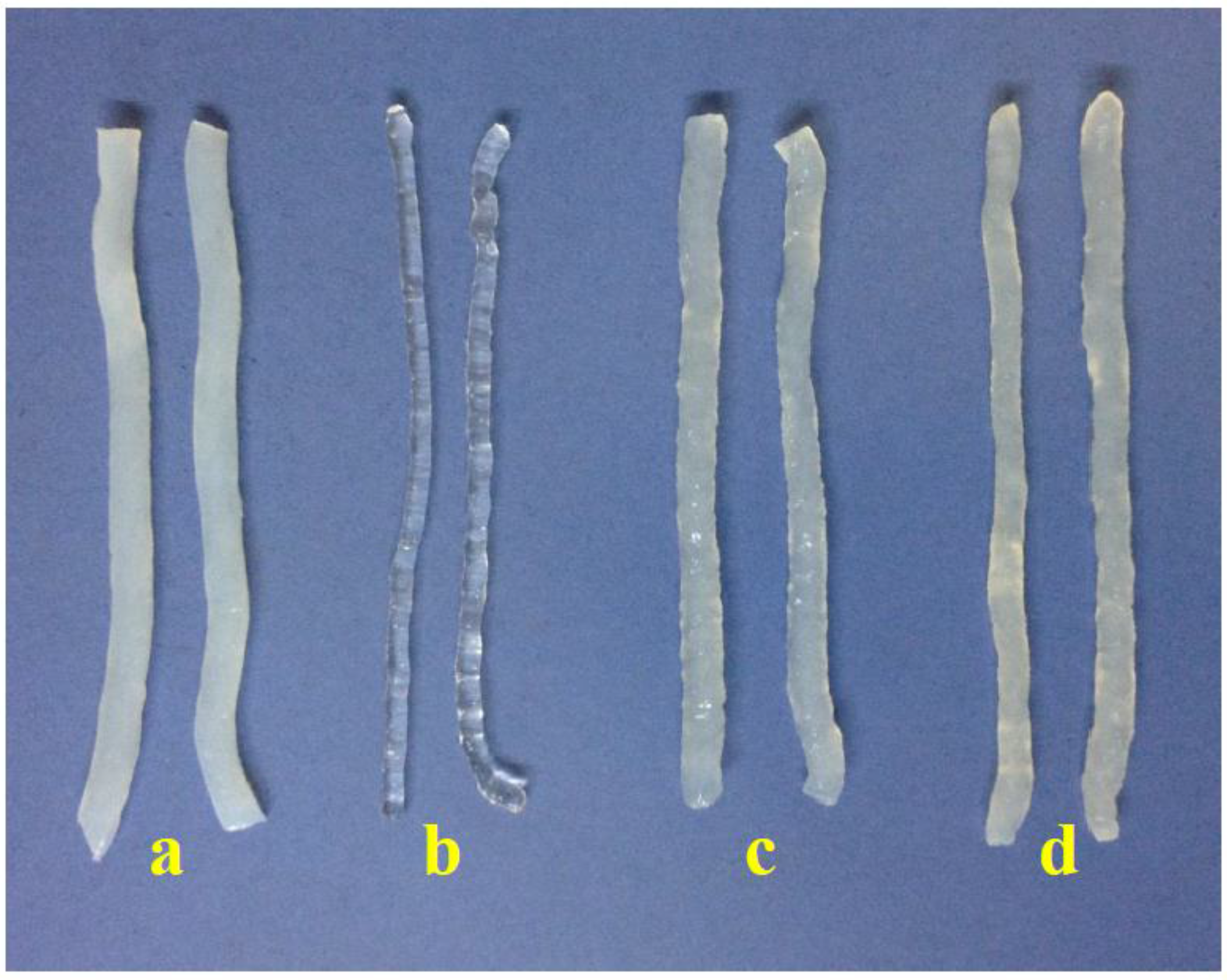

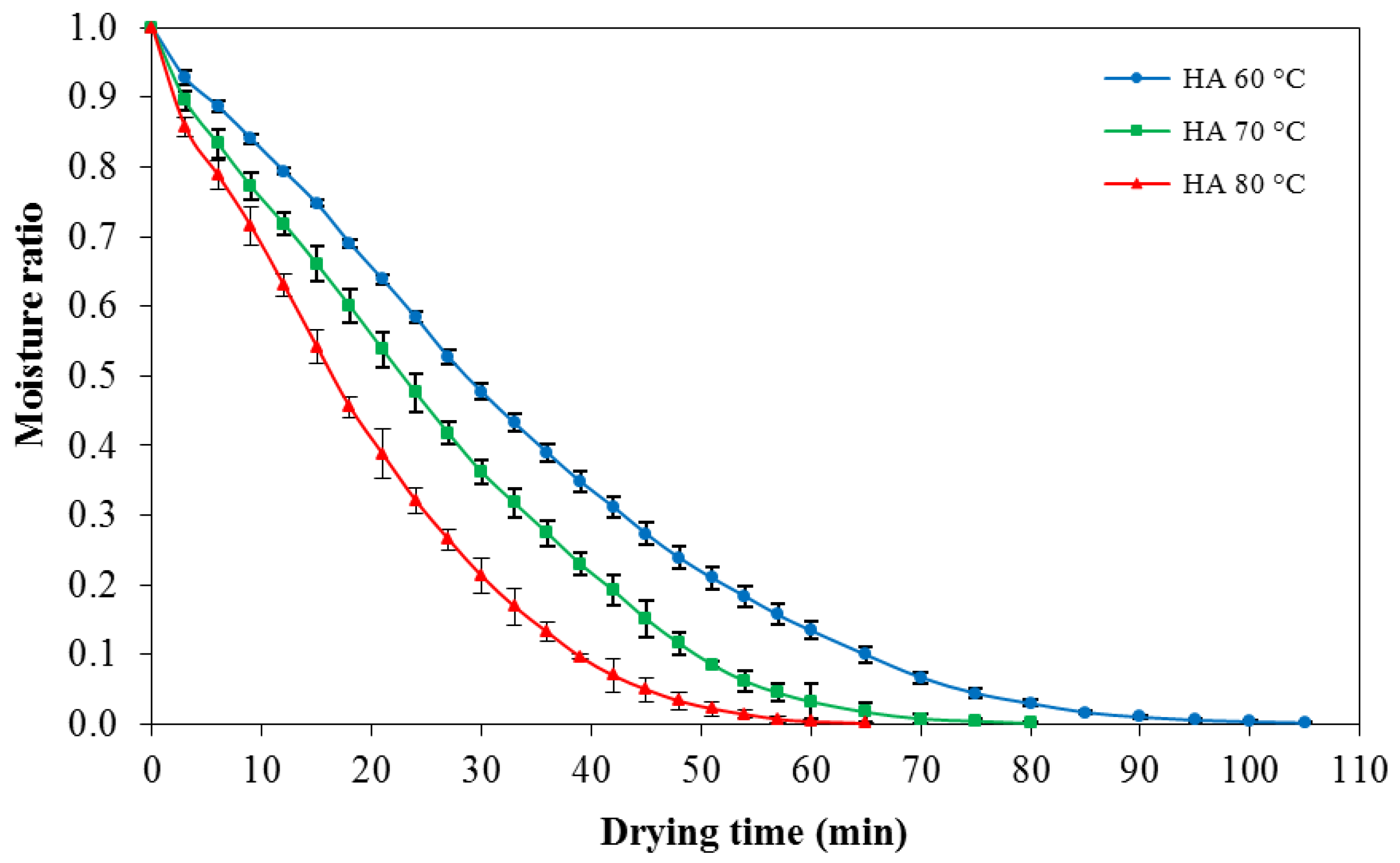
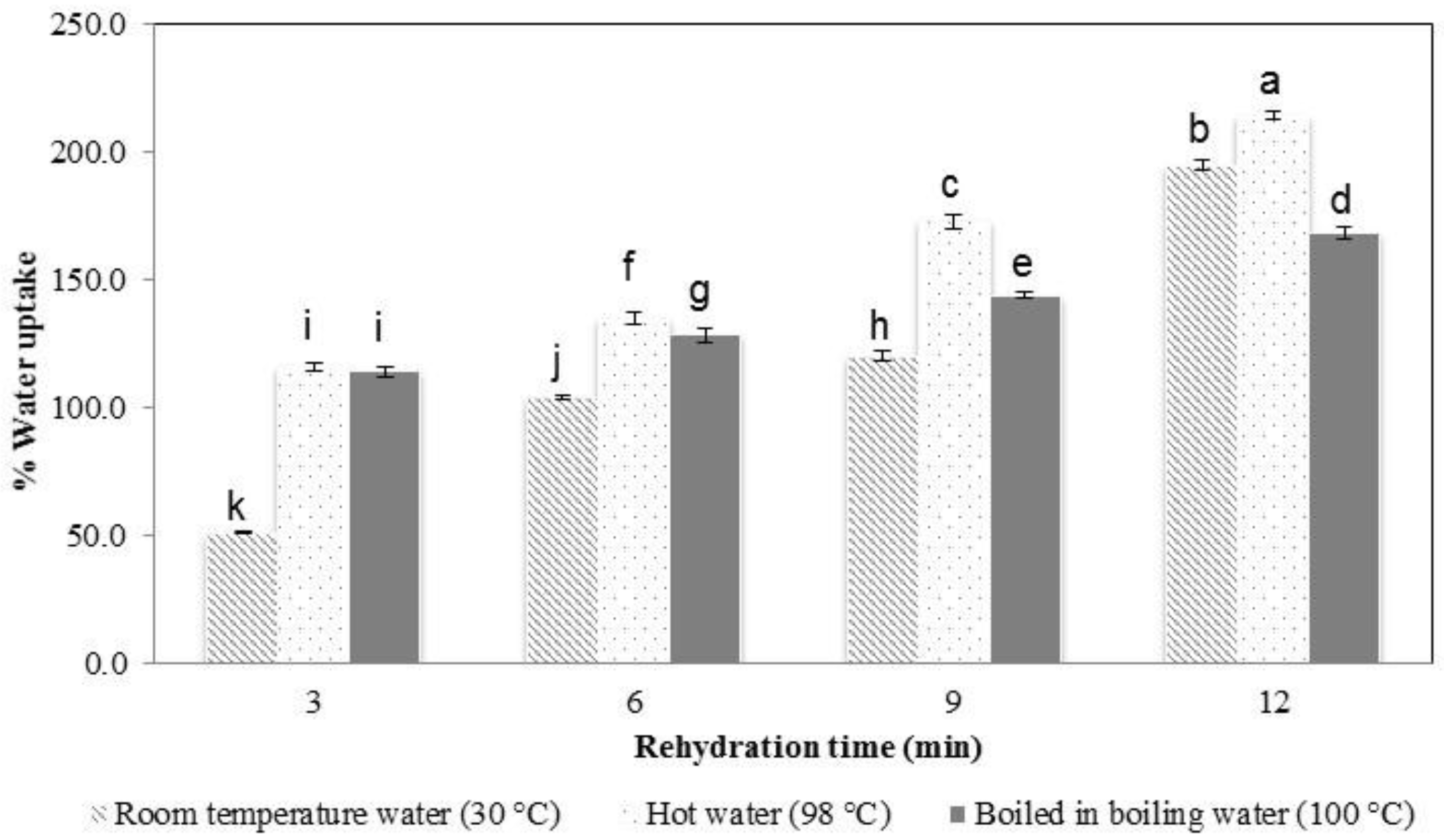

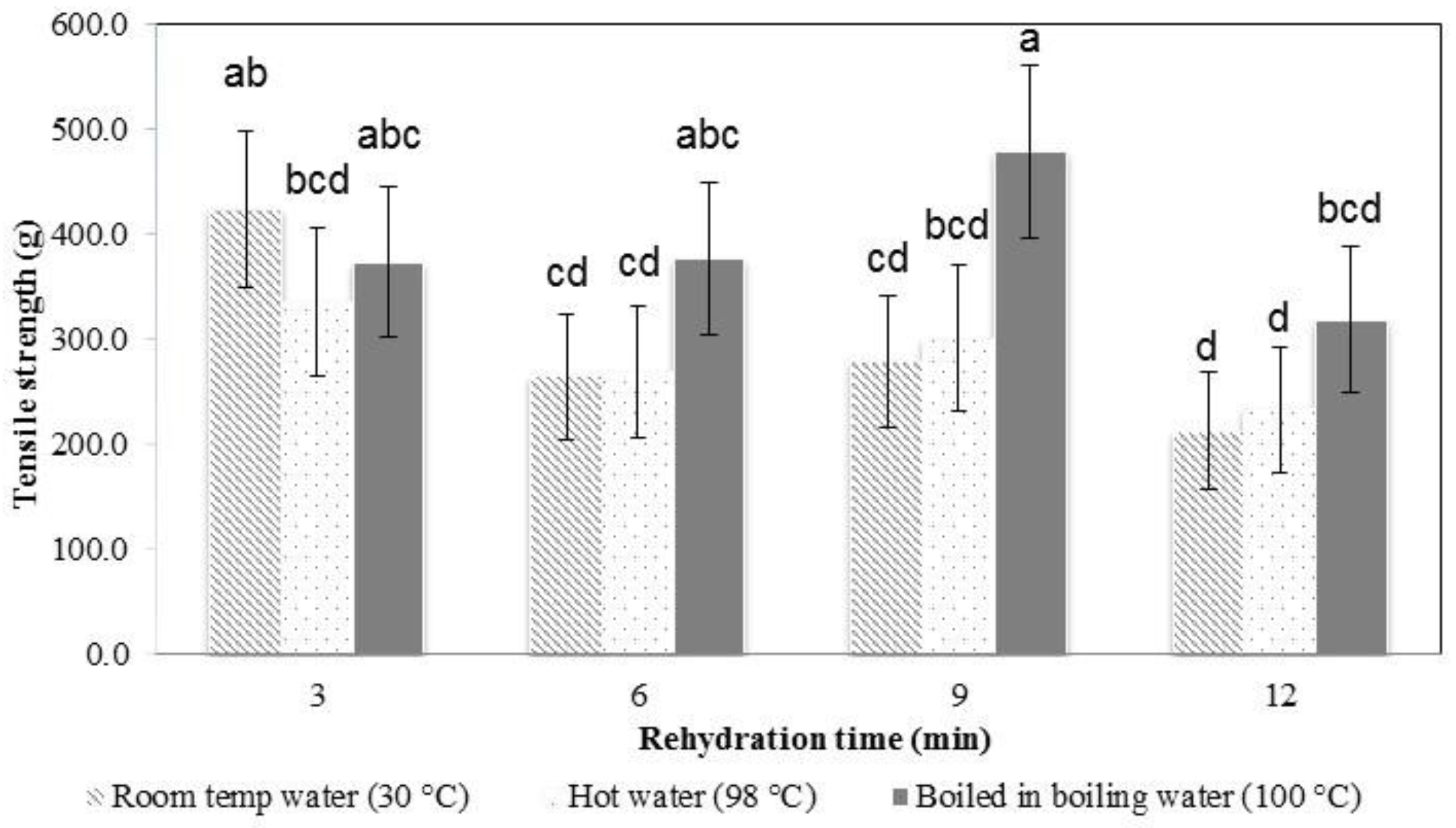
| KGM Sample | Springiness | Cohesiveness |
|---|---|---|
| Commercial product | 0.87 ± 0.03 abc | 0.75 ± 0.02 a |
| Limewater/15 min incubation | 0.89 ± 0.05 abc | 0.70 ± 0.02 bc |
| Limewater/20 min incubation | 0.89 ± 0.03 ab | 0.70 ± 0.02 bc |
| Limewater/25 min incubation | 0.88 ± 0.04 abc | 0.69 ± 0.03 bc |
| Limewater/30 min incubation | 0.90 ± 0.05 a | 0.74 ± 0.02 a |
| Limewater/35 min incubation | 0.84 ± 0.05 c | 0.68 ± 0.02 cd |
| Calcium hydroxide/0.5 h incubation | N/A | N/A |
| Calcium hydroxide/1 h incubation | N/A | N/A |
| Calcium hydroxide/1.5 h incubation | 0.84 ± 0.06 c | 0.71 ± 0.03 b |
| Calcium hydroxide/2 h incubation | 0.86 ± 0.02 abc | 0.66 ± 0.03 e |
| 0.25% sodium alginate | N/A | N/A |
| 0.5% sodium alginate | N/A | N/A |
| 1% sodium alginate | 0.85 ± 0.05 c | 0.66 ± 0.03 de |
| 1.25% sodium alginate | 0.85 ± 0.05 c | 0.63 ± 0.04 f |
| 1.5% sodium alginate | 0.85 ± 0.05 bc | 0.71 ± 0.04 b |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Impaprasert, R.; Piyarat, S.; Sophontanakij, N.; Sakulnate, N.; Paengkanya, S.; Borompichaichartkul, C.; Srzednicki, G. Rehydration and Textural Properties of Dried Konjac Noodles: Effect of Alkaline and Some Gelling Agents. Horticulturae 2017, 3, 20. https://doi.org/10.3390/horticulturae3010020
Impaprasert R, Piyarat S, Sophontanakij N, Sakulnate N, Paengkanya S, Borompichaichartkul C, Srzednicki G. Rehydration and Textural Properties of Dried Konjac Noodles: Effect of Alkaline and Some Gelling Agents. Horticulturae. 2017; 3(1):20. https://doi.org/10.3390/horticulturae3010020
Chicago/Turabian StyleImpaprasert, Rarisara, Sujika Piyarat, Natnicha Sophontanakij, Nattiya Sakulnate, Suwit Paengkanya, Chaleeda Borompichaichartkul, and George Srzednicki. 2017. "Rehydration and Textural Properties of Dried Konjac Noodles: Effect of Alkaline and Some Gelling Agents" Horticulturae 3, no. 1: 20. https://doi.org/10.3390/horticulturae3010020
APA StyleImpaprasert, R., Piyarat, S., Sophontanakij, N., Sakulnate, N., Paengkanya, S., Borompichaichartkul, C., & Srzednicki, G. (2017). Rehydration and Textural Properties of Dried Konjac Noodles: Effect of Alkaline and Some Gelling Agents. Horticulturae, 3(1), 20. https://doi.org/10.3390/horticulturae3010020





