Scientific and Pharmaceutical Aspects of Christensenella minuta, a Promising Next-Generation Probiotic
Abstract
1. Introduction
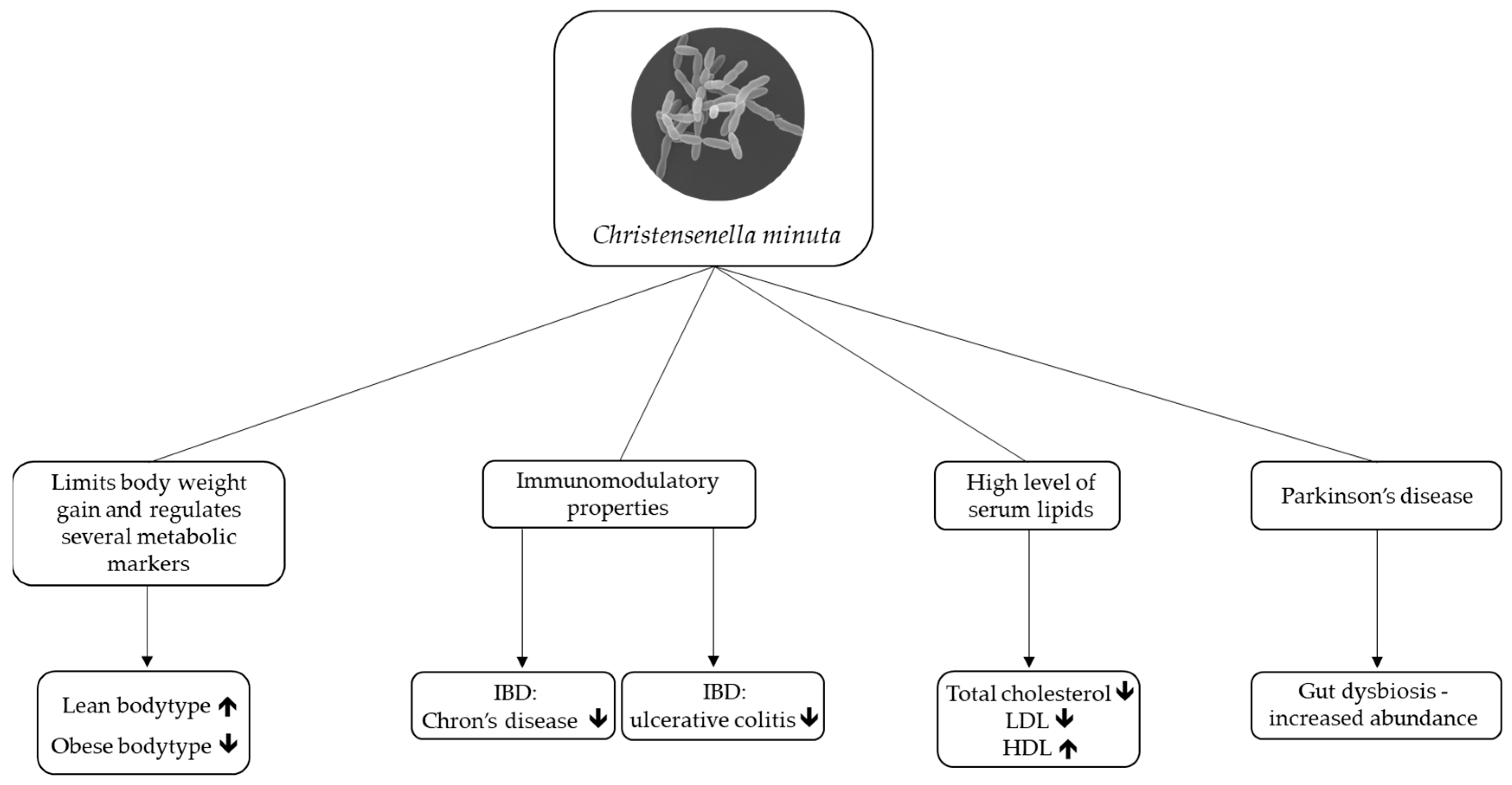
2. Christensenella minuta as a Potential Next-Generation Probiotic
3. Aspects of Metabolism
4. Possibilities of Large-Scale Cultivation
5. Strategies for Improved Oral Delivery
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- David, L.A.; Maurice, C.F.; Carmody, R.N.; Gootenberg, D.B.; Button, J.E.; Wolfe, B.E.; Ling, A.V.; Devlin, A.S.; Varma, Y.; Fischbach, M.A.; et al. Diet Rapidly and Reproducibly Alters the Human Gut Microbiome. Nature 2014, 505, 559–563. [Google Scholar] [CrossRef] [PubMed]
- Bermúdez-Humarán, L.G.; Salinas, E.; Ortiz, G.G.; Ramirez-Jirano, L.J.; Morales, J.A.; Bitzer-Quintero, O.K. From Probiotics to Psychobiotics: Live Beneficial Bacteria Which Act on the Brain-Gut Axis. Nutrients 2019, 11, 890. [Google Scholar] [CrossRef] [PubMed]
- Naseer, M.; Poola, S.; Uraz, S.; Tahan, V. Therapeutic Effects of Prebiotics on Constipation: A Schematic Review. Curr. Clin. Pharmacol. 2020, 15, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Davani-Davari, D.; Negahdaripour, M.; Karimzadeh, I.; Seifan, M.; Mohkam, M.; Masoumi, S.; Berenjian, A.; Ghasemi, Y. Prebiotics: Definition, Types, Sources, Mechanisms, and Clinical Applications. Foods 2019, 8, 92. [Google Scholar] [CrossRef] [PubMed]
- Martyniak, A.; Medyńska-Przęczek, A.; Wędrychowicz, A.; Skoczeń, S.; Tomasik, P.J. Prebiotics, Probiotics, Synbiotics, Paraprobiotics and Postbiotic Compounds in IBD. Biomolecules 2021, 11, 1903. [Google Scholar] [CrossRef]
- Abraham, B.P.; Quigley, E.M.M. Probiotics in Inflammatory Bowel Disease. Gastroenterol. Clin. N. Am. 2017, 46, 769–782. [Google Scholar] [CrossRef]
- Collinson, S.; Deans, A.; Padua-Zamora, A.; Gregorio, G.V.; Li, C.; Dans, L.F.; Allen, S.J. Probiotics for Treating Acute Infectious Diarrhoea. Cochrane Database Syst. Rev. 2020, 2020, CD003048. [Google Scholar] [CrossRef]
- Olaimat, A.N.; Aolymat, I.; Al-Holy, M.; Ayyash, M.; Abu Ghoush, M.; Al-Nabulsi, A.A.; Osaili, T.; Apostolopoulos, V.; Liu, S.-Q.; Shah, N.P. The Potential Application of Probiotics and Prebiotics for the Prevention and Treatment of COVID-19. NPJ Sci. Food 2020, 4, 17. [Google Scholar] [CrossRef] [PubMed]
- Williams, N.T. Probiotics. Am. J. Health Pharm. 2010, 67, 449–458. [Google Scholar] [CrossRef]
- Roselli, M.; Finamore, A. Use of Synbiotics for Ulcerative Colitis Treatment. Curr. Clin. Pharmacol. 2020, 15, 174–182. [Google Scholar] [CrossRef]
- Yadav, M.K.; Kumari, I.; Singh, B.; Sharma, K.K.; Tiwari, S.K. Probiotics, Prebiotics and Synbiotics: Safe Options for next-Generation Therapeutics. Appl. Microbiol. Biotechnol. 2022, 106, 505–521. [Google Scholar] [CrossRef]
- Simon, E.; Călinoiu, L.F.; Mitrea, L.; Vodnar, D.C. Probiotics, Prebiotics, and Synbiotics: Implications and Beneficial Effects against Irritable Bowel Syndrome. Nutrients 2021, 13, 2112. [Google Scholar] [CrossRef] [PubMed]
- Markowiak, P.; Śliżewska, K. Effects of Probiotics, Prebiotics, and Synbiotics on Human Health. Nutrients 2017, 9, 1021. [Google Scholar] [CrossRef] [PubMed]
- Sanders, M.E.; Merenstein, D.J.; Reid, G.; Gibson, G.R.; Rastall, R.A. Probiotics and Prebiotics in Intestinal Health and Disease: From Biology to the Clinic. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 605–616. [Google Scholar] [CrossRef]
- Wade, W.G.; Spratt, D.A.; Dymock, D.; Weightman, A.J. Molecular Detection of Novel Anaerobic species in Dentoalveolar Abscesses. Clin. Infect. Dis. 1997, 25, S235–S236. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chang, C.-J.; Lin, T.-L.; Tsai, Y.-L.; Wu, T.-R.; Lai, W.-F.; Lu, C.-C.; Lai, H.-C. Next-generation Probiotics in Disease Amelioration. J. Food Drug Anal. 2019, 27, 615–622. [Google Scholar] [CrossRef] [PubMed]
- López-Moreno, A.; Acuña, I.; Torres-Sánchez, A.; Ruiz-Moreno, Á.; Cerk, K.; Rivas, A.; Suárez, A.; Monteoliva-Sánchez, M.; Aguilera, M. Next-generation Probiotics for Neutralizing Obesogenic Effects: Taxa Culturing Searching Strategies. Nutrients 2021, 13, 1617. [Google Scholar] [CrossRef]
- Satokari, R. Modulation of Gut Microbiota for Health by Current and Next-Generation Probiotics. Nutrients 2019, 11, 1921. [Google Scholar] [CrossRef]
- O’Toole, P.W.; Marchesi, J.R.; Hill, C. Next-generation Probiotics: The Spectrum from Probiotics to Live Biotherapeutics. Nat. Microbiol. 2017, 2, 17057. [Google Scholar] [CrossRef]
- Langella, P.; Guarner, F.; Martín, R. Editorial: Next-Generation Probiotics: From Commensal Bacteria to Novel Drugs and Food Supplements. Front. Microbiol. 2019, 10, 1973. [Google Scholar] [CrossRef]
- Saarela, M.H. Safety Aspects of Next-generation Probiotics. Curr. Opin. Food Sci. 2019, 30, 8–13. [Google Scholar] [CrossRef]
- Parada Venegas, D.; De la Fuente, M.K.; Landskron, G.; González, M.J.; Quera, R.; Dijkstra, G.; Harmsen, H.J.M.; Faber, K.N.; Hermoso, M.A. Short Chain Fatty Acids (SCFAs)-Mediated Gut Epithelial and Immune Regulation and Its Relevance for Inflammatory Bowel Diseases. Front. Immunol. 2019, 10, 277. [Google Scholar] [CrossRef]
- Lee, S.; You, H.; Lee, M.; Kim, D.; Jung, S.; Park, Y.; Hyun, S. Different Reactions in Each Enterotype Depending on the Intake of Probiotic Yogurt Powder. Microorganisms 2021, 9, 1277. [Google Scholar] [CrossRef]
- Torp, A.M.; Bahl, M.I.; Boisen, A.; Licht, T.R. Optimizing Oral Delivery of Next-generation Probiotics. Trends Food Sci. Technol. 2022, 119, 101–109. [Google Scholar] [CrossRef]
- Amin, F.R.; Khalid, H.; El-Mashad, H.M.; Chen, C.; Liu, G.; Zhang, R. Functions of Bacteria and Archaea Participating in the Bioconversion of Organic Waste for Methane Production. Sci. Total Environ. 2021, 763, 143007. [Google Scholar] [CrossRef] [PubMed]
- Alonso, B.L.; Irigoyen von Sierakowski, A.; Sáez Nieto, J.A.; Rosel, A.B. First Report of Human Infection by Christensenella Minuta, a Gram-Negative, Strickly Anaerobic Rod That Inhabits the Human Intestine. Anaerobe 2017, 44, 124–125. [Google Scholar] [CrossRef] [PubMed]
- Morotomi, M.; Nagai, F.; Watanabe, Y. Description of Christensenella minuta Gen. Nov., Sp. Nov., Isolated from Human Faeces, Which Forms a Distinct Branch in the Order Clostridiales, and Proposal of Christensenellaceae Fam. Nov. Int. J. Syst. Evol. Microbiol. 2012, 62, 144–149. [Google Scholar] [CrossRef]
- Goodrich, J.K.; Waters, J.L.; Poole, A.C.; Sutter, J.L.; Koren, O.; Blekhman, R.; Beaumont, M.; Van Treuren, W.; Knight, R.; Bell, J.T.; et al. Human Genetics Shape the Gut Microbiome. Cell 2014, 159, 789–799. [Google Scholar] [CrossRef] [PubMed]
- Breton, J.; Galmiche, M.; Déchelotte, P. Dysbiotic Gut Bacteria in Obesity: An Overview of the Metabolic Mechanisms and Therapeutic Perspectives of Next-Generation Probiotics. Microorganisms 2022, 10, 452. [Google Scholar] [CrossRef]
- Lordan, C.; Thapa, D.; Ross, R.P.; Cotter, P.D. Potential for Enriching Next-Generation Health-Promoting Gut Bacteria through Prebiotics and Other Dietary Components. Gut Microbes 2020, 11, 1–20. [Google Scholar] [CrossRef]
- Oki, K.; Toyama, M.; Banno, T.; Chonan, O.; Benno, Y.; Watanabe, K. Comprehensive Analysis of the Fecal Microbiota of Healthy Japanese Adults Reveals a New Bacterial Lineage Associated with a Phenotype Characterized by a High Frequency of Bowel Movements and a Lean Body Type. BMC Microbiol. 2016, 16, 284. [Google Scholar] [CrossRef]
- Biagi, E.; Franceschi, C.; Rampelli, S.; Severgnini, M.; Ostan, R.; Turroni, S.; Consolandi, C.; Quercia, S.; Scurti, M.; Monti, D.; et al. Gut Microbiota and Extreme Longevity. Curr. Biol. 2016, 26, 1480–1485. [Google Scholar] [CrossRef]
- Bellais, S.; Nehlich, M.; Ania, M.; Duquenoy, A.; Mazier, W.; van den Engh, G.; Baijer, J.; Treichel, N.S.; Clavel, T.; Belotserkovsky, I.; et al. Species-Targeted Sorting and Cultivation of Commensal Bacteria from the Gut Microbiome Using Flow Cytometry under Anaerobic Conditions. Microbiome 2022, 10, 24. [Google Scholar] [CrossRef] [PubMed]
- Brooks, A.W.; Priya, S.; Blekhman, R.; Bordenstein, S.R. Gut Microbiota Diversity across Ethnicities in the United States. PLOS Biol. 2018, 16, e2006842. [Google Scholar] [CrossRef] [PubMed]
- Kong, F.; Hua, Y.; Zeng, B.; Ning, R.; Li, Y.; Zhao, J. Gut Microbiota Signatures of Longevity. Curr. Biol. 2016, 26, R832–R833. [Google Scholar] [CrossRef]
- Wang, F.; Yu, T.; Huang, G.; Cai, D.; Liang, X.; Su, H.; Zhu, Z.; Li, D.; Yang, Y.; Shen, P.; et al. Gut Microbiota Community and Its Assembly Associated with Age and Diet in Chinese Centenarians. J. Microbiol. Biotechnol. 2015, 25, 1195–1204. [Google Scholar] [CrossRef] [PubMed]
- Mazier, W.; Le Corf, K.; Martinez, C.; Tudela, H.; Kissi, D.; Kropp, C.; Coubard, C.; Soto, M.; Elustondo, F.; Rawadi, G.; et al. A New Strain of Christensenella Minuta as a Potential Biotherapy for Obesity and Associated Metabolic Diseases. Cells 2021, 10, 823. [Google Scholar] [CrossRef]
- Beaumont, M.; Goodrich, J.K.; Jackson, M.A.; Yet, I.; Davenport, E.R.; Vieira-Silva, S.; Debelius, J.; Pallister, T.; Mangino, M.; Raes, J.; et al. Heritable Components of the Human Fecal Microbiome Are Associated with Visceral Fat. Genome Biol. 2016, 17, 189. [Google Scholar] [CrossRef]
- Hibberd, A.A.; Yde, C.C.; Ziegler, M.L.; Honoré, A.H.; Saarinen, M.T.; Lahtinen, S.; Stahl, B.; Jensen, H.M.; Stenman, L.K. Probiotic or Synbiotic Alters the Gut Microbiota and Metabolism in a Randomised Controlled Trial of Weight Management in Overweight Adults. Benef. Microbes 2019, 10, 121–135. [Google Scholar] [CrossRef]
- Waters, J.L.; Ley, R.E. The Human Gut Bacteria Christensenellaceae Are Widespread, Heritable, and Associated with Health. BMC Biol. 2019, 17, 83. [Google Scholar] [CrossRef]
- Kropp, C.; Le Corf, K.; Relizani, K.; Tambosco, K.; Martinez, C.; Chain, F.; Rawadi, G.; Langella, P.; Claus, S.P.; Martin, R. The Keystone Commensal Bacterium Christensenella Minuta DSM 22607 Displays Anti-Inflammatory Properties Both In Vitro and In Vivo. Sci. Rep. 2021, 11, 11494. [Google Scholar] [CrossRef]
- Pellegrini, C.; Antonioli, L.; Colucci, R.; Blandizzi, C.; Fornai, M. Interplay among Gut Microbiota, Intestinal Mucosal Barrier and Enteric Neuro-Immune System: A Common Path to Neurodegenerative Diseases? Acta Neuropathol. 2018, 136, 345–361. [Google Scholar] [CrossRef]
- Borrelli, L.; Coretti, L.; Dipineto, L.; Bovera, F.; Menna, F.; Chiariotti, L.; Nizza, A.; Lembo, F.; Fioretti, A. Insect-Based Diet, a Promising Nutritional Source, Modulates Gut Microbiota Composition and SCFAs Production in Laying Hens. Sci. Rep. 2017, 7, 16269. [Google Scholar] [CrossRef]
- Navarro, S.L.; Levy, L.; Curtis, K.R.; Lampe, J.W.; Hullar, M.A.J. Modulation of Gut Microbiota by Glucosamine and Chondroitin in a Randomized, Double-Blind Pilot Trial in Humans. Microorganisms 2019, 7, 610. [Google Scholar] [CrossRef] [PubMed]
- Ruaud, A.; Esquivel-Elizondo, S.; de la Cuesta-Zuluaga, J.; Waters, J.L.; Angenent, L.T.; Youngblut, N.D.; Ley, R.E. Syntrophy via Interspecies H 2 Transfer between Christensenella and Methanobrevibacter Underlies Their Global Cooccurrence in the Human Gut. mBio 2020, 11, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Konikoff, T.; Gophna, U. Oscillospira: A Central, Enigmatic Component of the Human Gut Microbiota. Trends Microbiol. 2016, 24, 523–524. [Google Scholar] [CrossRef] [PubMed]
- Vartoukian, S.R. Cultivation Strategies for Growth of Uncultivated Bacteria. J. Oral Biosci. 2016, 58, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Nichols, D.; Cahoon, N.; Trakhtenberg, E.M.; Pham, L.; Mehta, A.; Belanger, A.; Kanigan, T.; Lewis, K.; Epstein, S.S. Use of Ichip for High-Throughput In Situ Cultivation of “Uncultivable” Microbial species. Appl. Environ. Microbiol. 2010, 76, 2445–2450. [Google Scholar] [CrossRef]
- Gao, X.; Zhang, Q.; Zhu, H. High Rejection Rate of Polysaccharides by Microfiltration Benefits Christensenella Minuta and Acetic Acid Production in an Anaerobic Membrane Bioreactor for Sludge Fermentation. Bioresour. Technol. 2019, 282, 197–201. [Google Scholar] [CrossRef]
- Gao, J.; Sadiq, F.A.; Zheng, Y.; Zhao, J.; He, G.; Sang, Y. Biofilm-Based Delivery Approaches and Specific Enrichment Strategies of Probiotics in the Human Gut. Gut Microbes 2022, 14, 2126274. [Google Scholar] [CrossRef]
- Rymovicz, A.U.M.; Souza, R.D.; Gursky, L.C.; Rosa, R.T.; Trevilatto, P.C.; Groppo, F.C.; Rosa, E.A.R. Screening of Reducing Agents for Anaerobic Growth of Candida Albicans SC5314. J. Microbiol. Methods 2011, 84, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Trabelsi, I.; Bejar, W.; Ayadi, D.; Chouayekh, H.; Kammoun, R.; Bejar, S.; Ben Salah, R. Encapsulation in Alginate and Alginate Coated-Chitosan Improved the Survival of Newly Probiotic in Oxgall and Gastric Juice. Int. J. Biol. Macromol. 2013, 61, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Łętocha, A.; Miastkowska, M.; Sikora, E. Preparation and Characteristics of Alginate Microparticles for Food, Pharmaceutical and Cosmetic Applications. Polymers 2022, 14, 3834. [Google Scholar] [CrossRef] [PubMed]
- Khosravi Zanjani, M.A.; Tarzi, B.G.; Sharifan, A.; Mohammadi, N. Microencapsulation of Probiotics by Calcium Alginate-Gelatinized Starch with Chitosan Coating and Evaluation of Survival in Simulated Human Gastro-Intestinal Condition. Iran. J. Pharm. Res. 2014, 13, 843–852. [Google Scholar] [PubMed]
- Wang, X.; Gao, S.; Yun, S.; Zhang, M.; Peng, L.; Li, Y.; Zhou, Y. Microencapsulating Alginate-Based Polymers for Probiotics Delivery Systems and Their Application. Pharmaceuticals 2022, 15, 644. [Google Scholar] [CrossRef]
- Sbehat, M.; Mauriello, G.; Altamimi, M. Microencapsulation of Probiotics for Food Functionalization: An Update on Literature Reviews. Microorganisms 2022, 10, 1948. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz Atay, H. Antibacterial Activity of Chitosan-Based Systems. In Functional Chitosan; Springer: Singapore, 2019; pp. 457–489. [Google Scholar]
- Sarao, L.K.; Arora, M. Probiotics, Prebiotics, and Microencapsulation: A Review. Crit. Rev. Food Sci. Nutr. 2017, 57, 344–371. [Google Scholar] [CrossRef]
- Krasaekoopt, W.; Bhandari, B.; Deeth, H. Evaluation of Encapsulation Techniques of Probiotics for Yoghurt. Int. Dairy J. 2003, 13, 3–13. [Google Scholar] [CrossRef]
- Dinakar, P.; Mistry, V.V. Growth and Viability of Bifidobacterium Bifidum in Cheddar Cheese. J. Dairy Sci. 1994, 77, 2854–2864. [Google Scholar] [CrossRef]
- Chen, M.-J.; Chen, K.-N. Applications of Probiotic Encapsulation in Dairy Products. In Encapsulation and Controlled Release Technologies in Food Systems; Blackwell Publishing: Ames, IA, USA, 2007; pp. 83–112. [Google Scholar]
- Sharma, H.; Sharma, S.; Bajwa, J.; Chugh, R.; Kumar, D. Polymeric Carriers in Probiotic Delivery System. Carbohydr. Polym. Technol. Appl. 2023, 5, 100301. [Google Scholar] [CrossRef]
- Mazzoni, C.; Nielsen, L.H. Microdevices to Successfully Deliver Orally Administered Drugs. In Nanotechnology for Oral Drug Delivery; Elsevier: Amsterdam, The Netherlands, 2020; pp. 285–315. [Google Scholar]
- Stenman, L.K.; Burcelin, R.; Lahtinen, S. Establishing a Causal Link between Gut Microbes, Body Weight Gain and Glucose Metabolism in Humans—Towards Treatment with Probiotics. Benef. Microbes 2016, 7, 11–22. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | Specific to C. minuta |
|---|---|
| Morphology | non-spore-forming, non-motile short rods |
| Gram staining | Gram-negative (however, Gram-positive dying behavior was observed as well) |
| Oxygen sensitivity | not extremely oxygen-sensitive |
| Beneficial metabolic relationships | Methanobacteriaceae M. smithii Oscillospira |
| Conditions | Specific to C. minuta |
|---|---|
| Optimal pH | 7.5 |
| Optimal temperature | 37–40 °C |
| Oxygen sensitivity | Anaerobic (it is able to tolerate oxygen for at least 24 h) |
| Utilized sugars | glucose, d-xylose, d-mannose, salicin, l-ramnose, and l-arabinose |
| Sugars that cannot be utilized | maltose, lactose, trehalose, sucrose, d-sorbitol, raffinose, d-mannitol, melesitol cellobiose |
| Enzymatic activity | β-galactosidase, naphthol-AS-BI-phosphohydrolase, α-arabinosidase, β-glucosidase, and glutamic acid decarboxylase |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pető, Á.; Kósa, D.; Szilvássy, Z.; Fehér, P.; Ujhelyi, Z.; Kovács, G.; Német, I.; Pócsi, I.; Bácskay, I. Scientific and Pharmaceutical Aspects of Christensenella minuta, a Promising Next-Generation Probiotic. Fermentation 2023, 9, 767. https://doi.org/10.3390/fermentation9080767
Pető Á, Kósa D, Szilvássy Z, Fehér P, Ujhelyi Z, Kovács G, Német I, Pócsi I, Bácskay I. Scientific and Pharmaceutical Aspects of Christensenella minuta, a Promising Next-Generation Probiotic. Fermentation. 2023; 9(8):767. https://doi.org/10.3390/fermentation9080767
Chicago/Turabian StylePető, Ágota, Dóra Kósa, Zoltán Szilvássy, Pálma Fehér, Zoltán Ujhelyi, Gabriella Kovács, István Német, István Pócsi, and Ildikó Bácskay. 2023. "Scientific and Pharmaceutical Aspects of Christensenella minuta, a Promising Next-Generation Probiotic" Fermentation 9, no. 8: 767. https://doi.org/10.3390/fermentation9080767
APA StylePető, Á., Kósa, D., Szilvássy, Z., Fehér, P., Ujhelyi, Z., Kovács, G., Német, I., Pócsi, I., & Bácskay, I. (2023). Scientific and Pharmaceutical Aspects of Christensenella minuta, a Promising Next-Generation Probiotic. Fermentation, 9(8), 767. https://doi.org/10.3390/fermentation9080767










