Sequencing Batch Reactor Performance Evaluation on Orthophosphates and COD Removal from Brewery Wastewater
Abstract
:1. Introduction
| Present Study | [1] | [16] | [17] | |||
|---|---|---|---|---|---|---|
| Parameter | Mean ± SD | Range | Range | Range | SA Discharge Limits | EU Discharge Limits |
| Temperature, °C | 31 ± 3.7 | 25.3–37 | 18–40 | - | <44 | - |
| pH | 6.5 ± 2.4 | 4.4–6.17 | 3–12 | 3–12 | 5.0–9.5 | - |
| Turbidity, NTU | 570 ± 164 | 303–1039 | - | - | - | - |
| Total COD, mg/L | 7687 ± 2030 | 3447–11,813 | 2000–6000 | 1800–5000 | 75 | 125 |
| BOD5, mg/L | - | - | 1200–3600 | 1005–3800 | - | 25 |
| Phosphates, mg/L | 343 ± 64 | 229–424 | 10–50 | 10–50 | 10 | 1–2 |
| TS, mg/L | 5951 ± 3387 | 2942–14,981 | 5100–8750 | 50–6000 | - | - |
| VSS, mg/L | 1799 ± 571 | 1043–2572 | - | - | - | - |
2. Materials and Methods
2.1. Sample Collection and Preparation
2.2. Activated Sludge
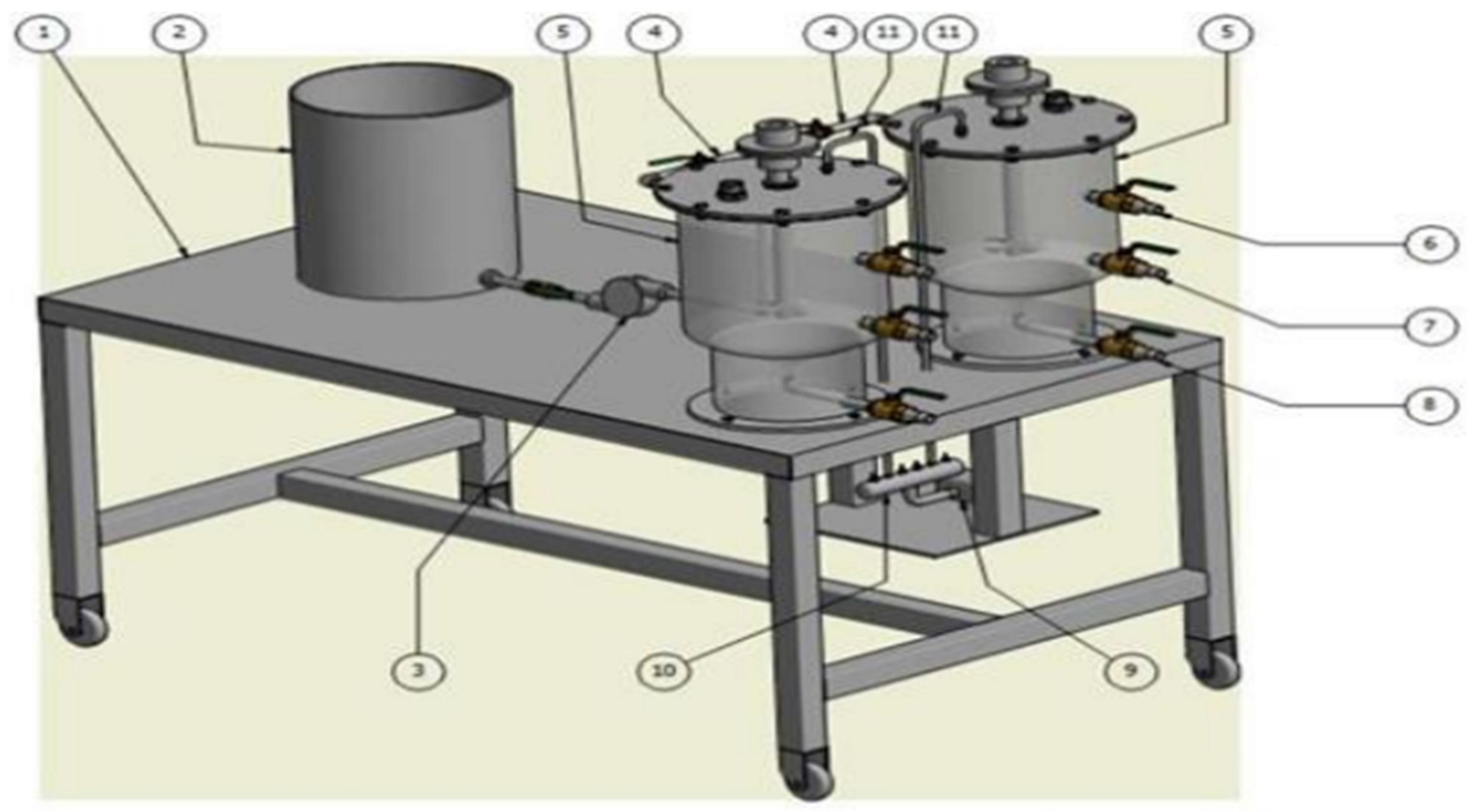
2.3. Sequencing Batch Reactor Design
2.4. Experimental Approach
- The filling phase—This was considered the first operational phase of the SBR system. The reactor was first seeded with 4 L of activated sludge under anaerobic conditions. Raw brewery wastewater was fed into the holding tank where suspended solids were allowed to settle by gravitational force for a period of 2 h. After the settling phase, 9 L of raw brewery wastewater supernatant was pumped into the reactor. The filling phase took place under anaerobic conditions; however, the stirrer was switched on and set to operate at 350 rpm to allow mixing. According to Tchobanoglous [23], only mixing during the filling stage promotes filamentous growth control thus improving sludge settling and thickening. The agitation speed of the stirrer was set to be at 350 rpm because it was observed that higher agitation speed resulted in sludge bulking, thus compromising the solids’ settleability. The filling phase on average for all experimental runs lasted for 5 min.
- Reaction phase—After the filling phase, the system was allowed to undergo an anaerobic phase which favored the polyphosphate-accumulating organisms, which lasted for a period of 4 h and thereafter the reaction phase was instigated. Oxygen was supplied using an aerator pump as depicted in Figure 1 at a flow rate of 7.5 L/min, maintaining a dissolved oxygen concentration of 3 mg/L. It is worth noting that for the current work, the effect of dissolved oxygen was not investigated. During the reaction phase, microorganisms consume substrate, i.e., orthophosphates under a controlled pH which was kept within the range of 4 to 9.5. According to Tchobanoglous [23], microbial activities are hindered at pH levels less than 4 and pH levels more than 9.5. The aeration duration and anaerobic phase duration were predetermined experimentally which lasted for 14 h and 4 h, respectively. Moreover, the SBR was operated at mesophilic temperature of ±25 °C.
- Settling phase—During this phase, bio-solids were allowed to separate gravitationally from the treated liquid under quiescent conditions resulting in a clear clarified supernatant. During this phase, the stirrer was switched off as well as the aeration system, and no influent was charged into the reactor nor effluent drawn. The settling period lasted for 2 h to enhance optimum settling of bio-solids containing biodegradable organic and biological pollutants, thus resulting in a clear clarified supernatant with minimum suspended solids.
- Drawing phase—This phase was considered the final treatment operational stage for the SBR system. During this phase, the clarified supernatant was sampled as the treated reactor effluent by tapping the reactor effluent into a 250 mL sterile glass bottle for laboratory analysis.
2.5. Laboratory Analysis
2.6. Data Analysis
3. Results and Discussion
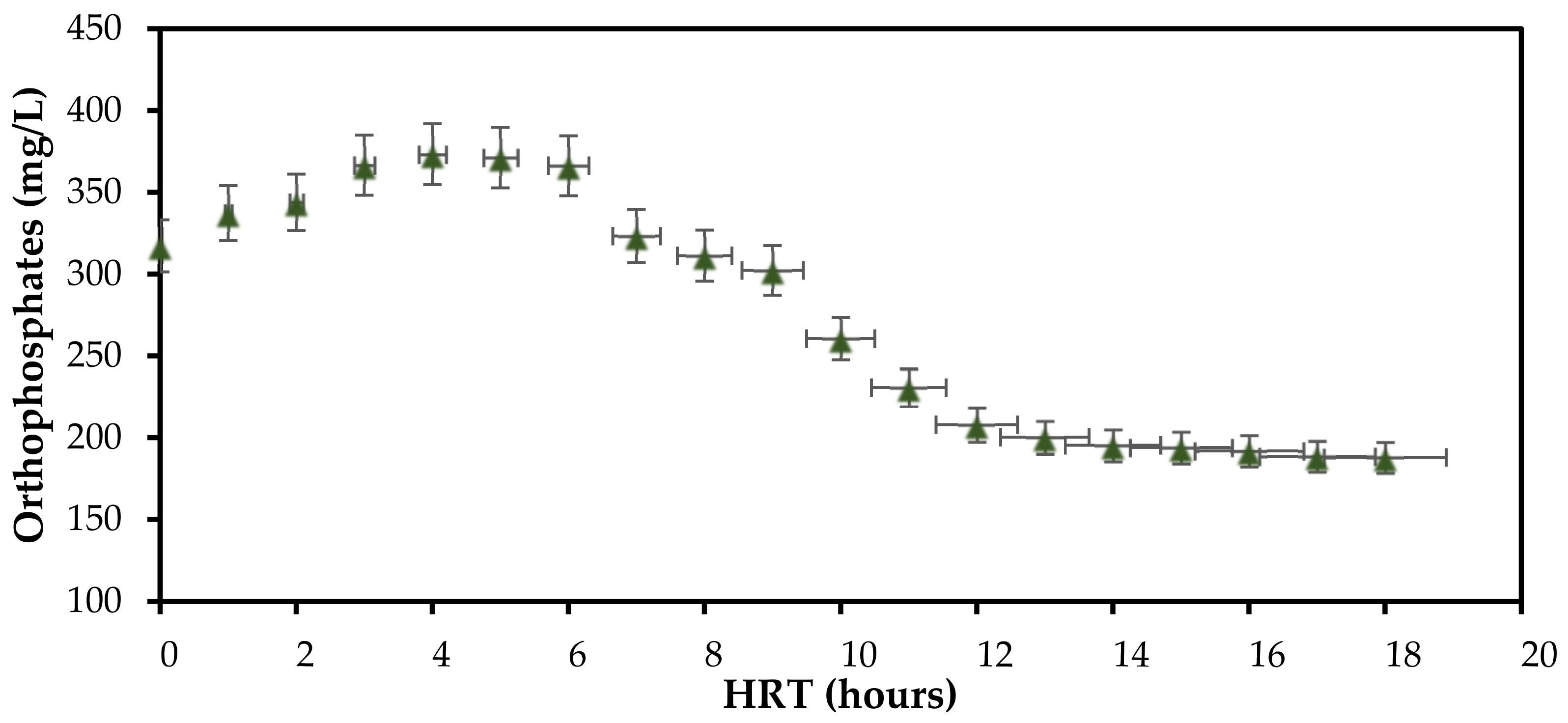
3.1. Effect of Hydraulic Retention Time (HRT) on Orthophosphate Removal
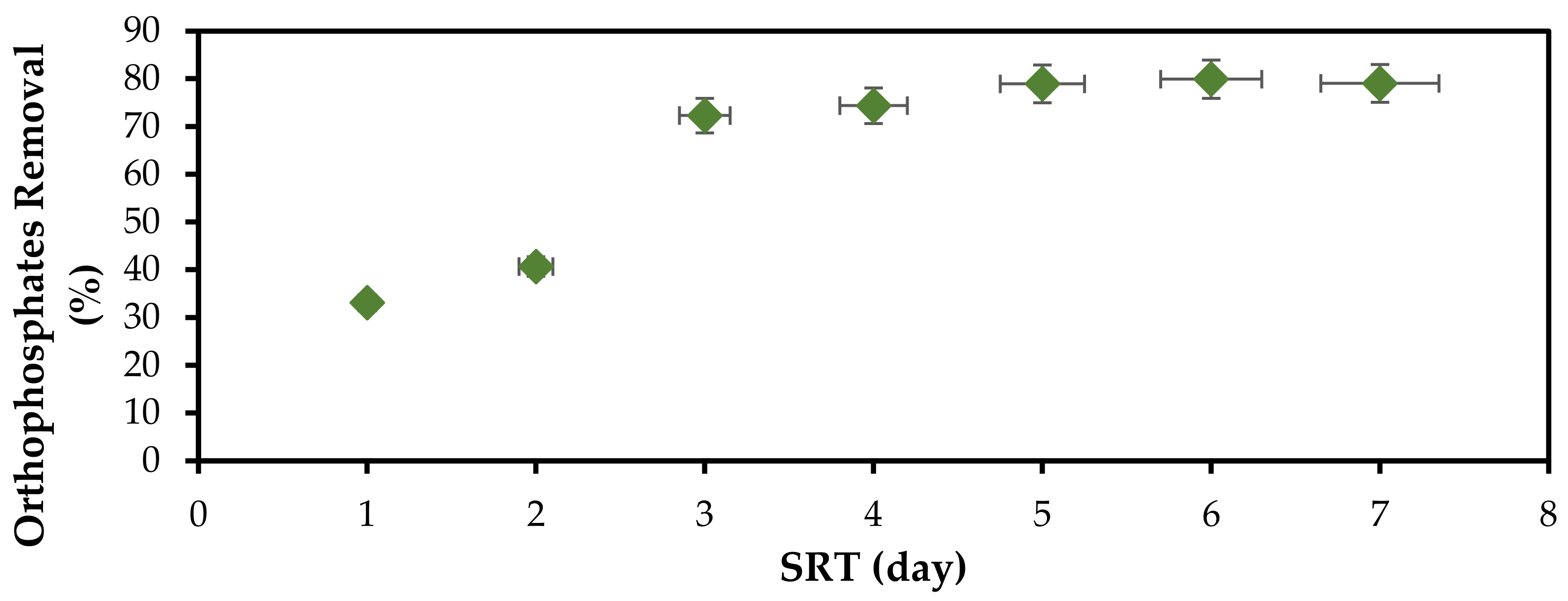
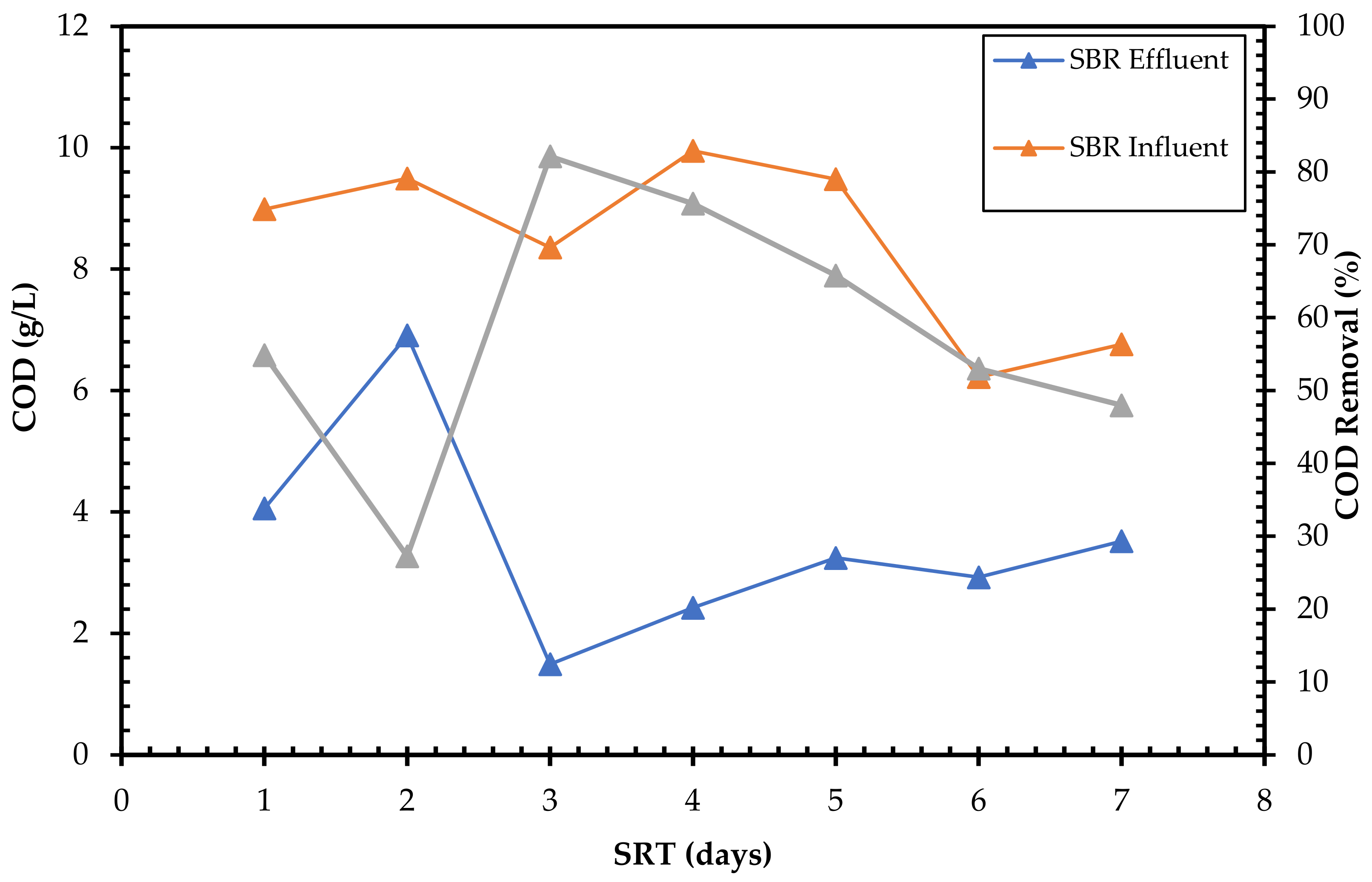
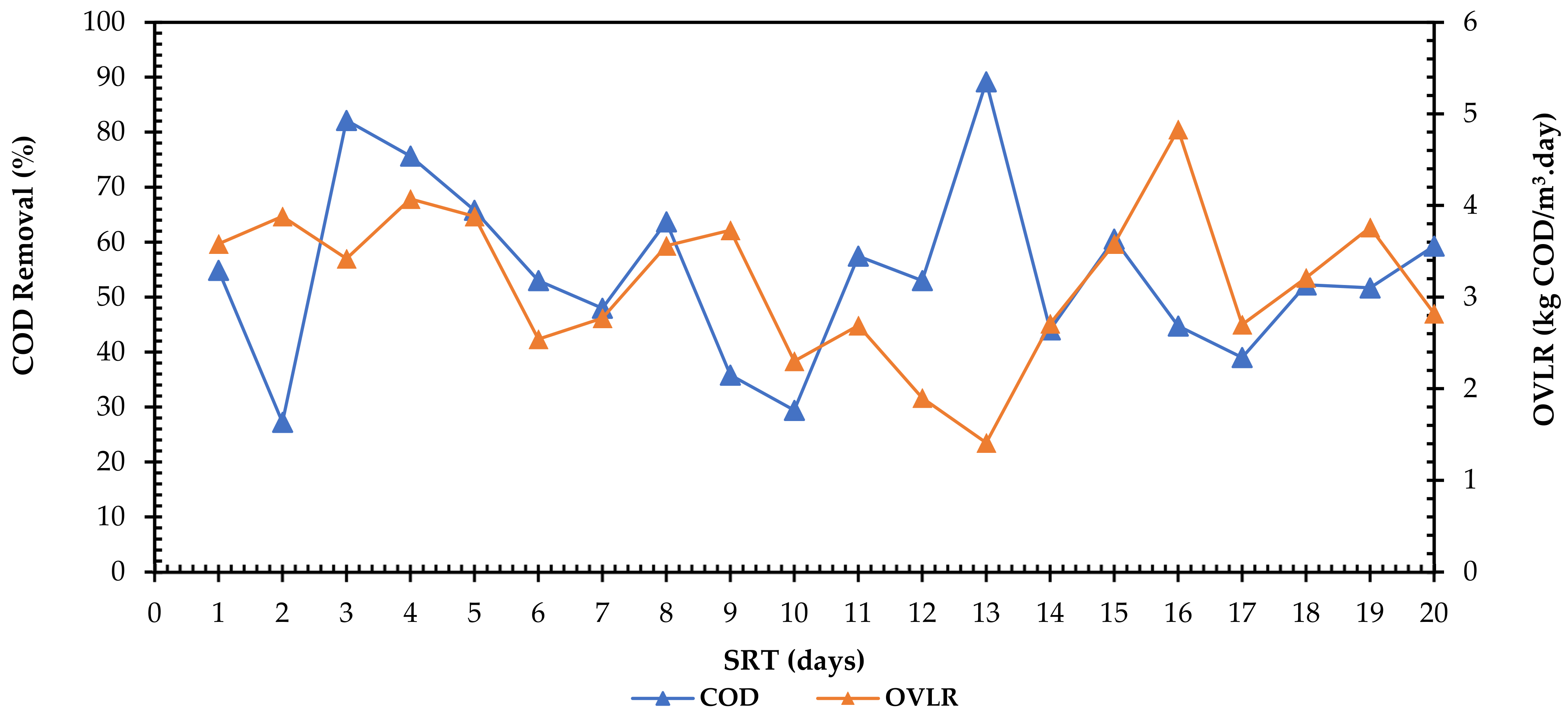
3.2. Effect of Solid Retention Time (STR) on Orthophosphate and COD Removal
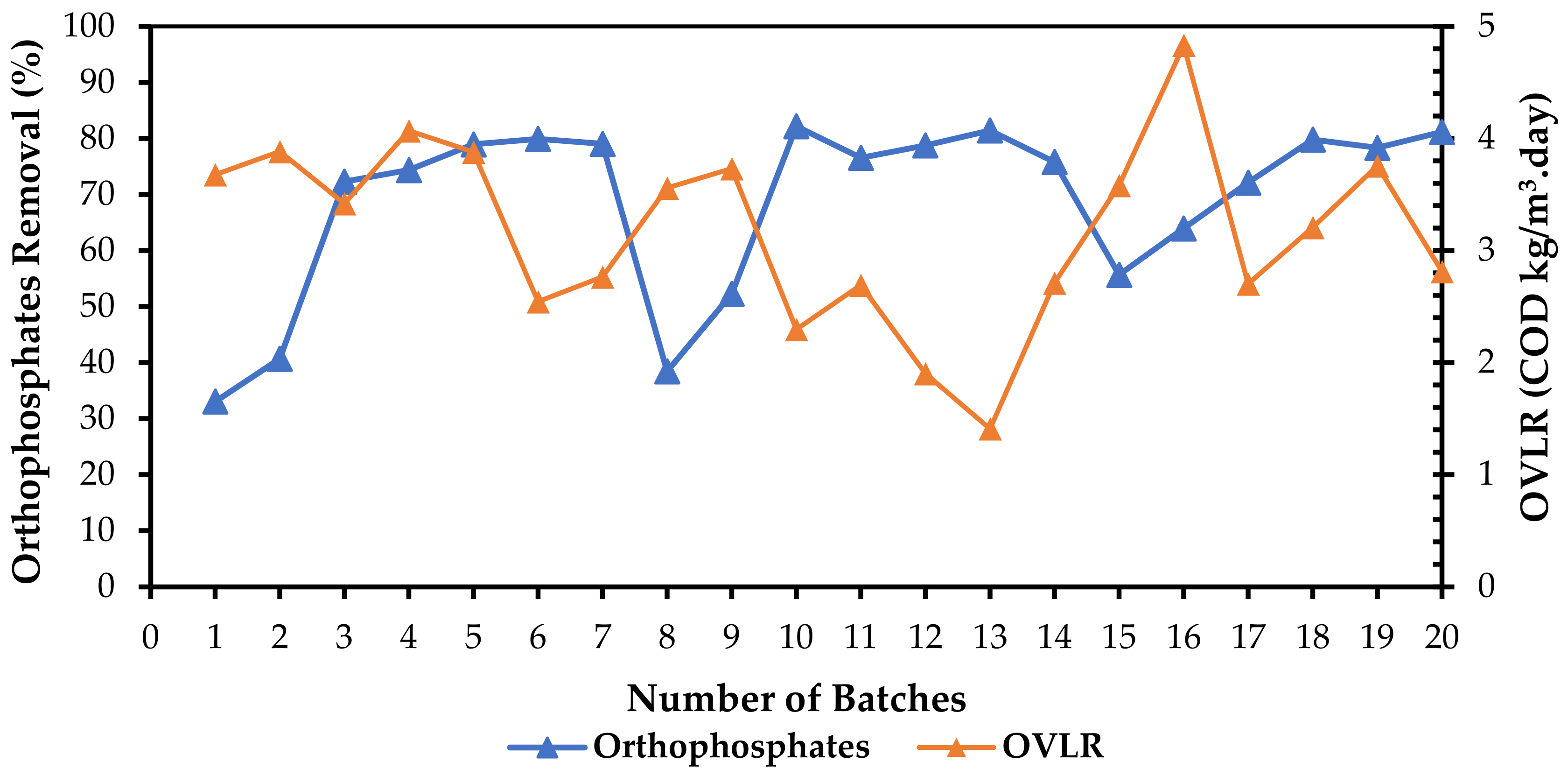
3.3. Orthophosphate and COD Removal with Variation in Organic Volumetric Loading Rate (OVLR)
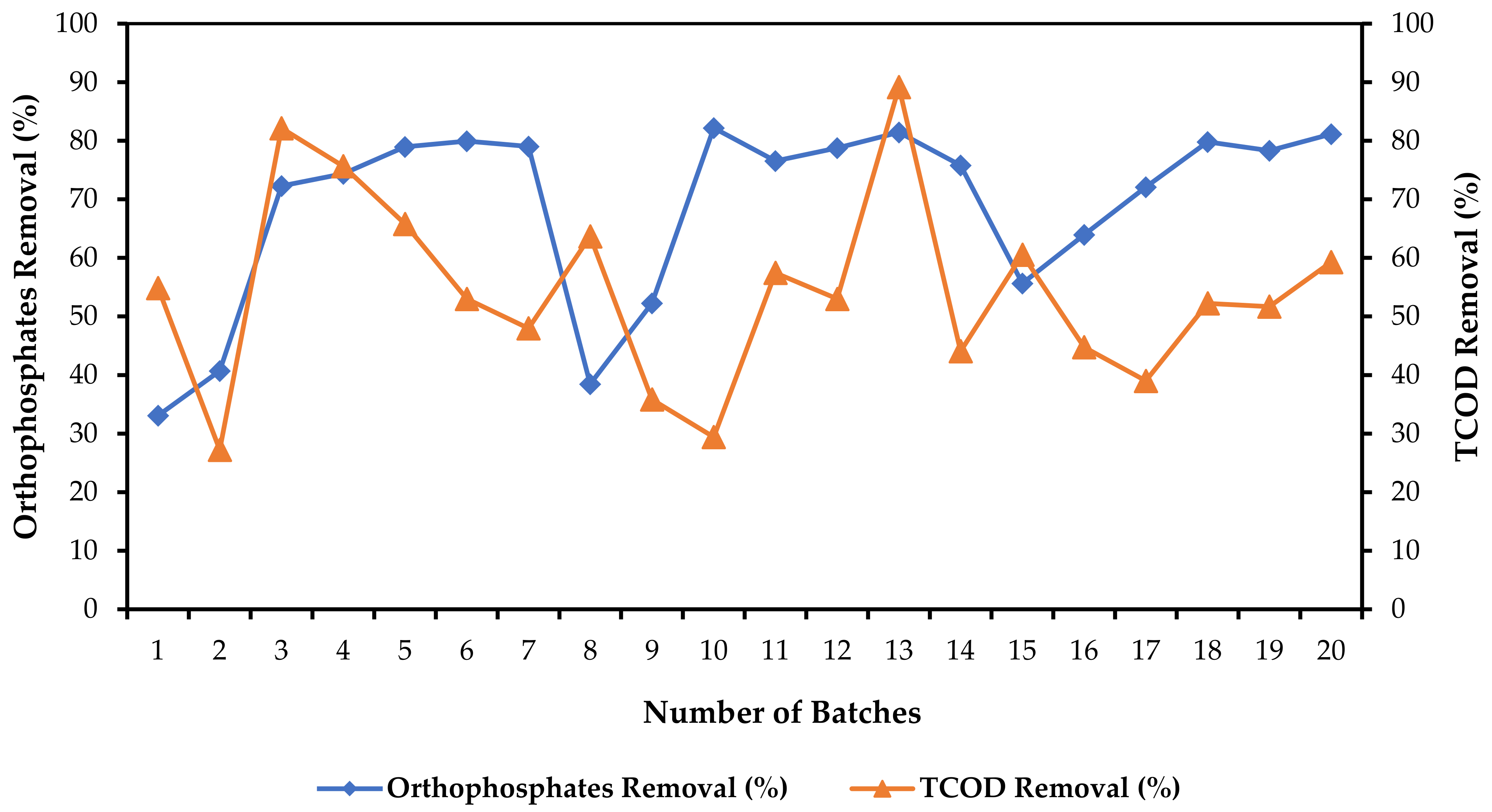
3.4. Orthophosphate and Total Chemical Oxygen Demand (TCOD) Removal
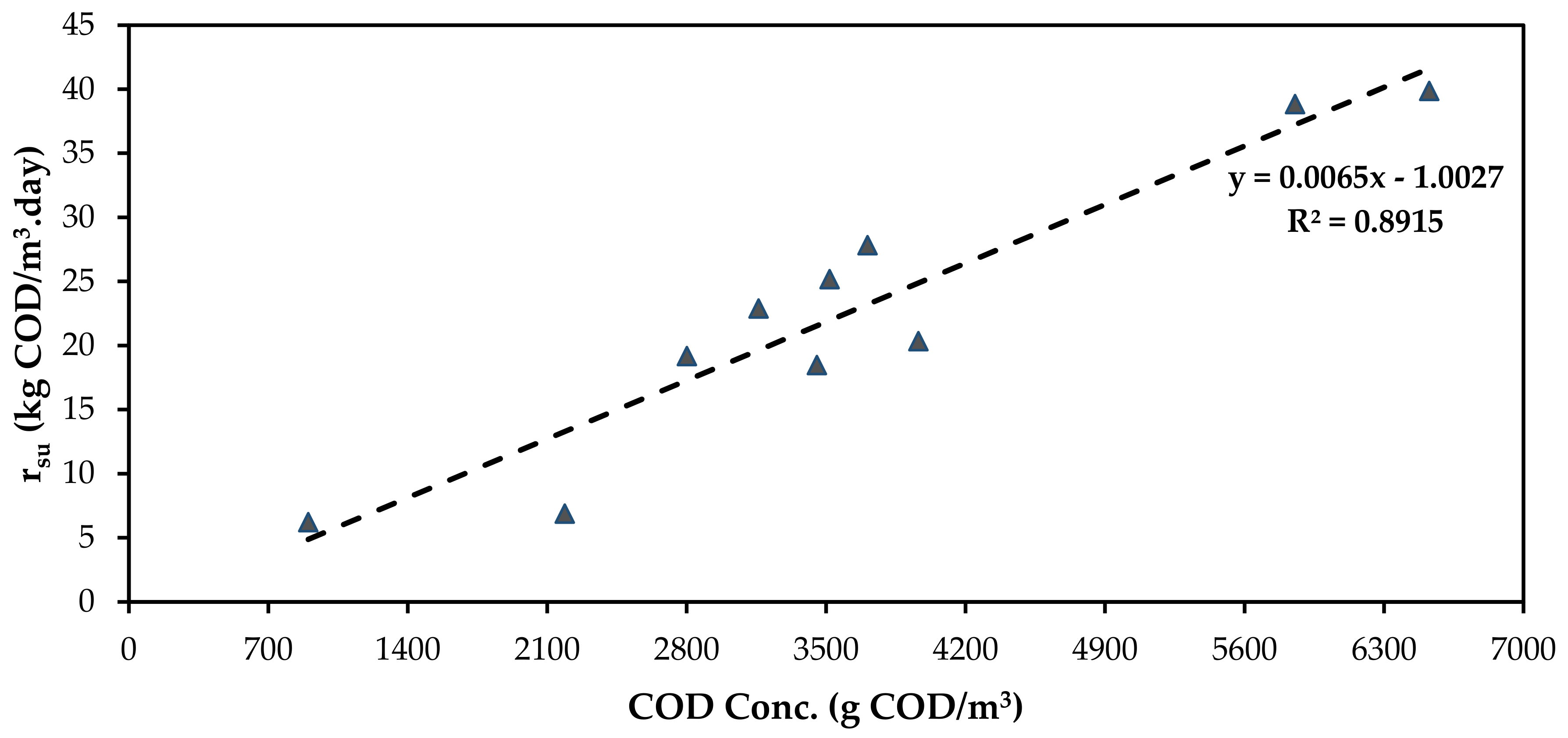
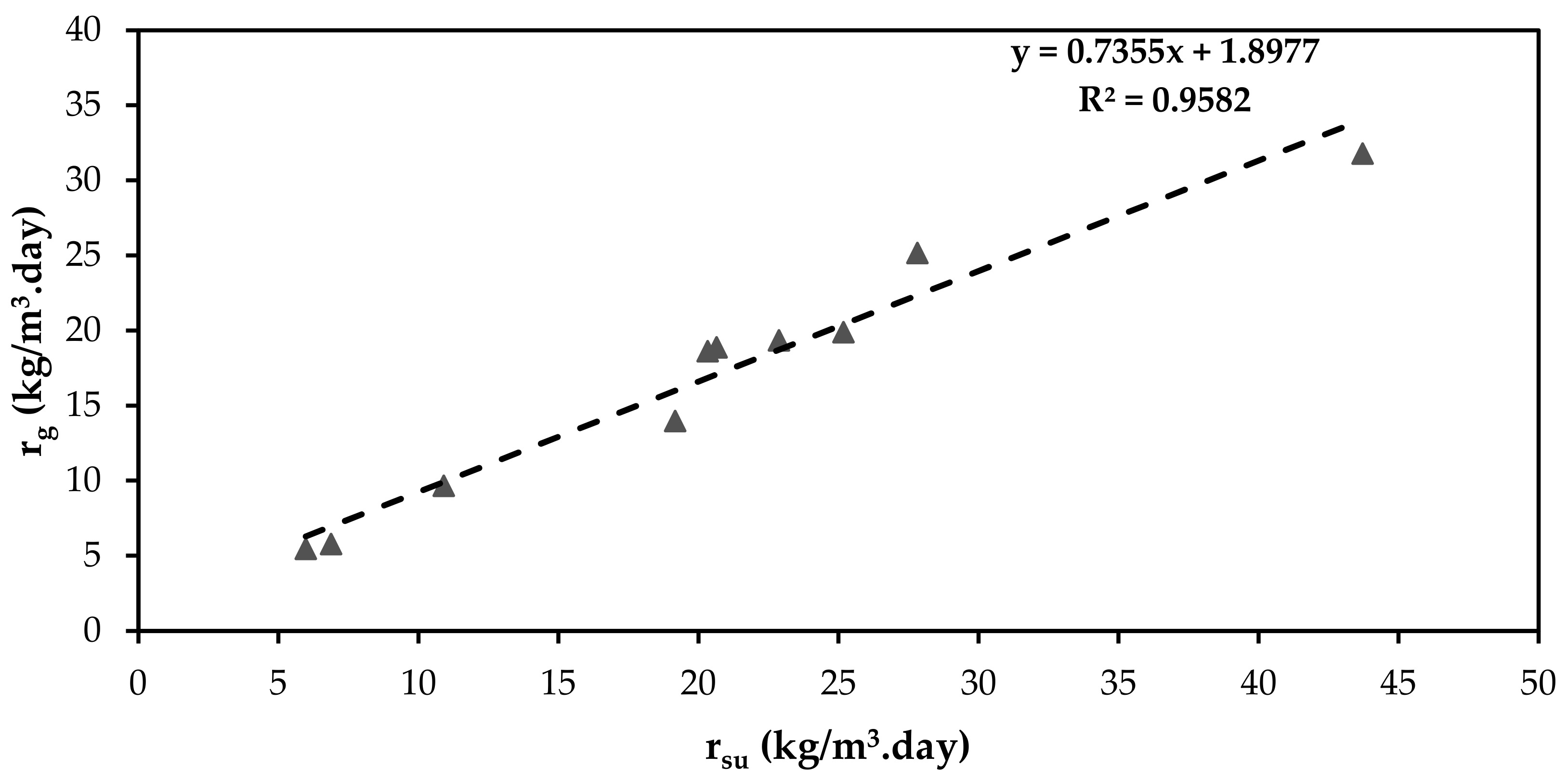
3.5. Substrate Utilisation Rate and Microbial Population Growth Late
4. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Simate, G.S.; Cluett, J.; Iyuke, S.E.; Musapatika, E.T.; Ndlovu, S.; Walubita, L.F.; Alvarez, A.E. The treatment of brewery wastewater for reuse: State of the art. Desalination 2011, 273, 235–247. [Google Scholar] [CrossRef]
- Dhakal, N.; Salinas-Rodriguez, S.G.; Hamdani, J.; Abushaban, A.; Sawalha, H.; Schippers, J.C.; Kennedy, M.D. Is Desalination a Solution to Freshwater Scarcity in Developing Countries? Membranes 2022, 12, 381. [Google Scholar] [CrossRef] [PubMed]
- Abd-Elaty, I.; Kuriqi, A.; Shahawy, A.E. Environmental rethinking of wastewater drains to manage environmental pollution and alleviate water scarcity. Nat. Hazards 2022, 110, 2353–2380. [Google Scholar] [CrossRef] [PubMed]
- Loza, V.; Perona, E.; Mateo, P. Specific responses to nitrogen and phosphorus enrichment in cyanobacteria: Factors influencing changes in species dominance along eutrophic gradients. Water Res. 2014, 48, 622–631. [Google Scholar] [CrossRef]
- Qin, B.; Gao, G.; Zhu, G.; Zhang, Y.; Song, Y.; Tang, X.; Xu, H.; Deng, J. Lake eutrophication and its ecosystem response. Chin. Sci. Bull. 2013, 58, 961–970. [Google Scholar] [CrossRef] [Green Version]
- Schindler, D.W.; Carpenter, S.R.; Chapra, S.C.; Hecky, R.E.; Orihel, D.M. Reducing phosphorus to curb lake eutrophication is a success. Environ. Sci. Technol. 2016, 50, 8923–8929. [Google Scholar] [CrossRef]
- Liu, D.H.; Lipták, B.G. Environmental Engineers’ Handbook, 2nd ed.; CRC Press Company: London, UK, 1997. [Google Scholar]
- Homayoun Aria, S.; Asadollahfardi, G.; Heidarzadeh, N. Eutrophication modelling of Amirkabir Reservoir (Iran) using an artificial neural network approach. Lakes Reserv. Res. Manag. 2019, 24, 48–58. [Google Scholar] [CrossRef] [Green Version]
- Atalay, S.; Ersöz, G. Advanced Oxidation Processes. In Novel Catalysts in Advanced Oxidation of Organic Pollutants; Springer: Cham, Swizerland, 2016; pp. 23–34. [Google Scholar]
- Lu, X.; Zhen, G.; Estrada, A.L.; Chen, M.; Ni, J.; Hojo, T.; Kubota, K.; Li, Y.Y. Operation performance and granule characterization of upflow anaerobic sludge blanket (UASB) reactor treating wastewater with starch as the sole carbon source. Bioresour. Technol. 2015, 180, 264–273. [Google Scholar] [CrossRef]
- Chen, H.; Chang, S.; Guo, Q.; Hong, Y.; Wu, P. Brewery wastewater treatment using an anaerobic membrane bioreactor. Biochem. Eng. J. 2016, 105, 321–331. [Google Scholar] [CrossRef]
- Corsino, S.F.; di Biase, A.; Devlin, T.R.; Munz, G.; Torregrossa, M.; Oleszkiewicz, J.A. Effect of extended famine conditions on aerobic granular sludge stability in the treatment of brewery wastewater. Bioresour. Technol. 2017, 226, 150–157. [Google Scholar] [CrossRef] [Green Version]
- Enitan, A.M.; Adeyemo, J.; Kumari, S.K.; Swalaha, F.M.; Bux, F. Characterization of brewery wastewater composition. Int. J. Environ. Ecol. Eng. 2015, 9, 1073–1076. [Google Scholar] [CrossRef]
- Khumalo, S.M. Biological Nutrient Removal from Industrial Wastewater Using a Sequencing Batch Reactor. Master’s Thesis, Durban University of Technology, Durban, South Africa, 2018. [Google Scholar]
- Goldammer, T. The Brewers’ Handbook; Keen Vision Publishing, LLC: Jordan, MN, USA, 1999. [Google Scholar]
- Werkneh, A.A.; Beyene, H.D.; Osunkunle, A.A. Recent advances in brewery wastewater treatment; approaches for water reuse and energy recovery: A review. Environ. Sustain. 2019, 2, 199–209. [Google Scholar] [CrossRef]
- Enitan, A.M.; Swalaha, F.M.; Adeyemo, J.; Bux, F. Assessment of brewery effluent composition from a beer producing industry in KwaZulu-Natal, South Africa. Fresenius Environ. Bull. 2014, 23, 693–701. [Google Scholar]
- Shao, X.; Peng, D.; Teng, Z.; Ju, X. Treatment of brewery wastewater using anaerobic sequencing batch reactor (ASBR). Bioresour. Technol. 2008, 99, 3182–3186. [Google Scholar] [CrossRef]
- Wang, S.G.; Liu, X.W.; Gong, W.X.; Gao, B.Y.; Zhang, D.H.; Yu, H.Q. Aerobic granulation with brewery wastewater in a sequencing batch reactor. Bioresour. Technol. 2007, 98, 2142–2147. [Google Scholar] [CrossRef]
- Rodrigues, A.C.; Brito, A.G.; Melo, L.F. Posttreatment of a brewery wastewater using a sequencing batch reactor. Water Environ. Res. 2001, 73, 45–51. [Google Scholar] [CrossRef] [Green Version]
- Ling, L.; Lo, K. Brewery wastewater treatment using suspended and attached growth sequencing batch reactors. J. Environ. Sci. Health A 1999, 34, 341–355. [Google Scholar] [CrossRef]
- Chan, C.; Guisasola, A.; Baeza, J.A. Enhanced Biological Phosphorus Removal at low Sludge Retention Time in view of its integration in A-stage systems. Water Res. 2017, 118, 217–226. [Google Scholar] [CrossRef]
- Tchobanoglous, G. Wastewater Engineering: Treatment and Resource Recovery Volume 2; McGraw-Hill: New York, NY, USA, 2014. [Google Scholar]
- Wang, X.; Wang, S.; Xue, T.; Li, B.; Dai, X.; Peng, Y. Treating low carbon/nitrogen (C/N) wastewater in simultaneous nitrification-endogenous denitrification and phosphorous removal (SNDPR) systems by strengthening anaerobic intracellular carbon storage. Water Res. 2015, 77, 191–200. [Google Scholar] [CrossRef]
- Ge, H.; Batstone, D.J.; Keller, J. Biological phosphorus removal from abattoir wastewater at very short sludge ages mediated by novel PAO clade Comamonadaceae. Water Res. 2015, 69, 173–182. [Google Scholar] [CrossRef] [Green Version]
- Koe, W.S.; Lee, J.W.; Chong, W.C.; Pang, Y.L.; Sim, L.C. An overview of photocatalytic degradation: Photocatalysts, mechanisms, and development of photocatalytic membrane. Environ. Sci. Pollut. Res. 2020, 27, 2522–2565. [Google Scholar] [CrossRef] [PubMed]
- Bhatkhande, D.S.; Pangarkar, V.G.; Beenackers, A.A.C.M. Photocatalytic degradation for environmental applications—A review. J. Chem. Technol. Biotechnol. 2002, 77, 102–116. [Google Scholar] [CrossRef]
- Jia, C.; Wang, Y.; Zhang, C.; Qin, Q. UV-TiO2 photocatalytic degradation of landfill leachate. Water Air Soil Pollut. 2011, 217, 375–385. [Google Scholar] [CrossRef]
- Cailotto, S.; Massari, D.; Gigli, M.; Campalani, C.; Bonini, M.; You, S.; Vomiero, A.; Selva, M.; Perosa, A.; Crestini, C. N-Doped Carbon Dot Hydrogels from Brewing Waste for Photocatalytic Wastewater Treatment. ACS Omega 2022, 7, 4052–4061. [Google Scholar] [CrossRef]
- Wani, A.; Bhasarkar, J.B.; Gaikwad, R. Photocatalytic Degradation of Sugar and Distillery Industry Effluent. J. Inst. Eng. Ser. E 2021, 103, 1–8. [Google Scholar] [CrossRef]
- Micheal, K.; Ayeshamariam, A.; Boddula, R.; Arunachalam, P.; AlSalhi, M.S.; Theerthagiri, J.; Prasad, S.; Madhavan, J.; Al-Mayouf, A.M. Assembled composite of hematite iron oxide on sponge-like BiOCl with enhanced photocatalytic activity. Mater. Sci. Energy Technol. 2019, 2, 104–111. [Google Scholar] [CrossRef]
- Ghernaout, D.; Elboughdiri, N. Advanced oxidation processes for wastewater treatment: Facts and future trends. Open Access Libr. J. 2020, 7, 1–15. [Google Scholar] [CrossRef]
- Chen, D.; Cheng, Y.; Zhou, N.; Chen, P.; Wang, Y.; Li, K.; Huo, S.; Cheng, P.; Peng, P.; Zhang, R.; et al. Photocatalytic degradation of organic pollutants using TiO2-based photocatalysts: A review. J. Clean. Prod. 2020, 268, 121725. [Google Scholar] [CrossRef]
- APHA. Standard Methods for the Examination of Water and Wastewater; American Public Health Association: Washington, DC, USA, 2005. [Google Scholar]
- Shabangu, K.P. Aerobic Sequencing Batch Reactor for the Treatment of Industrial Wastewater from the Brewery. Master’s Thesis, Durban University of Technology, Durban, South Africa, 2017. [Google Scholar]
- Shabangu, K.P.; Bakare, B.F. Study of an SBR Treating Brewery Wastewater: Case of COD-HRT and BOD Removal. In Proceedings of the World Congress on Engineering and Computer Science, San Francisco, CA, USA, 22–24 October 2019. [Google Scholar]
- Khumalo, S.M.; Bakare, B.F.; Rathilal, S.; Tetteh, E.K. Characterization of South African Brewery Wastewater: Oxidation-Reduction Potential Variation. Water 2022, 14, 1604. [Google Scholar] [CrossRef]
- Carucci, A.; Lindrea, K.; Majone, M.; Ramadori, R. Different mechanisms for the anaerobic storage of organic substrates and their effect on enhanced biological phosphate removal (EBPR). Water Sci. Technol. 1999, 39, 21–28. [Google Scholar] [CrossRef]
- Bakare, B.; Shabangu, K.; Chetty, M. Brewery wastewater treatment using laboratory scale aerobic sequencing batch reactor. S. Afr. J. Chem. Eng. 2017, 24, 128–134. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khumalo, S.M.; Bakare, B.F.; Tetteh, E.K.; Rathilal, S. Sequencing Batch Reactor Performance Evaluation on Orthophosphates and COD Removal from Brewery Wastewater. Fermentation 2022, 8, 296. https://doi.org/10.3390/fermentation8070296
Khumalo SM, Bakare BF, Tetteh EK, Rathilal S. Sequencing Batch Reactor Performance Evaluation on Orthophosphates and COD Removal from Brewery Wastewater. Fermentation. 2022; 8(7):296. https://doi.org/10.3390/fermentation8070296
Chicago/Turabian StyleKhumalo, Siphesihle Mangena, Babatunde Femi Bakare, Emmanuel Kweinor Tetteh, and Sudesh Rathilal. 2022. "Sequencing Batch Reactor Performance Evaluation on Orthophosphates and COD Removal from Brewery Wastewater" Fermentation 8, no. 7: 296. https://doi.org/10.3390/fermentation8070296
APA StyleKhumalo, S. M., Bakare, B. F., Tetteh, E. K., & Rathilal, S. (2022). Sequencing Batch Reactor Performance Evaluation on Orthophosphates and COD Removal from Brewery Wastewater. Fermentation, 8(7), 296. https://doi.org/10.3390/fermentation8070296









